Abstract
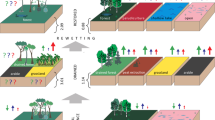
Tropical peat swamp degradation can modify net peat greenhouse gas (GHG) emissions even without drainage. However, current Intergovernmental Panel on Climate Change (IPCC) guidelines do not provide default emission factors (EF) for anthropogenically-degraded undrained organic soils. We reviewed published field measurements of peat GHG fluxes in undrained undegraded and degraded peat swamp forests in Southeast Asia (SEA) and Latin America and the Caribbean (LAC). Degradation without drainage shifted the peat from a net CO2 sink to a source in both SEA (− 2.9 ± 1.8 to 4.1 ± 2.0 Mg CO2–C ha−1 yr−1) and LAC (− 4.3 ± 1.8 to 1.4 ± 2.2 Mg CO2–C ha−1 yr−1). It raised peat CH4 emissions (kg C ha−1 yr−1) in SEA (22.1 ± 13.6 to 32.7 ± 7.8) but decreased them in LAC (218.3 ± 54.2 to 165.0 ± 4.5). Degradation increased peat N2O emissions (kg N ha−1 yr−1) in SEA forests (0.9 ± 0.5 to 4.8 ± 2.3) (limited N2O data). It shifted peat from a net GHG sink to a source in SEA (− 7.9 ± 6.9 to 20.7 ± 7.4 Mg CO2-equivalent ha−1 yr−1) and increased peat GHG emissions in LAC (9.8 ± 9.0 to 24.3 ± 8.2 Mg CO2-equivalent ha−1 yr−1). The large observed increase in net peat GHG emissions in undrained degraded forests compared to undegraded conditions calls for their inclusion as a new class in the IPCC guidelines. As current default IPCC EF for tropical organic soils are based only on data collected in SEA ombrotrophic peatlands, expanded geographic representation and refinement of peat GHG EF by nutrient status are also needed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Tropical peat swamp forests play an important role in regulating atmospheric concentrations of greenhouse gases (GHG). They are characterized by permanently or seasonally saturated soils and high litter inputs that exceed rates of organic matter decomposition, leading to substantial soil carbon and nitrogen storage. Drainage and conversion of tropical peat swamp forests to other uses are known to increase net GHG emissions from peat soils (Hergoualc’h and Verchot 2014; Inubushi et al. 2003; Swails et al. 2021). Anthropogenic activities that degrade peat swamp forests such as harvest (e.g., Basuki et al. 2021; Hergoualc’h et al. 2023), grazing (Eusse and Aide 1999), crop cultivation under the canopy (Swails et al. 2021) and fire (Astiani et al. 2018) without deforestation and conversion and regardless of drainage could also potentially exacerbate peat GHG emissions. The types of anthropogenic disturbances in undrained tropical peatlands and their associated impacts on peat GHG emissions are not well characterized, although some studies found substantial differences in GHG emissions between undegraded and degraded sites (Hergoualc’h et al. 2023; Swails et al. 2021; Sánchez et al. 2017).
Drainage of peatlands increases oxygen availability in the soil, which accelerates CO2 emissions and decreases CH4 fluxes from peat decomposition (Hergoualc’h and Verchot 2012; Hergoualc’h et al. 2017b; Itoh et al. 2017) while tending to increase N2O emissions (Pärn et al. 2018). With or without drainage, vegetation disturbance alters C inputs rates, directly impacting peat onsite CO2 emissions (difference in C inputs from litterfall and root mortality and C outputs from heterotrophic respiration; Drösler et al. 2014; Hergoualc’h and Verchot 2014). In addition changes to litter input quantity and quality can affect nutrient content of substrate for microbial decomposition (Allison and Vitousek 2004; Hobbie 2015; Lugo et al. 1990), potentially influencing heterotrophic respiration (Jauhiainen et al. 2016; Swails et al. 2018) as well as soil CH4 and N2O emissions (Hergoualc’h and Verchot 2014). Forest disturbance also modifies its microclimate (Blonder et al. 2018; Both et al. 2017; Marsh et al. 2022), which can affect biogeochemical cycling rates for example, reducing canopy cover increases air temperature potentially enhancing organic matter mineralization (Hoyt et al. 2019), however links between microclimate alteration and decomposition rates in tropical forests are unclear (Both et al. 2017). Tropical peat soils impacted by fire undergo extreme changes, including enrichment with cations (Ca, Mg, Mn, Fe, Na, Zn) (Könönen et al. 2015), which may enhance aerobic decomposition of soil organic matter over the short-term (Astiani et al. 2018; Lupascu et al. 2020). Ash remaining on site after fires raises soil pH, accelerating N loss (Certini 2005). Fire also increases the hydrophobicity of soil organic matter leading to additional nutrient losses due to erosion (Certini 2005). Nutrient content and microbial community in restored tropical peatlands indicate that these ecosystems are still influenced by historical land-use and management for years following restoration (Nurulita et al. 2016). Peat GHG emissions may be elevated in secondary peat swamp forests decades after initial disturbance (Swails et al. 2021).
Countries report their GHG emissions to the UNFCCC (United Nations Framework Convention on Climate Change) using guidelines developed by the Intergovernmental Panel on Climate Change (IPCC). The 2013 Wetland supplement provides in Chap. 2 (Drösler et al. 2014), default Tier 1 emission factors (EF) for drained forest on peat to quantify emissions of CO2 onsite, N2O emissions from peat decomposition, and peat CH4 emissions. Current IPCC guidelines do not provide methodological guidance for anthropogenically degraded organic soils that are undrained, which has implications for national GHG accounting in peat-rich countries where significant extents of undrained peatlands are degraded. For example, in Indonesia, forests subject to selective logging, shifting cultivation, fire, fuelwood collection, and livestock grazing account for 59% of total peat swamp forest area (Indonesian Ministry of Environment & Forestry 2021) with a substantial proportion unaffected by drainage (Dadap et al. 2021). Therefore, EF are needed to estimate peat emissions in undrained forests that are degraded by anthropogenic disturbance. Additionally, the potential influence on GHG fluxes of peat nutrient status (ombrotrophic vs. minerotrophic) (Drewer et al. 2010; Saarnio et al. 2007) and variation in climate (Ribeiro et al. 2021), forest vegetation (Wang et al. 2015), and management practices (Lilleskov et al. 2019) in the tropics calls for refinement of peat GHG EF by nutrient status and expanded geographic representation of the current IPCC default Tier 1 EF based only on data collected in SEA ombrotrophic peatlands.
In this paper we investigated peat GHG fluxes in undrained undegraded and degraded tropical peat swamp forests using observational data reported in the literature. We present peat CH4, N2O and onsite CO2 EF for undegraded and degraded forests in Southeast Asia (SEA) and Latin America and the Caribbean (LAC) and explore environmental variables controlling peat GHG fluxes. We address the following questions: (1) How do tropical peat EF (onsite CO2, CH4, N2O) and environmental variables differ between forest conditions (undegraded vs. degraded) and regions (SEA vs. LAC)? (2) Which environmental variables control peat GHG fluxes in these ecosystems? (3) How do the magnitudes of net peat GHG budgets and relative contributions of each EF (onsite CO2, CH4, N2O) vary according to forest conditions and regions? (4) What are research needs for further refinement of EF for undrained anthropogenically degraded tropical peat forests?
Methods
Data collection, calculation, and presentation
We compiled a database from international peer-reviewed publications (Table 1) of soil respiration and peat CH4 and N2O fluxes and controlling environmental variables in undrained peat swamp forests that were undegraded (UF) or degraded (UFDeg). The locations of study sites are displayed in Fig. 1. Undrained undegraded and degraded forests were identified based on study site descriptions provided in the publications included in our synthesis. We assumed that degraded sites were undrained if drainage or altered hydrology were not mentioned in the site description. Degraded sites included actively and previously logged forests as well as secondary forest regrowth following partial clearing for cultivation of crops under a reduced canopy (Table S1). Degraded palm swamp forests in Peru were subject to felling of Mauritia flexuosa palms for fruit collection, in addition to timber harvest (Hergoualc’h et al. 2020, 2023). All degraded sites were considered to be in an active or recovery stage of degradation as they had been monitored less than 50 years after anthropogenic disturbance. Indeed the time for degraded tropical forests to recover biomass and soil carbon stocks to the level of undisturbed forests is around a century (Martin et al. 2013).
