Abstract
In recent years, biologists and philosophers of science have argued that evolutionary theory should incorporate more seriously the idea of ‘reciprocal causation.’ This notion refers to feedback loops whereby organisms change their experiences of the environment or alter the physical properties of their surroundings. In these loops, in particular niche constructing activities are central, since they may alter selection pressures acting on organisms, and thus affect their evolutionary trajectories. This paper discusses long-standing problems that emerge when studying such reciprocal causal processes between organisms and environments. By comparing past approaches to reciprocal causation from the early twentieth century with contemporary ones in niche construction theory, we identify two central reoccurring problems: All of these approaches have not been able to provide a conceptual framework that allows (i) maintaining meaningful boundaries between organisms and environments, instead of merging the two, and (ii) integrating experiential and physical kinds of reciprocal causation. By building on case studies of niche construction research, we provide a model that is able to solve these two problems. It allows distinguishing between mutually interacting organisms and environments in complex scenarios, as well as integrating various forms of experiential and physical niche construction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
‘Reciprocity’ has become a buzzword in current biological parlance, especially in debates about the purported reciprocal nature of many developmental and evolutionary processes (e.g., Laland et al. 2011, 2013; Moczek 2015; Schwab et al. 2019; Uller and Laland 2019). Some biologists contend that the study of complex feedbacks between developing organisms and their environments, which have protracted evolutionary consequences, brings about important reconfigurations to standard evolutionary theory (see Laland et al. 2011, 2013, 2015). At the same time, a philosophical debate surrounding the novelty, limits and scope of the theoretical tenet of ‘reciprocal causation’ has emerged (e.g., Dickins and Barton 2013; Scholl and Pigliucci 2015; Svensson 2018; Baedke 2019; Fábregas-Tejeda and Vergara-Silva 2018a), primarily evaluating the concept’s epistemological, explanatory, and heuristic roles in biological practice (Buskell 2019). From the scientific side, niche construction theory (NCT; sensu Odling-Smee et al. 2003) has stood at the forefront of this debate (Uller and Helanterä 2019), but allied fields and research areas such as ecological evolutionary developmental biology (eco-evo-devo) and developmental systems theory have also contributed (see Schwab et al. 2019; Baedke and Gilbert 2020).
A common misassumption in this debate is that the reasoning about reciprocal interactions in evolution is a fairly recent development in the biosciences. However, reciprocity has been theorized in many ways throughout the history of biology. Firstly, a diversity of relata have been conceptualized as engaged in reciprocal relationships: for instance, organism-environment, gene-environment, gene-population, or population-population. Secondly, different kinds of reciprocal relationships have been posited for these relata. For example, for the organism-environment relationship, biologists have postulated ontological co-constitution, mutual structural fitting, concomitant reaction, and reciprocal causation, among others, as different hallmarks of reciprocity (for a different historical taxonomy, see Di Paolo 2020).
Ontological co-constitution holds that organisms and their environments are commingled and form a single interacting system that cannot be meaningfully disentangled (e.g., Haldane 1884, 1935; Levins and Lewontin 1985; Griffiths and Gray 1994; Oyama 2000; Walsh 2015:181, 2021; for an overview, see Baedke 2019; Pearce 2020). The view of mutual structural fitting between organism and environment can be found in Lawrence J. Henderson (1913), who argued that, besides considering organisms as structurally adapted to their surroundings, the specific physico-chemical properties of the environment should also be regarded as adapted, for they support the development of life: “The fitness of the environment is one part of a reciprocal relationship of which the fitness of the organism is the other” (Henderson 1913:271). Concomitant reaction refers to an event that concurrently alters an organism and its environment. It resembles John Stuart Mill’s (1843) law of coexistence. An example is the competitive exclusion principle in ecology, which states that species that have similar niches cannot stably coexist in the same place when their common resources are limiting (see Raerinne and Baedke 2015). Finally, reciprocal causation is usually defined as a feedback loop between two interacting, yet separate entities or processes: “According to the causal construal, organisms cause changes to the features of the environment external to them. […] These modified physical conditions, in turn, redound upon the organisms” (Walsh 2015:180).
Here, from this vast array of views on reciprocity in ecology and evolution, we will focus on organism-environment reciprocal causation, given that this has been the main target of recent heated discussions about NCT and the so-called ‘Extended Evolutionary Synthesis’ (Laland et al. 2015, 2017; see also Fábregas-Tejeda and Vergara-Silva 2018b). In recent years, some evolutionary biologists have highlighted reciprocal causation as a theoretical and modeling principle due to its alleged empirical aptness, claiming that reciprocal interactions are ubiquitous in evolutionary processes. Moreover, it could be used to explain stable, biased selection pressures acting on organisms, and provide a conceptual framework that could potentially extend the explanatory power and limited practices of standard evolutionary theory (Buskell 2019; see also Baedke et al. 2020a). Nevertheless, as Buskell (2019) appositely points out, this concept has remained ambiguous and it is still in need of further philosophical clarification.
In this paper, we provide guidance to unknot organism-environment reciprocal causation in current evolutionary and philosophical debates. We identify reoccurring problems in these views and introduce a model for solving them. We begin by providing a brief overview of some theorizations of organism-environment reciprocity in early twentieth-century biology. Our historical narrative contextualizes the study of reciprocity in novel ways by drawing on sources hitherto under-explored by scholars and by explaining the decline of studies on organism-environment reciprocity in evolutionary biology in the second half of the twentieth-century. We identify two main theoretical problems in these early conceptual frameworks that contributed to this development: They could not (i) maintain (meaningful) epistemic boundaries between organisms and environments, but instead merged the two, and (ii) failed to integrate physical kinds of reciprocal causation with those in which organisms’ experience of the environment plays a key role. These shortcomings led to methodological intractability and untranslatability to experimental interventions. Next, we show how these problems are reoccurring in present-day debates on reciprocal causation in evolutionary biology, especially in NCT. In this sense, we demonstrate that a better historical understanding of former debates on reciprocal causation can be a guide for exposing unsolved challenges in contemporary scientific practice. Finally, we propose a general conceptual and visual model of reciprocal causation that tackles these long-standing problems. By applying it to a number of niche construction (NC) cases, we illustrate how this model allows us to (i) distinguish between mutually interacting organisms and environments, and (ii) integrate various forms of experiential and physical NC. By doing so, we try to bridge the gap between conceptual frameworks of NC and scientific practice. In particular, we show that the organism-environment boundary is an epistemic necessity to understand complex causality in NC processes, including the causal roles each component plays in them.
Our model disentangles seemingly inextricable reciprocal relations by identifying characteristic causal patterns of different evolutionary relevant organism-environment interactions. This allows correlating different types of NC within one common causal framework. In this context, it is important to stress that we do not propose a new taxonomy of types of NC here. Rather, we move in a different direction: towards integrating and making sense of different processes that involve reciprocal causation between organism and environment. Our model is able to identify and distinguish between different kinds of reciprocal causal interactions, clarify their characteristic causal patterns and integrate them even in complex evolutionary scenarios that have been difficult to conceptualize thus far (e.g., those that involve interactions of two or more species, or those in which physical and experiential views of the environment must be integrated). This approach secures the comparability of NC methodologies and opens avenues for formalization to advance the debates in the field.
Organism-environment reciprocity in historical context
In order to clarify the concept of reciprocal causation, especially in NCT, we first need to understand how it was introduced and applied in the history of evolutionary thought. In fact, clarifying this poorly understood conceptual history sheds light on the theoretical challenges that reciprocal causation approaches to evolution still face today.
Svensson (2018) argues that the ideas of causal reciprocity, and of organisms as active modifiers of their selection pressures, mainly stem from the dialectical biology of Levins and Lewontin (1985; see also Lewontin 1983). However, these ideas are considerably older and theoretically richer. Already Haeckel (1866:I, 154; German original) claimed that “reciprocity of every single individual with its entire surrounding leads to the adaptation of its individual characters.” While he did not spell out this notion in detail, other scholars in the late nineteenth century and particularly the early twentieth century did. Especially organism-centered approaches to evolution, such as organicism, (German) holistic biology, and dialectical materialism (see Nicholson and Gawne 2015; Baedke 2019), developed sophisticated views of organism-environment reciprocity.
For example, J.S. Haldane (1884:32–33) highlighted: “The organism is thus no more determined by the surroundings than it at the same time determines them. The two stand to one another, not in the relation of cause and effect, but in that of reciprocity.” Holistic thinkers like Haldane expanded Immanuel Kant’s view of reciprocity on the organization of organisms to the relation between organism and environment. As Whitehead (1929: 185) puts it: “The relation of part to whole has the special reciprocity associated with the notion of organism […]; but this relation reigns throughout nature.” He contended that evolutionary research should acknowledge that “organisms can create their own environment” (Whitehead 1929: 140).
