Abstract
Segregating plant hybrids often have more ecological and molecular variability compared to parental species, and are therefore useful for studying relationships between different traits, and the adaptive significance of trait variation. Hybrid systems have been used to study the relationship between the expression of plant defense compounds and herbivore susceptibility. We conducted a western flower thrips (WFT) bioassay using a hybrid family and investigated the relationship between WFT resistance and pyrrolizidine alkaloid (PA) variation. The hybrid family consisted of two parental (Jacobaea vulgaris and Jacobaea aquatica) genotypes, two F1 genotypes, and 94 F2 hybrid lines. The J. aquatica genotype was more susceptible to thrips attack than the J. vulgaris genotype, the two F1 hybrids were as susceptible as J. aquatica, and susceptibility to WFT differed among F2 hybrid lines: 69 F2 lines were equally susceptible compared to J. aquatica, 10 F2 lines were more susceptible than J. aquatica and 15 F2 lines were as resistant as J. vulgaris or were intermediate to the two parental genotypes. Among 37 individual PAs that were derived from four structural groups (senecionine-, jacobine-, erucifoline- and otosenine-like PAs), the N-oxides of jacobine, jaconine, and jacoline were negatively correlated with feeding damage caused by WFT, and the tertiary amines of jacobine, jaconine, jacoline, and other PAs did not relate to feeding damage. Total PA concentration was negatively correlated with feeding damage. Among the four PA groups, only the total concentration of the jacobine-like PAs was negatively correlated with feeding damage. Multiple regression tests suggested that jacobine-like PAs play a greater role in WFT resistance than PAs from other structural groups. We found no evidence for synergistic effects of different PAs on WFT resistance. The relationship between PA variation and WFT feeding damage in the Jacobaea hybrids suggests a role for PAs in resistance to generalist insects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In plants, research into the role of hybridization in the evolution of novel traits and new species is gaining momentum (Barton, 2001; Seehausen, 2004; Abbott et al., 2008). Hybrids have been used increasingly in experimental studies in ecology and evolution in part because interspecific hybrids (specifically segregating generations) often show greater variation in traits compared to parental species. Furthermore, segregating hybrids frequently show greater independence between different traits than the parental species (Hochwender et al., 2000; Lexer et al., 2003; Orians et al., 2010). Interspecific hybrids can have novel patterns of secondary chemical expression or accumulation compared to parental species, and sometimes can be more resistant or susceptible to herbivores than parental species (Rieseberg and Ellstrand, 1993; Fritz, 1999; Orians, 2000). This makes hybrids useful for studying the relationship between secondary metabolite variation and herbivores (Hallgren et al., 2003; Leiss et al., 2009).
Hybridization occurs frequently in the Jacobaea (syn. Senecio, Asteraceae) genus (Vincent, 1996). Members of this genus have been used extensively to study plant-herbivore interactions, which are largely mediated by a diverse group of pyrrolizidine alkaloids (PAs; see reviews by Hartmann, 1999; Macel, 2011). Twenty-six PAs have been reported from 24 Jacobaea species (Pelser et al., 2005). Pyrrolizidine alkaloids are ester alkaloids composed of a necine base (amino alcohol moiety) and an alkyl, or rarely aralkyl, necic acid (Hartmann, 1999). PAs can occur in two forms in vivo, the tertiary amine (free base) or the N-oxide form (Hartmann et al., 1989; Rizk, 1991; Wiedenfeld et al., 2008; Joosten et al., 2011). In Jacobaea species, all PAs except for senecivernine are derived from senecionine N-oxide, which is synthesized in the roots, transported to the shoots, and diversified into other PA structures (Hartmann and Toppel, 1987). Variation in PA structure and form can lead to variation in the performance of generalist insects and other plant enemies such as nematodes (van Dam et al., 1995; Macel et al., 2005; Dominguez et al., 2008; Thoden et al., 2009).
Jacobaea vulgaris (tansy ragwort or common ragwort, syn. Senecio jacobaea) is native to Europe and west Asia but invasive in North America, Australia and New Zealand. Jacobaea aquatica (marsh ragwort, syn. Senecio aquaticus) is closely related to, but not a sister species of, J. vulgaris (Pelser et al., 2003). Natural hybrids between these species occur in at least one location in The Netherlands (Kirk et al., 2004). The two parental species are attacked by different suites of specialist and generalist herbivores (personal observation). A previous study showed that artificial hybridization between these two species can be used to produce F2 lines that are in some cases extremely susceptible, and in other cases extremely resistant, to generalist herbivores (Leiss et al., 2009).
