Abstract
Extracts from the biomass of Ruta graveolens and Ruta graveolens ssp. divaricata cultured in vitro under different light conditions (far-red, red and blue light, UV-A irradiation, in darkness and white light) were tested for the amounts of free phenolic acids and cinnamic acid (twelve compounds) as well as furanocoumarins and umbelliferone (seven compounds) using HPLC methods. Total amounts of the investigated groups of compounds in the cultures of both plants increased from 2.6 to 6.7 times, depending on light quality, and the maximum values reached were 106.50 and 1,276.74 mg 100 g−1 DW (in R. graveolens), and 106.97 and 262.54 mg 100 g−1 DW (in the subspecies), respectively. Both white light and blue light were equally beneficial for the total production of phenolic acids in cultures of both plants, whereas the total production of furanocoumarins was clearly better stimulated by blue light in R. graveolens and by darkness in the subspecies (i.e. the amounts were respectively 1.44 and 1.7 times higher than in the biomass cultivated under white light). The amounts of individual compounds in both plant cultures increased from about 2.2 to 26.3 times depending on light quality. The following bioactive compounds were obtained in quantities which are of interest from a practical perspective: in R. graveolens culture—protocatechuic acid (45 mg 100 g−1 DW), isopimpinellin (about 500 mg 100 g−1 DW) and bergapten (about 270 mg 100 g−1 DW), and in the subspecies culture: p-coumaric acid (70 mg 100 g−1 DW) and isopimpinellin (about 210 mg 100 g−1 DW).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phenolic acids are a group of plant metabolites highly valued in phytotherapy and cosmetology. These compounds exhibit numerous biological activities, the most important of which are their immunostimulating, antioxidant and anticancer properties (Abdel-Wahab et al. 2003; De Graff et al. 2003; Ekiert and Czygan 2007; Nakamura et al. 2001; Sroka and Cisowski 2003). Linear furanocoumarins have an established position in therapy as antiproliferative and photosensitizing agents, and some of them are calcium and potassium channel blockers (Bohuslavizki et al. 1994; Ekiert and Czygan 2007). Our earlier studies on phenolic acid accumulation in in vitro cultures of Ruta graveolens and Ruta graveolens ssp. divaricata focused on the effect of the concentration of plant growth regulators in the Linsmaier-Skoog (L-S) medium (Ekiert et al. 2008, 2009; Piekoszewska et al. 2008). The studies on linear furanocoumarins concentrated on the effect of culture type and on the dynamics of accumulation in culture growth cycles (Ekiert et al. 2001, 2005; Ekiert and Czygan 2005).
Apart from light intensity and photoperiod, the quality of light is one of the known ambient factors influencing metabolite accumulation in plant in vitro cultures (Ramawat and Mathur 2007). The majority of studies on the effect of light quality on the production of secondary metabolites have been concerned with other groups of compounds, e.g. alkaloids, anthocyanins glycosides and flavonoids (Ramawat and Mathur 2007). Also, recently the influence of light quality on the accumulation of other groups of plant pigments such as naphthoquinones and betacyanins has been studied (Zhang et al. 2010; Zhao et al. 2010). A few papers have dealt with the effect of light quality on the accumulation of phenolic acids, e.g. chlorogenic acid accumulation in Haplopappus sp. and Populus sp. cultures (Ramawat and Mathur 2007).
Our preliminary studies indicated a marked effect of monochromatic light on the accumulation of both phenolic acids and linear furanocoumarins in in vitro cultures of R. graveolens and its subspecies (Ekiert and Czygan 2007; Ekiert and Gomółka 1999).
The aim of our present experiments was to examine the effect of light with different wavelengths (far-red, red and blue, and UV-A irradiation), darkness and white light on the accumulation of phenolic acids and cinnamic acid, one of the parent compounds of these metabolites (twelve compounds), and linear furanocoumarins and their biogenetic precursor—umbelliferone (seven compounds) in the biomass from R. graveolens and its subspecies R. graveolens ssp. divaricata in vitro cultures and to propose the best conditions for the production of these two groups of bioactive metabolites.
