Abstract
Purpose
Stratification of patients with triple-negative breast cancer (TNBC) for anti-PD-L1 therapy is based on PD-L1 expression in tumor biopsies. This study sought to evaluate the risk of PD-L1 misclassification.
Methods
We conducted a high-resolution analysis on ten surgical specimens of TNBC. First, we determined PD-L1 expression pattern distribution via manual segmentation and measurement of 6666 microscopic clusters of positive PD-L1 immunohistochemical staining. Then, based on these results, we generated a computer model to calculate the effect of the positive PD-L1 fraction, aggregate size, and distribution of PD-L1 positive cells on the diagnostic accuracy.
Results
Our computer-based model showed that larger aggregates of PD-L1 positive cells and smaller biopsy size were associated with higher fraction of false results (P < 0.001, P < 0.001, respectively). Additionally, our model showed a significant increase in error rate when the fraction of PD-L1 expression was close to the cut-off (error rate of 12.1%, 0.84%, and 0.65% for PD-L1 positivity of 0.5–1.5%, ≤ 0.5% ,and ≥ 1.5%, respectively, P < 0.0001). Interestingly, false positive results were significantly higher than false negative results (0.51–22.62%, with an average of 6.31% versus 0.11–11.36% with an average of 1.58% for false positive and false negative results, respectively, P < 0.05). Furthermore, heterogeneous tumors with different aggregate sizes in the same tumor, were associated with increased rate of false results in comparison to homogenous tumors (P < 0.001).
Conclusion
Our model can be used to estimate the risk of PD-L1 misclassification in biopsies, with potential implications for treatment decisions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Triple-negative breast cancer (TNBC) accounts for 15–30% of breast cancer cases and is the most aggressive type of breast cancer with high rates of distant metastases and poor survival rates [1, 2]. It is defined by the lack of expression of estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 (HER2); hence it is insensitive to endocrine treatment and targeted therapies [3].
Immunotherapy in the matter of immune checkpoint inhibitors (ICIs) such as monoclonal antibodies targeting program death-ligand 1 (PD-L1) and program cell death 1 (PD-1) have reformed the treatment for numerous cancer types and recently, the addition of Atezolizumab to chemotherapy was approved for metastatic and unresectable locally advanced TNBCs [4,5,6,7]. PD-L1 positive tumors are defined according to immunohistochemical testing by PD-L1 expression on tumor-infiltrating immune cells accounting for at least 1% of the tumor area [8, 9]. It should be noted that different therapies requires different threshold of PD-L1 positivity and evaluation of PD-L1 positivity in both immune cells and tumor cells. While pembrolizumab threshold is PD-L1 positivity of > 10% and based on combined score of both tumor and inflammatory cells, Atezolizumab threshold is PDL-1 positivity of > 1% in immune cells [10].
Analysis of PD-L1 expression may be performed on tissue samples obtained from resection or on a core needle biopsy from either primary or metastatic sites [11], and hence, a biopsy should represent the entire tumor accurately. However, there are potential limitations that may lead to inaccurate classification of PD-L1 status, such as relatively small specimens or heterogeneous expression of PD-L1 within the tumor. Our study sought to characterize PD-L1 expression in immune cells on triple-negative breast cancer tumors and, using a computer-based algorithm, to estimate the risk of PD-L1 status misclassification (false negative and false positive results) in biopsies.
Methods
We used two complementary methods to evaluate the risk for false results in the analysis of PD-L1 in TNBC. First, we examined the pattern of PD-L1 expression using tumor surgical samples from patients with TNBC. Then, based on those PD-L1 patterns we used the MATLAB software to establish virtual samples, which were further evaluated to determine the risk of false PD-L1 status classification.
Clinical samples
Tumor tissue specimens were collected retrospectively from ten patients with TNBC, who had undergone surgical resection at Tel-Aviv Sourasky Medical Center. The study protocol was approved by the local ethics committee.
Immunohistochemical staining
Freshly cut, 4 micron slides were stained using the VENTANA PD-L1 (SP142) Assay (Ventana Medical Systems, Tucson, Arizona) according to manufacturer instructions. Stainings were performed on a Ventana BenchMark Ultra immuno stainer (Ventana Medical Systems, Tucson, Arizona). Normal tonsil tissue was used as a control for each case.
Slides were scanned using the Phillips UltraFast Scanners (Philips Digital Pathology Solutions, Best, the Netherlands) to obtain high-resolution whole slide digital images.
