Abstract
Objectives
We report here on the pre-clinical MRI characterization of an apoE−/− mouse model of stable and vulnerable carotid artery atherosclerotic plaques, which were induced by a tapered restriction (cast) around the artery. Specific focus was on the quantification of the wall shear stress, which is considered a key player in the development of the plaque phenotype.
Materials and methods
In vivo MRI was performed at 9.4 T. The protocol consisted of time-of-flight angiography, high-resolution T1- and T2-weighted black-blood imaging and phase-contrast flow velocity imaging as function of time in the cardiac cycle. Wall shear stress was determined by fitting the flow profile to a quadratic polynomial.
Results
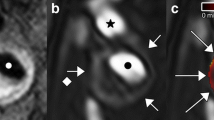
Time-of-flight angiography confirmed preservation of blood flow through the carotid arteries in all cases. T1- and T2-weighted MRI resulted in high-resolution images in which the position of the cast, luminal narrowing introduced by cast and plaque, as well as the arterial wall could be well identified. Laminar flow with low wall shear stress (11.2± 5.2 Pa) was measured upstream to the cast at the position of the vulnerable plaque. Downstream to the cast at the position of the stable plaque, the apparent velocities were low, which is consistent with vortices and an oscillatory nature of the flow.
Conclusions
Flow velocities and wall shear stress were successfully measured in this mouse model of stable and unstable plaque. The presented tools can be used to provide valuable insights in the pathogenesis of atherosclerosis.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Chien S (2008) Effects of disturbed flow on endothelial cells. Ann Biomed Eng 36(4): 554–562
Pedersen EM, Oyre S, Agerbaek M, Kristensen IB, Ringgaard S, Boesiger P, Paaske WP (1999) Distribution of early atherosclerotic lesions in the human abdominal aorta correlates with wall shear stresses measured in vivo. Eur J Vasc Endovasc Surg 18(4): 328–333
Buchanan JR, Kleinstreuer C, Truskey GA, Lei M (1999) Relation between non-uniform hemodynamics and sites of altered permeability and lesion growth at the rabbit aorto-celiac junction. Atherosclerosis 143(1): 27–40
Stone PH, Coskun AU, Kinlay S, Clark ME, Sonka M, Wahle A, Ilegbusi OJ, Yeghiazarians Y, Popma JJ, Orav J, Kuntz RE, Feldman CL (2003) Effect of endothelial shear stress on the progression of coronary artery disease, vascular remodeling, and in-stent restenosis in humans: in vivo 6-month follow-up study. Circulation 108(4): 438–444
Cheng C, Tempel D, van Haperen R, van der Baan A, Grosveld F, Daemen MJ, Krams R, de Crom R (2006) Atherosclerotic lesion size and vulnerability are determined by patterns of fluid shear stress. Circulation 113(23): 2744–2753
Mulder WJ, Strijkers GJ, Vucic E, Cormode DP, Nicolay K, Fayad ZA (2007) Magnetic resonance molecular imaging contrast agents and their application in atherosclerosis. Top Magn Reson Imaging 18(5): 409–417
Mulder WJ, Strijkers GJ, Briley-Saboe KC, Frias JC, Aguinaldo JG, Vucic E, Amirbekian V, Tang C, Chin PT, Nicolay K, Fayad ZA (2007) Molecular imaging of macrophages in atherosclerotic plaques using bimodal PEG-micelles. Magn Reson Med 58(6): 1164–1170
Windberger U, Bartholovitsch A, Plasenzotti R, Korak KJ, Heinze G (2003) Whole blood viscosity, plasma viscosity and erythrocyte aggregation in nine mammalian species: reference values and comparison of data. Exp Physiol 88(3): 431–440
Isobe S, Tsimikas S, Zhou J, Fujimoto S, Sarai M, Branks MJ, Fujimoto A, Hofstra L, Reutelingsperger CP, Murohara T, Virmani R, Kolodgie FD, Narula N, Petrov A, Narula J (2006) Noninvasive imaging of atherosclerotic lesions in apolipoprotein E-deficient and low-density-lipoprotein receptor-deficient mice with annexin A5. J Nucl Med 47(9): 1497–1505
Ni M, Zhang M, Ding SF, Chen WQ, Zhang Y (2008) Micro-ultrasound imaging assessment of carotid plaque characteristics in apolipoprotein-E knockout mice. Atherosclerosis 197(1): 64–71
Nahrendorf M, Zhang H, Hembrador S, Panizzi P, Sosnovik DE, Aikawa E, Libby P, Swirski FK, Weissleder R (2008) Nanoparticle PET-CT imaging of macrophages in inflammatory atherosclerosis. Circulation 117(3): 379–387
Fayad ZA, Fallon JT, Shinnar M, Wehrli S, Dansky HM, Poon M, Badimon JJ, Charlton SA, Fisher EA, Breslow JL, Fuster V (1998) Noninvasive in vivo high-resolution magnetic resonance imaging of atherosclerotic lesions in genetically engineered mice. Circulation 98(15): 1541–1547