Let us take a brief look at three early advocates of causal reciprocity–Jakob von Uexküll, Adolf Meyer-Abich, and Conrad Hal Waddington–to see how the above general ideas were developed into theoretical frameworks for biology. Uexküll (1928) conceptualized the environment (Umwelt) as constructed through the sensorial exploration of each organism. He argued that each organism is reciprocally connected with its own environment, which it subjectively perceives and acts upon. Thus, the environment contains a ‘perception world,’ which is experienced through sense receptors and processed neurally, and an ‘effect world,’ in which the organism causally acts on the environment through various traits and behaviors. The organism’s perceptions and actions together create a causal feedback loop or ‘function-circle’ (Funktionskreis) between the perception world and the effect world (see Fig. 1). For example, when organisms explore new environments, they attempt to establish new functional circles (e.g., by changing nutritional habits). If this action is not rewarded (i.e., a functional circle cannot be closed, e.g., when the new diet is not nutritious), the organism gives up this environment (switching back to old nutritional habits or exploring new ones). During this process, the environment causes the organism to rewire neural networks and physiological receptor-reaction connections.
Uexküll’s model of reciprocal organism-environment interaction. A simplified schema of the ‘function-circle.’ The organism (subject) perceives a ‘carrier of a feature’ of the environment (object) in its ‘perception world’ and reciprocally acts on and shapes the same environment (as a carrier of this effect) in its ‘effect world.’ (After Uexküll 1928:158)
Uexküll’s view of reciprocity conceptualizes the organism-environment relationship as highly individualized and mediated through experience (see Brentari 2015). It is likely because of this that it had only limited influence on evolutionary biology. One of the few scholars that drew extensively on Uexküll was Adolf Meyer-Abich (1943, 1964). His evolutionary reciprocity theory of holobiosis anticipated central elements of Lynn Margulis’ theory of endosymbiosis and introduced the holobiont concept around 50 years before her (Baedke et al. 2020b). Meyer-Abich argued that macroevolutionary change is driven by reciprocal processes between increasingly integrated organisms, which first constitute symbiotic partners, then holobionts, and finally systems of organs in a larger whole. At the crucial evolutionary stage of holobiosis, the formerly independent units can no longer develop on their own. Holobionts are able to realize novel traits and explore new environments. For example, in ‘lichen holobiosis,’ through the increasing interaction between green algae and/or blue-green algae and fungi, a new metabolic unit with new traits (novel metabolites and reproductive bodies) emerges over evolutionary time. This unit has a new energy-status, which allows accessing an unexplored ecological niche. In short, during holobiosis symbiotic organisms create new environments, which they both act on and experience in a novel manner. This view integrates previously distinct ‘function circles’ through organism-organism reciprocal causation (Meyer-Abich 1942). Organisms do not only construct their own individual environment (sensu Uexküll), but also construct integrated and shared environments for one another.
Another evolutionary take on reciprocal causation was developed by Conrad Hal Waddington. He argued that evolution involves changes in explorative behaviors (what he called ‘exploitive systems;’ see Waddington 1957:104–108, Waddington 1959a). These include the influence exerted by the organism on its environment, leading to feedback loops with selection pressures: “Animals […] themselves select the particular habitat in which their life will be passed. Thus the animal by its behaviour contributes in a most important way to determining the nature and intensity of the selective pressures which will be exerted on it” (Waddington 1959a:1635–1636). Waddington urged biologists to abandon two common views on the organism-environment relationship: First, “[n]atural selection is far from being as external a force as the conventional picture might lead one at first sight to believe” (1959a:1636). Second, “we have to think in terms of circular and not merely unidirectional causal sequences” (Waddington 1959b:400). As we will see below, it was these very two views, namely constructivism instead of externalism and causal reciprocity instead of unidirectional causality, that came under attack in evolutionary biology.
Theoretical problems and decline of organism-environment reciprocity
Traditional approaches of organism-environment reciprocal causation suffered from two main problems, already hinted by some scientists at the time, which contributed to their demise in the second half of the twentieth century: (i) Scholars were unsuccessful in retaining meaningful boundaries between organism and environment, and thus frequently proposed views of reciprocity in which organism and environment are merged, making them intractable for empirical studies; and (ii) there was little integration of experiential and physical views of the environment impinging on organisms. As we will see below, these problems are re-emerging in today’s debate regarding reciprocal causation and NCT.
(i) Many early twentieth-century authors held holistic viewpoints apropos the organism-environment relationship in which it is impossible “to distinguish separately the factors concerned,” and life is regarded as an “integrated unity covering both organism and its environment” (Haldane 1935:12; see also Uexküll 1909:196).Footnote 1 In that same line, Waddington (1957:189) claimed that “organism and environment are not two separable things.” But this stance of organism-environment inseparability, in which reciprocal interactions constitute each other into a single system, was also heavily criticized. Against Haldane, for instance, Joseph Needham (1936:10–11) highlighted: “we easily see that his demand for the unification of the organism and its surroundings is a methodologically impossible aim, for if no line can be drawn between organism and immediate surroundings, no better line can be drawn between immediate surroundings and far-off surroundings.” According to Needham, biologists need individuation criteria in order to carry on their research, otherwise they are unable to tell apart any organic system from another or environmental features relevant to the organism from those irrelevant to it. This was a common problem of strong views of reciprocity. For example, dialectical biologist Marcel Prenant (1943:61) spoke of an “organism-environment complex” in which causes and effects become indistinguishable. In general, this shared position led scientists to blur the boundary between organism and environment, resulting in methodological problems for biological practice. In that vein, many biologists saw organism-environment reciprocity as intractable (or harder to assay) in empirical studies, or maintained that explanations appealing to this principle have particular epistemic demands that call for different research strategies (e.g., Labouvie 1974).
(ii) Another main problem was that, for many biologists, the concept of ‘environment’ defended by reciprocity theories had shortcomings. Uexküll’s ‘sense physiological’ characterization of environment was deemed too narrow. Authors argued that the environment experienced by any organism (e.g., through its sensory receptors) is not the whole (relevant) environment for it (e.g., Hartmann 1950). Although not experienced, physical environmental factors can have causal consequences over organisms and their evolutionary trajectories. Moreover, the notion of experience was a heavily debated topic (e.g., Bierens de Haan 1947) and most researchers adamantly opposed to studying anything reminiscent of subjectivity (e.g., Tinbergen 1963). In sum, theories of organisms-environment reciprocity failed in providing a clear understanding of how experienced and physical environments are related, and how they can be integrated under one conceptual framework.
The decline of organism-environment reciprocity views in the second half of the twentieth century was also part of a more general trend that displaced the organism as the central, causally efficacious unit of biology, and supplanted it with the gene (Nicholson 2014; Baedke 2019). At least two stands can be identified in this trend: The marginalization of views of organism-environment reciprocity and a shift to study reciprocity in other relata; and the (re-)establishment of externalism as one of the main explanatory principles in evolutionary biology, thereby excluding notions of ‘individual experienced environments.’
First, a move away from studying organism-environment reciprocity was welcomed by classical and population geneticists. Clear boundaries between organisms and environments are a stipulation for fruitful research and “a practically and theoretically valuable abstraction” in those fields (Haldane 1936:349). Moreover, mainstream evolutionary biology was dominated by an asymmetric, unidirectional view of the organism-environment relationship (e.g., Williams 1992:484). The organism was downplayed as a causal agent that constructs its environment, and thus its own development and evolution.
Around the same time, gene-environment reciprocity was increasingly considered in population genetics (e.g., Haldane 1946; Lerner 1950; Falconer 1952), and studied through statistical tools like path analysis (Wright 1960). New models of reciprocal interactions of genes-populations and genes-environments (e.g., Fisher 1930; Kirkpatrick 1982; for overviews, see Reznick 2013 and Svensson 2018) focused, among other things, on positive and negative frequency-dependent selection (e.g., Fisher 1930; Wright 1969; Charlesworth 1971), population regulation by genetic feedbacks (e.g., Pimentel 1968), and eco-evolutionary dynamics (e.g., Thompson 1998). Their advantages notwithstanding, the vast majority of these evolutionary models did not encompass organism-environment reciprocal causation, despite what some scientists claim (Brodie III 2005; Svensson 2018). Reciprocity was usually studied for other relata, but not for whole organisms and their environments.
Second, an overt ‘externalist logic’ in evolutionary explanations reigned supreme (see Godfrey-Smith 1996; Walsh 2015), and the main concepts of environment employed in the field attest to that shift (see Brandon 1990; see also Antonovics et al. 1988): Views of ‘individual environments,’ like that of Uexküll, were replaced by populational vantage points. Brandon (2012) contends that, although no two organisms will ever interact with the same environment, they need to be seen as members of a homogeneous, shared selective environment (i.e. factors external to a population that affect its members’ relative reproductive success). Explaining evolutionary changes in genotype frequencies requires that two different genotypes share an external selective environment to which one is better adapted than the other (see also Walsh 2021).
This externalist logic impoverished the diverse roles previously granted to the environment in development and evolution. Evolutionists commonly overestimated the causal influence of the selective environment on organisms, and increasingly downgraded the influence of the non-selective environment: “Environment variance is a source of error that reduces precision in genetical studies, and the aim […] is therefore to reduce it as much as possible” (Falconer 1960:140). Waddington (1957:189) bitterly complained about this drawback: “Any further influence which the environment might have was degraded to the status of mere ‘noise’ in the system of genetic determination.”