Study locations (green circles) and peatland extent (black areas, Gumbricht et al. 2017) in Southeast Asia (n = 17) (a) and Latin America and the Caribbean (n = 4) (b). Study locations in Thailand and Micronesia are not shown (n = 2). There were no data for African peat swamp forests
Our analysis included only data collected using an experimental design which adequately covers spatiotemporal variability of GHG fluxes. Studies with a minimum of three spatial replicates per site, monitored at a minimum frequency of every two months over a period of at least one year were considered to meet this requirement. Whenever the fluxes were reported per unit hour or day, these were extrapolated to a full 365-day year. We considered each site as a replicate and computed a single mean annual value for multi-year studies.
To explore drivers of variation in soil GHG fluxes in undrained peat swamp forests, we investigated relationships among total soil respiration, peat CH4 and N2O fluxes and concurrently measured controlling environmental variables (rainfall, air and soil temperature, water table level, soil pH, C:N ratio, cation exchange capacity, base saturation, mineral N content, and peat minerotrophy or ombrotrophy status as defined by its Ca:Mg ratio, Lahteenoja et al. 2009). We classified peat with Ca:Mg ratio exceeding that of rainwater i.e. 6 (Berner and Berner 1996 in Weiss et al. 2002) as minerotrophic as this indicates minerotrophic Ca input from incoming surface or ground water in addition to atmospheric deposition (Weiss et al. 1997, 2002; Muller et al. 2006).
Following the IPCC guidelines (Drösler et al. 2014), onsite peat CO2 emission or uptake was calculated as the difference in C outputs from soil heterotrophic respiration and C inputs from litterfall and root mortality. We excluded offsite CO2 emissions via waterborne C losses as these are minimal in undrained peat swamp forests (Moore et al. 2013). Root exudates represent an important but uncertain onsite C input to soil (Jones et al. 2004), and they are excluded from peat onsite CO2 budgets by the IPCC. Following Hergoualc’h and Verchot (2011), litterfall observations restricted to fine litter [all apart from the studies by Hergoualc’h et al. (2023) and Ong et al. (2015)] were corrected to include large-branch fall as 30% of total C inputs (from litterfall, root production, and large branch-fall), as observed by Chimner and Ewel (2005). Annual root mortality was assumed to equal annual root production in some instances (Brady 1997; Chimner and Ewel 2004; Shimamura and Momose 2005), which is reasonable for understanding soil C dynamics over short timescales (Hertel et al. 2009). In the case of studies that did not measure litterfall and/or root mortality, we applied the average rates calculated for undrained undegraded and degraded forests in SEA and LAC. Among field studies measuring heterotrophic respiration with the trenching method, we considered only experimental designs with trenches dug to a depth at which no coarse roots are observed (approximately 1 m deep, Hergoualc’h et al. 2017b). For studies that measured only total soil respiration, heterotrophic respiration was calculated using the average partitioning ratios for undrained undegraded and degraded forests in SEA and LAC (Table S2).
All CH4 emission data were from chamber-based measurements except the studies of Deshmukh et al. (2021), Griffis et al. (2020), and Sakabe et al. (2018) where fluxes were measured by eddy covariance. The fluxes measured by Deshmukh et al. (2021) were reduced by 10% to exclude tree-mediated CH4 emissions (Sjögersten et al. 2020). Emissions of CH4 by Griffis et al. (2020) and Sakabe et al. (2018) were similar to soil chambers CH4 fluxes measured at the same locations by Hergoualc’h et al. (2020) and Hirano et al. (2009), respectively, indicating a minor contribution of trees to ecosystem-scale CH4 flux.
We calculated net onsite peat GHG budgets as the balance of onsite peat CO2 emission or uptake rate and the N2O and CH4 emission rates. The annual CH4 and N2O fluxes were converted to CO2-equivalent considering global warming potentials (GWP) with climate-carbon feedbacks over a 20-year time horizon (86 kg CO2-equivalent kg−1 CH4 for CH4 and 268 kg CO2-equivalent kg−1 N2O for N2O) and over a 100-year time horizon (34 kg CO2-equivalent kg−1 CH4 for CH4 and 298 kg CO2-equivalent kg−1 N2O for N2O)(Myhre et al. 2013). Though a 100-year time horizon is the convention for national GHG inventories, a 20-year horizon is more appropriate for evaluating impacts of forest degradation that typically occur over 20 to 30 years in the tropics. Additionally, a 20-year time horizon is more closely aligned with the urgent need to reduce emissions to meet temperature goals under the Paris Agreement (Abernethy and Jackson 2022). We calculated the relative contribution of each gas to net peat GHG budgets considering the absolute values of emissions and uptakes in CO2-equivalents.
Statistics
Statistical analysis was performed using R (V4.0.5) software with a probability threshold of 0.05 to determine significance. Uncertainties are reported as standard errors. The normality of residuals distribution for total soil respiration, peat CH4 and N2O fluxes, and controlling environmental variables was tested using the Shapiro–Wilk test. GHG fluxes and environmental variables were compared between regions (SEA vs. LAC) and between forest conditions (undegraded vs. degraded) in SEA by the t test or Mann–Whitney U test, depending on the normality of residuals distribution and availability of sufficient data points for a statistical test. No test was performed to compare forest condition in LAC due to the limited number of sites. Differences in annual precipitation between SEA and LAC were evaluated by comparing mean values at sites across the two regions regardless of forest condition. We assessed differences in water table seasonality between the two regions and between forest conditions by comparing the average difference in mean water table levels during the three shallowest and the three deepest consecutive months. Seasonality was determined per site where monthly water table level was available (data from Busman et al. 2023; Melling et al. 2005a, b, 2007; Swails et al. 2021, 2023 for SEA, and Hergoualc’h et al. 2020, 2023; Wright et al. 2013 for LAC) and the site-level seasonality values were averaged to calculate a mean value across forest conditions in each region and per condition across regions for comparison. Relationships between annual GHG fluxes and environmental variables were developed applying linear and curvilinear univariate regression to site-level values, and to mean forest condition values in each region (n = 4). For significant relationships, we selected the regression form that explained the most variation in the independent variable. We considered each site as a replicate to calculate the standard error of fluxes per forest condition and region. Gaussian error propagation was used for propagating uncertainties in calculations of peat CO2–C and GHG budgets (Lo 2005).
Results
Peat GHG fluxes and environmental variables
Mean peat GHG fluxes and environmental variables are presented per region and forest condition in Table 2. Average annual soil respiration tended to be greater in degraded than undegraded forests for both regions, and tended to be greater in SEA than LAC for both forest conditions. In SEA there was a tendency towards higher CH4 fluxes in degraded than undegraded forests, while in LAC the opposite was true. The average annual soil CH4 emission was 5 times higher in LAC than SEA in undegraded forests (p = 0.04), with a similar trend in degraded forests. Average annual peat N2O fluxes in SEA tended to be higher in degraded than undegraded forests, and the difference was marginally significant (p = 0.08). N2O emissions in LAC degraded and undegraded forests are not compared because differences in N2O fluxes between forest conditions were linked to soil moisture fluctuations that were unrelated to degradation (Hergoualc’h et al. 2020).
