Abstract
The role played by the polymorphism located in Toll-like Receptor 9 (TLR9) as a risk factor of cervical cancer remains elusive. Therefore, we studied the association of the TLR9 −1486 T/C (rs187084) and C2848T (rs352140) polymorphisms with cervical cancer. The TLR9 −1486 T/C and C2848T polymorphism was genotyped in 426 patients and 460 unrelated healthy females from the Polish population. Logistic regression analysis adjusting for age, pregnancy, oral contraceptive use, tobacco smoking, and menopausal status showed that both the TLR9 −1486 T/C and C2848T polymorphisms could be a genetic risk factor for cervical cancer. For the TLR9 −1486 T/C polymorphism, the adjusted OR for patients with the C/T genotype versus T/T genotype was 1.371 (95 % CI 1.021–1.842, p = 0.0361), the adjusted OR for the C/C genotype vs the T/T genotype was 1.300 (95 % CI 1.016–1.507, p = 0.0096), and the adjusted OR for the C/T or C/C genotype vs the T/T genotype was 1.448 (95 % CI 1.099–1.908, p = 0.0083). For the C2848T polymorphism, the adjusted OR for patients with the C/T genotype vs C/C genotype was 1.443 (95 % CI 1.019–2.043, p = 0.0380), the adjusted OR for the T/T genotype vs the C/C genotype was 1.237 (95 % CI 1.016–1.507, p = 0.0328), and the adjusted OR for the T/C or T/T genotype vs the C/C genotype was 1.345 (95 % CI 0.976–1.855, p = 0.0700). Our studies suggest that the TLR9 −1486 T/C and C2848T polymorphisms may be a genetic risk factor for cervical cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cervical carcinogenesis is a multi-step process associated with refractory infection by high-risk human papillomavirus (HPV) types [1, 2]. This includes the transformation of normal cervical epithelium to cervical intraepithelial neoplasia (CIN), which is transformed to invasive cervical carcinoma [1, 2]. The HPV oncoproteins E6 and E7 play a key role in cervical carcinogenesis via the disturbance of apoptosis, the cell cycle, and adaptive immune surveillance [3]. Only a minority of HPV infected women will develop CIN or cervical cancer, suggesting that HPV is not a sufficient separate factor responsible for tumorigenesis of the cervix [4]. Many epidemiological studies have also indicated that environmental factors, contraceptive use, smoking, and genetic factors may also contribute to cervical carcinogenesis [5–9].
Recently, several studies have demonstrated that HPV infections may change the expression of toll-like receptors (TLRs), which interferes with TLRs signaling during early and late malignant transformation of the cervix [10–13]. TLRs recognize exogenous pathogen-associated molecular patterns and they play an elementary role in the innate immune response [14].
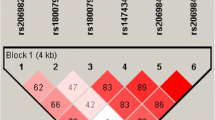
In humans, the family of TLRs includes ten members, which can be divided based on their localization in the cell [14]. TLR1, TLR2, TLR4, TLR5, TLR6, and TLR10 are usually situated on the cell surface, whereas TLR3, TLR7, TLR8, and TLR9 are localized almost exclusively in endosomes [14–17]. TLR9 have been shown to recognize unmethylated cytosine–phosphate–guanine (CpG) dinucleotide motifs located in bacterial, viral and fungal DNA [17, 18]. TLR9 is present in macrophages, dendritic cells and intestinal epithelium, as well as respiratory epithelial and keratinocytes cells [ 16, 17, 19–21 ]. It has been demonstrated that the −1486 T/C (rs187084), −1237T/C (rs5743836) and C2848T (rs352140) polymorphisms of TLR9 located in the same block of linkage disequilibrium (LD) may change TLR9 expression (HapMap CEU data http://hapmap.ncbi.nlm.nih.gov/) [22–24].
There are two studies on the association of the TLR9 1486 T/C (rs187084) and C2848T (rs352140) polymorphisms with cervical cancer in various populations, however, the results are inconsistent [25, 26]. Therefore, we aimed to study whether TLR9 −1486 T/C (rs187084) and C2848T (rs352140) can be a genetic risk factor of cervical cancer in the Polish population.
