Abstract
Aims
Growth reductions and yield losses from drought could be mitigated by developing rice genotypes with more efficient root systems. We examined spatiotemporal responses to drought in order to determine whether roots developing in upper vs. deeper soil layers respond differently to drought stress.
Methods
Root anatomical and architectural phenotypes of two rice genotypes, Azucena (drought tolerant) and IR64 (drought susceptible), were measured weekly in well-watered and vegetative-stage drought stress treatments in solid medium with stratified moisture availability. Basal and apical segments were collected from older, deeper nodal roots and apical segments from younger, shallow roots for assessment of anatomy and lateral rooting phenotypes. The relationship between root anatomy and root respiration rates was tested in solution culture and solid medium.
Results
Compared to IR64, Azucena had deeper root systems and larger diameter roots in both treatments but reduced its living tissue area in response to drought, while IR64 roots exhibited less plasticity in root diameter. Root respiration rates were positively correlated with root diameter and living tissue area, providing evidence that root anatomy affects the metabolic cost of tissues. In response to drought, Azucena showed reduced theoretical axial hydraulic conductance in shallow roots and at the base of deep roots but slightly greater conductance at the tip of deep roots, while IR64 displayed low plasticity in metaxylem phenotypes.
Conclusion
We propose that the plasticity of root phenotypes in Azucena contributes to its drought tolerance by reducing the metabolic cost of soil exploration and improving the efficiency of water transport.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rice (Oryza sativa) is the 2nd greatest cereal crop in global production (FAO 2018) and is the largest food source for populations in developing economies (McLean et al. 2013). Rice is extremely susceptible to yield loss from abiotic stresses such as drought and nutrient deficiency. Global rice production is dominated by irrigated and rainfed lowland paddy systems, which require greater water consumption than other cereals (McLean et al. 2013). Rice suffers greater yield loss than other cereals at similar levels of water stress (Daryanto et al. 2017). Rising global temperatures are expected to exacerbate drought-related yield losses in the future (Lobell and Gourdji 2012). Root phenotypes that improve water acquisition under drought can sustainably increase rice production and promote food security for low-input farmers and their communities.
Root phenotypes are key components of tolerance to edaphic stresses, including drought. Since growth and maintenance of living root tissue utilizes internal carbon and nutrients, efficient soil exploration requires optimized partitioning of these resources within the root system (Lynch 2013). The metabolic cost of root systems varies with root number and length and anatomical phenotypes, such as root diameter, the extent of aerenchyma formation, and the size of the cortex, stele, and vascular tissue. Phenes that reduce root system cost, including fewer nodal roots, reduced living tissue area via smaller root diameter, greater aerenchyma formation or root cortical senescence, greater cortical cell size, and reduced cortical cell file number, can promote greater root elongation, deeper soil exploration, and greater resource capture (Lynch 2013, 2019). Crops with anatomical traits that reduce metabolic cost per unit root length perform better under drought and low nutrient stress, as demonstrated in maize (Chimungu et al. 2014a, b; Saengwilai et al. 2014; Lynch 2015; Castañeda et al. 2018) and common bean (Strock et al. 2018, 2019; Strock and Lynch 2020). Though the benefit was not directly attributed to metabolic cost savings, rice lines that developed greater aerenchyma produced more lateral roots, greater biomass and yield under drought (Niones et al. 2012, 2013). The aforementioned root phenotypes have been shown to vary significantly among diverse rice genotypes (Terashima et al. 1987; Kondo et al. 2000; Uga et al. 2008, 2009; Vejchasarn et al. 2016), which suggests that root respiration requirements can also vary within rice. Although studies have shown a clear relationship between root anatomical phenotypes and root respiration rates in maize (Saengwilai et al. 2014; Castañeda et al. 2018; Strock et al. 2018), barley (Schneider et al. 2017b), and common bean (Phaseolus vulgaris) (Strock et al. 2018), this relationship has not been demonstrated in rice, which has much greater extent of aerenchyma formation than other crops.
A second factor influencing the efficiency of soil resource foraging is the placement of roots in soil domains where growth-limiting resources are most abundant. Rice genotypes that can take up water in deep soil are often more vigorous under drought (Gowda et al. 2011, 2012; Uga et al. 2013; Arai-Sanoh et al. 2014), since moisture is often still available in deep soil (Wasson et al. 2012; Lynch 2013). Deep rooting could be achieved by steeper root growth angle, fewer nodal roots with smaller diameter, fewer longer lateral roots, or lower metabolic cost per unit length of root tissue leading to deeper rooting (Lynch 2013). In maize and common bean, these ideotypes have been demonstrated as beneficial under nutrient and drought stress (Lynch 2019). In rice, deeper rooting via steeper root growth angle improves drought tolerance, and a gene, DRO1, which influences root growth angle, has been cloned and utilized in breeding programs (Uga et al. 2013; Kitomi et al. 2015). Greater lateral root proliferation from deep nodal roots may aid in water uptake from deep soils as well, and the benefits of greater lateral root length and density at depth have been documented in previous drought studies in rice (Kano et al. 2011; Gowda et al. 2012; Suralta et al. 2015; Hazman and Brown 2018).
Genotypes that not only access more water but use water more efficiently may have greater growth under drought stress. Variation in metaxylem size and number may affect water use efficiency and drought tolerance. Roots with smaller metaxylem vessels have less axial hydraulic conductance (Tyree and Ewers 1991), thereby allowing the plant to conserve soil water resources and maintain hydration of the root tip and surrounding rhizosphere (Lynch et al. 2014). Smaller metaxylem vessels are also more resistant to cavitation and collapse (Sperry and Saliendra 1994; Guet et al. 2015). Studies in wheat showed that smaller metaxylem vessels in seedling seminal roots improved yield under drought (Richards and Passioura 1989). Maize genotypes that reduced nodal root metaxylem area and axial hydraulic conductance had better performance under drought than genotypes that did not display this type of plasticity (Klein et al. 2020). In a greenhouse study, drought-tolerant rice cultivars had greater numbers of xylem vessels, and a greater increase in xylem vessel number with drought stress, than the drought susceptible cultivar (Abd Allah et al. 2010). In another pot study, an increase in xylem vessel number along with increased xylem vessel diameter was observed in response to drought in the Indica variety ‘Mudgo’, while NERICA4 (derived from O. sativa L. X O. glaberrima Steud.) reduced xylem vessel number but increased xylem vessel diameter with drought (de Bauw et al. 2019). In a diversity panel of Indica rice genotypes, there was genetic variation for several anatomical traits including late metaxylem vessel number and diameter in both control and water-deficit conditions, though the mean difference between water-deficit and control treatments was only -7% for late metaxylem vessel diameter and -2% for late metaxylem vessel number (Kadam et al. 2017).
Phenotypic variation along the root axis may also be important. For example, larger, fewer metaxylem vessels near the root tip and more, smaller metaxylem vessels near the root base occurs in some drought tolerant wheat varieties (Wasson et al. 2012; Kadam et al. 2015). Other studies have observed metaxylem constriction at the base of nodal and lateral roots in rice (Vejchasarn et al. 2016; Hazman and Brown 2018). Less axial hydraulic conductance in basal regions of roots may be a beneficial strategy in rice to moderate water movement to shoot to ensure that root tissues, particularly the growing tips and surrounding rhizosphere, maintain sufficient water for growth.
Cultivars in Indica and Japonica, the two main varietal groups within rice, have distinctly different root morphologies. Indica types tend to have small diameter, highly branched roots and smaller metaxylem vessels, while Japonica types tend to have larger diameter roots with less branching and larger metaxylem vessels (Yoshida and Hasegawa 1982; Gowda et al. 2011). Many high-yielding lowland varieties, such as IR64 (Indica type), perform best in high-input environments where water and nutrients are available in shallow soils. Upland varieties, such as Azucena (Japonica type), are often more resistant to abiotic stresses, though their yield potential is typically less. The relative drought tolerance of Azucena has often been attributed to its large diameter, deep roots, which improve penetration of hard soils and permit access to deep soil moisture (Gowda et al. 2011). It has been noted that Azucena responds to drought with increased root length density at depth (Henry et al. 2011), which likely results from both longer roots and more deep branching. Some studies in rice support the idea that root plasticity is beneficial for drought stress tolerance (Kano et al. 2011; Henry et al. 2012; Kadam et al. 2015; Suralta et al. 2016). However, the benefits of root plasticity may be limited to low-input agricultural systems (Schneider and Lynch 2020) and must be evaluated in managed, higher-input systems.
