Abstract
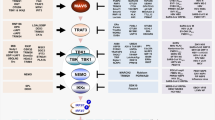
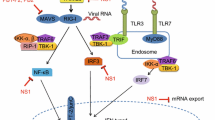
A wide range of host cellular signal transduction pathways can be stimulated by influenza virus infection. Some of these signal transduction pathways induce the host cell’s innate immune response against influenza virus, while others are essential for efficient influenza virus replication. This review examines the cellular signaling induced by influenza virus infection in host cells, including host pattern recognition receptor (PRR)-related signaling, protein kinase C (PKC), Raf/MEK/ERK and phosphatidy-linositol-3-kinase (PI3K)/Akt signaling, and the corresponding effects on the host cell and/or virus, such as recognition of virus by the host cell, viral absorption and entry, viral ribonucleoprotein (vRNP) export, translation control of cellular and viral proteins, and virus-induced cell apoptosis. Research into influenza virus-induced cell signaling promotes a clearer understanding of influenza virus-host interactions and assists in the identification of novel antiviral targets and antiviral strategies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Proud C G. PKR: A new name and new roles. Trends Biochem Sci, 1995, 20: 241–246, 1:CAS:528:DyaK2MXmtlaqu7w%3D, 7631421, 10.1016/S0968-0004(00)89025-8
Williams B R. PKR; a sentinel kinase for cellular stress. Oncogene, 1999, 18: 6112–6120, 1:CAS:528:DyaK1MXns1OqsrY%3D, 10557102, 10.1038/sj.onc.1203127
Dauber B, Martinez-Sobrido L, Schneider J, et al. Influenza B virus ribonucleoprotein is a potent activator of the antiviral kinase PKR. PLoS Pathog, 2009, 5: e1000473, 19521506, 10.1371/journal.ppat.1000473, 1:CAS:528:DC%2BD1MXnsFSqsbo%3D
Balachandran S, Barber G N. PKR in innate immunity, cancer, and viral oncolysis. Methods Mol Biol, 2007, 383: 277–301, 1:CAS:528:DC%2BD2sXhtleru7zE, 18217692, 10.1007/978-1-59745-335-6_18
Garcia M A, Meurs E F, Esteban M. The dsRNA protein kinase PKR: virus and cell control. Biochimie, 2007, 89: 799–811, 1:CAS:528:DC%2BD2sXnsFKrtbc%3D, 17451862, 10.1016/j.biochi.2007.03.001
Balachandran S, Roberts P C, Brown L E, et al. Essential role for the dsRNA-dependent protein kinase PKR in innate immunity to viral infection. Immunity, 2000, 13: 129–141, 1:CAS:528:DC%2BD3cXlsVyhtLY%3D, 10933401, 10.1016/S1074-7613(00)00014-5
Takizawa T, Ohashi K, Nakanishi Y. Possible involvement of double-stranded RNA-activated protein kinase in cell death by influenza virus infection. J Virol, 1996, 70: 8128–8132, 1:CAS:528:DyaK28Xmt1yqsbk%3D, 8892939
Balachandran S, Roberts P C, Kipperman T, et al. Alpha/beta interferons potentiate virus-induced apoptosis through activation of the FADD/Caspase-8 death signaling pathway. J Virol, 2000, 74: 1513–1523, 1:CAS:528:DC%2BD3cXmtFynuw%3D%3D, 10627563, 10.1128/JVI.74.3.1513-1523.2000
Li S, Min J Y, Krug R M, et al. Binding of the influenza A virus NS1 protein to PKR mediates the inhibition of its activation by either PACT or double-stranded RNA. Virology, 2006, 349: 13–21, 1:CAS:528:DC%2BD28Xks1Ontbc%3D, 16466763, 10.1016/j.virol.2006.01.005