Western flower thrips, Frankliniella occidentalis (hereafter WFT), is a key insect pest on a wide range of agricultural and horticultural crops globally (Kirk and Terry, 2003). Since this species is highly polyphagous and infests about 200 wild and cultivated host species (Yudin et al., 1986), F. occidentalis often is used as a representative generalist herbivore in studies of plant-insect interactions (e.g., Macel et al., 2005; Leiss et al., 2009). Previous studies have investigated the effects of PAs on WFT with experiments that used artificial diets (Macel et al., 2005), or demonstrated the relationship between PAs and WFT resistance in host plants (Macel, 2003; Leiss et al., 2009). These studies showed that PAs are toxic to WFT and play a role in the plant resistance against this insect. However, these authors incorporated only a limited number of PAs in their studies. Some PAs were not easily acquired for experiments. Only the major PAs were quantified in host plants, and PAs were measured without discrimination between the two forms due to technical limitations in analytical methods. The effects of PA variation in host plant on WFT resistance have not yet been tested.
The current study aimed to overcome the challenges associated with isolating many PA variants for diet studies by measuring WTF resistance in a segregating hybrid family, which is expected to demonstrate great variation in composition and concentration of secondary metabolites such as PAs. Additionally, technological advances now permit the detection of PAs that are present in extremely low concentrations or that demonstrate only slight structural variations compared to other PAs, thus allowing us to test the relationship between WFT resistance and PA composition using a comprehensive set of PAs in vivo.
We carried out WFT bioassays with an artificial hybrid family including one J. vulgaris genotype, one J. aquatica genotype, two F1 offspring, and 94 different F2 hybrid lines. We measured WFT feeding damage in the shoots of these genotypes, and investigated the relationship between PA variation and susceptibility to attack by WFT in the segregating F2 generation. We addressed the following questions: 1) Is there variation in WFT resistance among segregating Jacobaea hybrids? 2) Is WFT resistance explained by PA concentration and composition? and if so, 3) Do different structural PA variants affect WFT resistance differently? 4) Are there any interactions between the effects of different PAs on WFT resistance?
Methods and Material
Study System and Plant Growth
Jacobaea vulgaris seeds (collected at Meijendel Nature Reserve, 52° 7' 54" N, 4° 19' 46" E, The Netherlands) and J. aquatica seeds (collected at the Zwanenwater Reserve, 52° 48' 38" N, 4° 41' 7" E, The Netherlands) were germinated in glass vials. Clones were produced from tissue cultured seedlings, and several clones were subsequently grown in pots in climate rooms under standard conditions of 20°C, 70% relative humidity, light: dark 16 h: 8 h (Cheng et al., 2011). Potted plants were vernalized at 4°C with the standard light and humidity conditions for approximately 10 weeks to facilitate flowering. Both species are self-incompatible and crosses were performed by rubbing flower heads together (Kirk et al., 2005). Two rayed F1 offspring were selected from this initial cross, and were reciprocally crossed with each other to produce two sets of F2 offspring. One F2 set consisted of 56 individuals and the other consisted of 46 individuals. The parental, F1 and F2 individuals were maintained in tissue culture and were cloned to perform experiments using replicate genotypes.
We grew about 6 cloned replicates per F2 genotype and about 12 cloned replicates per parental and F1 genotype for the WFT bioassay. In addition we grew the same number of replicates of the genotypes for PA analysis. The PA data for these genotypes were used both to study WFT resistance as described in this paper and for an analysis of patterns of PA profiles in Jacobaea hybrid plants that was published elsewhere (Cheng et al., 2011). The clones were individually potted in 1.3 l pots filled with 95% sandy soil (collected from Meijendel), 5% potting soil (Slingerland Potgrond company, Zoeterwoude, The Netherlands) and 1.5 g/l Osmocote slow release fertilizer (N:P:K = 15:9:11, Scott®, Scotts Miracle-Gro, Marysville, OH, USA). Plants were kept in a climate room under standard conditions described above for 6 wk before the bioassay was initiated.
WFT Bioassay
We used 12 replicates of each parental and F1 genotype, and 3–6 replicates of each of 94 F2 hybrid genotypes (6 replicates were used for most genotypes, though less than 6 were used in cases where plants died or were too small compared to other plants of the same genotype). A total of 587 plants were placed randomly in a climate room and grown under standard conditions. About 5870 adult WFT, previously reared on chrysanthemum (Dendranthema grandiflora), were released at evenly spaced points in the climate room. During the 3 wk feeding period, plants were watered every 2 d without wetting or disturbing the leaves. After 3 wk, silver damage caused by feeding from WFT on both upper and lower leaf surfaces was visually scored in mm2 for each leaf, according to the methods developed by Leiss et al. (2009). Above ground plant parts (shoots) were harvested just above the root crown and dried for 3 d in an oven at 50°C before establishing the dry masses of the shoots.