One difficulty with identifying adaptive phenotypes for use in crop breeding is that both drought and root development are dynamic processes. In a typical terminal drought environment, moisture becomes increasingly stratified as upper soil layers dry while deeper layers may retain considerable moisture. Roots should be able to acquire water and nutrients during drought if they continue to explore deep soil. Although terminal drought is just one of a large number of potential drought scenarios, it is common enough that researchers and plant breeders have targeted deeper rooting as an important trait for drought tolerance in rice and other crops (Gowda et al. 2012; Lynch 2013; Arai-Sanoh et al. 2014). However, a deep rooting phenotype does not always ensure drought tolerance in rice, as exemplified by some varieties of upland rice that are identified as “deep rooting” that are also quite stress prone e.g. Moroberekan (Henry 2013). Careful characterization of root system phenes and spatiotemporal dynamics above and below ground will more accurately explain and predict drought tolerance in crop plants (Vadez 2014).
In this study, we explored root phenotypic responses to progressive drought in deep and shallow roots of two genotypes with contrasting drought tolerance. We compared responses of older, deeper roots with those of younger shallower roots, with weekly sampling to capture the dynamics of these responses. We hypothesized that apical segments of deep nodal roots would have greater axial hydraulic conductivity and greater lateral root branching density than shallow roots or basal segments of deep roots that developed earlier in the drought. We expected that these responses would accelerate over time as the upper soil strata became increasingly dry. We also tested the impact of root anatomy on root respiration rates in solid growth media and solution culture. We expected root diameter to decrease and aerenchyma area to increase during drought and that the respiration rate per unit root length would be positively associated with root diameter and negatively associated with aerenchyma area.
Materials and methods
Time course of drought responses
Two genotypes, Azucena (tropical japonica, upland, drought tolerant) and IR64 (indica, lowland, drought susceptible) (Supplementary Table 1) were grown in three biological replicates for each of five harvests of plants from drought and well-watered treatments. Plants were arranged in a randomized complete block design in the greenhouse.
Plastic mesocosms (15 cm diameter × 1.2 m tall) in greenhouses at Penn State University (University Park, PA, USA, 40° 48 7.4 N, 77° 51 46.5 W) were lined with a polyethylene liner and filled with a growth medium of 40% (by volume) medium-grade silica sand (0.3–0.5 mm, U.S. Silica, Berkeley Springs, WV, USA), 40% vermiculite (Griffin Greenhouse Supplies, Morgantown, PA, USA), 5% perlite (Griffin Greenhouse Supplies, Morgantown, PA, USA), 15% sifted field soil (Hagerstown silt loam; 64% silt, 21% clay, 15% sand; fine, mixed, semi-active, medic Typic Hapludult), and slow-release fertilizer pellets at 3.2 g/L (Osmocote Plus, 15% N, 9% P, 12% K, 1% Mg, 6% S, 0.02% B, 0.05% Cu, 0.4% Fe, 0.06% Mn, 0.02% Mo, 0.05% Zn, 3–4 month release). Seeds were manually de-hulled, sterilized with 10% (v/v) NaOCl in water, and rinsed with deionized water. Wire single-mesh baskets (14.3 cm diameter, 13 cm tall, Winco, NJ, USA) were placed into the top 10 cm of the mesocosm and filled with the same growth medium for measurement of nodal root angle. Seeds were planted directly into mesocosms inside the baskets. Temperature was maintained at 25–28 °C, and supplemental LED lights (200 μmol m−2 s−1 PAR) provided a 14-h day length when ambient light was less than 600 μmol m−2 s−1 PAR. Plants were watered daily with ~ 300 mL deionized water from the top of the mesocosm using a drip ring irrigation system and weekly with 100 mL 1 mM CaCl2. Mesocosms could drain through mesh at the bottom, providing aerobic conditions in both treatments. Two weeks after germination, drought treatment was imposed by withholding water except for weekly CaCl2 treatment to the top of the pot of both treatments. Volumetric water content was measured biweekly using time-domain reflectometry (TDR100, Campbell Scientific, Logan, UT, USA) with probes inserted 30.5 cm from the top and bottom of the mesocosm to measure surface soil drying as well as moisture content of deep wetter soil. Gravimetric moisture content was measured in 50 mL of medium collected at 30 cm from the top and bottom of mesocosms during each harvest using fresh and dry weight of the medium.
Three plant replicates were harvested from each genotype and treatment weekly from two to six weeks after germination. Stomatal conductance was measured on abaxial leaf tissue at 12 pm using a leaf porometer (SC-1, Meter Group Inc, Pullman, WA, USA) one day before harvest. Tiller number was counted manually. To measure sap exudation rate, shoots were cut 3 cm above the soil surface at 9 am and collected to be dried in a drying oven. The 3 cm stem segments were all immediately covered with a 50 mL pre-weighed polypropylene centrifuge tube filled with cotton. At 1 pm, tubes were removed, capped, and weighed. Sap exudation rate was calculated as sap weight exuded per hour (g/h). The root systems and media mix contained within the plastic liner were laid horizontal and pulled from the pots, and the liner was cut open to expose the roots and media. Media samples were taken at this time for gravimetric moisture content. The maximum depth of the root system was recorded, and the remaining media was gently washed from the root system. Clean root systems were preserved in bags with 70% (v/v) ethanol in water for later analysis.
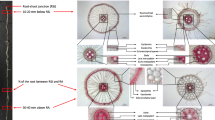
Nodal roots were classified into thick and thin classes based on root diameters, but only thick nodal root data is presented here. Mean cross-sectional areas of thick and thin nodal roots, respectively, were 0.423–0.565 mm2 and 0.154–0.181 mm2 in Azucena and 0.247–0.288 mm2 and 0.118–0.134 mm2 in IR64. Deep roots were those emerging between 50–90° from horizontal in the basket, while shallow roots emerged above 50° and had a length less than 50% of the maximum length of the root system.
From each plant, 2 cm segments of subapical root tissue were harvested for anatomy measurement at 10 and 20 cm from the root tip in three thick (Azucena mean: 0.827 mm2, IR64 mean: 0.455 mm2) shallow roots (Supplementary Fig. 1). In three thick deep nodal roots (length greater that 50% of the maximum root length of the root system), 2 cm samples were taken at 10 and 20 cm from the tip (subapical tissue) and 10 and 20 cm from the root base (basal tissue). Total nodal root number was counted, and the number of deep roots was measured by counting the number of roots emerging between 50–90° from horizontal in the basket. For lateral root measurements, one shallow and one deep (divided in half into basal and apical segments) intact, representative nodal root per plant were scanned on a flatbed scanner (Epson V700 Photo) and analyzed for S-type and L-type lateral root length (SLRL, LLRL) and density (SLRD, LLRD) using WinRHIZO Pro 2019 (Regent Instruments Inc, Quebec, Canada). Small lateral roots were classified as 0.10–0.25 mm diameter, and large lateral roots as roots 0.25–1.1 mm diameter for both genotypes as measured in WinRhizo.
