Abstract
The number of deaths caused by multidrug-resistant Pseudomonas aeruginosa has risen in the recent decade. The development of quorum sensing inhibition (QSI) is a promising approach for controlling Pseudomonas infection. Therefore, this study mainly aimed to investigate how a plant-source material inhibits QSI to produce an antipathogenic effect for fighting microbial infections. The QSI effect of Trigonella stellata was assessed by using Chromobacterium violaceum ATCC 12472 reporter strain. Trigonella stellata exhibited high QSI activity, and an ethanolic extract of T. stellata was prepared for phytochemical isolation of the most active QSI compound. Nine pure compounds were isolated and identified as kaempferitrin (1), soyasaponin I (2), β-sitosterol-3-O-glucoside (3), dihydromelilotoside (4), astrasikokioside I (5), methyl dihydromelilotoside (6), (3R, 4S)-4, 2′, 4′-trihydroxy-7-methoxy-4′-O-β-d-glucopyranosylisoflavan (7), (3S, 4R)-4, 2′, 4′-trihydroxy-7-methoxyisoflavan (8, TMF), and (+)-d-pinitol (9). These compounds were screened against C. violaceum ATCC 12472, and TMF exhibited a potent QSI. The effect of TMF at sub-minimum inhibitory concentrations (MICs) was assessed against P. aeruginosa virulence factors, including biofilm, pyocyanin formation protease and hemolysin activity. TMF induced significant elimination of QS-associated virulence behavior. In addition, TMF at sub-MICs significantly reduced the relative expression of lasI, lasR, rhlI, and rhlR compared with that in untreated cells. Furthermore, molecular docking was performed to predict structural basis of the QSI activity of TMF. The study demonstrated the importance of T. stellata as a signal modulator and inhibitor of P. aeruginosa pathogenesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pseudomonas aeruginosa is an opportunistic human pathogen that can be isolated from various ecosystems, including water and soil (Baltch and Smith 1994). Pseudomonas aeruginosa is prevalent in people who have cystic fibrosis, serious burns, or deep-wound infections, as well as those who have urinary tract infections (Turkina and Vikström 2019), and is a problematic pathogen due to the development of multiple drug resistance that extends to many antimicrobial agents. In addition, P. aeruginosa can form a biofilm that acts as a protective shield containing extra polysaccharide layers that prevent the penetration of antimicrobial agents. These factors create an urgent need for the development of novel approaches to manage Pseudomonas infection (Tolker‐Nielsen 2014).
Pseudomonas aeruginosa exhibits various virulence behaviors, including extracellular molecules such as rhamnolipids, lectins, lipase and aminopeptidase that facilitate microbial spreading. Furthermore, P. aeruginosa exhibits a pronounced ability to colonize hosts via adhesive microbial attachment and biofilm assembly. The majority of these virulence behaviors are regulated by quorum sensing (QS; Jimenez et al. 2012; Maraolo et al. 2020). QS is a cell-to-cell communication process mediated by the production of small molecules called autoinducers (AIs). These AIs function as infochemicals enabling bacterial cells to act as a group and facilitating crosstalk between two distinct bacterial species that share the same environment. Currently, four basic QS circuits in P. aeruginosa have been identified: LasI/LasR and RhlI/RhlR (Pesci et al. 1997), Pseudomonas quinolone signal (PQS) system (Dubern and Diggle 2008), and integrated QS system (Rampioni et al. 2016). On reaching a threshold level, these chemicals form a network that is interconnected and allows hierarchical control of each other’s activities and thereby controls the expression of genes linked to virulence factors (Passador et al. 1993).
QS inhibition (QSI) is one of the most promising approaches for reducing the pathogenicity of Pseudomonas infection via various mechanisms (Rémy et al. 2018; Naga et al. 2021). Plants are a potent source of therapeutic compounds and have attracted attention for as potential sources of compounds that could interfere with the QS signaling systems of P. aeruginosa and reduce associated pathogenesis. Natural products have long been thought of as a source of essential antibacterial agents that could be used to treat various pathogenic infections (Howes et al. 2020). For example, ascorbic acid is a purely natural compound that has been found to be a QSI (El‐Mowafy et al. 2014). In addition, QSIs have been identified from extracts of medicinal plants, including Adhatoda vasica Nees, Myoporum laetum (Zaki et al. 2013), Citrus sinensis, Laurus nobilis, Elettaria cardamomum, Allium cepa, and Coriandrum sativum (Al-Haidari et al. 2016).
Trigonella stellata is a palatable and nutraceutical herb that possesses anti-inflammatory activity, antioxidant and anticancer effects (Sindhu et al. 2012; Sheweita et al. 2020) and has been used as a traditional treatment for diarrhea and dysentery. The methanol extract of Trigonella foenum-graceum (fenugreek) seeds inhibited QS and biofilm formation in Aeromonas hydrophila and P. aeruginosa with activity associated with caffeine production by the plant (Husain et al. 2015). However, the antivirulence and the QSI activity of T. stellata have not yet been assessed. For instance, T. stellata could be a promising source for active derivatives that have potential QSI efficacy and antivirulence attributes against P. aeruginosa.
Therefore, this study aimed to evaluate the QSI effect of T. stellata total extract followed by purification and identification of active compounds. We evaluated the anti-QS activity of the pure compounds against P. aeruginosa clinical isolates and then assessed the phenotypic and genotypic analysis of QSI and potential for inhibition of virulence traits. Finally, we performed a modeling study to identify the probable mechanism of QSI in respect to an identified active component.
Materials and methods
Plant material collection and extract preparation
Trigonella stellata aerial parts were gathered from Daba, Matrouh, Egypt, in April 2013. The plant was identified by the Botany Department, Faculty of Sciences, Mansoura University. A voucher specimen with the code “TsT-4-13” was washed in tap water directly after collecting, air-dried for 6 weeks, and grounded using a miller to fine powder. Then, 50 g of dried powder was extracted using 70% ethanol and incubated overnight at 30 °C with shaking at 200 rpm. Plant extracts were filtrated and concentrated using a rotary evaporator at 40 °C.