The effects of light quality on the accumulation of phenolic acids and linear furanocoumarins in in vitro cultures of R. graveolens and R. graveolens ssp. divaricata have so far not been investigated in other research centers. However, the current literature contains several reports describing the significance of other factors, such as elicitors for the production of coumarins, including linear furanocoumarins in R. graveolens in vitro cultures (Orlita et al. 2008a, b).
Materials and methods
Plant material
The shoot culture of Ruta graveolens L. was initiated in our Department of Pharmaceutical Botany from hypocotyl segments of sterile seedlings—for details see Ekiert and Czygan (2007).
The shoot-differentiating callus culture of Ruta graveolens ssp. divaricata (Tenore) Gams. was established in the Institute for Biosciences of Würzburg University (Germany)—for details see Ekiert and Czygan (2007).
Experimental stationary liquid shoot cultures of R. graveolens were maintained on two variants of Linsmaier and Skoog medium (L-S) (1965) with different concentrations of plant growth regulators; variant I: 2 mg l−1 α-naphthaleneacetic acid (NAA) and 2 mg l−1 N6-benzyladenine (BA), variant II: 3 mg l−1 NAA and 1 mg l−1 BA, while the stationary shoot-differentiating liquid callus cultures of R. graveolens ssp. divaricata were maintained on a variant of L-S medium containing 2 mg l−1 NAA and 2 mg l−1 BA. The cultures were grown under different light conditions, at 25 ± 2 °C. They were subcultured every 4 weeks (three series).
Light conditions
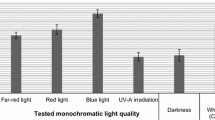
In vitro cultures of both plants were maintained under light of different spectra: far-red light (770–800 nm)—60 W incandescent light with standard filter no. 405 orange + standard filter no. 420, Compact Light B.V. Amsterdam, red light (647–770 nm)—Philips lamp TLD 36 W, blue light (450–492 nm)—Philips lamp TLD 36 W, UV-A irradiation (360–450 nm)—Philips lamp TLD 36 W, darkness and white light (390–760 nm)—Philips lamp TLD 36 W. Cultures grown under white light served as the control.
Extraction
The biomass from in vitro cultures (1.0 g) collected after 4-week growth cycles (three series) was subjected to extraction twice with boiling methanol (50 ml) under a reflux condenser for 3 h. The extracts were combined, condensed and evaporated to dryness. The residue was quantitatively dissolved in 5 ml of methanol and analyzed by HPLC.
HPLC analysis
In the methanolic extracts of the biomass, eleven phenolic acids and cinnamic acid as one group, and six linear furanocoumarins and umbelliferone as another group were quantified using RP-HPLC methods according to Ellnain-Wojtaszek and Zgórka (1999) with our modifications and those of Ekiert and Gomółka (1999), respectively.
Separation of both groups of metabolites was performed using a LiChrospher 100 RP-18 (4 mm × 20 cm) analytical column. The separation conditions for phenolic acids were as follows: solvent system composed of methanol: 0.5 % acetic acid—gradient elution, flow rate: 1 ml min−1, UV detector: λ = 254 nm, standards: caffeic, chlorogenic, cinnamic, protocatechuic, rosmarinic, salicylic, sinapic and syringic acids from Sigma, and p-coumaric, ferulic, p-hydroxybenzoic and vanillic acids from Fluka.
The separation conditions for coumarins were different: the solvent system contained methanol: water—gradient elution, flow rate: 1 ml min−1, detector UV: λ = 310 nm, standards: bergapten, imperatorin, psoralen, umbelliferone, xanthotoxin from Roth, and isopimpinellin, marmesin from Institute of Pharmacology, Polish Academy of Sciences, Kraków (Poland).
