Abstract
This paper reports the results of an international interlaboratory study led by the National Institute of Standards and Technology (NIST) on the measurement of high-pressure surface excess methane adsorption isotherms on NIST Reference Material RM 8850 (Zeolite Y), at 25 °C up to 7.5 MPa. Twenty laboratories participated in the study and contributed over one-hundred adsorption isotherms of methane on Zeolite Y. From these data, an empirical reference equation was determined, along with a 95% uncertainty interval (Uk=2). By requiring participants to replicate a high-pressure reference isotherm for carbon dioxide adsorption on NIST Reference Material RM 8852 (ZSM-5), this interlaboratory study also demonstrated the usefulness of reference isotherms in evaluating the performance of high-pressure adsorption experiments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Adsorbents have many potential applications, ranging from fluid catalytic cracking to gas separation and storage, to environmental remediation (Yang 2003). The performance of an adsorbent is especially determined by measuring an adsorption isotherm, a plot of gas uptake as a function of equilibrium pressure at a fixed temperature. Many of the aforementioned applications require measurements up to high-pressures; (Chareonsuppanimit et al. 2012; Darkrim et al. 2002; Liu et al. 2012; Mason et al. 2014; Menon 1968; Menon and Komarneni 1998; White et al. 2005) however, reproducibility of high-pressure measurements has proved to be a challenge, which has slowed the development of new materials and their applications (Broom and Hirscher 2016; Broom and Webb 2017; Espinal et al. 2013; Gasparik et al. 2014; Gensterblum et al. 2009, 2010; Goodman et al. 2004, 2007; Han et al. 2019; Hurst et al. 2016, 2019; Moretto et al. 2013; Park et al. 2017; Zlotea et al. 2009). In part, these experimental difficulties are linked to the lack of standardized measurement protocols, reference materials, and reference data. To address this situation, the National Institute of Standards and Technology (NIST), in partnership with the Department of Energy’s Advanced Research Projects Agency (ARPA-E), initiated a program to develop reference materials, reference data, and measurement protocols to improve adsorption metrology. Recently, the NIST Facility for Adsorbent Characterization and Testing (FACT Lab) led an interlaboratory study (hereafter, ILS1) on the measurement of high-pressure surface excess carbon dioxide (CO2) adsorption isotherms on NIST Reference Material RM 8852, ammonium ZSM-5, at 20 °C up to 4.5 MPa (Nguyen et al. 2018). The exercise involved eleven invited participants with recognized expertise in the adsorption sciences. The output was, for the first time, high-pressure adsorption reference data using a reference material. The CO2/ZSM-5 reference isotherm recently proved helpful for advancing modeling methods (Fang et al. 2020). Having companion high-pressure isotherm experiments showing high-quality results for a reference material would also be valuable to demonstrate reliability in high-pressure experiments on new adsorbents, particularly given the large number of porous materials that have not been replicated since they were first reported (Agrawal et al. 2020). The present study builds on the CO2/ZSM-5 reference isotherm work.
Working through Technical Working Group 39 of the Versailles Project on Advanced Materials and Standards (VAMAS),Footnote 1 the FACT Lab organized a second high-pressure interlaboratory study (hereafter, ILS2) to investigate the usefulness of the CO2/ZSM-5 reference isotherm in evaluating the performance of high-pressure adsorption instruments and to determine a reference isotherm for methane (CH4) adsorption on NIST Reference Material RM 8850, sodium Zeolite Y (ZY).Footnote 2 This study is different from ILS1 in two important ways. First, the study was open to any participants. However, those who had not participated in the ILS1 were required to replicate the reference isotherm derived from that study. Second, while a participant with a dataset statistically different from those of other participants was given an opportunity to remeasure or reprocess the submitted isotherms, the FACT Lab did not actively interact with the participant to identify the reasons for the difference.
To provide variety in materials, ZY, one of the three zeolitic reference materials at NIST was chosen for ILS2. ZY is more hygroscopic than ZSM-5 but less hygroscopic than Zeolite A (RM 8851). ZY (RM 8850) is completely microporous (see Fig. S1), whereas ZSM-5 (RM 8852) has a small amount of mesoporosity (Fang et al. 2020). ZY being a different material from ZSM-5 would also allow the true extent of the usefulness of the CO2/ZSM-5 reference isotherm to be determined when applied to measurements of other materials, as would be expected in real-world applications.