Bacterial strains and growth media
The QSI activity was assessed using the standard strain C. violaceum ATCC 12472 (McClean et al. 1997), which was grown for 24 h at 28 °C on Luria–Bertani (LB) media (0.5% w/v yeast extract, 1% w/v tryptone, and 1% w/v NaCl, pH 7) and 2% w/v agar (Bertani 2004). Pseudomonas aeruginosa clinical strains were collected from urine samples and termed Ps.A11, Ps.A12, Ps.A13, and Ps.A16. Collected samples were handled in accordance with the ethical guidelines of the Faculty of Medicine, Alexandria University, Egypt, and were identified as P. aeruginosa according to biochemical standards (Koneman et al. 2006) and the Vitik2 (bioMerieux, Inc., UAS) automated identification method. In addition, P. aeruginosa PA14 and PAO1 were used as the positive standard strains (Maura and Rahme 2017), while P. aeruginosa PAO-JP2 was used as a negative standard strain (Pearson et al. 1997). Standards and clinical P. aeruginosa strains were grown in LB medium and incubated overnight at 37 °C.
QSI assay of crude plant extract
Pure colonies of C. violaceum ATCC 12472 were inoculated in LB broth medium and cultivated at 28 °C with shaking at 200 rpm. Sterile LB agar molten at 55 °C was prepared and 15 mL was poured in the plates and left to solidify; the soft LB agar (5 mL) was then inoculated with 100 µL C. violaceum ATCC 12472 culture, and plates were allowed to completely solidify before wells were cut (10-mm diameter) in the agar using a cork borer. An aliquot of the concentrated T. stellata extract (100 μL) was added to the agar wells. After 24-h incubation at 28 °C, the extent of violet color around the well was evaluated and inhibition of this was determined as a positive QSI activity. Solvent was used as a negative control (McClean et al. 1997).
Isolation of the active compounds of T. stellata
A total of 470 g of air-dried and crushed aerial portions of T. stellata was extracted with 3 L of ethanol till exhaustion for 48 h at room temperature. The ethanolic extract was collected and dried at 45 °C under vacuum to yield a dry total extract with a final weight of 15 g. The entire dried extract was then disseminated in 250 mL water using sonication and then fractionated with hexane, ethyl acetate, methylene chloride, and butanol. The hexane, methylene chloride, ethyl acetate, and butanol fractions were dried to obtain weights of 3.9, 1.1, 1.53, and 2.5 g, respectively.
The ethyl acetate fraction (1.53 g) was spotted onto a 2.5 × 95 cm silica gel column and eluted with a mobile phase consisting of ethyl acetate:CHCl3:CH3OH:H2O at ratios of 15:8:4:1, 10:6:4:1, and 6:6:4:1; eluates were then examined using thin-layer chromatography (TLC). The subfractions of comparable spots were combined to yield eight fractions (TE1 to TE8). Fraction TE2 (185 mg) was chromatographed over RP-18 column and isocratically eluted with CH3OH:H2O (1:1) to provide two pure compounds: 6 and 7 (13.2 and 9.7 mg, respectively).
The methylene chloride extract (1.1 g) was purified using vacuum liquid chromatography (VLC) over RP-18 silica (20 × 2 cm) and eluted with two gradients of CH3OH:H2O (1:1 then 6:4) to obtain 11 fractions (TC1 to TC11). Fraction TC5 (118 mg) was applied to a standard silica gel column (2 × 95 cm) and eluted with ethyl acetate:CHCl3 (1:1) followed by ethyl acetate:CHCl3:CH3OH:H2O (10:6:4:1, then 6:4:4:1) to provide the purified compound 8 (10 mg). Fraction TC9 (529 mg) was chromatographed over a silica gel glass column (1.5 × 90 cm) and eluted with the gradients of ethyl acetate:CHCl3:CH3OH:H2O (15:8:4:1, 10:6:4:1, and 6:4:4:1) to yield compound 3 (23 mg).
The butanol extract (2.5 g) was passed over a VLC packed with RP-18 silica (25 × 4 cm) and eluted with the following mobile phase compositions H2O:CH3OH (1:0, 9:1, 7:3, 6:4, 5:5, 4:6, 3:7, 2:8, and 0:1), 1 L each, to yield 11 fractions (TB1 to TB11). Compound 9 (31 mg) was precipitated from the methanol-concentrated fraction TB4 (179 mg). The purification of two compounds 1 (8.1 mg) and 4 (9.8 mg) was achieved through column chromatography of fraction TB2 (108 mg) over RP-18. A white powder of purified compound 2 fraction TB6 (182 mg) was applied to an RP-18 column (2 × 30 cm) and eluted with gradients of H2O:CH3OH (1:1, 6:4, and 4:6). Compound 5 (6.2 mg) was obtained through the RP-18 column fractionation of TB7 (110 mg) and isocratic elution with H2O:CH3OH (6:4).
Characterization of isolated compounds by NMR spectroscopy
One- and two-dimensional (1D and 2D) NMR experiments were performed using a Varian 400 MHz, Bruker DRX-500, or Bruker Avance III 600 and 400 MHz spectrometer with deuterated solvents of pyridine, methanol, or dimethylsulfoxide (DMSO); the solvent signals served as an internal reference for adjustment. The Agilent Technologies 6200 series was used to record the positive and negative ion modes of compound masses. An AUTOPOL IV Automatic Polarimeter (Rudolph, Hackettstown, NJ, USA) was used to detect the specific rotations of the pure compounds. In addition, the Varian Cary 50 Bio UV–Vis spectrophotometer was used to measure the UV spectra. The silica gel (particles diameter 32–63 µm, Dynamic Adsorbents, Inc.) and RP-18 (Polar bond, J. T. Baker) were used for the preparation of column chromatography. TLC plates were monitored with silica gel F254 sheet (Fluka) or RP-18 (Merck). The spots of the tested compounds were visualized at 254 nm (UV) and then sprayed with vanillin (1% w/v) (Sigma) prepared in concentrated H2SO4:C2H5OH (10:90). All the chromatographic procedures were performed using analytical grade solvents (Fischer Chemical).
