Abstract
Monitoring population parameters of invasive species gains importance as these species continue to expand all over the world. Monitoring of invasive mammalian mesopredators is, however, complicated due to their nocturnal and secretive behaviour. In the European Union, the most common invasive mesopredator is the raccoon dog (Nyctereutes procyonoides), which causes concerns for native species, such as endangered waterfowl that may be subject to nest predation. We studied the density of mesopredators in southern Finland with wildlife cameras, using methodology of distance sampling. We deployed in total of 175 camera traps around 11 (spring 2020) and 16 (spring 2021) lakes or wetlands. We inferred densities for raccoon dogs, and for native mesopredators the red fox (Vulpes vulpes) and the European badger (Meles meles) for comparison. Raccoon dogs were found to have higher overall as well as site-specific densities (about 3.7 ind./km2) than badgers (1.2 ind./km2) and red foxes (0.6 ind./km2). The raccoon dogs also were present at every study wetland, while badgers were not found at all sites. The red fox showed more diurnal activity compared to raccoon dogs and badgers. Camera trap distance sampling enabled us to provide a density estimates on a rather small spatial and temporal scale for species of similar size and movement speed. It could therefore prove valuable as a long-term monitoring option, as climate trends are likely to further enable raccoon dog expansion. Currently this invasive species appears to be the most common mesopredator around wetlands in the southern boreal zone of southern Finland.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Estimating population density is a crucial instrument in wildlife management, and at the basis of many management decisions (Santini et al. 2022). It allows for insight and assessment of a population status and choosing for appropriate measures to support a healthy ecosystem. Based on such estimates, active measures may be taken to support, protect or supplement a struggling population. At the same time, it may also make the need for pest- or predator control measures apparent. This is especially important in handling invasive species with the potential to negatively affect or compete with local native species (Directorate-General for Environment 2022).
However, estimating density or abundance of a species may not be a straightforward task. Monitoring of mammalian mesopredators, for example, is complicated due to nocturnal and secretive living habits of these species (Sadlier et al. 2004; Podgórski et al. 2020). This is the case for the raccoon dog (Nyctereutes procyonoides), which in large parts of the European Union is the most common invasive mesopredator (Directorate-General for Environment 2022). Clear data on its density and abundanse is lacking, for example, in Finland, where raccoon dogs spread from the former Western Soviet Union where it was introduced from its original range in East Asia. Since then, raccoon dogs have been called one of the most successful alien carnivores introduced to Europe (Kauhala and Kowalczyk 2011).
Raccoon dogs are quick to adapt to new regions and individuals have previously been reported to have dispersed up to 300 km in a year. Coupled with a high reproductive rate, this gives them large potential to rapidly cover more area (Kauhala and Kowalczyk 2011). In an IUCN assessment from 2016, the adult spring population in Finland was given as an expert evaluation (with no quantitative census available) at 110,000–120,000 individuals (IUCN 2016). Climate change is also likely to further enable the spread of this species, as harsh winters are assumed to present a limiting factor to their habitable area (Melis et al. 2007). Raccoon dog is considered a harmful invasive species for many reasons (Kauhala and Kowalczyk 2011). It is an important vector of rabies in Europe, transmitting also fox tapeworm, sarcoptic mange and trichinellosis (Kauhala and Kowalczyk 2011). It is also suspected of causing damage to populations of ground-nesting birds, among them game birds such as waterfowl and grouse (Holopainen et al. 2021). This mainly refer to destruction of nests and clutches, potentially leading to or contributing to a decline in breeding success (Dahl and Åhlén 2019).
The density of predators is ultimately decisive in how much pressure is placed on native species in the area in question. It is hereby important to consider not just the invader in isolation, but to investigate its density in the context of the density of native species which inhabit the same niche or make use of overlapping food sources. Mesopredator species that are comparable to the raccoon dog in northern and central Europe are the red fox (Vulpes vulpes) and the European badger (Meles meles) (Jędrzejewska and Jędrzejewski 1998). While there may not necessarily be antagonistic interactions between these species, through interspecific competition the presence of the newcomer has potential to displace or suppress the native populations (Drygala et al. 2013). Certainly, the invasive species add to the exploitation of common food sources. Knowledge of population trends and spatial distribution of the raccoon dog are relevant for planning and inserting control measures or for reviewing their effectiveness. The assessments of raccoon dog population numbers have previously included surveys of hunting bag and roadkill numbers, or yearly game inquiries. However, the accuracy of such estimates largely depends on the collaboration and communication of several parties involved and may be prone to subjective bias (Balčiauskas et al. 2021). In particular, population estimates would be welcome in areas where invasive species are likely to cause harm for native species, such as areas surrounding wetlands in the case of the raccoon dog.