In the next section, we will turn to new views defending organism-environment reciprocal causation that are currently emerging in evolutionary biology. With a focus on NCT and the novel taxonomies of types of NC that have been recently proposed, we will show that the same problems (i) and (ii) that plagued early organism-environment reciprocity perspectives are still lingering in the field.
Niche construction theory and reoccurring problems
The idea of organism-environment reciprocity has recently made a comeback in evolutionary biology (Laland et al. 2011, 2013, 2015; Mesoudi et al. 2013; for discussion, see Fábregas-Tejeda and Vergara-Silva 2018a, b; Svensson 2018; Buskell 2019; Baedke 2019). This development has been stirred by a growing interest to again center evolutionary reasoning on the organism (Nicholson 2014; Laland et al. 2013, 2015, 2017; Moczek 2015; Sultan 2015; Walsh 2015), including organisms’ phenotypic plasticity and NC behaviors, which, as feedback circles, modify the natural selection pressures impinging upon them (Lewontin 1983; Sterelny 2001; Odling-Smee et al. 2003; 2011; Chiu and Gilbert 2015, 2020). NCT argues that organisms are not only effects of adaptation, but also causal starting points of evolutionary trajectories. This view of reciprocal causation has recently led to a number of different taxonomies distinguishing diverse kinds of feedback processes in NC.Footnote 2
According to Aaby and Ramsey (2019), during NC, organisms alter the feature-factor relationship between themselves and their environments in three different ways: by affecting environmental factors, by changing their features (traits), or by modifying the relation between organismic features and environmental factors. They call these external, constitutive and relational NC, respectively (for definitions, see Table 1, 1–3). In contrast, Chiu (2019) distinguishes physical NC from experiential NC (see Table 1:4–5), depending on whether organisms change the intrinsic physical properties of the environment or the way the environment is experienced, without, however, physically changing it. In this taxonomy, relocational NC and mediational NC are subtypes of experiential NC.
These taxonomies of types of NC–distinguishing physical/external, constitutive, relational, and experiential (including relocational and mediational) NC–have enriched our understanding of reciprocal organism-environment interactions in development and evolution. At the same time, however, despite these conceptual improvements the current debate about different types of NC still faces the same problems that theories of reciprocity could not solve in the early twentieth century: participants in this debate usually (i) do not spell out on what grounds meaningful boundaries between organisms and environments can be maintained and exploited for research purposes, and/or (ii) provide no guidance for how to integrate experiential and physical forms of reciprocal causation.
(i) By building on a rather externalist view of the niche (Brandon 1990), early NCT constructed organisms and environment as separate entities (Odling-Smee et al. 1996, 2003). Discounting relocation, every instance of NC was construed as an alteration of the physical properties of the environment. The role of organismal experience as an active causal factor in reciprocal interactions was not a central element of early NCT. Afterwards, influenced by developmental systems theory (Oyama 2000; Griffiths and Gray 2001) and its prominent assumption that “[t]here is no distinction between organism and environment” (Griffiths and Gray 2001: 207), more constructivist views of the environment emerged in NCT. Similar to J.S. Haldane’s or Uexküll’s earlier ideas, present-day strands of NCT often highlight the centrality of the organism in evolution, but, at the same time, merge organisms with environments and argue, more generally, that they are “engaged in reciprocally caused relationships […], entwined in, to coin a very apt phrase from developmental systems theory, ‘cycles of contingency’” (Laland and O’Brien 2011:193; see also Laland and Brown 2018:127).
Sonia Sultan holds that we have to accept the “difficulty of identifying a meaningful boundary between the organism and the environment [because] individual phenotypes inevitably affect both the external environment and the organism’s experience of that environment” (Sultan 2015:44–45). For example, she highlights that, when day length changes, pregnant meadow voles (Microtus pennsylvanicus) alter a key hormonal signal in their offspring. Due to a change in parental experience autumn-born pups develop thicker coats compared to summer-born pups, and this affects how they experience and interact with their environment. By acknowledging such reciprocal connections between the organism’s experience and external environment, Sultan expresses discomfort with the organism-environment distinction. Interestingly, she develops her critique along the same arguments about the interconnectedness of organisms’ ‘perception world’ and ‘effect world’ that already made Uexküll (1909:196) contend that the environment forms an “inextricable whole” with the organism.
Walsh (2021) links another inseparability view to reciprocal causation. He argues that “[t]wo causes, x and y, are reciprocal just if the effect that x has on y at time t is dependent upon the effect that y has on x at time t” (original emphasis). He asserts that when an environment’s effect on an organism is a concomitant consequence of the organism’s effect on that environment, one cannot explain the dynamics of the overall system by apportioning it to the effects of each component, thereby making the system inseparable. This view is, strictly speaking, not one of reciprocal causation, but one of synchronic ontological co-constitution, similar to that of Haldane (1884, 1935). However, Walsh understands his inseparability thesis to also be of relevance for temporally expanded processes of reciprocal NC (e.g., Laland et al. 2016).Footnote 3
These recent arguments for organism-environment inseparability open the way for anti-individualistic positions in NCT (see Baedke 2019). These views are methodologically problematic given that NCT needs to be able to clearly individuate the organism and its causal roles. This is because this approach tries to conceptualize and study the organism as a causal agent, different from its environment, which actively molds its own niche and evolutionary trajectory. Moreover, as noted in the previous section (see Needham 1936:10–11), the holistic stance of organism-environment inseparability is troublesome because it does not solve, ipso facto, the problem of ‘individuation’ of biological units. Claiming that organism and environment form an inextricable unit still carries the burden of proof of how to individuate such a system (i.e., how to separate it from other systems). Individuation is thus unavoidable for distinguishing between proximal and distal environments of a particular system (e.g., what elements of the physical world are included in this system and in what spatio-temporal scales) as well as for clarifying the units of physiological and ecological interaction, for recognizing which biological systems can legitimately claimed to be ‘causal agents,’ for distinguishing between one or more conspecifics in a community or population, and for individuating the components in multi-species collectives such as holobionts. In that sense, the holistic view of organism-environment inseparability does not address the problem of individuation of biological units, but simply pushes it aside.
(ii) Besides the old inseparability problem, a second challenge reoccurs in today’s reciprocity views on NC. It concerns the lack of integration between approaches of physical and experiential NC. While already Lewontin (Levins and Lewontin 1985:98–106; Lewontin 2000:55–68) (re)introduced the idea of experiential NC, this concept has not had a major impact on NCT to this day (see Chiu 2019). For example, critics have contended that ‘mediational NC’ is nothing but a phenotypic response to selective environmental pressures and that changes in environmental experiences should not be mixed up with changes in the environment (Brandon 1990; Godfrey-Smith 1996, 2001). Doing so might result in defending anti-realistic and insoluble holistic positions in which organisms subjectively construct environments.
While, in recent years, we have seen a number of approaches that try to strengthen the idea of experiential mediation of the environment in evolution (Walsh 2015, 2021; Sultan 2015; Chiu 2019; Chiu and Gilbert 2020), at least some of them suffer from the older Uexküllian stance of reinterpreting the whole environment of the organism as nothing but the experienced environment. For those that want to avoid this step, arguments have to be provided for how exactly the evolutionary relevant physical environment of organisms differs from their experienced one, why certain downstream physical effects on the environment can be distinguished and excluded from the processes of experiential NC, and how exactly experience affects evolution (e.g., population dynamics) differently from physical effects on selection pressures. These challenges, together with older anti-holistic objections, still lead to a lack of integration between approaches of physical/external and experiential NC, and of the application of experiential NC in evolutionary theory more generally.Footnote 4
Here, we do not argue for a conceptualization of the environment as “a wholly external, autonomous, causally unified entity” (Walsh 2021:3). In fact, we side with Walsh in pointing out that this construal of the environment is an abstraction that, according to the historical narrative of the previous section, solidified in evolutionary biology after the decline of organism-environment reciprocity theories in the second half of the twentieth century. Nevertheless, contra Walsh (2021), we contend that the proper apportioning and disentanglement of causal contributions in protracted interactions between organisms and environments is something important and feasible in the study of complex evolutionary scenarios.
We will now introduce a model that aims at solving these two long-standing problems of organism-environment reciprocal causation. It allows distinguishing between organisms and environments in all of the types of NC described in current taxonomies. To do so, this model does not develop a particular theory of biological boundaries.Footnote 5 However, it implies rejecting the general idea that boundaries between organisms and environments collapse due to reciprocal interactions over developmental and evolutionary time. In fact, it makes possible maintaining epistemically set boundaries between the two even in complex reciprocal evolutionary dynamics. In addition, it not only countenances distinguishing different types of experiential and physical NC, but provides a shared conceptual and visual framework that makes it possible to relate and integrate the two.
