Abstract
The neurotoxicity of NO2 exposure is well-known and potentially causes impaired of neural functions. This review aimed to estimate associations between prenatal NO2 exposure and neurodevelopment for children. Articles published until May 2019 reported prenatal NO2 exposure and children’s cognition, psychomotor, language, attention, IQ, and behavior function were searched according to all related terms. The main databases we retrieved included PubMed, Web of Science, Embase, and Cochrane Library. Coefficient was extracted, conversed, and synthesized by random effects meta-analysis. Meanwhile, qualitatively describe would be used for some studies which cannot be synthesized quantitatively for lack of quantity or methods inconsistency. Finally, a total of 3848 citations were searched, and only 10 studies were included. We estimated that per 10 μg/m3 increase of NO2 during pregnancy was associated with a − 0.76 point decrease in global psychomotor (95% CI, − 1.34, − 0.18) and a − 0.62 point decrease in fine psychomotor for children (95% CI, − 1.09, − 0.16). But no significant association found in general cognitive and language. In addition, through the literature review, it seemed that prenatal exposure to NO2 might cause adverse impacts on children’s attention, IQ, and different behaviors, but this requires confirmation from further researches. Our study indicated that prenatal exposure to NO2 seems to be associated with impaired neural development for children, especially for fine psychomotor. However, further studies are needed for determining the effects of prenatal air pollution exposure on attention, IQ, and behavior.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ambient air pollution is recognized as a global issue due to its adverse impacts on air quality and public health. According to statistics, the exposure concentration of air pollutants in many developed countries and most of developing countries were considerably higher than the guidelines recommended by the World Health Organization (WHO) (Organization 2007). Among them, nitrogen dioxide (NO2), a traffic-related air pollutant mainly derived from automobile exhaust and fuel combustion, still shows excessive exposure levels in many countries and even continues to increase. According to statistics, the exposure level of NO2 had increased 2.7 times from 1996 to 2012 in China (Seltenrich 2016).
The embryo and fetus, as the most sensitive period, are extremely susceptible to ambient NO2 exposure. Maternal exposure to NO2 can induce some abnormal reactions, including inflammation reaction, oxidative stress, and DNA methylation, which may cause adverse effects on organs and tissue of the offspring (Fiorito et al. 2018; Mirowsky et al. 2016). Some studies reported that maternal exposure to oxynitride might cause the oxidative damage of brain white and gray matter through oxidative stress (Anderson et al. 2018; Nagiah et al. 2015; Akhtar et al. 2017; Murray et al. 2015; Tonni et al. 2014). In addition, it also suggested that exposure to NO2 caused disruption of BBB and accumulation of amyloid β42 and α-synuclein starting in childhood through neural inflammation (Allen et al. 2014; Calderon-Garciduenas et al. 2015). Based on those evidences, it can be inferred that maternal exposure to NO2 may have adverse effects on children’s neural development.
Some studies and reviews have supported the hypothesis that exposure to ambient NO2 and other air pollution was associated with an increased risk of autism spectrum disorder (ASD) (Chun et al. 2020; Flores-Pajot et al. 2016) and attention deficit hyperactivity disorder (ADHD) for children (Donzelli and Carducci 2019; Donzelli et al. 2019; Forns et al. 2018; Min and Min 2017). However, the effects of prenatal exposure to NO2 on children’s neurodevelopment (including cognitive, psychomotor, language, and behavioral functions) are still unclear for different conclusions in related studies (Ren et al. 2019; Porta et al. 2016; Sentis et al. 2017). A study based on six European birth cohorts found that NO2 exposure during pregnancy was associated with delayed fine psychomotor development during childhood, but not cognition (Guxens et al. 2014). Nevertheless, some related studies did not find significant association between maternal exposure to NO2 and psychomotor development (Lin et al. 2014), and some studies considered it to have adverse effects on children’s cognitive function (Guxens et al. 2014; Porta et al. 2016). And for behavior, different studies also have drawn different conclusions (Ren et al. 2019; Yorifuji et al. 2017).
However, to our knowledge, there are no systematic reviews specifically addressing the association of prenatal NO2 exposure and children’s neural development. Therefore, we systematically review related literatures to explore and assess the incremental effect of 10 μg/m3 exposure to NO2 during gestation on children’s neurodevelopment, including cognition, language, fine and gross psychomotor, behavior, IQ, attention, and emotions.
Materials and methods
This review was developed in accordance with the PRISMA statement for systematic reviews (www.prisma-statement.org) and was registered with PROSPERO (www.crd.york.ac.uk/PROSPERO) under protocol number CRD42019125057.
PECO question
The research question was determined using the PECO strategy: population (children having undergone neurodevelopment evaluations and their mothers); exposure (10 μg/m3 exposure to NO2 during gestation); comparison (10 μg/m3 incremental increase); and outcome (children’s neurodevelopment, including cognition, fine and gross psychomotor, behavior, IQ, language, and attention). Based on this, we established research question as follows: For children, what is the incremental effect of 10 μg/m3 exposure to NO2 during gestation on children’s neurodevelopment, including cognition, language, fine and gross psychomotor, behavior, IQ, attention, and emotions.
