Abstract
It is possible to appropriately diagnose non-mass abnormalities by elucidating ultrasound non-mass abnormality findings and sharing the concept. If non-mass abnormalities can be diagnosed early, the number of curable cases could increase, leading to fewer breast cancer deaths. The Japan Society of Ultrasonics in Medicine (JSUM) Terminology/Diagnostic Criteria Committee has classified non-mass abnormalities into five subtypes: hypoechoic area in the mammary gland, abnormalities of the ducts, architectural distortion, multiple small cysts, and echogenic foci without a hypoechoic area. We herein define the findings for each of these subtypes and present a summary of the JSUM guidelines on non-mass abnormalities of the breast generated based on those findings.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
With respect to non-mass image-forming breast cancer on breast ultrasound (US), lesions that are difficult to recognize began to be identified as masses presenting a variety of morphologies, including the visualization of breast cancer with abnormal nipple discharge as mammary duct dilatation on US images in the 1980s [1, 2]. In addition, with the widespread adoption of mammography screening, many cases of breast cancer showing microcalcifications are being detected [3,4,5,6]; the morphology has been identified by collating lesions on mammography images with US images, and it is now possible to detect the US image characteristics of non-mass image-forming breast cancer [7, 8]. The subsequent widespread adoption of US breast screening as well as advances in diagnostic equipment have resulted in an increasing awareness of non-mass abnormalities [9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24].
The Breast Imaging Reporting and Data System (BI-RADS) 5th edition US lexicon includes masses, calcifications, and associated features, but it does not include the concept of non-mass abnormalities [25, 26].
Non-mass abnormalities present a variety of morphologies and findings on US images, but in the case of malignant lesions, not only ductal carcinoma in situ (DCIS) but also invasive lobular carcinoma (ILC), invasive ductal carcinoma (IDC), and other carcinomas present non-mass abnormality findings [21, 23, 26,27,28,29,30,31,32,33,34,35,36]. Appropriate diagnosis of invasive cancers presenting as non-mass abnormalities may help reduce breast cancer deaths; therefore, it is important to diagnose them by assessing US imaging findings and their distribution.
The Japan Society of Ultrasonics in Medicine (JSUM) Terminology/Diagnostic Criteria Committee has classified non-mass abnormalities into five subtypes: hypoechoic area in the mammary gland, abnormalities of the ducts, architectural distortion, multiple small cysts, and echogenic foci without a hypoechoic area. We herein define the findings for each of these subtypes and present a summary of the JSUM guidelines on non-mass abnormalities of the breast generated based on the findings that correspond to each subtype.
Definition of non-mass abnormalities
Non-mass abnormalities refer to lesions that are difficult to discern as masses on US images. Note that for a certain period of time when the concept of the term was first proposed, "non-mass image-forming lesion" was used, but the terminology has since been reexamined and is now unified as "non-mass abnormalities".
Note that a breast "mass" refers to a space-occupying lesion believed to be a lump formed by components that differ from the surrounding tissue, and corresponds to "mass image-forming lesion" in Guidelines for Ultrasound Diagnosis of Breast Disorders (2005) [9,10,11].
Classification of non-mass abnormalities
Hypoechoic area in the mammary gland
Definition: A hypoechoic area with properties that differ from those of the surrounding mammary gland or contralateral mammary gland, and which is difficult to discern as a mass.
The term "hypoechoic" used for non-mass abnormalities refers to an echo level that can be recognized as being lower than that of the surrounding mammary gland.
Note that the term "hypoechoic" used to express the echo level in a breast mass is generally based on the echo level of fat; therefore, it differs from the standard used for non-mass abnormalities.
In actual cases, it is sometimes difficult to assess whether a finding is a hypoechoic area or a mass, but there is no particular problem with the examiner or diagnostician subjectively making either assessment during the diagnosis process. Either way, the diagnosis should fundamentally be made with intraductal proliferative lesions in mind, both benign and malignant.
The differential diagnosis will primarily include, for example, DCIS and IDC or ILC with extensive intraductal component (EIC) in the case of malignant lesions, and ductal hyperplasia, adenosis, and sclerosing lesions in the case of benign lesions.
A hypoechoic area in the mammary gland is further classified into the following three subtypes, but not all can be distinctly classified.
-
①
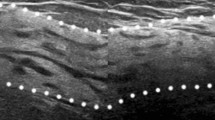
Patchy or mottled hypoechoic area: There are multiple relatively small hypoechoic areas that as a whole can be recognized as one lesion (Fig. 1).