Analysis of the clinical samples
The samples were analyzed according to VENTANA PD-L1 (SP142) assay. Thousands of stained immune cells as aggregates or single cells were manually captured on the digital slides assisted by QuPath software version 0.2.3. These detailed measurements along with total tumor area provided important information including PD-L1 expression percentage, number of stained immune cells, and the area and pattern of stained immune cells in each tumor.
Computer-based model
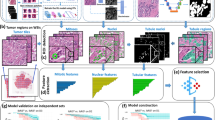
MATLAB software version R2017a was used to produce multiple matrices, where each matrix cell represented a typical immune cell, with a diameter of 10 µm, or calculated as an area of 100 µm2 as a matrix cell is a square. Each matrix represented a "tumor", with a dimension of 10 cm × 10 cm. Such a large "tumor" in the model was required for good representation of the different positive cell distribution, especially in cases with low PD-L1 positive cells fraction. An immune cell "expressing" PD-L1 protein received the value 1 whereas a cell that is negative for PD-L1 received the value 0. The "tumor" consisted of immune cells rather than tumor cells as VENTANA PD-L1 (SP142) assay calculate the proportion of tumor area that is occupied by PD-L1 staining tumor infiltrating immune cells. From each matrix or "tumor", a section was taken, to represent a "biopsy". A trial contained over a thousand "tumors" and "biopsies", which differed in the expression of PD-L1. The distribution of PD-L1 expression was based on our clinical finding and previous research [9]. 60% of cases had PD-L1 expression below 1%, approximately 20% of cases had PD-L1 expression between 1 and 5%, and 20% had PD-L1 expression of 5–20%. The model could either generate homogenous tumors, with the same aggregate or single cell sizes distributed in the entire tumor, or it could generate heterogenous tumors, with different sizes of aggregates, meaning each tumor had small and large aggregates in the same tumor, which were matched with the clinical sample findings (Fig. 1).
Simulated tumors by the computer-based model. a Simulated tumors differed by aggregates size, biopsy size and percentage of PD-L1 positivity. Left and middle—homogenous tumors, right—heterogenous tumor. Black—cells that are negative for PD-L1, white—cells that are positive for PD-L1, green—simulated biopsy, red—area magnified below. b Magnification of A
Several parameters that might lead to inaccurate results were evaluated in this model. The first is the size of the biopsy. The smaller the biopsy the less it represents the entire tumor and accordingly, may lead to increased risk for false results. The second parameter is the size of an aggregate. Aggregate represents a cluster of PDL-1 positive cells. Larger aggregates (higher number of positive PDL-1 cells adjacent to each other) might lead to more heterogeneous distribution of PD-L1 expression and hence might increase the risk for false results. Those parameters were statistically evaluated in the homogenous tumors, as each parameter could be isolated.
We ran the heterogenous modality ten times for each sample size and ran ten times each option for the homogenous modality. We received the following outputs: The percentage of expression of the PD-L1 in every tumor, the percentage of expression of the PD-L1 in every biopsy and the error rate, measured by the area under the curve. An error means discrepancies between the tumor treatment decision and the biopsy treatment decision, when the cutoff used for determining eligibility for anti-PD-L1 therapy was 1%. Additionally, false negative and false positive were measured.
A step-by-step description of the algorithm as well as the actual scripts can be observed in Supplementary material.
Statistical analysis
In the computer-based model we examined the effect of the biopsy size and the aggregate size on the error rate, using 2-sided, nonmatched t-test. P values < 0.05 were considered statistically significant. Additionally, we examined whether closer values of PD-L1 expression to the cutoff involves increase false results using Chi-square test.
Results
Clinical samples
Ten surgical samples of TNBC were analyzed in this study and a single FFPE slide from each case was stained for PD-L1. All slides were stained successfully with adequate positive and negative controls. Of the ten samples, two were positive cases and eight had low PD-L1 expression (< 1%). These cases were divided to three different groups based on PD-L1 status. Three cases had less than 0.1% of PD-L1 staining, four had 0.1–0.5% PD-L1 positivity and three cases had more than 0.75% PD-L1 expression. One case had low PD-L1 expression of 0.89% while two of them were positive with up to 10.2% of PD-L1 staining.
