Abstract
The introduction of non-native species with various ecological and functional traits to European forests may be a potential tool for mitigating climate risks. We analyzed the growth sensitivity to climate of seven alien (Acer rubrum, Betula maximowicziana, Castanea sativa, Cryptomeria japonica, Metasequoia glyptostroboides, Thuja plicata and Tsuga heterophylla) and two native (Fagus sylvatica and Picea abies) tree species on a productive forest site in western Germany in order to assess their potential suitability in light of climate change. Retrospective basal area increment at breast height (BAI) and total stem volume increment (VI) were statistically related to a total of 11 multiscale climatic variables showing minor discrepancies between BAI and VI in climatic sensitivity. Across the tree species, the strongest climate signals were found for the standardized precipitation evapotranspiration index (spei), vapor pressure deficit (vpd) and the number of frost days (nfr) representing water availability, heat and coldness, respectively. Growth of most species was positively affected by mild winters and growing season spei. Importance of the investigated variables differed among tree species with, e.g., C. sativa and T. plicata being mainly driven by winter vpd and A. rubrum, C. japonica and P. abies by water availability in the growing seasons. We conclude that particularly more anisohydric species (e.g., C. sativa) may profit from their low sensitivity to drier conditions. Highly sensitive and isohydric species (e.g., A. rubrum) will undergo significant growth reductions when climate becomes more variable.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the last two decades, adaptive forest management increasingly focuses on strategies to mitigate climate impacts on tree vitality and survival in European forests (Neumann et al. 2017; Thurm et al. 2018) and on a global scale (Allen et al. 2010). The ongoing climate change comprising climatic extremes (i.e., heat, drought and frost periods; IPCC 2013) is likely to increase environmental risks of European forest ecosystems (Bolte et al. 2010; Walentowski et al. 2017) such as declines in biodiversity (Archaux and Wolters 2006; Anderegg et al. 2013), shifting species composition and distribution (Dyderski et al. 2018; Buras and Menzel 2019) and increasing tree mortality (Anderegg et al. 2015; Neumann et al. 2017). One (active) measure to mitigate such climatic risks includes the introduction and cultivation of non-native tree species potentially better adapted to future climate conditions (Bolte et al. 2009). The recommended strategy basically focuses on the extension of the spectrum of tree species with various (different) ecological requirements and susceptibilities in order to prepare the forest ecosystems to various environmental future constellations. Thereby, the need for fulfilling multiple forest ecosystem services (e.g., economic yield, silvicultural integration, non-invasiveness) should be taken into account (Bolte et al. 2010; Eilmann and Rigling 2012).
The complexity and variety of (endogenous and exogenous) factors driving species’ climate sensitivity exacerbate the selection of appropriate tree species mitigating potential effects of climate change. In this context, the (evolutionary) long-term adaptation according to their ecological amplitudes in the natural range (Kölling et al. 2009; Kölling 2013) as well as morphological and functional traits (Bréda et al. 2006; Aubin et al. 2018) are of special importance (see Table 1 for details). For example, species distributed under broad ecological amplitudes such as the North American species Acer rubrum (Hart et al. 2012), Thuja plicata (Antos et al. 2016) or Tsuga heterophylla (Hennon 2011) are expected to possess large adaptive capacity to variable climate conditions. In particularly, the “super-generalist” (Abrams 1998) Acer rubrum is one of the most abundant and widespread tree species in eastern North America occurring under a huge ecological amplitude (Walters and Yawney 1990) indicating high adaptive potential. On the other hand, physiological and morphological traits and strategies have been considered as major factors controlling species-specific climate tolerance (Table 1; e.g., Brodribb et al. 2014, McCulloh et al. 2014). For example, the mesophilic and ring porous South European Castanea sativa (Bottacci 2011) follows a water spending strategy (anisohydric) and continues transpiration and wood formation when water resources become scarce (Gomes-Laranjo et al. 2012). This results in a general higher drought tolerance (e.g., Niinemets and Valladares 2006; Hoffmann et al. 2018a) or adaptive potential to new climatic conditions (Gomes-Laranjo et al. 2012; Thurm et al. 2018; Buras and Menzel 2019), even though during extreme drought events the species may tend to over-transpiration with increasing synergic risks (Conedera et al. 2010). A further anisohydric species, Betula maximowicziana (Ishii et al. 2013), originating from Northern Japan (Kurata 1971), is contrastingly characterized as a drought-intolerant species (e.g., Niinemets and Valladares 2006; Ranney et al. 1991). However, Hoffmann et al. (2018a) demonstrated a high growth stability during the European summer drought in 2003 on a West German forest site. Besides its heart-root system allowing extensive water uptake and its large climate amplitude including a higher summer drought and frost tolerance compared to European Betula species (Stephan and Liesebach 2011), this may indicate a general lower climate susceptibility. In contrast, isohydric and coniferous species such as Japanese Cryptomeria japonica (Kumagai et al. 2009) close stomata in early stages of water stress resulting in carbon starvation and lower wood productivity (McCulloh et al. 2014), in particular at dry sites (Matsumoto et al. 2006). A further isohydric tree species, the Chinese Metasequoia glyptostroboides, is characterized as a drought sensitive tree species (Vann 2005; Brodribb et al. 2014). Water stress produces high rates of membrane damage and low rates of root activity (Vann 2005). However, the widely cultivated M. glyptostroboides prospering in many geographic and climatic regions of the world is classified as climate-tolerant species, presumable due to its possibility to develop extensive, deeper root systems and to inhabit broad climatic amplitudes including regional extremes (Shen 2011; Williams 2005).
Climate impacts on tree growth can be determined by retrospective analyses of increment data. External stress situations (e.g., drought, heat, frost) may cause either cavitation-induced xylem disfunctions or a progressive depletion of stored carbohydrates, which both affect variation in stem growth (Choat et al. 2012; Eilmann and Rigling 2012). Besides classical climate variables (i.e., precipitation and temperature), a variety of climatic variables characterizing physiological stress conditions such as drought indicators or indices (WMO and GWP 2016) and evaporative demand may be more suitable to quantify growth reactions to climate events (Friedrichs et al. 2009). In addition, a complex interrelation of various environmental factors should be taken into account (Friedrichs et al. 2009; Scharnweber et al. 2011). Furthermore, the climate impact strongly depends on the temporal scale at which the climatic phenomena affect tree growth. So far, few climate–growth analyses have dealt with multiscale (monthly, cumulative and seasonal) climatic effects (e.g., Lévesque et al. 2013; Stojanović et al. 2018), or analyses were restricted to few timescales in the growing season such as 3- and 6-month periods. Information about both, dominant temporal scales and pervasive climatic parameters, may be crucial factors to derive complex relationships between climate variability and species-specific response patterns (Friedrichs et al. 2009; Lévesque et al. 2013).
Breast height series data can be related to diameter at breast height (DBH) and basal area (BA) measurements acting as useful predictors for approximating tree and stand characteristics (e.g., biomass production and stand density) in conventional forestry (Latte et al. 2016). In addition, several allometric models confirmed that tree taper stabilizes around breast height resulting in balanced and homogeneous growth (Chhin and Wang 2005; Kublin et al. 2013). As a consequence, climate-related interpretations in dendroecological research underly the assumption that breast height series approximately reflect the whole stem growth pattern or response to external factors (Chhin et al. 2010). However, a small number of comparative studies tested this hypothesis for European (e.g., Bouriaud et al. 2005; Sohn et al. 2012; Hoffmann et al. 2018a) or North American (Chhin et al. 2010) forest stands indicating both similarities and inconsistencies in climate–growth relationships between radial and volume increment (see Latte et al. 2016 for synthesis). Due to allometric discrepancies, additional measures connecting climatic impacts to volume-related tree growth are needed to better understand the species-specific climate sensitivity.
In our study, we analyzed the climate sensitivity of seven alien tree species and two native European tree species on a productive forest site in western Germany. Detailed stem analyses were applied to generate whole stem volume increment from stem disks at various tree heights. We hypothesized that climate sensitivity of tree growth is related to at least one of the following species-specific criteria: (1) ecological amplitude in the native range, (2) physiological traits and/or (3) morphological (external and internal) traits. Thus, a variety of species differing in these criteria was selected (see Table 1 for details). The objectives were to (1) identify most important climate factors controlling growth across tree species that can help evaluate the sensitivity of species to climate change, (2) detect dominant climatic temporal scales triggering species-specific growth and (3) evaluate whether climate–growth responses at breast height are good estimators for the whole stem responsiveness. Note: The same dataset was used in a previous study to analyze the sensitivity to specific, isolated drought events (e.g., 2003) at the study site providing additional information on drought tolerance of the mentioned species (see Hoffmann et al. 2018a for details).