Search strategy
To identify studies that estimated the effects of maternal exposure to NO2 on children’s neurodevelopment, related articles published until May 12, 2019, were searched using PubMed, Web of Science, Embase, and Cochrane Library. All related terms were used for retrieval, such as “maternal,” “air pollution,” “traffic-related pollution,” “nitrogen dioxide,” “neurodevelopment,” “cognition,” “psychomotor,” “language,” “behavior,” “attention,” and “child.” Full details were provided in the Appendix A. In addition, we manually searched the references in each included studies for additional publications.
Selection criteria and data collection
Studies with quantitative data on associations between maternal exposure to NO2 and children’s neurodevelopment were considered. Research articles were included if they (1) were written in English;( 2) were cohort or cross-sectional studies in human subjects; (3) measured maternal exposure to ambient NO2 throughout pregnancy or in trimester-specific periods; and (4) assessed children’s neurodevelopment outcomes such as cognition, language, psychomotor, emotions, behavior, emotions, and IQ levels. In addition, since our review only focused on the effects of maternal exposure to ambient NO2 on children’s development of neural function, some studies were excluded if (1) exposure window outside of pregnancy period and (2) outcome variables only included neurodevelopmental disorders, such as autistic spectrum disorder and attention deficit hyperactivity disorder.
The abstracts and titles screening, full-text screening, and data extraction were all carried out by two investigators independently. Discrepancies between the two investigators were resolved by discussion. The data extracted from each paper included study design, location, study period, sample size, methods and measurement, covariates, statistical analysis, and the ORs and 95% CIs used during statistical analyses.
Risk-of-bias assessment
The risk-of-bias (ROB) for each included study was assessed using a modified instrument specifically for examining associations between exposure to multiple air pollutants and autism spectrum disorder (Lam et al. 2016). This tool was developed based on the Cochrane Collaboration’s risk-of-bias tool (McHenry et al. 2018) and assesses the following 9 ROB domains: source population representation, blinding, exposure assessment (for air pollutants), outcome assessment, potential cofounding, incomplete outcome data, selective outcome reporting, conflict of interest, and other biases. Since the instructions for the outcome assessment domain were not fit for our review, prior to assessing ROB, we had modified the evaluation standard and instructions of outcome variable according to the criteria for the quality of the neuropsychological assessments created by McHenry et al. We rated each ROB domain as “low,” “probably low,” “probably high,” or “high” risk of bias or “not applicable” (risk of bias area not applicable to study) according to specific criteria as described in the modified ROB instruments (Appendix B). All included papers were independently evaluated by two investigators, and the contradictions were resolved through discussion.
Data synthesis and analysis
Meta-analysis was a preferred synthetic method when two or more unique studies reporting the same outcomes and provided sufficient quantitative data for meta-analysis. Prior to conducting the meta-analysis, coefficients and their confidence intervals (CIs) for the associations between NO2 exposure and outcome variables were extracted for meta-analysis. And standard errors (SEs) of the coefficients were calculated from the reported CIs under normal distribution assumption. And then the coefficients and SEs were converted to the same unit of 10 μg/m3 increase in NO2, which facilitate the consolidation of estimate from different studies. Increase of part per billion (ppb) was conversed to μg/m3 under standard atmosphere (101.325 Kpa) and standard temperature (25 °C), and the specific process of data conversion was shown in Appendix C. D-L random effects model meta-analysis was performed to merge effect value. And heterogeneity was quantified by I2 statistics. Meta-analysis was performed using “metafor” package in R version 3.5.2. Significance level was set at p < 0.05.
While other outcome variables were too heterogonous to meta-analysis, synthesized qualitatively in narrative was performed in accordance with the document Guidance on the Conduct of Narrative Synthesis in Systematic Reviews (Rodgers et al. 2019). In this process, we used tabulation and visual representations of data to simplify the key characteristics of included studies. And then, we synthesized evidence narratively. In the end, we compared our narrative results to those of other systematic reviews to judge the robustness of the results of this study.
Results
Search results
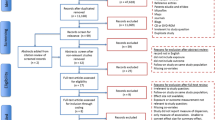
A total of 3848 citations (excluding duplicates, n = 647) were retrieved in our review, and 43 records were screened for full-text based on our study eligibility criteria independently. Finally, a total of 10 citations were included in this systematic review. The flowchart of the study selection process is depicted in Fig. 1.