Lu Y, Wambach M, Katze M G, et al. Binding of the influenza virus NS1 protein to double-stranded RNA inhibits the activation of the protein kinase that phosphorylates the elF-2 translation initiation factor. Virology, 1995, 214: 222–228, 1:CAS:528:DyaK2MXpslOmtb4%3D, 8525619, 10.1006/viro.1995.9937
Takeda K, Kaisho T, Akira S. Toll-like receptors. Annu Rev Immunol, 2003, 21: 335–376, 1:CAS:528:DC%2BD3sXjtF2isrg%3D, 12524386, 10.1146/annurev.immunol.21.120601.141126
Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol, 2004, 4: 499–511, 1:CAS:528:DC%2BD2cXlt1Cqur0%3D, 15229469, 10.1038/nri1391
Diebold S S, Kaisho T, Hemmi H, et al. Innate Antiviral responses by means of TLR7-Mediated recognition of single-stranded RNA. Science, 2004, 303: 1529–1531, 1:CAS:528:DC%2BD2cXhslCgsb4%3D, 14976261, 10.1126/science.1093616
Lund J M, Alexopoulou L, Sato A, et al. Recognition of sin gle-stranded RNA viruses by Toll-like receptor 7. Proc Natl Acad Sci USA, 2004, 101: 5598–5603, 1:CAS:528:DC%2BD2cXjsVagu7c%3D, 15034168, 10.1073/pnas.0400937101
Guillot L, Le Goffic R, Bloch S, et al. Involvement of toll-like receptor 3 in the immune response of lung epithelial cells to double-stranded RNA and influenza A virus. J Biol Chem, 2005, 280: 5571–5580, 1:CAS:528:DC%2BD2MXhtlCls7s%3D, 15579900, 10.1074/jbc.M410592200
Lee D C, Cheung C Y, Law A H, et al. p38 mitogen-activated protein kinase-dependent hyperinduction of tumor necrosis factor alpha expression in response to avian influenza virus H5N1. J Virol, 2005, 79: 10147–10154, 1:CAS:528:DC%2BD2MXntFCrtLo%3D, 16051807, 10.1128/JVI.79.16.10147-10154.2005
Maruoka S, Hashimoto S, Gon Y, et al. ASK1 regulates influenza virus infection-induced apoptotic cell death. Biochem Biophys Res Commun, 2003, 307: 870–876, 1:CAS:528:DC%2BD3sXls1Kqu7o%3D, 12878192, 10.1016/S0006-291X(03)01283-X
Kujime K, Hashimoto S, Gon Y, et al. p38 mitogen-activated protein kinase and c-jun-NH2-terminal kinase regulate RANTES production by influenza virus-infected human bronchial epithelial cells. J Immunol, 2000, 164: 3222–3228, 1:CAS:528:DC%2BD3cXhvVeks7Y%3D, 10706714
Ludwig S, Ehrhardt C, Neumeier E R, et al. Influenza virus-induced AP-1-dependent gene expression requires activation of the JNK signaling pathway. J Biol Chem, 2001, 276: 10990–10998, 1:CAS:528:DC%2BD3MXjvFCqu7Y%3D, 10.1074/jbc.M009902200
Kakugawa S, Shimojima M, Goto H, et al. The MAPK-activated kinase RSK2 plays a role in innate immune responses to influenza virus infection. J Virol, 2009
Kajiya T, Orihara K, Hamasaki S, et al. Toll-like receptor 2 expression level on monocytes in patients with viral infections: Monitoring infection severity. J Infect, 2008, 57: 249–259, 18657324, 10.1016/j.jinf.2008.06.009
Lee R M, White M R, Hartshorn K L. Influenza a viruses upregulate neutrophil toll-like receptor 2 expression and function. Scand J Immunol, 2006, 63: 81–89, 1:CAS:528:DC%2BD28XjtlCkt7Y%3D, 16476006, 10.1111/j.1365-3083.2005.01714.x
Whitmore M M, Iparraguirre A, Kubelka L, et al. Negative regulation of TLR-signaling pathways by activating transcription factor-3. J Immunol, 2007, 179: 3622–3630, 1:CAS:528:DC%2BD2sXpvFKls7o%3D, 17785797