Recently, camera traps (CT) have proven to be an effective tool in wildlife monitoring (Waern and Glover-Kapfer 2019). Methods to retrieve reliable population estimates from camera trap data are continuously being created and improved (Santini et al. 2022). For example, camera trap distance sampling (CT-DS) is a methodology developed to create estimates of density and/or abundance of species, without the need for individual recognition (CREEM 2022). This approach utilizes the distance of a recorded object to the observer during detection, to create a detection probability function (Thomas et al. 2010). Based on this information, inferences on abundance and density may be made (Miller et al. 2019). In its basic form, distance sampling is conducted using line transect surveys, but an adjustment to point transect set-ups is possible, in which case the CT functions as an observer. In this study, we use CT-DS to: (1) infer density of the invasive raccoon dog in lake shore areas in southern Finland where ground-nesting birds have experienced strong population declines, and (2) compare its density to similar native mesopredators, the red fox and the European badger. We used data from 2 years. In one of the years (2021), we had performed an experiment on chemical camouflage and conditioned food aversion as tools to limit predation of mesopredators on waterfowl nests (Selonen et al. 2022b). We here check whether this treatment affected density of mesopredators in the study areas. In comparison, we have data from a year (2020) without these treatments.
Material and methods
Study species
The invasive raccoon dog is an omnivorous canid mesopredator of approximately 50–65 cm head to body length and 7.5 kg body mass. They are monogamous and can produce litters of up to nine offspring each year, starting at the age of 1 year (Kauhala 1996). Their maximum life span in the wild is recorded at seven to 8 years. A study of their dietary composition based on morphological analysis of scats in Finland found a wide pallet ranging from voles and mice, birds and eggs to amphibians, invertebrates, and plants (Kauhala et al. 2006a).
The red fox weighs about 5–7 kg, with a head to body length of 50–90 cm. While also an opportunistic omnivore, the red fox tends to be more predatory than the raccoon dog, feeding more frequently on birds and small mammals, such as voles and hares (Dell’Arte et al. 2007). Reproduction in red foxes also starts at the age of 1 year, when they produce litters of on average five offspring. The European badger stand at 30 cm shoulder height and 56–81 cm head to body length and typically weighing 9–12 kg. Badgers usually have their first cubs in their second or third winter. They tend to live in social groups or clans and their diet is largely made up of amphibians and invertebrates, yet also includes birds and their eggs (Byrne et al. 2012).
Study area and data collection
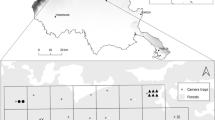
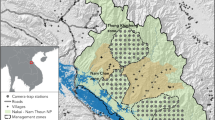
Data was collected at 11 and 16 wetlands in southern Finland during the springs of 2020 and 2021, respectively (Fig. 1). Seven study wetlands were included in both years while the rest were used only in 1 year. The nearest neighbour distance between the study wetlands was 14 ± 13 km (average ± SD, min 3 km, max 50 km inter-site distance) and the size of the wetlands, measured as distance of shore, was 3.0 ± 1.7 km. These wetlands were listed by the Helmi-programme of Finnish Ministry of the Environment as important waterbird breeding areas in the country. The wildlife cameras were set in shore forests and at the edges of agricultural fields and forests surrounding the wetland (10–200 m from the shore edge; the size of the study area is this buffer area surrounding each wetland). The shore forests in these areas are dominated by managed coniferous and mixed forests, with Norway spruce (Picea abies), scots pine (Pinus sylvestris), birches (Betula sp), aspen (Populus tremula), and alders (Alnus sp.) being the most abundant tree species. Agricultural areas covered about 30% of the landscape in the area. Human settlements are sparse in these areas, including mainly summer cottages. Wildlife cameras were not set close to human settlements and they were always placed with a permission from the landowner.