Average annual water table level was higher in degraded than undegraded forests in SEA (p = 0.02), with a comparable trend in LAC. Water level tended to be higher in LAC where it was above the soil surface than in SEA where it was below the soil surface, regardless of forest condition. The difference in water level between the two regions was significant in undegraded forests (p = 0.002). This trend might be explained by the 10% greater annual precipitation at LAC sites (2772 ± 58 mm) than SEA sites (2539 ± 88 mm) (p = 0.01). Additionally water table level seasonality tended to be greater in SEA forests (36.0 ± 8.1 cm) than LAC forests (25.1 ± 10.7) though difference between the two regions was unsignificant. Across regions water table level seasonality was similar in degraded (31.3 ± 8.3 cm) and undegraded forests (31.6 ± 11.1 cm). There were not sufficient data for statistical comparison of forest conditions within regions. LAC sites had predominantly minerotrophic peat while all SEA sites were ombrotrophic peatlands. Other variables were homogeneous across regions and not affected by degradation.
Across regions and regardless of degradation, average annual soil respiration increased with increasing soil ratio of \({\text{NO}}_{3}^{ - }\) to total mineral N (\({\text{NO}}_{3}^{ - }/{\text{NH}}_{4}^{ + }\)+\({\text{NO}}_{3}^{ - }\)) considering mean forest condition values in each region (Fig. 2). The relationship between soil respiration and mineral N considering site-level values was also significant (p < 0.05). Peat CH4 fluxes increased as average annual water table levels rose closer to the soil surface (Fig. 3). The link between CH4 and water table across forest conditions and regions was driven by the relationship in SEA forests where the average annual water table level remained at or below the soil surface. There was no relationship between CH4 and ground water levels in LAC, where the average annual water table tended to be at or above the soil surface and there was large variation in rates of annual emissions of CH4 among sites. N2O fluxes increased with increasing soil pH across forest conditions in SEA (Fig. 3) (soil pH was not available for LAC sites). However, with limited data the specific parameters of the correlation are not definitive. In SEA variation in annual emissions of N2O were very small in undegraded forests as compared to degraded conditions.
Relationship between total soil respiration (SR) and \({\text{NO}}_{3}^{ - }({\text{NH}}_{4}^{ + }/{\text{NO}}_{3}^{ - })\) ratio in undrained undegraded and degraded peat swamp forests (UF and UFDeg, respectively) in Southeast Asia (SEA) and Latin America and the Caribbean (LAC). Data points and error bars represent the mean per forest condition in each region and associated standard error. Regression was performed on 4 data points and relationship is significant at p < 0.05
Relationships between average annual soil CH4 fluxes and water table level (WT) (a) and soil N2O fluxes and peat pH (b) in undrained undegraded and degraded peat swamp forests (UF and UFDeg, respectively) in Southeast Asia (SEA) and Latin America and the Caribbean (LAC). Data points represent the measured value at a site. Relationships are significant at p < 0.05. There was no relationship between average annual soil CH4 fluxes and WT in LAC
Peat GHG budget
Annual peat onsite CO2 and net GHG budgets are summarized in Table 3. In both regions, the peat was a net CO2 sink in undegraded forest and a source in degraded forest, owing to elevated heterotrophic respiration and reduced C inputs to the peat under degraded conditions. Considering 20-year GWP of non-CO2 emissions, In SEA undegraded forests peat emissions of CH4 and N2O were offset by the uptake of CO2, while LAC undegraded forests were a net GHG source, owing to large emissions of CH4. Increased net peat GHG emissions in degraded forests as compared to undegraded conditions were driven by changes in the peat CO2 balance for both regions. In SEA, enhancement of peat CH4 and N2O fluxes in degraded compared to undegraded forests made additional contributions to increased net peat GHG emissions. The peat GHG budget in SEA forests was dominated by CO2 (79% and 72% in undegraded and degraded forests, respectively) while in LAC CH4 fluxes accounted for > 50% peat GHG budget regardless of forest condition. N2O emissions comprised 1% of the peat GHG budget in LAC forests, and contributed more substantially in SEA (4% and 10% for undegraded and degraded conditions, respectively).
Considering 100-year GWP of CH4 and N2O, net peat GHG emissions were reduced across forest conditions and regions compared to budgets based on 20-year GWP. In LAC, with high CH4 emisions, undegraded swamp forests were a net GHG source when considering the 20-yr GWP of CH4 (86 kg CO2-equivalent kg−1 CH4, Myhre et al. 2013) and a sink when using methane’s 100-year GWP value (34 kg CO2-equivalent kg−1 CH4, Myhre et al. 2013).
Discussion
Peat onsite CO2 emission and uptake
Our synthesis suggests that anthropogenic disturbance in undrained tropical peat forests alters the peat CO2 sink function primarily by increasing CO2 outputs from heterotrophic respiration, transforming the peat into a net CO2 source even without alleviation of oxygen constraints on aerobic respiration by drainage. Peat onsite CO2 emissions (Mg CO2–C ha−1 yr−1) in SEA and LAC degraded forests (4.1 ± 2.0 and 1.4 ± 2.2, respectively) were correspondingly 77% and 26% of the default IPCC EF for drained tropical peat forests (5.3) (Drösler et al. 2014). Without EF for undrained degraded forests, anthropogenic emissions from these ecosystems are either unaccounted, or the lack of disaggregation of degraded forests by drainage status, as in the Indonesian Forest Reference Emission Level (Budiharto et al. 2022), may overestimate peat onsite CO2 emissions, particularly in the case of the Indonesian EF (8.8 Mg C ha−1 yr−1, Novita et al. 2021b). Moreover, the latter Tier 2 EF is an overestimate of onsite CO2 emissions as it was based solely on heterotrophic respiration regardless of litter C inputs.
Mean annual total peat C inputs from litterfall and root mortality (Mg CO2–C ha−1 yr−1) in undegraded forests, which were similar in the two regions (8.6–9.0), were comparable to an average for undrained SEA peat forests (8.9 ± 1.4, Hergoualc’h and Verchot 2014) and slightly higher than a global estimate for tropical flooded forests on organic soils (8.0, Sjögersten et al. 2014). In both regions degradation tended to decrease total litter C inputs (6.4–8.1), but the impact of anthropogenic disturbance was inconsistent among sites in SEA where forests were impacted by different types of activities (logging, agroforestry) and were in different stages of recovery following disturbance (Table S1). For example, litter C inputs were higher in an Indonesian secondary peat swamp forest compared to a paired primary forest site in Central Kalimantan (Swails et al. 2021) but lower in previously logged than pristine forests in Sumatra (Brady 1997). LAC degraded forests were represented by a single location and disturbance type (cutting of M. flexuosa palms and timber harvest) (Table S1), therefore degradation-induced reduction in litter C inputs would need to be further evaluated. Consistent with our finding of variation in site-specific impacts of degradation on litter C inputs in peat swamp forests, litterfall in secondary tropical forests on mineral soils may be similar (Bambi et al. 2022; Burghouts et al. 1992; Herbohn and Congdon 1993), reduced (Riutta et al. 2021; Villela et al. 2006), or enhanced (Aryal et al. 2015; Ostertag et al. 2008) when compared to intact forests.
Total soil respiration (Mg CO2–C ha−1 yr−1) in undegraded SEA forests (10.4 ± 1.8) was in the range of earlier findings for undegraded peat swamp forests of the region (12.9 ± 2.1, Hergoualc’h and Verchot 2014). Likewise, total soil respiration in SEA degraded forests (15.1 ± 2.0) was similar to an average for degraded forests in the region (13.0 ± 1.5 Mg CO2–C ha−1 yr−1, Hergoualc’h and Verchot 2014), despite the fact that the earlier study disregarded drainage status. On the other hand, total soil respiration in LAC undegraded and degraded forests (7.9 ± 1.3 and 8.3 ± 1.8, respectively) was lower compared to SEA and slightly lower than a global estimate for wetlands on organic soils that included forest and non-forest ecosystems (8.8 ± 2.2, Sjögersten et al. 2014). The relationship between total soil respiration and the peat nitrate (\({\text{NO}}_{3}^{ - }\)) content (Fig. 2) links enhancement of soil respiration rate across forest conditions and regions to increased nitrate availability in the soil. Mineral N can stimulate respiration of both roots and microbial populations (Zhou et al. 2014), and enhancement of tropical peat soil respiration rates have previously been linked to increased soil nutrient content in field (Hergoualc’h and Verchot 2014; Sjögersten et al. 2011) and ex situ observations (Jauhiainen et al. 2016; Swails et al. 2018). Although NH4+ was abundant in soils (Table 2), it can form complexes with organic matter making it less available to plants and microbes (McNevin et al. 1999; Nommick 1965; Witter and Kirchmann 1989), while \({\text{NO}}_{3}^{ - }\) is relatively mobile in soil solutions (e.g., Bray 1954; Johnson and Cole 1980; Vitousek et al. 1982).
