Abstract
The subject of the study was silicate–phosphate glasses of NaCaPO4–SiO2 system which are precursors of glass–crystalline materials. Glass–crystalline materials of NaCaPO4–SiO2 system obtained via crystallization of glasses belong to a group of the so-called bioactive materials. In order to obtain glass–crystalline materials with pre-established parameters, it is necessary to conduct crystallization of glasses at specific conditions. In order to design direct crystallization process properly, it is necessary to know the structure and microstructure of the glassy precursor. Microscopic investigation showed that liquation takes place in all the studied glasses. Based on DSC examinations, it has been found out that crystallization of the glasses of NaCaPO4–SiO2 system is a multistep process. The presence of several clearly separated exothermic peaks in DSC curves of investigated glasses makes it possible to crystallize only the separated phase with the matrix remaining amorphous or vice versa. Conducted detailed X-ray and spectroscopic studies of the materials obtained by heating in a gradient furnace (in the temperature specified on the basis of DSC) showed that separated phase and matrix crystallizes separately. Therefore, bioactive glass–crystalline materials can be obtained due to the existence of the phase separation phenomenon and pre-established sizes of the crystalline phase.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Glasses and glass–crystalline materials of NaCaPO4–SiO2 system belong to a group of bioactive materials, capable of forming bonds with the tissue (e.g., [1–10]). The use of glass as a biomaterial makes it possible to take the advantage of specific properties of the glassy state, i.e., ease of obtaining practically any shape, ease of controlling properties by proper choice of chemical composition, possibility to apply various processing methods, as well as isotropic properties. However, the main feature of the silicate–phosphate glasses is also its fragility, which significantly limits their use as biomaterials. One of the best ways to improve the mechanical properties of glasses is to perform partial crystallization (devitrification) in order to obtain glass–crystalline materials. Such materials are characterized by the presence of very fine crystals of the crystalline phase, randomly distributed in the rest of the glassy matrix [11, 12]. This allows to combine advantages of both glassy and crystalline materials (high mechanical strength). As a result, the glass–crystalline materials are characterized by much higher mechanical strength compared to the glassy precursors. However, a problem arises because the growth of crystalline phases very adversely affects the bioactivity of the glasses [13, 14]. In the extreme case, uncontrolled crystallization can lead to the conversion of bioactive glass into completely inert material [14]. Thus, it is necessary to provide full control over the process. To design such process properly, it is necessary to know the structure and microstructure of the glassy precursors. Our previous studies concerning the structure and microstructure of glasses of XCaPO4–SiO2 system (where X = Na+ and or K+) have shown that glass phase separation exist in all obtained materials [15, 16].
We think that problems with uncontrolled growth of crystalline phases can be reduced in the silicate–phosphate glasses by using a glass phase separation phenomenon. The boundaries of separated phase–matrix phase may be a barrier limiting the growth of crystalline phases.
The main objective of this study was to determine the effect of NaCaPO4 content on the crystallization process and thermal resistance of glasses of NaCaPO4–SiO2 system.
Experimental
Synthesis of glasses is described in detail in our earlier article [17]. Heating of glasses at temperatures determined on the basis of thermal research (DSC) was carried out in a gradient furnace. The horizontal tube furnace (electric heating) with siliconite heating elements was used. Siliconite layer thickness is controlled temperature distribution, which is solid, wherein there is a maximum temperature in the center of the device and decreases progressively toward the ends of the tube. Pieces of glass was placed in a boat with kaolin-coated heat-resistant steel and then inserted into previously heating tube for 2 h. The highest temperature for the center of the furnace was determined from the differential scanning calorimetry (DSC) curve of glass (the maximum temperature of crystallization). The samples (after heating for 2 h) were then drawn from the furnace together with the boat and cooled.
The next step was to read the temperature at every 1 cm points along the axis of the furnace and determine the temperature distribution.
In order to identify the type of crystallizing phases during the crystallization process the detailed X-ray studies of the materials received after the devitrification have been carried out. XRD data were collected by PANalytical X’Pert Pro MD powder diffractometer using Kalfa1 radiation from Cu anode. The configuration was standard Bragg–Brentano setup with Ge (111) monochromator at the incident beam. All measurements were performed with the 0.008 step size at 3–75 scanning range and the 205 s of measurement time for each step. Measurements were carried out under room temperature and ambient pressure.
Middle infrared (MIR) spectroscopic measurements of the glasses were made with a Bio-Rad FTS 60V spectrometer. Transmission technique, samples as KBr pellets. Spectra were collected after 124 scans at 4 cm−1 resolution.
All glasses were subjected to DSC measurements conducted on Netzch STA 449 F1 Jupiter, operating in the heat flux DSC mode. Glass samples weighing 35 mg were heated in platinum crucibles at a rate of 10 °C min−1 in a dry nitrogen atmosphere up to 1,000 °C. The obtained samples were refined to the grain size below 0.1 mm.