Patients and methods
Patients and controls
The patient group was composed of 426 women with histologically determined cervical carcinoma according to the International Federation of Gynecology and Obstetrics (FIGO). All women were enrolled between April 2007 and July 2011 at the Department of Radiotherapy, Greater Poland Cancer Center in Poznan, Poland (Table 1). The controls included four hundred sixty unrelated healthy female volunteers who were matched by age to the patients (Table 1). Information about pregnancy, oral contraceptive use, tobacco smoking, and menopausal status was obtained as part of the patient history. Patients and controls were Caucasian, enrolled from the Wielkopolska area of Poland. All subjects provided written informed consent. The study was approved by the Local Ethical Committee of Poznan University of Medical Sciences.
Genotyping
DNA was obtained from peripheral leucocytes using a standard salting out procedure. Identification of the TLR9 -1486 T/C (rs187084) and C2848T (rs352140) polymorphic variant was performed by polymerase chain reaction-restriction fragment length polymorphism (PCR–RFLP). PCR was conducted employing primer pair 5′TTCATTCATTCAGCCTTCACTCA 3′, 5′ GAGTCAAAGCCACAGTCCACA 3′ and 5′ GCAGCACCCTCAACTTCACC 3′and 5′ GGCTGTGGATGTTGTTGTGG 3′, respectively.
The PCR-amplified fragments 565 bp in length bearing the TLR9 -1486 T/C (rs187084) polymorphism were digested with restriction enzyme Afl II (C/TTAAG) New England BioLabs, (Ipswich, USA). The TLR9 T allele was cleaved into 416 and 149 bp fragments, whereas the TLR9 C allele remained uncut. The PCR-amplified fragments 360 bp in length corresponding to the TLR9 C2848T polymorphism were subjected to digestion with the endonuclease BstUI (CG/CG) New England BioLabs, (Ipswich, USA). The TLR9 C allele was cleaved into 227 and 133 bp fragments, whereas the TLR9 T allele remained uncut. DNA fragments were separated by electrophoresis on 3 % agarose gel and visualized by ethidium bromide staining. The TLR9 1486 T/C and C2848T polymorphism was confirmed by repeated PCR–RFLP. Moreover, the restriction analysis was confirmed by commercial sequencing analysis.
Statistical analysis
The distribution of genotypes in patients and controls was examined for deviation from Hardy–Weinberg equilibrium using exact and log likelihood ratio χ2 tests [http://ihg.gsf.de/cgi-bin/hw/hwa1.pl]. The polymorphism was tested for association with cervical cancer using the χ2 test for trend (ptrend). The χ2 test was employed to examine differences in genotypic and allelic distribution between patients and controls. The odds ratio (OR) and 95 % confidence intervals (95 % CI) were calculated. Unconditional logistic regression analysis was used to adjust for the effect of confounders such as age, pregnancy, oral contraceptive use, tobacco smoking, and menopausal status. A p value of <0.05 was considered statistically significant.
Results
Prevalence of the TLR9 −1486 T/C polymorphism in women with cervical cancer
There was a higher frequency of the TLR9 CC genotype in women with cervical cancer compared to healthy individuals, which was 0.19 and 0.14, respectively (Table 2). We also found increased TLR9 C/T heterozygote frequency in patients than in controls, which was 0.48 and 0.44, respectively (Table 2). There was also an increased TLR9 C allele frequency in patients than in controls, which was 0.43 and 0.36, respectively (Table 2). The p value of the χ2 test of the trend observed for the TLR9 −1486 T/C polymorphism was statistically significant (ptrend = 0.0042). Logistic regression analysis showed a significant contribution of the TLR9 −1486 T/C polymorphism to cervical cancer (Table 2). The adjusted OR for patients with the C/T genotype vs T/T genotype was 1.371 (95 % CI 1.021–1.842, p = 0.0361), the adjusted OR for the C/C genotype vs the T/T genotype was 1.300 (95 % CI 1.016–1.507, p = 0.0096), and the adjusted OR for the C/T or C/C genotype vs the T/T genotype was 1.448 (95 % CI 1.099–1.908, p = 0.0083) (Table 2).