Intra-tumoral heterogeneity was observed upon cases examination and further analysis of aggregates' distribution showed heterogeneity of aggregates sizes in each tumor (Figs. 2, 3). Interestingly, in tumors with less than 0.1% PD-L1 expression the largest aggregate was 0.003 mm2, whereas in tumors with more than 0.5% PD-L1 expression there were aggregates reaching up to 0.473 mm2. Furthermore, in the cases with higher PD-L1 expression, although the majority of aggregates were small, the majority of PD-L1 positive immune cells came from large aggregates (Fig. 4). A further division of PD-L1 staining can be observed in supplementary Fig. 1.
Aggregates' distribution in three groups with different PD-L1 positivity status. Left—cases with less than 0.1% of PD-L1 staining, middle- cases with 0.1–0.5% of PD-L1 staining, right—cases with more than 0.75% of PD-L1 staining. a The number of aggregates per aggregate size. The circles' size represents the proportion of aggregate sizes in the entire PD-L1 positive area. b Percentage of each aggregate size in the entire PD-L1 positive area
Computer-based model
Biopsy accuracy is of paramount importance, as it guides us in choosing the most suitable treatment for TNBC. The computer-based model gave us the opportunity to examine how different parameters influence the error rate. This analysis showed us that both biopsy size and aggregate size of PD-L1 positive cells affect false results (Table 1).
In the homogenous tumors, the average error rate for aggregate size of 100 µm2, 200 µm2, 600 µm2, 1800 µm2, 4 00 µm2, and 18,000 µm2 were 1%, 1.2%, 2.1%, 3.4%, 4.7%, and 8%, respectively (P > 0.001). This finding indicate that larger aggregates would lead to higher false result rate.
The average error rate for biopsy of 1 mm2, 2 mm2, 5 mm2, 10 mm2, 20 mm2, and 50 mm2 were 6.3%, 4.7%, 3.7%. 2.7%, 1.9%, and 1.3%, respectively (P > 0.001). This finding is in acceptance with our hypothesis that larger samples would lead to more accurate results (Fig. 5).
False positive and negative results based on biopsy size and aggregate size. a False positive (left) and false negative (middle) depending on different aggregate sizes. Each line represents specific biopsy size. Table: paired t-tests between different aggregate sizes. Bold—P < 0.05, red—P < 0.01. b False positive (left) and false negative (middle) depending on different biopsy sizes. Each line represents specific aggregate size. Table: paired t-test between different biopsy sizes. Bold—P < 0.05, red—P < 0.01)
The analysis also showed that the closer the percentage of the PD-L1 expression to the cutoff value (1%), the greater the error rate. This was true across the different biopsy sizes and different aggregate sizes (Fig. 6, supplementary Fig. 2). For tumors with 0.5–1.5% positivity the error rate was 12.1% whereas the error rate for biopsies form tumors with ≤ 0.5% or ≥ 1.5% PD-L1 positivity was 0.84% and 0.65%, respectively (P < 0.0001, Chi-Square; Table 2).
In our study we noticed that false negative results were significantly lower than the false positive results as false negative ranged between 0.11 and 11.36%, with an average of 1.58% while false positive ranged between 0.51 and 22.62%, with an average of 6.31% (P < 0.05, t-test).
As demonstrated in the clinical section, in real-life samples the distribution of aggregates is not homogeneous. We, therefore, used the information from the clinical samples to generate virtual samples containing different size aggregates with different quantities, better representing what we would expect in real-life samples. In the heterogeneous model, based on the clinical samples, the average error rate for biopsies of 1 mm2, 2 mm2, 5 mm2, 10 mm2, 20 mm2, and 50 mm2 were 14.23%, 13.9%, 11.4%, 10.5%, 7.9%, and 5.9%, respectively, which is a significantly higher error rate compared to homogenous tumors (Chi-square P < 0.001).
Discussion
Immunotherapies such as monoclonal antibodies targeting PD-L1 and PD-1 are evolving and taking a significant role in cancer therapy in general and in breast cancer specifically [7, 9, 12]. However, it should be kept in mind that these treatments are not free from adverse events. These might include fatigue, pruritus, diarrhea, and rashes. Rarely, these therapies are related to death where the common causes are pneumonitis, pneumonia, sepsis, respiratory failure, and cardiovascular failure [13]. Therefore, finding the correct patients that will benefit from these therapies is essential, demonstrating the great importance of accurately classifying PD-L1 expression status in biopsies.