Assay of QSI activity of T. stellata isolated compounds with C. violaceum ATCC 12472
QSI activity of the nine isolated compounds from T. stellata was assessed using C. violaceum ATCC 12472. Compounds were dissolved to 5 mg/mL in DMSO. Aliquots of 50 μL of each compound were applied in each well (10 mm diameter), and then the plates were then incubated at 28 °C. After 24 h, the inhibition of induced violacein production around the wells was examined. DMSO was used as a control. Compounds possessing potential QSI with inhibition of violacein pigment production were estimated for their effect on virulence factors.
Antibacterial screening
The minimum inhibitory concentration (MIC) of the active compound TMF was calculated using the microtiter plate assay technique. Each well received 100 µL Mueller Hinton broth, and the TMF compound was serially diluted in twofold serial concentrations (512, 256, 128, 64, 32, 16, 8, 4, 1, and 0.5 µg/mL) and inoculated with 1 × 106 CFU/well of actively growing P. aeruginosa strains (Patel et al. 2015). The negative control consisted of wells containing media, whereas the positive control included wells containing media inoculated with the test strains. The MIC was defined as the lowest concentration of TMF compound that inhibited growth.
Effect of sub-MIC of TMF on the growth of P. aeruginosa strains
The viability of P. aeruginosa was investigated using the pour plate method after adding 1/2 MIC of TMF and comparing with that of the untreated culture. A volume of the overnight culture (1 mL) was collected and diluted 1:10. Diluted cultures were subcultured in molten LB agar, and the colonies were counted after overnight incubation at 37 °C. Pseudomonas aeruginosa growth curves (treated and nontreated) at an optical density (OD) at 600 nm were estimated at different time intervals.
Antivirulence effect of TMF on P. aeruginosa
Different virulence factors of P. aeruginosa strains were quantified after treatment by 1/2 and 1/4 MICs of TMF in triplicates. Assessment of virulence factors was performed with and without TMF (El-Mowafy et al. 2017). Similarly, control strains PAO1 and PA14 (positive controls) and PAO-JP2 (negative control) were measured under the same conditions (Musthafa et al. 2012).
Biofilm formation assay
Pseudomonas aeruginosa biofilm formation was assessed using 96-well flat-bottomed polystyrene plates. Aliquots of the treated and untreated cultures (100 μL) were distributed into the plates, which were then incubated at 37 °C for 24 h. Wells were washed three times with 200 μL of physiological saline before the attached bacteria were fixed for 15 min with 150 μL of absolute methanol. After emptying and drying the plates, the bacterial cells were stained with 150 μL of 1% (w/v) crystal violet. The excess stain was washed away, and the dye was dissolved in glacial acetic acid at 33% (v/v). The absorbance of the dyed cells was determined at 490 nm (Adonizio et al. 2008).
Pyocyanin assay
Pyocyanin was quantified using King A broth media (peptone 2% (w/v), K2SO4 1.0% (w/v), and MgCl2 0.14% (w/v)) (Essar et al. 1990). A volume of 500 μL of P. aeruginosa overnight culture was added to 5 mL of King A broth (treated and untreated media) and incubated with agitation at 200 rpm at 37 °C. The cultures were then centrifuged at 3000 rpm for 10 min at 4 °C to remove the cells. Pyocyanin was extracted with 3 mL of chloroform and vortexed until the color changed to greenish blue. The mixture was centrifuged at 3000 rpm for 10 min; 1 mL of 0.2 M HCl was added, and the mixture was shaken until the blue color turned pink. The absorbance of the pink layer was measured at 520 nm, and the concentration of pyocyanin in µg/mL was estimated using the formula: absorbance: absorbance × 17.072 (Essar et al. 1990).
Determination of hemolysin activity
The hemolytic activity of P. aeruginosa untreated and treated (1/2 and 1/4 MICs of TMF) culture-free supernatant was determined. In brief, 500 μL of supernatant was mixed with 700 μL of washed erythrocytes and incubated for 2 h at 37 °C. The reaction mixture was centrifuged at 8000 × g for 15 min at 4 °C (Rossignol et al. 2008), and the absorbance was measured at 540 nm (Annapoorani et al. 2012).
Total protease production
The proteolytic activity of all P. aeruginosa strains (treated and untreated) was estimated using the skimmed milk assay technique. The assay was performed by adding 1 mL of 1.25% (w/v) skimmed milk to 500 μL of culture supernatants and incubating for 1 h at 37 °C. The OD was measured at 600 nm and compared with that of the blank (skimmed milk). The reduction in the OD600 of the treated cells was compared with that of the untreated cells under the same conditions (Skindersoe et al. 2008).
Real-time PCR
The effect of TMF on the expression of QS regulatory genes lasI/R and rhlI/R in P. aeruginosa PAO1 was measured using RT-PCR. The control untreated PAO1 and cultures treated with 1/2 MIC of TMF were grown until an OD 600 nm of 0.4–0.5. Cells were collected via centrifugation at 6000 × g for 15 min, and RNA was extracted using TRIZOL reagent (Oxoid, Basingstoke, Hants, UK), according to the manufacturer’s instructions. The SensiFAST™ cDNA Synthesis Kit (Bioline Reagents Ltd., London, UK) was used to synthesize complementary DNA. The Rotor-Gene Q thermocycler (Qiagen, Valencia, CA, USA) was used to perform RT-PCR. The primers described in Table 1 were used in the amplification reaction with TOPrealTM qPCR 2 × PreMIX (SYBR Green with low ROX) (Enzynomics; Daejeon, Korea). The expression of each gene was normalized to that of the rpoD housekeeping gene, and the relative expression was determined using the formula 2−ΔΔCT (Livak and Schmittgen 2001). The expression of genes in PAO1 cultures treated with TMF was compared with that in control cultures without treatment.
Molecular docking
TMF was docked into the active site of P. aeruginosa LasR ligand-binding domain (Protein Data Bank ID: 2UV0) to evaluate the binding mechanism with LasR; this structure used 3-oxo-C12-HSL as an AI (Bottomley et al. 2007). All protein-bound water ligands were eliminated. ChemBioDraw was used to generate all components, which were then imported into the ChemBioOffice ultra v.14 programs. MM2, Jop type was used to minimize the energy, and docking was conducted using Molsoft (Abagyan et al. 1994).