Results
Increases in biomass in the cultures of both plants
Ruta graveolens shoot cultures were maintained on two variants of L-S medium (variant I: 2 mg l−1 NAA and 2 mg l−1 BA, variant II: 3 mg l−1 NAA and 1 mg l−1 BA) chosen as “productive media” and as good “growth media” based on the experiments concerning the influence of growth regulators on the accumulation of phenolic acids (Ekiert et al. 2009). As shown by our earlier studies on the accumulation of linear furanocoumarins in R. graveolens cultures, variant I was beneficial for the accumulation of this group of compounds and good for increasing the biomass (Ekiert 2004). The changes in the dry biomass of the shoots cultivated under different light conditions on the two variants of L-S medium were different; namely it increased 1.80–3.64 times and 1.93–3.77 times, respectively, within a 4-week growth cycle. The greatest increases were observed in the shoots growing under blue light, while extremely low growth in biomass occurred under far-red light. The L-S medium containing 2 mg l−1 NAA and 2 mg l−1 BA was selected in our earlier experiments as a “productive medium” for the accumulation of both furanocoumarins and phenolic acids, and as a good “growth medium” for R. graveolens ssp. divaricata in vitro culture (Ekiert 2004; Piekoszewska et al. 2008). In the previous experiment, increases in dry biomass on this medium varied, depending on light conditions, from 2.75 times under far-red light to 6.35 times under blue light.
Accumulation of phenolic acids and cinnamic acid
Ruta graveolens cultures
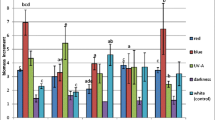
Ruta graveolens shoots growing on variant I of L-S medium showed an ability to synthesize five phenolic acids, namely: ferulic, p-coumaric, protocatechuic, syringic, vanillic, and also cinnamic acid (Table 1). The tested extracts did not contain caffeic, chlorogenic, rosmarinic, salicylic, sinapic, or p-hydroxybenzoic acids. Both blue light and white light were beneficial for the total accumulation of the compounds determined. Their total amounts depended on light quality and ranged from 34.41 mg 100 g−1 DW (UV-A irradiation) to 83.16 mg 100 g−1 DW (blue light), and 88.03 mg 100 g−1 DW (white light). Three phenolic acids, namely: protocatechuic, p-coumaric and syringic, were the main metabolites that accumulated in the highest amounts under blue light and white light. The maximum amounts of the bioactive protocatechuic acid were about 26 and 32 mg 100 g−1 DW, whereas the respective values for p-coumaric and syringic acids were similar and reached about 17–20 mg 100 g−1 DW. The accumulation of cinnamic acid was facilitated by monochromatic light: either blue or red or far-red, and UV-A irradiation, with the amounts of 10–14 mg 100 g−1 DW. The maximum amounts of vanillic and ferulic acids did not exceed 9.50 and 4.50 mg 100 g−1 DW, respectively.
The extracts from the biomass of R. graveolens cultivated on variant II of L-S medium contained cinnamic acid and a composition of phenolic acids identical to that of the biomass cultivated on variant I of that medium (Table 1). The highest total amounts of these compounds were also observed in the extracts from the biomass growing under blue light and white light (91.16 and 106.50 mg 100 g−1 DW, respectively). These amounts were 1.15 times greater than in the biomass cultured on variant I of L-S medium. The lowest total amounts of the metabolites determined were observed under far-red light (23.63 mg 100 g−1 DW). Two compounds dominated quantitatively, namely protocatechuic acid and p-coumaric acid. Their maximum amounts were high and of a similar order of magnitude, about 45 mg 100 g−1 DW. The amounts of syringic, vanillic, ferulic and cinnamic acids were lower and did not exceed 12 mg 100 g−1 DW for syringic acid and 10 mg 100 g−1 DW for the other compounds. Accumulation of all the phenolic acids and cinnamic acid was facilitated by blue light and white light too, while for the accumulation of cinnamic acid red light and far-red light were beneficial.