Location (grey stars) of the study wetlands and an example of placing of wildlife cameras (134 and 150 cameras in 2020 and 2021, respectively; 3–20 per site) to study densities of raccoon dogs, badgers and red foxes around wetlands in southern Finland. Coordinates for the wetlands can be seen in Supplement Table S21
A total of 134 and 150 camera traps (CT) were deployed for an average of 13 and 44 days between April and May in 2020 and 2021, respectively (175 CT in total across 20 wetlands, Supplement Table S1). The longer deployment period in 2021 was due to the experiment conducted that year. The CT set-up date varied across both years to ensure the survey began phenologically at the same time of spring in each wetland. To achieve this, the CT were set up a week or two earlier in the southwestern study wetlands, where spring arrives earlier compared to the northeastern parts of the study area (Fig. 1). The CT were set up approximately 200–300 m apart from each other, with the aim to encircle the chosen location. Depending on size of the area of waterbody, 3–20 CT were in use per wetland and they were placed to small forest openings or to edges of forests and fields to ensure camera view. Observations were gathered mostly in video and partially in pictures. All pictures and videos were split up into predetermined snapshot moments of 1 s. We used marked sticks at known distances to estimate distance from camera (see below). Sometimes animals responded to these sticks and the time that the individual was doing this was removed from the data. For raccoon dogs and red foxes these cases were rare, but badgers in a few sites often reacted to marker sticks by sniffing them.
We included 2 years of data, to have reference for the year 2021, the year the experiment was conducted in the study wetlands. This experiment investigated chemical camouflage (CC) and conditioned food aversion (CFA) as tools to limit predation of mesopredators on waterfowl nests (Selonen et al. 2022b). The experimental design consisted of six wetlands that received CC, six that received CFA, and six that were left as untreated control wetlands (see Supplement Table 3). Each treatment was applied to a specific wetland, and thus, all camera traps within that wetland were either CC, CFA or control in year 2021. The first treatment or control visits to the wetlands were made a week after the camera traps were set up in the study sites. Both treatments and control involved five separate visits to each wetland, with 5–7 days between visits (for more details, see Selonen et al. 2022b). In the CFA treatment, we added domestic mallard or goose eggs containing 100 mg of thiram to the study area. The thiram was injected into the eggs in the laboratory 1–2 days before taking them to the field. Thiram causes digestive discomfort (vomiting, nausea, and/or diarrhea) without other severe adverse health effects. The idea was that predators eating these eggs would learn to avoid eggs of the birds nesting in the wetland. In the CC treatment, we spread a mixture of selected waterfowl odour products in the areas (Selonen et al. 2022b). The odour was placed on the ground every 10 m in four directions from each wildlife camera, up to 150 m from the camera. The goal here was that predators would become desensitized or confused by the abundance of bird odour without a food reward and learn to associate the bird odours in the wetland as not indicating food. We test the potential effect of these treatments on the density estimates obtained in this study. This is done with a linear model (see below).
Distance sampling
Distance sampling method relaxes the assumption that all individuals within a survey plot are certainly detected. The underlying assumption is that the probability of detection of an object of interest will decrease the greater the distance from the observer becomes (Thomas et al. 2010). In the case of CT, the observer is assumed to be stationary (point transect) and surveys a small area in front of the CT. Animals in this area are recorded at a set of predetermined snapshot moments, separated by the time interval t. This is done to ensure that the distribution of detections is not biased by animal movement (Pal et al. 2021). Time interval t is chosen based on the movement speed of the species, and is here set at one second.
The radial distance of a passing animal to the camera trap functioning as an observer was estimated in reference to markers placed in the field of view for each snapshot moment recorded. Markers were mostly wooden sticks highlighted with tape, or tape bound to a tree branch, marking 3, 6, 9, and 12 m from the camera. Estimates of distance of the animal’s midpoint to the observer were rounded to the full meter. We counted individual animals separately, although raccoon dogs and badgers sometimes move as a pair (male and female). Mostly we however observed solitary animals, perhaps due to timing of the study in spring when pair members are likely to move solitarily if one is at the den (Kauhala et al. 1998).
The observer effort was computed for each camera based on the number of days the camera was operational. Not all cameras were functioning for the full duration. Effort was expressed in the number of seconds. Three types of camera traps were in use: UOVision 595 and UOVision UM785 with a view angle of 52°, as well as UOVision Compact 4GLTE with a field of view of 64°.