PA Data Acquisition
A Waters Acquity ultra performance liquid chromatographic system coupled to a Waters Quattro Premier XE tandem mass spectrometer (LC-MS/MS) (Waters, Milford, MA, USA) was used for PA analysis. Analysis was performed using a different set of the same tissue culture-derived clonal plants consisting of the same genotypes and number of clones as those used in the WFT bioassay, and grown under identical conditions. The plant shoots were harvested, stored at −80°C, and freeze-dried for 1 wk under vacuum with a collector temperature of −55°C. Dried plant material was ground to a fine powder and about 10 mg were extracted with 2% formic acid in a mass to volume ratio of 1:100. Heliotrine (Latoxan, Valence, France) was added as an internal standard to the extraction solvent at a concentration of 1 μg/ml. The extract was filtered through a 0.2 μm nylon membrane filter (Acrodisc, Pall Life Sciences, Ann Arbor, MI, USA). An aliquot (25 μl) of the filtered PA extract was diluted with water (975 μl) and injected into the LC-MS/MS system.
Seventeen individual PA standards were available for this study, representing the major PAs present in the plant extracts (Table 1). Senecionine, seneciphylline, retrorsine, and their corresponding N-oxides were obtained from Phytolab, Vestenbergsgreuth, Germany; senkirkine was obtained from Phytoplan, Heidelberg, Germany. Riddelliine and its N-oxide were obtained as authentic standards from Dr M. Chou (NCTR, Jefferson, AR, USA), and integerrimine was a gift of Dr. J. Trigo (UNICAMP, Campinas, Brasil). Jacobine and erucifoline were isolated from J. vulgaris plant material (PRISNA, Leiden, The Netherlands). The identity of the isolated standards was confirmed by 1H-NMR and LC-MS analysis and by comparison with literature data (Logie et al., 1994). Acetylseneciphylline was obtained by acetylation of seneciphylline with acetic anhydride and pyridine, according to the procedure described by He et al. (2010). Integerrimine N-oxide, jacobine N-oxide, erucifoline N-oxide, and acetylseneciphylline N-oxide were prepared by N-oxidation of the corresponding tertiary amine PAs according to the procedure described by Christie et al. (1949) and adapted by Chou et al. (2003). The purity of the obtained standards was checked by LC-MS analysis and was at least 90%.
The other PAs listed in Table 1 were tentatively identified on the basis of their retention time, molecular mass and fragmentation pattern, and on comparison with PA standards and literature data. The presence of PA N-oxides was confirmed by selective reduction to the corresponding tertiary amines according to the method of Joosten et al. (2010). All PAs included in this study have been reported before as constituents of J vulgaris and/or J aquatica (Hartmann and Witte, 1995; Joosten et al., 2011; Langel et al., 2011) and no new PAs were identified.
Data were recorded in multiple monitoring mode (MRM) using two selected precursor ions to product ion transitions per compound. The MS settings are shown in Table 1. For quantification, the sum of the two peak areas obtained for each compound was normalized against the peak area of the internal standard. Quantification was performed against a standard solution (100 μg/l) of the PAs in an extraction of tansy (Tanacetum vulgare), a plant known to be free of PAs. The use of a PA standard solution in blank plant extract was considered to be a more reliable approach than quantification against a PA solution in solvent only. This PA standard extraction was injected into every 30 samples and the averaged response was used for quantification. For those PAs without standards available, a semi quantitative (indicative) value was obtained by comparison with the most closely related analogue (e.g., an isomer) as indicated in Table 1. Data processing was conducted with Masslynx 4.1 software (Waters, Milford, MA, USA).
PA expression is genetically controlled under standard growth conditions, and PA production is not induced in shoots by aboveground herbivory in Jacobaea plants (Vrieling and Bruin, 1987; van Dam et al., 1993; Vrieling et al., 1993). Therefore, we averaged the concentration of each PA across all clones of each genotype and used the genotypic mean concentrations in the analyses presented here. The 37 PAs identified from the Jacobaea hybrids could be classified into four types, according to their structural characteristics, biosynthetic pathways, and expression pattern: senecionine-like PAs, jacobine-like PAs, erucifoline-like Pas, and otonecine-like PAs (Pelser et al., 2005; Cheng et al, 2011). We followed this classification in this study (Tables 1–2). The total PA concentration as well as the amount for each structural group was calculated by summing the concentrations of the individual PAs.