The maximum total amounts of the metabolites in the extracts from the biomass cultured on variant II of L-S medium were greater in comparison with the biomass from variant I. The maximum amounts of therapeutically interesting compounds, i.e. protocatechuic acid (45.44 mg 100 g−1 DW—blue light) and p-coumaric acid (43.14 mg 100 g−1 DW—white light) were also higher. A more efficient accumulation of the examined metabolites on variant II of L-S medium confirmed our decision to test the two “productive media” selected in earlier studies (Ekiert et al. 2009).
Ruta graveolens ssp. divaricata cultures
Extracts of Ruta graveolens ssp. divaricata biomass cultivated on variant I of L-S medium showed a different composition of phenolic acids in comparison with the biomass of Ruta graveolens (Table 2). Apart from protocatechuic and p-coumaric acids and cinnamic acid, they also contained caffeic and chlorogenic acids. Ferulic, rosmarinic, salicylic, sinapic, syringic, vanillic and p-hydroxybenzoic acids were not detected in the tested extracts. The total amounts of the main compounds were highest under blue light and white light. The total amounts of the above-mentioned metabolites varied considerably, from 15.92 mg 100 g−1 DW (far-red light) to 99.76 mg 100 g−1 DW (blue light) and 106.97 mg 100 g−1 DW (white light). The biomass of R. graveolens ssp. divaricata was characterized by a marked ability to accumulate one compound, p-coumaric acid, which confirmed the results of our earlier studies (Ekiert et al. 2008; Piekoszewska et al. 2008). The maximum amounts of this compound, interesting from a practical perspective, equal to 38.86 mg 100 g−1 DW (blue light) and 74.33 mg 100 g−1 DW (white light), indicate potential applicability of these results. The maximum amounts of the remaining phenolic acids did not exceed 10 mg 100 g−1 DW. Only cinnamic acid was accumulated in higher quantities; its maximum amount was 38.68 mg 100 g−1 DW (blue light).
Accumulation of linear furanocoumarins
Ruta graveolens cultures
Four of the seven coumarin compounds tested were found in the biomass extracts from Ruta graveolens cultured on variant I of L-S medium irrespective of the light conditions: bergapten, imperatorin, isopimpinellin and psoralen (Table 3). None of the extracts contained marmesin, umbelliferone or xanthotoxin. The total amounts of determined compounds were highest under blue light. They depended on light quality and ranged from 372.01 mg 100 g−1 DW (red light) to 1,276.74 mg 100 g−1 DW (blue light). The total amount of the compounds was also significant in the extracts from the biomass grown under white light (884.03 mg 100 g−1 DW), but 1.44 times lower than in that grown under blue light. The amounts of individual furanocoumarins fluctuated within a wide range, from several milligrams to about 0.5 g 100 g−1 DW. Their amounts increased from 2.20 to 6.10 times depending on light quality. Blue light was the most beneficial type of illumination for the accumulation of all the compounds determined. The maximum amounts of psoralen and isopimpinellin were very high, as high as about 500 mg 100 g−1 DW, the amounts of bergapten were also high, reaching about 270 mg 100 g−1 DW. The imperatorin content was much lower, equalling 14.02 mg 100 g−1 DW. The psoralen content was also the highest in the presence of white light (about 370 mg 100 g−1 DW).
The extracts from the biomass growing on the other test variant of L-S medium (variant II) showed the same composition of the metabolites (Table 3). Blue light was also more beneficial for the total amount of linear furanocoumarins than white light.
The total amounts of the compounds were generally lower than in the extracts from the biomass cultured on variant I of L-S medium. They were also dependent on light quality and varied considerably, from 164.22 mg 100 g−1 DW (far-red) to 993.75 mg 100 g−1 DW (blue light). A significant amount of the compounds was demonstrated to occur also in the extracts from the biomass growing under white light (724.85 mg 100 g−1 DW).