ZY, a faujasite, was introduced as an acidic zeolitic catalyst for the cracking of hydrocarbons in the 1960s (Cejka et al. 2017). The microporous zeolite has a three-dimensional pore structure, with pores running in mutually orthogonal directions. The pore diameter is ≈ 0.8 nm, which is defined by a twelve-member oxygen ring and leads into a cavity of diameter ≈1.2 nm (Baerlocher et al. 2007). The cavity is surrounded by ten sodalite cages connected on their hexagonal faces, in a tetrahedral, three-dimensional structure in which every sodalite cage has four uniformly distributed nearest neighbors as binding partners (Baerlocher et al. 2007; Cejka et al. 2017; Julbe and Drobek 2016). Methane, the major component of natural gas, meanwhile, was chosen as the adsorbate because of interest in using adsorbents for methane storage and transportation at safe pressure levels (Beckner and Dailly 2016; Keskin Avci and Erucar 2018; Menon and Komarneni 1998). The CH4/ZY pair was selected because of its commercial relevance (Cejka et al. 2017) and because it provides a reference isotherm for a supercritical fluid.
2 Experimental and data analysis methods
2.1 Methods
As mentioned, ILS2 involved measurement of CH4/ZY surface excess adsorption isotherms at 25 °C for pressures up to 7.5 MPa, using NIST RM 8850 as the adsorbent. This reference material is highly homogenized and characterized with reference values for elemental composition (e.g., Si/Al = 2.547 ± 0.037, Na/Al = 0.997 ± 0.018) and certain other physicochemical properties (e.g., loss-on-fusion ≈ loss-on-ignition ≈ 0.25), and information values for a range of structural properties (Turner et al. 2008). Sufficient units of this material are in stock at NIST (https://www.nist.gov/srm) to ensure availability for the foreseeable future.
In total, 20 laboratories participated in ILS2; seven of the 20 were participants in ILS1. The measurement capabilities of these laboratories included both commercial and custom-built manometric and gravimetric instruments.
The measurement protocol instructions were minimal. For CO2/ZSM-5 measurements, the protocol and conditions developed in ILS1 were prescribed (Nguyen et al. 2018). For the CH4/ZY, the measurement protocol specified a minimum purity of the adsorptive (≥ 99.999%), the sample pretreatment (activation at 350 °C for at least 12 h using a turbomolecular pump), the pressure range (7.5 MPa or the maximum capability of the instrument), the recommended equilibrium pressure points, the temperature (25 °C), and the number of isotherms to be measured (two isotherms each for two separate aliquots, totaling four isotherms for CH4). It was recommended to perform a blank run (i.e., an isotherm in the absence of the adsorbent) to subtract from the isotherm measured with the adsorbent present (Nguyen et al. 2017). Each participant was provided with one unit (40 g) each of RM 8852 and RM 8850. Participants were asked to submit an experimental report, which detailed their experimental procedures and data processing steps, and to submit the isotherms as surface excess uptake in units of millimoles of adsorbed fluid per gram of activated zeolite in a provided template. For the most part, the participants followed the prescribed protocols, although there were some small deviations. Details on various experimental parameters and procedures for each dataset can be found in Table 1.
2.2 Data evaluation
Each participant in ILS2 submitted at least one high-pressure dataset. For clarity, a CH4/ZY dataset is composed of four adsorption isotherms from two aliquots of ZY (aliquot 1—isotherm 1, aliquot 1—isotherm 2, aliquot 2—isotherm 1, aliquot 2—isotherm 2). In general, the intralaboratory isotherms were highly reproducible (except for DS 14 and 15; see Figs. S6–S10 in the Supplemental Information). Twenty-six high-pressure CH4/ZY datasets were submitted, for a total of 104 isotherms. In addition, two participants submitted 5 isotherms over the pressure range of 1 kPa to 100 kPa. These low-pressure data were useful in constraining the empirical reference function at low-pressure.