Barral P M, Sarkar D, Su Z-z, et al. Functions of the cytoplasmic RNA sensors RIG-I and MDA-5: Key regulators of innate immunity. Pharmacol Ther, 2009, 124: 219–234, 1:CAS:528:DC%2BD1MXhtFGktLnO, 19615405, 10.1016/j.pharmthera.2009.06.012
Yoneyama M, Fujita T. RIG-I family RNA helicases: Cytoplasmic sensor for antiviral innate immunity. Cytokine Growth Factor Rev, 2007, 18: 545–551, 1:CAS:528:DC%2BD2sXpvFGrs7Y%3D, 17683970, 10.1016/j.cytogfr.2007.06.023
Takeuchi O, Akira S. MDA5/RIG-I and virus recognition. Curr Opin Immunol, 2008, 20: 17–22, 1:CAS:528:DC%2BD1cXisVWhu7g%3D, 18272355, 10.1016/j.coi.2008.01.002
Le Goffic R, Pothlichet J, Vitour D, et al. Cutting edge: Influenza A virus activates TLR3-dependent inflammatory and RIG-I-dependent antiviral responses in human lung epithelial cells. J Immunol, 2007, 178: 3368–3372, 17339430
Siren J, Imaizumi T, Sarkar D, et al. Retinoic acid inducible gene-I and mda-5 are involved in influenza A virus-induced expression of antiviral cytokines. Microbes Infect, 2006, 8: 2013–2020, 1:CAS:528:DC%2BD28XpsVOmu7w%3D, 16797201, 10.1016/j.micinf.2006.02.028
Matikainen S, Siren J, Tissari J, et al. Tumor necrosis factor alpha enhances influenza A virus-induced expression of antiviral cytokines by activating RIG-I gene expression. J Virol, 2006, 80: 3515–3522, 1:CAS:528:DC%2BD28Xjt1Kis74%3D, 16537619, 10.1128/JVI.80.7.3515-3522.2006
Hale B G, Randall R E, Ortin J, et al. The multifunctional NS1 protein of influenza A viruses. J Gen Virol, 2008, 89: 2359–2376, 1:CAS:528:DC%2BD1cXht1ektbfP, 18796704, 10.1099/vir.0.2008/004606-0
Talon J, Horvath C M, Polley R, et al. Activation of interferon regulatory factor 3 is inhibited by the influenza A virus NS1 protein. J Virol, 2000, 74: 7989–7996, 1:CAS:528:DC%2BD3cXlsl2gu7c%3D, 10933707, 10.1128/JVI.74.17.7989-7996.2000
Ehrhardt C, Seyer R, Hrincius E R, et al. Interplay between influenza A virus and the innate immune signaling. Microbes Infect, 2010, 12: 81–87, 1:CAS:528:DC%2BC3cXosFKr, 19782761, 10.1016/j.micinf.2009.09.007
Guo Z, Chen L M, Zeng H, et al. NS1 protein of influenza A virus inhibits the function of intracytoplasmic pathogen sensor, RIG-I. Am J Respir Cell Mol Biol, 2007, 36: 263–269, 1:CAS:528:DC%2BD2sXjtlWjuro%3D, 17053203, 10.1165/rcmb.2006-0283RC
Mibayashi M, Martinez-Sobrido L, Loo Y M, et al. Inhibition of retinoic acid-inducible gene I-mediated induction of beta interferon by the NS1 protein of influenza A virus. J Virol, 2007, 81: 514–524, 1:CAS:528:DC%2BD2sXms1ShsA%3D%3D, 17079289, 10.1128/JVI.01265-06
Opitz B, Rejaibi A, Dauber B, et al. IFNbeta induction by influenza A virus is mediated by RIG-I which is regulated by the viral NS1 protein. Cell Microbiol, 2007, 9: 930–938, 1:CAS:528:DC%2BD2sXktFCmsLg%3D, 17140406, 10.1111/j.1462-5822.2006.00841.x
Gack M U, Albrecht R A, Urano T, et al. Influenza A virus NS1 targets the ubiquitin ligase TRIM25 to evade recognition by the host viral RNA sensor RIG-I. Cell Host Microbe, 2009, 5: 439–449, 1:CAS:528:DC%2BD1MXosVClu7s%3D, 19454348, 10.1016/j.chom.2009.04.006
Nemeroff M E, Barabino S M L, Li Y, et al. Influenza virus NS1 protein interacts with the cellular 30 kDa subunit of CPSF and inhibits 3′ end formation of cellular pre-mRNAs. Mol Cell, 1998, 1: 991–1000, 1:CAS:528:DyaK1cXktlKktr4%3D, 9651582, 10.1016/S1097-2765(00)80099-4