Prevalence of the TLR9 C2848T polymorphism in women with cervical cancer
We observed an increased frequency of the TLR9 TT genotype in patients than controls, which was 0.26 and 0.22, respectively (Table 2). The TLR9 C/T heterozygote frequency in women with cervical cancer was also increased compared to healthy individuals and amounted to 0.54 and 0.51, respectively (Table 2). We also observed a higher TLR9 T allele frequency in patients then healthy individuals, which was 0.53 and 0.48, respectively (Table 2). The p value of the χ2 test of the trend observed for the TLR9 C2848T polymorphism was statistically significant (ptrend = 0.0449). Logistic regression analysis showed a significant association of the TLR9 C2848T polymorphism with cervical cancer (Table 2). The adjusted OR for patients with the C/T genotype vs C/C genotype was 1.443 (95 % CI 1.019–2.043, p = 0.0380), the adjusted OR for the T/T genotype vs the C/C genotype was 1.237 (95 % CI 1.016–1.507, p = 0.0328), and the adjusted OR for the T/C or T/T genotype vs the C/C genotype was 1.345 (95 % CI 0.976–1.855, p = 0.0700) (Table 2).
Discussion
TLR9 plays a crucial role in pathogen recognition and activation of innate immunity [15, 18]. Stimulation of TLR9 activates human B cells and plasmacytoid dendritic cells, causing T helper-1 type immune responses and antitumor responses [18, 27–31]. The role of the TLR9 pathway in anticancer treatment has been considered in animal models and patients with renal carcinoma, malignant melanoma, and non-Hodgkin’s lymphoma [27–31]. Moreover, the significance of the TLR9 pathway has been demonstrated in vaccine treatment of cervical carcinoma in murine model [28]. Mansilla et al. [28] demonstrated that intratumoral administration of EDA-HPVE7 fusion protein in combination with TLR9 ligand CpG-B was able to eradicate large established cervical tumors.
HPV infects primitive basal keratinocytes; however, its abundant expression and viral assembly take place only in the upper layers of the stratum spinosum and granulosum of squamous epithelia [32]. Keratinocytes bear TLR9, and stimulation of TLR9 may result in the production of a spectrum of mediators influencing the function of immune cells [21]. Some studies have suggested that changes occur in TLR9 expression during cervical carcinogenesis [11, 13]. Recently, Hasimu et al. (2007) found that the expression of TLR9 can be up-regulated by HPV16 infection in CIN and in cervical squamous carcinoma cells [11]. They also suggested that TLR9 may play important roles in the development and progression of CIN and cervical carcinoma [11]. Adding to the above findings are those of Hasan et al. [13], who demonstrated that HPV16 infection of human primary keratinocytes reduced TLR9 transcription and lead to a functional loss of TLR9-regulated pathways. This may suggest that polymorphisms of TLR9 that modulate the expression of TLR9 may have an effect on the etiopathogenesis of cervical cancer [33].
We observed a contribution of the TLR9 −1486 T/C (rs187084) and C2848T (rs352140) polymorphisms to the risk of cervical cancer in a Polish population. Recent studies conducted by Chen et al. [25] demonstrated that the TLR9 −1486 T/C (rs187084) polymorphism, located in the LD block with rs352140, was associated with a significantly increased risk of cervical cancer. In contrast, Pandey et al. [26] showed that the TT genotype of TLR9 (rs352140) displayed borderline significance in increased risk for advanced cervical cancer in a North India population.
These differences in the effect of TRL9 polymorphism on the susceptibility to cervical cancer development between our and Hindu populations may result from racial heterogeneity, the size of the studied groups, and the action of distinct behavioral and environmental factors [33].