Characteristics of included studies
All of 10 studies were published between 2012 and 2019. Table 1 presents the characteristics of included studies. Ages of children ranged from 6 months to 8 years of age. Sample sizes ranged from 438 (Lertxundi et al. 2015) to 33,890 (Yorifuji et al. 2017). In addition to one cross-sectional study (Ren et al. 2019) and one longitudinal survey (Yorifuji et al. 2017), the remaining 8 studies were based on prospective birth cohort design. And 5 studies were conducted in Europe (Guxens et al. 2012, 2014; Lertxundi et al. 2015, 2019; Sentis et al. 2017), such as Italy and Spanish, while the remaining 5 studies were conducted separately in Asian countries including China (Lin et al. 2014; Porta et al. 2016; Ren et al. 2019), South Korea (Kim et al. 2014), and Japan (Yorifuji et al. 2017). In particular, Guxens et al. (Guxens et al. 2014) had published a study which analyzed data from 6 European population-based birth cohorts conducted in 11 different regions, and the effect values for each subproject were provided in the supplemental material. So those 11 subprojects were also included in our meta-analysis.
Risk of bias assessment
Overall, most studies were rated as “low” or “probably low” ROB in most domains other than “potential confounding” and “selective outcome reporting”(Fig. 2). The incomplete outcome data in half of the studies were not well described and not adequately addressed. Otherwise, 6 studies were considered as “probably high” ROB for potential confounding that failed to adjust for many of the important confounders, such as maternal IQ which is significantly associated with children’s neurodevelopment (Kim et al. 2014). It is worth noting that one study was rated as “high” ROB in NO2 exposure assessment, because it utilized the air pollution concentrations in kindergarten as the surrogate of maternal exposure during pregnancy.
Meta-analysis
After preliminary synthesis, we found that studies involving general cognition, language, global psychomotor, fine psychomotor, and gross psychomotor could be quantitatively synthesized.
General cognitive
Two citations (Guxens et al. 2014; Kim et al. 2014) (including 8 cohort studies from different regions) estimated the effect of maternal exposure to NO2 on children’s general cognition. Overall, it showed that a 10 μg/m3 increase in maternal NO2 exposure was not significantly associated with general cognition for children (ES = − 0.33; 95% CI, − 1.02, 0.37), with a median heterogeneity (I2 = 48.1%) (Fig. 3a). The potential confounding and incomplete outcome data of those two studies are all considered as “probably high” ROB for lacking of important covariates.
Language
Nine cohort studies of 2 citations (Guxens et al. 2014; Lin et al. 2014) had assessed the effects of maternal NO2 exposure on children’s language. The summary effect estimate was − 0.13 (95% CI, − 0.34, 0.60), and evidence of statistical heterogeneity was low (I2 = 14.16%) (Fig. 3b). Except for consistent probably high ROB in potential confounding and incomplete data, cohort study in Taiwan also had probably high risk in the assessment of NO2 exposure and outcome variable.
Psychomotor outcomes
Three of the 10 citations included assessed the effects of maternal exposure to NO2 on the children’s psychomotor-related outcomes (Guxens et al. 2014; Kim et al. 2014; Lin et al. 2014), including global psychomotor, gross psychomotor, and fine psychomotor. Table 1 summarizes the cognition assessment tools used in each study and specific psychomotor outcome variables. The age of child cognitive testing ranged from less than 6 months to 7 years old. Among them, two citations (Guxens et al. 2014; Kim et al. 2014) (including 12 cohort studies from different regions) estimated the effect of maternal exposure to NO2 on children’s global psychomotor. The results of the meta-analysis showed that a 10 μg/m3 increase in maternal NO2 exposure was significantly associated with children’s global psychomotor with combined ES which was − 0.76, (95% CI, − 1.34, − 0.18), with moderate heterogeneity (I2 = 36.98%) (Fig. 4a). Moreover, children’s fine psychomotor was also associated significantly with 10 μg/m3 increases in maternal NO2 exposure (ES = 0.62; 95% CI, − 1.09, − 0.16; I2 = 0.00%) according to summary estimate of 8 cohort studies among 2 citations (Fig. 4b). But no significant association was found between maternal exposure to NO2 and children’s gross psychomotor (ES = − 0.38; 95% CI, − 0.90, 0.14; I2 = 16.68%) in meta-analysis (Fig. 4c). These studies have a relatively consistent potential high ROB among incomplete data and potential confounding, but low ROB in the assessment of NO2 exposure and outcomes. Another study conducted in four regions of Spanish also reported that prenatal exposure to NO2 was related with decrease fine motor in boys.
Synthesized qualitatively in narrative
We developed Table 1 to visually represent the preliminary comprehensive results, which contribute to simply list studies on the effect of NO2 on children’s attention, IQ, behavior, and emotion.
Attention
Only two studies (Sentis et al. 2017; Yorifuji et al. 2017) estimated the effect of maternal exposure to NO2 on children’s attentional problem. A longitudinal survey of 33,890 samples conducted in Japan found that per 10.8 ppm increase in NO2 exposure during pregnancy may cause increased risk for “failure to pay attention when crossing a street” for children aged 8 (adjust OR = 1.10; 95% CI, 1.02–1.19). But no obviously impact on the risk for interrupting people and inability to wait his/her turn during play was reported (Yorifuji et al. 2017). But it is worth noting that this study had really high ROB in the assessment of NO2 exposure since they assessed maternal NO2 exposure level just based on municipality-representative monthly average concentrations. Another cohort study (Sentis et al. 2017) of 1298 children in Spanish showed a lower risk of overall bias. It used the Kiddie-Conners Continuous Performance Test (K-CPT) to assess attention function and found that prenatal exposure to NO2 was associated with an impaired standard error of the hit reaction time (HRT(SE)) (increase of 1.12 ms [95% CI, 0.22–2.02] per 10 μg/m3 increase in prenatal NO2) and increased omission errors (increase of 1.06 ms [95% CI, 1.01–1.11] per 10 μg/m3 increase in prenatal NO2), which were both indicative inattentiveness.