-
②
Geographic hypoechoic area: It looks as if a patchy or mottled hypoechoic area has fused together (Fig. 2).
-
③
Indistinct or ill-defined hypoechoic area: It is difficult to express as either patchy/mottled or geographic, and it cannot be recognized as a mass due to having an indistinct or ill-defined border (Fig. 3a).
A segmental or focal hypoechoic area in the mammary gland indicates malignant potential. If echogenic foci suggestive of calcification are found in a hypoechoic area, the malignant potential is higher (Fig. 3b). If blood flow is clearly increased in a hypoechoic area with thickening of the mammary gland, the presence of intraductal proliferative changes should be kept in mind when making the diagnosis.
Abnormalities of the ducts
Definition: The properties of the ducts such as caliber, wall thickness, or regularity are different from those of normal ducts.
Abnormalities of the ducts are classified into the following three types.
-
①
Duct dilatation: A state in which ducts are clearly dilated beyond the extent of the areola as compared with other ducts.
(Explanation) Ducts within the extent of the areola are not considered an abnormality based on a finding of dilatation alone as dilatation is also found in normal cases. Moreover, it is not considered an abnormality based on a finding of duct dilatation alone during late gestation or lactation (Fig. 4). Note that the bilateral or diffuse linear high echoes sometimes seen in the mammary glands of young individuals are believed to be ducts and are not considered an abnormality.
-
②
Ducts with internal echoes: The following are examples of US findings seen in the ducts.
-
Solid echoes: A finding that suggests a solid lesion such as a hypoechoic component seen in the ducts.
-
Echogenic foci: Hyperechoic spots in the ducts.
-
Floating echoes: A finding in which breast milk, abscess, blood, or other liquid is seen in the ducts and its fluidity is observed.
Note: Attention is required when there are internal echoes in the case of focal or segmental dilated ducts. If the starting point of the solid component in the dilated ducts is steep, there is a high likelihood of it being a benign lesion such as an intraductal papilloma (Fig. 5), and when the beginning is gentle or the solid component in the ducts is continuous, there is a high likelihood of it being a malignant lesion, such as DCIS (Fig. 6).
-
③
Irregularity of ductal caliber (Fig. 7)
Note: It is somewhat difficult to evaluate irregularity of ductal caliber based on static images alone, but it is considered to be a finding that leans towards malignancy.
Architectural distortion
Definition: Findings of mammary gland structure-concentrated tightening/distortion at one spot or in a localized area in the mammary gland.
Architectural distortion, which is thought to be due to convergent changes in tissue, is found not only in malignant lesions but also in benign lesions.
Multiple small cysts
Definition: A finding in which multiple lesions recognized as small cysts several millimeters in size are observed in the mammary gland.
They are small hypoechoic masses that cannot be confirmed to be cysts (internal anechoic area), and they include masses that cannot be confirmed to be solid masses such as those without internal blood flow. In cases where clustering of microcysts is observed, the term "clustered microcysts" can be used.
A detailed examination is not needed when screening reveals multiple small cysts alone. This is because the frequency of malignancy is extremely low when the only finding is multiple small cysts (clustered microcysts), because they are often indolent even if malignant.
In rare cases, they indicate the presence of DCIS (Fig. 12), but most cases are benign lesions, such as apocrine metaplasia (Fig. 13).
Echogenic foci without a hypoechoic area (Fig. 14)
Definition: A lesion in which multiple hyperechoic foci thought to be microcalcifications are present focally or segmentally in the mammary gland, without involvement of a clear hypoechoic area or abnormalities of the ducts in the surrounding area.
This finding is not an independent US finding. As a rule, it is predicated on observation of calcifications that requires differentiation of benignancy/malignancy using mammography. It is used in cases, where the site of hyperechoic foci on US does not contradict the site of calcifications on mammography.
The presence of this finding may indicate DCIS, but they are helpful in determining the target for needle biopsy and other procedures.
6) The following are ancillary findings.
-
a.
Echogenic foci: Echogenic foci are defined as multiple point-like hyperechogenicity within the hypoechoic area or within the solid part of the abnormalities of the ducts. The presence of echogenic foci, suggesting multiple calcifications within the lesion, is an important finding, because it increases the likelihood of malignancy.
-
b.