To date, the TLR9 C2848T (rs352140) polymorphism has also been associated with Hodgkin’s lymphoma, periodontitis, ulcerative colitis, and systemic lupus erythematosus [34–37]. The other TLR9 variants have also been found to be risk factors of atopic eczema, tuberculosis, Helicobacter pylori-induced gastritis, and rheumatoid arthritis [38–41]. Moreover, these TLR9 variants may influence the clinical course of HIV-1 infection, and the development of endometrial cancer, osteoarthritis, and non-Hodgkin lymphoma [42–45].
The role of the rs352140 polymorphism on TLR9 expression has been suggested by Kikuchi et al. [24],who demonstrated that the TLR9 2848 TT genotype was associated with a higher expression of TLR9 and an increased frequency of IgM + B cells. It has been believed that chronic inflammation may lead to cancer development and progression (33). The increased expression of the TLR9 2848 T variant in precursor malignant lesion cells combined with infection by various pathogens might support inflammation and cervical cancer development (33).
Our studies suggest that the C2848T (rs352140) polymorphism might be a risk factor of cervical cancer in Polish women. Our genetic evaluation is the first in a Caucasian cohort; therefore this study should be replicated in a larger and independent cohort.
References
Walboomers JM, Jacobs MV, Manos MM et al (1999) Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol 189:12–19
Muñoz N, Bosch FX, de Sanjosé S, Herrero R, Castellsagué X, Shah KV et al (2003) Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med 348:518–527
Münger K, Howley PM (2002) Human papillomavirus immortalization and transformation functions. Virus Res 89:213–228
Giuliano AR, Harris R, Sedjo RL, Baldwin S, Roe D, Papenfuss MR et al (2002) Incidence, prevalence, and clearance of type-specific human papillomavirus infections: the Young Women’s Health Study. J Infect Dis 186:462–469
Magnusson PK, Sparen P, Gyllensten UB (1999) Genetic link to cervical tumours. Nature 400:29–30
Castellsague X, Munoz N (2003) Chap. 3: Cofactors in human papillomavirus carcinogenesis–role of parity, oral contraceptives, and tobacco smoking. J Natl Cancer Inst Monogr 20–28
Magnusson PK, Lichtenstein P, Gyllensten UB (2000) Heritability of cervical tumours. Int J Cancer 88:698–701
Hemminki K, Chen B (2006) Familial risks for cervical tumors in full and half siblings: etiologic apportioning. Cancer Epidemiol Biomarkers Prev 15:1413–1414
Moreno V, Bosch FX, Muñoz N, Meijer CJ, Shah KV, Walboomers JM et al (2002) Effect of oral contraceptives on risk of cervical cancer in women with human papillomavirus infection: the IARC multicentric case-control study. Lancet 359:1085–1092
DeCarlo CA, Rosa B, Jackson R, Niccoli S, Escott NG, Zehbe I (2012) Toll-like receptor transcriptome in the HPV-positive cervical cancer microenvironment. Clin Dev Immunol 2012:785825
Hasimu A, Ge L, Li QZ, Zhang RP, Guo X (2011) Expressions of Toll-like receptors 3, 4, 7, and 9 in cervical lesions and their correlation with HPV16 infection in Uighur women. Chin J Cancer 30:344–350
Kim WY, Lee JW, Choi JJ, Choi CH, Kim TJ, Kim BG et al (2008) Increased expression of Toll-like receptor 5 during progression of cervical neoplasia. Int J Gynecol Cancer 18:300–305
Hasan UA, Bates E, Takeshita F, Biliato A, Accardi R, Bouvard V et al (2007) TLR9 expression and function is abolished by the cervical cancer-associated human papillomavirus type 16. J Immunol 178:3186–3197
