Abstract
Objectives
To increase the efficiency of enzymatic hydrolysis for plant biomass conversion into renewable biofuel and chemicals.
Results
By overexpressing the point mutation A824 V transcriptional activator Xyr1 in Trichoderma reesei, carboxymethyl cellulase, cellobiosidase and β-d-glucosidase activities of the best mutant were increased from 1.8 IU/ml, 0.1 IU/ml and 0.05 IU/ml to 4.8 IU/ml, 0.4 IU/ml and 0.3 IU/ml, respectively. The sugar yield of wheat straw saccharification by combining enzymes from this mutant and the Aspergillus niger genetically modified strain ΔcreA/xlnR c/araR c was improved up to 7.5 mg/ml, a 229 % increase compared to the combination of wild type strains.
Conclusions
Mixing enzymes from T. reesei and A. niger combined with the genetic modification of transcription factors is a promising strategy to increase saccharification efficiency.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Depleting fossil energy sources and related environmental issues have stimulated great interest in alternative renewable biofuel sources (Nigam and Singh 2011). Plant biomass is a promising source of raw material for biofuel production (Klemm et al. 2005). A key bottleneck for the bioconversion of plant biomass into biofuels is the high cost of enzymes and their insufficient efficiency in releasing sugars from lignocellulose (Falkoski et al. 2012). To achieve cost-effective bioconversions, maximizing the enzyme yield and optimizing enzyme mixtures is considered an attractive approach with great potential (Pal and Chakraborty 2013).
Trichoderma reesei and Aspergillus niger are established industrial producers of enzymes that degrade plant biomass (Dos Santos Castro et al. 2014). The main activators of cellulolytic and xylanolytic gene expression in T. reesei and A. niger are Xyr1 and XlnR, respectively (Stricker et al. 2006) (van Peij et al. 1998). Constitutive expression of Xyr1 in T. reesei leads to a significant elevation of the production of the main biomass degrading enzymes (Mach-Aigner et al. 2008). The mutation A824V of Xyr1 improves xylanase and basal cellulase gene expression in T. reesei (Derntl et al. 2013). In A. niger, constitutive activation of XlnR also results in constant xylanase production even under repressing conditions (Hasper et al. 2004). A. niger contains a second hemicellulolytic transcriptional activator, AraR, which has sequence homology to XlnR but is only present in the Eurotiales (Battaglia et al. 2011). This regulator controls the production of extracellular L-arabinose releasing enzymes in A. niger.
Although T. reesei produces an efficient set of cellulolytic enzymes, β-d-glucosidase production is low, while the opposite was reported for A. niger (van den Brink and de Vries 2011). Therefore, plant biomass hydrolysis can be improved by using T. reesei cellulase complemented with enzymes from other organism (Tabka et al. 2006). In this study, improvement of wheat straw saccharification was achieved by overexpressing a constitutive version of xyr1 in T. reesei and combining the enzymes produced by this strain with those from an A. niger strain in which creA was deleted and constitutive versions of araR and xlnR were inserted.
Materials and methods
Microbial strains, plasmids and cultivation
Escherichia coli DH5α was used for cloning. Agrobacterium tumefaciens AGL-1 served as a T-DNA donor for T. reesei transformation. All fungal strains are listed in Supplementary Table 1. A uridine auxotrophic ura5- negative strain 2-3 was used for overexpression of native or mutated A824V xyr1. A T-DNA binary vector, Npxbthg, containing the selecting marker gene ura5 coding for orotate phosphoribosyltransferase from Penicillium oxalicum under control of the Aspergillus nidulans gpdA promoter and trpC terminator, was used to construct the transformation vectors for T. reesei.
Transformants of T. reesei were selected as described previously (Gruber et al. 1990), with cefotaxime (300 μM) and 0.2 % Triton X-100 (v/v). Each positive transformant was used to generate genetically stable and correct mono-conidia, which were confirmed by PCR. All Aspergillus strains were cultivated in Aspergillus minimal medium (de Vries et al. 2004). Wheat straw, xylan from beechwood, Avicel, glucose and xylose were used as carbon sources. An A. niger strain with a creA deletion was used (van den Brink et al. 2014).
The expression of araR and xlnR was made constitutive by deleting the C-terminus from Pro646 and Leu668, respectively (Hasper et al. 2004). The A. niger ΔcreA background was used in both case.