Labs who had not participated in ILS1 were asked to provide two CO2/ZSM-5 isotherms (20 °C), one isotherm each from two separate sample aliquots. In general, these intralaboratory isotherms were also highly reproducible (except for DS 14, see Figs. S3–S5 in the Supplemental Information). The purpose of this exercise was to demonstrate the measurement capabilities of these laboratories. Successful replication of the CO2/ZSM-5 isotherm was a criterion for inclusion of CH4/ZY isotherms from these labs in determination of the reference isotherm for ILS2. The CO2/ZSM-5 isotherms were evaluated with two metrics. First, the residuals from the reference isotherm (reference isotherm minus measured isotherm) were determined. Ideally, all residuals should be within the prediction interval of the CO2/ZSM-5 reference isotherm (Uk=2= ± 0.075 mmol/g). This metric provides a qualitative measure of replication of the CO2/ZSM-5 reference isotherm. A second more quantitative metric of replication of the CO2/ZSM 5 reference isotherm, the “goodness-of-fit” (GOF), was also computed. For this test, the reference function from ILS1, which defines the amount of gas adsorbed versus pressure, was compared to each of the CO2/ZSM-5 datasets for new participants in ILS2. The GOF metric was based on the residuals to the reference function, using a Bayesian, Markov Chain Monte Carlo algorithm. This calculation included the uncertainty in the estimates of the reference function as well as the uncertainty of the submitted datasets. The lower the value of this test, the better the dataset replicates the CO2/ZSM-5 reference isotherm. When this test was applied to the datasets of ILS1, the average GOF value was 0.078, with a standard deviation of 0.048. With this range as a guide, it was decided that CO2/ZSM-5 isotherms submitted to ILS2 with a GOF value less than 0.07 would be considered to have replicated the reference isotherm, those with a value between 0.07 and 0.12 would be evaluated on a case-by-case basis, and those with a value greater than 0.12 would be considered as failing to replicate the reference isotherm and submitted CH4/ZY isotherm data would not be included in determining the reference function for ILS2. Of course, passing the GOF test was a necessary—but not sufficient—criterion for a CH4/ZY dataset to be included in the determination of the reference isotherm for ILS2. The GOF calculation was done using the OpenBUGS (Lunn et al. 2009) code shown in the Supplemental Information.
2.3 Dataset display
To clearly display a plot including datasets from all participants, the average of the isotherms for each dataset was determined. These are shown in the figures in the text. [There were two datasets (DS 14 & DS 15) for which averaging was not possible. In these cases, one representative isotherm was selected for display.] In determination of the reference isotherm, the full datasets shown in the Supplemental Information were used–not the averaged datasets shown in the figures of the body of the text. The number of each dataset is random and does not correspond to the numeric listing of authors.
2.4 Reference function determination
The CH4/ZY isotherms were fit collectively to the function,
[nex—surface excess uptake (mmol/g), P—equilibrium pressure (MPa), a, b, c, d, and e being empirical parameters shown in Table 2]. The values of a, b, c, d, and e and the associated 95% uncertainty interval to that fit were determined using a Bayesian, Markov Chain Monte Carlo method (Gelman 2013; Possolo and Toman 2007). The 95% uncertainty interval is the expanded uncertainty (Uk=2) with k = 2, or 2 times the uncertainty of the reference value, which equates to 95% coverage probability or 95% level of confidence that the true value lies within the interval (Taylor and Kuyatt 2001). The OpenBUGS code used for the fit is given in the Supplemental Information. This function was selected because it replicated the form of the measured isotherms. No physical significance should be associated with the function or its parameters, following the practice of ILS1.
3 Results and discussions
3.1 CO2/ZSM-5 isotherms
In total, 17 CO2 datasets were submitted by 12 labs, with one lab submitting four datasets, and two labs submitting two datasets (see Figs. S2–S6). The residuals of the datasets (reference isotherm minus measured data) are displayed in Fig. 1. From visual inspection two datasets (DS 4 & DS 14) are noticeably outside of the uncertainty interval of the CO2/ZSM-5 reference function. The datasets were also evaluated for their goodness-of-fit to the CO2/ZSM-5 reference data and placed into one of three categories based on their goodness-of-fit values: pass (GOF < 0.07), borderline (GOF between 0.07 and 0.12), and fail (GOF > 0.12). Among those participating in ILS2 who had not participated in ILS1, four (DS 1, DS 4, DS 10 & DS 14) were identified as having failed the goodness-of-fit test. Seven CO2/ZSM-5 datasets (DS 2, DS 6, DS 7, DS 13, DS 16, DS 17 & DS 18) passed the goodness-of-fit test. Six datasets (DS 3, DS 5 DS 8, DS 9, DS 11 & DS 12) fell into the borderline category. The quality of the residuals of the datasets shown in Fig. 1 correlate well with the goodness-of-fit values (see Supplemental Information). Datasets that failed and some datasets borderline in the GOF test exhibit slightly greater variability in the intralaboratory replicate isotherms (Figs. S3–S5), although in general, intralaboratory isotherms have good reproducibility.
