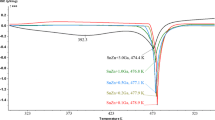
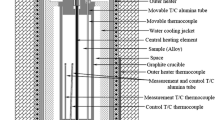
Abstract
This work is aimed at comparing several methods for the measurement of physical properties for molten Sn and Sn–Ag alloys, namely, surface tension, density, and viscosity. The method used for viscosity in this work is the modified capillary method. For surface tension and density, the data used for comparison were previously measured using the maximum bubble pressure method and the dilatometer technique, respectively, for four Sn–Ag alloys having (3.8, 32, 55, and 68) at% Ag. The results are compared with those obtained using a new method based on a fluid draining from a crucible under the influence of gravity, designated the Roach–Henein (RH) method. This new method enables the determination of these three physical properties in one set of measurements. Liquid Sn was used as well as two liquid Sn–Ag alloys having (3.8 and 34.6) at% Ag with the RH method. It was determined that the RH method may be used to simultaneously obtain surface tension, viscosity, and density and that the errors associated with these measurements were similar to those obtained using traditional and separate techniques. Comparisons of the measured viscosity and surface tension to those predicted using thermodynamic models will also be presented. Finally a comparison of mixing model predictions with the experimentally measured alloy surface tension and viscosity is also presented.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- A :
-
Molar surface area in a monolayer of species i, m2
- A o :
-
Pre-exponential in Eq. 39, mPa · s
- B :
-
Starting liquid height H s in the reservoir, m
- C m :
-
Cumulative mass, kg
- D(T):
-
Diameter of lower container in CM method as a function of temperature T, m
- G :
-
Free energy, J · mol−1
- G*:
-
Activation energy of the alloy, J · mol−1
- ΔG′:
-
Change in activation energy, J · mol−1
- H s :
-
Height of liquid at which measurement started, initial height, m
- H f :
-
Height of liquid at which measurement ended, final height, m
- H m :
-
Average height, m
- H m :
-
Molar enthalpy of mixing for the liquid alloy, J · mol−1
- L :
-
Constant set to 1.091 for liquid metals assuming close-packed structures
- M :
-
Molecular weight, kg · mol−1
- N 0 :
-
Avogadro’s number, mol−1
- N s :
-
Depth of capillary immersion at the beginning of measurement, m
- N f :
-
Depth of capillary immersion at the end of measurement, m
- N m :
-
Substitute depth of measurement, m
- Q exp :
-
Experimental volumetric flow rate, m3 · s−1
- R :
-
Universal gas constant, J · mol−1 · K−1
- S :
-
Area under the curve between H s and H f, m · s
- T :
-
Temperature, K
- V :
-
Volume of draining fluid, volume liquid, m3
- V i :
-
Molar volume of pure liquid i, m3
- V s :
-
Volume of liquid drawn into the reservoir at the start of measurement, m3
- ΔV E :
-
Excess molar volume of alloy components
- W i,j :
-
Interaction coefficient for binary alloys
- X :
-
Mole fraction, dimensionless
- Z :
-
Coordination number
- a :
-
Polynomial constant describing slope of the discharge coefficient curve, dimensionless
- b :
-
Polynomial constant describing the y-intercept of the discharge coefficient curve dimensionless
- g:
-
Acceleration of gravity, m · s−2
- h :
-
Planck’s constant, J · s
- h exp :
-
Liquid head above a point of reference in RH method, m
- l :
-
Length of capillary, m
- Δp :
-
Pressure difference, Pa
- r :
-
Capillary radius in MC method, m
- r c :
-
Crucible radius, m
- r o :
-
Orifice radius in RH method, m
- t :
-
Time, s
- u :
-
Velocity, m · s−1
- α :
-
Constant
- β :
-
Parameter corresponding to the ratio of the coordination number in the surface phase to that in the bulk phase (Z S/Z B)
- η :
-
Viscosity, mPa · s
- η H :
-
Hagenbach correction for viscosity, mPa · s
- ρ :
-
Density, kg · m−3
- σ :
-
Surface tension, mN · m−1
- Φ:
-
Diameter of the bottom tank (crucible), m
- Bo :
-