Unknotting reciprocal causation in physical niche construction
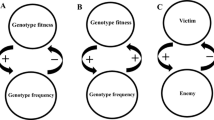
Previous conceptualizations of the relationship between organisms and environments have been strongly driven by visual approaches, especially by diagrams representing their cyclical connection (see, e.g., Fig. 1; see also Di Paolo 2020). We tie in with this tradition, as visual representations can indeed play powerful epistemic and heuristic roles. They allow organizing and guiding scientific and philosophical reasoning by supporting the articulation of novel concepts, pointing to relations and anomalies not expressed verbally in theories, and encouraging the clarification of implicit assumptions. In addition, they can guide scientific practice by coordinating methodological strategies and facilitating formalization (Waddington 1977; Griesemer 1991; Baedke and Schöttler 2017).
Despite these general advantages, past and present visual models of the relationship between organisms and their environments typically show some limitations. For example, in line with the long-standing problems discussed above, they usually depict organism and environment as partaking in a seemingly inextricable reciprocal loop (see Fig. 2a; see also Levis and Pfennig 2017; Laland et al. 2017; Di Paolo 2020). Therefore, a first step towards unknotting organism-environment reciprocal interactions would require distinguishing between the two components. This means that some causal processes occurring in the organism are relatively autonomous from the environment, and vice versa (see Toepfer 2012). Thus, in addition to causal pathways connecting organism and environment, we have to incorporate others that start and end within the limits of the organism and within the limits of the environment (see Fig. 2b). The resulting model of reciprocal causation, nevertheless, is still conceptually intractable as such depictions of loops cannot convey an understanding of how causal processes in and between organism and environment occur over time. A second unknotting step, then, consists of unrolling the organism-environment cycle to make explicit the sequential character in which reciprocal causation occurs (see Fig. 2c). In that sense, if we epistemically grant that organism and environment are distinct, we can articulate the complex causal relations between them as well as how they are modified through each component and unfold over life histories.
We propose a refined version of this ‘open-loop’ model to represent organism-environment reciprocity (see Fig. 2d). Its components are two ordered series of states of the organism and the environment (O and E, respectively), and arrows representing causal processes between their states, as well as between successive states of the organism and successive states of the environment.Footnote 6 For instance, the interaction between organism and environment at state n (symbolized as On and En) causally contributes to the next state of the organism, the environment, or both (On+1 and En+1). Accordingly, the diagram includes an arrow from En to On+1, one from On to En+1, or both. If we represent all the possible causal relations, we obtain a basic unknotted causal model of organism-environment reciprocity (see Fig. 2d).Footnote 7 The causal relations between, say, an organism state and a successive environment state can be conceptualized as being invariant under a range of counterfactual interventions on the organism state. In short, an arrow connecting two states of organism and environment indicates that these states are causally related in an interventionist sense (Woodward 2003).
This kind of diagram is rather unilluminating unless we weigh the arrows according to their relevance for a particular causal explanation. This process leads to the identification of certain invariant causal paths that connect particular changes in states of the organism and the environment. In this view, some arrows (but not others) represent meaningful explanantia addressing regularly occurring causal couplings of organisms and environments. The precise interpretation of the states and relations depends on the causal narrative a scientist is putting forward, given a particular research case. It should be kept in mind that highlighting certain pathways (i.e. including them in an explanans) does not imply that non-highlighted causal arrows do not exist, but instead that we can (to some degree) abstract from their causal effects. Thus, the totality of causal relations is always at play.
Unknotting organism-environment reciprocal causation. a Organism (O) and environment (E) partake in a loop of reciprocal interaction. b Additional loops represent intrinsic causal processes in the organism and environment. c Organism-environment interactions as in (b) but depicted in a sequential manner. d Model of organism-environment reciprocal causation with a succession of states. Subscripts indicate different states of the organism and the environment, and arrows represent causal contributions. e Application of the model (d) to the case of physical niche construction in reef-building corals. The highlighted arrows and states of corals (O) and their environment (E) constitute a causal path that matches a sequence of steps (1–3) in a causal narrative. For details, see text
Let us now illustrate this visual and conceptual model of reciprocal causation by applying it to a well-known case of external/physical NC (sensu Chiu 2019; Aaby and Ramsey 2019). Reef-building corals dramatically alter the physical and chemical conditions of their environments mainly by secreting calcium carbonate skeletons. These accumulate and form complex structures that constitute habitats for hundreds of other species (Jones et al. 1994). These environmental changes have reciprocal effects on corals. For instance, some macroalgae and sponges that settle in coral reefs compete with corals for space, light, or food resources (see Sultan 2015 and references therein). This NC case is represented in Fig. 2e. The causal narrative underlying the explanation of physical NC in these corals is highlighted in the figure (sequence 1–3). Corals (On−1) in a given environment (En−1) secrete calcium carbonate. This (1) causes changes in the species composition of the environment, which in the next state (En) includes some species that newly compete with corals (On), thus having (2) causal downstream effects on corals’ survival, development, or reproduction, i.e., the next state of the corals (On+1). Corals, in turn, may respond to these environmental changes by (3) modifying the environment in a new round of NC. This simplified narrative could be enriched by adding intermediate steps within the causal path. Depending on the case under study, the sequence could also start with an environmental change that alters the organism, with that organism affecting again the environment at a later time point.
In the next section, we will explore how our model can be expanded to incorporate the causal processes of experiential NC. This extension will be crucial to tackle the second problem of reciprocity theories, namely how to integrate physical and experiential NC.
Experienced environments and niche construction
In recent years, there have been increasing efforts to show that experienced environments and experiential NC do not, in fact, necessarily open the door to spurious, subjectivist, anti-realist and insoluble holistic views on the organism-environment relation (Walsh 2015, 2021; Sultan 2015; Chiu 2019). In addition, outside the NCT literature there exists a large body of fruitful ecological projects that study, for example, how landscape features drive avoidance behaviors through experiences of fear and disgust (Sheriff and Thaler 2014; Gaynor et al. 2019). While these developments suggest that problems of integrating physical and experiential reciprocal processes between organism and environment might not be as widespread in ecology as they are in evolutionary biology, unfortunately, these new conceptual frameworks have not yet exerted influence on evolutionary methodologies. For instance, it remains largely unclear how shifts in organisms’ experiences can change selection pressures acting on them or establish individualized niches (but see Sultan 2015; Chiu and Gilbert 2020; Müller et al. 2020). We believe this is not least due to the fact that advocates of experiential NC often endorse a view of organism-environment inseparability (Levins and Lewontin 1985; Sultan 2015; Walsh 2015, 2021), which can make methodological implementation difficult.
We try to bridge this gap between conceptual frameworks of experiential NC, on the one hand, and scientific practice, on the other, by integrating experienced environments into the above model–however, without merging the whole environment with the organism. In our model, the ‘experienced environment,’ variable Ex (see Fig. 3a), represents a mediating interface between organism and physical environment. It constitutes the sum of environmental cues (temperature, pressure, location, etc.) that can causally affect this interface and thus the organism.Footnote 8Ex is meant to convey four basic ideas. First, what is a cue depends on the organism’s sensory system and the way the organism modulates its behavior to choose certain environmental factors (see Sultan 2015). Second, experienced cues are transduced into chemical and cellular processes (which regulate, e.g., gene expression patterns or microbiome composition), and finally lead to metabolic, morphological or behavioral changes. Third, a difference in Ex between two organisms living in the same environment E means that E is experienced differently by each organism (e.g., as favorable or unfavorable, as stressful or non-stressful). Individual experiences are then directly linked to the ecological performance of these organisms in E, and hence affect their distribution and potentially their evolutionary trajectories. Finally, and most importantly, a change in Ex means a change in the relation of the organism to its physical environment, without alterations of the intrinsic properties of the external environment (Chiu 2019).Footnote 9
Let us now discuss how this expanded model represents different types of experiential NC, i.e. constitutive, mediational, relational and relocational NC.Footnote 10 Organisms that conduct constitutive NC actively construct the experiences of their environments through their plastic behavior and development. Aaby and Ramsey (2019) exemplify constitutive NC with a lion (On−1) that changes its size, strength, and coordination as it develops (see Fig. 3b, sequence 1–3). Through this maturation and changes in bodily constitution (1), the experienced space of possible prey (Exn) is transformed (2). The ingestion of larger prey then affects the constitution of the lion (3), which has ecological downstream effects. Aaby and Ramsey (2019:11) assert that “the development of the lion is thus partly responsible for the construction of its niche.” In short, experienced environments are in part created by intrinsic developmental processes.
Different from that, mediational NC describes changes in the impact and significance of the environment for the organism that are due to novel ways of how the environment is experienced, but without physical modifications of it (see Chiu 2019). Mediational NC can happen within or across generations, but all instances show the same causal pattern (see Fig. 3c, sequence 1–3).Footnote 11 An example of mediational NC is nutrient foraging in Arabidopsis thaliana (see Giehl and von Wirén 2014; Sultan 2015). Plants are able to respond to nutrient shortage or localized nutrient availability by altering their root system architecture to efficiently explore certain zones of the soil that contain limited resources. As Sultan (2015:80) notes: “plants are able to experience an environment that is consistently high in nutrients despite this resource patchiness because of the developmental and physiological plasticity of root systems.” Low levels of nutrients in the soil (En−1) produce a shift in the way the plant (On−1) experiences the environment (Exn−1), i.e. as unfavorable (1). This causes developmental and physiological changes in the plant, i.e. primary or lateral root elongation (2), which then affect how the plant experiences its environment, i.e. as nutrient-rich (3). This experience can then stop further physiological changes in the plant (4).