Overexpression of native or constitutive T. reesei xyr1
The native and mutated A824V xyr1 PCR fragment were amplified from genomic DNA of QM9414 by using primers in Supplementary Table 2 and cloned between the pdc promoter and trpC terminator into the BamHI site of Npxbthg using the ClonExpress MultiS kit (Vazyme Biotech) resulting in vector Npxbthg-OEXyr1 and Npxbthg-OEmXyr1. These vectors were transformed into the recipient strain using A. tumefaciens-mediated fungal transformation.
Cellulase activity, SDS-PAGE and silver staining
Activity of carboxymethyl cellulase (CMCase), cellobiosidase (pNPCase) and β-d-glucosidase (pNPGase) were assayed as described previously (Ma et al. 2011). Significance analysis was performed by Student’s t-test using a two-tailed test. SDS-PAGE (12 %, w/v) with silver staining was performed to analyze extracellular protein profiles. Due to interference of wheat straw with the usual protein determination methods, the total protein determination was estimated by visual inspection of the SDS-PAGE loaded with 20 μl of samples.
Saccharification of wheat straw
Saccharification was performed in 10 ml containing 2 ml enzyme mixtures, 8 ml saline/sodium acetate buffer (pH 5, 50 mM) and 0.3 g washed wheat straw, and incubated at 30 °C and 200 rpm. The ratio of the enzyme mixtures from T. reesei and A. niger was 1:1 taken from wheat straw liquid (3 %, w/v) after 4 days of cultivation and filtered through a 0.22 µm nylon filter (van den Brink et al. 2014). For control measurements, 2 ml saline/sodium acetate buffer (pH 5, 50 mM) substituted the enzyme mixtures. The reducing sugar yield was quantified daily using the DNS method (Ma et al. 2011). HPLC was used to determine individual sugar concentrations. Sugar analysis was performed on a Thermo Scientific 5000 + HPLC-PAD system. A multi-step gradient was used to analyze the sugars. A flow rate of 0.3 ml/min was used on a CarboPac PA1 column. Guard column: Dionex CarboPac PA1 BioLC 2 × 50 mm. Main column: Dionex CarboPac PA1 BioLC 2 × 250 mm. A pre flow of 18 mM NaOH was used before injection to equilibrate the column. During a total running time of 50 min, the following solutions were used: A, water; B, 100 mM NaOH; C, 100 mM NaOH with 1 M sodium acetate. 18 % B was used for the first 20 min followed by a 10 min linear gradient to 40 % C, 0 % B. 100 % C for 5 min. To rinse the acetate, 100 % B was used for 5 min. 10 min 18 % B was used for 10 min to rinse the column. Quantification was performed by external standard calibration. Reference sugars (Sigma-Aldrich) were used from 2.5 to 200 µM. The data obtained are the results of two independent biological replicates and for each replicate three technical replicates were assayed.
Results and discussion
Overexpression of native and constitutive xyr1 leads to significant increase of enzyme yields
The three T. reesei strains: wild type, overexpressed xyr1 and overexpressed constitutive xyr1, were grown on minimal medium with 1 % carbon source (xylan, xylose, glucose, wheat straw and Avicel). No significant growth improvement between the T. reesei wild type and Xyr1 mutant strains was observed on glucose, Avicel or wheat straw (Fig. 1). In contrast, on xylan and xylose, the colony size of the Xyr1 mutants was considerably increased compared to the wild type. This correlates with the previously demonstrated role of Xyr1 in xylan and xylose utilization (Stricker et al. 2006). Furthermore, overexpression of the constitutive xyr1 resulted in better growth than that of native xyr1, suggesting the point mutation (A824V) in the gene encoding Xyr1 enhances the utilization ability of xylan and xylose by T. reesei.
Growth profiles of the parental strain (WT), QM9414 uridine auxotrophic mutant; OExyr1-1 and OExyr1-2, overexpressed xyr1; OEmxyr1, overexpressed constitutive xyr1 (A804V). 1000 spores in 2 µl ACES were spotted on the center of minimal medium plates with 1 % carbon source (W/V, xylan from beechwood, xylose, glucose, wheat straw and Avicel, respectively) and incubated at 30 °C. Pictures were taken three days (3d) after inoculation. The arrows show the edges of the colonies
Cellulase activities of T. reesei wild type and the native or constitutive xyr1 transformants OExyr1-1, OExyr1-2, OEmxyr1 were measured in supernatants from Avicel and wheat straw cultures. All three strains produced a larger amount of secreted proteins than the wild type (Fig. 2). This result contrasts with the overexpression of XlnR in Aspergillus vadensis where additional copies of the transcriptional activator had no effect on protein production (Bouzid and de Vries 2014). OEmxyr1 produced the highest protein level and showed the earliest protein production, particularly in Avicel (Fig. 2a). The other two mutants showed an earlier secretion of proteins with wheat straw than with Avicel (Fig. 2b). This may be linked to a higher fungal biomass formation as suggested by their growth profiles (Fig. 1).