Bond number \({\left({\frac{\rho gr_0 h_{\rm exp}}{\sigma}}\right)}\), dimensionless
- C d :
-
Discharge coefficient, dimensionless
- Fr :
-
Froude number \({({\rm Q}_{\rm exp}^{2}/(2\pi g{\rm r}_{\rm o}^{3}))}\), dimensionless
- Re :
-
Reynolds number \({\left({\frac{2\rho Q_{\rm exp}}{\pi r_0 \eta}}\right)}\), dimensionless
- B:
-
Bulk
- E:
-
Excess
- S:
-
Surface
- ex:
-
Excess
- Ag:
-
Silver
- Sn:
-
Tin
References
Moser Z., Gąsior W., Pstruś J.: J. Phase Equilib. 22, 254 (2001)
Gąsior W., Moser Z., Pstruś J., Kucharski M.: Arch. Metall. 46, 23 (2001)
Roach S.J., Henein H.: Can. Metall. Quart. 42, 175 (2003)
Roach S.J., Henein H.: Metall. Mater. Trans. B 36, 667 (2005)
Henein H.: Can. Metall. Quart. 44, 261 (2004)
Rothwell E.: J. Inst. Met. 90, 389 (1962)
Kisun’ko V.Z., Novokhatskii I.A., Beloborodov A.Z., Bychkov Y.B., Pogorelov A.I.: Tsvetnte Metally (Non-Ferrous) 24, 84 (1983)
Z. Moser, W. Gąsior, A. Dębski, J. Pstruś, Database of Lead-free Soldering Materials (Institute of Metallurgy and Materials Science PAS, Krakow, 2007), ISBN 83-60768-01-3
Schpilrajn E.E., Fomin W.A., Scovorodko S.N., Sokol G.F.: Isledowanie Wiazkosti Zidkich Metalov. Moskwa, Nauka (1983)
Iida T., Guthrie R.I.L.: The Physical Properties of Liquid Metals, pp. 77. Clarendon Press, Oxford (1988)
Chhabra R., Sheth D.: Z. Metallkd. 81, 264 (1990)
Gebhardt E., Becker M., Tragner E.: Z. Metallkd. 46, 379 (1955)
Mills K.C., Su Y.C.: Int. Mater. Rev. 51, 329 (2006)
Lide, D.R. (ed.): CRC Handbook of Chemistry and Physics, 88th edn. CRC Press, Boca Raton, FL (2007)
Miller R.R.: Liquid Metals Handbook. Atomic Energy Commission and the Department of Navy, Washington, DC (1952)
Alchagirov B.B., Chochaeva A.M.: High Temp. 38, 44 (2000)
Keene B.J.: Int. Mater. Rev. 38, 157 (1993)
Lee J., Shimoda W., Tanaka T.: Mater. Trans. 45, 2864 (2004)
Kucharski M., Fima P.: Monasch. Chem. 136, 1841 (2005)
Butler J.A.V.: Proc. R. Soc. Lond., Ser. A CXXXV, 348 (1932)
Tanaka T., Hack K., Iida T., Hara S.: Z. Metallkd. 87, 380 (1996)
Ohnuma I., Liu X.J., Ishida K.: J. Electron. Mater. 28, 1164 (1998)
I. Ohnuma, X.J. Liu, Y. Inohana, K. Ishida, Z. Moser, W. Gąsior, J. Pstruś, W. Zakulski, in Proceedings of CALPHAD XXIX (Massachusetts Institute of Technology, Cambridge, MA, 2000), p. 15
Kucharski M.: Arch. Hut. 22, 181 (1977)
Ohtani H., Miyashita M., Ishida K.: J. Jpn. Inst. Met. 63, 685 (1999)
Gąsior W., Moser Z., Pstruś J.: J. Phase Equilib. 24, 504 (2003)
Gąsior W., Pstruś J., Moser Z., Krzyżak A., Fitzner K.: J. Phase Equilib. 24, 40 (2003)
Berthou P.E., Tougas R.: Metall. Trans. 1, 2978 (1970)
J. Pstruś, Physico-Chemical Properties of New Solder Alloys on Example of Sn-Zn-In System, Ph.D. Thesis, Krakow, 2008
Gale, W.F., Totemeier, T.C. (eds): Smithells Metals Reference Book. Elsevier, Butterworth-Heinemann, Oxford (2004)
Kaban I., Mhiaoui S., Hoyer W., Gasser J.-G.: J. Phys.: Condens. Matter 17, 7867 (2005)
E.A. Moelwyn-Hughes, Phys. Chem. 434 (1970)
Sichen D., Bygdén J., Seetharaman S.: Metall. Mater. Trans. B 25, 519 (1994)
Seetharaman S., Sichen D.: Metall. Mater. Trans. B 25, 589 (1994)
Kozlov L.Y., Romanov L.M., Petrov N.N.: Izv. Vuzov. Chernaya Metall. 3, 7 (1983)
G. Kaptay, in Proceeedings of MicroCAD 2003, International Conference Section: Metallurgy (University of Miskolc, Hungary, 2003), p. 23
Acknowledgments
The authors wish to acknowledge the Natural Science and Engineering Research Council for funding the experimental work and analysis of the Edmonton effort. The authors from IMIM PAS wish to thank the Ministry of Science and Higher Education for financing the project on enlarging the SURDAT database.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Gancarz, T., Moser, Z., Gąsior, W. et al. A Comparison of Surface Tension, Viscosity, and Density of Sn and Sn–Ag Alloys Using Different Measurement Techniques. Int J Thermophys 32, 1210–1233 (2011). https://doi.org/10.1007/s10765-011-1011-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10765-011-1011-1