Data Analysis
We used general linear models to determine whether WFT resistance differed according to plant genotype. Feeding damage (dependent variable) was log- transformed to achieve normality, and plant genotype was defined as the independent variable with plant dry mass as covariate. Normal distributions and homogenous variances were confirmed by testing the residuals of the models using Shapiro tests and Bartlett tests respectively. Two models were set up: in the first, J. vulgaris was used as a reference, and in the second, J. aquatica was used. All other genotypes were compared to the reference in the model. Differences between the hybrid and parental genotypes were evaluated by using the regression coefficient matrices of the two models. In each matrix, the estimated coefficient of a hybrid indicated whether it had suffered more or less damage than the reference genotype, and the P value showed whether the difference was significant (Crawley, 2005). This is similar to a post-hoc test of an ANOVA model, however, such a post-hoc test includes all pair-wise comparisons between groups, and we were interested only in testing for differences between hybrid and parental genotypes. The difference between the two parental genotypes was also tested by using the same regression coefficient matrices. WFT resistance of each hybrid genotype was categorized according to these definitions: ND—no difference, leaf damage area of the hybrid was not different from that of both parents; A—additive, damage was intermediate between that of the parents; Ds—susceptible-dominant, damage was similar to that of the susceptible parent; Dr—resistant-dominant, damage was similar to that of the resistant parent, S—susceptible, damage was greater than that of the susceptible parent; R—resistant, damage was less than that of the resistant parent (Supplementary Table 1).
For correlation tests and principal components analysis, we included only data from F2 genotypes, since we were interested in using the variation from this segregating generation to search for underlying relationships between WTF resistance and PA expression. We were not able to test for differences in these relationships between the different generations described in this study because only a limited number of genotypes were included from the parental and F1 generations. However, the parental and F1 plants provided reference points for WFT resistance comparison. We used log-transformed genotypic mean values of feeding damage and PA concentrations to carry out correlation analyses. Either Spearman (for six minor PAs that did not have normally distributed concentrations) or Pearson correlation tests were carried out to test the relationship between feeding damage and the concentrations of individual PAs, pooled concentrations of each of the four PA groups and total PA (see details in Table 2 and Fig. 3).
Pyrrolizidine alkaloids from within structural groups were closely correlated with each other, and it was not possible, therefore, to investigate the interactions between them. The PAs from different structural groups, however, were generally expressed independently. The sum concentrations of the PAs from the four groups were not correlated with one another (Cheng et al., 2011). We used a multiple-regression model to test for interactions between the effects of different PA structural classes on feeding damage. In this model, feeding damage (represented by log-transformed genotypic mean values) was defined as the dependent variable, and the sum concentrations of each of the four PA structural groups (log-transformed and centered genotypic mean concentrations) were defined as independent variables.
The principal component analysis (PCA) was carried out by using log-transformed genotypic mean concentrations of all individual PAs except the six minor PAs that did not have normally distributed concentrations. Compared to the major PAs, these six PAs were present at very low concentrations (on average less than 1% of total PA concentration). Pearson correlation tests were carried out between the first six principle components (PCs) from the PCA and feeding damage. In order to evaluate the contribution of each PA to each PC (in other words the loading), Pearson correlation tests were carried out between individual PAs and the first 3 PCs, since PCs four to six accounted for a low proportion of the total variation and were not correlated with WFT feeding damage. The P-values were adjusted using the sequential Bonferroni method when multiple tests were carried out.
All analyses were conducted in R version 2.10.0 (R Development Core Team, 2009)
Results
Variation in Feeding Damage
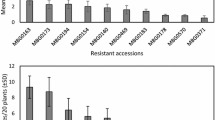
Feeding damage was genotype-dependent (d.f. = 97,488; F = 5.30; P < 0.001). Plant mass also had effect on feeding damage (d.f. = 1,488; F = 18.44; P < 0.001). Among the two parental genotypes, J. aquatica suffered more feeding damage than J. vulgaris (d.f. = 1, 22; t = 6.18; P < 0.001). Both of the F1 lines were as susceptible as J. aquatica. Among the 94 F2 hybrids, 69 were as susceptible as J. aquatica, 10 were more susceptible than J. aquatica, 15 showed intermediate resistance, 9 were as resistant as J. vulgaris, and none were more resistant than J. vulgaris (Fig. 1, see statistical details in Supplementary Table 1).