Worthley SG, Helft G, Fuster V, Zaman AG, Fayad ZA, Fallon JT, Badimon JJ (2000) Serial in vivo MRI documents arterial remodeling in experimental atherosclerosis. Circulation 101(6): 586– 589
Choudhury RP, Aguinaldo JG, Rong JX, Kulak JL, Kulak AR, Reis ED, Fallon JT, Fuster V, Fisher EA, Fayad ZA (2002) Atherosclerotic lesions in genetically modified mice quantified in vivo by non-invasive high-resolution magnetic resonance microscopy. Atherosclerosis 162(2): 315–321
Dietrich T, Hucko T, Bourayou R, Jahnke C, Paetsch I, Atrott K, Stawowy P, Grafe M, Klein C, Schnackenburg B, Fleck E, Graf K (2009) High resolution magnetic resonance imaging in atherosclerotic mice treated with ezetimibe. Int J Cardiovasc Imaging 25: 827–836
Mulder WJ, Douma K, Koning GA, van Zandvoort MA, Lutgens E, Daemen MJ, Nicolay K, Strijkers GJ (2006) Liposome-enhanced MRI of neointimal lesions in the ApoE-KO mouse. Magn Reson Med 55(5): 1170–1174
Nahrendorf M, Jaffer FA, Kelly KA, Sosnovik DE, Aikawa E, Libby P, Weissleder R (2006) Noninvasive vascular cell adhesion molecule-1 imaging identifies inflammatory activation of cells in atherosclerosis. Circulation 114(14): 1504–1511
Kelly KA, Nahrendorf M, Yu AM, Reynolds F, Weissleder R (2006) In vivo phage display selection yields atherosclerotic plaque targeted peptides for imaging. Mol Imaging Biol 8(4): 201–207
McAteer MA, Schneider JE, Ali ZA, Warrick N, Bursill CA, von zur Muhlen C, Greaves DR, Neubauer S, Channon KM, Choudhury RP (2008) Magnetic resonance imaging of endothelial adhesion molecules in mouse atherosclerosis using dual-targeted microparticles of iron oxide. Arterioscler Thromb Vasc Biol 28(1): 77–83
Burtea C, Laurent S, Murariu O, Rattat D, Toubeau G, Verbruggen A, Vansthertem D, Vander Elst L, Muller RN (2008) Molecular imaging of alpha v beta3 integrin expression in atherosclerotic plaques with a mimetic of RGD peptide grafted to Gd-DTPA. Cardiovasc Res 78(1): 148–157
Beckmann N (2000) High resolution magnetic resonance angiography non-invasively reveals mouse strain differences in the cerebrovascular anatomy in vivo. Magn Reson Med 44: 252–258
Yang YM, Feng X, Yao ZW, Tang WJ, Liu HQ, Zhang L (2008) Magnetic resonance angiography of carotid and cerebral arterial occlusion in rats using a clinical scanner. J Neurosci Methods 167(2): 176–183
Jacoby C, Böring Y, Beck A, Zernecke A, Aurich V, Weber C, Schrader J, Flögel U (2008) Dynamic changes in murine vessel geometry assessed by high-resolution magnetic resonance angiography: a 9.4T study. J Magn Reson Imaging 28(3): 637–645
Miraux S, Franconi J, Thiaudière E (2006) Blood velocity assessment using 3D bright-blood time-resolved magnetic resonance angiography. Magn Reson Med 56(3): 469–473
Parzy E, Miraux S, Franconi JM, Thiaudière E (2009) In vivo quantification of blood velocity in mouse carotid and pulmonary arteries by ECG-triggered 3D time-resolved magnetic resonance angiography. NMR Biomed 22(5): 532–537
Rensen SS, Niessen PM, van Deursen JM, Janssen BJ, Heijman E, Hermeling E, Meens M, Lie N, Gijbels MJ, Strijkers GJ, Doevendans PA, Hofker MH, De Mey JG, van Eys GJ (2008) Smoothelin-B deficiency results in reduced arterial contractility, hypertension, and cardiac hypertrophy in mice. Circulation 118(8): 828–836
Herold V, Wellen J, Ziener CH, Weber T, Hiller KH, Nordbeck P, Rommel E, Haase A, Bauer WR, Jakob PM, Sarkar SK (2009) In vivo comparison of atherosclerotic plaque progression with vessel wall strain and blood flow velocity in apoE(−/−) mice with MR microscopy at 17.6 T. Magn Reson Mater Phys 22(3): 159–166
Zhao X, Pratt R, Wansapura J (2009) Quantification of aortic compliance in mice using radial phase contrast MRI. J Magn Reson Imaging 30(2): 286–291
Acknowledgments
This study was funded in part by the BSIK program entitled Molecular Imaging of Ischemic Heart Disease (project number BSIK03033), by the EC-FP6-project DiMI, LSHB-CT-2005-512146, and by the Dutch Heart Foundation (NHS) project number 2006T106. Jo Habets and Leonie Niesen are acknowledged for biotechnical assistance.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
The Below is the Electronic Supplementary Material.
ESM 2 (MOV 55 kb)
ESM 3 (MOV 65 kb)
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
van Bochove, G.S., Straathof, R., Krams, R. et al. MRI-determined carotid artery flow velocities and wall shear stress in a mouse model of vulnerable and stable atherosclerotic plaque. Magn Reson Mater Phy 23, 77–84 (2010). https://doi.org/10.1007/s10334-010-0200-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-010-0200-4