Anatomy samples were placed in plastic histocaps and dried using a critical point drier (Leica EM CPD 300, Leica Microsystems Inc, Buffalo Grove, IL, USA) to maintain root structure. Root cross-sectional images were taken using laser ablation tomography (Hall et al. 2019). For laser ablation, samples were moved into a 355-nm Avia 7000 pulsed laser beam that vaporizes the samples in the plane of focus of a Canon T3i camera with a 53 micro lens (MP-E65 mm). Cross-sectional images of roots were measured with Mipar® software to quantify total root cross-sectional area (RXSA), stele area (TSA), median late metaxylem vessel area (MXA), number of late metaxylem vessels (MXV), and aerenchyma area (AA). Cortical area (TCA) was calculated as: RXSA – TSA. Percent aerenchyma area (percAA) was calculated as a proportion of cortical area: AA/(TCA)*100. Living tissue area (LTA) was calculated as: RXSA – AA – MXA. Theoretical root axial hydraulic conductance (hydraulic conductance, kg m MPa−1 s−1) was calculated using the modified Hagen-Poiseuille Law:
where d is the mean diameter of each metaxylem vessel (m), ρ is the xylem sap fluid density assumed to be water (1000 kg m−3), and η is the viscosity of the xylem sap assumed to be water (1 × 10–9 MPa s) (Tyree and Ewers 1991). Shallow, subapical, and basal anatomy measurements represented the mean value from samples at 10 and 20 cm from either the root tip or base. All data preparation and statistics including ANOVA and pair-wise t-tests were performed in R v 3.5.3 (R Core Team 2016). The ANOVA was carried out on a linear model (Treatment*Genotype) to see whether the terms of the model were significant using the anova() and lm() functions from the base R package. For correlations, a linear regression model was fit to the data, the r (and R2) statistics calculated, and then this correlation was tested for significance using both r and n, all with the ggpmisc R package (Aphalo 2021). The corrplot package (Wei and Simko 2017) was used to generate correlation matrix plots, and the ggplot2 package (Wickham 2016) was used to generate all other plots.
Measurement of root respiration in solution culture
Genotypes Azucena and IR64 were grown in 4 biological replicates for this solution culture experiment. Seeds were dehulled, sterilized in 10% (v/v) NaOCl in water, rinsed with deionized water, and soaked in deionized water for 2–4 h. Seeds were germinated on rolled germination paper (10 seeds per roll, 40.6 X 20.3 cm, Anchor Paper Company, St. Paul, MN, USA) that was soaked with 0.5 M CaSO4 at 28 °C for 3 days in the dark, and seedlings were then moved to a light chamber for 4 days. Plants were placed in a hydroponic tank with 100L of half-strength Yoshida nutrient solution (Yoshida et al. 1976), changed every three days and pH adjusted daily, in a greenhouse maintained at 20–23 °C air temperature and 22–26° solution temperature with supplemental LED light (200 μmol m−2 s−1 PAR) for a 14-h day. Plants were arranged by genotype in two blocks within one tub.
At 4 weeks after transplanting, shoots were collected and dried, and 2 cm root samples were taken from each nodal root on the plant (ranging from 4–9 roots) at 5 cm from the root tip (subapical tissue) and 10 cm from the root base (basal tissue) in thick and thin nodal roots separately. Thick and thin nodal root classifications were determined by visually comparing root diameters within each root system (means at 5 cm from tip and 10 cm from base: Azucena 0.423–0.565 mm2; IR64 0.247–0.288 mm2) and three thin (Azucena 0.154–0.181 mm2, IR64 0.118–0.134 mm2). Any lateral roots were excised from the nodal root samples, which were kept hydrated in a damp paper towel before measurement. Root samples were placed in a root respiration chamber attached to a Licor 6400XT (Licor Inc., Lincoln, NE, USA) closed system in a 27 °C water bath, and the rate of CO2 evolution was recorded for 5 min. Data were later scaled by total length of root tissue in the chamber (µmol CO2 cm−1 s−1). Root samples were then transferred to histocaps in 70% (v/v) ethanol in water, dried in a critical point drier (Leica EM CPD 300, Leica Microsystems Inc, Buffalo Grove, IL, USA), and ablated on the laser ablation system to obtain root cross-section images. Root images were analyzed with Mipar® software to quantify RXSA, TSA, TCA, MXA, MXV, AA, percAA, LTA, and axial hydraulic conductance. All data preparation and statistics were performed in R v 3.5.3 as described above.
Root respiration of roots grown in solid medium
Based on previous phenotyping of the Rice Diversity Panel (RDP1) (Vejchasarn 2014), twelve genotypes were chosen for variation in root cross-sectional area within each of the Indica and Japonica varietal groups, for a total of 24 genotypes (Supplementary Table 1).
Plastic pots (42 height × 20 diameter) were filled with a 40% vermiculite (Griffin Greenhouse Supplies, Morgantown, PA, USA) and 60% silica sand (0.3–0.5 mm, U.S. Silica, Berkeley Springs, WV, USA). Phosphorus treatment was applied using alumina-buffered P buffering at 1 µM for low P and 320 µM for high P (Lynch et al. 1990) mixed directly into the media. Fertilizer was applied twice daily using 500 mL per application of modified Yoshida nutrient solution containing 1 μM P at pH 5.5 (Yoshida et al. 1976) and once per week substituting with plain water. Genotypes and treatments were arranged in a randomized complete block design in the greenhouse with four biological replicates. Two seeds were planted directly into pots and thinned to one plant per pot one week after germination, on April 3, 2012 and grown under natural light in a greenhouse at 25–28 °C for 11 weeks.
At 11 weeks after germination, root systems were extracted and washed over wire mesh and root samples were taken for root respiration rates. Four nodal roots that reached at least 2/3 the depth of the pot were excised, and lateral roots were cut off. Roots were then cut into three segments: 0–5 cm, 5–12 cm, and 12–20 cm from the root tip. Root segments were then placed into a root respiration chamber attached to a Licor 6400XT (Licor Inc., Lincoln, NE, USA) in the closed configuration, and respiration rates were measured as described above. Root segments were placed back into 70% (v/v) ethanol in water and stored for anatomy measurement. Laser ablation tomography (Hall et al. 2019) was used to take high-resolution transverse cross-sectional images of each root sample. Root anatomical measurements including RXSA, TSA, TCA, MXA, MXV, AA, percAA, and LTA were quantified from images using Mipar® image analysis software. Data preparation and statistics were performed in R v 3.5.3 as described above. Raw data, including shoot and root traits by genotype and treatment, are shown in Supplementary Table 1.
Samples of shoot tissue were dried, ground, and ashed at 495 °C for 12 h. Ashed tissue was dissolved in 100 mL HCl and analyzed spectrophotometrically for P concentration using the Murphy-Riley method (Murphy and Riley 1962). Total P content per plant shoot was calculated as P concentration in tissue multiplied by shoot dry biomass.
Results
Effects of water stress on shoot growth and root architectural phenotypes in Azucena and IR64
In mesocosms in the drought treatment, soil moisture decreased in shallow soil in both genotypes but was greater in deep soil at the bottom of the mesocosms (Supplementary Fig. 2). There were no differences between genotypes in volumetric or gravimetric water content at the top or bottom of the mesocosms.
The drought tolerant genotype Azucena maintained 55% greater shoot dry biomass and 54% fewer tillers than IR64 under well-watered conditions at week 6 (Fig. 1, Table 1, Supplementary Table 2). Differences in shoot dry biomass between drought and well-watered treatments did not emerge until week 5, when shoot dry biomass of Azucena was reduced by 67% with drought, though it was not significantly different in IR64 (Supplementary Table 2). By week 6, drought stress had reduced shoot dry biomass by 61% in Azucena and 39% in IR64, but nevertheless, Azucena still maintained 33% greater shoot biomass and 33–354% fewer tillers than IR64.
Measurements of sap exudation rates showed significant effects of drought by week 3, though genotypic differences were not detected under drought at any time (Supplementary Table 2). Stomatal conductance fell in both genotypes over the course of the drought, with significant treatment effects detectable by 4 weeks (Supplementary Table 2).
Nodal root number and deep root number (roots emerging at angles greater than 50° from horizontal) were both reduced due to drought treatment, but deep root number was more strongly affected (Table 1). At early stages of growth under well-watered conditions, Azucena had 43–45% fewer nodal roots than IR64, but nodal root numbers were comparable between genotypes by 5 weeks after emergence (Fig. 1, Supplementary Table 2). Under drought, Azucena and IR64 had similar numbers of nodal roots at most sampling times, except at week 5 when IR64 increased nodal root number coincidently with an increase in tiller number (Fig. 1, Supplementary Table 2).