Experienced environments and niche construction (I). a Basic model of reciprocal causation between organism (O) and environment (E) including experienced environment (Ex). b Application of the model to an example of constitutive NC, in which the development of a lion (O) changes its experience (Ex) of potential prey in its environment (E). c Example of mediational NC, where the experience (Ex) of low levels of nutrients in the soil (E) causes physiological and developmental changes in a plant (O), which subsequently change the plant’s experience. For the numerated sequence of steps along these paths, see text
Yet another causal pattern comes to light in the case of relational NC (Aaby and Ramsey 2019), where organism-organism interaction mediates changes in the experienced environment, without organisms modifying their constitution or their environment. These biotic interactions can be represented as two interlinked sequences of reciprocal organism-environment interactions (see Fig. 4a, sequence 1–3). One example is mice that pile up to keep warm (Aaby and Ramsey 2019). If their nest gets cold, mice (On−1 and O’n−1) physically interact with one another, which affects the rates of heat loss just by existing in physical proximity (1). This causes mice to experience their environment (Exn and Ex’n) as warmer than it actually is (2), as this behavior does not actually change the physical temperature of the nest.Footnote 12 The individual experience of mice then again modulates their behavior to continue or stop piling up (3).
Finally, our model can represent relocational NC as a break in the continuity of the sequence of environmental states. This occurs during an experience-mediated transition of the organism from one physical environment to a new one. One example is the migration of anadromous fish from the ocean to rivers to spawn (Fig. 4b, sequence 1–5). Migrating behavior (On) can be triggered by fish’s experience (Exn−1) of certain environmental conditions in the ocean (En−1), like population density, food availability or climatic variability (1). This exposes the fish to a series of transitional environments (Eτ) during migration (2), the experience of which (Exτ) continuously modulates its behavior (3). Finally, after continuous migration (4), the experience (Exn+1) of a favorable environment, a certain river (E’n+1), triggers a change in fish’s behavior, which stops migrating and chooses that environment to spawn (5).
Experienced environments and niche construction (II). a Example of relational NC. In a cold nest, mice (O and O’) pile up to change the experience of the temperature of their environment (Ex and Ex’), but not the actual temperature in the nest (E and E’). b Example of relocational NC. Environmental cues (E) cause migration of anadromous fish from the ocean to rivers, during which fish experience (Exτ) different transitional environments (Eτ), out of which the organism chooses one (E’) in which it spawns. See details in the text
This model has several advantages. First, as the above examples show, it allows representing highly different forms of experiential NC in a single conceptual and visual framework. It makes it possible to identify these forms through their characteristic causal patterns, which emerge from particular reciprocal organism-environment interactions. Second, this model does not need to merge organism’s physical and experienced environments, but can keep them conceptually distinct. It recognizes that there are cases of physical NC that are not experiential NC, and vice versa.Footnote 13 In other words, it avoids the persistent stance that reduces all ecologically and evolutionary relevant physical factors to individual experienced environments. As an alternative, we hold that there are instances of relevant physical effects on the organism that are not directly experienced by it. For example, some wavelengths of radiation cannot be experienced by most organisms but can cause evolutionary-relevant genetic mutations in them. In the case of many mammals, ionizing radiation affects DNA directly, without receptor or sensory mediation, which can have consequences in their evolutionary trajectories (e.g., Adewoye et al. 2015; Kesäniemi et al. 2020).Footnote 14
Third, and related to the previous point, this model avoids an inflationary construal of organismic experience, in which every single influence on the organism causing some internal reaction is mediated by inbuilt sensory filters. Instead, it presupposes that Ex shows a sufficient degree of sensory specificity (which evolved biologically or was acquired through biological or cultural transmission) for detecting particular changes in the environment in such a way that the organism can react functionally to them in a (minimally) directed manner. Such reactions range from purposeful behavioral responses to environmentally-induced developmental biases. Finally, this model allows clearly distinguishing not only physical and experienced environments, but the causal roles different types of experiential and physical NC play as well. Even more importantly, as we show below, it can integrate experiential and physical NC in a common framework applicable to complex empirical scenarios.
Integrating different types of niche construction
Our visual and conceptual model can identify the precise causal patterns which are characteristic of each type of NC identified by current taxonomies. This allows drawing clear-cut distinctions between different kinds of causal narratives about NC processes. However, each of these narratives on their own have limitations when applied on more complex phenomena, in which two or more organisms (from different species) interact in reciprocal ways that include more than one type of NC. In such cases, the present taxonomies do not provide clear guidelines on how to integrate different NC processes, especially if they include both physical and experiential forms of NC. We hold that our model serves as a tool for relating and integrating different kinds of NC. This enables addressing more comprehensive causal explanations of complex reciprocal organism-environment interactions, which are the norm rather than the exception in ecological and evolutionary scenarios. We illustrate this with two examples.
Our first example is the acceleration of flower production in Solanum melongena (eggplant) as a consequence of active leaf damage by Bombus terrestris bumblebees (Pashalidou et al. 2020). When faced with a shortage of pollen, bumblebee workers actively damage the leaves of flowerless plants, which accelerates flower production. In this way, bumblebees increase the local availability of their nutritional resources (see Fig. 5a, sequence 1–7). A shortage of pollen in the environment of the bumblebees (En−2) is experienced (Exn−2) by them (On−2) as nutrient scarcity (1). This modifies the behavior of bumblebees (2), which start damaging the leaves of eggplants (3). The damaged plants (O’n) experience their environment as threatening (4) and thus alter their constitution by allocating resources to the production of flowers (5). Here, we see two different physiological processes at play, with distinct locations in the plant and different functions: plant experience refers to a particular sensory-cue interface which detects with sufficient specificity damage to leaves, and plant constitution refers to the morphogenetic processes that lead to flower production in meristems. The availability of flowers, in turn, alters the behavior of bumblebees, which cease damaging the plants and start collecting pollen (6). Further reciprocal interactions between bumblebees and plants could then continue (7).
This case can be framed as physical NC: bumblebees actively modify their environment (which includes plants) and the consequences of their activity reciprocally impact them. But it is important to note that also experience is included here as a key component, contrary to the way physical/external NC has traditionally been conceptualized. In addition, and more importantly, this case of reciprocal interaction is only possible by virtue of the NC capacities of plants. Indeed, from the perspective of the eggplant, this case is an instance of mediational NC, whereby plants accelerate flower production due to a change in their experienced environment, without altering intrinsic features of the environment. In short, a proper understanding of this case requires the integration of two kinds of NC and of experience into physical NC within one causal explanation. Our model allows achieving such integrations.
We now move to a more challenging example: the transition to herbivory in ruminants and the role symbiotic microbes play in this evolutionary shift (Chiu and Gilbert 2020). This example involves two processes. On the one hand, microbes colonize the digestive system of the animal and help develop the rumen, which is the organ that houses them and allows them to function. On the other hand, these developmental changes cause a dietary shift to herbivory in the animal, which now perceives plants as edible. According to Chiu and Gilbert (2020), the former is an instance of physical NC (though they label it as ‘perturbational,’ following Odling-Smee et al. 2003), whereby microbes help construct their own environment–the rumen. The latter is an instance of mediational NC, i.e. a change in the animal’s experience of its environment without physically changing it. Further, Chiu and Gilbert state that (a) ruminants also engage in physical NC by contributing to the development of their microbes’ niche, and that (b) the holobiont as a whole (i.e. the ruminant host plus its microbiome), and not just the animal, is the unit that engages in mediational NC. Taken together, these statements are problematic, since (a) implies that microbes and host are separate units, each partaking in the environment of the other, whereas (b) assumes that both the host and its microbiome behave as one unit that shares an experience of the environment.
Let us represent the case by means of our model (Fig. 5b, sequence 1–6). We consider the rumen microbes, taken as a collective, and the animal host as distinct organisms (O’ and O, respectively). The rumen is the microbiome’s environment (E’) and the host’s environment is its external environment (E). After colonizing the rumen during birth, the microbes (O’n−2) proliferate and release compounds (particularly butyrate) that cause the growth and differentiation of this organ (1). The altered rumen (E’n−1) provides a suitable environment for further microbial proliferation and diversification of the microbial community (2). The development of the microbial community modifies the constitution of the animal (3) and this, in turn, leads the animal to start perceiving the plants in its environment as edible and digestible (4). The dietary shift of the animal alters its development (5) and, consequently, reacts upon the rumen microbiome’s composition and diversity (6). Further downstream reciprocal processes would, for instance, include microbes continuing to mold the rumen and causally contribute to the animal’s development by producing cellulose-digesting enzymes that allow the host to digest plant material, or by neutralizing plant defense chemicals that would otherwise be toxic for the host (Chiu and Gilbert 2020).