The observed trend towards increased total soil respiration rate in degraded forests as compared to undegraded conditions across the two regions could be due to enhancement of heterotrophic and/or autotrophic respiration rates. However, degradation increased the contribution of heterotrophic respiration to total respiration in both SEA and LAC forests (Table S2), based on limited data, suggesting either a decline of autotrophic activity, an enhancement of heterotrophic activity, or both. For example, lowered autotrophic respiration in degraded forests compared to undegraded conditions was associated with a decline in root biomass in paired LAC palm swamp forests, with increased magnitude of total soil respiration attributed to elevation of heterotrophic respiration (Dezzeo et al. 2021; Hergoualc’h et al. 2023). Stimulation of heterotrophic respiration can result from increased quantity of soil C and/or nutrient inputs (Chambers et al. 2000; Wilcke et al. 2005) which in disturbed forests can be caused by increased necromass (Carlson et al. 2017; Palace et al. 2007; Rozak et al. 2018) and decreased litter C:N ratio associated with alteration of species composition (Uriarte et al. 2015). Coarse litter decomposition could be a significant driver of heterotrophic respiration in LAC palm-dominated swamp forests, particularly in degraded sites where the quantity of downed woody debris was increased by palm cutting (Bhomia et al. 2019). Such enhancement can last for decades, as observed by Riutta et al. (2021) in previously logged upland tropical rainforests in Indonesia. Acceleration of soil organic matter decomposition in degraded tropical forests leading to increased soil CO2 efflux has also been linked to alteration of microbial community composition (Zhou et al. 2018) though no data were from tropical peat swamp forests. Further investigations are needed to clarify links among vegetation disturbance, soil chemistry, and CO2 emissions from peat decomposition in degraded undrained forests.
Peat CH4 fluxes
Mean annual peat CH4 fluxes tended to be greater in degraded than undegraded conditions in SEA, while the opposite was true in LAC based on observations at one single location in Peru (Hergoualc’h et al. 2020). The tendency towards higher CH4 emissions in degraded than undegraded conditions in SEA may be explained by an increase in mean annual water table level in the disturbed forests compared to intact sites (Table 2). Evapotranspiration, a major determinant of water table level in tropical peatlands, is decreased by vegetation loss (Hirano et al. 2015). Selective harvest can raise the water table level by as much as 20 cm in drained boreal peatland forests (Leppä et al. 2020). The activity of methanotrophs is mainly limited by oxygen availability (Le Mer and Roger 2001), thus higher water table levels favor methane production and CH4 fluxes increased across forest conditions and regions when mean annual water table level was closer or above the soil surface (Fig. 3).
However, the opposite trends in the impact of degradation on peat CH4 emissions in the two regions, and the low explanatory power of water table level, suggest either additional controls on methanogenesis or a more nuanced, non-monotonic impact of water level. Turetsky et al. (2014) noted that deeper inundation, as at the LAC degraded sites, can inhibit CH4 emissions for several reasons—limited diffusion through deeper water, enhanced oxidation in deeper water, especially if flowing, and reduced labile C inputs. Plant residues are known to be a major substrate for methanogens (Le Mer and Roger 2001) and microbial decomposition in wetlands often correlates well with carbon quality index (Bridgham et al. 2013).
In SEA CH4 fluxes (kg C ha−1 yr−1) in degraded forests (32.7 ± 7.8) were elevated compared to the previous estimate by Hergoualc’h and Verchot (2014) of 28.6 ± 9.7 (n = 8) and relatively lower in undegraded sites (22.1 ± 13.6). In LAC, where wetlands contribute a third of global wetland emissions of methane as determined by satellite observations (Stavert et al. 2022), annual fluxes of CH4 (165.0 ± 4.5 and 218.3 ± 54.2 kg C ha−1 yr−1 in degraded and undegraded forests, respectively) tended to be an order of magnitude higher compared to SEA (Table 2) but lower than a global estimate for tropical wetlands (401 ± 17, Sjögersten et al. 2014). In addition to lower annual precipitation, at SEA sites precipitation was typically reduced during a period of 4–7 months (Ali et al. 2006; Hirano et al. 2007; Ishikura et al. 2019) while at LAC sites the dry season is shorter (2 months, Griffis et al. 2020) or there is no pronounced dry season (Wright et al. 2013). Diminished seasonality of water table levels in LAC may reflect differences in distribution of rainfall throughout the year, as well as differences in watershed hydrology between SEA and LAC sites. The difference in peat CH4 emissions between SEA and LAC sites irrespective of forest condition highlights a key challenge to the development of globally relevant soil EF for tropical peatlands.
Peat N2O fluxes
Peat N2O fluxes (kg N ha−1 yr−1) in SEA undegraded forests (0.9 ± 0.5) tended to be lower than previously estimated for undrained forests (2.7 ± 1.9, n = 7, Hergoualc’h and Verchot 2014) while in degraded forests they were higher (4.8 ± 2.3). In LAC mean annual N2O fluxes across forest conditions (1.0 ± 0.2), based on measurements from three sites at a single location in Peru (Hergoualc’h et al. 2020), tended to be similar to fluxes from undegraded forests in SEA.
Changes in pH have been identified as a significant driver of changes to soil microbial communities (Shi et al. 2011) and N2O production by soils was linked to pH in SEA forests (p = 0.02, Fig. 3). Where nitrification is the main N2O-forming process, emissions tend to increase as the pH increases (Sahrawat 1982; Granli and Bockman 1996) via its influence on microbial community structure (Jiang et al. 2015). However, the driver of the trend towards increased N2O emissions in degraded undrained SEA forests compared to undegraded conditions was not clear. Variation in N2O production by tropical peat soils is related to changes in nitrogen availability (van Lent et al. 2015), and soil N inputs could be increased in secondary forest from enhanced litter inputs (Aryal et al. 2015), lower litter C:N ratio (Yang and Luo 2011; Zou et al. 2021), or both, promoting higher N2O production rates. Though there was a small increase in aboveground litter inputs in SEA degraded forests compared to undegraded conditions, root mortality decreased with degradation leading to lowered total litter inputs (Table 3). The associated trends towards higher peat N2O emissions and increased soil \({\text{NO}}_{3}^{ - }\) content and \({\text{NO}}_{3}^{ - }({\text{NH}}_{4}^{ + }/{\text{NO}}_{3}^{ - })\) in degraded compared to undegraded conditions in SEA (Table 2) suggests higher N2O emissions from soils may be related to \({\text{NO}}_{3}^{ - }\) substrate-induced enhancement of denitrification rather than total mineral N (\({\text{NH}}_{4}^{ + }\) and \({\text{NO}}_{3}^{ - }\)) availability per se. Vegetation species composition can also influence soil N2O fluxes in tropical forests (Soper et al. 2018), and changes to species assemblages at degraded sites compared to paired undegraded sites were noted by studies in both regions (Basuki et al. 2021; Bhomia et al. 2019; Novita et al. 2021a). However, variation in N2O fluxes at LAC degraded sites were linked to differences in soil moisture unassociated with degradation (Hergoualc’h et al. 2020) and the relationship between increased soil N2O fluxes and species composition in degraded SEA sites is unclear.