Materials and methods
Study site and species selection
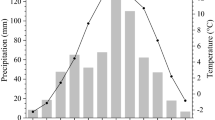
The study was carried out in the Arboretum Burgholz in North Rhine-Westphalia in western Germany (51° 13′ 7.226″ N, 7° 7′ 8.479″ E). In this forest district, different native and alien tree species have been cultivated under comparable site conditions since the late nineteenth century (Becker 2009). The Arboretum is characterized by advantageous climatic conditions (oceanic influence) for tree growth with high annual precipitation sums and a mild annual temperature average (1131 mm and 9.8 °C for the period 1960–2014, Fig. 1; DWD 2015). For the considered total growth period of the study trees (1960–2014), a clear warming trend during the last five decades was observed (Fig. S1). The forest soil in the Arboretum can be classified as a podzolic Cambisol, characterized by a poor to moderate nutrient supply, silty loam, good water supply and a mor humus layer.
a Location of the Arboretum Burgholz (blue cross), b distribution of the study stands in the Arboretum and c mean monthly precipitation sum (± standard error) and mean monthly temperature (maximum, mean, minimum) for the period 1985–2012 (DWD 2015). (Color figure online)
For this study, the alien tree species Acer rubrum L., Betula maximowicziana Regel, Castanea sativa Mill., Cryptomeria japonica D. Don, Metasequoia glyptostroboides Hu et Cheng, Thuja plicata Donn and Tsuga heterophylla (Raf.) Sarg. were selected due to their favorable economic criteria and to their purported high climatic tolerances (e.g., Schenck 1939a; Burns and Honkala 1990a, b; Roloff et al. 2011; Praciak et al. 2013) as a function of their ecological properties and functional traits (see Table 1 for details). Additionally, two economically important native species, Fagus sylvatica L. and Picea abies H. Karst, were chosen as reference species. Within the Arboretum, the different tree species were mainly established as monospecific to mixed stands (dominated by the target tree) of 23 to 75 m2 ha−1 in basal area (Table S1). Due to homogenous (soil and climatic) site conditions and the proximity of the selected stands (Fig. 1), the tree species grow under comparable environmental conditions. All stands were evenly aged (34–58 years) and had undergone the same silvicultural treatment (planting, thinning from above; Hoffmann et al. 2018b). The species names will be abbreviated by their genus name in the following (e.g., Castanea sativa = Castanea).
Growth data
We selected six to ten dominant or codominant (Kraft classes 1 and 2, Kraft 1884) trees per species. Cross-sectional stem disks were cut along the felled trees at 0.5 m, 1.3 m and subsequently every 2 m up to the tree top (max. height = 29.3 m). A total of 922 dried stem disks were progressively polished and scanned at 1200 dpi resolution (A3 scanner 2400S, Mustek). Annual radial increments were measured to the nearest 0.01 mm (Rinn 2006) along four radii at the main cardinal directions using LignoVision™ software (version 1.38e, Rinntech e. K., Heidelberg, Germany). Crossdating accuracy was checked visually and statistically by the parameters Gleichläufigkeit (Eckstein and Bauch 1969), t-value (Baillie and Pilcher 1973) and Gleichläufigkeit-significance (Rinn 2005) in a stepwise approach considering radial directions within a stem disk, different heights within a tree and different individuals per tree species (see Hoffmann et al. 2018a for details) using TSAP software (TSAP-Win professional version 4.68e Rinntech e. K., Heidelberg, Germany). A total of 19 stem disks of two trees that did not meet the statistical criteria were omitted from analyses.
Radial increment data were converted into (raw) basal area increment (BAIr) to increase representation of whole stem growth using high-dimensional units (Hinckley and Lassoie 1981; Biondi and Qeadan 2008). Furthermore, (raw) total stem volume increment (VIr) was calculated by the package treeglia using the function “StemAnalysis” (Bascietto and Scarascia-Mugnozza 2004) from the open-source software R (version 3.2.5, R Core Team 2016). Considering all height sections (stem heights) of individual trees, the application firstly interpolated annual stem heights (Carmean 1972). Annual volume increment was then computed according to both, Smalian’s and cone’s formulas (Bascietto and Scarascia-Mugnozza 2004). For comparative analyses, we restricted BAIr series data to measurements at standard breast height (1.3 m, in the following denoted by BAI). Additionally, VIr data were used as a reference for the total stem growth (Table 3). Differences between tree species in mean BAIr and VIr were assessed by analysis of variance (ANOVA; BAIr) and the nonparametric Kruskal–Wallis test by rank (VIr), since homogeneity of variance of residuals or normal distribution of the values were not given for mean VIr.
The individual BAIr and VIr series were detrended and standardized using a cubic smoothing spline (Cook and Peters 1981) with a 50% frequency cutoff at 67% of the series length (e.g., Zang et al. 2012; Restaino et al. 2016). Regarding the various tested detrending techniques, our results have been found to be robust if using fixed spline wavelengths (e.g., 10 years). We performed autoregressive modeling of the standardized series to remove first-order autocorrelation validated by Akaike information criterion (AIC). The resulting detrended residual series of each increment variable (BAI and VI) and tree species were averaged to site chronologies by a robust biweight mean (Cook 1985). Thus, a total of 18 chronologies for two increment variables and 9 tree species were constructed (Fig. 2). The common growth period from 1985–2012 was selected for statistical climate–growth analyses in order to reach comparability among species in series length. The chronologies were finally evaluated and compared using the descriptive statistics mean sensitivity (ms, a measure to quantify chronologies’ interannual variation; Fritts 1976), effective chronology signal (es, an estimator to quantify signal strength between and within trees; Cook and Pederson 2011), expressed population signal (eps, which determines the representation of the theoretical population by the chronology; Buras 2017) as well as Pearson correlation coefficient between VI and BAI chronologies (RVI) and across the tree species. For consideration of temporal changes in the relationships of the chronologies, the common period was divided into two additional periods: 1985–1997 and 1998–2012.
Climate data
All climate data were accessed from the closest meteorological station “Wuppertal-Buchenhofen” (ID 5717, 51° 13.488′ N, 7° 6.318′ E, 130 m a.s.l.; National Meteorological Service of Germany, DWD 2015), located in the center of the Arboretum Burgholz (approximately 2.3 km distance to the furthermost study stand, Fig. 1). The climate records from 1960–2014 included daily average, maximum and minimum temperatures (tavg, tmax and tmin in °C), precipitation sums (prec in mm) and vapor pressure (vp in hpa). tavg, tmax and tmin were averaged to monthly values for climate–growth analyses. Additionally, the following physiological important monthly variables were computed: potential evapotranspiration (pet), vapor pressure deficit (vpd in hPa), degree day sum (> 5 °C and > 10 °C, sum∆t05 and sum∆t10), number of frost days (nfr), de Martonne aridity index (dmi; de Martonne 1926) and the standardized precipitation evapotranspiration index (spei; Vicente-Serrano et al. 2010; Table 2). For details on climate variables including calculations and individual cross-correlations, see Supplementary Information (SI Methods, Appendix 2: Climate variables; Eq. S1, S2; Fig. S2–S4; Table S2).
In addition to the monthly timescale, 2- to 6-month seasonal periods of climate variables were considered by averaging tavg, tmax, tmin, pet, vpd and dmi values, and summing prec, sum∆t05, sum∆t10, spei and nfr data ranging from April of the previous growth year to September of the current year. To account for potential vulnerability to coldness and (late) frost, tmin and nfr variables were restricted to the cold months from October of the previous year to April of the current year. Here, we excluded May data since solely two years (1996 and 2012) with one frost day each were detected. In total, 11 climate variables summing up to 1011 single- and multimonth possible predictor variables were obtained for climate–growth analyses.
The 11 climate variables were assigned to one of the three general categories according to its climatic origin and relevance for climate compatibility or vulnerability: (1) water availability (indicator for drought tolerance or demand), (2) temperature (indicator for heat tolerance or demand) and (3) frost (indicator for cold and [late] frost tolerance or demand; Table 2).
Climate–growth analyses
Climate–growth relationships between the species-specific BAI and VI chronologies and single monthly and seasonal climate variables were assessed using classical correlation functions. Bootstrapped correlation coefficients (bcc) according to Biondi and Waikul (2004) with a 95% confidence interval were quantified by the R package treeclim (Zang and Biondi 2015) for the common period 1985–2012. To identify the most contributing climate factors across tree species, we firstly extracted lowest negative and highest positive bcc by species and growth variable. The highest total frequency of a climate variable (among species and increment variable) within each category (water availability, temperature or frost) was then assumed to be most appropriate to explain tree growth across species.
Relationships between the growth chronologies (BAI and VI) and multiple climate variables were modeled using multiple stepwise regression analyses with forward selection for the same period 1985–2012. For these analyses, we used standardized climate variables (except for nfr) where the mean = 0 and standard deviation = 1 (Latte et al. 2016). The standardization enhanced the comparability of individual regression terms. The initial linear regression with growth data was performed using the “dredge”-function of the R package MuMIn (Barton 2016) for each climate variable and timescale (single- to 6-month) to preselect best-fit combinations of months (model selection of best-fit months of individual timescales). Here, the maximum number of regression terms in a single model was set to 4 and the regression model was selected by minimum AICc (Akaike information criterion, corrected for small sample sizes; Burnham and Anderson 2010). The best-fit predictor variables (months of individual timescales) were secondly merged for all timescales of individual climate variables (months of all timescales) used for subsequent linear regression with growth data. The regression models were determined by the same above-mentioned analyses with selection (“dredge,” maximum number of terms = 4) of variable sets by minimizing the value of AICc (model selection of best-fit months of all timescales we will name subset). We thirdly applied the function “selMod” (R package pgirmess; Giraudoux 2017) to compare the selected subset models of different climate variables within the predefined categories (water availability, temperature and frost; model selection of best-fit climate variable). Similar to correlation functions, the climate variable with highest frequency within a category was assumed to be the best explaining climate variable across the tree species. Final regression models were determined by combining the pool of significant variables of the three categories using the “dredge”-function (maximum number of terms = 9). To identify and, consequently, reduce multicollinearity in models, we used stepwise variance inflation factor VIF selection (Zuur et al. 2007). Here, climate variables with highest VIF values were removed stepwise. The selection was repeated until all VIF values fell below the defined threshold of VIF = 1.5. The relative importance of an individual predictor variable (RIV) in each regression model was calculated by the function “calc.relimp” of the package relaimpo (Grömping 2006) using the “lmg”-method (i.e., R2 contribution averaged over orderings among the regressors, e.g., Chevan and Sutherland 1991).