Analysis followed the approach of Howe et al. (2017) as detailed by Howe and Rexstad (2022). Briefly, a detection function is fitted to the distance data using the function ds in the R package distance (Miller et al. 2019). The detection in CT may decline at large distances and be lowered very close to camera (Howe et al. 2017). After visual inspections of the distance data, we truncated data such that no observations below 1 m and above 12 m were used because detection was overall lowered above this limit in all species. The detection function describes the underlying relationship between the distance and the probability of detection. As the decline of detection with distance is unknown, the aim is to find a function that most parsimoniously describes this decline. There are several possible formulations for the detection function. Here we considered the uniform, half-normal and hazard-rate key function. Adjustments terms (cosine for uniform and half-normal and polynomial for hazard rate) were included to increase flexibility and achieve the best fit to the data (Laake et al. 1996). We selected for each key function (uniform, half-normal, hazard rate) the most parsimonious formulation in terms of adjustment using QAIC, but omit key functions with adjustments of such magnitude that detection declined non-monotonically with distance. We then in a second step selected across the set of most parsimoneous key functions the one with lowest c-hat (Howe and Rexstad 2022). This procedure was followed to select the detection function for each of the three species for each of the two study years.
Density estimates
We used the above selected detection function to infer density using the dht2 function in the R package distance (Miller et al. 2019). Truncation distances were as described above. Animals may not be active, and hence available for detection, during the entire day. We used the time stamps of the camera detections to model the daily availability and its uncertainty (standard error) for each species in each study year using the R package activity (Rowcliffe 2022) and took both availability and its uncertainty into account for inferring density. We further took the restricted view of the camera (either 52 or 64 degrees, as described above) into account as the sample fraction. The different study sites were modelled as separate geographical strata allowing to obtain study wetland specific density estimates as well as overall density across all study sites combined. The uncertainty of the density estimate was computed using the design-based approach (method “P2” of Fewster et al. 2009).
Finally, we tested whether the above described treatments, chemical camouflage and conditioned food aversion, affected density estimates in 2021. We built a loglinear model (SAS, Glimmix 9.3) where the density estimates in 2021, were explained with two class variables, the treatment (CC = chemical camouflage, CFA = conditioned food aversion, and C = control) and mesopredator species (raccoon dog, red fox, and badger). Cases that CTs in the wetland did not produce observations of a species were included as zero density in the model. Unfortunately, the R package distance does not allow multiple strata and we could not model the treatment, along with the site, using that approach.
Results
Data description
Raccoon dogs were in both years detected in all study wetlands, red foxes in all but one wetland, while badgers remained undetected in several study wetlands (Table 1). The number of detections was much larger for raccoon dogs compared to the other species (Table 1). However, there were also clear differences in availability, as the red fox showed more diurnal activity and hence had a greater availability for detection compared to badger and raccoon dog (Supplementary Fig. S1).
Detection function and density estimates
We selected the most parsimonious function to describe the decline in detection with distance from the CT for each species and year (Supplementary Table S2, Fig. S2). The inferred estimates of overall density were larger for raccoon dogs than for badgers and for red foxes in both years (Fig. 2; on average for 2 years; raccoon dog: 3.7 ind./km2; red fox: 0.6 ind./km2; badger: 1.2 ind./km2). For study wetlands where all the three species were detected together, the wetland-specific point estimates for density of the raccoon dog were higher than those for badger and red fox (Supplementary Table S3—2020: 8/8 sites; 2021: 10/12 sites, sign tests: p < 0.001).
Density in individuals per km2 of the badger (in black), the raccoon dog (dark grey) and the red fox (light grey) with their 95% confidence interval (lines) for the two study years. See Table S3 for the overall density estimates plotted here, and note that estimates for 2021 may be overestimates (see main text and Table S3)
Densities seem overall higher in 2021 than 2020 (Fig. 2), as there tended to be an increase in the densities of the year 2021 due to the treatments described in methods (estimates: control = − 1.1 ± 0.6, CC = 0.1 ± 0.6, CFA = 0: Fdf = 2.82,43, p = 0.07). The predicted density estimates from this model for the year 2021 were for raccoon dog 3.3 ind./km2, for badger 0.30 ind./km2, and for red fox 0.91 ind./km2 (difference between species Fdf = 11.452,43, p = 0.0001; compare to year 2021 in Fig. 2).
Discussion
We used camera trap distance sampling (CT-DS) to infer densities of the invasive raccoon dog and two native mesopredators, the badger and the red fox, in wetland areas in southern Finland. We found that only raccoon dogs were detected in all wetlands studied, had an overall higher density than the other two species, and the highest local density in sites where all species co-occur. Our findings therefore provide quantitative underpinning for the assertion that the raccoon dog is a highly successful mesopredator. Given its high density and overall presence, the raccoon dog likely exerts competition over shared food resources with the native mesopredator species, the red fox and the badger, and creates additional pressures for the prey populations.