Variation in western flower thrips (WFT) feeding damage (mm2) on Jacobaea aquatica, Jacobaea vulgaris, 2 F1 and 94 F2 hybrids. a Mean feeding damage for one J. aquatica genotype (JA), one J. vulgaris genotype (JV), and 2 F1 (F1-A and F1 -B) genotypes. Error bars are standard errors, N = 12. J. vulgaris was significantly different from the other genotypes at * P < 0.05. b Distribution frequency for genotypic mean WFT feeding damage of 94 F2 hybrids. N = 3–6 for each genotype. In total, 587 plants were used in WFT bioassay
Relationship Between Feeding Damage and PA Concentration
Correlation tests between feeding damage and individual PAs showed that feeding damage was negatively correlated with the concentrations of the N-oxides of two jacobine-like PAs (jaconine and jacoline). Jacobine N-oxide concentration was marginally correlated with feeding damage, and the correlations between the free bases of jacobine-like PAs and feeding damage were not significant after correction for multiple testing. No other individual PAs were correlated with feeding damage (Table 2). Total PA concentration also was correlated with feeding damage (Fig. 2a). Of the four structural groups of PAs, only the sum concentration of jacobine-like PAs was significantly correlated with feeding damage by WFT (Fig. 2c). The sum concentrations of the other three groups were not correlated with feeding damage (see the statistical results for senecionine-and erucifoline-like PAs in Fig. 2b,d; for otosenine-like PAs: d.f. = 92, r = 0.35, P = 0.77).
Relationship between feeding damage by western flower thrips (WFT) (mm2) and the concentration of total pyrrolizidine alkaloid (PA), senecionine-like, jacobine-like, and erucifoline-like PAs (μg/g dw) of F2 hybrids of Jacobaea aquatica and Jacobaea vulgaris. The data for WFT feeding damage and concentrations are the log-transformed genotypic mean values. In each panel, the results of the Pearson correlation tests between feeding damage and the PA concentrations are provided; in all cases, d.f. = 92
The multiple regression models showed that among the four PA groups only jacobine-like PAs had significant negative effects on feeding. There were no two-way interactions between the groups. A three-way interaction between senecionine-like, jacobine-like and erucifoline-like PAs and an interaction between the four PA groups were present. However, these were only marginally significant (0.05 < P < 0.1, Table 3).
Relationships Between Feeding Damage and PA Composition
We used principal component analysis (PCA) to reduce the PA data set to a smaller number of uncorrelated axes. PC1 explained 44%, PC2 explained 19% and PC3 explained 12% of the variation in the data. More than 90% of the total variation was accounted for by the first 6 PCs. Among first 6 PCs, PC1 was negative correlated (d.f. = 92, r = −0.32, P = 0.002) and PC3 was positively correlated with feeding damage (d.f. = 92, r = −0.23, P = 0.04). No other PCs were correlated with feeding damage (data not shown). Correlation tests between each PC and individual PAs’ concentrations allowed us to identify which PAs were associated with each PC. Jacobine-like PAs (except jacozine and its N-oxide) were strongly correlated with PC1, such that individuals with high PC1 scores had high concentrations of jacobine-like PAs. Variation in some senecionine-like, erucifoline-like PAs and otosenine-like PAs contributed strongly to PC3 (individuals with high PC3 scores had low/high concentrations of these PAs; Supplementary Table 2). A plot of PC1 vs. PC2 shows that F2 hybrids can be divided roughly into different groups. Feeding damage, indicated by the size of the dots, is not clearly clustered either on the plot of PC1 vs. PC2 or the plot of PC1 vs. PC3 (Fig. 3).
Principal component analysis (PCA) of the pyrrolizidine alkaloid (PA) profiles of F2 hybrids of Jacobaea aquatica and Jacobaea vulgaris. PCA was performed on the log-transformed genotypic mean concentrations of all individual PAs excluding six minor PAs that did not have normally distributed concentrations (see Table 2). One dot represents one of 94 F2 hybrid genotypes. Size of each dot represents mean WFT feeding damage for that genotype. The genotypic mean concentrations are the average value of the three to six replicates from the same genotype
Discussion
Segregating hybrids are sometimes used to study correlations and trade-offs between different ecologically important traits in plants, because they exhibit greater variation than parental species, and greater independence between traits (e.g., Orians et al., 2010). We showed that there is high variation in the WFT susceptibility among F2 hybrids of J. vulgaris and J. aquatica. Although most F2 hybrids were as susceptible as or even more susceptible than J. aquatica (73% and 11% among all F2 hybrids respectively), there were still some hybrids with resistance similar to J. vulgaris (10%) or intermediate to the two parents (6%). The expression of PAs among the F2 hybrid generation was highly variable (Cheng et al., 2011, but also see Fig. 2 and Fig. 3), and this variation provided an opportunity to investigate the in vivo effects of PA composition on plant resistance to a generalist herbivore.