Conenello G M, Palese P. Influenza A virus PB1-F2: A small protein with a big punch. Cell Host Microbe, 2007, 2: 207–209, 1:CAS:528:DC%2BD2sXht1CntLvO, 18005736, 10.1016/j.chom.2007.09.010
Mitzner D, Dudek S E, Studtrucker N, et al. Phosphorylation of the influenza A virus protein PB1-F2 by PKC is crucial for apoptosis promoting functions in monocytes. Cell Microbiol, 2009, 11: 1502–1516, 1:CAS:528:DC%2BD1MXhtFOgs7%2FK, 19523156, 10.1111/j.1462-5822.2009.01343.x
Zamarin D, Garcia-Sastre A, Xiao X, et al. Influenza virus PB1-F2 protein induces cell death through mitochondrial ANT3 and VDAC1. PLoS Pathog, 2005, 1: e4, 16201016, 10.1371/journal.ppat.0010004, 1:CAS:528:DC%2BD2MXhtFGls7rP
McAuley J L, Hornung F, Boyd K L, et al. Expression of the 1918 influenza A virus PB1-F2 enhances the pathogenesis of viral and secondary bacterial pneumonia. Cell Host Microbe, 2007, 2: 240–249, 1:CAS:528:DC%2BD2sXht1CntLvE, 18005742, 10.1016/j.chom.2007.09.001
Zamarin D, Ortigoza M B, Palese P. Influenza A virus PB1-F2 protein contributes to viral pathogenesis in mice. J Virol, 2006, 80: 7976–7983, 1:CAS:528:DC%2BD28Xot1yrtrY%3D, 16873254, 10.1128/JVI.00415-06
Mazur I, Wurzer W J, Ehrhardt C, et al. Acetylsalicylic acid (ASA) blocks influenza virus propagation via its NF-kB-inhibiting activity. Cell Microbiol, 2007, 9: 1683–1694, 1:CAS:528:DC%2BD2sXnslyhsrc%3D, 17324159, 10.1111/j.1462-5822.2007.00902.x
Toker A. Signaling through protein kinase C. Front Biosci, 1998, 3: D1134–1147, 1:CAS:528:DyaK1cXntlKns7k%3D, 9792904
Hoffmann H H, Palese P, Shaw M L. Modulation of influenza virus replication by alteration of sodium ion transport and protein kinase C activity. Antiviral Res, 2008, 80: 124–134, 1:CAS:528:DC%2BD1cXht1ensLjE, 18585796, 10.1016/j.antiviral.2008.05.008
Kunzelmann K, Beesley A H, King N J, et al. Influenza virus inhibits amiloride-sensitive Na+ channels in respiratory epithelia. Proc Natl Acad Sci USA, 2000, 97: 10282–10287, 1:CAS:528:DC%2BD3cXmtlehtLg%3D, 10920189, 10.1073/pnas.160041997
Lazrak A, Iles K E, Liu G, et al. Influenza virus M2 protein inhibits epithelial sodium channels by increasing reactive oxygen species. FASEB J, 2009, 23: 3829–3842, 1:CAS:528:DC%2BD1MXhtlymtrzI, 19596899, 10.1096/fj.09-135590
Sieczkarski S B, Whittaker G R. Characterization of the host cell entry of filamentous influenza virus. Arch Virol, 2005, 150: 1783–1796, 1:CAS:528:DC%2BD2MXos1eksL8%3D, 15959836, 10.1007/s00705-005-0558-1
Constantinescu S N, Cernescu C D, Popescu L M. Effects of protein kinase C inhibitors on viral entry and infectivity. FEBS Lett, 1991, 292: 31–33, 1:CAS:528:DyaK38XhsFCjuw%3D%3D, 1659999, 10.1016/0014-5793(91)80826-O
Arora D J, Gasse N. Influenza virus hemagglutinin stimulates the protein kinase C activity of human polymorphonuclear leucocytes. Arch Virol, 1998, 143: 2029–2037, 1:CAS:528:DyaK1cXnvFyjur0%3D, 9856090, 10.1007/s007050050439
Root C N, Wills E G, McNair L L, et al. Entry of influenza viruses into cells is inhibited by a highly specific protein kinase C inhibitor. J Gen Virol, 2000, 81: 2697–2705, 1:CAS:528:DC%2BD3cXnvFWgurY%3D, 11038382