Vascularity: When color Doppler clearly detects a large number of blood flow signals in or near the lesion, it is a helpful finding to suspect malignancy.
Comparison with the contralateral mammary gland serves as a reference in the assessment.
-
c.
Elasticity: Malignant lesions often have increased stiffness over the surrounding tissue, which is helpful in distinguishing between benign from malignant.
Lesion distribution
As for lesion distribution, assess and record the lesion site in the following order.
-
①
Bilateral, unilateral
-
②
Focal (clustered), segmental, diffuse
It is easy to grasp segmental distribution when making the assessment by envisioning an image that extends radially with the nipple as the peak. The term clustered is sometimes used for multiple small cysts.
A bilateral or diffuse (scattered) distribution usually indicates a benign lesion. Attention is required in the case of a segmental or focal (clustered) distribution as it may indicate malignant potential. In the case of a distinct segmental distribution, in particular, the possibility of DCIS should be considered. However, breast cancer that presents a finding of multiple small cysts alone is very rare even if the distribution is segmental or focal (clustered), and it is not thought to affect the vital prognosis; therefore, thorough examination is not necessary when screening does not reveal other findings such as hypoechoic area in the mammary gland and abnormalities of the ducts [9,10,11].
Conclusion
Lesions that present non-mass abnormality findings on US include a variety of lesions, from benign to highly malignant breast cancers. The JSUM Terminology/Diagnostic Criteria Committee has classified US findings of non-mass abnormalities into five subtypes. Diagnostic accuracy may be improved by keeping in mind the classification and distribution of non-mass abnormalities when diagnosing lesions presenting as non-mass abnormalities.
Data availability statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
Ueno E, Tohno E, Ito K. Classification and diagnosis criteria in breast echography. J Med Ultrason. 1986;13:19–31.
Ueno E, Tohno E, Soeda S, et al. Dynamic tests in real-time breast echography. Ultrasound Med Biol. 1988;14:53–7.
Ciatto S, Cataliotti L, Distante V. Nonpalpable lesions detected with mammography: review of 612 consecutive cases. Radiology. 1987;165:99–102.
Hall FM, Storella JM, Silverston DZ, et al. Nonpalpable breast lesions: recommendations for biopsy based on suspicion of carcinoma at mammography. Radiology. 1988;167:353–8.
White E, Lee CY, Kristal AR. Evaluation of the increase in breast cancer incidence in relation to mammography use. J Natl Cancer Inst. 1990;82:1546–52.
Leung JWT, Sickles EA. Developing asymmetry identified on mammography: correlation with imaging outcome and pathologic findings. AJR Am J Roentgenol. 2007;188:667–75.
Shetty MK, Watson AB. Sonographic evaluation of focal asymmetric density of the breast. Ultrasound Q. 2002;18:115–21.
Giess CS, Chesebro AL, Chikarmane SA. Ultrasound features of mammographic developing asymmetries and correlation with histopathologic findings. AJR Am J Roentgenol. 2018;210:W29–38.
Japan association of breast and thyroid sonology. Guideline for breast ultrasound management and diagnosis. Tokyo: Nankodo.; 2004.
Morishima I, Ueno E, Tohno E, et al. Ultrasonic Diagnosis of Non-Mass Image-Forming Breast Cancer. In: Ueno E, Shiina T, Kubota M, et al., editors. Research and Development in Breast Ultrasound. Tokyo: Springer; 2005. p. 127–34.
Endo T, Kubota M, Konishi Y, et al. Draft diagnosis guidelines for non-mass image forming lesions by the Japan association of breast and thyroid sonology(JABTS) and Japan society of ultrasonics in medicine. Research and Development in Breast Ultrasound. Tokyo: Springer; 2005. p.89–100.
Tohno E, Ueno E. Ultrasound(US) diagnosis of nonpalpable breast cancer. Breast Cancer. 2005;12:267–71.
Sotome K, Yamamoto Y, Hirano A, et al. The role of contrast enhanced MRI in the diagnosis of non-mass-image-forming lesions on breast ultrasonography. Breast Cancer. 2007;14:371–80.
Takei J, Tsunoda-Shimizu H, Kikuchi M, et al. Clinical implications of architectural distortion visualized by breast ultrasonography. Breast Cancer. 2009;16:132–5.
Berg WA, Campassi CI, Ioffe OB. Cystic lesions of the breast: sonographic-pathologic correlation. Radiology. 2003;227:183–91.