Results
Growth chronologies
For the period 1985–2012, mean (raw) BAIr differed significantly from 12.2 (Acer) to 34.7 (Thuja) cm2 a−1, and mean VIr varied significantly from 10.9 (Acer) to 59.9 (Tsuga) dm3 a−1 among tree species (Fig. 2; Table 3, all p < 0.001). The eps and es values of the detrended series ranged from 0.88 to 0.97 and 0.48 to 0.81, respectively, indicating a strong common signal and within- and between-tree correlation (Table 3). Comparing the increment variables, ms varied between the investigated species, with highest ms for BAI measures for six of the nine tree species. VI chronologies highly corresponded to BAI series presenting high correlation coefficients within individual species (Table 3, RVI = 0.77–0.96, p < 0.001). Comparing tree species, the correlation between BAI (R = − 0.02–0.69) or VI (0.20–0.77) chronologies varied strongly across individual species with highest similarity for Cryptomeria and Picea trees (Fig. S4). The overall mean correlation across species’ VI chronologies (1985–2012: R = 0.52) increased from R = 0.28 (1985–1997) to R = 0.64 (1998–2012), indicating a higher synchronicity and growth response to similar climatic factors with increasing tree age (see Hoffmann et al. 2018a for detailed analyses for the period 1998–2012).
Climate–growth relationships
Climate factors across tree species
Bootstrapped correlation functions in association with multiple linear regressions revealed species-specific differences for correlation and response patterns of the growth indices (BAI and VI) to climatic variables. Thus, growth was specifically driven by individual climate variables at individual month(s) and timescales (see Table S3 for details as an example for the correlation functions). Across the tree species and increment variables (BAI, VI), climate–growth analyses identified spei to be most important within the category water availability (including also prec and dmi) showing the largest number of highest bootstrapped correlation coefficients (bcc) and of best explaining variables, respectively (Table 4). For the six primarily temperature-controlled variables (tavg, tmax, pet, vpd, sum∆t05, sum∆t10), the most frequently and strongest relationships with tree growth were found for vpd. Within the climatic category frost (nfr, tmin), we found nfr as an overall reliable frost-indicator associating single and multiple climate–growth relationships (Table 4).
Species-specific response to single climate variables (correlation functions)
For BAI and VI detrended series, bootstrapped correlation functions indicated Betula, Castanea, Fagus and Thuja to be less sensitive to spei when compared with the remaining species (Fig. 3; see also Fig. S5; Table S4). Growth of Tsuga, Cryptomeria, Picea and Acer (July) trees was negatively correlated with growing season vpd of the current year. For Metasequoia, Acer and Betula, nfr during October to April strongly inhibited the increment variables (BAI or VI; most frequently in January to April). In total, highest number of Pearson’s correlation coefficients was found for Fagus (n = 105, primary positive relationships with previous vpd) as well as Metasequoia (n = 72) and Acer (n = 68), both revealing multiple correlations with spei, vpd and nfr. Lowest number of relationships was identified for Castanea (n = 12), Betula (n = 27) and Thuja (n = 51) trees indicating a general lower sensitivity to various climate constellations.
Species-specific correlation coefficients between increment variables (basal area increment, BAI, and volume increment, VI) and monthly (timescale = 1) or seasonal (timescale = 2–6) standardized precipitation evapotranspiration index (spei), vapor pressure deficit (vpd) and the number of frost days (nfr) for the period 1985–2012 (p < 0.05). Lowercase abbreviations (a–d) denote months of the previous year (April–December), and uppercase abbreviations (J–S) indicate months of the current year (January–September). The month(s) and timescale at which the highest correlation was reached by species, increment variable and climate variable are framed in black. In the case of seasonal overlapping time windows, the respective subsequent correlation coefficients are indicated. (Color figure online)
Multiple climate–growth relationships
Taking into account combinations and the partial contributions of the three selected climatic variables (spei, vpd, nfr), multiple regression models demonstrated significant coefficients of all predictor variables (p < 0.05) after variance inflation factor (VIF) selection (Table 5; see Table S5 for the relative importance values of predictor variables, RIV).
The species Castanea, Thuja, Fagus and Tsuga were predominantly affected by the temperature-based vpd. In case of Castanea, vpd in December to January was the most influential factor according to the ranking of RIV (Table 5; Fig. 4). In addition, with positive effects of spei in current July on BAI and VI, climate variables explained the lowest variance in growth (29% and 36% for BAI and VI, respectively) when compared with the other tree species. VI of Thuja trees was predominantly facilitated by high winter vpd (January), followed by vpd in April–May of the previous and spei in spring (March–July) of the current year. As demonstrated by the single correlation functions (Fig. 3), vpd prior to the year of wood formation was the most promoting factor for Fagus trees (in combination with winter vpd: R2 = 0.64 and R2 = 0.58). Growth of Tsuga was primarily negatively affected by vpd in summer of the current year (July) revealing strong relationships between increment and together with secondary climate variables (summer or winter spei and vpd in January; R2 = 0.53 and R2 = 0.55).
Graphical illustration of the linear regression models relating species-specific increment variables (incr) to the best explaining climate variables (spei, vpd, nfr) at the month(s) and timescale of the year at which the lowest AICc was reached. For each model, climate variables promoting growth (positive relationships) are presented in blue, and variables inhibiting growth (negative relationships) are highlighted in red. The numerals (1, 2, 3) indicate the hierarchical importance of the climate variable as a function of its listed relative importance of the predictor variable (RIV) in each regression model in descending order (1 = highest RIV, 3 = lowest RIV, all p < 0.05). Lowercase abbreviations (a–d) denote months of the previous year (April–December), and uppercase abbreviations (J–S) indicate months of the current year (January–September). R2 = multiple R2. Significance levels of the models: p < 0.001***, p < 0.01**, p < 0.05*. (Color figure online)
In contrast, growth of Acer, Cryptomeria, Picea and Betula was triggered more dominantly by water availability (i.e., values of spei, Fig. 4; Table 5). spei in late summer of the current growth year promoted growth of Acer trees. In combination with a negative growth response to nfr in January (VI) and positive effects of winter or spring vpd, strong climate–growth relationships were found (R2 = 0.54 and R2 = 0.49). Climate response patterns of Betula were most different to the other tree species. High water availability (spei) in the current August (primarily VI and subordinately BAI) as well as in preceding December (BAI) negatively affected growth with secondary negative (early spring nfr) or positive (winter vpd positively) influencing variables of lower importance. Growth of Cryptomeria was predominantly related to 6-month water availability (spei) in February–July in the year of tree ring formation with second-order variables being winter and previous spring vpd. Similarly to Cryptomeria, annual increment of Picea was limited by spei during growing seasons in the prior (BAI and VI) and current year (VI) as well as vpd in the preceding November to January.
Multiple regression analyses of Metasequoia confirmed the observed vulnerability of growth to frost occurrence (nfr) in February–April (Fig. 4). Furthermore, summer spei in both, previous and current years, positively influenced the annual increment.
Differences between breast height and stem increment in response to climate
Climate sensitivity slightly varied between BAI and VI depending on the tree species (Figs. 3, 4). However, across tree species no clear trend in growth response to climate regarding the considered increment variables was found. VI slightly differed in terms of the greater impact of temperature (vpd) in the spring and winter season prior to the year of wood formation (Acer, Betula, Castanea, Fagus, Picea, Thuja, Tsuga). Furthermore, VI appeared to be marginally more sensitive to water availability (spei) in the current growing season (Acer, Betula, Picea, Thuja) with no sensitivity of VI to current year summer spei for Fagus and Tsuga trees. BAI tended to be more affected by water availability (spei) in the preceding growing season (Picea, Thuja) or by nfr (Betula) when compared with VI. The overall strongest discrepancies in climate sensitivity between BAI and VI were found for Betula, Picea and Thuja trees (ΔR2 = 0.09, 0.12 and 0.09, respectively, Fig. 4). The lowest discrepancies in response to climate between BAI and VI were observed for Cryptomeria (ΔR2 = 0.01) and Tsuga (ΔR2 = 0.02) trees (Fig. 4).
Discussion
Our analysis confirmed that growth was significantly driven by climatic factors that function at different time periods at the mesic forest site. Temperature-controlled vapor pressure deficit (vpd) in preceding cold periods tended to be the most important driver across the tree species for current growth. Additionally, water availability represented by the standardized precipitation evapotranspiration index (spei) in the growing seasons (prior and current to the year of wood formation) strongly affected tree growth.