Sieczkarski S B, Brown H A, Whittaker G R. Role of protein kinase C betaII in influenza virus entry via late endosomes. J Virol, 2003, 77: 460–469, 1:CAS:528:DC%2BD3sXkt1Cn, 12477851, 10.1128/JVI.77.1.460-469.2003
Pleschka S. RNA viruses and the mitogenic Raf/MEK/ERK signal transduction cascade. Biol Chem, 2008, 389: 1273–1282, 1:CAS:528:DC%2BD1cXht1ekt7%2FK, 18713014, 10.1515/BC.2008.145
Huang X, Liu T, Muller J, et al. Effect of influenza virus matrix protein and viral RNA on ribonucleoprotein formation and nuclear export. Virology, 2001, 287: 405–416, 1:CAS:528:DC%2BD3MXmsVGiurs%3D, 11531417, 10.1006/viro.2001.1067
Sakaguchi A, Hirayama E, Hiraki A, et al. Nuclear export of influenza viral ribonucleoprotein is temperature-dependently inhibited by dissociation of viral matrix protein. Virology, 2003, 306: 244–253, 1:CAS:528:DC%2BD3sXitVKmtrY%3D, 12642098, 10.1016/S0042-6822(02)00013-2
Ludwig S, Wolff T, Ehrhardt C, et al. MEK inhibition impairs influenza B virus propagation without emergence of resistant variants. FEBS Letters, 2004, 561: 37–43, 1:CAS:528:DC%2BD2cXhvFagu7w%3D, 15013748, 10.1016/S0014-5793(04)00108-5
Pleschka S, Wolff T, Ehrhardt C, et al. Influenza virus propagation is impaired by inhibition of the Raf/MEK/ERK signalling cascade. Nat Cell Biol, 2001, 3: 301–305, 1:CAS:528:DC%2BD3MXitFemsLw%3D, 11231581, 10.1038/35060098
Marjuki H, Alam M I, Ehrhardt C, et al. Membrane accumulation of influenza A virus hemagglutinin triggers nuclear export of the viral genome via protein kinase Calpha-mediated activation of ERK signaling. J Biol Chem, 2006, 281: 16707–16715, 1:CAS:528:DC%2BD28XlsV2lt7Y%3D, 16608852, 10.1074/jbc.M510233200
Marjuki H, Yen H L, Franks J, et al. Higher polymerase activity of a human influenza virus enhances activation of the hemagglutinin-induced Raf/MEK/ERK signal cascade. Virol J, 2007, 4: 134, 18053252, 10.1186/1743-422X-4-134, 1:CAS:528:DC%2BD1cXjvVWlsrs%3D
Beaton A R, Krug R M. Selected host cell capped RNA fragments prime influenza viral RNA transcription in vivo. Nucleic Acids Res, 1981, 9: 4423–4436, 1:CAS:528:DyaL38Xks1yhtQ%3D%3D, 7301581, 10.1093/nar/9.17.4423
Krug R M. Priming of influenza viral RNA transcription by capped heterologous RNAs. Curr Top Microbiol Immunol, 1981, 93: 125–149, 1:CAS:528:DyaL3MXmtFOgtLY%3D, 6269803
Chen Z, Li Y, Krug R M. Influenza A virus NS1 protein targetspoly( A)-binding protein II of the cellular 3[prime]-end processing machinery. EMBO J, 1999, 18: 2273–2283, 1:CAS:528:DyaK1MXivVOmsrc%3D, 10205180, 10.1093/emboj/18.8.2273
Lu Y, Qian X Y, Krug R M. The influenza virus NS1 protein: a novel inhibitor of pre-mRNA splicing. Genes Dev, 1994, 8: 1817–1828, 1:CAS:528:DyaK2cXlsFWntrs%3D, 7958859, 10.1101/gad.8.15.1817
Garfinkel M S, Katze M G. Translational control by influenza virus. Selective translation is mediated by sequences within the viral mRNA 5′-untranslated region. J Biol Chem, 1993, 268: 22223–22226, 1:CAS:528:DyaK3sXmsVOjsL8%3D, 8226725
de la Luna S, Fortes P, Beloso A, et al. Influenza virus NS1 protein enhances the rate of translation initiation of viral mRNAs. J Virol, 1995, 69: 2427–2433, 7884890