CO2 adsorption on ZSM-5. Residuals [nex, ref(P) – Measured] for those who did not participate (1–18) in the previous interlaboratory study (ILS1). Other participants (19–28) participated in an earlier study (ILS1), demonstrating their measurement capabilities. (Isotherms are shown in the Supplemental Information). Residuals have units of mmol/g
For the interested reader, ILS1 is a great resource providing pitfalls and recommendations for high-pressure CO2 measurement (Nguyen et al. 2018). The participants of the failed datasets (DS 1, DS 4, DS 10, and DS 14) were unable to remeasure, reprocess, or (in one case) improve the data to identify the origin for the deviation. However, two failed datasets came from labs that each had at least one other CO2/ZSM-5 dataset that didn’t fail the GOF test, suggesting an issue with their instrument or method rather than with the material. Evaluation of the experimental details indicated the activation protocol was not followed for one case (DS 14), which could explain the lower uptake. The importance of following the activation protocol was explicitly highlighted in ILS1(Nguyen et al. 2018), and modeling work based on the CO2/ZSM-5 reference data underscored that varying the pretreatment temperature of ammonium ZSM-5 affects what cations are in the zeolite (Fang et al. 2020).
3.2 CH4/ZY isotherms
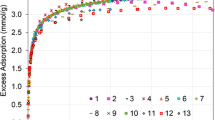
All participants were allowed to submit CH4/ZY isotherms, although CH4/ZY datasets from labs and/or instruments that failed to replicate the CO2/ZSM 5 isotherm would automatically be excluded in the determination of the reference isotherm for ILS2. The as-submitted CH4/ZY datasets, excluding DS 14, which did not follow the prescribed activation protocol, were plotted together and Eq. (1) was fitted to them. This process identified ten datasets that were statistical outliers (DS 1, DS 4, DS 9, DS 10, DS 11, DS 12, DS 14, DS 15, DS 16 & DS 27), with datapoints lying outside the 95% uncertainty interval. These participants were given the opportunity to remeasure or reprocess their results. Four datasets (DS 4, DS 9, DS 11 & DS 12) were resubmitted after review of the initial result indicated they were outliers. All final submitted datasets are shown in Fig. 2. In general, the intralaboratory isotherms for the datasets were highly reproducible (except for DS 14 and 15; see Figs. S6–S10 in the Supplemental Information). Four datasets (DS 1, DS 4, DS 10 & DS 14) failed the CO2 test, and one participant (DS 15) failed to replicate the CO2/ZSM-5 isotherm by not submitting CO2 isotherm data. The remaining 23 datasets were fitted to Eq. (1), which along with expert judgement, re-identified two datasets as statistical outliers (DS 16 & DS 27), while 21 datasets appear to be in good agreement. DS 27 is a participant in ILS1 who used a different instrument in ILS2 and did not have CO2/ZSM-5 isotherms for the new instrumentation. Only one (DS 16) of the outlying CH4/ZY datasets successfully reproduced the CO2/ZSM-5 isotherm. The resubmission led to improvements in three datasets (DS 9, DS 11, and DS 12) with identified reasons for the observed variation in the outlying surface excess datasets including temperature instability (leading to volume calibration, and sample and blank isotherms being performed under different conditions), inaccurate mass measurement (due to balance mass drift), and error in application of blank subtraction, stressing the importance of following the recommendations outlined in the ILS1 paper. After eliminating the datasets that did not pass the proficiency test or had another identified issue, 21 datasets remained, as shown in Fig. 3.