Effects of climate factors on tree growth
Our findings underline the effects of vapor pressure deficits (vpd) on tree growth observed by different authors. Williams et al. (2012) and Restaino et al. (2016) reported a consistent and strong impact of growing season deficits on the growth of coniferous tree species throughout western US forests. In contrast to temperature that limits growth as a function of decreasing photosynthesis at given thresholds of leaf temperature, vpd is linked to stomatal closure which ultimately reduces growth due to prevented CO2 uptake (Restaino et al. 2016). Thus, growing season vpd seems to be a reliable indicator for physiological plant stress that is more directly associated with tree productivity. However, the inverse and positive effects of vpd in non-growing season do not reflect the stress-related evaporative demand, particularly astonishing for deciduous species ceasing photosynthesis in winter. For the coniferous evergreen species, the lower saturation pressure in winter months (in comparison with growing season) and consequently weaker deficit might be sufficient to induce transpiration processes without drought damage due to available water supply at the study site. In this regard, vpd might be as low to prevent stomatal closure. Because of the nonlinear (exponential) response of saturation vapor pressure to temperature, increasing temperature increasingly affects vpd given constant relative humidity and even more strongly given constant absolute humidity. This is why we consider vpd as a matrix for temperature. The strong sensitivity of vpd to air temperature (tavg; Fig. 3) might be a further generalized explanation for the across species’ dependency on high vpd in cold months. The importance of winter temperatures and temperature-related variables to tree growth had been reported for various tree species in Central (e.g., Feliksik and Wilczyński 2009; Lebourgeois et al. 2010; van der Maaten 2012; Land et al. 2017) and Southern Europe (e.g., Granda et al. 2013; Martin-Benito et al. 2013; Żywiec et al. 2017). The general positive growth response to winter vpd might be related to decreasing winter embolism with increasing temperature in cold months (Lebourgeois et al. 2010; van der Maaten 2012). The risk of cavitation restraining water transport processes is of larger importance for ring porous tree species with large vessels (Améglio et al. 2002) such as Castanea sativa. In diffuse-porous species, the number of embolized xylem conduits can either increase (e.g., in Acer or Fagus trees) or decrease during winter, while coniferous species are less vulnerable to winter embolism (Améglio et al. 2002). A warm winter may further encourage soil warming and mycorrhizal root growth as well as promote bud burst phenology and needle and shoot maturation, thus prolonging the growing season of the following year (Lebourgeois et al. 2010). As supported by the findings of Restaino et al. (2016), the lagged impact of temperature-driven variables such as vpd in non-growing seasons is higher for regions where water is less limiting. Thus, the relatively high dependence of tree growth on previous vpd may be related to the general high water supply of our study site.
An increasing number of comparative dendroecological studies of various tree species recently confirmed that water availability and drought stress during the growing season rank among the dominant climatic factors driving growth of European forests (e.g., Zang et al. 2014; Bhuyan et al. 2017; Vitali et al. 2017; Stojanović et al. 2018). The use of extended drought indices considering such physiological stress conditions has greatly increased. The applied standardized precipitation evapotranspiration index (spei) benefits from the involvement of a water balance giving thresholds for regional water availability above or below average conditions for a given climate record (Vicente-Serrano et al. 2010). However, recent studies emphasize that negative spei do not represent sites’ water shortage per se, suggesting not to apply spei as a standalone index for detection of drought severity, especially in a global context (Zang et al. 2020). In this study, the response to spei particularly differed between tree species with respect to the temporal scale of water availability or shortage, respectively (Figs. 3, 4). When water becomes low, xylem water potentials decrease, and leaf stomata closure causes a rapid cessation of photosynthetic CO2 assimilation (Choat et al. 2018). As a consequence, carbon storage and wood formation are inhibited. Over longer drought periods, low photosynthesis rates in association with cavitation-induced gas emboli throughout the xylem reduce hydraulic conductance and water transport, and carbohydrate pools can be depleted (Choat et al. 2018). Since strategies to avoid water stress (e.g., anisohydric or isohydric) strongly differ among tree species (e.g., Thomsen et al. 2013; Fardusi et al. 2016) as a function of drought duration, this emphasizes the need for considering cumulative effects of water availability. As reported by Bhuyan et al. (2017), spei of short- and intermediate-term scales seems to be appropriate to capture drought signals of tree growth in temperate forests.
In dendroecology research, the rare application of the number of frost days (e.g., Gazol et al. 2019) or derived spring frost measures based on accumulated degree day sums (e.g., Vanoni et al. 2016; Vitasse et al. 2019) may be linked to the fact that this meteorological parameter simplifies the absolute values of (minimum) temperature records. Furthermore, calibration (of frost day signals in growth series) over summer months is impaired due to lack of days below freezing point. However, our analyses demonstrated that the cumulative occurrence of frost can have significant impacts on tree growth when restricted to cold month including spring data (Figs. 3, 4). As frost events are expected to become more frequent in earlier starting and longer lasting growing seasons, especially in Europe (Liu et al. 2018), this variable will gain increasing importance in the future when evaluating tree response to climate.
In total, interactions of climate parameters considering water- and temperature-related (including frost occurrence) conditions seem to play a major role in species-specific growth dynamics (Table 5; Fig. 4). The combination of high vpd in preceding winter periods with high water availability during growth periods was the most significant complex of factors in the growth variation across the tree species. On the other hand, the multiple approaches demonstrated individualistic climate response of species to a unique set of climate parameters.
Species-specific response to climate factors
The climate–growth analyses revealed species-specific patterns with respect to growth dominating climatic parameters and timescales. Tree growth of Castanea, Thuja and Fagus was mainly affected by vpd in winter or spring (Fagus) with slightly lower dependency on spei. For Acer, Cryptomeria and Picea, water availability during growing seasons was the dominant factor facilitating tree growth. High vpd in current July reduced growth of Tsuga, and Betula was negatively affected by high water availability in current August. Growth of Metasequoia was strongly limited by frost occurrence in the beginning of a year. These climate–growth patterns may result from specific ecological properties or morphological and functional traits. The species’ water-use strategies seem to be a crucial factor regarding the sensitivity to water availability and drought. Thus, anisohydric and water spending species such as Betula, Castanea and Fagus were generally less affected by spei in comparison with the isohydric species. This effect can be supported by morphological traits such as the development of deep root systems facilitating the exploitation of water resources. The long-term adaption due to the ecological amplitudes might additionally control the species-specific climate sensitivity, e.g., when the mesic conditions of the study site coincide with optimum climate conditions of a species’ range (e.g., Fagus). On the other hand, if the study site represents the ecological distributional edge of a species (e.g., in terms of temperature), a higher sensitivity was found (e.g., sensitivity to frost of Metasequoia). As the individual characteristics may specifically trigger climate sensitivity, all criteria should be considered. For a comprehensive assessment of species’ climate vulnerability, we further suggest the incorporation of all three analyzed climatic aspects weighted in the following order: water availability (spei), temperature (vpd) and frost (nfr).
Our analyses shows a positive effect of water availability on anisohydric Castanea sativa growth only in late summer, while the positive effect of winter vpd was stronger (Fig. 4). Due to its affinity on high summer temperatures (22–30 °C, Gomes-Laranjo et al. 2012) or degree days (Bottacci 2011), Castanea is expected to shift northwards to Central Europe where future conditions will become more favorable, e.g., in terms of milder winters (Broadmeadow et al. 2005; Thurm et al. 2018; Buras and Menzel 2019). In total, Castanea trees provided the lowest sensitivity to multiple climate factors with relatively low dependency on water availability.
For a further anisohydric species, Betula maximowicziana, water availability even affected tree growth negatively (Fig. 4). At mesic sites where water is less limiting such as the study site Burgholz, oversupply of precipitation and waterlogging can cause oxygen deprivation inhibiting root functioning and growth with negative feedbacks on transpiration and photosynthesis, leading to potential growth losses (Repo et al. 2016). The high sensitivity to waterlogging of Betula species such as Betula pendula (Wang et al. 2016) and, in particular, Betula maximowicziana (Niinemets and Valladares 2006) may be linked to our observed negative impact of spei in August. This, in turn, indicates a lower demand for water and coincides with the reported high resistance to extreme summer drought in 2003 with particularly low precipitation amounts in August (Hoffmann et al. 2018a).
Additionally, the European native Fagus sylvatica seems to benefit from its water spending (anisohydric) behavior (e.g., Pretzsch et al. 2013) resulting in a low growth response to water availability (Figs. 3, 4). As shown in a previous study (Hoffmann et al. 2018a), beech trees were of rather medium to high drought tolerance supporting the secondary dependence on water availability at the study site. In contrast, temperature and vpd in the preceding year strongly triggered tree growth. These (first-order) lags between temperature and growth response of Fagus had been observed in several studies. Thus, temperature in months of the preceding summer to autumn (e.g., Čufar et al. 2008; Castagneri et al. 2014; Dulamsuren et al. 2017) or winter (e.g., Lebourgeois et al. 2005; van der Maaten 2012; van der Maaten-Theunissen and Bouriaud 2012) positively influenced Fagus growth at different European forest sites. However, no further analyses supporting such (positive) ongoing heating effects were found. We hypothesize that the mesic study site with deep soils and high precipitation amounts reflects nearly optimal climate conditions for Fagus trees where additional heat and transpiration do not limit (stress), but facilitate photosynthesis and wood formation. However, the currently increasing number of observations of beech crown dieback after the extraordinary summer drought in 2018 (e.g., Asche and Meier zu Beerentrup 2019; Buras et al. 2020) may indicate a rapid increase in drought vulnerability when such climate extremes accumulate. This “new phenomena” may particularly hold for shallow and sandy soils with low water holding capacity (personal observation) and is in line with projected decrease in beech abundance over large parts of Central Europe (Buras and Menzel 2019).
In our analysis, water availability of the isohydric (McCulloh et al. 2014) and drought-resistant (Grossnickle and Russell 2010; Hoffmann et al. 2018a) Thuja plicata was of lower importance for tree growth with little response of volume increment (Fig. 4). Comparable to the growth reaction of Fagus, the greater importance of vpd in the previous year may be related to the favorable mesic conditions of the study site facilitating ongoing transpiration and photosynthesis. As reported by Schaberg et al. (2010), root sensitivity to frost damages due to a limited cold tolerance and shallow rooting may further explain the positive effect of vpd in winter months. The wide ecological amplitude of the shadow-tolerant (Minore 1990) Thuja trees might furthermore explain the general lower climate sensitivity (Fig. 4).