Enami K, Sato T A, Nakada S, et al. Influenza virus NS1 protein stimulates translation of the M1 protein. J Virol, 1994, 68: 1432–1437, 1:CAS:528:DyaK2cXitlygu7k%3D, 7508995
Katze M G, DeCorato D, Krug R M. Cellular mRNA translation is blocked at both initiation and elongation after infection by influenza virus or adenovirus. J Virol, 1986, 60: 1027–1039, 1:CAS:528:DyaL2sXhs1Gjug%3D%3D, 3023655
Park Y W, Katze M G. Translational control by influenza virus. Identification of cis-acting sequences and trans-acting factors which may regulate selective viral mRNA translation. J Biol Chem, 1995, 270: 28433–28439, 1:CAS:528:DyaK2MXps1ahtLk%3D, 7499349, 10.1074/jbc.270.47.28433
Aragon T, de la Luna S, Novoa I, et al. Eukaryotic translation initiation factor 4GI is a cellular target for NS1 protein, a translational activator of influenza virus. Mol Cell Biol, 2000, 20: 6259–6268, 1:CAS:528:DC%2BD3cXlvFegurY%3D, 10938102, 10.1128/MCB.20.17.6259-6268.2000
Burgui I, Aragon T, Ortin J, et al. PABP1 and eIF4GI associate with influenza virus NS1 protein in viral mRNA translation initiation complexes. J Gen Virol, 2003, 84: 3263–3274, 1:CAS:528:DC%2BD3sXpvFartLg%3D, 14645908, 10.1099/vir.0.19487-0
Feigenblum D, Schneider R J. Modification of eukaryotic initiation factor 4F during infection by influenza virus. J Virol, 1993, 67: 3027–3035, 1:CAS:528:DyaK3sXks12gt74%3D, 8098776
Katze M G. Regulation of the interferon-induced PKR: can viruses cope? Trends in Microbiology, 1995, 3: 75–78, 1:STN:280:DyaK2M3ksFKjtA%3D%3D, 7537157, 10.1016/S0966-842X(00)88880-0
Goodman A G, Smith J A, Balachandran S, et al. The cellular protein P58IPK regulates influenza virus mRNA translation and replication through a PKR-Mediated mechanism. J Virol, 2007, 81: 2221–2230, 1:CAS:528:DC%2BD2sXitVOgsr4%3D, 17166899, 10.1128/JVI.02151-06
Hatada E, Hasegawa M, Mukaigawa J, et al. Control of influenza virus gene expression: quantitative analysis of each viral RNA species in infected cells. J Biochem, 1989, 105: 537–546, 1:CAS:528:DyaL1MXitV2itLY%3D, 2760014
Shapiro G I, Gurney T, Jr Krug R M. Influenza virus gene expression: control mechanisms at early and late times of infection and nuclearcytoplasmic transport of virus-specific RNAs. J Virol, 1987, 61: 764–773, 1:CAS:528:DyaL2sXhtFKgsb4%3D, 3806797
Yamanaka K, Nagata K, Ishihama A. Temporal control for translation of influenza virus mRNAs. Arch Virol, 1991, 120: 33–42, 1:CAS:528:DyaK38XhvVWqtQ%3D%3D, 1929878, 10.1007/BF01310947
Duronio V. The life of a cell: apoptosis regulation by the PI3K/PKB pathway. Biochem J, 2008, 415: 333–344, 1:CAS:528:DC%2BD1cXht1CmsrnP, 18842113, 10.1042/BJ20081056
Franke T F, Hornik C P, Segev L, et al. PI3K/Akt and apoptosis: size matters. Oncogene, 2003, 22: 8983–8998, 1:CAS:528:DC%2BD3sXpsFOmsrg%3D, 14663477, 10.1038/sj.onc.1207115
Ji W T, Liu H J. PI3K-Akt signaling and viral infection. Recent Pat Biotechnol, 2008, 2: 218–226, 1:CAS:528:DC%2BD1cXhtlOgtrzN, 19097333, 10.2174/187220808786241042
Hale B G, Batty I H, Downes C P, et al. Binding of influenza A virus NS1 protein to the inter-SH2 domain of p85 suggests a novel mechanism for phosphoinositide 3-kinase activation. J Biol Chem, 2008, 283: 1372–1380, 1:CAS:528:DC%2BD1cXks1ektA%3D%3D, 18029356, 10.1074/jbc.M708862200