We demonstrated that concentrations of total PA and jacobine-like PAs were negatively correlated with feeding damage by using correlation tests. The multiple regression and PCA also indicated that concentrations of jacobine-like PAs were more closely related to WFT resistance than concentrations of the other PAs. The important role of jacobine-like PAs in WFT resistance of Jacobaea plants also has been supported by previous studies. Macel (2003) found that WFT feeding damage was negatively correlated with total PA concentration and with jacobine (both N-oxide and free base) concentration in J. vulgaris plants. Leiss et al. (2009) found that resistant Jacobaea hybrids had higher concentrations of jacobine N-oxide and jaconine N-oxide than susceptible hybrids. To develop a better understanding of the deterrent effects of different PAs on WFT, bioassays should be conducted using pure samples of different PAs.
Macel et al. (2005) tested WFT larval survival on artificial diets containing six individual PAs including senecionine, seneciphylline, retrorsine, senkirkine, heliotrine, and monocrotaline, or mixtures of senecionine, seneciphylline, and retrorsine. The experiment indicated that toxic effects of PAs on WFT larva differed among the individual PAs. Furthermore, higher PA concentrations had more potent toxic effects, and no synergistic effects resulted from PA mixtures. These findings support the results of our study, with the caveat that our analysis revealed a potential weak interaction between the different kinds of PAs. However, the interactions were slight (0.05 < P < 0.1, Table 3), and it is difficult to interpret interactions between more than two predictors.
PA variation accounted for a relatively low proportion of the variation in feeding damage (R 2 = 0.17, Table 3). Therefore, other factors likely play roles in plant susceptibility to WFT. These factors may include plant physical characteristics such as plant size, which was found to be a significant covariate in this study. Total PA concentration and plant size together explained a slightly higher proportion of the total variation (R 2 = 0.20). Other secondary metabolites have been reported from these species and their hybrids, including flavonoids, kaempferol glucoside, and chlorogenic acid (Leiss et al., 2009; Kirk et al., 2011), and other phytochemicals such as sesquiterpene lactones may be present but remain unreported. These metabolites may also play a role in resistance to herbivores, individually or in interaction with PAs.
References
Abbott, R. J., Ritchie, M. G., and Hollingsworth, P. M. 2008. Speciation in plants and animals: pattern and process—Introduction. Phil. Trans. R. Soc. Lond. B. 363:2965–2969.
Barton, N. H. 2001. The role of hybridization in evolution. Mol. Ecol. 10:551–568.
Cheng, D., Kirk, H., Mulder, P. P. J., Vrieling, K., and Klinkhamer, P. G. L. 2011. Pyrrolizidine alkaloid variation in shoots and roots of segregating hybrids between Jacobaea vulgaris and Jacobaea aquatica. New Phytol. doi: 10.1111/j.1469-8137.2011.03841.x.
Chou, M. W., Wang, Y. P.,Yan, J., Yang, Y. C., Beger, R. D., Williams, L. D., Doerge, D. R., and Fu, P. P. 2003. Riddelliine N-oxide is a phytochemical and mammalian metabolite with genotoxic activity that is comparable to the parent pyrrolizidine alkaloid riddelliine. Toxicology Letters. 145:239–247.
Christie, S. M. H., Kropman, M., Leisegang, E. C., and Warren, F. L. 1949. The senecio alkaloids. Part III. The structure of retrorsine and isatidine, and the isomerism of retronecic acid and isatinecic acid. J. Chem. Soc. 1700–1702.
Crawley, M. J. 2005. Statistics: An Introduction Using R. John Wiley & Sons, Ltd., Longdon.
Dominguez, D. M., Reina, M., Santos-Guerra, A., Santana, O., Agullo, T., Lopez-Balboa, C., and Gonzalez-Coloma, A. 2008. Pyrrolizidine alkaloids from Canarian endemic plants and their biological effects. Biochem. Syst. Ecol. 36:153–166.
Fritz, R. S. 1999. Resistance of hybrid plants to herbivores: genes, environment, or both? Ecology 80:382–391.
Hallgren, P., Ikonen, A., Hjalten, J., and Roininen, H. 2003. Inheritance patterns of phenolics in F1, F2, and back-cross hybrids of willows: implications for herbivore responses to hybrid plants. J. Chem. Ecol. 29:1143–1158.
Hartmann, T. 1999. Chemical ecology of pyrrolizidine alkaloids. Planta. 207:483–495.
