Abstract
While cardiac magnetic resonance (CMR) is the reference method to evaluate left and right ventricular functions, volumes and masses, there is no widely accepted method for the quantitative analysis of trabeculae and papillary muscles (TPM). The aim of this study was to investigate the effect of TPM quantification on left and right ventricular CMR values in a normal cohort and to investigate interobserver variability of threshold-based (TB) analysis by three independent observers with variant experience in CMR. At our clinic, 60 healthy volunteers (30 males, mean age 25.6 ± 4.7 years) underwent CMR scan performed on a 1.5T Philips Achieva MR machine. On short-axis cine images, endo- and epicardial contours were detected by three independent observers with variable experience in CMR (low- ca. 120, mid- > 800, high-experienced > 5000 original CMR cases). Using Conv and TB methods (Medis 7.6 QMass software Leiden, The Netherland), we measured LV and RV ejection fractions, end-diastolic, end-systolic, stroke volumes and masses. We used TB method for quantifying TPM in ventricles using epicardial contour layers. Interobserver variability was evaluated, and the observer’s experience as an impact on variability of each investigated parameters was assessed. Comparing Conv and TB quantification methods’ significant difference were detected for all LV and RV parameters in case of all observers (H, M and L p < 0.0001). The global intraclass correlation coefficient (G-ICC) representing interobserver agreement for all investigated parameters was lower with Conv method (G-ICCConv vs. G-ICCTB 0.86 vs. 0.92 p < 0.0001). The ICC of LV parameters was higher using TB quantification (LV-ICCConv vs. LV-ICCTB 0.92 vs. 0.96 p < 0.0001), and for the evaluation of RV values, the TB method also had significantly higher interobserver agreement (RV-ICCConv vs. RV-ICCTB 0.80 vs. 0.89 p < 0.0001). The TB algorithm could be a consistent method to assess LV and RV CMR values, and to measure trabeculae and papillary muscles quantitatively in various level of experience in CMR.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cardiovascular magnetic resonance (CMR) is the reference method to evaluate left (LV) and right (RV) ventricular volumes and masses [1, 2], although conventional 2D cine methods are known to cause partial volume artefacts due to the thickness of the slices.
Due to the high spatial resolution and excellent myocardial-blood contrast, CMR provide an accurate evaluation of small myocardial structures like myocardial trabeculae and papillary muscles (TPM) [3, 4]. Quantitative analysis of ventricular trabeculation and papillary muscles is controversial. There is neither generally accepted evaluation for TPM quantification, nor data available about the distribution of CMR performing laboratories regarding TPM quantification. It is difficult to compare literature data because of the diverse evaluation of TPM volumes. The majority of prior studies presented data that was either papillary muscle or trabecular mass measurement in isolation [5,6,7,8]; few studies are available which reported the sum of papillary and trabecular mass [9,10,11,12].
According to literature data, quantitative analysis of TPM could alter normal LV values [10, 13]. Although structural and functional evaluation of RV is challenging, CMR is an accurate method to evaluate thin free wall and trabeculation of right ventricle [1]. The right ventricular trabeculation are pronounced, but limited data is available regarding quantitative measurement of these structures in a normal population [9, 14, 15] or in pathological conditions [6, 16]. Large population based studies provide normal RV values excluding small intracavitary structures from RV mass [17,18,19].
Cardiovascular magnetic resonance is a rapidly maturing imaging modality. Indications of CMR examinations have continuously expanded, and an increasing number of centers perform CMR scans. Based on UK data, in half of the CMR performing laboratories are working less experienced physicians and a significant proportion of these centers do not have formal training programs for their trainees or supervised by a mentor with Level 3 CMR certification [14]. For trainees with moderate experience, the attributes of the clinically used quantification software are highly important to gain the most optimal interobserver and intraobserver agreement.
In this study we used a threshold-based (TB), semi-automatic cardiac MR quantification software (7.6 QMass Medis, Leiden, The Netherland) for the measurement of TPM. The software could delineate the myocardium from blood pool based on their different signal intensities in the absence of endocardial contour [20]. The algorithm has improved accuracy evaluating left ventricular volumetric parameters compared to flow measurement as a reference [20]. TB quantification also reported less time consuming than conventional (Conv) method regarding LV evaluation [20].
The aim of this study was to investigate the effect of TPM quantification on left and right ventricular parameters in a normal cohort and explore how varying experience in utilizing CMR influences reproducibility.