Hale B G, Jackson D, Chen Y H, et al. Influenza A virus NS1 protein binds p85beta and activates phosphatidylinositol-3-kinase signaling. Proc Natl Acad Sci USA, 2006, 103: 14194–14199, 1:CAS:528:DC%2BD28XhtVClsr3L, 16963558, 10.1073/pnas.0606109103
Hale B G, Randall R E. PI3K signalling during influenza A virus infections. Biochem Soc Trans, 2007, 35: 186–187, 1:CAS:528:DC%2BD2sXjt1Wrs7o%3D, 17371234, 10.1042/BST0350186
Shin Y K, Li Y, Liu Q, et al. SH3 binding motif 1 in influenza A virus NS1 protein is essential for PI3K/Akt signaling pathway activation. J Virol, 2007, 81: 12730–12739, 1:CAS:528:DC%2BD2sXhtl2jtb%2FJ, 17881440, 10.1128/JVI.01427-07
Shin Y K, Liu Q, Tikoo S K, et al. Influenza A virus NS1 protein activates the phosphatidylinositol 3-kinase (PI3K)/Akt pathway by direct interaction with the p85 subunit of PI3K. J Gen Virol, 2007, 88: 13–18, 1:CAS:528:DC%2BD2sXmslegtQ%3D%3D, 17170431, 10.1099/vir.0.82419-0
Ehrhardt C, Wolff T, Pleschka S, et al. Influenza A virus NS1 protein activates the PI3K/Akt pathway to mediate antiapoptotic signaling responses. J Virol, 2007, 81: 3058–3067, 1:CAS:528:DC%2BD2sXjslWrtbk%3D, 17229704, 10.1128/JVI.02082-06
Zhirnov O P, Klenk H D. Control of apoptosis in influenza virus-infected cells by up-regulation of Akt and p53 signaling. Apoptosis, 2007, 12: 1419–1432, 1:CAS:528:DC%2BD2sXmvVyqurs%3D, 17468837, 10.1007/s10495-007-0071-y
Lu X Y, Masic A, Li Y, et al. The PI3K/Akt pathway inhibits influenza A virus-induced Bax-mediated apoptosis by negatively regulating the JNK pathway via ASK1. J Gen Virol, 2010, 91: 1439–1449, 1:CAS:528:DC%2BC3cXnsFKhtb4%3D, 20130137, 10.1099/vir.0.018465-0
Ehrhardt C, Ludwig S. A new player in a deadly game: influenza viruses and the PI3K/Akt signalling pathway. Cell Microbiol, 2009, 11: 863–871, 1:CAS:528:DC%2BD1MXmslShtbs%3D, 19290913, 10.1111/j.1462-5822.2009.01309.x
Ehrhardt C, Marjuki H, Wolff T, et al. Bivalent role of the phosphatidylinositol-3-kinase (PI3K) during influenza virus infection and host cell defence. Cell Microbiol, 2006, 8: 1336–1348, 1:CAS:528:DC%2BD28XptlKgt7c%3D, 16882036, 10.1111/j.1462-5822.2006.00713.x
Shin Y K, Liu Q, Tikoo S K, et al. Effect of the phosphatidylinositol 3-kinase/Akt pathway on influenza A virus propagation. J Gen Virol, 2007, 88: 942–950, 1:CAS:528:DC%2BD2sXivVyqurc%3D, 17325368, 10.1099/vir.0.82483-0
Flory E, Kunz M, Scheller C, et al. Influenza virus-induced NF-kappaB-dependent gene expression is mediated by overexpression of viral proteins and involves oxidative radicals and activation of IkappaB kinase. J Biol Chem, 2000, 275: 8307–8314, 1:CAS:528:DC%2BD3cXit1entL8%3D, 10722660, 10.1074/jbc.275.12.8307
Tenoever B R, Ng S L, Chua M A, et al. Multiple functions of the IKK-related kinase IKKepsilon in interferon-mediated antiviral immunity. Science, 2007, 315: 1274–1278, 1:CAS:528:DC%2BD2sXitFChsrc%3D, 17332413, 10.1126/science.1136567
Pahl H L, Baeuerle P A. Expression of influenza virus hemagglutinin activates transcription factor NF-kappa B. J Virol, 1995, 69: 1480–1484, 1:CAS:528:DyaK2MXjslemt7s%3D, 7853480