Hartmann, T. and Toppel, G. 1987. Senecionine N-oxide, the primary product of pyrrolizidine alkaloid biosynthesis in root cultures of Senecio vulgaris. Phytochemistry 26:1639–1643.
Hartmann, T. and Witte, L. 1995. Pyrrolizidine alkaloids: Chemical, biological and chemoecological aspects, pp. 155–233, in S. W. Pelletier (eds.), Alkaloids: Chemical and Biological Perspectives, vol. 9. Pergamon Press, Oxford, UK.
Hartmann, T., Ehmke, A., Eilert, U., Von Borstel, K., and Theuring, C. 1989. Sites of synthesis, translocation and accumulation of pyrrolizidine alkaloid N-oxides in Senecio vulgaris L. Planta 177:98–107.
He, Y.-Q., Yang, L., Liu, H.X., Zhang, J.-W., Liu, Y., Fong, A., Xiong, A.-Z., Lu, Y.-L., Yang, L., Wang, C.-H., and Wang, Z.-T. 2010. Glucuronidation, a new metabolic pathway for pyrrolizidine alkaloids. Chem. Res. Toxicol. 23:591–599.
Hochwender, C. G., Fritz, R. S., and Orians, C. M. 2000. Using hybrid systems to explore the evolution of tolerance to damage. Evol. Ecol. 14:509–521.
Joosten, L., Mulder, P. P. J., Vrieling, K., Van Veen, J. A. V., and Klinkhamer, P. G. L. 2010. The analysis of pyrrolizidine alkaloids in Jacobaea vulgaris; a comparison of extraction and detection methods. Phytochem. Anal. 21:197–204.
Joosten, L., Cheng, D., Mulder, P. P. J., Vrieling, K., Van Veen, J. A. V., and Klinkhamer, P. G. L. 2011. The genotype dependent presence of pyrrolizidine alkaloids as tertiary amine in Jacobaea vulgaris. Phytochemistry 70:214–222.
Kirk, W. D. J., and Terry, L. I. 2003. The spread of the western flower thrips Frankliniella occidentalis (Pergande). Agric. For. Entomol. 5:301–310.
Kirk, H., Macel, M., Klinkhamer, P. G. L., and Vrieling, K. 2004. Natural hybridization between Senecio jacobaea and Senecio aquaticus: molecular and chemical evidence. Mol. Ecol. 13:2267–2274.
Kirk, H., Vrieling, K., and Klinkhamer, P. G. L. 2005. Reproductive fitness of hybrids between Senecio jacobaea and S. aquaticus (Asteraceae). Am. J. Bot. 92:1467–1473.
Kirk, H., Cheng, D., Choi, Y. H., Vrieling, K., and Klinkhamer, P. G. L. 2011. Transgressive segregation of primary and secondary metabolites in F2 hybrids between Jacobaea aquatica and J. vulgaris. Metabolomics doi: 10.1007/s11306-011-0301-8
Langel, D. Ober, D. and Pelser, P. B. 2011. The evolution of pyrrolizidine alkaloid biosynthesis and diversity in the Senecioneae. Phytochem. Rev. 10:3–74.
Leiss, K. A., Choi, Y. H., Abdel-Farid, I. B., Verpoorte, R., and Klinkhamer, P. G. L. 2009. NMR metabolomics of thrips (Frankliniella occidentalis) resistance in Senecio hybrids. J. Chem. Ecol. 35:219–229.
Lexer, C., Randell, R. A., and Rieseberg, L. H. 2003. Experimental hybridization as a tool for studying selection in the wild. Ecology 84:1688–1699.
Logie, C. G., Grue, M. R., and Liddell, J. R. 1994. Proton NMR speectroscopy of pyrrolizidine alkaloids. Phytochemistry, 37:43–109.
Macel, M. 2003. On the evolution of the diversity of pyrrolizidine alkaloids: The role of insects as selective forces. PhD Dissertation. University of Leiden.
Macel, M. 2011. Attract and deter: a dual role for pyrrolizidine alkaloids in plant–insect interactions. Phytochem. Rev. 1–8.
Macel, M., Bruinsma, M., Dijkstra, S. M., Ooijendijk, T., Niemeyer, H. M., and Klinkhamer, P. G. L. 2005. Differences in effects of pyrrolizidine alkaloids on five generalist insect herbivore species. J. Chem. Ecol. 31:1493–1508.
Orians, C. M. 2000. The effects of hybridization in plants on secondary chemistry: implications for the ecology and evolution of plant-herbivore interactions. Am. J. Bot. 87:1749–1756.