We identified a few factors that might lead to inaccurate evaluation of PD-L1 expression. The major factors include the sample size and the aggregates size. The effect of biopsy size on accurate representation of the whole tumor has been evaluated in many fields of medicine including breast cancer [14, 15]. For example, a previous study in a series of 300 breast cancer tissue showed a significant improvement in agreement between core needle biopsies and surgical excision biopsies as the biopsy size increased [15]. This is in accordance with our computer-based model that showed smaller biopsies are associated with greater false results. Additionally, our study showed that larger aggregates increase the risk for false results.
As demonstrated in the computer-based model, PD-L1 expression levels near the cutoff value were associated with higher error rate.
Interestingly, the computer-based model showed that the error rate was higher in the low PD-L1 expression cases (false positive results) compared to the positive cases (false negative results). Importantly, both false positive and false negative results can impair patients' treatment. False positive results can lead to overtreatment, adverse effects, and potential financial burden, while false negative results would make a patient not eligible for a potentially effective therapy. The frequency of these type of error should be evaluated in further studies.
Intra-tumoral heterogeneity was widely researched in non-small cell lung cancer and melanoma while only a few researches have addressed this matter in breast cancer [16,17,18,19,20]. Dill et al. found a discordance of 50% for tumoral PD-L1 staining of 245 breast cancer patients and 59% concordance of PD-L1 staining in the immune stroma in 91 patients. However, this research included a variety of breast cancers and did not exclusively address TNBC [21]. Additionally, Stovgaard et al. showed there was substantial heterogeneity of PD-L1 expression in 110 patients with TNBC, whereas heterogeneity was greater in immune cells rather than tumor cells [22]. Although it should be noted, Stovgaard et al. used 22C3 PD-L1 clone rather than SP142. These findings are in accordance with our clinical samples. Hence, we illustrated heterogenous tumors in our computer-based model, which demonstrated significant higher false results compared to homogenous tumors.
In this study we found a few factors that affect the reliability of biopsies in representing the tumor's PD-L1 expression. Accordingly, we developed a model that can predict the risk of false results based on the fraction of PD-L1 positive cells in the biopsy and the biopsy size (Table 3, Fig. 7). As false positive cases can result in overtreatment and false negative cases miss patients that can benefit from immune checkpoint therapy, a potential clinical implication of our algorithm is to minimize these cases. Although further clinical trials are required to validate this research, our algorithm can be an addition to the oncologist toolkit that could also be taken into account for making the best treatment decision for the patient.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Doepker MP, Holt SD, Durkin MW et al (2018) Triple-negative breast cancer: a comparison of race and survival. Am Surg 84:881–887. https://doi.org/10.1177/000313481808400636
den Brok WD, Speers CH, Gondara L et al (2017) Survival with metastatic breast cancer based on initial presentation, de novo versus relapsed. Breast Cancer Res Treat 161:549–556. https://doi.org/10.1007/s10549-016-4080-9
Yin L, Duan JJ, Bian XW, Yu SC (2020) Triple-negative breast cancer molecular subtyping and treatment progress. Breast Cancer Res 22:1–13. https://doi.org/10.1186/s13058-020-01296-5
Narayan P, Wahby S, Gao JJ et al (2020) FDA approval summary: atezolizumab plus paclitaxel protein-bound for the treatment of patients with advanced or metastatic TNBC whose tumors express PD-L1. Clin Cancer Res 26:2284–2289. https://doi.org/10.1158/1078-0432.CCR-19-3545
Csőszi T, Fülöp A, Gottfried M et al (2016) Pembrolizumab versus chemotherapy for PD-L1–positive non–small-cell lung cancer. N Engl J Med. https://doi.org/10.1056/NEJMoa1606774
Zhang T, Xie J, Arai S et al (2016) The efficacy and safety of anti-PD-1/PD-L1 antibodies for treatment of advanced or refractory cancers: a meta-analysis. Oncotarget 7:73068–73079. https://doi.org/10.18632/oncotarget.12230
Philips GK, Atkins M (2015) Therapeutic uses of anti-PD-1 and anti-PD-L1 antibodies. Int Immunol 27:39–46. https://doi.org/10.1093/intimm/dxu095