IQ
For IQ, only a cohort study of 719 has been reported (Tzivian et al. 2016). It found that a 10 μg/m3 higher NO2 exposure during pregnancy was associated with 1.4 fewer points (95% CI, − 2.6, − 0.20) of verbal IQ and 1.4 fewer points (95% CI, − 2.7, − 0.20) of verbal comprehension IQ, but no significant associations with full-scale IQ and performance IQ. However, this study was not control for potential confounders for maternal IQ level.
Behavior
Two studies (Ren et al. 2019; Yorifuji et al. 2017) estimated the effect of maternal exposure to NO2 on children’s behavior, but the outcome variables between them were different. The study conducted in Japan mainly estimated effects on aggressive behaviors including lying, destroying toys and/or books, hurting other people, and causing disturbances in public. But no significant association was found in this study. Another study of 657 samples using behavior difficult assessed by the Strengths and Difficulties Questionnaire as outcome variable and observed positive associations between exposure to NO2 (aOR = 1.204; 95% CI, 1.042, 1.392) and total difficulties, especially in the first trimester (aOR = 1.039; 95% CI, 1.013, 1.066). Both those studies had a probably high ROB. The longitudinal survey in Japan collected main information by mail-related questionnaire, so it caused a probably high ROB in source population representation, the assessment of variables, and incomplete outcome data. Another retrospective survey mainly aimed at kindergarten children and caused high ROB in NO2 exposure assessment.
Emotion
We only retrieved one study that included emotion as the outcome variable, and it found no significant correlation between maternal exposure to NO2 and children’s emotion symptoms (aOR = 0.937; 95% CI, 0.854, 1.108) (Ren et al. 2019). But this study had high ROB in the assessment of NO2 exposure since it utilized the air pollution concentrations in kindergarten as the surrogate of maternal exposure during pregnancy.
We were unable to compare our results to those of other systematic reviews because no other reviews have dealt with this topic, as far as we are aware.
Discussion
Our meta-analysis has found evidence suggestive of a relationship between prenatal exposure to NO2 and the development of psychomotor, especially in global psychomotor and fine psychomotor for children. But there is no significant association in language and cognitive development. Through the literature review, only several studies with high ROB have reported possible negative effects, so the relationship between prenatal exposure to NO2 and children’s attention function, behavior difference, IQ, and emotion is still unclear. Overall, most studies were rated as “low” or “probably low” risk of bias in most domains, but half of the studies were at probably high ROB in incomplete outcome data and potential confounding.
Our review has found a negative association between prenatal NO2 exposure and psychomotor in children, especially in fine psychomotor. This finding was consistent with previous six European birth cohorts (Guxens et al. 2014). Lertxundi et al. also found adverse effects of prenatal NO2 exposure on motor scores in women living close to (< 300 m) to metal processing activities (Lertxundi et al. 2015). In addition, a birth cohort conducted in South Korea had suggested that NO2 exposure on psychomotor development index (PDI) may be especially stronger at an earlier age since it only found significant adverse effects at 6 months of age, but not at 12 and 24 months (Kim et al. 2014). However, our review found no significant difference while stratified by age (results were not show). Our meta-analysis showed that there was no significant difference between NO2 exposure during pregnancy and cognitive development, which was consisted with Guxens’ studies (Guxens et al. 2012; Guxens et al. 2014). However, Lertxundi et al. pointed out different results that prenatal exposure to NO2 may decrease children’s mental score, and this negative effect could be higher in the proximity of metal processing plants (Lertxundi et al. 2015). The same negative associations were found in another study after adjusting maternal IQ (Kim et al. 2014). It indicated that maternal IQ might be an important confounding factor between air pollution and neural development in offspring, but was not considered in most included studies. However, there is not enough evidence to support the effect of prenatal NO2 exposure on children’s IQ, behavior, and emotion for the lack or contradiction of study. So further studies about utero air pollution exposure on those neural function are generally needed, since some studies suggested that exposure to air pollution during non-pregnancy may cause impairment for IQ and behavior (Calderon-Garciduenas et al. 2015; Sunyer et al. 2015; Tzivian et al. 2016). One study also pointed that NO2 exposure during the first trimester was more significantly associated with behavioral (Ren et al. 2019), so the key exposure period should also be addressed in future studies.