Integrating different kinds of niche construction. a Causal diagram of the acceleration of flower production in Solanum melongena (O’) as a consequence of active leaf damage by Bombus terrestris bumblebees (O). Bumblebees’ behavior is triggered by the perceived (Ex) shortage of pollen in the environment (E). Flower production in plants is mediated by their perception (Ex’) of their environment (E’) as threatening. In this case, both organisms are part of each other’s (partly overlapping) environments that causally affect them. b Causal diagram of the transition to herbivory in ruminants (O) in a given environment (E). This transition is partly explained by changes in the rumen (E’) driven by microbes (O’), which cause the animal to experience plants as edible (Ex). For details, see text
What we can learn from the application of our model to this complex case is that we can trace these different but entangled processes of NC only by considering the microbes as organisms distinct from their host and by studying the reciprocal interactions of these components with their unique environments individually. If, on the contrary, we consider the holobiont as an indivisible whole, we would not be allowed to identify the NC activities of the microbes, simply because NC is a dialectical relation between organisms and environments, and we would only be left with the environment of the holobiont (not of microbes). Consequently, embracing the ‘holobiont-as-individual’ view would imply collapsing O’ into O, thus reducing our diagram to steps 3–6. The result of this operation would be indistinguishable from a case of constitutive NC (see, e.g., the causal pattern in the development of lions; Fig. 3b).
To sum up, these examples show that our model allows for the conceptual and visual integration of different kinds of physical and experiential NC. The model provides a consistent framework that can relate and compare causal explanations of organism-environment reciprocity in multi-species interaction networks, which are more complex and heterogeneous than those described by current NC taxonomies. More generally, it calls attention to implicit assumptions and overlooked questions in NCT. For instance, regarding the second example, we may ask what exactly an environment is for a holobiont, and what causal role the microbial environment plays in the host’s environment. Also, what does it tell us to see similar causal patterns arising on the level of individual organisms (e.g., constitutive NC) compared to those in integrated collectives, where causal roles and phases of the whole causal path are distributed across different organisms? In the final section, we discuss the heuristic roles of our model for scientific research.
Conclusions and outlook
In this paper we identified long-standing problems about reciprocal causation in evolutionary theory, and offered solutions to them. We first showed that, contrary to what is assumed in current evolutionary debates, theoretical viewpoints that argue for organism-environment reciprocal causation have a long pedigree in the history of evolutionary thought, especially in early twentieth-century. We then recounted how accounts of organism-centered reciprocity were marginalized in the second half of the twentieth century, and how the study of reciprocity between other causal relata (e.g., gene-environment, gene-population) gained the upper hand. This shift was accompanied by a consistent avoidance of the concept of ‘individual environments’ and the establishment of externalism as an important explanatory principle in evolutionary biology.
The decline of traditional approaches of organism-environment reciprocity was also related to two, still today unsolved theoretical problems in these views: (i) Meaningful boundaries between organism and environment were often blurred, which leads to each component’s intractability for empirical studies, and (ii) there was little theoretical integration of experiential and physical views of the environment. The latter prevented the clarification of the manifold causal consequences that take place through reciprocal organism-environment interactions, as it cannot spell out the exact causal links between organism-driven changes in experienced and physical environments. We showed that these problems are re-emerging in today’s debate on reciprocal causation in ecological and evolutionary processes. It is important to mention that their reappearance is not restricted to new approaches that combine ideas from developmental systems theory and NCT. Instead, more generally, these problems result from certain views of causal reciprocity (wherein organisms are merged with their environments and all physical interactions are understood as mediated through experience) that are present in a number of evolutionary frameworks.
In addition, we presented a conceptual and visual model that is able to tackle the aforementioned problems. Instead of scattering causes and effects across inextricable organism-environment systems, we showed that the organism-environment boundary is an epistemic necessity to understand the complex causality and causal contributions of each component in reciprocal NC processes. Our model unknots seemingly inextricable reciprocal causation by underscoring the sequential character of these interactions. Then, it clarifies the kind of interactions by identifying characteristic causal patterns in sequences of organism-environment relations. This allows mapping all types of reciprocal causation currently identified in different NC taxonomies (external or physical, constitutive, mediational, relational and relocational NC) onto one common framework. In turn, this makes possible to identify and distinguish the causal patterns different kinds of reciprocal processes play in ecology and evolution. Most importantly, with respect to the above two problems, (i) our model does not require the assumption that reciprocally interacting organisms and environments are inseparable. It allows distinguishing between organisms and environments by clarifying each component’s specific causal role in their mutual interactions. In addition, (ii) it can integrate cases of organisms’ (experience and) experiential NC with those of physical NC. This allows applying the present framework even to highly complex empirical scenarios, in which both processes work together. It also demystifies the role of organismic experience in ecology and evolution.
We have not attempted to provide a metaphysical account of reciprocal causation, but an epistemic tool that can partition causal contributions of organisms and environments. This should allow biologists to develop conceptually well-grounded and feasible methodologies for probing into complex evolutionary settings. We contend that no matter what one’s ontological position regarding the nature of the organism-environment relationship is, one needs to provide an answer on how to translate this position into scientific practice. We addressed this challenge not by drawing organism-environment boundaries per se, but by tracing and distinguishing different kinds of relations between organism and environment and to apportion their causal contributions. This makes it possible to theoretically sustain epistemically set boundaries even in complex reciprocal evolutionary dynamics, in which other approaches would rather drop the idea of boundaries altogether (i.e., cases in which, e.g., many organisms from different species and their different experiences and environments are involved).
Besides these conceptual and theoretical advantages, our model may also play important heuristic roles in experimental research on NC. By building on (and being compatible with) interventionist views of causation (Woodward 2003), it allows the clear identification of causal paths and relevant counterfactual dependencies between organisms and environments, which could help designing experimental setups and selecting suitable variables to intervene on. Of course, more fine-grained manipulations or simulations would require formalization and mathematical modeling. Recently, several mathematical models have been proposed to address reciprocal interactions between organisms and environments, and some of them explicitly deal with NC (e.g., Torres et al. 2009). Following Lewontin’s (1983) early proposal, some formalizations represent organism-environment interaction by means of systems of coupled differential equations (e.g Gurney and Lawton 1996; Krakauer et al. 2009; Tanaka et al. 2020), while others incorporate elements of game theory (Fort 2020) or make use of causal graph theory (e.g., Ay and Löhr 2015; Otsuka 2015). By drawing on the latter set of approaches as well as interventionist interpretations of causality in path analysis (Pearl 2009), we will explore the formalization of our conceptual model elsewhere. In fact, the diagrams presented in this paper can be seen as directed acyclic graphs, whereby the states of the organism and the environment are vertices and the causal relations are edges.
For now, we ask philosophers of biology and evolutionary biologists to become aware of (and finally overcome) long-standing problematic assumptions about reciprocal causation between organisms and environments. These relations can, in fact, be conceptually clarified and investigated. Therefore, we need to unknot the seemingly inextricable bundle between organisms and environments, and integrate organismic experience more seriously to understand how organisms shape their ecological contexts as well as their evolutionary trajectories.
Notes
In a similar manner, American pragmatists like John Dewey defended a view of organism-environment inseparability (Pearce 2014, 2020). They often adopted an ontological reading where organism and environment could not be disentangled, unless abstracted. For example, Dewey claimed in his ‘The Reflex Arc Concept in Psychology’ that stimulus and response are not separate entities (see Pearce 2020:180). In this paper we do not discuss this kind of ontological co-constitution between organism and environment (what Pearce labels the ‘dual aspects view’), but reciprocal causation.
We focus on the works of Aaby and Ramsey (2019) and Chiu (2019). Both can be seen as conceptual updates to the original taxonomy of NC types by Odling-Smee et al. (2003:44–45). We should stress, however, that outside of NCT and its taxonomies, there exists a broader trend to study organism-environment reciprocity in evolutionary biology and ecology (from ecosystem engineering and habitat choice, to developmental plasticity and active sensory mediation of environmental cues).
Below we do not aim at solving cases of synchronic inseparability, i.e. ontological co-constitution. Rather, within a Humean view of causation (in which effects follow causes over time), we address cases of NC in which diachronic organism-environment relations are considered to be inextricable. We take these problems to be the cornerstone of current explanatory efforts in evolutionary biology that link constructive behaviors and environmental properties, rather than the metaphysical thesis of co-constitution.
We understand integration of experiential and physical NC to depend, first, on identifying and distinguishing the two (rather than substituting one by the other), and, second, on providing clear guidance and criteria on how both types of NC work together and are linked in complex evolutionary scenarios.
Although we are not trying to define a particular boundary between organisms and environments, our model offers a way of demarcating biological individuals from their surroundings by clarifying the causal structure of each interacting component. For a similar strategy, see Sterner (2017).
Please note that although time is implicit in these causal arrows, a series of states should not be interpreted as a series of regular time intervals, since the time scale at which organismic and environmental processes occur might vary within one series.
Krakauer et al. (2020) recently proposed a similar diagram, but with the different aim of identifying biological individuals as quantitative patterns of information flow. For a different diagram of organism-environment relationships, see Ay and Löhr (2015). In addition, our causal diagrams should not be understood as representing inter-level relations as those traced in neo-mechanistic approaches of explanation. In more complex scenarios of NC (see Figs. 3–5 below) it is neither clear what is a higher or lower level nor whether experience qualifies as a level of organization at all.