Top: Datasets used to extract the reference isotherm for CH4 adsorption on Zeolite Y, with the associated 95% confidence interval (Isotherms shown here are the average of all four submitted, see Supplemental Information for full datasets). Bottom: Residuals from reference function [nex,ref(P) – Measured]. Residuals have units of mmol/g
An empirical surface excess reference function was determined by optimizing the fit of Eq. (1) to the final remaining datasets and is also shown in Fig. 3. The optimized parameters are given in Table 2. This function is predictive up to 7.5 MPa and has expanded uncertainty, Uk=2, for the excess uptake of approximately 0.09 mmol/g over the full pressure range. Beneath the isotherms are the residuals (reference function minus measured isotherm) along with Uk=2. The residuals show that the reference function adequately represents the final set of isotherms over the full pressure range of the study. The datasets and the reference isotherm are available through the NIST Database of Novel and Emerging Adsorbent Materials.Footnote 3
4 Conclusions and outlook
This work provides an empirical reference surface excess isotherm function for high-pressure CH4 adsorption on Zeolite Y (NIST Reference Material 8850) at 25 °C up to 7.5 MPa, the second data produced in such a way by the FACT Lab through an interlaboratory study. This reference isotherm should prove useful for researchers interested in working with CH4 or supercritical adsorption measurements. The study also demonstrated the usefulness of reference isotherms and reference materials for evaluating the reliability of high-pressure adsorption experiments; specifically, a lab’s inability to replicate the CO2/ZSM-5 reference isotherm was highly correlated with outlier CH4/ZY data.
The FACT Lab plans to continue efforts to develop reference materials, reference data, and measurement protocols to improve adsorption metrology. Forthcoming interlaboratory studies will explore different adsorptive gases and vapors (H2, N2, H2O, etc.), other temperature ranges (e.g., cryogenic), as well as multicomponent adsorption measurements.
Notes
References
Agrawal, M., Han, R., Herath, D., Sholl, D. S.: Does repeat synthesis in materials chemistry obey a power law? Proc. Nat. Acad. Sci. 117(2), 877 (2020). https://doi.org/10.1073/pnas.1918484117
Baerlocher, C., McCusker, L.B., Olson, D.H.: Atlas of Zeolite Framework Types, 6th edn. Elsevier Science, Amsterdam (2007)
Beckner, M., Dailly, A.: Hydrogen and methane storage in adsorbent materials for automotive applications. Int. J. Energy Res. 40(1), 91–99 (2016). https://doi.org/10.1002/er.3324
Broom, D.P., Hirscher, M.: Irreproducibility in hydrogen storage material research. Energy Environ. Sci. 9(11), 3368–3380 (2016). https://doi.org/10.1039/c6ee01435f
Broom, D.P., Webb, C.J.: Pitfalls in the characterisation of the hydrogen sorption properties of materials. Int. J. Hydrogen Energy 42(49), 29320–29343 (2017). https://doi.org/10.1016/j.ijhydene.2017.10.028
Cejka, J., Morris, R.E., Nachtigall, P.: Zeolites in Catalysis: Properties and Applications. Royal Society of Chemistry, Cambridge (2017)
Chareonsuppanimit, P., Mohammad, S.A., Robinson, R.L., Gasem, K.A.M.: High-pressure adsorption of gases on shales: measurements and modeling. Int. J. Coal Geol. 95, 34–46 (2012). https://doi.org/10.1016/j.coal.2012.02.005
Darkrim, F.L., Malbrunot, P., Tartaglia, G.P.: Review of hydrogen storage by adsorption in carbon nanotubes. Int. J. Hydrogen Energy 27(2), 193–202 (2002). https://doi.org/10.1016/S0360-3199(01)00103-3
Espinal, L., Poster, D.L., Wong-Ng, W., Allen, A.J., Green, M.L.: Measurement, standards, and data needs for CO2 capture materials: a critical review. Environ. Sci. Technol. 47(21), 11960–11975 (2013). https://doi.org/10.1021/es402622q