Exo-protein profiles of the wild type and mutant strains grown on Avicel and wheat straw. The strains were inoculated at 106 spores/ml into 250 ml flasks containing 50 ml minimal medium with a Avicel PH-101(1 %, W/V) or b wheat straw (3 %, W/V) and cultivated at 30 °C, 200 rpm. Culture supernatants of the fungal strains were collected after four (4d) and seven (7d) days by centrifugation at 4 °C and 12,000×g for 10 min
Consistent with extracellular protein production, cellulase activity of the mutant strains was considerably improved compared to the wild type, in agreement with earlier reports (Mach-Aigner et al. 2008; Derntl et al. 2013). Interestingly, the overexpressed constitutive mutant OEmxyr1, had an even greater effect. After 4 days of growth, both in Avicel and wheat straw, only OEmxyr1 produced high cellulase activity. Moreover, after 7 days’ growth on Avicel, OEmxyr1 showed increased CMCase, pNPCase and pNPGase activities from 1.8 IU/ml, 0.1 IU/ml and 0.05 IU/ml to 4.8 IU/ml, 0.4 IU/ml and 0.3 IU/ml, respectively, (Fig. 3a–c). No significant differences were observed for the pNPGase in wheat straw medium, but OEmxyr1 produced more of this activity in Avicel medium (Fig. 3c), demonstrating the influence of the carbon source on the regulation of the β-d-glucosidase in the constitutive Xyr1 strain (Castro et al. 2014).
Cellulase activities measured from the wild type and the mutant strains grown on Avicel and wheat straw. a CMCase, b pNPCase and c pNPGase activities. The culture condition and the sample treatment were the same as for Fig. 1. Vertical bars indicate standard deviation of biological duplicates
Enhancing wheat straw saccharification
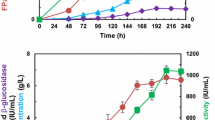
To evaluate the effect of the overexpressed constitutive Xyr1, saccharification experiments were performed on wheat straw using supernatants of wheat straw-grown cultures. T. reesei OExyr1 and OEmxyr1 showed higher release of sugar compared to the reference strain (Fig. 4). The OEmxyr1 released by far the largest amount of sugar which is in line with the measured activities. Interestingly, this increased saccharification capacity is mainly due to xylose release and not due to release of glucose (Fig. 4b).
A triple mutant from Aspergillus niger was further added to the experiment. This triple mutant is a double constitutive araR and xlnR made from a ΔcreA background (ΔcreA/xlnR c/araR c).
Combining the enzyme mixtures of these strains with the enzyme mixtures of the T. reesei wild type significantly improved saccharification. Combining enzyme mixtures of OExyr1 and OEmxyr1 with A. niger ΔcreA/xlnR c/araR c resulted in even better wheat straw saccharification and the best sugar yield (7.51 mg/ml) was achieved by combining enzymes from OEmxyr1 and mutant ΔcreA/xlnR c/araR c. This is 229 % of the combination of wild type strains (Fig. 4), which goes beyond results obtained previously (van den Brink et al. 2014).
In conclusion, we significantly increased the saccharification of wheat straw by combining the enzyme mixtures of two engineered strains: T. reesei OEmxyr1 and A. niger ΔcreA/xlnR c/araR c.