Orians, C. M., Hochwender, C. G., Fritz, R. S., and Snall, T. 2010. Growth and chemical defense in willow seedlings: trade-offs are transient. Oecologia 163:283–290.
Pelser, P. B., Gravendeel, B., and Van Der Meijden, R. 2003. Phylogeny reconstruction in the gap between too little and too much divergence: the closest relatives of Senecio jacobaea (Asteraceae) according to DNA sequences and AFLPs. Mol. Phylogenet. Evol. 29:613–628.
Pelser, P. B., De Vos, H., Theuring, C., Beuerle, T., Vrieling, K., and Hartmann, T. 2005. Frequent gain and loss of pyrrolizidine alkaloids in the evolution of Senecio section Jacobaea (Asteraceae). Phytochemistry 66:1285–1295.
R DEVELOPMENT CORE TEAM. 2009. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org
Rieseberg, L. H. and Ellstrand, N. C. 1993. What can molecular and morphological markers tell us about plant hybridization. Crit. Rev. Plant Sci. 12:213–241.
Rizk, A. M. 1991. Naturally Occurring Pyrrolizidine Alkaloids. Boca Raton, Florida, USA: CRC Press. 240 p.
Seehausen, O. 2004. Hybridization and adaptive radiation. Trends Ecol. Evol. 19:198–207.
Thoden, T. C., Boppre, M., and Hallmann, J. 2009. Effects of pyrrolizidine alkaloids on the performance of plant-parasitic and free-living nematodes. Pest Manage. Sci. 65:823–830.
Van Dam, N. M., Van Der Meijden, E., and Verpoorte, R. 1993. Induced responses in 3 alkaloid-containing plant-species. Oecologia. 95:425–430.
Van Dam, N. M., Vuister, L. W. M., Bergshoeff, C., De Vos, H., and Van Der Meijden, E. D. 1995. The 'raison d'etre' of pyrrolizidine alkaloids in Cynoglossum officinale deterrent effects against generalist herbivores. J. Chem. Ecol. 21:507–523.
Vincent, P. L. D. 1996. Progress on clarifying the generic concept of Senecio based on an extensive world-wide sample of taxa, pp. 597–611 in: D. Hind, H. Beentje (eds.), Compositae: Systematics. Proceedings of the International Compositae Conference. Royal Botanic Gardens, Kew.
Vrieling, K., and Bruin, J. 1987. Induction of pyrrolizidine alkaloids after artificial damage of rosette-plants of Senecio jacobaea L. Rijksuniversiteit Faculteit Landbouwwetenschappen Gent. 52: 1321–1326.
Vrieling, K., De Vos, H., and Van Wijk, C. A. M. 1993. Genetic analysis of the concentrations of pyrrolizidine alkaloids in Senecio Jacobaea. Phytochemistry 32:1141–1144.
Wiedenfeld, H., Roeder, E., Bourauel, T., and Edgar, J. 2008. Pyrrolizidine Alkaloids: Structure and Toxicity. V&R unipress GmbH, Bonn.
Yudin, L. S., Cho, J. J., and Mitchell, W. C. 1986. Host range of western flower thrips, Frankliniella occidentalis (Thysanoptera, Thripidae), with special reference to Leucaena glauca. Environ. Entomol. 15:1292–1295.
Acknowledgements
Dandan Cheng thanks the China Scholarship Council (CSC) of the Ministry of Education for financial support. We thank Cilke Hermans, Karin van der Veen-van Wijk and Henk Nell for technical assistance in plants growth and WFT bioassay; Lotte Joosten for help in PA extraction, measurements, and analysis; Kirsten Leiss, Mohammed Mirnezhad, Kelly Stolk and Anniek de Vreede for help in feeding damage scoring. We also thank two anonymous reviewers for suggestions on manuscript writing.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Table 1
General linear models of the thrips resistance indicator (thrips feeding damage, mm2, for the models: d.f. = 97, 489; F = 5.30; P < 0.001) (DOC 185 kb)
Supplemental Table 2
Statistics results of correlation tests between the first three PCs and individual pyrrolizidine alkaloids (PAs) from PCA in the shoots of 94 F2 hybrid genotypes of Jacobaea aquatica, Jacobaea vulgaris (DOC 63 kb)
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Cheng, D., Kirk, H., Vrieling, K. et al. The Relationship between Structurally Different Pyrrolizidine Alkaloids and Western Flower Thrips Resistance in F2 Hybrids of Jacobaea vulgaris and Jacobaea aquatica . J Chem Ecol 37, 1071–1080 (2011). https://doi.org/10.1007/s10886-011-0021-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-011-0021-6