Generally speaking, our review found that prenatal exposure to NO2 has negative impact in neural development for children and this finding was biologically explicable. It was speculated that impaired immune function, such as oxidative stress (Calderon-Garciduenas 2016; Li et al. 2015, 2016, 2019) and inflammatory responses (Ehsanifar et al. 2019; Ransohoff et al. 2015; Rychlik et al. 2019), may be a potential pathway by which prenatal exposure to NO2 may cause an impact on neural functional development. But there was no sufficient evidence indicated the mediated proportion of impaired immune function. In addition, from the perspective of genetics, we speculated that DNA adducts were also a potential pathway, because some studies suggested that DNA adducts in cord blood were associated with children’s behavior and motor scores (Perera et al. 2011; Tang et al. 2008), while it may be higher when maternal exposure to PM2.5 and NO2 during pregnancy (Pedersen et al. 2009; Pedersen et al. 2015).
Moreover, by meta-analysis, only psychomotor function was found to be impaired. We attributed it to the abnormal release of dopamine in the cerebral cortex. Some animal studies supported evidences, which suggested that a facilitated release of dopamine in the prefrontal cortex or in the striatum was triggered by the diesel exhaust particle exposure during pregnancy (Suzuki et al. 2010; Yokota et al. 2009). The dopamine and noradrenaline systems in the prefrontal cortex had an important role in the control of motor activity through VTA-accumbency-dopamine activity (Oades et al. 1986). In humans, a double-blind randomized crossover study was carried out, in which volunteers were exposed to dilute diesel exhaust or filtered air for 1 h. There was increased activity of the frontal cortex during and after diesel exhaust exposure (Cruts et al. 2008). The frontal cortex controls the actions of the body through its motor areas such as the primary motor or the premotor cortex. Some related animal studies also supported the results of this review. Some studies have found that diesel exhaust particles, black carbon, or NO2 exposure during pregnancy decreased the motor function in the offspring mice, attributed this impact to the change of the neurochemical monoamine metabolism of several regions of the brain (Suzuki et al. 2010; Yokota et al. 2009).
To our knowledge, our study is the first meta-analysis and systematic review to estimate comprehensively the effect of prenatal exposure to NO2 on children’s neural functional development. Extensive outcome variables were evaluated by our review, including cognition, psychomotor, language, IQ, behavior, and emotion. To avoid repetition bias, we also excluded some estimate values for the same outcome variables that were concomitant in six European cohorts and another article.
One of the limitations of this review is that we cannot carry out meta-analysis on some neural function including attention, behavior, and IQ, since there are few studies with IQ as outcome variables, and the measurement with behavior and attention was inconsistent. However, we summarized and described the previous research results by systematic review and pointed out possible effects and the direction of further research. In addition, as the scope of NO2 exposure was not reported in some studies, our review could not conduct stratified analysis based on the regional pollution level although we surmised that the effects in high-polluted areas might be stronger. Publication bias must be considered in our meta-analysis. Scientific investigations that do not find any significant results often fail to be published. The results of egg’s test showed low report bias in each sub-study included in the meta-analysis, but overestimation of the effects value still cannot be ignored.
In our review, we encountered a great variety of methods used for measuring specific neurocognitive abilities. Thus a more homogenous deployment of measurement methods for outcome variable is suggested to future studies. Second, the effect of prenatal air pollution exposure on children’s attention, behavior difference, IQ, and emotion is still unclear and still needs further assessment. In addition, it is necessary to report the average concentration for main pollutants, which can contribute to the subgroup analysis to estimate the effects of prenatal exposure to air pollution on children neural function in high polluted areas. This review only focused on prenatal exposure to NO2 and found strong evidence. But air pollution is a mixture contained of various components, including gases (such as NO2), particulate matter (PM), metals, and organic compounds that have been found may affect children’s neurodevelopment (Flores-Pajot et al. 2016; Donzelli and Carducci 2019; Donzelli et al. 2019), so it is necessary to conduct more systematic reviews about other pollutants which also may cause adverse impact on neural development.
Conclusions
The results from our study suggest that prenatal exposure to NO2 might be associated with psychomotor, especially in fine psychomotor for children, but not in language and cognitive development. In addition, the relationship between prenatal exposure to NO2 and children’s attention, behavior difference, IQ, and emotion is still unclear and requires more confirmation from further research. Based on this, we suggested more homogenous deployment of measurement methods for outcome variable to future studies.