Data analysis and statistics
Experiments were performed in triplicate, and Excel was used to determine the means, standard deviations, and standard errors. Statistical analysis was calculated using Welch’s t-test. A significant difference between treated and untreated cultures was considered when the probability value was ***p ≤ 0.001, **p ≤ 0.01, or *p ≤ 0.05.
Results
Screening for QSI activity of T. stellata
The T. stellata ethanolic extract showed a potent QSI activity against C. violaceum ATCC 12472 after 24 h at 28 °C as shown by the inhibition of violacein pigment production to a diameter of 20 mm.
Fractionation and structure elucidation of isolated compounds in T. stellata
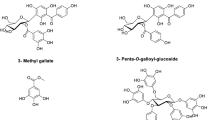
The phytochemical analysis of T. stellata extracts in methylene chloride, ethyl acetate, and butanol resulted in isolation and identification of nine known compounds. Their structures were determined by analyzing 1D and 2D NMR spectra and mass spectrometry and by comparing their spectroscopic data with that published data (Ngoc et al., 2012; Shams Eldin et al., 2018).
The structures of isolated compounds (Fig. 1) were identified as kaempferitrin (1) (Figs. S1–S4) (Pereira et al. 2011), soyasaponin I (2) (Figs. S5–S11), β-sitosterol-3-O-glucoside (3) (Khan and Hossain 2015), dihydromelilotoside (4) (Figs. S12–S17), astrasikokioside I (5) (Figs. S18–S23) (Yahara et al. 2000), methyl dihydromelilotoside (6) (Figs. S24–S28), (3R, 4S)-4, 2′, 4′-trihydroxy-7-methoxy-4′-O-β-d-glucopyranosylisoflavan (7) (Figs. S29–S35), (3S, 4R)-4, 2′, 4′-trihydroxy-7-methoxyisoflavan (Figs. S36–S43), and (+)-d-pinitol (9) (Figs. S4–S47) (Ganbaatar et al. 2016). Interestingly, compounds 5, 6, and 9 have not been previously described from T. stellate.
Structures of nine isolated compounds from T. stellata, kaempferitrin (1), soyasaponin I (2), β-sitosterol-3-O-glucoside (3), dihydromelilotoside (4), astrasikokioside I (5), methyl dihydromelilotoside (6), (3R,4S)-4,2`,4`-trihydroxy-7-methoxy-4`-O-β-D-glucopyranosylisoflavan (7), (3S,4R)-4,2`,4`-trihydroxy-7-methoxyisoflavan (8, TMF), and (+)-D-pinitol (9)
Chromobacterium violaceum ATCC 12472 bioassay of QSI activity of compounds isolated from T. stellata
The QSI activity of nine compounds isolated from T. stellata was screened using C. violaceum ATCC 12472. Compound 8, TMF, exhibited potent anti-QS activity with an inhibition in the diameter of the violet pigmentation by 15 mm. Compounds 1, 3, 5, 6, and 9 showed weak QSI effect with inhibition of the violet pigment diameter by 2, 5, 5, 3, and 2 mm, respectively. Therefore, fraction 8 was chosen for further investigations of antipathogenic potential against P. aeruginosa standard and clinical strains (Table 2).
Antibacterial screening
The MIC of TMF against P. aeruginosa strains Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, PAO1, and PAO-JP2 was 512 μg/mL. Sub-MICs at 1/2 and 1/4 strength were calculated as 256 and 128 μg/mL, respectively (Table 3).
Effect of the sub-MIC of TMF on P. aeruginosa viability
The antimicrobial effect of TMF on P. aeruginosa viability was determined before and after supplying growing cells with 1/2 MIC (256 μg/mL) for 24 h at 37 °C. The bacterial count from untreated cultures was 112, 94, 126, 113, 98, 133, and 103 × 107 CFU/mL for Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, PAO1, and PAO-JP2, respectively. The count from the treated cultures was 108, 95, 121, 107, 96, 129, and 100 × 108 CFU/mL for Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, PAO1, and PAO-JP2, respectively. There was no significant difference between the bacterial count between untreated and treated cultures. Furthermore, no effect on bacterial growth was observed in cultures treated with 1/2 MIC of TMF in comparison with that in untreated cultures (Fig. S48).
Antivirulence effect of TMF on P. aeruginosa strains
Treatment with both the sub-MICs of TMF compound (1/2 and 1/4) significantly inhibited formation of biofilm and pyocyanin and activity of hemolysin and protease in Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, and PAO1 as compared with that in untreated cultures. PAO-JP2 had the lowest activity among the tested strains for all virulence factors.
Inhibition of biofilm formation
A sub-MIC of TMF (256 μg/mL) significantly reduced biofilm formation in Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, and PAO1 by 76.5%, 75.3%, 79.6%, 76.3%, 81%, and 81.8%, respectively. Furthermore, 1/4 MIC significantly decreased biofilm by 77.2%, 73.7%, 79.6%, 74.9%, 80%, and 77.3% in Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, and PAO1, respectively (Fig. 2; Table S1).
Effect of 1/4 and 1/2 sub-MICs of (3S,4R)-4,2`,4`-trihydroxy-7-methoxyisoflavan (TMF) isolated from T. stellata on biofilm formation by P. aeruginosa strains; Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, PAO1, and PAO-JP2 compared to the untreated cultures. Error bars show standard deviation of three replicates, ***p ≤0.001, **p ≤ 0.01
Pyocyanin inhibition
Low concentrations of TMF at sub-MICs (1/2 and 1/4) significantly inhibited pyocyanin production in all tested P. aeruginosa strains. TMF at 1/2 MIC decreased pyocyanin by 78.6%, 69.6%, 71.7%, 70.7%, 61%, and 63.2%, respectively, in all tested strains (Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, and PAO1) (p ≤ 0.001), also the 1/4 MIC of TMF significantly decreased pyocyanin by 77.7%, 68.3%, 73.4%, 65.5%, 57.7%, and 61.8%, respectively, in these strains as well (p ≤ 0.001, Fig. 3; Table S2).