Fang, H., Findley, J., Muraro, G., Ravikovitch, P.I., Sholl, D.S.: A strong test of atomically detailed models of molecular adsorption in zeolites using multilaboratory experimental data for CO2 adsorption in ammonium ZSM-5. J. Phys. Chem. Lett. 11(2), 471–477 (2020). https://doi.org/10.1021/acs.jpclett.9b02986
Gasparik, M., Rexer, T.F.T., Aplin, A.C., Billemont, P., De Weireld, G., Gensterblum, Y., et al.: First international inter-laboratory comparison of high-pressure CH4, CO2 and C2H6 sorption isotherms on carbonaceous shales. Int. J. Coal Geol. 132, 131–146 (2014). https://doi.org/10.1016/j.coal.2014.07.010
Gelman, A.C., Stern, H., Dunson, D., Vehtari, A., Rubin, D.: Bayesian Data Analysis. Chapman & Hall, Boca Raton (2013)
Gensterblum, Y., van Hemert, P., Billemont, P., Busch, A., Charriere, D., Li, D., et al.: European inter-laboratory comparison of high pressure CO2 sorption isotherms. I: activated carbon. Carbon 47(13), 2958–2969 (2009). https://doi.org/10.1016/j.carbon.2009.06.046
Gensterblum, Y., van Hemert, P., Billemont, P., Battistutta, E., Busch, A., Krooss, B.M., et al.: European inter-laboratory comparison of high pressure CO2 sorption isotherms II: natural coals. Int. J. Coal Geol. 84(2), 115–124 (2010). https://doi.org/10.1016/j.coal.2010.08.013
Goodman, A.L., Busch, A., Duffy, G.J., Fitzgerald, J.E., Gasem, K.A.M., Gensterblum, Y., et al.: An inter-laboratory comparison of CO2 isotherms measured on Argonne premium coal samples. Energy Fuels 18(4), 1175–1182 (2004). https://doi.org/10.1021/ef034104h
Goodman, A.L., Busch, A., Bustin, R.M., Chikatamarla, L., Day, S., Duffy, G.J., et al.: Inter-laboratory comparison II: CO2 isotherms measured on moisture-equilibrated Argonne premium coals at 55 degrees C and up to 15 MPa. Int. J. Coal Geol. 72(3–4), 153–164 (2007). https://doi.org/10.1016/j.coal.2007.01.005
Han, R., Walton, K.S., Sholl, D.S.: Does chemical engineering research have a reproducibility problem? Annu. Rev. Chem. Biomol. Eng. 10(1), 43–57 (2019). https://doi.org/10.1146/annurev-chembioeng-060718-030323
Hurst, K.E., Parilla, P.A., O'Neill, K.J., Gennett, T.: An international multi-laboratory investigation of carbon-based hydrogen sorbent materials. Appl. Phys. A 122(1), 1–9 (2016). https://doi.org/10.1007/s00339-015-9537-x
Hurst, K.E., Gennett, T., Adams, J., Allendorf, M.D., Balderas-Xicohténcatl, R., Bielewski, M., et al.: An international laboratory comparison study of volumetric and gravimetric hydrogen adsorption measurements. ChemPhysChem 20(15), 1997–2009 (2019). https://doi.org/10.1002/cphc.201900166
Julbe, A., Drobek, M.: Zeolite Y type. In: Drioli, E., Giorno, L. (eds.) Encyclopedia of Membranes, pp. 2060–2061. Springer, Berlin, Heidelberg (2016)
Keskin Avci, S., Erucar, I.: 2.7 porous materials. In: Dincer, I. (ed.) Comprehensive Energy Systems, pp. 182–203. Elsevier, Oxford (2018)
Kunz, O., Wagner, W.: The GERG-2008 wide-range equation of state for natural gases and other mixtures: an expansion of GERG-2004. J. Chem. Eng. Data 57(11), 3032–3091 (2012). https://doi.org/10.1021/je300655b
Liu, J., Thallapally, P.K., McGrail, B.P., Brown, D.R., Liu, J.: Progress in adsorption-based CO2 capture by metal–organic frameworks. Chem. Soc. Rev. 41(6), 2308–2322 (2012). https://doi.org/10.1039/C1CS15221A
Lunn, D., Spiegelhalter, D., Thomas, A., Best, N.: The BUGS project: evolution, critique and future directions. Stat. Med. 28(25), 3049–3067 (2009). https://doi.org/10.1002/sim.3680
Mason, J.A., Veenstra, M., Long, J.R.: Evaluating metal–organic frameworks for natural gas storage. Chem. Sci. 5(1), 32–51 (2014). https://doi.org/10.1039/C3SC52633J
Menon, P.G.: Adsorption at high pressures. Chem. Rev. 68(3), 277–294 (1968). https://doi.org/10.1021/cr60253a002