Azucena generally exhibited deeper rooting than IR64. Azucena developed deep roots earlier, and by six weeks had 21-fold more deep roots than IR64 under well-watered conditions and threefold more deep roots with drought (Fig. 1, Supplementary Table 2). Azucena increased the number of deep roots by ~ ninefold in well-watered and by ~ 3.75-fold under drought from week 2 to 6, but IR64 maintained a small number of deep roots throughout the experiment with no treatment effect. Azucena maintained a greater proportion of deep nodal roots (45–63%) compared to IR64 (6–22%) under both treatments. IR64 increased its proportion of deep nodal roots in drought-treated plants compared to well-watered plants, but not significantly until week 6, while Azucena had a slightly reduced proportion of deep roots with drought (15% less at week 6) (Supplementary Table 2).
Lateral root growth was greater near deep root apices than in shallow roots
L-type lateral roots continued to grow during drought on the apical segments of deep (older) nodal roots (L-type lateral root length, Fig. 2, Supplementary Tables 3 and 4). Azucena had greater L-type lateral root length than IR64 at 5 weeks in well-watered conditions, but there were no significant genotypic differences under drought at any time. L-type lateral root density of deep apical segments increased in response to drought at week 5 in Azucena and at week 6 in IR64 (Supplementary Table 3). Apical segments of deep roots bore 88–95% greater L-type lateral root length than those of shallow roots at all sampling times and over both treatments and genotypes, despite few differences in L-type lateral root branching density (Fig. 2, Supplementary Table 4), indicating that shallow roots had very short lateral roots.
L-type lateral root length (a, b) and branching density (c, d) of thick nodal roots of Azucena (dashed line) and IR64 (solid line) under well-watered or drought stress treatments in greenhouse mesocosms. In plots a and c, apical samples were collected 10–20 cm from the tip of deep nodal roots (triangle) and shallow nodal roots (circle). In plots b and d, apical segments collected 10–20 cm from the tip (triangle) are compared with basal segments collected 10–20 cm from the base (square) of deep nodal roots. Branching density is expressed as number of L-type lateral roots per cm nodal root length. Error bars represent standard error
When apical versus basal segments of deep roots were compared, there were no significant differences in L-type lateral root length in either genotype or treatment (Fig. 2, Supplementary Table 4). Genotype and treatment had little effect on L-type lateral root length of basal segments, with only a few significant effects at p-value < 0.1 (Supplementary Table 4). L-type lateral root density was not significantly different between apical and basal segments in the well-watered treatment, but in IR64, L-type lateral root density rose more quickly in apical segments than in basal segments over the course of the drought, so that at week 5 IR64 had greater L-type lateral root density near the apex. A similar pattern was suggested in Azucena but variability was too great to detect significant differences.
S-type lateral root length increased over time in apical segments of deep roots in the well-watered treatment, but in the drought treatment this increase was attenuated in Azucena and eliminated in IR64 (Fig. 3, Supplementary Table 3). S-type lateral root length was usually less in apical segments of shallow roots than in apical segments of deep roots in both treatments. At week 6, S-type lateral root length was 72–75% greater in apical segments of deep roots compared to those of shallow roots in both treatments for Azucena but greater only in the well-watered treatment in IR64. In contrast, S-type lateral root density was consistently greater in apical segments of shallow roots than deep roots. Basal segments of deep roots increased S-type lateral root length over time in both treatments to a greater extent than apical segments of deep roots, but there were no differences between basal and apical segments in S-type lateral root density (Fig. 3, Supplementary Table 4). Drought reduced S-type lateral root density on basal segments of IR64 but not Azucena (Fig. 3, Supplementary Table 3).
S-type lateral root length (a, b) and branching density (c, d) of thick nodal roots of Azucena (dashed line) and IR64 (solid line) under well-watered or drought stress treatments in greenhouse mesocosms. In plots a and c, apical samples were collected 10–20 cm from the tip of deep nodal roots (triangle) and shallow nodal roots (circle). In plots b and d, apical segments collected 10–20 cm from the tip (triangle) are compared with basal segments collected 10–20 cm from the base (square) of thick deep nodal roots. Branching density is expressed as number of S-type lateral roots per cm nodal root length. Error bars represent standard error
Root anatomical phenotypes depend on root position and age
Root anatomical phenotypes were measured in thick nodal roots in shallow and deep parts of the root system over time in well-watered and drought conditions. Similar data was collected for thin nodal roots with nearly identical results (not shown). Here, we compare 1) subapical anatomy (10 and 20 cm from the root tip) of shallow vs. deep roots (Fig. 4), where shallow roots were defined as emerging above 50° from the basket and having less than half the length of the entire root system, while deep roots were longer and emerged below 50°; and 2) subapical (10 and 20 cm from the root tip) vs. basal (10 and 20 cm from the root base) anatomy of deep roots (Supplementary Fig. 3). In the well-watered treatment, each of these segments developed in similarly moist medium and variation was likely due to earlier (basal segments) vs. later (apical segments) development. However, in the drought treatment, basal segments developed during early stages of drought, while apical segments of deep roots developed in moist deep soil later in drought, and apical segments of shallow roots developed in dry shallow soil later in drought (Supplementary Fig. 2).
Root anatomical phenotypes of Azucena (dashed line) and IR64 (solid line) at 10–20 cm from the root tip of deep (triangle) and shallow (circle) thick nodal roots under drought stress in greenhouse mesocosms. a root cross-sectional area, b living tissue area, c stele area, d cortical area, e aerenchyma area, f late metaxylem vessel number, g median late metaxylem vessel area, and h theoretical axial hydraulic conductance. Error bars represent standard error
Anatomical phenes were significantly affected by treatment, cultivar, harvest date, and sampling position, with the exception of percent aerenchyma, which was only affected by sampling position (Table 1, Fig. 5). Under both well-watered and drought conditions, most anatomical phenotypes were positively correlated (Supplementary Fig. 3). Percent aerenchyma was unrelated to area phenotypes in deep roots and was negatively correlated in shallow roots. Correlations were similar in well-watered plants, with the exception that there were fewer and weaker significant correlations of percent aerenchyma with other anatomical phenotypes (Supplementary Fig. 3).
Subapical segments of shallow roots usually had greater diameter than those of deep roots, a pattern reflected in all the area phenotypes (Fig. 4). This difference was attenuated in roots from drought treated plants. In deep roots, root cross-sectional area was less in drought-treated than in well-watered plants, but subapical segments were less affected than basal segments in Azucena (Fig. 6, Supplementary Table 4). In IR64, cross-sectional area was similar between subapical and basal segments, but living tissue area was less in basal segments in both treatments (Fig. 6). Percent aerenchyma was greater in basal compared with subapical segments in both genotypes and treatments (Fig. 6).
Root anatomy of subapical versus basal segments of deep nodal roots. Root anatomical phenotypes of Azucena (dashed line) and IR64 (solid line) in basal (square) and subapical (triangle) deep thick nodal root tissue under drought stress in greenhouse mesocosms. a root cross-sectional area, b living tissue area, c stele area, d cortical area, e aerenchyma area, f late metaxylem vessel number, g median late metaxylem vessel area, and h theoretical axial hydraulic conductance. Error bars represent standard error. Apical tissue was measured at 10 and 20 cm from the tip, so if roots were < 20 cm, basal tissue anatomy was not measured
Xylem phenotypes were affected by sampling position in both genotypes. Late metaxylem vessel number was generally greater in shallow than in deep roots, while median metaxylem area was similar, especially with drought (Fig. 4). Median metaxylem area in shallow roots was less drought-treated than in well-watered plants by 5 weeks in both genotypes (Supplementary Table 3). Metaxylem vessel number was greater in basal segments than in subapical segments of IR64, and median metaxylem vessel area increased during the drought in Azucena in basal segments but remained mostly consistent in subapical segments (Fig. 6).