The increases in the amounts of individual linear furanocoumarins varied even more than those in the extracts from variant I of L-S medium, and ranged from 4.50 to 23.00 times, depending on light quality. Blue light was also the most effective illumination in stimulating the accumulation of the individual metabolites. Two compounds dominated quantitatively: psoralen and isopimpinellin. The maximum amount of psoralen, of about 500 mg 100 g−1 DW, was almost identical to that for variant I of L-S medium. Isopimpinellin was also accumulated in considerable amounts, but its maximum content of about 290 mg 100 g−1 DW was 1.69 times lower than on variant I of L-S medium. The accumulation of bergapten was also less efficient (max. 179.50 mg 100 g−1 DW). In contrast, the imperatorin content (23.00 mg 100 g−1 DW) was 1.64 times higher than on L-S medium variant I. Blue light was also the most effective in stimulating the accumulation of all four compounds. High isopimpinellin content of 288.16 mg 100 g−1 DW was also determined in the extracts of the biomass cultured in the dark.
Ruta graveolens ssp. divaricata cultures
Extracts of Ruta graveolens ssp. divaricata growing on L-S medium variant I were demonstrated to contain a qualitatively richer composition of linear furanocoumarins in comparison with Ruta graveolens biomass (Table 4). Five of the seven compounds tested were identified, namely bergapten, imperatorin, isopimpinellin, marmesin and xanthotoxin. The presence of psoralen and umbelliferone was not confirmed. In general, the highest total amount of linear furanocoumarins was produced in the dark. The total amounts of the metabolites were much lower than in the biomass from R. graveolens in vitro cultures and also fluctuated within a wide range depending on light quality, from 78.26 mg 100 g−1 DW (far-red light) to 262.54 mg 100 g−1 DW (darkness). The total amount of linear furanocoumarins in the extracts from the biomass growing under white light was relatively high and amounted to 172.74 mg 100 g−1 DW. Among the linear furanocoumarins determined, isopimpinellin dominated quantitatively. Its amount increased, depending on light quality, 7.76 times and reached a maximum value in the dark (206.39 mg 100 g−1 DW). The amounts of the remaining four metabolites increased from 3.20 to 26.30 times depending on light quality. Maximum amounts of other therapeutically important compounds were observed for xanthotoxin in the dark (21.37 mg 100 g−1 DW), and for bergapten and imperatorin under white light (about 37 and 20 mg 100 g−1 DW, respectively).
Discussion
Our studies demonstrated that blue light and white light were the most effective in stimulating the accumulation of free phenolic acids in extracts from the biomass of both Ruta graveolens and Ruta graveolens ssp. divaricata in vitro cultures. The total amounts of these compounds accumulated in the biomass of both plants cultured in vitro under the two types of illumination were comparable.
The blue light and white light were also shown to have a beneficial effect on the total accumulation of linear furanocoumarins in R. graveolens cultures. The amount of furanocoumarins obtained in the presence of blue light was 1.44 times higher than under white light.
All the other lighting conditions were much less advantageous for the accumulation of the majority of the metabolites under study. Only cinnamic acid was accumulated more efficiently under red light and far-red light.
The beneficial effect of white light and blue light on the accumulation of plant metabolites is known. White light has been demonstrated to produce a beneficial effect on the accumulation of alkaloids in R. graveolens cultures and of flavonoids in Citrus aurantium cultures, as well as on the production of cardiac glycosides by Digitalis lanata or indole alkaloids and anthocyanin glycosides in Catharanthus roseus cultures (Ramawat and Mathur 2007). White light also stimulates the accumulation of betacyanins in callus cultures of Suaeda salsa (Zhao et al. 2010).
Blue light facilitates the accumulation of anthocyanins glycosides in Haplopappus gracilis and Populus sp. cultures. However, red light has been found beneficial for the accumulation of chlorogenic acid in in vitro culture of Haplopappus gracilis (Ramawat and Mathur 2007).
In contrast, the maximum accumulation of linear furanocoumarins in R. graveolens ssp. divaricata cultures occurred in the dark. This result is not surprising. Our earlier studies on furanocoumarin accumulation in Ammi majus cultures proved that the absence of light stimulated the accumulation of bergapten (Ekiert 1988, 1993). Studies of different groups of alkaloids, conducted in other research centers, have demonstrated a disadvantageous effect of white light on the accumulation of caffeine in Camellia sinensis cultures, nicotine in Nicotiana tabacum cultures and tropane alkaloids in Hyoscyamus muticus cultures (Aly et al. 2010). Recently, it has been demonstrated that white light inhibits the biosynthesis of naphthoquinone pigments, shikonin and its derivatives in cell cultures of Lithospermum erythrorhizon (Zhang et al. 2010).