Density estimates
Our overall estimates of raccoon dog densities (Fig. 2) were higher compared to previous estimates from the literature, where density estimates range between 0.8 and 2.2 ind./km2 in Finland (Kauhala et al. 2010). It is possible that raccoon dog density has increased in Finland since the study of Kauhala et al. (2010). However, these earlier estimates are based on extrapolation of home range sizes inferred on the basis of a limited number of animals over larger areas and not on direct density estimation of individuals. Furthermore, we here quantified densities in the surroundings of wetlands or lakes, which present limited range of habitat types present in Finland and can have higher densities due to the wetland. While raccoon dogs can adjust to a wide range of conditions, Kauhala et al. (2010) found that the population density was highest with up to two ind./km2, when there was a small-scaled landscape mosaic of meadows and fields, small mixed forests, or gardens. The landscape near our CT ranged from field edges and pastures to wetlands, flood meadows, and forests. Those habitat types provide a variety of food resources, as well as undergrowth for shelter preferred by the raccoon dogs (Kauhala and Auttila 2010).
Overall the density estimates seemed to be higher in 2021 than 2020. The difference between study years was that in 2021 we conducted an experiment investigating chemical camouflage (CC) and conditioned food aversion (CFA) within study wetlands (Selonen et al. 2022b). While this treatment had only a marginally significant effect on mesopredator density, it suggested a trend towards increasing density. This suggests that, in studies using CC or CFA to limit predation, at least a short-term increase in predator activity might be expected. However, this is acceptable if the targeted prey remains protected. This was the case in Selonen et al. (2022b) as in particular CC effectively safeguarded artificial nests. Apparently, the predators became desensitized to odour cues leading to nests (Selonen et al. 2022b). Nevertheless, the reliable density estimates for the study species may be closer to those of year 2020 than 2021 in our study. Another difference between the years was that the study period was notably shorter in 2020 (13 days) than 2021 (44 days). This does not affect the density estimation if the detection probability does not change in time, but there might be some changes. Particularly red foxes are quite wary and if they detected the camera, they may have in the beginning of the camera trapping period avoided approaching it. This may have decreased the density estimate for red foxes in 2020 when the CT period was short.
We were able to create density estimates for all three species of interest with distance sampling on a rather small spatial and temporal scale. In comparison to other methods for creating density estimates without individual recognition, like Random Encounter Model (Jones et al. 2015), distance sampling does not require estimates of animal’s movement speed or angle to the camera, which can be difficult to accurately assess. The caveat of the distance sampling, like other CT-based density estimates, is that the placement of CTs in the landscape is rarely entirely random. This leads to estimates skewed towards habitats where the CTs are located, in our case, shore forests of wetlands. However, in scenarios where monitoring predator populations is crucial, distance sampling offers a valuable tool. It provides a quick framework for long-term monitoring of population trends, requiring relatively low effort and maintenance. Once a camera trap set-up is established, the data-collection can be periodically re-run with little additional effort. The continuous expansion of this methodology and the potential development of the raccoon dog’s dynamics in the community of native mesopredators in the coming years would make this an intriguing long-term monitoring project.
Comparison to native species
Raccoon dogs were found to have the highest density of the three mesopredators, not only in the overall estimates of all detections combined, but also in all except two study locations. It is notable that these two study locations were within a wolf territory, a factor that has been earlier found to decrease raccoon dog observations more than those of red foxes and badgers (Selonen et al. 2022a). Unfortunately, wolf occurrence was too scarce in the current data to be included in the analysis. Nevertheless, our results clearly demonstrate that the invasive mesopredator has successfully reached a population density higher than that of native mesopredators in southern Finnish lakeshore areas and habitats surrounding wetlands. The same was found to be the case in Poland in Bialowieza, where the raccoon dog is observed to be more common than native mesopredators with reported densities of 0.7 ind./km2 for raccoon dogs, 0.2–0.5 ind./km2 for red foxes and 0.2 ind./km2 for badgers (Jędrzejewska and Jędrzejewski 1998; Kowalczyk et al. 2003). Instead, Goszczyński (1999) reported similar spring densities in North Eastern Poland for red foxes (0.27 ind./km2), badgers (0.36 ind./km2) and raccoon dogs (0.37 ind./km2). These estimates are, however, based on methods such as track censuses, den site surveys and extrapolation of home range sizes.