For Tsuga heterophylla, our findings confirm the frequently observed strong sensitivity (negative influence) to growing season temperature (Ettinger et al. 2011) and vpd (Hoffmann et al. 2018a) with highest impact in July (Figs. 3, 4). Heat-induced evaporative demand and subsequently early stomatal closure (isohydric) seem to be of special importance for the growth of Tsuga trees. Furthermore, water availability (spei) in the current growth year with focus on the vegetation period is a further climatic factor limiting annual tree growth. For the mesic study site, the observed strong climate sensitivity does not coincide with findings primary classifying Tsuga as a rather climate-tolerant tree species (Ettinger et al. 2011) due to its distribution under broad ecological amplitudes (Burns and Honkala 1990a).
Climate–growth relationships of Metasequoia glyptostroboides indicate a strong sensitivity to various climate constellations (particularly given by the high number correlations; Fig. 3) which does scarcely match with its common characterization as a climate-tolerant tree species (Williams 2005; Shen 2011). Our findings suggest a strong limitation of annual increment in response to frost occurrence in January to April of the current growth year (Figs. 3, 4). Thus, the reported risk to late frost damage due to early bud break (Burrichter and Schoenwald 1968) could be partially confirmed, while its cold tolerance could not (Williams 2005). The reduced drought tolerance (Vann 2005) is furthermore reflected by the growth response to spei in the previous growing season. However, negative effects of (too) strong transpiration (indicated by high vpd) on tree growth (Burrichter and Schoenwald 1968) were not found and may be explained by the general good water supply (Fig. 1).
The response patterns of European native Picea abies to climate parameters largely corresponded with those of Cryptomeria trees (Fig. 3). This isohydric species has widely been classified as susceptible to water stress during drought in European forests (e.g., Pretzsch et al. 2013; Zang et al. 2014; Vitali et al. 2017) and is expected to largely disappear from Central European lowlands under future scenarios (Buras and Menzel 2019). Our analysis suggests that accumulated water availability (long-term spei) during the growing season is the dominant factor driving tree growth, even for sites with a favorable water supply (Fig. 4).
The strong sensitivity to water availability during growing seasons (Fig. 4) of the isohydric Cryptomeria japonica corresponds with findings (or classifications) of various studies and expert assessments (e.g., Schenck 1939b; Schober 1978; Matsumoto et al. 2006; Tange et al. 2009). Therefore, its high water demand, but low ability to transport water from the soil according to its large hydraulic resistance (Matsumoto et al. 2006), may explain the observed drought vulnerability (Hoffmann et al. 2018a). Besides strong regulation of leaf water potential (Kumagai et al. 2009), a shallow root system (Konôpka et al. 2006) additionally inhibits water uptake from deeper soil layers during drought. For Cryptomeria trees, high values of vpd in the current growing season may induce physiological stress caused by high evaporative demand (Fig. 3). However, mild conditions in the preceding winter positively influenced growth, which may be related to reduced winter embolisms or prolonged vegetation periods.
Against common characterizations as an adaptable, undemanding and climate-tolerant tree species (Walters and Yawney 1990; Abrams 1998), Acer rubrum surprisingly tended to be very sensitive to all investigated climatic parameters (spei, vpd and nfr, Figs. 3, 4). Growth was strongly driven by water availability in summer that may be related to the early cease of transpiration by stomatal closure when water becomes short (isohydric, Thomsen et al. 2013). vpd in winter largely promoted growth, whereas the occurrence of frost had a negative effect. This resulted in a complex interaction of climatic conditions indicating uncertainty in estimates of growth response. Our findings strongly differ from results of Teets et al. (2018) concluding Acer to exhibit a greater plasticity to fluctuating climate conditions due to the lack of significant climate–growth relationships in the most northeastern USA (Maine). However, findings of Hart et al. (2012) in the more central eastern USA (Tennessee) indicate some accordance with our results as the authors found a positive growth response to temperature in winter and to precipitation in spring, but negative correlations with summer temperature.
Comparison of breast height and stem volume response to climate factors
Overall, BAI and VI responded similarly to climate events. Climate sensitivity only marginally and species-specifically differed within trees. We found no universal response pattern that would give indications for growth allocation in relation to climate factors. VI appeared to be marginally more sensitive to temperature-driven vpd in spring or winter of the previous year and to water availability (spei) in the current year growing season. BAI tended to be slightly more sensitive to spei in the previous year growing season. Considering that VI represents growth response in upper stem segments stronger when compared with BAI (as VI incorporates a relatively higher amount of increment measurements above breast height), two general mechanisms may explain growth allocation (e.g., Gower et al. 1995; Sevanto et al. 2003; Ryan et al. 2006): 1) carbon allocation and 2) hydraulic limitations:
During unfavorable growth conditions (e.g., drought), reduced carbon assimilates are used for respiration first, and remaining assimilates are exploited for wood formation. Upper stem parts, close to the organs of photosynthesis (foliage), profit most (allocation at tree tops) and the basipetal (top-down) transport of carbohydrates is reduced (Gower et al. 1995; van der Maaten-Theunissen and Bouriaud 2012). Consequently, relatively low growth rates at lower stem sections lead to higher growth variations under more variable and unfavorable conditions. The lagged growth response at breast height to spei may be related to a reduced availability of carbohydrate reserves and limited allocation to the lower stem. On the other hand, during favorable conditions represented by mild winter and spring (high vpd), the maximum growth shifts downwards resulting in a relative increase in increment on the lower bole (Larson 1963) or, in turn, in a higher sensitivity of upper stem represented by VI.
In contrast, hydraulic and gravitational limitations are proposed as a potential mechanism reducing water availability at the top of tall trees (Ryan et al. 2006; Kerhoulas and Kane 2012; Latte et al. 2016). Thus, climate sensitivity is expected to increase with stem height (Kerhoulas and Kane 2012) and may explain the observed stronger sensitivity of species’ VI to current year spei.
Our findings suggest that mechanisms (carbon allocation and hydraulic limitation) driving growth allocation are not uniform across tree species and may interact (Ryan and Yoder 1997). Allocations may be further influenced by other factors such as microclimate, fruit production and cambial reactivation and cessation (Chhin et al. 2010; van der Maaten-Theunissen and Bouriaud 2012; Latte et al. 2016). The general strong allometric relationships between increment at breast height and of the whole stem support the overall similarity of growth response to climate parameters for BAI and VI (corresponding with Corona et al. 1995; Latte et al. 2016). In spite of marginal species-specific differences in climate sensitivity between BAI and VI, radial growth at breast height seems to be a reliable estimate for tree sensitivity.
Methodical limitations
We consider our results to be representative for mesic forest sites with a strong oceanic influence and deep, well water-supplied, but nutrient-poor soils as represented by our Arboretum site (Fig. 1). Thus, implications regarding a general growth behavior of the tree species should be limited to comparable site conditions. The inclusion of site-specific gradients by extension of study sites with, e.g., decreasing water supply or altitudes (e.g., Vanoni et al. 2016; Dulamsuren et al. 2017) would be helpful to derive species-specific climatic site suitability. Further physiological mechanisms difficult to reconstruct such as fruit production and defense mechanisms can influence and superimpose growth responses to climate (Hacket-Pain et al. 2015, 2018).
On chronology-level, the climatic signal component is strongly affected by the number of observations and period length (Schweingruber 1983). In this study, new information about whole stem response to climate had been derived at the expense of the sample size. Despite its general reliable signal strength (estimated by, e.g., eps), a short series length may additionally mask a certain sub-period referring to juvenile growth. Thus, superimpositions of age-related effects on species’ climate response in the first years cannot be completely excluded hindering the transferability to old-growth forest stands (in particular for the younger stands of M. glyptostroboides or B. maximowicziana). We applied a similar detrending method (67% spline) to maintain a maximum comparability across tree species.
Our study only weakly considered the influence of tree size and stand density (including competition) on annual stem growth, even though these factors may bias climate signals in growth series (e.g., Zang et al. 2012; Sohn et al. 2016; Pretzsch et al. 2018). As our aim was to minimize local endogenous and stand level exogenous effects on increment by selecting dominant individuals, we emphasize to take these factors into account when analyzing climate susceptibility of different tree species in future studies.
Conclusion and implications for alien tree species
Focusing on site-specific climate factors, the parameters spei, vpd and nfr were the most important climatic variables explaining growth patterns across seven alien and two native tree species at a productive study site in western Germany. By finding universal variables for describing climate sensitivity, tree responses to climate can be compared between species. We could show that mild winter conditions strongly promote tree growth of several species at mesic forest sites where water availability is less limiting. However, cumulative water shortage in the growing seasons may negatively affect tree growth.
Castanea sativa and Betula maximowicziana showed the lowest sensitivity to climatic factors, may therefore exceed climate tolerance levels of native species and may show great potentials to future wood production facing climate change. For B. maximowicziana, it was further shown that extreme drought events such as the European summer drought in 2003 are likely to be compensated for (Hoffmann et al. 2018a). For both species, growth seems to be promoted by a water spending, anisohydric physiology during drought when considering the investigated time period. Furthermore, the anisohydric Fagus sylvatica seems to benefit from a relatively low vulnerability to variations in water availability in our dataset. For these three anisohydric species, however, increasing (in frequency and strength) severe drought may lead to irreversible damages of essential organs with follow-up dieback phenomena as already observed for European beech stands (particularly at shallow soils). Due to high drought tolerance (Hoffmann et al. 2018a) with major limitations of growth by vpd, Thuja plicata and Tsuga heterophylla can be classified as tree species with moderate climate sensitivity. Metasequoia glyptostroboides, Picea abies, Cryptomeria japonica and Acer rubrum, on the other hand, revealed a strong sensitivity to complex climate conditions and are least suitable among those species that we investigated to mitigate climatic risks.