The effect of light quality on morphogenesis in in vitro cultures is widely known. The influence of light quality on the induction and expression of embryogenesis in in vitro cultures of Agave tequilana var. Azul (Rodríguez-Sahagún et al. 2011) and Campanula punctata var. rubriflora (Sivanesan et al. 2011) and the induction of protocorm-like bodies in in vitro cultures of orchids – Dendrobium officinale (Lin et al. 2011) and Oncidium sp. (Mengxi et al. 2011) represent good examples. Experiments concerning the influence of light quality on the accumulation of primary and secondary plant metabolites are rare. Thus, our present results are of great value from the point of view of basic research as they document the effect of light quality on the accumulation of bioactive secondary metabolites such as phenolic acids, cinnamic acid and linear furanocoumarins. The study also proves that the optimal light quality for the accumulation of different compounds should be chosen empirically.
Some of the results obtained may have practical value. The most important findings include the demonstration that higher total amounts of biologically active linear furanocoumarins can be obtained from cultures maintained under blue light (R. graveolens cultures) or in the dark (R. graveolens ssp. divaricata cultures) than under white light. Such lighting conditions, namely continuous blue light or darkness, can be easily applied to in vitro cultures and the biomass of both plants cultivated in vitro under such conditions can be proposed as a rich, potential source of the therapeutically important metabolites investigated in our study.
The maximum total amounts of the phenolic acids confirmed in the biomass from the in vitro cultures of both plants were 1.60 times greater than (R. graveolens culture) or equal to (R. graveolens ssp. divaricata) their amounts in the above-ground parts of plants growing under natural conditions (Ekiert et al. 2009; Piekoszewska et al.—unpublished). The maximum total amounts of coumarins determined in R. graveolens cultures were 2.20 times greater than in the above-ground parts of plants growing in vivo (Ekiert and Gomółka 1999). Only the maximum total amount of coumarins in in vitro cultures of R. graveolens subspecies was lower than in the above-ground parts of plants growing in vivo (Ekiert et al. 2005).
The present data confirm that in vitro cultures of both plants are an abundant source of some therapeutically important compounds, namely: R. graveolens cultures—protocatechuic acid (45 mg 100 g−1 DW), bergapten (about 270 mg 100 g−1 DW) and isopimpinellin (about 500 mg 100 g−1 DW), and R. graveolens ssp. divaricata cultures—p-coumaric acid (70 mg 100 g−1 DW) and isopimpinellin (about 210 mg 100 g−1 DW). Higher amounts of the metabolites determined here can be obtained from cultures with a higher degree of organogenesis, i.e. R. graveolens shoot cultures. Our results confirm this known dependence in plant biotechnology (Charlwood et al. 1990).
Conclusions
The results obtained in the present study have shown that the production of free phenolic acids and cinnamic acid, and of linear furanocoumarins is dependent on light quality. White light and blue light stimulated the total production of phenolic acids in cultures of both test plants. The highest total amounts of furanocoumarins were found in Ruta graveolens culture under blue light and in the subspecies culture in the absence of light. The maximum amounts of some bioactive metabolites are of interest from a practical point of view.