Red foxes reached lower overall estimate than badgers (Fig. 2). However, the density of badgers appeared more variable than that of other study species, as badger detections were grouped at few locations, and several had no sighting at all. Despite this, forests near wetlands may be more favored habitat for badgers than for red foxes in Finland, as badgers prefer spruce forests with rich soil type (Kauhala and Kowalczky 2012). In contrast, a study investigating space use and interaction between red foxes and raccoon dogs in agricultural landscapes of northeast Germany concluded that red foxes displayed little preference or otherwise avoidance of specific types of habitats (Drygala and Zoller 2013). Previous estimates of red fox density in Finland based on home range size and snow track counts amounted to 0.35 and 0.44 ind./km2 (Kauhala et al. 2006b), which is only slightly lower to the overall density of 0.6 ind./km2 (average for 2 years) found via distance sampling in the current study. However, comparing these values is complicated by differences in methods, locations and possible population trends in time.
The badger densities in Finland do not appear to have declined during the time that raccoon dog has invaded the country (Kauhala 1995). Instead, it has been hypothesized that clear felling of forests as a frequent form of forest management in Finland benefits the raccoon dog while negatively impacting the native badger (Kauhala and Kowalczyk 2011). The presence of badgers may even be a contributing factor to the raccoon dog density. Raccoon dogs, like red foxes, use dens made by badgers to hibernate and raise their young (Kowalczyk et al. 2008). It is hypothesized that use of badger dens as a refuge from cold temperatures enlarges the realized niche and aids the spread of raccoon dogs (Kowalczyk et al. 2008), but in Finland this likely is not the case because raccoon dogs have spread much further north than badgers.
Introducing a new predator to an environment increases predation pressure, posing an additional threat to prey populations. Our results suggest that the invasive raccoon dog may have a greater impact on prey species in wetland habitats compared to native mesopredators due to their higher densities. Many wetland-associated bird species show declining trends (McMahon et al. 2020; Pöysä and Linkola 2021). The role of raccoon dog in possible declines of these avian species is debated (Kauhala and Kowalczyk 2011), but clearly the threat in the form of nest predation does exist (Dahl and Åhlén 2019). In addition to wetland birds, amphibians are also vulnerable to raccoon dog predation, as frogs have a major role in their spring diet (Kauhala and Kowalczyk 2011). Thus, the high densities of raccoon dogs do raise concerns related to wetland conservation.
Conclusion
Based on the methodology of camera trap distance sampling, in our data the invasive raccoon dog reached higher densities than the ecologically comparable native mesopredators, the red fox and the European badger. This indicates that this invasive canid has since its introduction surpassed the abundance of both native mesopredators in lakeshore regions of southern Finland. Thus, in areas close to important breeding areas of many declining waterbird species the invasive species could currently be a bigger threat to nesting waterfowl than the two native mesopredators. Further studies on the foraging and predatory behaviors of these species would however give a more accurate picture of the pressures they pose to the prey populations.
Data availability
The datasets analysed during the current study together with the R script for the analyses can be obtained from https://doi.org/10.5281/zenodo.11108107.
References
Balčiauskas L, Startford J, Balčiauskienė L, Kučas A (2021) Roadkills as a method to monitor raccoon dog populations. Animals 11:3147. https://doi.org/10.3390/ani11113147
Byrne A, O’Keeffe J, Sleeman D, Davenport J, Martin S (2012) The ecology of the European badger (Meles meles) in Ireland—a review. Biol Environ Proc Roy Irish Acad 112:115–132
CREEM (2022) DistanceSampling.org-Online distance sampling course. (C. f. Modelling, Editor) Retrieved March 2022, from University of St Andrews: https://workshops.distancesampling.org/online-course/syllabus/Chapter1/