Emission factors for undrained degraded tropical peat swamp forests
Although drainage is widely considered the dominant driver of increased soil GHG emissions in tropical peatlands (Couwenberg et al. 2010; Prananto et al. 2020), vegetation and soil processes are tightly linked. Our results indicated enhancement of peat GHG emissions in disturbed peat swamp forests even without lowering of the water table. In SEA, the net peat GHG emissions budget (Mg CO2-equivalent ha−1 yr−1) in undrained degraded forests (20.7 ± 7.4) was similar to that for drained tropical peat forests computed as the sum of default peat onsite CO2 and CH4 and N2O IPCC EF expressed in CO2 equivalents (21.7, using a 20-year GWP) and a drastic shift from net peat GHG uptake by undegraded forest in the region (− 7.9 ± 6.9). The net peat GHG budget in LAC undrained degraded forests (24.3 ± 8.2) was greater, albeit marginally, than the IPCC drained forest default, which is based on measurements of Indonesian ombrotrophic peats.
Across forest conditions and regions, peat C fluxes as methane were small compared to CO2 uptake and emission and when expressed on a Mg C ha−1 yr−1 basis undegraded sites were a net peat C sink. Although the IPCC encourages countries to report their GHG based on 100-year GWP, Abernethy and Jackson (2022) recommend using a shorter time-horizon GWP to better address challenges to achieving temperature goals under the Paris Agreement. Independently of GWP time horizon both SEA and LAC degraded peat swamp forests were net C sources and net GHG sources (Table 3).
Given the differences in peat GHG fluxes across forest conditions and regions, refinement of tropical peat GHG emission factors for undrained degraded forests is required to improve accuracy of country peat GHG emissions inventories. Countries are encouraged to develop Tier 2 (regional) EF for GHG inventories, given that differences in environmental controls on peat GHG fluxes (e.g., management practices, soils and vegetation properties, geologic and geomorphological differences, etc.) may drive differences in net peat GHG emissions. While data from a few arbitrary locations may not be representative of a given region, our synthesis does highlight bias in data availability among regions and its implications. For example, substantial differences in peat CH4 flux rates between the SEA and LAC forests in this study suggest that application of Tier 1 default EF based on SEA peatlands may underestimate soil emissions of CH4 in LAC forests. Further investigations of links between vegetation disturbance and changes in organic matter dynamics, soil nutrient cycling, and peat GHG emissions are needed to identify appropriate proxies for differentiating undrained degraded forest. We were limited in our analysis to disturbance types and controlling variables of soil GHG fluxes measured in the studies included in our synthesis; additional exploration of the influence of fire, rainfall history, and vegetation density on peat GHG budgets is needed. Considering the large influence of soil respiration partitioning ratios on estimates of heterotrophic respiration, investigations of the contribution of heterotrophic and autotrophic respiration to total soil respiration in undegraded and degraded forests are critical. Additional measurements that adequately cover spatial and temporal variability in peat GHG fluxes are particularly needed in minerotrophic peats, and Africa is strongly underrepresented in studies of tropical peat GHG fluxes. A substantial proportion of studies in tropical peatlands do not contribute towards reducing uncertainty in annual peat GHG fluxes due to low monitoring frequency and duration. Out of the 39 studies we reviewed on total soil respiration and peat CH4 and N2O fluxes in undrained tropical peat swamp forests, 16 (or 41% of the total) were excluded because they did not meet our criteria (see Methods) for measurement frequency or study duration.
Anthropogenic disturbance in undrained tropical peat swamp forests is widespread geographically (Hergoualc’h et al. 2017a; Miettinen et al. 2016) and therefore a major threat to ecosystem function globally, though the total area of undrained degraded peat swamp forests in the tropics is uncertain. Degradation without drainage is an important category of anthropogenic disturbance of peat swamp forests in Southeast Asia, where an estimated 74% of degraded peat swamp forests are hydrologically intact based on mapping of drainage canals (Dadap et al. 2021). Further investigation is needed to determine the extent of tropical peat swamp forest degradation with and without drainage in South America and Africa. Technologies for remote sensing of water table levels (Burden et al. 2020; Swails et al. 2019) and vegetation disturbance (Miettinen et al. 2012; Miettinen and Liew 2010; Hergoualc’h et al. 2017a) in forested peatlands offer potential methods for quantifying the extent of undrained degraded peat swamp forests over large scales. The observed increases in peat GHG emissions in undrained degraded tropical peat swamp forest as compared to undegraded conditions call for their inclusion as a new class in the IPCC guidelines to support countries in their development of GHG inventories.
Data availability
The datasets generated and analyzed during the current study can be found at https://doi.org/10.17528/CIFOR/DATA.00291.
References
Abernethy S, Jackson RB (2022) Global temperature goals should determine the time horizons for greenhouse gas emission metrics. Environ Res Lett 17(2):024019
Ali M, Taylor D, Inubushi K (2006) Effects of environmental variations on CO2 efflux from a tropical peatland in eastern Sumatra. Wetlands 26(2):612–618
Allison SD, Vitousek PM (2004) Rapid nutrient cycling in leaf litter from invasive plants in Hawai’i. Oecologia 141(4):612–619
Aryal DR, De Jong BH, Ochoa-Gaona S et al (2015) Successional and seasonal variation in litterfall and associated nutrient transfer in semi-evergreen tropical forests of SE Mexico. Nutr Cycl Agrosyst 103(1):45–60
Astiani D, Curran LM, Burhanuddin et al (2018) Fire-driven biomass and peat carbon losses and post-fire soil CO2 emission in a West Kalimantan Peatland forest. J Trop for Sci 30(4):570–575
Bambi P, Tonin AM, Rezende RDS et al (2022) The legacy of forest logging on organic matter inputs and storage in tropical streams. Biotropica 55(1):40–52
Basuki I, Kauffman JB, Peterson JT et al (2021) Land cover and land use change decreases net ecosystem production in tropical peatlands of West Kalimantan, Indonesia. Forests 12(11):1587
Berner EK, Berner RA (1996) Global environment: water, air and geochemical cycles. Prentice Hall, Hoboken
Bhomia RK, van Lent J, Rios JMG et al (2019) Impacts of Mauritia flexuosa degradation on the carbon stocks of freshwater peatlands in the Pastaza-Marañón river basin of the Peruvian Amazon. Mitig Adapt Strat Glob Change 24(4):645–668
Blonder B, Both S, Coomes DA et al (2018) Extreme and highly heterogeneous microclimates in selectively logged tropical forests. Front Forests Global Change 1:5
Both S, Elias DM, Kritzler UH et al (2017) Land use not litter quality is a stronger driver of decomposition in hyperdiverse tropical forest. Ecol Evol 7(22):9307–9318
Brady MA (1997) Organic matter dynamics of coastal peat deposits in Sumatra, Indonesia, Ph.D thesis, University of BC, Vancouver, BC, Canada, p 259
Bray RH (1954) A nutrient mobility concept of soil-plant relationships. Soil Sci 78(1):9–22