Effect of 1/4 and 1/2 sub-MICs of (3S,4R)-4,2`,4`-trihydroxy-7-methoxyisoflavan (TMF) on pyocyanin production by P. aeruginosa strains; Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, PAO1, and PAO-JP2 compared to the untreated cultures. Error bars show standard deviation of three replicates, ***p ≤ 0.001, **p ≤ 0.01
Decrease in hemolysin activity
TMF at sub-MICs significantly inhibited hemolysin activity (p ≤ 0.001) in isolates Ps.A11, Ps.A12, Ps.A13, Ps.A16, PAO1, and PA14. The 1/2 MIC decreased hemolysin production by Ps.A11, Ps.A12, Ps.A13, Ps.A16, PAO1, and PA14 by 53.8%, 53.8%, 40.8%, 52.1%, 61.8%, and 56%, respectively, whereas the 1/4 MIC (128 μg/mL) lowered hemolysin activity by 48.9%, 52.2%, 41.2%, 47.4%, 56.1%, and 55%, respectively (Fig. 4; Table S3).
Effect of 1/4 and 1/2 sub-MICs of (3S,4R)-4,2`,4`-trihydroxy-7-methoxyisoflavan (TMF) isolated from T. stellata on hemolysin activity by P. aeruginosa strains; Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, PAO1, and PAO-JP2 compared to the untreated cultures. Error bars show standard deviation of three replicates, **p ≤ 0.01, *p ≤ 0.05
Decrease in protease activity
Protease production was significantly reduced at 1/2 MIC of TMF by 40.2%, 39.7%, 35.4%, 33.7%, 36.1%, and 43.2% in Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, and PAO1, respectively, compared with that in untreated cultures. The 1/4 MIC significantly reduced protease activity by 42.1%, 38%, 34.4%, 30.6%, 28%, and 41% in Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, and PAO1, respectively (Fig. 5; Table S4).
Effect of 1/4 and 1/2 sub-MICs of (3S,4R)-4,2`,4`-trihydroxy-7-methoxyisoflavan (TMF) on protease production by P. aeruginosa strains; Ps.A11, Ps.A12, Ps.A13, Ps.A16, PA14, PAO1, and PAO-JP2 compared to the untreated cultures. Error bars show standard deviation of three replicates, ***p ≤ 0.001, **p ≤ 0.01
Suppression of the QS regulatory genes
We investigated the effect of TMF at 1/2 MIC on gene expression involved in the QS circuit of P. aeruginosa. RT-PCR was used to assess the relative expression levels of lasI, lasR, rhlI, and rhlR genes. TMF significantly (p < 0.01) lowered the mRNA level of lasI and lasR by 64.5% and 34%, respectively, in the standard strain PAO1 compared with that in the untreated cultures (Fig. 6).
Moreover, the relative expression of rhlI and rhlR genes in P. aeruginosa PAO1 treated with 1/2 MIC of TMF were significantly reduced compared with that in the untreated cells. The relative expression of rhlI and rhlR mRNA were significantly reduced by 57% and 56.9%, respectively (p < 0.01) compared with that in the untreated PAO1 (Fig. 6).
Binding affinity analysis for LasR and ligands by molecular docking
The LasR ligand, 3-oxo-C12-HSL, was redocked with LasR to assess the docking technique. This showed that the ICM score was − 107.47 and that three H-bonds were formed with Asp73, Trp60, and Ser129. Thus, these three amino acids are crucial for binding with the LasR active site. Docking of TMF with the LasR had an ICM score of − 74.35 (Table 4).
TMF formed six H-bonds, one each with both Leu110 and Ser129 and two each with Thr75 and Asp73 (Table 5; Fig.
7).
Discussion
QS is the key regulatory mechanism that manages bacterial pathogenesis via gene expression and virulence behavior. Treatment of P. aeruginosa infection is challenging due to the ability of this bacterium to form a biofilm that is resistant to conventional antibiotics (Pérez-Pérez et al. 2017). The antimicrobial resistance of Pseudomonas is also associated with the production of degradative enzymes that can inactivate antimicrobials or the production of debasing chemicals and efflux pumps from chromosomally encoded antibiotic-resistant genes (Du et al. 2013). Accordingly, developing new strategies to manage Pseudomonas infection is essential. One approach is to focus on inhibiting QS crosstalk as this could critically inhibit virulent effects. Violacein production in C. violaceum ATCC 12472 is controlled by N-acyl-l-homoserine lactones mainly N-(3-hydroxydecanoyl)-l-homoserine lactone (AHL). In addition, other signaling molecules, such as 3-oxo-C10-HSL and 3-oxo-C12-HSL, can enhance violacein production in this bacterium (Morohoshi et al. 2008, 2010; Mion et al. 2021). In previous research, C. violaceum ATCC 12472 has been used to assay violacein inhibition by glyceryl trinitrate (Abbas and Shaldam 2016), silver nanoparticles (Shah et al. 2019), and Spirulina platensis (Lewis Oscar et al. 2018).
Trigonella stellata belongs to the genus Trigonella, which can be found in dry places across the Mediterranean (Turki et al. 2013). In China, Egypt, and India, Trigonella has a long history of traditional use as a medicine (Saxena and Albert 2005). Trigonella seeds have been traditionally used as a laxative and antipyretic (Yoshikawa et al. 1998) and have been used to treat gastrointestinal diseases, hyperlipidemia, inflammation, and diabetes for centuries (Shah et al. 2006). Trigonella can induce diverse pharmacological effects that are primarily due to the presence of diosgenin, flavonoids, trigonelline, coumarin, furostanol, and flavonol glycosides (Sheweita et al. 2020). In this study, we validated T. stellata for anti-QS activities. Extracts from T. stellata were highly potent in inhibiting violet pigment formation, which indicated possible interference with AHL-based signaling (Table 2). The bioassay of QSI activity of the nine purified compounds from T. stellata revealed that the highest QSI was mediated by the TMF compound. Therefore, TMF was assessed for its ability to inhibit QS-related virulence factors.
Pseudomonas aeruginosa causes nosocomial infections that are typically associated with the formation of a biofilm that is resistant to most antimicrobials (Pérez-Pérez et al. 2017). Herein, TMF could significantly reduce biofilm formation with all pseudomonad strains tested (Fig. 2), which would consequently make the bacteria more vulnerable to the response of the immune system (Rasmussen et al. 2005). TMF compound is considered a flavonoid derivative. The antibiofilm effects of flavonoids, such as apigenin, luteolin, quercetin, fisetin, and chrysin, on Staphylococcus aureus have also been reported (Cho et al. 2015).