Emens LA, Adams S, Barrios CH et al (2021) First-line atezolizumab plus nab-paclitaxel for unresectable, locally advanced, or metastatic triple-negative breast cancer: IMpassion130 final overall survival analysis. Ann Oncol 32:983–993. https://doi.org/10.1016/j.annonc.2021.05.355
Schmid P, Adams S, Rugo HS et al (2018) Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N Engl J Med 379:2108–2121. https://doi.org/10.1056/nejmoa1809615
Di Spazio L, Rivano M, Cancanelli L et al (2022) The degree of programmed death-ligand 1 (PD-L1) positivity as a determinant of outcomes in metastatic triple-negative breast cancer treated with first-line immune checkpoint inhibitors. Cureus 1:1–5. https://doi.org/10.7759/cureus.21065
Badve SS, Penault-Llorca F, Reis-Filho JS et al (2021) Determining PD-L1 status in patients with triple-negative breast cancer: lessons learned From IMpassion130. JNCI J Natl Cancer Inst 00:1–12. https://doi.org/10.1093/jnci/djab121
Schmid P, Cortes J, Pusztai L et al (2020) Pembrolizumab for early triple-negative breast cancer. N Engl J Med 382:810–821. https://doi.org/10.1056/nejmoa1910549
Wang Y, Zhou S, Yang F et al (2019) Treatment-related adverse events of PD-1 and PD-L1 inhibitors in clinical trials: a systematic review and meta-analysis. JAMA Oncol 5:1008–1019. https://doi.org/10.1001/jamaoncol.2019.0393
Gagné A, Wang E, Bastien N et al (2019) Impact of specimen characteristics on PD-L1 testing in non-small cell lung cancer: validation of the IASLC PD-L1 testing recommendations. J Thorac Oncol 14:2062–2070. https://doi.org/10.1016/j.jtho.2019.08.2503
Focke CM, Decker T, Van Diest PJ (2016) The reliability of histological grade in breast cancer core needle biopsies depends on biopsy size: a comparative study with subsequent surgical excisions. Histopathology 69:1047–1054. https://doi.org/10.1111/his.13036
McLaughlin J, Han G, Schalper KA et al (2016) Quantitative assessment of the heterogeneity of PD-L1 expression in non–small-cell lung cancer. JAMA Oncol 2:46–54. https://doi.org/10.1001/jamaoncol.2015.3638
Munari E, Zamboni G, Lunardi G et al (2018) PD-L1 expression heterogeneity in non-small cell lung cancer: defining criteria for harmonization between biopsy specimens and whole sections. J Thorac Oncol 13:1113–1120. https://doi.org/10.1016/j.jtho.2018.04.017
Madore J, Vilain RE, Menzies AM et al (2015) PD-L1 expression in melanoma shows marked heterogeneity within and between patients: Implications for anti-PD-1/PD-L1 clinical trials. Pigment Cell Melanoma Res 28:245–253. https://doi.org/10.1111/pcmr.12340
Ben Dori S, Aizic A, Sabo E, Hershkovitz D (2020) Spatial heterogeneity of PD-L1 expression and the risk for misclassification of PD-L1 immunohistochemistry in non-small cell lung cancer. Lung Cancer 147:91–98. https://doi.org/10.1016/j.lungcan.2020.07.012
Gniadek TJ, Li QK, Tully E et al (2017) Heterogeneous expression of PD-L1 in pulmonary squamous cell carcinoma and adenocarcinoma: implications for assessment by small biopsy. Mod Pathol 30:530–538. https://doi.org/10.1038/modpathol.2016.213
Dill EA, Gru AA, Atkins KA et al (2017) PD-L1 expression and intratumoral heterogeneity across breast cancer subtypes and stages: an assessment of 245 primary and 40 metastatic tumors. Am J Surg Pathol 41:334–342. https://doi.org/10.1097/PAS.0000000000000780
Stovgaard ES, Bokharaey M, List-Jensen K et al (2020) PD-L1 diagnostics in the neoadjuvant setting: implications of intratumoral heterogeneity of PD-L1 expression in triple negative breast cancer for assessment in small biopsies. Breast Cancer Res Treat 181:553–560. https://doi.org/10.1007/s10549-020-05655-w
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
SBD, AZ, AA, and DH conceived the research and designed its structure. SBD and AZ collected the digital pathology slides and performed annotations. All authors participated in data analysis. SBD and DH prepared the manuscript. All authors read and approved the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved and informed consent was waived by the local ethics committee at Tel-Aviv Sourasky medical center. Approval number: 0660-16-TLV.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ben Dori, S., Aizic, A., Zubkov, A. et al. The risk of PD-L1 expression misclassification in triple-negative breast cancer. Breast Cancer Res Treat 194, 297–305 (2022). https://doi.org/10.1007/s10549-022-06630-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06630-3