Root anatomical phenotypes exhibited greater plasticity in response to drought treatment in Azucena than in IR64
Azucena consistently had larger diameter roots and more, larger metaxylem vessels than IR64. Azucena had 30–55% greater root cross-sectional area, 29–54% greater living tissue area, 33–58% greater metaxylem vessel area, 22–49% greater ratio of total metaxylem vessel area to root cross-sectional area, 14–43% greater metaxylem vessel number, and 71–86% greater axial hydraulic conductance than IR64 under both treatments, while there was no difference in percent aerenchyma between genotypes (Figs. 4–6, Supplementary Table 3). All root anatomical phenotypes were significantly affected by drought treatment except percent aerenchyma (Table 1). Azucena maintained greater trait values than IR64 in most cases, even considering the greater reductions with drought.
Compared with IR64, Azucena exhibited greater reductions in root cross-sectional area and living tissue area in response to drought in subapical segments of deep roots (30–42% reduction in Azucena at week 6, 0–25% reduction in IR64 at week 6), while shallow roots of both genotypes had 34–44% reduction with drought at week 6 (Fig. 4, Supplementary Table 3). In shallow roots, IR64 had 30% greater percent aerenchyma area with drought treatment, whereas Azucena showed no differences between drought and well-watered plants. In contrast, deep roots had 18–25% less percent aerenchyma with drought at week 6 in both genotypes.
Azucena showed greater plasticity of metaxylem vessel number and axial hydraulic conductance than IR64 in both shallow and deep roots (Figs. 4, 6, Supplementary Table 3). Although metaxylem vessel number is generally correlated with area phenotypes (Supplementary Fig. 3), at week 6 it was 9% greater in shallow roots and 22% less in deep roots in drought compared with the well-watered treatment in Azucena. Root axial hydraulic conductance was 49% less in shallow and 44% less in deep roots in response to drought in Azucena but not different between treatments in IR64 (Fig. 4). Median metaxylem vessel area of shallow roots was 25–29% less in both genotypes in response to drought, counteracting the greater vessel number in Azucena. Median metaxylem vessel area was 21% less with drought in deep roots of IR64.
In the well-watered treatment, metaxylem area and axial hydraulic conductance were greater in shallow roots than in deep roots in Azucena but not IR64 (Fig. 4, Supplementary Table 4). At week 6, Azucena shallow roots had 11% greater median metaxylem vessel area and 31% greater axial hydraulic conductance than deep roots.
Root anatomical phenotypes affect root respiration rates
In order to assess the impact of variation in root anatomical phenotypes on root metabolic cost, we measured root respiration rates and root anatomical phenotypes in rice roots grown in solid growth media under high and low P treatment and in solution culture, since insufficient P was previously shown to result in thinner nodal roots (Vejchasarn et al. 2016). Plants were grown with moderate phosphorus stress, with 38% less biomass and 38% fewer tillers than plants grown in high phosphorus (Table 2). Genotypic variation and phosphorus treatment effects resulted in variation in root cross-sectional area and living tissue area (Table 2, Supplementary Table 1), which were expected to affect respiration rates. Respiration rates were positively related to root cross-sectional area and its components, cortical and stele areas, and living tissue area, but negatively associated with percent aerenchyma (Table 3). Across all genotypes, root respiration rates were 17% greater in apical segments of roots grown in low P compared with those grown in high P (Table 2, Supplementary Fig. 4).
In solution culture, respiration rates per unit length of root tissue had even stronger positive correlations with root anatomical phenotypes than in solid medium, with strong correlations in subapical segments of both thick and thin nodal roots (Table 3). In Azucena, thick nodal roots consistently had greater root respiration per length compared to IR64 due to its greater root cross-sectional, living tissue, cortical, and stele area (Supplementary Fig. 5). Root cross-sectional area, stele area, cortical area, and living tissue area correlated positively with respiration rates per unit length in apical segments, while there were no relationships between anatomical phenotypes and respiration rates calculated per gram of tissue (p-values > 0.05, data not shown). Percent aerenchyma area was negatively associated with respiration rate only when all segments were combined (Table 3).
Discussion
In this study, we investigated the dynamic responses of root architecture and anatomy to drought in two genotypes representing the Japonica and Indica varietal groups of rice. IR64 is a high-yielding Indica cultivar with minimal drought tolerance, while Azucena is a lower yielding but moderately drought-tolerant (Guiderdoni et al. 2006; Venuprasad et al. 2007) deep-rooting Japonica cultivar (Gowda et al. 2012). Azucena is adapted to upland conditions (Guiderdoni et al. 2006) and benefits from the ability of its thick nodal roots to penetrate dry soils and reach deep water in a field setting. We confirmed in these experiments that Azucena has a greater number of deep nodal roots of larger diameter than IR64 even under drought (Figs. 1, 4). Azucena and IR64 had similar lateral root branching patterns (Figs. 2, 3), but the greater length and depth of nodal roots of Azucena would place more lateral roots in deeper soil, where more moisture is available.
If the “steep, cheap, and deep” ideotype proposed for maize (Lynch 2013) applies to rice, then fewer nodal roots should contribute to performance under drought by conserving resources for elongation rather than proliferation of nodal roots, resulting in greater root depth (Gao and Lynch 2016). Both cultivars significantly reduced nodal root number with drought (Fig. 1), which may redirect resources to root elongation and deep root proliferation for both cultivars, in addition to reflecting reduced overall growth. The greater depth of nodal roots in Azucena would increase the efficiency of the remaining nodal roots for water and nutrient uptake as drought progresses.
Lateral root plasticity
We expected lateral root length and density to be greater in deep roots in response to drought, as has been observed in other studies with rice, including in the field (Kano et al. 2011; Gowda et al. 2012; Suralta et al. 2015). Increased development of L-type lateral roots at depth is beneficial in most drought scenarios, and QTLs and alleles have been identified for lateral root phenotypes in rice (Lyu et al. 2013; Niones et al. 2015; Suralta et al. 2015). Interestingly, both cultivars in this experiment continued to elongate L-type lateral roots over time in both basal and apical segments of deep nodal roots, even under drought. L-type lateral roots can become quite long and could contribute substantially to deeper soil exploration even if they arise from the more basal segments of the nodal root. In previous research using the same growth system, four rice cultivars grown with drought stress had much greater lateral root length and branching density on apical segments of nodal roots compared to well-watered plants (Hazman and Brown 2018). Neither of the cultivars in the present study exhibited as strong a difference between treatments. L-type lateral root density and length in apical segments of nodal roots under drought were similar in the two studies, but in the present study, nodal roots had greater lateral root branching in the well-watered treatment, possibly due to cultivar differences. Deep apical segments of Azucena had a notable increase in L-type lateral root density at week 5 of drought, though density, but not length, of its L-type lateral roots then declined from weeks five to six (Fig. 2). Reallocation of resources to development of fewer but longer L-type lateral roots could be advantageous under drought, since this could potentially place growing lateral roots in deeper soil. The continued development of L-type lateral roots in Azucena and IR64 probably benefits both genotypes under drought stress but may provide greater benefit to Azucena because of the deeper angle and greater length of the nodal roots on which the lateral roots are borne. The responses of Azucena provide an example of phene synergism, where the combined effects of nodal root length combined with lateral root length and density would most likely exceed the additive effects of these phenes individually.
Shallow nodal roots, on the other hand, showed little increase in L-type or S-type lateral root length over time in either treatment, despite greater branching density of S-type lateral roots compared with deep nodal roots in both treatments and cultivars (Figs. 2, 3). This difference in lateral root development between shallow and deep nodal roots could not be attributed to a drier growth medium, since the moisture content of shallow soil was just as great as deep soil in the irrigated treatment (Supplementary Fig. 2). The difference in branching between shallow and deep nodal roots therefore appears to be developmentally controlled rather than a response to the local environment.
Living tissue area was related to root metabolic cost
Reduced living tissue area has been proposed as a desirable trait for reducing the cost of soil exploration in crops, since roots with fewer living cells would require less maintenance respiration per unit length (Lynch 2011, 2013). Root cortical senescence in barley (Schneider et al. 2017b, 2017a) and reduced living tissue area via fewer cortical cells or development of root cortical aerenchyma in maize (Chimungu et al. 2014b; Saengwilai et al. 2014; Castañeda et al. 2018) were associated with less root respiration and better plant performance with water and nutrient stress. Phenotypes that reduce root metabolic cost may also contribute to drought tolerance in rice. Azucena showed greater reduction in root cross-sectional area and living tissue area in response to drought stress compared to IR64 (Fig. 4, Supplementary Fig. 5, Supplementary Table 3), which would be expected to reduce the metabolic cost of root exploration. The plasticity of living tissue area in Azucena compared to IR64 was driven by cortical, stele, and percent aerenchyma areas. Greater root cross-sectional area and living tissue area reductions may have allowed Azucena to conserve metabolic energy to permit continued axial root growth to access deep water under drought, despite the fact that metabolic cost per unit root length was still greater for Azucena even after drought-related reductions.