Results and discussion
Glasses belonging to NaCaPO4–SiO2 systems whose composition are given in Table 1 have been selected for the studies. The composition was selected to compensate the negative charge of [PO4]3− and [SiO4]4− by Na+ and Ca2+ cations.
EDX studies showed that the chosen glasses separated phase and the matrix chemical compositions differ significantly among themselves [15, 16]. They must therefore also have different crystallization temperature. Structural studies (MIR, MAS NMR) revealed that both separated phase and matrix are characterized by a domain structure [18]. As it was mentioned above, knowledge of microstructure and structure of glasses provides a basis for the design of direct crystallization process. Of course, in order to plan the direct crystallization process it is necessary to determine characteristic temperatures for the glassy state, in particular the devitrification temperature.
The results of thermal studies (DSC) of glasses are presented in Figs. 1, 2, 3, 4, and 5. Examination of the DSC curves can trace the effect of the NaCaPO4 content on the thermal resistance and the process of crystallization of the glasses of NaCaPO4–SiO2 system. As you can see the crystallization of investigated glasses (except of 1NA glass) takes place in two stages–peaks in the range of 826–917 and 892–964 °C. One step crystallization process of 1NA glass is related to the almost invisible phase separation [15, 16] and very small amount of NaCaPO4. DSC curves of 2Na—5NA glasses are all alike—which demonstrate that the crystallization of these glasses is very similar. Earlier reported glass phase separation suggests that the matrix and separated phase crystallize separately. The increased amount of NaCaPO4 in liquidation silicate–phosphate glasses of the tested systems (2Na–5NA) results only in increased heat resistance of glasses. There is a clear, systematic shift of peaks to higher temperatures (Table 2), reflecting the increasing resistance to crystallization. To determine in detail the crystallization process of tested glasses, the direct crystallization process (in gradient furnace) of selected glass (2Na) was conducted.
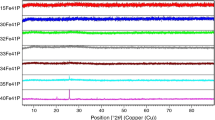
The 2Na glass has been selected for detailed studies because it is characterized by the presence of spherical nano-sized separated phase [15, 16]. The DSC curve of glass 2Na (Fig. 2) shows two distinct peaks (at 826 and 892 °C), so heating process was performed at 800, 825, 870, and 890 °C. After the heating process, obtained materials were analyzed by MIR and XRD methods. Figure 6 shows the MIR spectra of the starting glass and materials received after the heating process. Assignment of the individual bands to the respective types of vibration is shown in Table 3. On the basis of earlier detailed spectroscopic studies of model glasses, it was found that in the glasses of NaCaPO4–SiO2 system the glass matrix has a mixed domain structure—there are domains of NaCaPO4 and cristobalite structure (submicroheterogenity). While separated phase have a homogeneous domain structure—only domains of cristobalite structure occur [18]. According to Görlich [19], devitrification of glasses is based on reorientation of domains during heating. Thus domains can be treated as crystallization nuclei, which will decide of the type of crystallizing phase [17]. This information allows to precisely describe the devitrification process. Analyzing MIR spectra shown in Fig. 6, it can be said that the phosphate phase crystallizes as the first one. Characteristic bending vibrations O–P–O in phospho-oxygen tetrahedrons at about 575 cm−1 appear at 800 °C and then its intensity increase at 825 and 870 °C [20]. In connection with the previously presented information it can be concluded that the phosphate phase crystallized only from the matrix—phosphate phase was present only in the matrix of glass. Further heat treatment (at 890 °C) results in much sharpens of band at about 800 cm−1, and appear of additional bands at about 610 and 1,197 cm−1. These bands are characteristic for cristoballite [21]. As it can therefore be concluded the cristoballite crystallized in the second step—separated phase and the remainder of the matrix crystallized. To accurately determine the type of crystallizing phases, X-ray studies were performed. Figure 7 shows a X-ray patterns of glasses heated at appropriate temperatures. X-ray patterns show mainly amorphous halo and a few weak reflections in the 30–35° 2θ. The phase analysis has allowed to link this reflections with phosphate phase (NaCaPO4). As you can see in the case of glasses heated at temperatures of 800, 825, and 870 °C, we get a glass–crystalline material. This confirms that, in the case of investigated glasses, in the first stage phosphate phase (NaCaPO4) crystallized from the matrix. The heating of glass at 890 °C leads to obtain a completely crystalline material, in which the dominant phase is cristoballite. It can be concluded that properly conducted crystallization can lead to partial crystallization of the matrix in complete amorphous of separated phase.