References
Battaglia E, Visser L, Nijssen A, van Veluw GJ, Wosten HAB, de Vries RP (2011) Analysis of regulation of pentose utilisation in Aspergillus niger reveals evolutionary adaptations in Eurotiales. Stud Mycol 69:31–38
Bouzid O, de Vries RP (2014) Overexpression of XlnR regulated hemicellulase-encoding genes in Aspergillus vadensis. Curr Biotechnol 3:252–256
Castro LD, Antonieto AC, Pedersoli WR, Rocha RS, Persinoti GF, Silva RN (2014) Expression pattern of cellulolytic and xylanolytic genes regulated by transcriptional factors XYR1 and CRE1 are affected by carbon source in Trichoderma reesei. Gene Expr Patt 14:88–95
de Vries RP, Burgers K, van de Vondervoort PJI, Frisvad JC, Samson RA, Visser J (2004) A new black Aspergillus species, A. vadensis, is a promising host for homologous and heterologous protein production. Appl Environ Microbiol 70:3954–3959
Derntl C, Gudynaite-Savitch L, Calixte S, White T, Mach RL, Mach-Aigner AR (2013) Mutation of the Xylanase regulator 1 causes a glucose blind hydrolase expressing phenotype in industrially used Trichoderma strains. Biotechnol Biofuel 6:62
Dos Santos Castro L, Pedersoli WR, Antonieto AC, Steindorff AS, Silva-Rocha R, Martinez-Rossi NM, Rossi A, Brown NA, Goldman GH, Faca VM et al (2014) Comparative metabolism of cellulose, sophorose and glucose in Trichoderma reesei using high-throughput genomic and proteomic analyses. Biotechnol Biofuel 7:41
Falkoski DL, Guimaraes VM, de Almeida MN, Alfenas AC, Colodette JL, de Rezende ST (2012) Characterization of cellulolytic extract from Pycnoporus sanguineus PF-2 and its application in biomass saccharification. Appl Biochem Biotechnol 166:1586–1603
Gruber F, Visser J, Kubicek CP, de Graaff LH (1990) The development of a heterologous transformation system for the cellulolytic fungus Trichoderma reesei based on a pyrG-negative mutant strain. Curr Genet 18:71–76
Hasper AA, Trindade LM, van der Veen D, van Ooyen AJ, de Graaff LH (2004) Functional analysis of the transcriptional activator XlnR from Aspergillus niger. Microbiology 150:1367–1375
Klemm D, Heublein B, Fink HP, Bohn A (2005) Cellulose: fascinating biopolymer and sustainable raw material. Angew Chem Int Ed 44:3358–3393
Ma L, Zhang J, Zou G, Wang C, Zhou Z (2011) Improvement of cellulase activity in Trichoderma reesei by heterologous expression of a beta-glucosidase gene from Penicillium decumbens. Enzym Microb Technol 49:366–371
Mach-Aigner AR, Pucher ME, Steiger MG, Bauer GE, Preis SJ, Mach RL (2008) Transcriptional regulation of xyr1, encoding the main regulator of the xylanolytic and cellulolytic enzyme system in Hypocrea jecorina. Appl Environ Microbiol 74:6554–6562
Nigam PS, Singh A (2011) Production of liquid biofuels from renewable resources. Prog Energy Combust Sci 37:52–68
Pal RK, Chakraborty S (2013) A novel mixing strategy for maximizing yields of glucose and reducing sugar in enzymatic hydrolysis of cellulose. Biores Technol 148:611–614
Stricker AR, Grosstessner-Hain K, Wurleitner E, Mach RL (2006) Xyr1 (xylanase regulator 1) regulates both the hydrolytic enzyme system and D-xylose metabolism in Hypocrea jecorina. Eukaryot Cell 5:2128–2137
Tabka MG, Herpoel-Gimbert I, Monod F, Asther M, Sigoillot JC (2006) Enzymatic saccharification of wheat straw for bioethanol production by a combined cellulase xylanase and feruloyl esterase treatment. Enzym Microb Technol 39:897–902
van den Brink J, de Vries RP (2011) Fungal enzyme sets for plant polysaccharide degradation. Appl Microbiol Biotechnol 91:1477–1492
van den Brink J, Maitan-Alfenas GP, Zou G, Wang C, Zhou Z, Guimaraes VM, de Vries RP (2014) Synergistic effect of Aspergillus niger and Trichoderma reesei enzyme sets on the saccharification of wheat straw and sugarcane bagasse. Biotechnol J 9:1329–1338
van Peij N, Gielkens MMC, de Vries RP, Visser J, de Graaff LH (1998) The transcriptional activator XlnR regulates both xylanolytic and endoglucanase gene expression in Aspergillus niger. Appl Environ Microbiol 64:3615–3619
Acknowledgments
AVD, JvdB and IB were supported by a grant of The Netherlands Organisation for Scientific Research (NWO) of the China–Netherlands Joint Scientific Thematic Research Programme (jstp.10.005) to RPdV. IB was supported by a grant of the Dutch Technology Foundation STW, applied science division of NWO, and the Technology Program of the Ministry of Economic Affairs 016.130.609 to RPdV.
Supporting information
Supplementary Table 1—List of T. reesei and A. niger strains.
Supplementary Table 2—List of primers.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Jiang, Y., Duarte, A.V., van den Brink, J. et al. Enhancing saccharification of wheat straw by mixing enzymes from genetically-modified Trichoderma reesei and Aspergillus niger . Biotechnol Lett 38, 65–70 (2016). https://doi.org/10.1007/s10529-015-1951-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-015-1951-9