We found a positive relationship between root respiration rates per unit root length and phenotypes associated with root diameter: cortical area, stele area, and living tissue area, and a negative relationship with root cortical aerenchyma (Table 3), while there was no correlation between these root phenotypes and respiration rates per gram of root tissue (data not shown). Because it has larger diameter roots, respiration rates per unit length for nodal roots of Azucena were greater than those of IR64 (Supplementary Fig. 5), while there was little difference between genotypes when comparing respiration rates per gram of root tissue (data not shown). The greater metabolic cost per unit length of Azucena roots likely drove the greater drought-induced plasticity of root diameter, which resulted in production of relatively cheaper roots under stress to permit greater axial root elongation for deeper soil exploration. In the solid medium experiment, both genotype and low phosphorus were used to create a range of nodal root diameters, since low phosphorus was previously reported to reduce root diameter (Vejchasarn et al. 2016; de Bauw et al. 2019). Similar effects of root anatomy on respiration were found in both solid medium and hydroponic experiments (Table 3). An exception to this general rule occurred in the apical segment of nodal roots grown in solid medium, which had less cross-sectional area with low phosphorus, yet also had greater respiration rates than high phosphorus roots (Table 2). Low phosphorus tends to favor axial growth rather than lateral root branching (Borch et al. 1999; Hanlon et al. 2018), and the respiration rate of the axial root tip could reflect preferential allocation of resources to the tip to support exploration for more phosphorus.
We expected that the extent of aerenchyma formation would be negatively correlated with respiration rate per unit root length, as it is for maize (Zhu et al. 2010; Saengwilai et al. 2014; Castañeda et al. 2018). We found only weak relationships in mature nodal root tissues of rice, but stronger correlations when the less aerenchymatous apical tissue was included (Tables 2, 3). Rice has considerably greater aerenchyma formation than maize, comprising 42–75% of cortical area in rice (Supplementary Table 4) versus 0–20% of cortical area in greenhouse or field-grown maize (Zhu et al. 2010). Rice therefore has proportionately less living tissue area in the cortex than maize, and much of its living tissue area is found in the outer cell layers (epidermis, exodermis) and in the stele. Even so, the relationship between root respiration per root length and root diameter-associated anatomical phenotypes indicates that root anatomy can have a significant impact on root system metabolic cost and therefore drought tolerance in rice.
Xylem responses to drought were stronger in shallow roots
Previous research on the effects of drought on xylem phenotypes in rice has been mixed, with some studies showing no effect (Kadam et al. 2015), and others showing that some cultivars have smaller and fewer late metaxylem vessels with drought (Yambao et al. 1992; Mostajeran and Rahimi-Eichi 2008; Henry et al. 2012; Hazman and Brown 2018). In this study, we found that plasticity of metaxylem phenotypes was greater in shallow than in deep nodal roots. In subapical and basal segments of deep nodal roots, median xylem vessel area was barely affected by drought, and metaxylem vessel number was significantly reduced only in Azucena, resulting in 28% less theoretical axial conductance in basal segments and 40% reduction in subapical segments after six weeks of drought (Fig. 5, Supplementary Table 3). The number of xylem vessels was almost always greater in shallow than in deep roots in both cultivars, but their median area was only greater in well-watered Azucena plants (Fig. 4). Shallow roots of Azucena had significantly less metaxylem area with drought treatment, which resulted in less axial hydraulic conductance (Fig. 4, Supplementary Table 3). These changes were consistent with the hypothesis that a larger number of smaller metaxylem vessels would provide more hydraulic safety in drier soil, such as that experienced by the shallow roots. The fact that even subapical segments of deep nodal roots displayed reductions in theoretical axial hydraulic conductance suggests that metaxylem trait plasticity could be responding to both global and local signals.
Summary
Root phenotypes have a significant impact on drought tolerance of crop plants. We demonstrate here that in addition to cultivar differences in root phenotypes, it is important to consider the temporal and spatial aspects of root responses to drought. Even within the same root class, i.e. nodal roots, we observed differences in architectural and anatomical phenotypes depending on the age and developmental stage of the root, as reflected in the differing anatomy, lateral root branching phenotypes, and drought responses of shallow (younger, shorter) compared with deep (older, longer) nodal roots. As drought progresses, the apices of the growing roots of these two classes are exposed to very different soil environments. We therefore expected much stronger drought responses in shallow roots compared with deep roots. For some traits, shallow roots had much greater plasticity than deep roots, e.g. root cross-sectional area, median metaxylem vessel area, and L-type lateral root branching, but in other cases the shallow roots had similar or less plasticity than deep roots, e.g. metaxylem vessel number and S-type lateral root branching. The phenotypic responses to drought in shallow and deep roots were consistent with proposed strategies to reduce the metabolic cost of soil exploration, with greater diameter reductions in larger-diameter shallow roots and in Azucena compared with IR64. The greater plasticity of root phenotypes in Azucena in combination with deeper, longer nodal roots likely contributes to its drought tolerance.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AA:
-
Aerenchyma area
- TCA:
-
Cortical area
- HP:
-
High phosphorus treatment
- LTA:
-
Living tissue area
- LP:
-
Low phosphorus treatment
- MXA:
-
Median metaxylem vessel area
- MXV:
-
Number of metaxylem vessels
- percAA:
-
Percent aerenchyma area
- RXSA:
-
Root cross-sectional area
- TSA:
-
Stele area
References
Abd Allah AA, Badawy S, Zayed B, El-Gohary A (2010) The Role of Root System Traits in the Drought Tolerance of Rice (Oryza sativa L.). J Plant Prod 1(4):621–631. https://doi.org/10.21608/JPP.2010.86384
Aphalo PJ (2021) ggpmisc: miscellaneous extensions to 'ggplot2'. R package version 0.4.4. https://CRAN.R-project.org/package=ggpmisc. Accessed 4 Aug 2021
Arai-Sanoh Y, Takai T, Yoshinaga S, Nakano H, Kojima M, Sakakibara H, Kondo M, Uga Y (2014) Deep rooting conferred by DEEPER ROOTING 1 enhances rice yield in paddy fields. Sci Rep 4:5563. https://doi.org/10.1038/srep05563
Borch K, Bouma TJ, Lynch JP, Brown KM (1999) Ethylene: A regulator of root architectural responses to soil phosphorus availability. Plant Cell Environ 22:425–431. https://doi.org/10.1046/j.1365-3040.1999.00405.x
Castañeda TG, Brown KM, Lynch JP (2018) Reduced root cortical burden improves growth and grain yield under low phosphorus availability in maize. Plant Cell Environ 41:1579–1592. https://doi.org/10.1111/pce.13197
Chimungu J, Brown KM, Lynch JP (2014a) Large root cortical cell size improves drought tolerance in maize (Zea mays L.). Plant Physiol 166:2166–2178. https://doi.org/10.1104/pp.114.250449