References
Akhtar F, Rouse CA, Catano G, Montalvo M, Ullevig SL, Asmis R, Kharbanda K, Maffi SK (2017) Acute maternal oxidant exposure causes susceptibility of the fetal brain to inflammation and oxidative stress. J Neuroinflammation 14:195. https://doi.org/10.1186/s12974-017-0965-8
Allen JL et al (2014) Developmental exposure to concentrated ambient ultrafine particulate matter air pollution in mice results in persistent and sex-dependent behavioral neurotoxicity and glial activation. Toxicol Sci 140:160–178. https://doi.org/10.1093/toxsci/kfu059
Anderson SM, Naidoo RN, Ramkaran P, Phulukdaree A, Muttoo S, Asharam K, Chuturgoon AA (2018) The effect of nitric oxide pollution on oxidative stress in pregnant women living in Durban, South Africa. 74:228–239. https://doi.org/10.1007/s00244-017-0465-y
Calderon-Garciduenas L (2016) Smoking and cerebral oxidative stress and air pollution: a dreadful equation with particulate matter involved and one more powerful reason not to smoke anything! J Alzheimers Dis 54:109–112. https://doi.org/10.3233/jad-160510
Calderon-Garciduenas L et al (2015) Decreases in short term memory, IQ, and altered brain metabolic ratios in urban apolipoprotein epsilon4 children exposed to air pollution. J Alzheimers Dis 45:757–770. https://doi.org/10.3233/jad-142685
Chun H, Leung C, Wen SW, McDonald J, Shin HH (2020) Maternal exposure to air pollution and risk of autism in children: a systematic review and meta-analysis. Environ Pollut (Barking, Essex : 1987) 256:113307. https://doi.org/10.1016/j.envpol.2019.113307/fncel.2015.00018
Cruts B, van Etten L, Tornqvist H, Blomberg A, Sandstrom T, Mills NL, Borm PJ (2008) Exposure to diesel exhaust induces changes in EEG in human volunteers. Part Fibre Toxicol 5:4. https://doi.org/10.1186/1743-8977-5-4
Donzelli G, Carducci A (2019) The association between lead and attention-deficit/hyperactivity disorder: a systematic review. 16:16. https://doi.org/10.3390/ijerph16030382
Donzelli G, Llopis-Gonzalez A, Llopis-Morales A, Cioni L (2019) Particulate matter exposure and attention-deficit/hyperactivity disorder in children:aA systematic review of epidemiological studies. 17:17. https://doi.org/10.3390/ijerph17010067
Ehsanifar M, Jafari AJ, Nikzad H, Zavareh MS, Atlasi MA, Mohammadi H, Tameh AA (2019) Prenatal exposure to diesel exhaust particles causes anxiety, spatial memory disorders with alters expression of hippocampal pro-inflammatory cytokines and NMDA receptor subunits in adult male mice offspring. Ecotoxicol Environ Saf 176:34–41. https://doi.org/10.1016/j.ecoenv.2019.03.090
Fiorito G et al (2018) Oxidative stress and inflammation mediate the effect of air pollution on cardio- and cerebrovascular disease: a prospective study in nonsmokers. 59:234–246. https://doi.org/10.1002/em.22153
Flores-Pajot MC, Ofner M, Do MT, Lavigne E, Villeneuve PJ (2016) Childhood autism spectrum disorders and exposure to nitrogen dioxide, and particulate matter air pollution: a review and meta-analysis. Environ Res 151:763–776. https://doi.org/10.1016/j.envres.2016.07.030
Forns J et al (2018) Air pollution exposure during pregnancy and symptoms of attention deficit and hyperactivity disorder in children in Europe. Epidemiology (Cambridge, Mass) 29:618–626. https://doi.org/10.1097/ede.0000000000000874
Guxens M, Aguilera I, Ballester F, Estarlich M, Fernández-Somoano A, Lertxundi A, Lertxundi N, Mendez MA, Tardón A, Vrijheid M, Sunyer J, on behalf of the INMA (INfancia y Medio Ambiente) Project (2012) Prenatal exposure to residential air pollution and infant mental development: modulation by antioxidants and detoxification factors. Environ Health Perspect 120:144–149. https://doi.org/10.1289/ehp.1103469
Guxens M et al (2014) Air pollution during pregnancy and childhood cognitive and psychomotor development: six European birth cohorts. Epidemiology (Cambridge, Mass) 25:636–647. https://doi.org/10.1097/ede.0000000000000133
Kim E, Park H, Hong YC, Ha M, Kim Y, Kim BN, Kim Y, Roh YM, Lee BE, Ryu JM, Kim BM, Ha EH (2014) Prenatal exposure to PM(1)(0) and NO(2) and children's neurodevelopment from birth to 24 months of age: mothers and Children's Environmental Health (MOCEH) study. Sci Total Environ 481:439–445. https://doi.org/10.1016/j.scitotenv.2014.01.107
Lam J, Sutton P, Kalkbrenner A, Windham G, Halladay A, Koustas E, Lawler C, Davidson L, Daniels N, Newschaffer C, Woodruff T (2016) A systematic review and meta-analysis of multiple airborne pollutants and autism spectrum disorder. PLoS One 11:e0161851. https://doi.org/10.1371/journal.pone.0161851