Bridgham SD, Cadillo-Quiroz H, Keller JK et al (2013) Methane emissions from wetlands: Biogeochemical, microbial, and modeling perspectives from local to global scales. Glob Change Biol 19:1325–1346
Budiharto, Krisnawati H, Manuri S et al (2022) National Forest Reference Level for Deforestation, Forest Degradation and Enhancement of Forest Carbon Stock. 2nd Submission to the United Nations Framework Convention on Climate Change
Burdun I, Bechtold M, Sagris V et al (2020) A comparison of three trapezoid models using optical and thermal satellite imagery for water table depth monitoring in Estonian bogs. Remote Sens 12(12):1980
Burghouts T, Ernsting G, Korthals G et al (1992) Litterfall, leaf litter decomposition and litter invertebrates in primary and selectively logged dipterocarp forest in Sabah, Malaysia. Ser B: Biol Sci 335(1275):407–416Philosophical Transactions of the Royal Society of London
Busman NA, Melling L, Goh KJ et al (2023) Soil CO2 and CH4 fluxes from different forest types in tropical peat swamp forest. Sci Total Environ 858:159973
Carlson BS, Koerner SE, Medjibe VP et al (2017) Deadwood stocks increase with selective logging and large tree frequency in Gabon. Glob Change Biol 23(4):1648–1660
Certini G (2005) Effects of Fire on properties of forest soils: a review. Oecologia 143:1–10
Chambers JQ, Higuchi N, Schimel JP et al (2000) Decomposition and carbon cycling of dead trees in tropical forests of the central Amazon. Oecologia 122(3):380–388
Chimner RA, Ewel KC (2004) Differences in carbon fluxes between forested and cultivated Micronesian tropical peatlands. Wetlands Ecol Manage 12:419–427
Chimner RA, Ewel KC (2005) A tropical freshwater wetland: II. Production, decomposition, and peat formation. Wetlands Ecol Manage 13:671–684
Couwenberg J, Dommain R, Joosten H (2010) Greenhouse gas fluxes from tropical peatlands in south-east Asia. Glob Change Biol 16(6):1715–1732
Dadap NC, Hoyt AM, Cobb AR et al (2021) Drainage canals in Southeast Asian peatlands increase carbon emissions. AGU Adv 2(1):e2020AV000321
Deshmukh CS, Julius D, Desai AR et al (2021) Conservation slows down emission increase from a tropical peatland in Indonesia. Nat Geosci 14(7):484–490
Dezzeo N, Grandez-Rios J, Martius C et al (2021) Degradation-driven changes in fine root carbon stocks, productivity, mortality, and decomposition rates in a palm swamp peat forest of the Peruvian Amazon. Carbon Balance Manag 16(1):1–14
Drewer J, Lohila A, Aurela M et al (2010) Comparison of greenhouse gas fluxes and nitrogen budgets from an ombotrophic bog in Scotland and a minerotrophic sedge Fen in Finland. Eur J Soil Sci 61(5):640–650
Drösler M, Verchot LV, Freibauer A et al (2014) Drained inland organic soils. In: 2013 supplement to the 2006 IPCC guidelines for National Greenhouse Gas inventories: wetlands: Methodological Guidance on lands with Wet and Drained soils, and constructed wetlands for Wastewater Treatment. Intergovernmental Panel on Climate Change, Geneva
Eusse AM, Aide TM (1999) Patterns of litter production across a salinity gradient in a Pterocarpus officinalis tropical wetland. Plant Ecol 145(2):307–315
Granli T, Bøckman OC (1996) Nitrous oxide (N2O) emissions from soils in warm climates. In: N. Ahmad (Ed.), Nitrogen economy in tropical soils: Proceedings of the international symposium on nitrogen economy in tropical soils, held in Trinidad, WI, January 9–14, 1994. Springer, Heidelberg, pp. 159–164
Griffis TJ, Roman DT, Wood JD et al (2020) Hydrometeorological sensitivities of net ecosystem carbon dioxide and methane exchange of an amazonian palm swamp peatland. Agric for Meteorol 295:108167
Gumbricht T, Roman-Cuesta RM, Verchot L et al (2017) An expert system model for mapping tropical wetlands and peatlands reveals South America as the largest contributor. Glob Change Biol 23(9):3581–3599
Hadi A, Inubushi K, Furukawa Y et al (2005) Greenhouse gas emissions from tropical peatlands of Kalimantan, Indonesia. Nutr Cycl Agrosyst 71(1):73–80
Harrison ME, Cheyne SM, Sulistiyanto Y et al (2007) Biological effects of smoke from dry-season fires in non‐burnt areas of the Sebangau peat swamp forest, central Kalimantan, Indonesia, paper presented at International Symposium and Workshop Carbon‐Climate‐ Human Interactions: Carbon Pools, Fire, Mitigation, Restoration and Wise Use, Yogyakarta, Indonesia, 27–31 August
Herbohn JL, Congdon RA (1993) Ecosystem dynamics at disturbed and undisturbed sites in north Queensland wet tropical rain forest. II Litterfall Journal of Tropical Ecology 9(3):365–380
Hergoualc’h K, Verchot LV (2011) Stocks and fluxes of carbon associated with land use change in southeast Asian tropical peatlands: a review. Glob Biogeochem Cycles. https://doi.org/10.1029/2009GB003718
Hergoualc’h KA, Verchot LV (2012) Changes in soil CH4 fluxes from the conversion of tropical peat swamp forests: a meta-analysis. J Integr Environ Sci 9(2):93–101
Hergoualc’h K, Verchot L (2014) Greenhouse gas emission factors for land use and land-use change in southeast Asian peatlands. Mitig Adapt Strat Glob Change 19:789–807
Hergoualc’h K, Gutiérrez-Vélez VH, Menton M et al (2017a) Characterizing degradation of palm swamp peatlands from space and on the ground: an exploratory study in the Peruvian Amazon. For Ecol Manag 393:63–73
Hergoualc’h K, Hendry DT, Murdiyarso D et al (2017b) Total and heterotrophic soil respiration in a swamp forest and oil palm plantations on peat in Central Kalimantan, Indonesia. Biogeochemistry 135(3):203–220
Hergoualc’h K, Dezzeo N, Verchot LV et al (2020) Spatial and temporal variability of soil N2O and CH4 fluxes along a degradation gradient in a palm swamp peat forest in the Peruvian Amazon. Glob Change Biol 26(12):7198–7216
Hergoualc’h K, van Lent J, Dezzeo N et al (2023) Major CO2 losses from degradation of Mauritia flexuosa peat swamp forests in western Amazonia. This issue
Hertel D, Harteveld MA, Leuschner C (2009) Conversion of a tropical forest into agroforest alters the fine root-related carbon flux to the soil. Soil Biol Biochem 41(3):481–490
Hirano T, Segah H, Harada T et al (2007) Carbon dioxide balance of a tropical peat swamp forest in Kalimantan, Indonesia. Glob Change Biol 13(2):412–425
Hirano T, Jauhiainen J, Inoue T et al (2009) Controls on the carbon balance of tropical peatlands. Ecosystems 12(6):873–887
Hirano T, Kusin K, Limin S et al (2015) Evapotranspiration of tropical peat swamp forests. Glob Change Biol 21(5):1914–1927
Hobbie SE (2015) Plant species effects on nutrient cycling: revisiting litter feedbacks. Trends Ecol Evol 30(6):357–363
Hoyos-Santillan J, Lomax BH, Large D et al (2019) Evaluation of vegetation communities, water table, and peat composition as drivers of greenhouse gas emissions in lowland tropical peatlands. Sci Total Environ 688:1193–1204
Hoyt AM, Gandois L, Eri J et al (2019) CO2 emissions from an undrained tropical peatland: interacting influences of temperature, shading and water table depth. Glob Change Biol 25(9):2885–2899
Indonesian Ministry of Environment & Forestry (2021) Rekalkulasi Penutupan Lahan Indonesia Tahun 2020 (Land-Cover Indonesia 2020). Direkorat Inventarisasi dan Pemantauan Sumber Daya Hutan. Direktorat Jenderal Planologi Kehutanan dan Tata Lingkungan. Kementerian Lingkungan Hidup dan Kehutanan. Jakarta
Inubushi K, Hadi A, Okazaki M et al (1998) Effect of converting wetland forest to sago palm plantations on methane gas flux and organic carbon dynamics in tropical peat soil. Hydrol Process 12(13–14):2073–2080