Methods
Patients’ characteristics
A total of 30 male and 30 female Caucasian healthy individuals (age 25.6 ± 4.7 years) underwent cardiovascular magnetic resonance imaging. Volunteers’ personal and family history were obtained using a uniform patient questionnaire focusing on cardiovascular diseases and risk factors. All subjects were free of complaints and had no known cardiovascular diseases. All volunteers underwent on a detailed medical check-up included physical examination, 12-lead resting electrocardiography and echocardiography examination. The study protocol was approved by the national ethics committee and informed consent was obtained from all individual participants included in the study.
Image acquisition
CMR examinations were conducted on a 1.5 T MRI scanner (Achieva, Philips Medical Systems) with a 5-channel cardiac coil. Retrospectively-gated, balanced steady-state free precession (bSSFP) cine images were acquired in conventional 2-chamber, 3-chamber and 4-chamber views. Short-axis cine images with full coverage of the left and right ventricle and left and right ventricular outflow tract (LVOT, RVOT) movies were also obtained. Slice thickness was 8 mm without interslice gap, field of view was 350 mm on average adapted to body size.
Image analysis
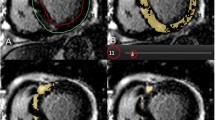
On cine short axis images end-diastolic and end-systolic cardiac phases were identified (Fig. 1). The most basal section was required to show ≥ 50% visible myocardial circumference in order to be included. For the correct visualization of LV and RV contours, additional LVOT and RVOT cine images were performed.
During conventional contouring (a, c) epi- and endocardial layers were manually traced and trabeculae and papillary muscles (TPM) included in the ventricular cavity. For threshold-based quantification we used the same end-systolic and end-diastolic phases and TPM were measured as part of the ventricular mass (b, d)
During conventional contouring epi- and endocardial layers were manually traced. Endocardial layer was detected along the compact myocardium resulting that TPM being included in the ventricular cavity (Fig. 1). We measured the following CMR parameters in left and right ventricles: ejection fraction (EF), end-diastolic (EDV), end-systolic (ESV), stroke volumes (SV), cardiac output (CO) and myocardial mass in end-diastolic (EDM) and end-systolic (ESM) phases. Parameters corrected for body surface area were calculated.
For TB quantification, we used the same end-systolic and end-diastolic phases. A thresholding algorithm (MassK 7.6, Medis, Leiden, The Netherlands) was used to discriminate between chamber blood and myocardium based on their alter signal intensity. The algorithm calculates blood percentage value for each pixels with the previously described equation [7]. For the evaluation of TPM, we used the application default 50% thresholding value for both left and right ventricles without manual correction and TPM, TPMi (TPM/BSA), TPM% (TPM/EDM × 100) were calculated (Fig. 1).
Three independent observers evaluated CMR images both with conventional (Conv) and threshold-based (TB) methods. The most experienced reader has 15 years of experience with more than 5000 original CMR cases and Level3 certification proved by the European Association of Cardiovascular Imaging. The mid-experienced reader had more than 800 individual original CMR cases during the last 5 years and low-experienced reader independently analyzed ca. 120 CMR cases.
Statistical analysis
Statistical analyses were performed using MedCalc 13.2.2 (MedCalc Software, Ostend, Belgium). The Kolmogorov–Smirnov test was used to assess the normal distribution of the data. Continuous variables were reported as mean ± SD. p values of less than 0.05 were considered significant. The comparison of conventional and threshold-based methods was performed with repeated measures one-way ANOVA with a within-subjects design and Tukey–Kramer post hoc multi-comparison test. Equality of left and right ventricular stroke volumes was tested with paired samples t-tests. The effect of gender on the outcome of the evaluation methods was investigated with a multi-level repeated measured ANOVA. The interobserver agreement of the three MRI evaluation methods was examined with the intraclass correlation coefficients (ICC score). ICC was interpreted as follows: less than 0.4, poor; 0.4 to 0.75, fair to good; and greater than 0.75, excellent.