Lertxundi A, Baccini M, Lertxundi N, Fano E, Aranbarri A, Martínez MD, Ayerdi M, Álvarez J, Santa-Marina L, Dorronsoro M, Ibarluzea J (2015) Exposure to fine particle matter, nitrogen dioxide and benzene during pregnancy and cognitive and psychomotor developments in children at 15 months of age. Environ Int 80:33–40. https://doi.org/10.1016/j.envint.2015.03.007
Lertxundi A, Andiarena A, Martínez MD, Ayerdi M, Murcia M, Estarlich M, Guxens M, Sunyer J, Julvez J, Ibarluzea J (2019) Prenatal exposure to PM2.5 and NO2 and sex-dependent infant cognitive and motor development. Environ Res 174:114–121. https://doi.org/10.1016/j.envres.2019.04.001
Li R, Kou X, Xie L, Cheng F, Geng H (2015) Effects of ambient PM2.5 on pathological injury, inflammation, oxidative stress, metabolic enzyme activity, and expression of c-fos and c-jun in lungs of rats. Environ Sci Pollut Res Int 22:20167–20176. https://doi.org/10.1007/s11356-015-5222-z
Li W, Wilker EH, Dorans KS, Rice MB, Schwartz J, Coull BA, Koutrakis P, Gold DR, Keaney JF Jr, Lin H, Vasan RS, Benjamin EJ, Mittleman MA (2016) Short-term exposure to air pollution and biomarkers of oxidative stress: the Framingham Heart Study. J Am Heart Assoc 5:5. https://doi.org/10.1161/jaha.115.002742
Li X, Zhang X, Zhang Z, Han L, Gong D, Li J, Wang T, Wang Y, Gao S, Duan H, Kong F (2019) Air pollution exposure and immunological and systemic inflammatory alterations among schoolchildren in China. Sci Total Environ 657:1304–1310. https://doi.org/10.1016/j.scitotenv.2018.12.153
Lin CC, Yang SK, Lin KC, Ho WC, Hsieh WS, Shu BC, Chen PC (2014) Multilevel analysis of air pollution and early childhood neurobehavioral development. Int J Environ Res Public Health 11:6827–6841. https://doi.org/10.3390/ijerph110706827
McHenry MS et al (2018) Neurodevelopment in young children born to HIV-infected mothers: a meta-analysis. Pediatrics 141:141. https://doi.org/10.1542/peds.2017-2888
Min JY, Min KB (2017) Exposure to ambient PM10 and NO2 and the incidence of attention-deficit hyperactivity disorder in childhood. Environ Int 99:221–227. https://doi.org/10.1016/j.envint.2016.11.022
Mirowsky JE, Dailey LA, Devlin RB (2016) Differential expression of pro-inflammatory and oxidative stress mediators induced by nitrogen dioxide and ozone in primary human bronchial epithelial cells. Inhal Toxicol 28:374–382. https://doi.org/10.1080/08958378.2016.1185199
Murray KN, Parry-Jones AR, Allan SM (2015) Interleukin-1 and acute brain injury. Front Cell Neurosci 9, 18. https://doi.org/10.3389/fncel.2015.00018
Nagiah S, Phulukdaree A, Naidoo D, Ramcharan K, Naidoo RN, Moodley D, Chuturgoon A (2015) Oxidative stress and air pollution exposure during pregnancy: a molecular assessment. Hum Exp Toxicol 34:838–847. https://doi.org/10.1177/0960327114559992
Oades RD, Taghzouti K, Rivet JM, Simon H, Le Moal M (1986) Locomotor activity in relation to dopamine and noradrenaline in the nucleus accumbens, septal and frontal areas: a 6-hydroxydopamine study. Neuropsychobiology 16:37–42. https://doi.org/10.1159/000118294
Organization WH (2007) Air quality guidelines: global update 2005. Particulate matter, ozone, nitrogen dioxide and sulfur dioxide. Indian J Med Res 4:492–493
Pedersen M et al (2009) Increased micronuclei and bulky DNA adducts in cord blood after maternal exposures to traffic-related air pollution. Environ Res 109:1012–1020. https://doi.org/10.1016/j.envres.2009.08.011
Pedersen M, Mendez MA, Schoket B, Godschalk RW, Espinosa A, Landström A, Villanueva CM, Merlo DF, Fthenou E, Gracia-Lavedan E, van Schooten FJ, Hoek G, Brunborg G, Meltzer HM, Alexander J, Nielsen JK, Sunyer J, Wright J, Kovács K, de Hoogh K, Gutzkow KB, Hardie LJ, Chatzi L, Knudsen LE, Anna L, Ketzel M, Haugen M, Botsivali M, Nieuwenhuijsen MJ, Cirach M, Toledano MB, Smith RB, Fleming S, Agramunt S, Kyrtopoulos SA, Lukács V, Kleinjans JC, Segerbäck D, Kogevinas M (2015) Environmental, dietary, maternal, and fetal predictors of bulky DNA adducts in cord blood: a European mother-child study (NewGeneris). Environ Health Perspect 123:374–380. https://doi.org/10.1289/ehp.1408613
Perera FP, Wang S, Vishnevetsky J, Zhang B, Cole KJ, Tang D, Rauh V, Phillips DH (2011) Polycyclic aromatic hydrocarbons-aromatic DNA adducts in cord blood and behavior scores in New York city children. Environ Health Perspect 119:1176–1181. https://doi.org/10.1289/ehp.1002705
Porta D et al (2016) Air pollution and cognitive development at age 7 in a prospective Italian birth cohort. Epidemiology (Cambridge, Mass) 27:228–236. https://doi.org/10.1097/ede.0000000000000405