Akira S, Takeda K (2004) Toll-like receptor signalling. Nat Rev Immunol 4:499–511
Ahmad-Nejad P, Häcker H, Rutz M, Bauer S, Vabulas RM, Wagner H (2002) Bacterial CpG-DNA and lipopolysaccharides activate Toll-like receptors at distinct cellular compartments. Eur J Immunol 32:1958–1968
Heil F, Ahmad-Nejad P, Hemmi H, Hochrein H, Ampenberger F, Gellert T et al (2003) The Toll-like receptor 7 (TLR7)-specific stimulus loxoribine uncovers a strong relationship within the TLR7, 8 and 9 subfamily. Eur J Immunol 33:2987–2997
Latz E, Schoenemeyer A, Visintin A, Fitzgerald KA, Monks BG, Knetter CF et al (2004) TLR9 signals after translocating from the ER to CpG DNA in the lysosome. Nat Immunol 5:190–198
Hemmi H, Takeuchi O, Kawai T, Kaisho T, Sato S, Sanjo H et al (2000) A Toll-like receptor recognizes bacterial DNA. Nature 408:574–740
Schmausser B, Andrulis M, Endrich S, Lee SK, Josenhans C, Müller-Hermelink HK et al (2004) Expression and subcellular distribution of toll-like receptors TLR4, TLR5 and TLR9 on the gastric epithelium in Helicobacter pylori infection. Clin Exp Immunol 136:521–526
Akhtar M, Watson JL, Nazli A, McKay DM (2003) Bacterial DNA evokes epithelial IL-8 production by a MAPK-dependent, NF-kappaB-independent pathway. FASEB J 17:1319–1321
Sugita K, Kabashima K, Atarashi K, Shimauchi T, Kobayashi M, Tokura Y (2007) Innate immunity mediated by epidermal keratinocytes promotes acquired immunity involving Langerhans cells and T cells in the skin. Clin Exp Immunol 147:176–183
Tao K, Fujii M, Tsukumo S, Maekawa Y, Kishihara K, Kimoto Y et al (2007) Genetic variations of Toll-like receptor 9 predispose to systemic lupus erythematosus in Japanese population. Ann Rheum Dis 66:905–909
Lange NE, Zhou X, Lasky-Su J, Himes BE, Lazarus R, Soto-Quirós M et al (2011) Comprehensive genetic assessment of a functional TLR9 promoter polymorphism: no replicable association with asthma or asthma-related phenotypes. BMC Med Genet 12:26
Kikuchi K, Lian ZX, Kimura Y, Selmi C, Yang GX, Gordon SC et al (2005) Genetic polymorphisms of toll-like receptor 9 influence the immune response to CpG and contribute to hyper-IgM in primary biliary cirrhosis. J Autoimmun 24:347–352
Chen X, Wang S, Liu L, Chen Z, Qiang F, Kan Y et al. (2011) A genetic variant in the promoter region of toll-like receptor 9 and cervical cancer susceptibility. DNA Cell Biol. doi:10.1089/dna.2011.1427
Pandey S, Mittal B, Srivastava M, Singh S, Srivastava K, Lal P et al (2011) Evaluation of Toll-like receptors 3 (c.1377C/T) and 9 (G2848A) gene polymorphisms in cervical cancer susceptibility. Mol Biol Rep 38:4715–4721
Carpentier AF, Chen L, Maltonti F, Delattre JY (1999) Oligodeoxynucleotides containing CpG motifs can induce rejection of a neuroblastoma in mice. Cancer Res 59:5429–5432
Mansilla C, Berraondo P, Durantez M, Martínez M, Casares N, Arribillaga L et al. (2011) Eradication of large tumors expressing human papillomavirus E7 protein by therapeutic vaccination with E7 fused to the extra domain a from fibronectin. Int J Cancer. doi: 10.1002/ijc.26412
Hofmann MA, Kors C, Audring H, Walden P, Sterry W, Trefzer U (2008) Phase 1 evaluation of intralesionally injected TLR9-agonist PF-3512676 in patients with basal cell carcinoma or metastatic melanoma. J Immunother 31:520–527
Leonard JP, Link BK, Emmanouilides C, Gregory SA, Weisdorf D, Andrey J et al (2007) Phase I trial of toll-like receptor 9 agonist PF-3512676 with and following rituximab in patients with recurrent indolent and aggressive non Hodgkin’s lymphoma. Clin Cancer Res 13:6168–6174