Inubushi K, Furukawa Y, Hadi A et al (2003) Seasonal changes of CO2, CH4 and N2O fluxes in relation to land-use change in tropical peatlands located in coastal area of South Kalimantan. Chemosphere 52(3):603–608
Ishida T, Suzuki S, Nagano T et al (2001) CO2 emission rate from a primary peat swamp forest ecosystem in Thailand. Environ Control Biology 39:305–312
Ishikura K, Hirata R, Hirano T et al (2019) Carbon dioxide and methane emissions from peat soil in an undrained tropical peat swamp forest. Ecosystems 22(8):1852–1868
Itoh M, Okimoto Y, Hirano T et al (2017) Factors affecting oxidative peat decomposition due to land use in tropical peat swamp forests in Indonesia. Sci Total Environ 609:906–915
Jauhiainen J, Silvennoinen H, Hämäläinen R et al (2012) Nitrous oxide fluxes from tropical peat with different disturbance history and management. Biogeosciences 9:1337–1350
Jauhiainen J, Silvennoinen H, Könönen M et al (2016) Management driven changes in carbon mineralization dynamics of tropical peat. Biogeochemistry 129(1):115–132
Jiang X, Hou X, Zhou X et al (2015) pH regulates key players of nitrification in paddy soils. Soil Biol Biochem 81:9–16
Johnson DW, Cole DW (1980) Anion mobility in soils: relevance to nutrient transport from forest ecosystems. Environ Int 3(1):79–90
Jones DL, Hodge A, Kuzyakov Y (2004) Plant and mycorrhizal regulation of rhizodeposition. New Phytol 163(3):459–480
Könönen M, Jauhiainen J, Laiho R et al (2015) Physical and chemical properties of tropical peat under stabilised land uses. Mires and Peat 16(8):1–13
Lähteenoja O, Ruokolainen K, Schulman L et al (2009) Amazonian floodplains harbour minerotrophic and ombrotrophic peatlands. CATENA 79(2):140–145
Lau SYL, Midot F, Dom SP et al (2022) Application of ammonium sulfate affects greenhouse gases and microbial diversity of an oil palm plantation on tropical peat. Archives of Agronomy and Soil Science, pp 1–14
Le Mer J, Roger P (2001) Production, oxidation, emission and consumption of methane by soils: a review. Eur J Soil Biol 37(1):25–50
Leppä K, Hökkä H, Laiho R et al (2020) Selection cuttings as a tool to control water table level in boreal drained peatland forests. Front Earth Sci 8:428
Lilleskov E, McCullough K, Hergoualc’h K et al (2019) Is Indonesian peatland loss a cautionary tale for Peru? A two-country comparison of the magnitude and causes of tropical peatland degradation. Mitig Adapt Strat Glob Change 24:591–623
Lo E (2005) Gaussian error propagation applied to ecological data: post-ice-storm-downed woody biomass. Ecol Monogr 75:451–466
Lugo AE, Cuevas E, Sanchez MJ (1990) Nutrients and mass in litter and top soil of ten tropical tree plantations. Plant Soil 125(2):263–280
Lupascu M, Akhtar H, Smith TE et al (2020) Post-fire carbon dynamics in the tropical peat swamp forests of Brunei reveal long‐term elevated CH4 flux. Glob Change Biol 26(9):5125–5145
Marsh CD, Hill RA, Nowak MG et al (2022) Measuring and modelling microclimatic air temperature in a historically degraded tropical forest. Int J Biometeorol 66(6):1283–1295
Martin PA, Newton AC, Bullock JM (2013) Carbon pools recover more quickly than plant biodiversity in tropical secondary forests. Proc Royal Soc B: Biol Sci 280(1773):20132236
Mata DI, Moreno-Casasola P, Madero-Vega C (2012) Litterfall of tropical forested wetlands of Veracruz in the coastal floodplains of the Gulf of Mexico. Aquat Bot 98(1):1–11
McNevin D, Barford J, Hage J (1999) Adsorption and biological degradation of ammonium and sulfide on peat. Water Res 33(6):1449–1459
Melling L, Hatano R, Goh KJ (2005a) Methane fluxes from three ecosystems in tropical peatland of Sarawak, Malaysia. Soil Biol Biochem 37(8):1445–1453
Melling L, Hatano R, Goh KJ (2005b) Soil CO2 flux from three ecosystems in tropical peatland of Sarawak, Malaysia. Tellus B: Chemical and Physical Meteorology 57(1):1–11
Melling L, Hatano R, Goh KJ (2007) Nitrous oxide emissions from three ecosystems in tropical peatland of Sarawak, Malaysia. Soil Sci Plant Nutr 53(6):792–805
Miettinen J, Liew SC (2010) Degradation and development of peatlands in Peninsular Malaysia and in the islands of Sumatra and Borneo since 1990. Land Degrad Dev 21(3):285–296
Miettinen J, Hooijer A, Wang J et al (2012) Peatland degradation and conversion sequences and interrelations in Sumatra. Reg Envriron Chang 12(4):729–737
Miettinen J, Shi C, Liew SC (2016) Land cover distribution in the peatlands of Peninsular Malaysia, Sumatra and Borneo in 2015 with changes since 1990. Global Ecol Conserv 6:67–78
Moore S, Evans CD, Page SE et al (2013) Deep instability of deforested tropical peatlands revealed by fluvial organic carbon fluxes. Nature 493(7434):660–663
Muller J, Wüst RA, Weiss D et al (2006) Geochemical and stratigraphic evidence of environmental change at Lynch’s Crater, Queensland, Australia. Glob Planet Change 53(4):269–277
Myhre G, Shindell D, Bréon FM et al (2013) Anthropogenic and natural Radiative forcing. In: Stocker TF, Qin D, Plattner GK, Tignor M, Allen SK, Boschung J et al (eds) Climate Change 2013: the physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge and New York
Nommik H (1965) Ammonium fixation and other reactions involving a nonenzymatic immobilization of mineral nitrogen in soil. Soil Nitrogen 10:198–258
Novita N, Kauffman JB, Hergoualc’h K et al (2021a) Carbon stocks from peat swamp forest and oil palm plantation in Central Kalimantan, Indonesia. Climate Change Research, Policy and Actions in Indonesia: Science, Adaptation and Mitigation, pp. 203–227
Novita N, Lestari NS, Lugina M et al (2021b) Geographic setting and groundwater table control carbon emission from Indonesian peatland: a meta-analysis. Forests 12(7):832
Nurulita Y, Adetutu EM, Gunawan H et al (2016) Restoration of tropical peat soils: the application of soil microbiology for monitoring the success of the restoration process. Agric Ecosyst Environ 216:293–303
Ong CS, Juan JC, Yule CM (2015) Litterfall production and chemistry of Koompassia malaccensis and Shorea uliginosa in a tropical peat swamp forest: plant nutrient regulation and climate relationships. Trees 29(2):527–537
Ostertag R, Marin-Spiotta E, Silver W (2008) Litterfall and decomposition in relation to soil carbon pools along a secondary forest chronosequence in Puerto Rico. Ecosystems 11:701–714
Palace M, Keller M, Asner GP et al (2007) Necromass in undisturbed and logged forests in the Brazilian Amazon. For Ecol Manag 238(1–3):309–318
Pärn J, Verhoeven JT, Butterbach-Bahl K et al (2018) Nitrogen-rich organic soils under warm well-drained conditions are global nitrous oxide emission hotspots. Nat Commun 9(1):1–8
Prananto JA, Minasny B, Comeau LP et al (2020) Drainage increases CO2 and N2O emissions from tropical peat soils. Glob Change Biol 26(8):4583–4600
Rahajoe JS, Kohyama T, Limin SH (2000) Litter decomposition process in two contrastive nutrient limited forest types in central Kalimantan, in Proceedings of the International Symposium on Tropical Peatlands, Bogor, Indonesia, 22–23 November 1999, edited by T. Iwakuma, Hokkaido Univ. and Indonesian Inst. of Sci., Bogor, Indonesia, pp. 223–231
Ribeiro K, Pacheco FS, Ferreira JW et al (2021) Tropical peatlands and their contribution to the global carbon cycle and climate change. Glob Change Biol 27(3):489–505
Riutta T, Kho LK, Teh YA et al (2021) Major and persistent shifts in below-ground carbon dynamics and soil respiration following logging in tropical forests. Glob Change Biol 27(10):2225–2240