Dahl F, Åhlén N (2019) Nest predation by raccoon dog Nyctereutes procyonoides in the archipelago of northern Sweden. Biol Invasions 21:743–755. https://doi.org/10.1007/s10530-018-1855-4
Dell’Arte GA, Laaksonen T, Norrdahl K, Korpimäki E (2007) Variation in the diet composition of a generalist predator, the red fox, in relation to season and density of main prey. Acta Oecologica 31:276–281
Directorate-General for Environment (2022) European Commision-Nature and Biodiversity. Retrieved March 2022, from Invasive Alien Species: https://ec.europa.eu/environment/nature/invasivealien
Drygala F, Zoller H (2013) Spatial use and interaction of the invasive raccoon dog and the native red fox in Central Europe: competition or coexistence? Eur J Wildl Res 59:683–691. https://doi.org/10.1007/s10344-013-0722-y
Drygala F, Werner U, Zoller H (2013) Diet composition of the invasive raccoon dog (Nyctereutes procyonoides) and the native red fox (Vulpes vulpes) in north-east Germany. Hystrix 24:190–194. https://doi.org/10.4404/hystrix-24.2-8867
Fewster RM, Buckland ST, Burnham KP, Borchers DL, Jupp PE, Laake JL, Thomas L (2009) Estimating the encounter rate variance in distance sampling. Biometrics 65:225–236
Goszczyński J (1999) Fox, raccoon dog and badger densities in North Eastern Poland. Acta Theriol 44:413–420
Holopainen S, Väänänen V-M, Vehkaoja M, Fox A (2021) Do alien predators pose a particular risk to duck nests in Northern Europe? Results from an artificial nest experiment. Biol Invasion 23:3795–3807. https://doi.org/10.1007/s10530-021-02608-2
Howe E, Buckland S, Després-Einspenner M, Kühl H (2017) Distance sampling with camera traps. Methods Ecol Evol 8:1558–1565. https://doi.org/10.1111/2041-210X.12790
Howe E, Rexstad E (2022) Analysis of camera trapping data. Example analysis with Ivory Coast Maxwell’s duiker. http://examples.distancesampling.org/Distance-cameratraps/camera-distill.html#ref-howeetal (version dated Nov 3, 2022; Accessed 20 April 2023)
IUCN (2016) IUCN Red List. Retrieved from https://www.canids.org/species/view/PREKLY462191
Jędrzejewska B, Jędrzejewski W (1998) Predation invertebrate communities: the Białowieża Primeval Forestas a case study. Ecological Studies, vol 135. Springer, Berlin
Jones K, Freeman R, Rowcliffe M, Lucas T, Moorcroft E (2015) A generalised random encounter model for estimating animal density with remote sensor data. Methods Ecol Evol 6:500–509. https://doi.org/10.1111/2041-210X.12346
Kauhala K (1995) Changes in distribution of the European badger Meles meles in Finland during the rapid colonization of the raccoon dog. Ann Zool Fenn 23:183–191
Kauhala K (1996) Reproductive strategies of the raccoon dog and the red fox in Finland. Acta Theriol 41:51–58. https://doi.org/10.4098/AT.arch.96-4
Kauhala K, Auttila M (2010) Habitat preferences of the native badger and the invasive raccoon dog in southern Finland. Acta Theriol 55:231–240. https://doi.org/10.4098/j.at.0001-7051.040.2009
Kauhala K, Kowalczky R (2012) The Raccoon dog (Nyctereutes procyonides) in the community of medium-sized carnivores in Europe: its adaptations, impact on native fauna and management of the population. In: Kauhala K, Kowalczyk R (eds) Species, conservation and management. Nova Publishers
Kauhala K, Kowalczyk R (2011) Invasion of the raccoon dog Nyctereutes procyonoides in Europe: history of colonization, features behind its success, and threats to native fauna. Curr Zool 57:584–598. https://doi.org/10.1093/czoolo/57.5.584
Kauhala K, Helle E, Pietilä H (1998) Time allocation of male and female raccoon dogs to pup rearing at the den. Acta Theriol 43:301–310. https://doi.org/10.1016/j.mambio.2006.10.006
Kauhala K, Holmala K, Lammers W, Schregel J (2006a) Home ranges and densities of medium-sized carnivores in south-east Finland, with special reference to rabies spread. Acta Theriol 51:1–13. https://doi.org/10.1007/BF03192650
Kauhala K, Laukkanen P, von Rége I (2006b) Summer food composition and food niche overlap of the raccoon dog, red fox and badger in Finland. Ecography 21:457–463. https://doi.org/10.1111/j.1600-0587.1998.tb00436.x
Kauhala K, Schregel J, Auttila M (2010) Habitat impact on raccoon dog Nyctereutes procyonoides home range size in southern Finland. Acta Theriol 55:371–380. https://doi.org/10.1007/BF03193240
Kowalczyk R, Zalewski A, Jędrzejewska B, Jędrzejewski W (2003) Spatial organization and demography of badgers Meles meles in Białowieża Forest (Poland) and the influence of earthworms on badger densities in Europe. Can J Zool 81:74–87