Based on this study and responses to drought investigated by Hoffmann et al. (2018a), the investigated species can be ranked by an increasing climatic susceptibility as follows: C. sativa < B. maximowicziana < T. plicata < F. sylvatica < T. heterophylla < M. glyptostroboides < P. abies < C. japonica < A. rubrum . Our species assessment may facilitate decision making when it comes to increasing the share of non-native species in European temperate forests.
References
Abrams MD (1998) The Red Maple Paradox. Bioscience 48:355–364
Allen CD, Macalady AK, Chenchouni H et al (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manag 259:660–684
Améglio T, Bodet C, Lacointe A, Cochard H (2002) Winter embolism, mechanisms of xylem hydraulic conductivity recovery and springtime growth patterns in walnut and peach trees. Tree Physiol 22:1211–1220
Anderegg WR, Kane JM, Anderegg LD (2013) Consequences of widespread tree mortality triggered by drought and temperature stress. Nat Clim Change 3:30–36
Anderegg WR, Hicke JA, Fisher RA et al (2015) Tree mortality from drought, insects, and their interactions in a changing climate. New Phytol 208:674–683
Antos JA, Filipescu CN, Negrave RW (2016) Ecology of western redcedar (Thuja plicata). Implications for management of a high-value multiple-use resource. For Ecol Manag 375:211–222
Archaux F, Wolters V (2006) Impact of summer drought on forest biodiversity. What do we know? Ann For Sci 63:645–652
Asche N, Meier zu Beerentrup H (2019) Buchensterben im Raum Detmold – Trocknis und Buchenborkenkäfer als Ursache? Report available via https://www.waldwissen.net. Accessed 9 March 2020
Aubin I, Boisvert-Marsh L, Kebli H et al (2018) Tree vulnerability to climate change. Improving exposure-based assessments using traits as indicators of sensitivity. Ecosphere 9:e02108
Baillie MGL, Pilcher JR (1973) A simple crossdating program for tree-ring research. Tree-Ring Bulletin 33:7–14
Barton K (2016) MuMIn: multi-model inference. R package version 1.15.6. Available via https://CRAN.R-project.org/package=MuMIn. Accessed 30 Aug 2018
Bascietto M, Scarascia-Mugnozza G (2004) A collection of functions to determine annual tree carbon increment via stem-analysis. Ann For Sci 61:597–602
Becker A (2009) Wie es zum Anbau der fremdländischen Baumarten in Burgholz kam. In: Landesbetrieb Wald und Holz NEW (ed) Burgholz - Geschichte und Perspektiven eines Versuchsreviers im Zeichen des Klimawandels. Landesbetrieb Wald und Holz NRW, Münster, pp 13–16
Bennett JN, Andrew B, Prescott CE (2002) Vertical fine root distributions of western redcedar, western hemlock, and salal in old-growth cedar–hemlock forests on northern Vancouver Island. Can J For Res 32:1208–1216
Bhuyan U, Zang C, Menzel A (2017) Different responses of multispecies tree ring growth to various drought indices across Europe. Dendrochronologia 44:1–8
Biondi F, Qeadan F (2008) A theory-driven approach to tree-ring standardization: defining the biological trend from expected basal area increment. Tree-Ring Res 64:81–96
Biondi F, Waikul K (2004) DENDROCLIM2002: a C++ program for statistical calibration of climate signals in tree-ring chronologies. Comput Geosci 30:303–311
Bolte A, Ammer C, Löf M, Madsen P, Nabuurs G-J, Schall P, Spathelf P, Rock J (2009) Adaptive forest management in central Europe. Climate change impacts, strategies and integrative concept. Scand J For Res 24:473–482
Bolte A, Ammer C, Löf M, Nabuurs G-J, Schall P, Spathelf P (2010) Adaptive forest management. A prerequisite for sustainable forestry in the face of climate change. In: Spathelf P (ed) Sustainable forest management in a changing world. A European perspective. Springer, Dordrecht
Bottacci A (2011) Castanea sativa Miller. In: Roloff A, Schütt P, Lang UM, Stimm B (eds) Enzyklopädie der Holzgewächse. Handbuch und Atlas der Dendrologie, Wiley-VCH, Weinheim
Bouriaud O, Bréda N, Dupouey J-L, Granier A (2005) Is ring width a reliable proxy for stem-biomass increment? A case study in European beech. Can J For Res 35:2920–2933
Bréda N, Huc R, Granier A, Dreyer E (2006) Temperate forest trees and stands under severe drought. A review of ecophysiological responses, adaptation processes and long-term consequences. Ann For Sci 63:625–644
Broadmeadow MSJ, Ray D, Samuel CJA (2005) Climate change and the future for broadleaved tree species in Britain. For Int J For Res 78:145–161
Brodribb TJ, McAdam SA, Jordan GJ, Martins SC (2014) Conifer species adapt to low-rainfall climates by following one of two divergent pathways. Proc Natl Acad Sci USA 111:14489–14493
Buras A (2017) A comment on the expressed population signal. Dendrochronologia 44:130–132
Buras A, Menzel A (2019) Projecting tree species composition changes of European Forests for 2061–2090 under RCP 4.5 and RCP 8.5 scenarios. Front Plant Sci 9:1986
Buras A, Rammig A, Zang CS (2020) Quantifying impacts of the drought 2018 on European ecosystems in comparison to 2003. Biogeosciences 17:1655–1672. https://doi.org/10.5194/bg-17-1655-2020
Burnham KP, Anderson DR (2010) Model selection and multimodel inference. A practical information-theoretic approach. Springer, New York
Burns RM, Honkala BH (1990a) Silvics of North America. Volume 1, Conifers. Agriculture Handbook 654. U.S. Dept. of Agriculture, Forest Service, Washington, D.C
Burns RM, Honkala BH (1990b) Silvics of North America. Volume 2, Hardwoods. Agriculture Handbook 654. U.S. Dept. of Agriculture, Forest Service, Washington, D.C
Burrichter E, Schoenwald W (1968) Forstliche Anbauversuche mit der Metasequoia glyptostroboides im Raum Westfalen. VS Verlag für Sozialwissenschaften, Wiesbaden
Carmean WH (1972) Site index curves for Upland Oaks in the Central States. Forest Science 18:109–120
Castagneri D, Nola P, Motta R, Carrer M (2014) Summer climate variability over the last 250 years differently affected tree species radial growth in a mesic Fagus–Abies–Picea old-growth forest. For Ecol Manag 320:21–29
Chevan A, Sutherland M (1991) Hierarchical partitioning. Am Stat 45:90–96
Chhin S, Wang GG (2005) The effect of sampling height on dendroclimatic analysis. Dendrochronologia 23:47–55
Chhin S, Hogg EH, Ted Lieffers VJ, Huang S (2010) Growth-climate relationships vary with height along the stem in lodgepole pine. Tree Physiol 30:335–345
Choat B, Jansen S, Brodribb TJ et al (2012) Global convergence in the vulnerability of forests to drought. Nature 491:752–755
Choat B, Brodribb TJ, Brodersen CR, Duursma RA, López R, Medlyn BE (2018) Triggers of tree mortality under drought. Nature 558:531–539
Conedera M, Barthold F, Torriani D, Pezzatti GB (2010) Drought sensitivity of Castanea sativa: case study of summer 2003 in the southern Alps. In: Proceedings of the 1st European congress on chestnut Castanea 2009, Cuneo-Torino, Italy, October 13–16, 2009. Acta Horticulturae, vol 866. pp 297–302
Cook ER (1985) A time series analysis approach to tree-ring standardization. Dissertation, University of Arizona, Tucson
Cook ER, Pederson N (2011) Uncertainty, emergence, and statistics in dendrochronology. In: Diaz HF, Hughes MK, Swetnam TW (eds) Dendroclimatology. Progress and prospects. Springer, Dordrecht, pp 77–112
Cook ER, Peters K (1981) The smoothing spline: a new approach to standardizing forest interior tree-ring width series for dendroclimatic studies. Tree-Ring Bull 41:45–53
Core Team R (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Corona P, Romagnoli M, Torrini L (1995) Stem annual increments as ecobiological indicators in Turkey oak (Quercus cerris L.). Trees 10:13–19
Čufar K, Prislan P, de Luis M, Gričar J (2008) Tree-ring variation, wood formation and phenology of beech (Fagus sylvatica) from a representative site in Slovenia, SE Central Europe. Trees 22:749–758
de Martonne E (1926) Une nouvelle fonction climatologique: L’indice d’aridité. La Météorologie 2:449–458
Dulamsuren C, Hauck M, Kopp G, Ruff M, Leuschner C (2017) European beech responds to climate change with growth decline at lower, and growth increase at higher elevations in the center of its distribution range (SW Germany). Trees 31:673–686
DWD (2015) Deutscher Wetterdienst. Wetter und Klima aus einer Hand. Available via http://www.dwd.de/; ftp://ftp-cdc.dwd.de/pub/CDC/observations_germany/climate/. Accessed 10 June 2015
Dyderski MK, Paź S, Frelich LE, Jagodziński AM (2018) How much does climate change threaten European forest tree species distributions? Glob Change Biol 24:1150–1163
Eckstein D, Bauch J (1969) Beitrag zur Rationalisierung eines dendrochronologischen Verfahrens und zur Analyse seiner Aussagesicherheit. Forstwissenschaftliches Centralblatt 88:230–250
Eilmann B, Rigling A (2012) Tree-growth analyses to estimate tree species’ drought tolerance. Tree Physiol 32:178–187