Rozak AH, Rutishauser E, Raulund-Rasmussen K et al (2018) The imprint of logging on tropical forest carbon stocks: a bornean case-study. For Ecol Manag 417:154–166
Saarnio S, Morero M, Shurpali NJ et al (2007) Annual CO2 and CH4 fluxes of pristine boreal mires as a background for the lifecycle analyses of peat energy. Boreal Environ Res 12:101–113
Sahrawat KL (1982) Nitrification in some tropical soils. Plant Soil 65:281–286
Sakabe A, Itoh M, Hirano T et al (2018) Ecosystem-scale methane flux in tropical peat swamp forest in Indonesia. Glob Change Biol 24:5123–5136
Sánchez ME, Chimner RA, Hribljan JA et al (2017) Carbon dioxide and methane fluxes in grazed and undisturbed mountain peatlands in the Ecuadorian Andes. Mires and Peat 19(20):1–18
Saragi-Sasmito MF, Murdiyarso D, June T et al (2019) Carbon stocks, emissions, and aboveground productivity in restored secondary tropical peat swamp forests. Mitig Adapt Strat Glob Change 24(4):521–533
Shi S, Richardson AE, O’Callaghan M et al (2011) Effects of selected root exudate components on soil bacterial communities. FEMS Microbiol Ecol 77(3):600–610
Shimamura T, Momose K (2005) Organic matter dynamics control plant species coexistence in a tropical peat swamp forest. Proc Royal Soc B: Biol Sci 272(1571):1503–1510
Sjögersten S, Cheesman AW, Lopez O et al (2011) Biogeochemical processes along a nutrient gradient in a tropical ombrotrophic peatland. Biogeochemistry 104(1):147–163
Sjögersten S, Black CR, Evers S et al (2014) Tropical wetlands: a missing link in the global carbon cycle? Glob Biogeochem Cycles 28(12):1371–1386
Sjögersten S, Siegenthaler A, Lopez OR et al (2020) Methane emissions from tree stems in neotropical peatlands. New Phytol 225(2):769–781
Soper FM, Sullivan BW, Nasto MK et al (2018) Remotely sensed canopy nitrogen correlates with nitrous oxide emissions in a lowland tropical rainforest. Ecology 99(9):2080–2089
Stavert AR, Saunois M, Canadell JG et al (2022) Regional trends and drivers of the global methane budget. Glob Change Biol 28(1):182–200
Sulistiyanto Y (2004) Nutrient dynamics in different sub-types of peat swamp forest in central Kalimantan, Indonesia, Ph.D. thesis, University of Nottingham, Nottingham, UK, p. 351
Sundari S, Hirano T, Yamada H et al (2012) Effect of groundwater level on soil respiration in tropical peat swamp forests. J Agricultural Meteorol 68(2):121–134
Swails E, Jaye D, Verchot L et al (2018) Will CO2 emissions from drained tropical peatlands decline over time? Links between soil organic matter quality, nutrients, and C mineralization rates. Ecosystems 21(5):868–885
Swails E, Yang X, Asefi S et al (2019) Linking soil respiration and water table depth in tropical peatlands with remotely sensed changes in water storage from the gravity recovery and climate experiment. Mitig Adapt Strat Glob Change 24(4):575–590
Swails E, Hergoualc’h K, Verchot L et al (2021) Spatio-temporal variability of peat CH4 and N2O fluxes and their contribution to peat GHG budgets in Indonesian forests and oil palm plantations. Front Environ Sci 48:617828
Swails E, Drewer J, Hartill J et al (2023) Soil nitrous oxide fluxes from a land-use change transition in an Indonesian peatland. Biogeochemistry. https://doi.org/10.1007/s10533-023-01070-7
Turetsky MR, Kotowska A, Bubier J et al (2014) A synthesis of methane emissions from 71 northern, temperate, and subtropical wetlands. Glob Change Biol 20(7):2183–2197
Uriarte M, Turner BL, Thompson J et al (2015) Linking spatial patterns of leaf litterfall and soil nutrients in a tropical forest: a neighborhood approach. Ecol Appl 25(7):2022–2034
van Lent J, Hergoualc’h K, Verchot LV (2015) Reviews and syntheses: soil N2O and NO emissions from land use and land-use change in the tropics and subtropics: a meta-analysis. Biogeosciences 12(23):7299–7313
Vijayanathan J, Ishak MF, Parlan I et al (2021) Temporal patterns control carbon balance in forest and agricultural tropical peatlands in North Selangor, Malaysia. iForest-Biogeosciences and Forestry 14(4):362
Villela DM, Nascimento MT, de Aragao LEO et al (2006) Effect of selective logging on forest structure and nutrient cycling in a seasonally dry Brazilian Atlantic forest. J Biogeogr 33(3):506–516
Vitousek PM, Gosz JR, Grier CC et al (1982) A comparative analysis of potential nitrification and nitrate mobility in forest ecosystems. Ecol Monogr 52(2):155–177
Wang H, Richardson CJ, Ho M (2015) Dual controls on carbon loss during drought in peatlands. Nat Clim Change 5(6):584–587
Weiss D, Shotyk W, Cheburkin AK et al (1997) Atmospheric lead deposition from 12,400 to ca. 2,000 yrs BP in a peat bog profile, Jura Mountains, Switzerland. Water Air Soil Pollut 100(3):311–324
Weiss D, Shotyk W, Rieley J et al (2002) The geochemistry of major and selected trace elements in a forested peat bog, Kalimantan, SE Asia, and its implications for past atmospheric dust deposition. Geochim Cosmochim Acta 66(13):2307–2323
Wilcke W, Hess T, Bengel C et al (2005) Coarse woody debris in a montane forest in Ecuador: mass, C and nutrient stock, and turnover. For Ecol Manag 205(1–3):139–147
Witter E, Kirchmann H (1989) Peat, zeolite and basalt as adsorbents of ammoniacal nitrogen during manure decomposition. Plant Soil 115:43–52
Wright EL, Black CR, Turner BL et al (2013) Environmental controls of temporal and spatial variability in CO2 and CH4 fluxes in a neotropical peatland. Glob Change Biol 19(12):3775–3789
Yang Y, Luo Y (2011) Carbon: nitrogen stoichiometry in forest ecosystems during stand development. Glob Ecol Biogeogr 20(2):354–361
Zhou L, Zhou X, Zhang B et al (2014) Different responses of soil respiration and its components to nitrogen addition among biomes: a meta-analysis. Glob Change Biol 20(7):2332–2343
Zhou Z, Wang C, Luo Y (2018) Effects of forest degradation on microbial communities and soil carbon cycling: a global meta-analysis. Glob Ecol Biogeogr 27(1):110–124
Zou J, Wu J, Osborne B et al (2021) Nitrogen accumulation, rather than carbon: nitrogen stoichiometric variation, underlies carbon storage during forest succession. Environ Res Lett 16(2):02405
Acknowledgements
We would like to thank Novi Wahyuni and Aoife O’Donoghue for their assistance in identifying literature for inclusion in this study. The concept for this paper was presented at the workshop “Peatlands for climate change mitigation in agriculture” that took place in Aarhus, Denmark, on 4–5 October 2022, and which was sponsored by the Organisation for Economic Co-operation and Development (OECD) Co-operative Research Programme: Sustainable Agricultural and Food Systems.
Funding
This research was conducted under the Sustainable Wetlands Adaptation and Mitigation Program (SWAMP) and was generously supported by the governments of the United States of America (Grant MTO-069033) and Norway (QZA-21/-124). It was undertaken as part of the CGIAR research program on Climate Change, Agriculture and Food Security (CCAFS).
Author information
Authors and Affiliations
Contributions
Literature search and data analysis were performed by ES. The first draft of the manuscript was written by ES and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Klaus Butterbach-Bahl.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Swails, E., Frolking, S., Deng, J. et al. Degradation increases peat greenhouse gas emissions in undrained tropical peat swamp forests. Biogeochemistry 167, 59–74 (2024). https://doi.org/10.1007/s10533-023-01110-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-023-01110-2