Results
Comparison of conventional and threshold-based methods
The various CMR parameters were compared against each other to detect differences between conventional and threshold-based methods and systematic differences were found. All of the investigated LV and RV parameters were significantly different: volumetric values were lower and masses were higher with the TB method in case of all observers (H, M, L observers p < 0.001). LV mass differed substantially by method with absolute difference of 34.4–43.8% and relative difference of 30.4– 40.9% (p < 0.001). RV mass differed by method with absolute difference of 83.0–121.6% and relative difference of 86.4–114.3% (p < 0.001). Mean differences between Conv and TB CMR values are shown in Table 1. Left and right ventricular stroke volumes over all three observers were different using the conventional method [(1) High vs. Mid LVSVi: 51.7 ± 6.5 vs. 54.9 ± 5.9 p < 0.001 (2) High vs. Low-experienced LVSVi: 51.7 ± 6.5 vs. 56.4 ± 6.2 p < 0.001; same significance for RV parameters (1) RVSVi: 50.0 ± 6.0 vs. 53.8 ± 6.6 p < 0.01 (2) 50.0 ± 6.0 vs. 52.9 ± 7.0 p < 0.01]. Equality of left ventricular stroke volumes was excellent with the TB method, and no significant differences were found between observers (High vs. Mid vs. Low-experienced LVSVi: 48.8 ± 5.9 vs. 50.9 ± 5.5 vs. 50.3 ± 5.3). We found that the lowest deviation between the two measurement in case of the distinct parameters were not consequently associated to one observer.
Trabeculation and papillary muscles in left and right ventricles
The average LV-TPM in end-diastolic phase was around one-third of the left ventricular mass in all observers (LV-TPM% of observers: H 30.3 ± 3.9%, M 30.0 ± 4.04%, L 26.5 ± 4.0%) while TPM in the right ventricle was measured as approximately half of the right ventricular mass (RV-TPM% of observers: H 54.6 ± 6.6%, M 48.3 ± 5.8%, L 47.0 ± 9.4%). The LV-TPM in end-diastolic and end-systolic phases was not different in observers regardless of the level of experience.
In both the left and right ventricles, the expert reader measured the highest TPMi compared to other observers (LV-EDTPMi H vs. M and L-experienced 22.5 ± 4.9 vs. 19.9 ± 4.7 and L 19.4 ± 3.4 RV-EDTPMi H vs. M-experienced 18.2 ± 4.1 vs. 15.8 ± 2.9 p < 0.001).
Gender differences
The volumetric and masses parameters of male subjects were significantly higher than that of the concomitant female parameter, across all methods and observers (p < 0.001). Differences between male and female identical CMR parameters were determined to be independent from different methods and observers (e.g., H-experienced male vs. female LVMiConv 57.9 ± 7.3 vs. 43.3 ± 4.2 LVMiTB 52.7 ± 6.1 vs. 39.5 ± 5.1 p < 0.001).
Interobserver reliability of different MRI methods
Comparing the two methods, interobserver variability of LV parameters was lower using TB analysis and for the evaluation of RV values TB method had superiority against Conv analysis (Table 2). Global intraclass correlation (G-ICC) value represents interobserver agreement of all investigated LV and RV parameters. G-ICCConv and G-ICCTB were excellent, G-ICC of TB quantification was higher than G-ICC of Conv method (Table 2).
Regarding distinct CMR parameters, interobserver agreement was excellent for volumetric parameters with both methods (ICC of LV-EDViConv 0.978 and LV-EDViTB 0.969, RV-EDViConv 0.947 and RV-EDViTB 0.960). When measuring LV and RV masses, TB quantification seems to be more reliable (ICC of LV-EDMiConv 0.868 and LV-EDMiTB 0.936, RV-EDMiConv 0.748 and RV-EDMiTB 0.920). It should be mentioned, however, that based on the data in Table 3 the ICC scores of the methods showed variability within the different parameters: the above observed trend is not reflected for the case of RVESVi, RVCOi, and RVEF and the ICC scores of RVEF have large confidence intervals.
Discussion
Left and right ventricular parameters can change depending on a different contouring process resulting in diverse normal CMR values.
In this study, we compared two different LV and RV measurements focusing on interobserver variability of readers with different experience. We determined the global ICC score for all measured CMR parameters and the TB method showed significantly higher interobserver agreement. TB-method surpassed the first method significantly with G-ICC scores of over 0.9. The threshold-based method has improved accuracy comparing aortic flow measurement as a reference [20]; accordingly, our data proved excellent equality of left and right ventricular stroke volumes with TB analysis compared to the conventional method.
Left ventricular mass is a widely accepted morphological parameter to assess and predict clinical and cardiovascular outcomes, respectively [15, 21]. Our data regarding the sum of papillary muscle and trabecular mass in the left ventricle was consistent with large population based literature data in all observers [10].