Gern J E, French D A, Grindle K A, et al. Double-stranded RNA induces the synthesis of specific chemokines by bronchial epithelial cells. Am J Respir Cell Mol Biol, 2003, 28: 731–737, 1:CAS:528:DC%2BD3sXks1Kit7k%3D, 12600836, 10.1165/rcmb.2002-0055OC
Severa M, Fitzgerald K A. TLR-mediated activation of type I IFN during antiviral immune responses: fighting the battle to win the war. Curr Top Microbiol Immunol, 2007, 316: 167–192, 1:CAS:528:DC%2BD1cXhs12rt7c%3D, 17969448, 10.1007/978-3-540-71329-6_9
Nimmerjahn F, Dudziak D, Dirmeier U, et al. Active NF-kappaB signalling is a prerequisite for influenza virus infection. J Gen Virol, 2004, 85: 2347–2356, 1:CAS:528:DC%2BD2cXmvVKhtrY%3D, 15269376, 10.1099/vir.0.79958-0
Kumar N, Xin Z T, Liang Y, et al. NF-kappaB signaling differentially regulates influenza virus RNA synthesis. J Virol, 2008, 82: 9880–9889, 1:CAS:528:DC%2BD1cXht1amtr3J, 18701591, 10.1128/JVI.00909-08
Wurzer W J, Ehrhardt C, Pleschka S, et al. NF-kappaB-dependent induction of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and Fas/FasL is crucial for efficient influenza virus propagation. J Biol Chem, 2004, 279: 30931–30937, 1:CAS:528:DC%2BD2cXlslOktL0%3D, 15143063, 10.1074/jbc.M403258200
Berghall H, Siren J, Sarkar D, et al. The interferon-inducible RNA helicase, mda-5, is involved in measles virus-induced expression of antiviral cytokines. Microbes Infection, 2006, 8: 2138–2144, 10.1016/j.micinf.2006.04.005, 1:CAS:528:DC%2BD28XpsVOlsb4%3D
Liu P, Jamaluddin M, Li K, et al. Retinoic acid-inducible gene I mediates early antiviral response and Toll-like receptor 3 expression in respiratory syncytial virus-infected airway epithelial cells. J Virol, 2007, 81: 1401–1411, 1:CAS:528:DC%2BD2sXhtFCnsr0%3D, 17108032, 10.1128/JVI.01740-06
Thompson A J, Locarnini S A. Toll-like receptors, RIG-I-like RNA helicases and the antiviral innate immune response. Immunol Cell Biol, 2007, 85: 435–445, 1:CAS:528:DC%2BD2sXpvFWgur8%3D, 17667934, 10.1038/sj.icb.7100100
Pothlichet J, Chignard M, Si-Tahar M. Cutting edge: innate immune response triggered by influenza A virus is negatively regulated by SOCS1 and SOCS3 through a RIG-I/IFNAR1-dependent pathway. J Immunol, 2008, 180: 2034–2038, 1:CAS:528:DC%2BD1cXhsVSht7w%3D, 18250407
Rintahaka J, Wiik D, Kovanen P E, et al. Cytosolic antiviral RNA recognition pathway activates caspases 1 and 3. J Immunol, 2008, 180: 1749–1757, 1:CAS:528:DC%2BD1cXotlKiuw%3D%3D, 18209072
Karlas A, Machuy N, Shin Y, et al. Genome-wide RNAi screen identifies human host factors crucial for influenza virus replication. Nature, 2010, 463: 818–822, 1:CAS:528:DC%2BC3cXntVCltQ%3D%3D, 20081832, 10.1038/nature08760
Konig R, Stertz S, Zhou Y, et al. Human host factors required for influenza virus replication. Nature, 2010, 463: 813–817, 20027183, 10.1038/nature08699, 1:CAS:528:DC%2BC3cXhvVKrtb4%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Dai, X., Zhang, L. & Hong, T. Host cellular signaling induced by influenza virus. Sci. China Life Sci. 54, 68–74 (2011). https://doi.org/10.1007/s11427-010-4116-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4116-z