Abbreviations
- BA:
-
N6-Benzyladenine
- DW:
-
Dry weight
- HPLC:
-
High-pressure liquid chromatography
- L-S:
-
Linsmaier and Skoog
- NAA:
-
α-Naphthaleneacetic acid
References
Abdel-Wahab MH, El-Mahdy MA, Abd-Ellah MF, Helal GK, Khalifa F, Hamada FM (2003) Influence of p-coumaric acid on doxorubicin-induced oxidative stress in rat’s heart. Pharmacol Res 48:461–465
Aly UI, El-Shabrawi HM, Hanafy M (2010) Impact of culture conditions on alkaloid production from undifferentiated cell suspension cultures of Egyptian henbane. Aust J Basic Appl Sci 4:4717–4725
Bohuslavizki KH, Hänsel W, Kneip A, Koppenhöfer E, Niemöller E, Sanmann K (1994) Mode of action of psoralens, benzofurans, acridinons, and coumarins on the ionic currents in intact myelinated nerve fibres and its significance in demyelinating diseases. Gen Physiol Biophys 13:309–328
Charlwood BV, Charlwood SK, Molina-Tores JM (1990) Accumulation of secondary compounds by organized plant cultures. In: Charlwood BV, Rhodes MJC (eds) Secondary products from plant tissue cultures. Clarendon Press, Oxford, pp 167–200
De Graff WG, Myers LS, Mitchell JB, Hahn SM (2003) Protection against adriamycin cytotoxicity and inhibition of DNA topoisomerase II activity by 3,4-dihydroxybenzoic acid. Int J Oncol 23:159–163
Ekiert H (1988) Badania nad metabolitami hodowli tkankowej Ammi majus L. Doctor’s thesis, Medical Academy in Kraków (Poland), in polish
Ekiert H (1993) Ammi majus L (Bishop’s weed): In vitro culture and the production of coumarin compounds. In: Bajaj YPS (ed) Biotechnology in agriculture and forestry. Medicinal and aromatic plants IV. Springer, Berlin, pp 1–17
Ekiert H (2004) Accumulation of biologically active furanocoumarins within in vitro cultures of medicinal plants. In: Ramawat KG (ed) Biotechnology of medicinal plants, Vitalizer and therapeutic. Science Publishers, Enfield, pp 267–296
Ekiert H, Czygan FC (2005) Accumulation of biologically active furanocoumarins in agitated cultures of Ruta graveolens L. and Ruta graveolens ssp. divaricata (Tenore) Gams. Pharmazie 60:623–626
Ekiert H, Czygan FC (2007) Secondary metabolites in in vitro cultures of Ruta graveolens L. and Ruta graveolens ssp. divaricata (Tenore) Gams. In: Ramawat KG, Merillon JM (eds) Biotechnology: secondary metabolites. Plants and microbes. Science Publishers, Plymouth, pp 445–482
Ekiert H, Gomółka E (1999) Effect of light on contents of coumarin compounds in shoots of Ruta graveolens L. cultured in vitro. Acta Soc Bot Polon 68:197–200
Ekiert H, Chołoniewska M, Gomółka E (2001) Accumulation of furanocoumarins in Ruta graveolens L. shoot culture. Biotechnol Lett 23:543–545
Ekiert H, Abou-Mandour AA, Czygan FC (2005) Accumulation of biologically active furanocoumarins in Ruta graveolens ssp. divaricata (Tenore) Gams in vitro culture. Pharmazie 60:66–68
Ekiert H, Piekoszewska A, Szewczyk A, Baczyńska S, Kuś A (2008) Isolation and identification of selected phenolic acids from biomass of Ruta graveolens L. and Ruta graveolens ssp. divaricata (Tenore) Gams cultured in vitro. Acta Biochim Polon 55:61
Ekiert H, Szewczyk A, Kuś A (2009) Free phenolic acids in Ruta graveolens L. in vitro culture. Pharmazie 64:694–696
Ellnain-Wojtaszek M, Zgórka G (1999) High-performance liquid chromatography and thin-layer chromatography of phenolic acids from Ginkgo biloba L. leaves collected within vegetative period. J Liq Chrom Rel Tech 22:1457–1471
Lin Y, Li J, Li B, He T, Chun Z (2011) Effects of light quality on growth and development of protocorm-like bodies of Dendrobium officinale in vitro. Plant Cell Tiss Organ Cult 105:329–335