Although right ventricular morphology and function have a diagnostic and prognostic value in cardiovascular [22,23,24,25] and pulmonary diseases [18, 24, 26], TPM and TPM corrected RV parameters are not-well understood. Trabeculation in RV significantly affects quantifications of volumes and masses; indeed, in our study ca. half of the right ventricular mass was measured as TPM which is not a negligible fraction. As we previously mentioned, right ventricular trabeculation is physiologically more pronounced than the left ventricular, and the evaluation of the right ventricular contours are more challenging. Delineating the highly trabeculated endocardial surface from the ventricular cavity is more complicated in the RV than along the smooth LV surface. Manually contouring the epicardial surface of the thin RV wall is also more difficult than is the case with the thick LV wall. With the TB method after epicardial contouring, the software defines the endocardial surface based on the different signal intensity of myocytes and blood. These anatomical and technical differences could explain the large differences regarding RV mass measurement.
Threshold-based semi-automatic quantification systems are a user-friendly, accurate and consistent method for evaluating left and right ventricular CMR parameters. As it was apparent based on the above, the experience of the evaluator did not have any considerable effect on either LV or RV CMR parameters while using the TB quantification method.
Limitations of the study: the limited patient number and the lack of scan-rescan reproducibility. The high differences of ICC values between the conventional and TB methods partially may come from the altering ranges of the measured parameters.
The precise and standard measurement of left and right ventricular CMR parameters is crucial and plays a major role during patient follow-up. This study highlights the necessity of a consistent method for evaluation of papillary muscle and trabecula mass in ventricles to uniform normal CMR values, and to avoid misinterpretation of various methods and inaccurate clinical decision-making. The superior reproducibility of TPM corrected LV and RV parameters regardless of the readers’ experience favors this TB algorithm for clinical use.
References
Farber NJ, Reddy ST, Doyle M, Rayarao G, Thompson DV, Olson P et al (2014) Ex vivo cardiovascular magnetic resonance measurements of right and left ventricular mass compared with direct mass measurement in excised hearts after transplantation: a first human SSFP comparison. J Cardiovasc Magn Reson 16:74
Lorenz CH, Walker ES, Morgan VL, Klein SS, Graham TP (1999) Normal human right and left ventricular mass, systolic function, and gender differences by cine magnetic resonance imaging. J Cardiovasc Magn Reson 1(1):7–21
Moon JC, Lorenz CH, Francis JM, Smith GC, Pennell DJ (2002) Breath-hold FLASH and FISP cardiovascular MR imaging: left ventricular volume differences and reproducibility. Radiology 223(3):789–797
Fieno DS, Jaffe WC, Simonetti OP, Judd RM, Finn JP (2002) TrueFISP: assessment of accuracy for measurement of left ventricular mass in an animal model. J Magn Reson Imaging 15(5):526–531
Sievers B, Kirchberg S, Bakan A, Franken U, Trappe HJ (2004) Impact of papillary muscles in ventricular volume and ejection fraction assessment by cardiovascular magnetic resonance. J Cardiovasc Magn Reson 6(1):9–16
Vogel-Claussen J, Finn JP, Gomes AS, Hundley GW, Jerosch-Herold M, Pearson G et al (2006) Left ventricular papillary muscle mass: relationship to left ventricular mass and volumes by magnetic resonance imaging. J Comput Assist Tomogr 30(3):426–432
Alfakih K, Plein S, Thiele H, Jones T, Ridgway JP, Sivananthan MU (2003) Normal human left and right ventricular dimensions for MRI as assessed by turbo gradient echo and steady-state free precession imaging sequences. J Magn Reson Imaging 17(3):323–329
Hudsmith LE, Petersen SE, Francis JM, Robson MD, Neubauer S (2005) Normal human left and right ventricular and left atrial dimensions using steady state free precession magnetic resonance imaging. J Cardiovasc Magn Reson 7(5):775–782
Andre F, Burger A, Lossnitzer D, Buss SJ, Abdel-Aty H, Gianntisis E et al (2015) Reference values for left and right ventricular trabeculation and non-compacted myocardium. Int J Cardiol 185:240–247
Chuang ML, Gona P, Hautvast GL, Salton CJ, Blease SJ, Yeon SB et al (2012) Correlation of trabeculae and papillary muscles with clinical and cardiac characteristics and impact on CMR measures of LV anatomy and function. JACC Cardiovasc Imaging 5(11):1115–1123