Conclusions
Thermal studies of liquation silicate–phosphate glasses of NaCaPO4–SiO2 system allow to conclude that crystallization of glasses of this system, containing more than 20 % NaCaPO4 is a multistep process. Crystallization of all glasses in which, there is the phenomenon of phase separation is very similar—the two-stage crystallization. The increase of NaCaPO4 amount in studied glasses causes a significant increase of their resistance to crystallization.
Structural studies of materials obtained after heating in gradient furnace shown that the first one to crystallize was the glass matrix (phosphate phase—NaCaPO4), then the separated phase and the remainder of the matrix (cristobalite). Thus, based on the investigated glasses it is possible to obtain glass–crystalline materials by appropriate selection of crystallization conditions.
References
Hench LL, Splinter RJ, Greenlee TK, Allen WC. Bonding mechanism at the interface of ceramic prosthetic materials. J Biomed Res. 1971;2:117–41.
Bellantone M, Coleman NJ, Hench LL. Bacteriostatic action of a novel four-component bioactive glass. J Biomed Mat Res. 2000;51:484–90.
Hill RG, Stamboulis A, Law RV, Clifford A, Towler MR, Crowley C. The influence of strontium substitution in fluorapatite glasses and glass ceramics. J Non-Cryst Solids. 2004;336:223–9.
Lusvardi G, Malavasi G, Menabue L, Menziani MC, Pedone A, Segre U. Density of multicomponent silica-based potential bioglasses: quantitative structure-property relationships (QSPR) analysis. J Eur Ceram Soc. 2007;27:499–504.
Lusvardi G, Malavasi G, Menabue L, Aina V, Morterra C. Fluoride-containing bioactive glasses: surface reactivity in simulated body fluids solutions. Acta Biomater. 2009;5:3548–62.
Kansal I, Tulyaganov DU, Goel A, Pascual MJ, Ferreira JMF. Structural analysis and thermal behavior of diopside-fluor apatite-wollastonite-based glasses and glass-ceramics. Acta Biomater. 2010;6:4380–8.
Fredholm YC, Karpukhina N, Law RV, Hill RG. Strontium containing bioactive glasses: glass structure and physical properties. J Non-Cryst Solids. 2010;356:2546–51.
Aina V, Bertinetti L, Cerrato G, Cerruti M, Lusvardi G, Malavasi G, Morterra C, Tacconi L. On the dissolution/reaction of small-grain bioglass 45S5 and F-modified bioactive glasses in artificial saliva (AS). Appl Surf Sci. 2011;257:4185–95.
Brauer DS, Mneimne M, Hill RG. Fluoride-containing bioactive glasses: fluoride loss during melting and ion release in tris buffer solution. J Non-Cryst Solids. 2011;357:3328–33.
Goel A, Rajagopal RR, Ferreira JMF. Influence of strontium on structure, sintering and biodegradation behaviour of CaO–MgO–SrO–SiO2–P2O5–CaF2 glasses. Acta Biomate. 2011;7:4071–80.
Stand Z. Glass-ceramic materials. Amsterdam: Elsevier; 1986.
Holand W, Beall G. Glass-ceramic technology. Ohio: The American Ceramic Society; 2002.
Peitl O, Torre G, Hench LL. Effect of crystallization on apatite-layer formation of bioactive glass 45S5. J Biomed Mater Res. 1996;30:509–14.
Li P, Zhang F, Kokubo TJ. The effect of residual glassy phase in a bioactive glass-ceramic on the formation of its surface apatite layer in vitro. Mater Sci Mater Med. 1992;3:452–6.
Sitarz M, Rokita M, Handke M, Galuskin EW. Structural studies of the NaCaPO4–SiO2 sol-gel derived materials. J Mol Struct. 2003;651–653:489–98.
Sitarz M. Influence of modifying cations on the structure and texture of silicate–phosphate glasses. J Mol Struct. 2008;887:237–48.
Sitarz M, Szumera M. Crystallization of silico-phosphate glasses. J Therm Anal Calorim. 2008;91:255–60.
Sitarz M. The structure of liquation silico-phosphate glasses. J Mol Struct. 2008;887:229–36.
Görlich E. Glassy state. Cracow: AGH no. 155; 1989 (in Polish).
Jastrzębski W, Sitarz M, Rokita M, Bułat K. Infrared spectroscopy of different phosphates structures. Spectrochim Acta Part A. 2011;79:722–7.
Sitarz M, Handke M, Mozgawa W. Identification of silicooxygen rings in SiO2 based on IR spectra. Spectrochim Acta Part A. 2000;56:1819–23.
Acknowledgments
The project was funded by the National Science Center awarded on the basis of the decision number DEC-2011/01/N/ST8/07425.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Sitarz, M., Bulat, K., Wajda, A. et al. Direct crystallization of silicate–phosphate glasses of NaCaPO4–SiO2 system. J Therm Anal Calorim 113, 1363–1368 (2013). https://doi.org/10.1007/s10973-013-3240-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-013-3240-y