The RhlI/R and PQS signaling systems of P. aeruginosa also regulate the production of a distinct green pigment called pyocyanin (Gupta et al. 2011). The absence of this green pigment indicates lower regulatory control of pyocyanin production (Fig. 3). Treatment of all tested P. aeruginosa strains with TMF at low concentrations (128–256 μg/mL) resulted in a significant reduction in pyocyanin levels, indicating inhibition of the C4-HSL and pqsR signaling molecules of the RhlI/R and PQS signaling systems, respectively.
Pseudomonas aeruginosa produces extracellular enzymes such as protease and hemolysin that allow the bacteria to spread inside host tissues and to resist host immunity (Gupta et al. 2011). We found that TMF significantly suppressed the activity of these factors, which are regulated by the LasI/LasR system, to varying degrees (Figs. 4, 5). Similarly, the flavonoid fraction of Psidium guajava leaves extract was shown to decrease P. aeruginosa PAO1 virulence factors (Vasavi et al. 2014). Flavonoids, such as taxifolin, naringenin, catechin, and flavanes-3-ol, have been shown to have a significant effect on the Las system in P. aeruginosa PAO1 (Vandeputte et al., 2011; Rasamiravaka et al., 2013; Bouyahya et al., 2017), and flavonoids from Centella asiatica plant have also been shown to reduce the proteolytic activities of this strain (Vasavi et al., 2016).
In P. aeruginosa, LasI/R act as transcriptional activators that manipulate the expression of QS cascades while RhlI/R and RqsR regulate the expression of the associated virulence factors [80, 81]. In this study, the relative mRNA expression of lasI/lasR and rhlI/rhlR was significantly reduced by treatment with TMF at sub-MIC (Fig. 6). The low levels of these factors were associated with QS interruption, confirming the influence of QSI on production of virulence factors of the tested strains.
The molecular docking was performed to evaluate the structural basis of QSI activity of TMF compound (Abagyan et al. 1994). In P. aeruginosa, the Las system is the main QS regulator and is activated by the 3-oxo-C12-HSL signaling molecule, which controls the QS network (Pesci et al. 1997; Venturi 2006). We retrieved the structure of LasR receptor protein (PDB ID 2UV0) (https://www.rcsb.org/structure/2UV0). The scoring functions and hydrogen bonds produced by the LasR active site with the surrounding amino acids could be used to determine the binding mode, affinity, and orientation of TMF (Fig. 7). Analysis using Pdbsum revealed that the important amino acids that bind with LasR active site are Cys79, Tyr56, Ser129, Ala50, Trp60, Thr75, Tyr93, Leu110, Asp73, Trp88, Ala105, Tyr64, and Gly126 (http://www.ebi.ac.uk/thornton-srv/databases/cgi-bin/pdbsum /GetPage.pl). In 2017, pyridoxal lactohydrazone was discovered to reduce QS-related virulence factors in P. aeruginosa and to establish hydrogen bonds with Thr75, Trp60, Ser129, and Arg61 in the active site of LasR (Heidari et al. 2017). Furthermore, fenaclon competitively binds to LasR via the formation of hydrogen bonds with Asp73, Ser129, and Tyr56 (Pattnaik et al. 2018). In this study, TMF formed six H-bonds, one each with Leu110 and Ser129 and two each with Thr75 and Asp73; these bonds would hinder the binding of the natural ligand 3-oxo-C12-HSL with its receptor and interrupt the signaling cascade (Table 5).
Conclusion
This study highlights the importance of the medicinal plant T. stellate and its active metabolite TMF as a potent QSI of P. aeruginosa without any effect on bacterial viability. This inhibitory effect could be attributed to the effect of TMF at sub-inhibitory concentrations on the relative expression of QS genes. Reduction in the relative expression of lasI/lasR and rhlI/rhlR was associated with a significant inhibition of virulence factors (biofilm formation, pyocyanin production, and hemolysin and protease activity). Furthermore, these in vitro results were confirmed using a molecular docking study that calculated the binding affinity of TMF with LasR. Overall, our study shows that TMF can combat the pathogenesis and dissemination of Pseudomonas infection through QS interference. Further studies need to be performed to evaluate the in vivo antipathogenic effects of TMF.
Data availability
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
References
Abagyan R, Totrov M, Kuznetsov D (1994) ICM—a new method for protein modeling and design: applications to docking and structure prediction from the distorted native conformation. J Comput Chem 15:488–506
Adonizio A, Kong K-F, Mathee K (2008) Inhibition of quorum sensing-controlled virulence factor production in Pseudomonas aeruginosa by South Florida plant extracts. Antimicrob Agents Chemother 52:198–203
Al-Haidari RA, Shaaban MI, Ibrahim SRM, Mohamed GA (2016) Anti-quorum sensing activity of some medicinal plants. Afr J Tradit Complement Altern Med 13:67–71
Annapoorani A, Parameswari R, Pandian SK, Ravi AV (2012) Methods to determine antipathogenic potential of phenolic and flavonoid compounds against urinary pathogen Serratia marcescens. J Microbiol Methods 91:208–211
Baltch AL, Smith RP (1994) Pseudomonas aeruginosa: infections and treatment. In: Infectious disease and therapy series. Marcel Dekker, New York
Bertani G (2004) Lysogeny at mid-twentieth century: P1, P2, and other experimental systems. J Bacteriol 186:595–600
Bottomley MJ, Muraglia E, Bazzo R, Carfì A (2007) Molecular insights into quorum sensing in the human pathogen Pseudomonas aeruginosa from the structure of the virulence regulator LasR bound to its autoinducer. J Biol Chem 282:13592–13600