In some organisms, environmental cues are compared with internal organismic cues (e.g., body growth, change in hormone levels, hunger, etc.) to adjust physiological or behavioral responses.
In many cases of experiential NC, however, there are also causal downstream effects on the physical environment.
For reasons of simplicity, in the examples discussed in this section we do not consider (and abstract from) direct physical effects on the organism that are not mediated through organisms’ experience (but see Fig. 5b below).
An example of transgenerational mediational NC is the case of pregnant meadow voles discussed above. Here a change in day length during pregnancy leads to pups with thicker coats that are born in autumn, which again affects how they experience their environment.
Note that in this example the environments Ex and Ex’ are the same.
Walsh (2015, 2021) has defended the view that every instance of physical NC is one of experiential NC (but not vice versa). For his theory that relies on the notion of affordances, all changes in the physical environment are experienced in some way or another by organisms. Affordances emerge from organism-environment systems as a whole, providing organisms different ‘graspable tools’ to react to (for discussions on the evolutionary roles of affordances in NCT, see Heras-Escribano 2020).
Additionally, in the history of life some physical properties of the environment emerged before becoming experienced cues of organisms. For example, in the global NC process in which cyanobacteria made oxygen accumulate in the ocean/atmosphere (as a byproduct of photosynthesis), oxygen was not immediately an experienced cue for all life forms present at that time.
References
Aaby BH, Ramsey G (2019) Three kinds of niche construction. Br J Philos. https://doi.org/10.1093/bjps/axz054
Adewoye AB, Lindsay SJ, Dubrova YE et al (2015) The genome-wide effects of ionizing radiation on mutation induction in the mammalian germline. Nat Commun 6:6684. https://doi.org/10.1038/ncomms7684
Antonovics J, Ellstrand NC, Brandon RN (1988) Genetic variation and environmental variation: expectations and experiments. In: Gottlieb LD, Jain SK (eds) Plant evolutionary biology. Springer, Netherlands, Dordrecht, pp 275–303
Ay N, Löhr W (2015) The Umwelt of an embodied agent—a measure-theoretic definition. Theory Biosci 134(3):105–116
Baedke J (2019) O organism, where art thou? Old and new challenges for organism-centered biology. J Hist Biol 52(2):293–324
Baedke J, Schöttler T (2017) Visual metaphors in the sciences: the case of epigenetic landscape images. J Gen Philos Sci 48(2):173–194
Baedke J, Fábregas-Tejeda A, Vergara-Silva F (2020a) Does the extended evolutionary synthesis entail extended explanatory power? Biol Philos 35(1):20. https://doi.org/10.1007/s10539-020-9736-5
Baedke J, Fábregas-Tejeda A, Nieves Delgado A (2020b) The holobiont concept before Margulis. J Exp Zool B Mol Dev Evol 334(3):149–155
Baedke J, Gilbert SF (2020) Evolution and development. In: Zalta EN (ed) The Stanford encyclopedia of philosophy, Fall 2020. Metaphysics Research Lab, Stanford University. https://stanford.library.sydney.edu.au/archives/fall2020/entries/evolution-development/
Bierens de Haan JA (1947) Animal psychology and the science of animal behaviour. Behaviour 1(1):71–80
Brandon RN (1990) Adaptation and environment. Princeton University Press, Princeton
Brandon RN (2012) The concept of the environment in evolutionary theory. In: Kabasenche WP, O’Rourke M, Slater MH (eds) The environment: philosophy, science, and ethics. MIT Press, Cambridge, MA, pp 19–35
Brentari C (2015) Jakob von Uexküll: the discovery of the Umwelt between biosemiotics and theoretical biology. Springer, Dordrecht
Brodie ED III (2005) Caution: niche construction ahead. Evolution 59(1):249–251
Buskell A (2019) Reciprocal causation and the extended evolutionary synthesis. Biol Theory 14(4):267–279
Charlesworth B (1971) Selection in density-regulated populations. Ecology 52(3):469–474
Chiu L (2019) Decoupling, commingling, and the evolutionary significance of experiential niche construction. In: Uller T, Laland KN (eds) Evolutionary causation: biological and philosophical reflections. MIT Press, Cambridge, MA, pp 299–322
Chiu L, Gilbert SF (2015) The birth of the holobiont: multi-species birthing through mutual scaffolding and niche construction. Biosemiotics 8(2):191–210
Chiu L, Gilbert SF (2020) Niche construction and the transition to herbivory: phenotype switching and the organization of new nutritional modes. In: Levine H, Jolly MK, Kulkarni P, Nanjundiah V (eds) Phenotypic switching. Academic Press, Cambridge, MA, pp 459–482
Di Paolo EA (2020) Picturing organisms and their environments: interaction, transaction, and constitution loops. Front Psychol 11:1912. https://doi.org/10.3389/fpsyg.2020.01912
Dickins TE, Barton RA (2013) Reciprocal causation and the proximate–ultimate distinction. Biol Philos 28(5):747–756
Fábregas-Tejeda A, Vergara-Silva F (2018a) Hierarchy theory of evolution and the extended evolutionary synthesis: some epistemic bridges, some conceptual rifts. Evol Biol 45(2):127–139
Fábregas-Tejeda A, Vergara-Silva F (2018b) The emerging structure of the extended evolutionary synthesis: where does Evo-Devo fit in? Theory Biosci 137(2):169–184
Falconer DS (1952) The problem of environment and selection. Am Nat 86(830):293–298
Falconer DS (1960) Introduction to quantitative genetics. Ronald Press Company, New York
Fisher RA (1930) The genetical theory of natural selection. Clarendon Press, Oxford
Fort H (2020) Combining niche and game theories to address interspecific cooperation in ecological communities. Community Ecol 21(1):13–24
Gaynor KM, Brown JS, Middleton AD, Power ME, Brashares JS (2019) Landscapes of fear: spatial patterns of risk perception and response. Trends Ecol Evol 34:355–368
Giehl RFH, von Wirén N (2014) Root nutrient foraging. Plant Physiol 166(2):509–517
Godfrey-Smith P (1996) Complexity and the function of mind in nature. Cambridge University Press, Cambridge
Godfrey-Smith P (2001) On the status and explanatory structure of developmental systems theory. In: Oyama S, Griffiths P, Gray RD (eds) Cycles of contingency. MIT Press, Cambridge, MA, pp 283–297
Griesemer JR (1991) Must scientific diagrams be eliminable? The case of path analysis. Biol Philos 6(2):155–180
Griffiths PE, Gray RD (1994) Developmental systems and evolutionary explanation. J Philos 91(6):277–304
Griffiths P, Gray R (2001) Darwinism and developmental systems. In: Oyama S, Griffiths P, Gray R (eds) Cycles of contingency: developmental systems and evolution. MIT Press, Cambridge, MA, pp 195–218
Gurney WSC, Lawton JH (1996) The population dynamics of ecosystem engineers. Oikos 76(2):273–283
Haeckel E (1866) Generelle Morphologie der Organismen, 2 vols. Reimer, Berlin
Haldane JS (1884) Life and mechanism. Mind 9(33):27–47
Haldane JS (1935) The physiology of Descartes and its modern developments. Acta Biotheor 1(1):5–16
Haldane JBS (1936) Some principles of causal analysis in genetics. Erkenntnis 6:346–357
Haldane JBS (1946) The interaction of nature and nurture. Ann Eugen 13(1):197–205
Hartmann N (1950) Philosophie der Natur: abriss der speziellen Kategorienlehre. De Gruyter, Berlin
Henderson LJ (1913) The fitness of the environment: an inquiry into the biological significance of the properties of matter. Macmillan, New York
Heras-Escribano M (2020) The evolutionary role of affordances: ecological psychology, niche construction, and natural selection. Biol Philos 35(2):30. https://doi.org/10.1007/s10539-020-09747-1
Jones CG, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 69(3):373–386
Kesäniemi J, Lavrinienko A, Tukalenko E et al (2020) Exposure to environmental radionuclides alters mitochondrial DNA maintenance in a wild rodent. Evol Ecol 34:163–174
Kirkpatrick M (1982) Sexual selection and the evolution of female choice. Evolution 36(1):1–12
Krakauer DC, Page KM, Erwin DH (2009) Diversity, dilemmas, and monopolies of niche construction. Am Nat 173(1):26–40
Krakauer D, Bertschinger N, Olbrich E, Flack JC, Ay N (2020) The information theory of individuality. Theory Biosci 139(2):209–223
Labouvie EW (1974) Developmental causal structures of organism-environment interactions. Hum Dev 17(6):444–452
Laland K, Brown G (2018) The social construction of human nature. In: Lewens T, Hannon E (eds) Why we disagree about human nature. Oxford University Press, Oxford, pp 127–144
Laland KN, O’Brien MJ (2011) Cultural niche construction: an introduction. Biol Theory 6(3):191–202
Laland KN, Sterelny K, Odling-Smee J, Hoppitt W, Uller T (2011) Cause and effect in biology revisited: is mayr’s proximate-ultimate dichotomy still useful? Science 334(6062):1512–1516