Ettinger AK, Ford KR, HilleRisLambers J (2011) Climate determines upper, but not lower, altitudinal range limits of Pacific Northwest conifers. Ecology 92:1323–1331
Fardusi MJ, Ferrio JP, Comas C, Voltas J, Resco de Dios V, Serrano L (2016) Intra-specific association between carbon isotope composition and productivity in woody plants: a meta-analysis. Plant Sci 251:110–118
Feliksik E, Wilczyński S (2009) The effect of climate on tree-ring chronologies of native and nonnative tree species growing under homogenous site conditions. Geochronometria 33:49–57
Friedrichs DA, Büntgen U, Frank DC, Esper J, Neuwirth B, Löffler J (2009) Complex climate controls on 20th century oak growth in Central-West Germany. Tree Physiol 29:39–51
Fritts HC (1976) Tree rings and climate. Academic Press, London
Gazol A, Camarero JJ, Colangelo M, de Luis M, Martínez del Castillo E, Serra-Maluquer X (2019) Summer drought and spring frost, but not their interaction, constrain European beech and Silver fir growth in their southern distribution limits. Agric For Meteorol 278:107695
Giraudoux P (2017) pgirmess: Data Analysis in Ecology. R package version 1.6.7. https://CRAN.R-project.org/package=pgirmess. Accessed 16 Aug 2018
Gomes-Laranjo J, Dinis L-T, Martins L et al (2012) Characterization of chestnut behavior with photosynthetic traits. In: Najafpour M (ed) Applied photosynthesis. InTech, Rijeka, pp 47–80
Gower ST, Isebrands JG, Sheriff DW (1995) Carbon allocation and accumulation in conifers. In: Smith WK, Hinckley TM, Roy J (eds) Resource physiology of conifers. Acquisition, allocation, and utilization. Elsevier Science, Burlington, pp 217–254
Granda E, Camarero JJ, Gimeno TE, Martínez-Fernández J, Valladares F (2013) Intensity and timing of warming and drought differentially affect growth patterns of co-occurring Mediterranean tree species. Eur J For Res 132:469–480
Grömping U (2006) Relative Importance for Linear Regression in R. The Package relaimpo. J Stat Softw 17:1–27
Grossnickle SC, Russell JH (2010) Ecophysiological processes of western redcedar (Thuja plicata) and yellow-cedar (Callitropsis nootkatensis). In: Harrington CA (ed) A tale of two cedars: international symposium on Western Redcedar and Yellow-Cedar. Gen. Tech. Rep. PNW-GTR-828, pp 33–36
Hacket-Pain AJ, Friend AD, Lageard JG, Thomas PA (2015) The influence of masting phenomenon on growth-climate relationships in trees: explaining the influence of previous summers’ climate on ring width. Tree Physiol 35:319–330
Hacket-Pain AJ, Ascoli D, Vacchiano G et al (2018) Climatically controlled reproduction drives interannual growth variability in a temperate tree species. Ecol Lett 21:1833–1844
Hart JL, Buchanan ML, Clark SL, Torreano SJ (2012) Canopy accession strategies and climate-growth relationships in Acer rubrum. For Ecol Manage 282:124–132
Hennon PE (2011) Tsuga heterophylla. In: Roloff A, Schütt P, Lang UM, Stimm B (eds) Enzyklopädie der Holzgewächse. Handbuch und Atlas der Dendrologie. Wiley-VCH, Weinheim, pp 1–8
Hinckley, TM, Lassoie, JP (eds) (1981) Radial growth in coniferous and deciduous trees: A comparison. Mitteil. der Forstl. Bundesversuchsanstalt Wien, 142/1: 17–56, Wien
Hoffmann N, Schall P, Ammer C, Leder B, Vor T (2018a) Drought sensitivity and stem growth variation of nine alien and native tree species on a productive forest site in Germany. Agric For Meteorol 256–257:431–444
Hoffmann N, Seidel D, Ammer C, Leder B, Vor T (2018b) Quantifizierung der Zuwachsreaktion fremdländischer Baumarten auf Konkurrenz und Freistellungsmaßnahmen. Allgemeine Forst- und Jagdzeitung 189(3/4):57–87. https://doi.org/10.23765/afjz0002021
IPCC (2013) Summary for Policymakers. In: Stocker TF, Qin D, Plattner GK, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Ishii H, Azuma W, Nabeshima E (2013) The need for a canopy perspective to understand the importance of phenotypic plasticity for promoting species coexistence and light-use complementarity in forest ecosystems. Ecol Res 28:191–198
Kerhoulas LP, Kane JM (2012) Sensitivity of ring growth and carbon allocation to climatic variation vary within ponderosa pine trees. Tree Physiol 32:14–23
Kölling C (2013) Nichtheimische Baumarten – Alternativen im klimagerechten Waldumbau? LWF aktuell 96:4–11
Kölling C, Knoke T, Schall P, Ammer C (2009) Überlegungen zum Risiko des Fichtenanbaus in Deutschland vor dem Hintergrund des Klimawandels. Forstarchiv 80:42–54
Konôpka B, Noguchi K, Sakata T, Takahashi M, Konôpková Z (2006) Fine root dynamics in a Japanese cedar (Cryptomeria japonica) plantation throughout the growing season. For Ecol Manag 225:278–286
Kraft G (1884) Beiträge zur Lehre: Von den Durchforstungen, Schlagstellungen und Lichtungshieben. Klindworth, Hannover
Kublin E, Breidenbach J, Kändler G (2013) A flexible stem taper and volume prediction method based on mixed-effects B-spline regression. Eur J For Res 132:983–997
Kumagai T’o, Aoki S, Otsuki K, Utsumi Y (2009) Impact of stem water storage on diurnal estimates of whole-tree transpiration and canopy conductance from sap flow measurements in Japanese cedar and Japanese cypress trees. Hydrol Process 23:2335–2344
Kurata S (1971) Illustrated important forest trees of Japan. Vols. 1 and 2. Chikyu Shuppan, Tokyo
Land A, Remmele S, Schönbein J, Küppers M, Zimmermann R (2017) Climate-growth analysis using long-term daily-resolved station records with focus on the effect of heavy precipitation events. Dendrochronologia 45:156–164
Larson PR (1963) Stem form development of forest trees. Forest science Monograph 5, Society of American Foresters
Latte N, Lebourgeois F, Claessens H (2016) Growth partitioning within beech trees (Fagus sylvatica L.) varies in response to summer heat waves and related droughts. Trees 30:189–201
Lebourgeois F, Bréda N, Ulrich E, Granier A (2005) Climate-tree-growth relationships of European beech (Fagus sylvatica L.) in the French Permanent Plot Network (RENECOFOR). Trees 19:385–401
Lebourgeois F, Rathgeber CBK, Ulrich E (2010) Sensitivity of French temperate coniferous forests to climate variability and extreme events (Abies alba, Picea abies and Pinus sylvestris). J Veg Sci 21:364–376
Lévesque M, Saurer M, Siegwolf R, Eilmann B, Brang P, Bugmann H, Rigling A (2013) Drought response of five conifer species under contrasting water availability suggests high vulnerability of Norway spruce and European larch. Glob Change Biol 19:3184–3199
Liu Q, Piao S, Janssens IA et al (2018) Extension of the growing season increases vegetation exposure to frost. Nat Commun 9:426
Martin-Benito D, Beeckman H, Cañellas I (2013) Influence of drought on tree rings and tracheid features of Pinus nigra and Pinus sylvestris in a mesic Mediterranean forest. Eur J For Res 132:33–45
Matsumoto Y, Shigenaga H, Miura S, Nagakura J, Taoda H (2006) Mapping of Japanese Cedar (Cryptomeria japonica) Forests Vulnerable to Global Warming in Japan. Glob Environ Res 10:181–188
McCulloh KA, Johnson DM, Meinzer FC, Woodruff DR (2014) The dynamic pipeline: hydraulic capacitance and xylem hydraulic safety in four tall conifer species. Plant Cell Environ 37:1171–1183
Minore D (1990) Thuja plicata Donn ex D. Don, Western Redcedar. In: Burns RM, Honkala BH (eds) Silvics of North America. Volume 1, Conifers. Agriculture Handbook 654, U.S. Dept. of Agriculture, Forest Service, Washington, D.C., pp 590–600
Neumann M, Mues V, Moreno A, Hasenauer H, Seidl R (2017) Climate variability drives recent tree mortality in Europe. Glob Change Biol 23:4788–4797
Niinemets Ü, Valladares F (2006) Tolerance to shade, drought, and waterlogging of temperate Northern Hemisphere Trees and Shrubs. Ecol Monogr 76:521–547
Praciak A, Pasiecznik N, Sheil D et al (2013) The CABI encyclopedia of forest trees. CABI, Wallingford
Pretzsch H, Schütze G, Uhl E (2013) Resistance of European tree species to drought stress in mixed versus pure forests: evidence of stress release by inter-specific facilitation. Plant Biol (Stuttg) 15:483–495
Pretzsch H, Schütze G, Biber P (2018) Drought can favour the growth of small in relation to tall trees in mature stands of Norway spruce and European beech. For Ecosyst 5:20
Ranney TG, Bir RE, Skroch WA (1991) Comparative drought resistance among six species of birch (Betula): influence of mild water stress on water relations and leaf gas exchange. Tree Physiol 8:351–360
Repo T, Launiainen S, Lehto T et al (2016) The responses of Scots pine seedlings to waterlogging during the growing season. Can J For Res 46:1439–1450
Restaino CM, Peterson DL, Littell J (2016) Increased water deficit decreases Douglas fir growth throughout western US forests. Proc Natl Acad Sci USA 113:9557–9562