Usami Y, Tsunoda H, Kajiura Y, et al. Ultrasonographic evaluation of multiple cystic lesions in breast sonography. Jpn J Med Ultrasonics. 2011;38:455–60.
Uematsu T. Non-mass like lesions on breast ultrasonography: a systematic review. Breast Cancer. 2012;19:295–301.
Kim SJ, Park YM, Jung HK. Nonmasslike lesions on breast sonography: comparison between benign and malignant lesions. J Ultrasound Med. 2014;33:421–30.
Ko KH, Hsu HH, Yu JC, et al. Non-mass-like breast lesions at ultrasonography: feature analysis and BI-RADS assessment. Eur J Radiol. 2015;84:77–85.
Wang ZL, Li N, Li M, Wan WB. Non-mass-like lesions on breast ultrasound: classification and correlation with histology. Radiol Med (Torino). 2015;120:905–10.
Lee J, Lee JH, Baik S, et al. Non-mass lesions on screening breast ultrasound. Med Ultrason. 2016; 18: 446–451
Watanabe T, Yamaguchi T, Tsunoda H, et al. Ultrasound image classification of ductal carcinoma in situ (DCIS) of the breast: analysis of 705 DCIS lesions. Ultrasound Med Biol. 2017;43:918–25.
Park JW, Ko KH, Kim EK, et al. Non-mass breast lesions on ultrasound: final outcomes and predictors of malignancy. Acta Radiol. 2017; 58: 1054–60.
Ban K, Tsunoda H, Watanabe T, et al. Characteristics of ultrasonographic images of ductal carcinoma in situ with abnormalities of the ducts. J Med Ultrason. 2020;47:107–15.
Mendelson EB, Bohm-Velez M, Berg WA, et al. BI-RADS: ultrasound. In: D’Orsi CJ, Sickles EA, Mendelson EB, et al, eds. Breast Imaging Reporting and Data System: ACR BI-RADS Atlas. Reston, Va: American College of Radiology. 2013.
Raza S, Goldkamp AL, Chikarmane SA, et al. US of breast masses categorized as BI-RADS 3, 4, and 5: pictorial review of factors influencing clinical management. Radiographics. 2010;30:1199–213.
Moon WK, Myung JS, Lee YJ, et al. US of ductal carcinoma in situ. RadioGraphics. 2002;22:269–80; discussion 280–81.
Shin HJ, Kim HH, Kim SM, et al. Screening-detected and symptomatic ductal carcinoma in situ: differences in the sonographic and pathologic features. AJR Am J Roentgenol. 2008;190:516–25.
Park JS, Park YM, Kim EK, et al. Sonographic findings of high-grade and non-high-grade ductal carcinoma in situ of the breast. J Ultrasound Med. 2010;29:1687–97.
Izumori A, Takebe K, Sato A. Ultrasound fndings and histological features of ductal carcinoma in situ detected by ultrasound examination alone. Breast Cancer. 2009;17:136–41.
Wang LC, Sullivan M, Du H, et al. US appearance of ductal carcinoma in situ. Radiographics. 2013;33:213–28.
Li JK, Wang HF, He Y, et al. Ultrasonographic features of ductal carcinoma in situ: analysis of 219 lesions. Gland Surg. 2020;9:1945–54.
Gunawardena DS, Burrows S, Taylor DB. Non-mass versus mass-like ultrasound patterns in ductal carcinoma in situ: is there an association with high-risk histology? Clin Radiol. 2020;75:140–7.
Moschetta M, Sardaro A, Nitti A, et al. Ultrasound evaluation of ductal carcinoma in situ of the breast. J Ultrasound. 2022;25:41–5.
Porter AJ, Evans EB, Foxcroft LM, et al. Mammographic and ultrasound features of invasive lobular carcinoma of the breast. J Med Imaging Radiat Oncol. 2014;58:1–10.
Selinko VL, Middleton LP, Dempsey PJ. Role of sonography in diagnosing and staging invasive lobular carcinoma. J Clin Ultrasound. 2004;32:323–32.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical statements
All procedures followed the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Ito, T., Ueno, E., Endo, T. et al. The Japan Society of Ultrasonics in Medicine guidelines on non-mass abnormalities of the breast. J Med Ultrasonics 50, 331–339 (2023). https://doi.org/10.1007/s10396-023-01308-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10396-023-01308-9