Bouyahya A, Dakka N, Et-Touys A et al (2017) Medicinal plant products targeting quorum sensing for combating bacterial infections. Asian Pac J Trop Med 10:729–743
Cho HS, Lee J-H, Cho MH, Lee J (2015) Red wines and flavonoids diminish Staphylococcus aureus virulence with anti-biofilm and anti-hemolytic activities. Biofouling 31:1–11
Du D, Venter H, Pos KM, Luisi BF (2013) The machinery and mechanism of multidrug efflux in gram-negative bacteria. In: Microbial efflux pumps: current research. Caister Academic Press, Norfolk
Dubern J-F, Diggle SP (2008) Quorum sensing by 2-alkyl-4-quinolones in Pseudomonas aeruginosa and other bacterial species. Mol Biosyst 4:882–888
El-Mowafy SA, Shaaban MI, Abd El Galil KH (2014) Sodium ascorbate as a quorum sensing inhibitor of Pseudomonas aeruginosa. J Appl Microbiol 117:1388–1399
El-Mowafy SA, Abd El Galil KH, Habib E-SE, Shaaban MI (2017) Quorum sensing inhibitory activity of sub-inhibitory concentrations of β-lactams. Afr Health Sci 17:199–207
Essar DW, Eberly L, Hadero A, Crawford IP (1990) Identification and characterization of genes for a second anthranilate synthase in Pseudomonas aeruginosa: interchangeability of the two anthranilate synthases and evolutionary implications. J Bacteriol 172:884–900
Ganbaatar C, Gruner M, Tunsag J et al (2016) Chemical constituents isolated from Zygophyllum melongena Bunge growing in Mongolia. Nat Prod Res 30:1661–1664
Gupta RK, Setia S, Harjai K (2011) Expression of quorum sensing and virulence factors are interlinked in Pseudomonas aeruginosa: an in vitro approach. Am J Biomed Sci 3:116–125
Heidari A, Noshiranzadeh N, Haghi F, Bikas R (2017) Inhibition of quorum sensing related virulence factors of Pseudomonas aeruginosa by pyridoxal lactohydrazone. Microb Pathog 112:103–110
Howes MR, Quave CL, Collemare J et al (2020) Molecules from nature: reconciling biodiversity conservation and global healthcare imperatives for sustainable use of medicinal plants and fungi. Plants People Planet 2:463–481
Husain FM, et al (2015) Trigonella foenum-graceum (Seed) extract interferes with quorum sensing regulated traits and biofilm formation in the strains of Pseudomonas aeruginosa and Aeromonas hydrophila. Evidence-Based Complementary and Alternative Medicine
Jimenez PN, Koch G, Thompson JA et al (2012) The multiple signaling systems regulating virulence in Pseudomonas aeruginosa. Microbiol Mol Biol Rev 76:46–65
Khan N, Hossain MS (2015) Scopoletin and β-sitosterol glucoside from roots of Ipomoea digitata. J Pharmacogn Phytochem 4:5–7
Koneman E, Winn WJ, Allen S et al (2006) Chapter 6. In: Koneman’s color atlas and textbook of diagnostic microbiology. Lippincott Williams and Wilkins, New York
Lewis Oscar F, Nithya C, Alharbi SA, et al (2018) In vitro and in silico attenuation of quorum sensing mediated pathogenicity in Pseudomonas aeruginosa using Spirulina platensis. Microb Pathog 116:246–256
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Maraolo AE, Mazzitelli M, Trecarichi EM et al (2020) Ceftolozane/tazobactam for difficult-to-treat Pseudomonas aeruginosa infections: a systematic review of its efficacy and safety for off-label indications. Int J Antimicrob Agents 55:105891
Maura D, Rahme LG (2017) Pharmacological inhibition of the Pseudomonas aeruginosa MvfR quorum-sensing system interferes with biofilm formation and potentiates antibiotic-mediated biofilm disruption. Antimicrob Agents Chemother. https://doi.org/10.1128/AAC.01362-17
McClean KH, Winson MK, Fish L et al (1997) Quorum sensing and Chromobacterium violaceum: exploitation of violacein production and inhibition for the detection of N-acylhomoserine lactones. Microbiology 143:3703–3711
Mion S, Carriot N, Lopez J et al (2021) Disrupting quorum sensing alters social interactions in Chromobacterium violaceum. NPJ Biofilms Microbiomes 7:1–16
Morohoshi T, Nakazawa S, Ebata A et al (2008) Identification and characterization of N-acylhomoserine lactone-acylase from the fish intestinal Shewanella sp. strain MIB015. Biosci Biotechnol Biochem 72:1887–1893
Morohoshi T, Fukamachi K, Kato M et al (2010) Regulation of the violacein biosynthetic gene cluster by acylhomoserine lactone-mediated quorum sensing in Chromobacterium violaceum ATCC 12472. Biosci Biotechnol Biochem 74:2116–2119
Musthafa KS, Sivamaruthi BS, Pandian SK, Ravi AV (2012) Quorum sensing inhibition in Pseudomonas aeruginosa PAO1 by antagonistic compound phenylacetic acid. Curr Microbiol 65:475–480
Naga NG, El-Badan DE-S, Rateb HS et al (2021) Quorum sensing inhibiting activity of cefoperazone and its metallic derivatives on Pseudomonas aeruginosa. Front Cell Infect Microbiol. https://doi.org/10.3389/fcimb.2021.716789
Ngoc TM, Khoi NM, Nhiem NX et al (2012) Xanthine oxidase inhibitory activity of constituents of Cinnamomum cassia twigs. Bioorg Med Chem Lett 22:4625–4628
Passador L, Cook JM, Gambello MJ et al (1993) Expression of Pseudomonas aeruginosa virulence genes requires cell-to-cell communication. Science (80- ) 260:1127–1130
Patel JB, Cockerill FR, Bradford PA (2015) Performance standards for antimicrobial susceptibility testing: twenty-fifth informational supplement. CLSI, Wayne
Pattnaik SS, Ranganathan S, Ampasala DR et al (2018) Attenuation of quorum sensing regulated virulence and biofilm development in Pseudomonas aeruginosa PAO1 by Diaporthe phaseolorum SSP12. Microb Pathog 118:177–189
Pearson JP, Pesci EC, Iglewski BH (1997) Roles of Pseudomonas aeruginosa las and rhl quorum-sensing systems in control of elastase and rhamnolipid biosynthesis genes. J Bacteriol 179:5756–5767