Thompson JA, Kuzel T, Drucker BJ, Urba WJ, Bukowski RM (2009) Safety and efficacy of PF-3512676 for the treatment of stage IV renal cell carcinoma: an open-label, multicenter phase I/II study. Clin Genitourin Cancer 7:E58–E65
Doorbar J (2005) The papillomavirus life cycle. J Clin Virol 32:S7–S15
El-Omar EM, Ng MT, Hold GL (2008) Polymorphisms in Toll-like receptor genes and risk of cancer. Oncogene 27:244–252
Holla LI, Vokurka J, Hrdlickova B, Augustin P, Fassmann A (2010) Association of Toll-like receptor 9 haplotypes with chronic periodontitis in Czech population. J Clin Periodontol 37:152–159
Fuse K, Katakura K, Sakamoto N, Ohira H (2010) Toll-like receptor 9 gene mutations and polymorphisms in Japanese ulcerative colitis patients. World J Gastroenterol 16:5815–5821
Mollaki V, Georgiadis T, Tassidou A, Ioannou M, Daniil Z, Koutsokera A et al (2009) Polymorphisms and haplotypes in TLR9 and MYD88 are associated with the development of Hodgkin’s lymphoma: a candidate-gene association study. J Hum Genet 54:655–659
Xu CJ, Zhang WH, Pan HF, Li XP, Xu JH, Ye DQ (2009) Association study of a single nucleotide polymorphism in the exon 2 region of toll-like receptor 9 (TLR9) gene with susceptibility to systemic lupus erythematosus among Chinese. Mol Biol Rep 36:2245–2248
Novak N, Yu CF, Bussmann C, Maintz L, Peng WM, Hart J et al (2007) Putative association of a TLR9 promoter polymorphism with atopic eczema. Allergy 62:766–772
Velez DR, Wejse C, Stryjewski ME, Abbate E, Hulme WF, Myers JL et al (2010) Variants in toll-like receptors 2 and 9 influence susceptibility to pulmonary tuberculosis in Caucasians, African-Americans, and West Africans. Hum Genet 127:65–73
Ng MT, Van’t Hof R, Crockett JC, Hope ME, Berry S, Thomson J et al (2010) Increase in NF-kappaB binding affinity of the variant C allele of the toll-like receptor 9–1237T/C polymorphism is associated with Helicobacter pylori-induced gastric disease. Infect Immun 78:1345–1352
Etem EO, Elyas H, Ozgocmen S, Yıldırım A, Godekmerdan A (2011) The investigation of toll-like receptor 3, 9 and 10 gene polymorphisms in Turkish rheumatoid arthritis patients. Rheumatol Int 31:1369–1374
Bochud PY, Hersberger M, Taffé P, Bochud M, Stein CM, Rodrigues SD et al (2007) Polymorphisms in Toll-like receptor 9 influence the clinical course of HIV-1 infection. AIDS 21:441–446
Ashton KA, Proietto A, Otton G, Symonds I, McEvoy M, Attia J et al (2010) Toll-like receptor (TLR) and nucleosome-binding oligomerization domain (NOD) gene polymorphisms and endometrial cancer risk. BMC Cancer 10:382
Su SL, Yang HY, Lee CH, Huang GS, Salter DM, Lee HS (2012) The (-1486T/C) promoter polymorphism of the TLR-9 gene is associated with end-stage knee osteoarthritis in a Chinese population. J Orthop Res 30:9–14
Carvalho A, Cunha C, Almeida AJ, Osório NS, Saraiva M, Teixeira-Coelho M et al. (2011) The rs5743836 polymorphism in TLR9 confers a population-based increased risk of non-Hodgkin lymphoma. Genes Immun. doi:10.1038/gene.2011.59
Acknowledgments
Supported by Grant No 502-01-01124182-07474, Poznań University of Medical Sciences. The technical assistance of Ms. Monika Świerczyńska is gratefully acknowledged.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Roszak, A., Lianeri, M., Sowińska, A. et al. Involvement of toll-like receptor 9 polymorphism in cervical cancer development. Mol Biol Rep 39, 8425–8430 (2012). https://doi.org/10.1007/s11033-012-1695-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-012-1695-8