Ransohoff RM, Schafer D, Vincent A, Blachere NE, Bar-Or A (2015) Neuroinflammation: ways in which the immune system affects the brain. Neurotherapeutics 12:896–909. https://doi.org/10.1007/s13311-015-0385-3
Ren Y et al (2019) Outdoor air pollution pregnancy exposures are associated with behavioral problems in China's preschoolers. Environ Sci Pollut Res Int 26:2397–2408. https://doi.org/10.1007/s11356-018-3715-2
Rodgers M, Arai L, Britten N, Petticrew M, Popay M, Roberts H, Sowden A (2019) Guidance on the conduct of narrative synthesis in systematic reviews: a comparison of guidance-led narrative synthesis versus meta-analysis
Rychlik KA et al (2019) In utero ultrafine particulate matter exposure causes offspring pulmonary immunosuppression. 116:3443–3448. https://doi.org/10.1073/pnas.1816103116
Seltenrich N (2016) A satellite view of pollution on the ground: long-term changes in global nitrogen dioxide. Environ Health Perspect 124:A56
Sentis A et al (2017) Prenatal and postnatal exposure to NO2 and child attentional function at 4-5years of age. Environ Int 106:170–177. https://doi.org/10.1016/j.envint.2017.05.021
Sunyer J, Esnaola M, Alvarez-Pedrerol M, Forns J, Rivas I, López-Vicente M, Suades-González E, Foraster M, Garcia-Esteban R, Basagaña X, Viana M, Cirach M, Moreno T, Alastuey A, Sebastian-Galles N, Nieuwenhuijsen M, Querol X (2015) Association between traffic-related air pollution in schools and cognitive development in primary school children: a prospective cohort study. PLoS Med 12:e1001792. https://doi.org/10.1371/journal.pmed.1001792
Suzuki T, Oshio S, Iwata M, Saburi H, Odagiri T, Udagawa T, Sugawara I, Umezawa M, Takeda K (2010) In utero exposure to a low concentration of diesel exhaust affects spontaneous locomotor activity and monoaminergic system in male mice. Part Fibre Toxicol 7:7. https://doi.org/10.1186/1743-8977-7-7
Tang D, Li TY, Liu JJ, Zhou ZJ, Yuan T, Chen YH, Rauh VA, Xie J, Perera F (2008) Effects of prenatal exposure to coal-burning pollutants on children's development in China. Environ Health Perspect 116:674–679. https://doi.org/10.1289/ehp.10471
Tonni G, Leoncini S, Signorini C, Ciccoli L, De Felice C (2014) Pathology of perinatal brain damage: background and oxidative stress markers. Arch Gynecol Obstet 290:13–20. https://doi.org/10.1007/s00404-014-3208-6
Tzivian L et al. (2016) Long-term air pollution and traffic noise exposures and cognitive function:a cross-sectional analysis of the Heinz Nixdorf recall study J Toxicol Environ Health A 79:1057–1069. doi:https://doi.org/10.1080/15287394.2016.1219570
Yokota S, Mizuo K, Moriya N, Oshio S, Sugawara I, Takeda K (2009) Effect of prenatal exposure to diesel exhaust on dopaminergic system in mice. Neurosci Lett 449:38–41. https://doi.org/10.1016/j.neulet.2008.09.085
Yorifuji T, Kashima S, Diez MH, Kado Y, Sanada S, Doi H (2017) Prenatal exposure to outdoor air pollution and child behavioral problems at school age in Japan. Environ Int 99:192–198. https://doi.org/10.1016/j.envint.2016.11.016
Funding
The present study was funded by the Social Development of Science and Technology of Shaanxi Province [grant number 2019SF-100], the Bureau of Xi’an Science and Technology [grant number 201805098YX6SF32(1)], and the Center for Maternal and Child Health, Chinese Center for Disease Control and Prevention [grant number 2015FYJ002]. The funder did not participate in any part of the study from design to approval of the manuscript, except for supporting this project.
Author information
Authors and Affiliations
Contributions
LS and WY had the idea for the article. LS, LY, LH, and WY reviewed the literature. LY, CQ, ZY, ZF, and MC checked and managed the data. LS and MC performed data analysis. LS, LY, LH, and WY drafted the manuscript. CQ, ZY, ZF, and MC provided expert review of the manuscript and reviewed and approved the manuscript. All authors contributed to the research article and approved the final version.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no actual or potential conflict of interests.
Ethical approval and consent to participate
Not applicable.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shang, L., Yang, L., Yang, W. et al. Effects of prenatal exposure to NO2 on children’s neurodevelopment: a systematic review and meta-analysis. Environ Sci Pollut Res 27, 24786–24798 (2020). https://doi.org/10.1007/s11356-020-08832-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08832-y