Linsmaier EM, Skoog F (1965) Organic growth factor requirements of tobacco tissue cultures. Physiol Plant 18:100–127
Mengxi L, Zhigang X, Yang Y, Yijie F (2011) Effects of different spectral lights on Oncidium PLBs induction, proliferation, and plant regeneration. Plant Cell Tiss Organ Cult 106:1–10
Nakamura Y, Torikai K, Ohigashi H (2001) A catechol antioxidant protocatechuic acid potentiates inflammatory leukocyte-derived oxidative stress in mouse skin via a tyrosinase bioactivation pathway. Free Rad Biol Med 30:967–978
Orlita A, Sidwa-Gorycka M, Kumirska J, Maliński E, Siedlecka EM, Gajdus J, Łojkowska E, Stepnowski P (2008a) Identification of Ruta graveolens L. metabolites accumulated in the presence of abiotic elicitors. Biotechnol Prog 24:128–133
Orlita A, Sidwa-Gorycka M, Paszkiewicz M, Malinski E, Kumirska J, Siedlecka EM, Łojkowska E, Stepnowski P (2008b) Application of chitin and chitosan as elicitors of coumarins and fluoroquinolone alkaloids in Ruta graveolens L. (common rue). Biotechnol Appl Biochem 51:91–96
Piekoszewska A, Baczyńska S, Ekiert H (2008) Free phenolic acids in stationary liquid culture of Ruta graveolens ssp. divaricata (Tenore) Gams. Acta Biochim Polon 55:57
Ramawat KG, Mathur M (2007) Factors affecting the production of secondary metabolites. In: Ramawat KG, Merillon JM (eds) Biotechnology: secondary metabolites. Plants and microbes. Science Publishers, Plymouth, pp 59–102
Rodríguez-Sahagún A, Acevedo-Hernández G, Rodríguez-Domínguez JM, Rodríguez-Garay B, Cervantes-Martínez J, Castellanos-Hernández OA (2011) Effect of light quality and culture medium on somatic embryogenesis of Agave tequilana Weber var. Azul. Plant Cell Tiss Organ Cult 104:271–275
Sivanesan I, Lim MY, Jeong BR (2011) Somatic embryogenesis and plant regeneration from leaf and petiole explants of Campanula punctata Lam. var. rubriflora Makino. Plant Cell Tiss Organ Cult 107:365–369
Sroka Z, Cisowski W (2003) Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem Toxicol 41:753–758
Zhang WJ, Su J, Tan MY, Liu GL, Pang YJ, Shen HG, Qi JL, Yang Y (2010) Expression analysis of shikonin-biosynthetic genes in response to M9 medium and light in Lithospermum erythrorhizon cell cultures. Plant Cell Tiss Organ Cult 101:135–142
Zhao SZ, Sun HZ, Chen M, Wang BS (2010) Light-regulated betacyanin accumulation in euhalophyte Suaeda salsa calli. Plant Cell Tiss Organ Cult 102:99–107
Acknowledgments
The in vitro culture of Ruta graveolens ssp. divaricata was obtained in cooperation with the Institute of Biosciences, Würzburg University (Germany)—Prof. F.Ch. Czygan, Dr. A. Abou-Mandour. The effect of wavelength was investigated in a biotechnological laboratory of the Department of Ornamental Plants, University of Agriculture in Kraków, and we would like to express our gratitude to the Head of this Department, Prof. Anna Bach. The authors are also grateful to Dr. R. Wróbel for translating this article into English and Dr. M. Paszt for proffessional English correction.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated memory to Professor Dr. Franz-Christian Czygan, Head of Department of Pharmaceutical Biology, Würzburg University, Honorary Doctor of I. Kant University, Kaliningrad, dead on 16th January 2012.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Szopa, A., Ekiert, H., Szewczyk, A. et al. Production of bioactive phenolic acids and furanocoumarins in in vitro cultures of Ruta graveolens L. and Ruta graveolens ssp. divaricata (Tenore) Gams. under different light conditions. Plant Cell Tiss Organ Cult 110, 329–336 (2012). https://doi.org/10.1007/s11240-012-0154-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-012-0154-5