Pereira DA, Dalmarco JB, Wisniewski A Jr et al (2011) Lotus corniculatus regulates the inflammation induced by bradykinin in a murine model of pleurisy. J Agric Food Chem 59:2291–2298
Pérez-Pérez M, Jorge P, Pérez Rodríguez G et al (2017) Quorum sensing inhibition in Pseudomonas aeruginosa biofilms: new insights through network mining. Biofouling 33:128–142
Pesci EC, Pearson JP, Seed PC, Iglewski BH (1997) Regulation of las and rhl quorum sensing in Pseudomonas aeruginosa. J Bacteriol 179:3127–3132
Rampioni G, Falcone M, Heeb S et al (2016) Unravelling the genome-wide contributions of specific 2-alkyl-4-quinolones and PqsE to quorum sensing in Pseudomonas aeruginosa. PLoS Pathog 12:e1006029
Rasamiravaka T, Jedrzejowski A, Kiendrebeogo M et al (2013) Endemic Malagasy Dalbergia species inhibit quorum sensing in Pseudomonas aeruginosa PAO1. Microbiology 159:924–938
Rasmussen TB, Bjarnsholt T, Skindersoe ME et al (2005) Screening for quorum-sensing inhibitors (QSI) by use of a novel genetic system, the QSI selector. J Bacteriol 187:1799–1814
Rémy B, Mion S, Plener L et al (2018) Interference in bacterial quorum sensing: a biopharmaceutical perspective. Front Pharmacol 9:203
Rossignol G, Merieau A, Guerillon J et al (2008) Involvement of a phospholipase C in the hemolytic activity of a clinical strain of Pseudomonas fluorescens. BMC Microbiol 8:1–14
Saxena VK, Albert S (2005) Quercetin-3-O-alpha-l-rhamnopyranoside from the seeds of Trigonella foenum-graecum. Proc Natl Acad Sci India A 75:287
Shah SN, Bodhankar SL, Bhonde R, Mohan V (2006) Hypoglycemic activity of the combination of active ingredients isolated from Trigonella foenum-graecum in alloxan induced diabetic mice. PharmacologyOnLine 1:65–82
Shah S, Gaikwad S, Nagar S, et al (2019) Biofilm inhibition and anti-quorum sensing activity of phytosynthesized silver nanoparticles against the nosocomial pathogen Pseudomonas aeruginosa. Biofouling 35:34–49
Shams Eldin SM, Radwan MM, Wanas AS et al (2018) Bioactivity-guided isolation of potential antidiabetic and antihyperlipidemic compounds from Trigonella stellata. J Nat Prod 81:1154–1161
Sheweita SA, ElHady SA, Hammoda HM (2020) Trigonella stellata reduced the deleterious effects of diabetes mellitus through alleviation of oxidative stress, antioxidant-and drug-metabolizing enzymes activities. J Ethnopharmacol 256:112821
Sindhu G, Ratheesh M, Shyni GL et al (2012) Anti-inflammatory and antioxidative effects of mucilage of Trigonella foenum graecum (Fenugreek) on adjuvant induced arthritic rats. Int Immunopharmacol 12:205–211
Skindersoe ME, Alhede M, Phipps R et al (2008) Effects of antibiotics on quorum sensing in Pseudomonas aeruginosa. Antimicrob Agents Chemother 52:3648–3663
Tolker-Nielsen T (2014) Pseudomonas aeruginosa biofilm infections: from molecular biofilm biology to new treatment possibilities. APMIS 122:1–51
Turki Z, El-Shayeb F, Abozeid A (2013) Seed morphology of some Trigonella L. species (Fabaceae) and its taxonomic significance. Int J Sci Res 3:940–948
Turkina MV, Vikström E (2019) Bacteria–host crosstalk: sensing of the quorum in the context of Pseudomonas aeruginosa infections. J Innate Immun 11:263–279
Vandeputte OM, Kiendrebeogo M, Rasamiravaka T et al (2011) The flavanone naringenin reduces the production of quorum sensing-controlled virulence factors in Pseudomonas aeruginosa PAO1. Microbiology 157:2120–2132
Vasavi HS, Arun AB, Rekha P (2014) Anti-quorum sensing activity of Psidium guajava L. flavonoids against Chromobacterium violaceum and Pseudomonas aeruginosa PAO1. Microbiol Immunol 58:286–293
Vasavi HS, Arun AB, Rekha PD (2016) Anti-quorum sensing activity of flavonoid-rich fraction from Centella asiatica L. against Pseudomonas aeruginosa PAO1. J Microbiol Immunol Infect 49:8–15
Venturi V (2006) Regulation of quorum sensing in Pseudomonas. FEMS Microbiol Rev 30:274–291
Yahara S, Kohjyouma M, Kohoda H (2000) Flavonoid glycosides and saponins from Astragalus shikokianus. Phytochemistry 53:469–471
Yoshikawa M, Murakami T, Komatsu H et al (1998) Medicinal foodstuffs. VIII. fenugreek seed. (2): Structures of six new furostanol saponins, trigoneosides IVa, Va, Vb, VI, VIIb, and VIIIb, from the seeds of Indian Trigonella foenum-graecum L. Heterocycles 1:397–405
Zaki AA, Shaaban MI, Hashish NE et al (2013) Assessment of anti-quorum sensing activity for some ornamental and medicinal plants native to Egypt. Sci Pharm 81:251–258
Acknowledgements
All appreciation to Professor Mona El-Sayed Mabrouk, Department of Botany and Microbiology, Damanhour University.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This research did not receive any specific grant from funding agencies in the public, copyediting service funding was provided by The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Conceptualization: KMG, MIS; Methodology: NGN, AAZ, MIS; Formal analysis and investigation: NGN, AAZ, DEE-B, HSR, KMG, MIS; Writing—original draft preparation: NGN, AAZ, HSR, KMG, MIS; Writing—review and editing: AAZ, DEE-B, HSR, KMG, MIS; Resources: NGN, AAZ, MIS; Supervision: AAZ, DEE-B, HSR, KMG, MIS.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This study was reviewed and deemed exempt by our IRB, No. 0201472 (18th March 2021), Faculty of Medicine, Alexandria University, Egypt.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Naga, N.G., Zaki, A.A., El-Badan, D.E. et al. Methoxyisoflavan derivative from Trigonella stellata inhibited quorum sensing and virulence factors of Pseudomonas aeruginosa. World J Microbiol Biotechnol 38, 156 (2022). https://doi.org/10.1007/s11274-022-03337-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-022-03337-x