Rinn F (2005) TSAP-Win™—time series analysis and presentation for Dendrochronology and related applications. User Reference, Heidelberg
Rinn F (2006) Lignovision™—Scanning of wood and tree-ring recognition. User Reference, Heidelberg
Roloff A, Schütt P, Lang UM, Stimm B (2011) Enzyklopädie der Holzgewächse. Handbuch und Atlas der Dendrologie. Wiley-VCH, Weinheim
Ryan MG, Yoder BJ (1997) Hydraulic limits to tree height and tree growth. Bioscience 47:235–242
Ryan MG, Phillips N, Bond BJ (2006) The hydraulic limitation hypothesis revisited. Plant Cell Environ 29:367–381
Schaberg PG, Hennon PE, D’Amore DV, Halman JM (2010) Comparisons of the cold tolerance and rooting depth of yellow-cedar, western redcedar, western hemlock, mountain hemlock and sitka spruce growing together in Ketchikan, Alaska. In: Harrington CA (ed) A tale of two cedars: international symposium on Western Redcedar and Yellow-Cedar. Gen. Tech. Rep. PNW-GTR-828, p 76
Scharnweber T, Manthey M, Criegee C, Bauwe A, Schröder C, Wilmking M (2011) Drought matters—Declining precipitation influences growth of Fagus sylvatica L. and Quercus robur L. in north-eastern Germany. For Ecol Manag 262:947–961
Schenck CA (1939a) Fremdländische Wald- und Parkbäume. Ein Buch für alle Forstwirte und Dendrologen. Parey, Berlin
Schenck CA (1939b) Fremdländische Wald- und Parkbäume. Bd. 2: Die Nadelhölzer. Ein Buch für alle Forstwirte und Dendrologen. Parey, Berlin
Schober R (1978) Erste Ergebnisse von Anbauversuchen mit Tannen und anderen Koniferen aus Japan im Vergleich mit nordamerikanischen und europäischen Nadelhölzern. Teil II: die Versuche und ihre Ergebnisse. Allgemeine Forst- und Jagdzeitung 149:197–223
Schweingruber FH (1983) Der Jahrring. Standort, Methodik, Zeit und Klima in der Dendrochronologie. P. Haupt, Bern, p 234
Sevanto S, Vesala T, Perämäki M, Nikinmaa E (2003) Sugar transport together with environmental conditions controls time lags between xylem and stem diameter changes. Plant Cell Environ 26:1257–1265
Shen Z (2011) Metasequoia glyptostroboides. In: Roloff A, Schütt P, Lang UM, Stimm B (eds) Enzyklopädie der Holzgewächse. Handbuch und Atlas der Dendrologie. Wiley-VCH, Weinheim, pp 1–10
Sohn JA, Kohler M, Gessler A, Bauhus J (2012) Interactions of thinning and stem height on the drought response of radial stem growth and isotopic composition of Norway spruce (Picea abies). Tree Physiol 32:1199–1213
Sohn JA, Saha S, Bauhus J (2016) Potential of forest thinning to mitigate drought stress: a meta-analysis. For Ecol Manag 380:261–273
Stephan BR, Liesebach M (2011) Betula maximowicziana. In: Roloff A, Schütt P, Lang UM, Stimm B (eds) Enzyklopädie der Holzgewächse. Handbuch und Atlas der Dendrologie. Wiley-VCH, Weinheim, pp 1–12
Stojanović DB, Levanič T, Matović B, Stjepanović S, Orlović S (2018) Growth response of different tree species (oaks, beech and pine) from SE Europe to precipitation over time. Dendrobiology 79:97–110
Tange T, Yanaga K, Osawa H, Masumori M (2009) Effects of evening and nighttime leaf wetting on stomatal behavior of Cryptomeria japonica growing in dry soil. Photosynthetica 47:313–316
Teets A, Fraver S, Weiskittel AR, Hollinger DY (2018) Quantifying climate-growth relationships at the stand level in a mature mixed-species conifer forest. Glob Change Biol 24:3587–3602
Thomsen J, Bohrer G, Matheny A, Ivanov V, He L, Renninger H, Schäfer K (2013) Contrasting hydraulic strategies during dry soil conditions in Quercus rubra and Acer rubrum in a sandy site in Michigan. Forests 4:1106–1120
Thurm EA, Hernandez L, Baltensweiler A et al (2018) Alternative tree species under climate warming in managed European forests. For Ecol Manag 430:485–497
van der Maaten E (2012) Climate sensitivity of radial growth in European beech (Fagus sylvatica L.) at different aspects in southwestern Germany. Trees 26:777–788
van der Maaten-Theunissen M, Bouriaud O (2012) Climate–growth relationships at different stem heights in silver fir and Norway spruce. Can J For Res 42(5):958–969
Vann DR (2005) Physiological ecology of Metasequoia glyptostroboides Hu et Cheng. In: LePage BA, Williams CJ, Yang H (eds) The geobiology and ecology of Metasequoia. Springer, Dordrecht, pp 305–330
Vanoni M, Bugmann H, Nötzli M, Bigler C (2016) Drought and frost contribute to abrupt growth decreases before tree mortality in nine temperate tree species. For Ecol Manag 382(15):51–63
Vicente-Serrano SM, Beguería S, López-Moreno JI (2010) A multiscalar drought index sensitive to global warming. The standardized precipitation evapotranspiration index. J Clim 23:1696–1718
Vitali V, Büntgen U, Bauhus J (2017) Silver fir and Douglas fir are more tolerant to extreme droughts than Norway spruce in south-western Germany. Glob Change Biol 23:5108–5119
Vitasse Y, Bottero A, Cailleret M, Bigler C, Fonti P, Gessler A, Lévesque M, Rohner B, Weber P, Rigling A, Wohlgemuth T (2019) Contrasting resistance and resilience to extreme drought and late spring frost in five major European tree species. Glob Change Biol 25(11):3781–3792
Wagenführ R (1999) Anatomie des Holzes. Strukturanalytik, Identifizierung, Nomenklatur, Mikrotechnologie. DRW-Verlag, Leinfelden-Echterdingen, p 188
Walentowski H, Falk W, Mette T, Kunz J, Bräuning A, Meinardus C, Zang C, Sutcliffe LME, Leuschner C (2017) Assessing future suitability of tree species under climate change by multiple methods: a case study in southern Germany. Ann. For. Res. 60(1):101–126
Walters RS, Yawney HW (1990) Acer rubrum L., Red Maple. In: Burns RM, Honkala BH (eds) Silvics of North America. Volume 2, Hardwoods. Agriculture Handbook 654, U.S. Dept. of Agriculture, Forest Service, Washington, D.C., pp 60–69
Wang A-F, Roitto M, Sutinen S, Lehto T, Heinonen J, Zhang G, Repo T (2016) Waterlogging in late dormancy and the early growth phase affected root and leaf morphology in Betula pendula and Betula pubescens seedlings. Tree Physiol 36:86–98
Williams CJ (2005) Ecological characteristics of Metasequoia glyptostroboides. In: LePage BA, Williams CJ, Yang H (eds) The geobiology and ecology of Metasequoia. Springer, Dordrecht, pp 285–302
Williams AP, Allen CD, Macalady AK et al (2012) Temperature as a potent driver of regional forest drought stress and tree mortality. Nat Clim Change 3:292–297
WMO, GWP (2016) Handbook of Drought Indicators and Indices (M. Svoboda and B.A. Fuchs), 45. http://www.droughtmanagement.info/literature/GWP_Handbook_of_Drought_Indicators_and_Indices_2016.pdf. Accessed 03 Feb 2018
Zang C, Biondi F (2015) treeclim. An R package for the numerical calibration of proxy-climate relationships. Ecography 38:431–436
Zang C, Pretzsch H, Rothe A (2012) Size-dependent responses to summer drought in Scots pine, Norway spruce and common oak. Trees 26:557–569
Zang C, Hartl-Meier C, Dittmar C, Rothe A, Menzel A (2014) Patterns of drought tolerance in major European temperate forest trees: climatic drivers and levels of variability. Glob Change Biol 20:3767–3779
Zang C, Buras A, Esquivel-Muelbert A, Jump AS, Rigling A, Rammig A (2020) Standardized drought indices in ecological research: why one size does not fit all. Glob Change Biol 26(2):322–324
Zuur AF, Ieno EN, Smith GM (2007) Analysing ecological data. Springer, New York
Żywiec M, Muter E, Zielonka T, Delibes M, Calvo G, Fedriani JM (2017) Long-term effect of temperature and precipitation on radial growth in a threatened thermo-Mediterranean tree population. Trees 31:491–501
Acknowledgements
This study is part of the project “Documentation and evaluation of alien tree species in North Rhine-Westphalia under climate change” and was funded by the Ministry for Climate Protection, Environment, Agriculture, Conservation and Consumer Protection of the State of North Rhine-Westphalia. The authors are grateful to Christian Ammer and Bertram Leder for supervision the project part. We would like to thank Norbert Tennhoff, Wolfgang Müller and Herbert Dautzenberg for supplemental site data and active participation at site logistics in Burgholz. We are further grateful Ulrike Westphal, Michael Unger, Dirk Böttger and Ray Wollenzien for their helpful technical assistance in the forest stands, workshop and laboratory.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Miren del Rio.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised due to a retrospective Open Access order.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hoffmann, N., Heinrichs, S., Schall, P. et al. Climatic factors controlling stem growth of alien tree species at a mesic forest site: a multispecies approach. Eur J Forest Res 139, 915–934 (2020). https://doi.org/10.1007/s10342-020-01295-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-020-01295-3