Kowalczyk R, Jędrzejewska B, Zalewski A, Jedrzejewski W (2008) Facilitative interactions between the Eurasian badger (Meles meles), the red fox (Vulpes vulpes) and the invasive raccoon dog (Nyctereutes procyonoides) in Białowiez˙a Primeval Forest, Poland. Can J Zool 86:1389–1396. https://doi.org/10.1139/Z08-127
Laake J, Buckland S, Anderson D, Brunham K (1996) Distance User’s Guide-Version 2.2. Retrieved April 2021, from distancesampling.org: http://distancesampling.org/Distance/old-versions/downloads/guide.pdf
McMahon BJ, Doyle S, Gray A, Kelly S, Redpath SM (2020) European bird declines: do we need to rethink approaches to the management of abundant generalist predators? J Appl Ecol 57:1885–1890
Melis C, Nordgård H, Herfindal I, Kauhala K, Åhlen P-A, Strann KB, Andersen R (2007) Raccoondogs in Norway—potential expansion rate, distribution area and management implications. NTNU Vitenskapsmuseet Rapp Zool Ser 3:1–49
Miller D, Rexstad E, Thomas L, Marshall L, Laake J (2019) Distance sampling in R. J Stat Softw. https://doi.org/10.18637/jss.v089.i01
Pal R, Bhattacharya T, Qureshi Q, Buckland S, Sathyakumar S (2021) Using distance sampling with camera traps to estimate the density of group-living and solitary mountain ungulates. Oryx 55:668–676. https://doi.org/10.1017/S003060532000071X
Podgórski T, Acevedo P, Apollonio M, Berezowska-Cnota T, Bevilacqua C et al (2020) Guidance on estimation of abundance and density of wild carnivore population: methods, challenges, possibilities. EFSA Support Publ. https://doi.org/10.2903/sp.efsa.2020.EN-1947,17,11
Pöysä H, Linkola P (2021) Extending temporal baseline increases understanding of biodiversity change in European boreal waterbird communities. Biol Conserv 257:109139
Rowcliffe M (2022) Activity: animal activity statistics. R package version 1.3.2. https://CRAN.R-project.org/package=activity
Sadlier LMJ, Webbon CC, Baker PJ, Harris S (2004) Methods of monitoring red foxes Vulpes vulpes and badgers Meles meles: are field signs the answer? Mammal Rev 34:75–98
Santini G, Abolaffio M, Ossi F, Franzetti B, Cagnacci F, Focardi S (2022) Population assessment without individual identification using camera-traps: a comparison of four methods. Basic Appl Ecol 61:68–81. https://doi.org/10.1016/j.baae.2022.03.007
Selonen V, Banks P, Tobajas J, Laaksonen T (2022b) Protecting prey by deceiving predators: a field experiment testing chemical camouflage and conditioned food aversion. Biol Cons 275:109749. https://doi.org/10.1016/j.biocon.2022.109749
Selonen V, Brommer J, Holopainen S, Kauhala K, Krüger H, Poutanen J, Väänänen V-M, Laaksonen T (2022a) Invasive species control with apex predators: increasing presence of wolves is associated with reduced occurrence of the alien raccoon dog. Biol Invasions 24:3461–3474. https://doi.org/10.1007/s10530-022-02850-2
Thomas L, Buchland S, Rexstad E, Laake J, Strindberg S et al (2010) Distance software: design and analysis of distance sampling surveys for estimating population size. J Appl Ecol 47:5–14. https://doi.org/10.1111/j.1365-2664.2009.01737.x
Waern O, Glover-Kapfer P (2019) Snap happy: camera traps are an effective sampling tool when compared with alternative methods. Roy Soc Open Sci 6:3. https://doi.org/10.1098/rsos.181748
Acknowledgements
We thank Mikaela Honkala, Tuija Koivisto, Kaisa Merimaa, Jorma Nurmi, Jenni Poutanen, Elina Tuomikoski and Linda Uusihakala participating in the data collection, and the landowners who gave the permissions to place wildlife cameras on their property.
Funding
Open Access funding provided by University of Turku (including Turku University Central Hospital). This work was supported by grants by Finnish Ministry of Agriculture and Forestry (Sotka-project) to Toni Laaksonen and Vesa Selonen and Research Council of Finland (project no. 357199) to Vesa Selonen.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics approval
Ethics approval was not required for this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Selonen, V., Brommer, J.E., Klangwald, C. et al. Successful invasion: camera trap distance sampling reveals higher density for invasive raccoon dog compared to native mesopredators. Biol Invasions 26, 1–10 (2024). https://doi.org/10.1007/s10530-024-03323-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-024-03323-4