Menon, V.C., Komarneni, S.: Porous adsorbents for vehicular natural gas storage: a review. J. Porous Mater. 5(1), 43–58 (1998). https://doi.org/10.1023/A:1009673830619
Moretto, P., Zlotea, C., Dolci, F., Amieiro, A., Bobet, J.L., Borgschulte, A., et al.: A Round Robin test exercise on hydrogen absorption/desorption properties of a magnesium hydride based material. Int. J. Hydrogen Energy 38(16), 6704–6717 (2013). https://doi.org/10.1016/j.ijhydene.2013.03.118
Nguyen, H.G.T., Horn, J.C., Thommes, M., van Zee, R.D., Espinal, L.: Experimental aspects of buoyancy correction in measuring reliable high-pressure excess adsorption isotherms using the gravimetric method. Meas. Sci. Technol. 28(12), 125802 (2017)
Nguyen, H.G.T., Espinal, L., van Zee, R.D., Thommes, M., Toman, B., Hudson, M.S.L., et al.: A reference high-pressure CO2 adsorption isotherm for ammonium ZSM-5 zeolite: results of an interlaboratory study. Adsorption 24(6), 531–539 (2018). https://doi.org/10.1007/s10450-018-9958-x
Park, J., Howe, J.D., Sholl, D.S.: How reproducible are isotherm measurements in metal-organic frameworks? Chem. Mater. 29(24), 10487–10495 (2017). https://doi.org/10.1021/acs.chemmater.7b04287
Possolo, A., Toman, B.: Assessment of measurement uncertainty via observation equations. Metrologia 44(6), 464–475 (2007). https://doi.org/10.1088/0026-1394/44/6/005
Setzmann, U., Wagner, W.: A new equation of state and tables of thermodynamic properties for methane covering the range from the melting line to 625 K at pressures up to 1000 MPa. J. Phys. Chem. Ref. Data 20(6), 1061–1155 (1991). https://doi.org/10.1063/1.555991
Span, R., Wagner, W.: A new equation of state for carbon dioxide covering the fluid region from the triple-point temperature to 1100 K at pressures up to 800 MPa. J. Phys. Chem. Ref. Data 25(6), 1509–1596 (1996). https://doi.org/10.1063/1.555991
Span, R., Wagner, W.: Equations of state for technical applications. II. Results for nonpolar fluids. Int. J. Thermophys. 24(1), 41–109 (2003). https://doi.org/10.1023/A:1022310214958
Taylor, B. N., & Kuyatt, C. E. (2001). NIST Technical Note 1297: Guidelines for Evaluating and Expressing the Uncertainty of NIST Measurement Results. https://physics.nist.gov/TN1297. Accessed 2 July 2020
Turner, S., Sieber, J.R., Vetter, T.W., Zeisler, R., Marlow, A.F., Moreno-Ramirez, M.G., et al.: Characterization of chemical properties, unit cell parameters and particle size distribution of three zeolite reference materials: RM 8850-zeolite Y, RM 8851-zeolite A and RM 8852-ammonium ZSM-5 zeolite. Microporous Mesoporous Mater. 107(3), 252–267 (2008). https://doi.org/10.1016/j.micromeso.2007.03.019
White, C.M., Smith, D.H., Jones, K.L., Goodman, A.L., Jikich, S.A., LaCount, R.B., et al.: Sequestration of carbon dioxide in coal with enhanced coalbed methane recovery: a review. Energy Fuels 19(3), 659–724 (2005). https://doi.org/10.1021/ef040047w
Yang, R.T.: Adsorbents: Fundamentals and Applications. Wiley, Hoboken (2003)
Zlotea, C., Moretto, P., Steriotis, T.: A round robin characterisation of the hydrogen sorption properties of a carbon based material. Int. J. Hydrogen Energy 34(7), 3044–3057 (2009). https://doi.org/10.1016/j.ijhydene.2009.01.079
Acknowledgements
The instruments of the Facility for Adsorbent Characterization and Testing were funded by the Advanced Research Projects Agency-Energy (ARPA-E) through Interagency Agreement № 1208-0000.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nguyen, H.G.T., Sims, C.M., Toman, B. et al. A reference high-pressure CH4 adsorption isotherm for zeolite Y: results of an interlaboratory study. Adsorption 26, 1253–1266 (2020). https://doi.org/10.1007/s10450-020-00253-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-020-00253-0