Chimungu JG, Brown KM, Lynch JP (2014b) Reduced root cortical cell file number improves drought tolerance in maize. Plant Physiol 166:1943–1955. https://doi.org/10.1104/pp.114.249037
Daryanto S, Wang L, Jacinthe PA (2017) Global synthesis of drought effects on cereal, legume, tuber and root crops production: A review. Agric Water Manag 179:18–33. https://doi.org/10.1016/j.agwat.2016.04.022
de Bauw P, Vandamme E, Lupembe A, Mwakasege L, Senthilkumar K, Merckx R (2019) Architectural root responses of rice to reduced water availability can overcome phosphorus stress. Agronomy 9:1–24. https://doi.org/10.3390/agronomy9010011
FAO (2016) Crops and livestock products. License: CC BY-NC-SA 3.0 IGO. https://www.fao.org/faostat/en/#data/QCL. Accessed 1 Apr 2018
Gao Y, Lynch JP (2016) Reduced crown root number improves water acquisition under water deficit stress in maize (Zea mays L.). J Exp Bot 67:4545–4557. https://doi.org/10.1093/jxb/erw243
Gowda VRP, Henry A, Vadez V, Shashidhar HE, Serraj R (2012) Water uptake dynamics under progressive drought stress in diverse accessions of the OryzaSNP panel of rice (Oryza sativa). Funct Plant Biol 39:402–411. https://doi.org/10.1071/FP12015
Gowda VRP, Henry A, Yamauchi A, Shashidhar HE, Serraj R (2011) Root biology and genetic improvement for drought avoidance in rice. Field Crops Res 122:1–13. https://doi.org/10.1016/j.fcr.2011.03.001
Guet J, Fichot R, Lédée C, Laurans F, Cochard H, Delzon S, Bastien C, Brignolas F (2015) Stem xylem resistance to cavitation is related to xylem structure but not to growth and water-use efficiency at the within-population level in Populus nigra L. J Exp Bot 66:4643–4652. https://doi.org/10.1093/jxb/erv232
Guiderdoni E, Courtois B, Huang N, McCouch SR, Ghesquière A, Lorieux M, Filloux D, Albar L, Ahmandi N, Tharreau D, Notteghem JL (2006) The IR64-Azucena mapping population. In: Rocchi D, Despréaux D, Frison EA, Hubert B, Lantin MM (eds) France and the CGIAR: Delivering Scientific Results for Agricultural Development. CGIAR Secretariat Publications, Rome, pp 34–38
Hall B, Lanba A, Lynch JP (2019) Three-dimensional analysis of biological systems via a novel laser ablation technique. J Laser Appl 31:022602. https://doi.org/10.2351/1.5096089
Hanlon MT, Ray S, Saengwilai P, Luthe D, Lynch JP, Brown KM (2018) Buffered delivery of phosphate to Arabidopsis alters responses to low phosphate. J Exp Bot. https://doi.org/10.1093/jxb/erx454
Hazman M, Brown KM (2018) Progressive drought alters architectural and anatomical traits of rice roots. Rice 11:1–16. https://doi.org/10.1186/s12284-018-0252-z
Henry A (2013) IRRI’s drought stress research in rice with emphasis on roots: accomplishments over the last 50 years. Plant Root 7:5–19. https://doi.org/10.3117/plantroot.7.5
Henry A, Cal AJ, Batoto TC, Torres RO, Serraj R (2012) Root attributes affecting water uptake of rice (Oryza sativa) under drought. J Exp Bot 63:695–709. https://doi.org/10.1093/jxb/err313
Henry A, Gowda VRP, Torres RO, McNally KL, Serraj R (2011) Variation in root system architecture and drought response in rice (Oryza sativa): Phenotyping of the OryzaSNP panel in rainfed lowland fields. Field Crops Res 120:205–214. https://doi.org/10.1016/j.fcr.2010.10.003
Kadam NN, Tamilselvan A, Lawas L, Quinones C, Bahuguna R, Thomson M, Dingkuhn M, Muthurajan R, Struik P, Yin X, Jagadish S (2017) Genetic Control of Plasticity in Root Morphology and Anatomy of Rice in Response to Water Deficit. Plant Physiol 174(4):2302–2315. https://doi.org/10.1104/pp.17.00500
Kadam NN, Yin X, Bindraban PS, Struik PC, Jagadish KSV (2015) Does morphological and anatomical plasticity during the vegetative stage make wheat more tolerant of water deficit stress than rice? Plant Physiol 167:1389–1401. https://doi.org/10.1104/pp.114.253328
Kano M, Inukai Y, Kitano H, Yamauchi A (2011) Root plasticity as the key root trait for adaptation to various intensities of drought stress in rice. Plant Soil 342:117–128. https://doi.org/10.1007/s11104-010-0675-9
Kitomi Y, Kanno N, Kawai S, Mizubayashi T, Fukuoka S, Uga Y (2015) QTLs underlying natural variation of root growth angle among rice cultivars with the same functional allele of DEEPER ROOTING 1. Rice 8:16. https://doi.org/10.1186/s12284-015-0049-2
Klein SP, Schneider HM, Perkins AC, Brown KM, Lynch JP (2020) Multiple integrated root phenotypes are associated with improved drought tolerance. Plant Physiol 183:1011–1025. https://doi.org/10.1104/pp.20.00211
Kondo M, Aguilar A, Abe J, Morita S (2000) Anatomy of nodal roots in tropical upland and lowland rice varieties. Plant Prod Sci 3:437–445. https://doi.org/10.1626/pps.3.437
Lobell DB, Gourdji SM (2012) The influence of climate change on global crop productivity. Plant Physiol 160:1686–1697. https://doi.org/10.1104/pp.112.208298
Lynch JP (2013) Steep, cheap and deep: an ideotype to optimize water and N acquisition by maize root systems. Ann Bot 112:347–357. https://doi.org/10.1093/aob/mcs293
Lynch JP (2019) Root phenotypes for improved nutrient capture: an underexploited opportunity for global agriculture. New Phytol 223:548–564. https://doi.org/10.1111/nph.15738
Lynch JP (2015) Root phenes that reduce the metabolic costs of soil exploration: opportunities for 21st century agriculture. Plant Cell Environ 38:1775–1784. https://doi.org/10.1111/pce.12451
Lynch JP (2011) Root phenes for enhanced soil exploration and phosphorus acquisition: Tools for future crops. Plant Physiol 156:1041–1049. https://doi.org/10.1104/pp.111.175414
Lynch JP, Chimungu JG, Brown KM (2014) Root anatomical phenes associated with water acquisition from drying soil: targets for crop improvement. J Exp Bot 65:6155–6166. https://doi.org/10.1093/jxb/eru162
Lynch JP, Epstein E, Lauchli A, Weigt GI (1990) An automated greenhouse sand culture system suitable for studies of P nutrition. Plant Cell Environ 13:547–554. https://doi.org/10.1111/j.1365-3040.1990.tb01071.x
Lyu J, Zhang S, Dong Y, He W, Zhang J, Deng X, Zhang Y, Li X, Li B, Huang W, Wan W, Yu Y, Li Q, Li J, Liu X, Wang B, Tao D, Zhang G, Wang J, Xu X, Hu F, Wang W (2013) Analysis of elite variety tag SNPs reveals an important allele in upland rice. Nat Commun 4:2138. https://doi.org/10.1038/ncomms3138
McLean J, Hardy B, Hettel G (2013) Rice Almanac, 4th edition. Source book for one of the most important economic activities on earth. International Rice Research Institute, Los Baños, Philippines
Mostajeran A, Rahimi-Eichi V (2008) Drought stress effects on root anatomical characteristics of rice cultivars (Oryza sativa L.). Pak J Biol Sci 11:2173–2183. https://doi.org/10.3923/pjbs.2008.2173.2183
Murphy J, Riley J (1962) A modified single solution reagent for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36. https://doi.org/10.1016/S0003-2670(00)88444-5
Niones JM, Inukai Y, Suralta RR, Yamauchi A (2015) QTL associated with lateral root plasticity in response to soil moisture fluctuation stress in rice. Plant Soil 391:63–75. https://doi.org/10.1007/s11104-015-2404-x
Niones JM, Suralta RR, Inukai Y, Yamauchi A (2013) Roles of root aerenchyma development and its associated QTL in dry matter production under transient moisture stress in rice. Plant Prod Sci 16:205–216. https://doi.org/10.1626/pps.16.205
Niones JM, Suralta RR, Inukai Y, Yamauchi A (2012) Field evaluation on functional roles of root plastic responses on dry matter production and grain yield of rice under cycles of transient soil moisture stresses using chromosome segment substitution lines. Plant Soil 359:107–120. https://doi.org/10.1007/s11104-012-1178-7