Papavassiliu T, Kuhl HP, Schroder M, Suselbeck T, Bondarenko O, Bohm CK et al (2005) Effect of endocardial trabeculae on left ventricular measurements and measurement reproducibility at cardiovascular MR imaging. Radiology 236(1):57–64
Park EALW., Kim HK, Chung JW (2015) Effect of papillary muscles and trabeculae on left ventricular measurement using cardiovascular magnetic resonance imaging in patients with hypertrophic cardiomyopathy. Korean J Radiol 16(1):4–12
Weinsaft JW, Cham MD, Janik M, Min JK, Henschke CI, Yankelevitz DF et al (2008) Left ventricular papillary muscles and trabeculae are significant determinants of cardiac MRI volumetric measurements: effects on clinical standards in patients with advanced systolic dysfunction. Int J Cardiol 126(3):359–365
Antony R, Daghem M, McCann GP, Daghem S, Moon J, Pennell DJ et al (2011) Cardiovascular magnetic resonance activity in the United Kingdom: a survey on behalf of the British Society of Cardiovascular Magnetic Resonance. J Cardiovasc Magn Reson 13:57
Levy D, Garrison RJ, Savage DD, Kannel WB, Castelli WP (1990) Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med 322(22):1561–1566
Winter MM, Bernink FJ, Groenink M, Bouma BJ, van Dijk AP, Helbing WA et al (2008) Evaluating the systemic right ventricle by CMR: the importance of consistent and reproducible delineation of the cavity. J Cardiovasc Magn Reson 10:40
Foppa M, Arora G, Gona P, Ashrafi A, Salton CJ, Yeon SB et al (2016) Right ventricular volumes and systolic function by cardiac magnetic resonance and the impact of sex, age, and obesity in a longitudinally followed cohort free of pulmonary and cardiovascular disease: the Framingham Heart Study. Circ Cardiovasc Imaging 9(3):e003810
Kawut SM, Horn EM, Berekashvili KK, Garofano RP, Goldsmith RL, Widlitz AC et al (2005) New predictors of outcome in idiopathic pulmonary arterial hypertension. Am J Cardiol 95(2):199–203
Maceira AM, Prasad SK, Khan M, Pennell DJ (2006) Reference right ventricular systolic and diastolic function normalized to age, gender and body surface area from steady-state free precession cardiovascular magnetic resonance. Eur Heart J 27(23):2879–2888
Varga-Szemes A, Muscogiuri G, Schoepf UJ, Wichmann JL, Suranyi P, De Cecco CN et al (2016) Clinical feasibility of a myocardial signal intensity threshold-based semi-automated cardiac magnetic resonance segmentation method. Eur Radiol 26(5):1503–1511
Bluemke DA, Kronmal RA, Lima JA, Liu K, Olson J, Burke GL et al (2008) The relationship of left ventricular mass and geometry to incident cardiovascular events: the MESA (Multi-Ethnic Study of Atherosclerosis) study. J Am Coll Cardiol 52(25):2148–2155
de Groote P, Millaire A, Foucher-Hossein C, Nugue O, Marchandise X, Ducloux G et al (1998) Right ventricular ejection fraction is an independent predictor of survival in patients with moderate heart failure. J Am Coll Cardiol 32(4):948–954
Di Salvo TG, Mathier M, Semigran MJ, Dec GW (1995) Preserved right ventricular ejection fraction predicts exercise capacity and survival in advanced heart failure. J Am Coll Cardiol 25(5):1143–1153
Driessen MM, Baggen VJ, Freling HG, Pieper PG, van Dijk AP, Doevendans PA et al (2014) Pressure overloaded right ventricles: a multicenter study on the importance of trabeculae in RV function measured by CMR. Int J Cardiovasc Imaging 30(3):599–608
Polak JF, Holman BL, Wynne J, Colucci WS (1983) Right ventricular ejection fraction: an indicator of increased mortality in patients with congestive heart failure associated with coronary artery disease. J Am Coll Cardiol 2(2):217–224
Goldhaber SZ, Visani L, De Rosa M (1999) Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet 353(9162):1386–1389
Funding
This project was supported by a grant from the National Research, Development and Innovation Office (NKFIH) of Hungary (K 120277). Project No. NVKP_16-1-2016-0017 has been implemented with the support provided from the National Research, Development and Innovation Fund of Hungary, financed under the NVKP_16 funding scheme.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the National Research Committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Csecs, I., Czimbalmos, C., Suhai, F.I. et al. Left and right ventricular parameters corrected with threshold-based quantification method in a normal cohort analyzed by three independent observers with various training-degree. Int J Cardiovasc Imaging 34, 1127–1133 (2018). https://doi.org/10.1007/s10554-018-1322-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-018-1322-4