Laland KN, Odling-Smee J, Hoppitt W, Uller T (2013) More on how and why: cause and effect in biology revisited. Biol Philos 28(5):719–745
Laland KN, Uller T, Feldman MW, Sterelny K, Müller GB, Moczek A, Jablonka E, Odling-Smee J (2015) The extended evolutionary synthesis: its structure, assumptions and predictions. Proc Royal Soc B 282(1813):20151019. https://doi.org/10.1098/rspb.2015.1019
Laland K, Matthews B, Feldman MW (2016) An introduction to niche construction theory. Evol Ecol 30(2):191–202
Laland K, Odling-Smee J, Endler J (2017) Niche construction, sources of selection and trait coevolution. Interf Focus 7(5):20160147. https://doi.org/10.1098/rsfs.2016.0147
Lerner IM (1950) Population genetics and animal improvement. Cambridge University Press, Cambridge
Levins R, Lewontin R (1985) The dialectical biologist. Harvard University Press, Cambridge, MA
Levis NA, Pfennig DW (2017) Organisms and their environment: an evolving relationship. Evolution 71(2):503–504
Lewontin RC (1983) Gene, organism, and environment. In: Bendall D (ed) Evolution: from molecules to men. Cambridge University Press, Cambridge, pp 273–285
Lewontin RC (2000) The triple helix: gene, organism, and environment. Harvard University Press, Cambridge, MA
Mesoudi A, Blanchet S, Charmantier A, Danchin É, Fogarty L, Jablonka E, Laland KN, Morgan TJH, Müller GB, Odling-Smee FJ, Pujol B (2013) Is non-genetic inheritance just a proximate mechanism? A corroboration of the extended evolutionary synthesis. Biol Theory 7(3):189–195
Meyer-Abich A (1942) Kant und das biologische Denken. Acta Biotheor 6(3):185–211
Meyer-Abich A (1943) Beiträge zur Theorie der Evolution der Organismen. I. Das typologische Grundgesetz und seine Folgerungen für Phylogenie und Entwicklungsphysiologie. Acta Biotheor 7(1):1–80
Meyer-Abich A (1964) The historico-philosophical background of the modern evolution-biology. E. J. Brill, Leiden
Mill JS (1843) A system of logic, ratiocinative and inductive, being a connected view of the principles of evidence, and the methods of scientific investigation, vol 2. John W Parker, West Strand
Moczek AP (2015) Re-evaluating the environment in developmental evolution. Front Ecol Evol 3:7. https://doi.org/10.3389/fevo.2015.00007
Müller C, Caspers BA, Gadau J, Kaiser S (2020) The power of infochemicals in mediating individualized niches. Trends Ecol Evol 35(11):981–989
Needham J (1936) Order and life. Yale University Press, Yale
Nicholson DJ (2014) The return of the organism as a fundamental explanatory concept in biology. Philos Compass 9(5):347–359
Nicholson DJ, Gawne R (2015) Neither logical empiricism nor vitalism, but organicism: what the philosophy of biology was. Hist Philos Life Sci 37(4):345–381
Odling-Smee FJ, Laland KN, Feldman MW (1996) Niche construction. Am Nat 147:641–64
Odling-Smee FJ, Laland KN, Feldman MW, Feldman MW (2003) Niche construction: the neglected process in evolution. Princeton University Press, Princeton
Otsuka J (2015) Using causal models to integrate proximate and ultimate causation. Biol Philos 30(1):19–37
Oyama S (2000) The ontogeny of information: developmental systems and evolution. Duke University Press, Durham
Pashalidou FG, Lambert H, Peybernes T, Mescher MC, Moraes CMD (2020) Bumble bees damage plant leaves and accelerate flower production when pollen is scarce. Science 368(6493):881–884
Pearce T (2014) The dialectical biologist, circa 1890: John Dewey and the Oxford Hegelians. J Hist Philos 52:747–778
Pearce T (2020) Pragmatism’s evolution: organism and environment in american philosophy. University of Chicago Press, Chicago
Pearl J (2009) Causality. Cambridge University Press, Cambridge
Pimentel D (1968) Population regulation and genetic feedback: evolution provides foundation for control of herbivore, parasite, and predator numbers in nature. Science 159(3822):1432–1437
Prenant M (1943) Biology and marxism. International Publishers, New York
Raerinne J, Baedke J (2015) Exclusions, explanations, and exceptions: on the causal and lawlike status of the competitive exclusion principle. Philos Theory Biol 7:e602 https://doi.org/10.3998/ptb.6959004.0007.002
Reznick DN (2013) A critical look at reciprocity in ecology and evolution: introduction to the symposium. Am Nat 181(S1):S1–S8
Scholl R, Pigliucci M (2015) The proximate–ultimate distinction and evolutionary developmental biology: causal irrelevance versus explanatory abstraction. Biol Philos 30(5):653–670
Schwab DB, Casasa S, Moczek AP (2019) On the reciprocally causal and constructive nature of developmental plasticity and robustness. Front Genet 9:735 https://doi.org/10.3389/fgene.2018.00735
Sheriff MJ, Thaler JS (2014) Ecophysiological effects of predation risk; an integration across disciplines. Oecologia 176:607–611
Sterelny K (2001) Niche construction, developmental systems and the extended replicator. In: Oyama S, Griffiths P, Gray RD (eds) Cycles of contingency. MIT Press, Cambridge, MA, pp 333–350
Sterner B (2017) Individuality and the control of life cycles. In: Lidgard S, Nyhart LK (eds) Biological individuality: integrating scientific, philosophical, and historical perspectives. University of Chicago Press, Chicago, pp 84–108
Sultan SE (2015) Organism and environment: ecological development, niche construction, and adaptation. Oxford University Press, Oxford
Svensson EI (2018) On reciprocal causation in the evolutionary process. Evol Biol 45(1):1–14
Tanaka MM, Godfrey-Smith P, Kerr B (2020) The dual landscape model of adaptation and niche construction. Philos Sci 87(3):478–498
Thompson JN (1998) Rapid evolution as an ecological process. Trends Ecol Evol 13(8):329–332
Tinbergen N (1963) On aims and methods of ethology. Z Tierpsychol 20(4):410–433
Toepfer G (2012) Teleology and its constitutive role for biology as the science of organized systems in nature. Stud Hist Philos Biol Biomed Sci 43(1):113–119
Torres J-L, Pérez-Maqueo O, Equihua M, Torres L (2009) Quantitative assessment of organism–environment couplings. Biol Philos 24(1):107–117
Uller T, Helanterä H (2019) Niche construction and conceptual change in evolutionary biology. Br J Philos Sci 70(2):351–375
Uller T, Laland KN (2019) Evolutionary causation: biological and philosophical reflections. MIT Press, Cambridge, MA
von Uexküll J (1909) Umwelt und Innenwelt der Tiere. Springer, Berlin
von Uexküll J (1928) Theoretische Biologie. Springer, Berlin Heidelberg, Berlin
Waddington CH (1957) The strategy of the genes: a discussion of some aspects of theoretical biology. Allen and Unwin, London
Waddington CH (1959a) Evolutionary systems–animal and human. Nature 183(4676):1634–1638
Waddington CH (1959b) Evolutionary adaptation. In: Tax S (ed) Evolution after Darwin, the University of Chicago centennial. University of Chicago Press, Chicago, pp 381–402
Waddington CH (1977) Tools for thought. Jonathan Cape, Frogmore
Walsh DM (2015) Organisms, agency, and evolution. Cambridge University Press, Cambridge
Walsh DM (2021) Environment as abstraction. Biol Theory. https://doi.org/10.1007/s13752-020-00367-2
Whitehead AN (1929) Science and the modern world. Macmillan, New York (Original: 1925)
Williams GC (1992) Gaia, nature worship and biocentric fallacies. Q Rev Biol 67(4):479–486
Woodward J (2003) Making things happen. Oxford University Press, Oxford
Wright S (1960) The treatment of reciprocal interaction, with or without lag, in path analysis. Biometrics 16(3):423–445
Wright S (1969) Evolution and the genetics of populations, vol 2: theory of gene frequencies. University of Chicago Press, Chicago
Acknowledgements
We thank Grant Ramsey, Bendik Aaby, Rose Trappes, Azita Chellappoo, and Daniel Brooks for constructive comments on earlier versions of this paper. We express gratitude to two anonymous reviewers of this journal for the comments and feedback provided. Moreover, we thank the attendees of the online workshop ‘Niche Construction and Other Mechanisms in Ecology and Evolution’ (Bielefeld University, 2020) and of a session at the Oslo Meeting of the International Society for the History, Philosophy, and Social Studies of Biology (ISHPSSB, 2019) for their useful feedback on presentations on this topic. We gratefully acknowledge the financial support from the German Research Foundation (DFG; project no. BA 5808/2-1).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Baedke, J., Fábregas-Tejeda, A. & Prieto, G.I. Unknotting reciprocal causation between organism and environment. Biol Philos 36, 48 (2021). https://doi.org/10.1007/s10539-021-09815-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10539-021-09815-0