R Core Team (2016) R: a language and environment for statistical computing
Richards RA, Passioura JB (1989) A breeding program to reduce the diameter of the major xylem vessel in the seminal roots of wheat and its effect on grain yield in rain-fed environments. Aust J Agric Res 40:943–950. https://doi.org/10.1071/AR9890943
Saengwilai P, Nord EA, Chimungu JG, Brown KM, Lynch JP (2014) Root cortical aerenchyma enhances nitrogen acquisition from low-nitrogen soils in maize. Plant Physiol 166:726–735. https://doi.org/10.1104/pp.114.241711
Schneider H, Lynch J (2020) Should root plasticity be a crop breeding target? Front Plant Sci 11:1–16. https://doi.org/10.3389/fpls.2020.00546
Schneider HM, Wojciechowski T, Postma JA, Brown KM, Lücke A, Zeisler V, Schreiber L, Lynch JP (2017a) Root cortical senescence decreases root respiration, nutrient content and radial water and nutrient transport in barley. Plant Cell Environ 1392–1408. https://doi.org/10.1111/pce.12933
Schneider H, Wojciechowski T, Postma J a, Brown KM, Lucke A, Zeisler V, Schreiber L, Lynch JP (2017b) Root cortical senescence decreases root metabolic costs and radial water and nutrient transport in barley. Plant Cell Environ 40:1392–1408. https://doi.org/10.1111/pce.12933
Sperry JS, Saliendra NZ (1994) Intra- and inter-plant variation in xylem embolism in Betula occidentalis. Plant Cell Environ 17:1233–1241. https://doi.org/10.1111/j.1365-3040.1994.tb02021.x
Strock CF, Burridge J, Massas ASF, Beaver J, Beebe S, Camilo SA, Fourie D, Jochua C, Miguel M, Miklas PN, Mndolwa E, Nchimbi-Msolla S, Polania J, Porch TG, Rosas JC, Trapp JJ, Lynch JP (2019) Seedling root architecture and its relationship with seed yield across diverse environments in Phaseolus vulgaris. Field Crops Res 237:53–64. https://doi.org/10.1016/j.fcr.2019.04.012
Strock CF, Lynch JP (2020) Root secondary growth: an unexplored component of soil resource acquisition. Ann Bot 126:205–218. https://doi.org/10.1093/aob/mcaa068
Strock CF, Morrow de la Riva L, Lynch J (2018) Reduction in Root Secondary Growth as a Strategy for Phosphorus Acquisition. Plant Physiol 176:691–703. https://doi.org/10.1104/pp.17.01583
Suralta RR, Kano-Nakata M, Niones JM, Inukai Y, Kameoka E, Tran TT, Menge D, Mitsuya S, Yamauchi A (2016) Root plasticity for maintenance of productivity under abiotic stressed soil environments in rice: Progress and prospects. Field Crops Res 220:57–66. https://doi.org/10.1016/j.fcr.2016.06.023
Suralta RR, Lucob NB, Perez L, Niones JM, Nguyen H (2015) Developmental and quantitative trait loci analyses of root plasticity in response to soil moisture fluctuation in rice. Philipp J Crop Sci 40:12–24
Terashima K, Hiraoka H, Nishiyama I (1987) Varietal difference in the root of rice plant. Jpn J Crop Sci 56:521–529. https://doi.org/10.1626/JCS.56.521
Tyree MT, Ewers FW (1991) The hydraulic architecture of trees and other woody plants. New Phytol 119:345–360. https://doi.org/10.1111/j.1469-8137.1991.tb00035.x
Uga Y, Ebana K, Abe J, Morita S, Okuno K, Yano M (2009) Variation in root morphology and anatomy among accessions of cultivated rice (Oryza sativa L.) with different genetic backgrounds. Breed Sci 59:87–93. https://doi.org/10.1270/jsbbs.59.87
Uga Y, Okunoi K, Yano M (2008) QTLs underlying natural variation in stele and xylem structures of rice root. Breed Sci 58:7–14. https://doi.org/10.1270/jsbbs.58.7
Uga Y, Sugimoto K, Ogawa S, Rane J, Ishitani M, Hara N, Kitomi Y, Inukai Y, Ono K, Kanno N, Inoue H, Takehisa H, Motoyama R, Nagamura Y, Wu J, Matsumoto T, Takai T, Okuno K, Yano M (2013) Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nat Genet 45:1097–1102. https://doi.org/10.1038/ng.2725
Vadez V (2014) Root hydraulics: The forgotten side of roots in drought adaptation. Field Crops Res 165:15–24. https://doi.org/10.1016/j.fcr.2014.03.017
Vejchasarn P (2014) Nutritional and genetic architecture of root traits in rice. [Doctoral dissertation, The Pennsylvania State University]. PSU Campus Repository. https://etda.libraries.psu.edu/catalog/21425
Vejchasarn P, Lynch JP, Brown KM (2016) Genetic variability in phosphorus responses of rice root phenotypes. Rice 9:29. https://doi.org/10.1186/s12284-016-0102-9
Venuprasad R, Lafitte H, Atlin G (2007) Response to direct selection for grain yield under drought stress in rice. Crop Sci 47(1):285–293. https://doi.org/10.2135/cropsci2006.03.0181
Wasson AP, Richards RA, Chatrath R, Misra SC, Prasad SVS, Rebetzke GJ, Kirkegaard JA, Christopher J, Watt M (2012) Traits and selection strategies to improve root systems and water uptake in water-limited wheat crops. J Exp Bot 63:3485–3498. https://doi.org/10.1093/jxb/ers111
Wei T, Simko V (2017) R package “corrplot”: Visualization of a correlation matrix (Version 0.84) Accessed 4 Aug 2021
Wickham H (2016) ggplot2: Elegant graphics for data analysis. Springer-Verlag, New York
Yambao EB, Ingram KT, Real JG (1992) Root xylem influence on the water relations and drought resistance of rice. J Exp Bot 43:925–932. https://doi.org/10.1093/jxb/43.7.925
Yoshida S, Forno DA, Cock JH, Gomez KA (1976) Routine procedure for growing rice plants in culture solution. In: Laboratory Manual for Physiological Studies of Rice, 3rd edn. The International Rice Research Institute, Los Baños, Philippines
Yoshida S, Hasegawa S (1982) The rice root system: its development and function. In: Drought resistance in crops with emphasis on rice. The International Rice Research Institute, Los Baños, Philippines
Zhu J, Brown KM, Lynch JP (2010) Root cortical aerenchyma improves the drought tolerance of maize (Zea mays L.). Plant Cell Environ 33:740–749. https://doi.org/10.1111/j.1365-3040.2009.02099.x
Acknowledgements
This work was supported by a NIPGR travel grant to JG, a Royal Thai Government Scholarship to PV, and the USDA National Institute of Food and Agriculture Federal Appropriations under project PEN04732. Any opinions, findings, and conclusions or recommendations expressed in this publication are those of the authors and do not necessarily reflect the views of the USDA National Institute of Food and Agriculture. The authors thank Christopher Strock for his careful feedback on this manuscript.
Funding
This work was supported by a NIPGR travel grant to JG, a Royal Thai Government Scholarship to PV, and the USDA National Institute of Food and Agriculture Federal Appropriations under project PEN04732. Any opinions, findings, and conclusions or recommendations expressed in this publication are those of the authors and do not necessarily reflect the views of the USDA National Institute of Food and Agriculture.
Author information
Authors and Affiliations
Contributions
JR and KB conceived and designed the drought time course and solution culture respiration study, and JG, PV, and KB conceived and designed the solid media respiration study. JR executed and collected data for the drought time course and solution culture respiration study, and JG and PV executed and collected data for the solid media respiration study. JR analyzed and prepared data with guidance from KB. JR and KB interpreted results and wrote manuscript with input from JL. JG and PV approved the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor: Matthias Wissuwa.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fonta, J.E., Giri, J., Vejchasarn, P. et al. Spatiotemporal responses of rice root architecture and anatomy to drought. Plant Soil 479, 443–464 (2022). https://doi.org/10.1007/s11104-022-05527-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-022-05527-w