Abstract
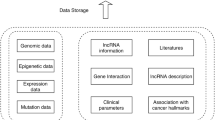
The functional impact of several long intergenic non-coding RNAs (lincRNAs) has been characterized in previous studies. However, it is difficult to identify lincRNAs on a large-scale and to ascertain their functions or predict their structures in laboratory experiments because of the diversity, lack of knowledge and specificity of expression of lincRNAs. Furthermore, although there are a few well-characterized examples of lincRNAs associated with cancers, these are just the tip of the iceberg owing to the complexity of cancer. Here, by combining RNA-Seq data from several kinds of human cell lines with chromatin-state maps and human expressed sequence tags, we successfully identified more than 3000 human lincRNAs, most of which were new ones. Subsequently, we predicted the functions of 105 lincRNAs based on a coding-non-coding gene co-expression network. Finally, we propose a genetic mediator and key regulator model to unveil the subtle relationships between lincRNAs and lung cancer. Twelve lincRNAs may be principal players in lung tumorigenesis. The present study combines large-scale identification and functional prediction of human lincRNAs, and is a pioneering work in characterizing cancer-associated lincRNAs by bioinformatics.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Guttman M, Amit I, Garber M, et al. Chromatin signature reveals over a thousand highly conserved large non-coding RNAs in mammals. Nature, 2009, 458: 223–227
Khalil A M, Guttman M, Huarte M, et al. Many human large intergenic noncoding RNAs associate with chromatin-modifying complexes and affect gene expression. Proc Natl Acad Sci USA, 2009, 106: 11667–11672
Cabili M N, Trapnell C, Goff L, et al. Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes Dev, 2011, 25: 1915–1927
Trapnell C, Williams B A, Pertea G, et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol, 2010, 28: 511–515
Guttman M, Garber M, Levin J Z, et al. Ab initio reconstruction of cell type-specific transcriptomes in mouse reveals the conserved multi-exonic structure of lincRNAs. Nat Biotechnol, 2010, 28: 503–510
Sone M, Hayashi T, Tarui H, et al. The mRNA-like noncoding RNA Gomafu constitutes a novel nuclear domain in a subset of neurons. J Cell Sci, 2007, 120: 2498–2506
Mercer T R, Dinger M E, Sunkin S M, et al. Specific expression of long noncoding RNAs in the mouse brain. Proc Natl Acad Sci USA, 2008, 105: 716–721
Mercer T R, Dinger M E, Mattick J S. Long non-coding RNAs: insights into functions. Nat Rev Genet, 2009, 10: 155–159
Wilusz J E, Sunwoo H, Spector D L. Long noncoding RNAs: functional surprises from the RNA world. Genes Dev, 2009, 23: 1494–1504
Yamasaki C, Murakami K, Fujii Y, et al. The H-Invitational Database (H-InvDB), a comprehensive annotation resource for human genes and transcripts. Nucleic Acids Res, 2008, 36: D793–799
Harrow J, Denoeud F, Frankish A, et al. GENCODE: producing a reference annotation for ENCODE. Genome Biol, 2006, 7: S41–49
Pruitt K D, Tatusova T, Maglott D R. NCBI reference sequences (RefSeq): a curated non-redundant sequence database of genomes, transcripts and proteins. Nucleic Acids Res, 2007, 35: D61–65
Bono H, Kasukawa T, Furuno M, et al. FANTOM DB: database of Functional Annotation of RIKEN Mouse cDNA Clones. Nucleic Acids Res, 2002, 30: 116–118
Guttman M, Amit I, Garber M, et al. Chromatin signature reveals over a thousand highly conserved large non-coding RNAs in mammals. Nature, 2009, 458: 223–227
Khalil A, Guttman M, Huarte M, et al. Many human large intergenic noncoding RNAs associate with chromatin-modifying complexes and affect gene expression. Proc Natl Acad Sci USA, 2009, 106: 11667
Guttman M, Garber M, Levin J Z, et al. Ab initio reconstruction of cell type-specific transcriptomes in mouse reveals the conserved multi-exonic structure of lincRNAs. Nat Biotechnol, 2010, 28: 503–510
Cabili M N, Trapnell C, Goff L, et al. Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes Dev, 2011, 25: 1915–1927
Rinn J, Kertesz M, Wang J, et al. Functional demarcation of active and silent chromatin domains in human HOX loci by noncoding RNAs. Cell, 2007, 129: 1311–1323
Nagano T, Mitchell J A, Sanz L A, et al. The Air noncoding RNA epigenetically silences transcription by targeting G9a to chromatin. Science, 2008, 322: 1717–1720
Pandey R R, Mondal T, Mohammad F, et al. Kcnq1ot1 antisense noncoding RNA mediates lineage-specific transcriptional silencing through chromatin-level regulation. Mol Cell, 2008, 32: 232–246
Huarte M, Guttman M, Feldser D, et al. A large intergenic noncoding RNA induced by p53 mediates global gene repression in the p53 response. Cell, 2010, 142: 409–419
Liao Q, Liu C, Yuan X, et al. Large-scale prediction of long non-coding RNA functions in a coding-non-coding gene co-expression network. Nucleic Acids Res, 2011, 39: 3864–3878
Gupta R A, Shah N, Wang K C, et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature, 2010, 464: 1071–1076
Huarte M, Rinn J L. Large non-coding RNAs: missing links in cancer? Hum Mol Genet, 2010, 19: R152–161
Chen G, Wang Z, Wang D, et al. LncRNADisease: a database for long noncoding RNA associated diseases. Nucleic Acids Res, 2013, 41: D983–986
Bernstein B E, Birney E, Dunham I, et al. An integrated encyclopedia of DNA elements in the human genome. Nature, 2012, 489: 57–74
Fejes A P, Robertson G, Bilenky M, et al. FindPeaks 3.1: a tool for identifying areas of enrichment from massively parallel short-read sequencing technology. Bioinformatics, 2008, 24: 1729–1730
Kong L, Zhang Y, Ye Z Q, et al. CPC: assess the protein-coding potential of transcripts using sequence features and support vector machine. Nucleic Acids Res, 2007, 35: W345–349
Dunham I, Kundaje A, Aldred S F, et al. An integrated encyclopedia of DNA elements in the human genome. Nature, 2012, 489: 57–74
Zhang B, Horvath S. A general framework for weighted gene co-expression network analysis. Stat Appl Genet Mol Biol, 2005, 4: Article17
Nayak R R, Kearns M, Spielman R S, et al. Coexpression network based on natural variation in human gene expression reveals gene interactions and functions. Genome Res, 2009, 19: 1953–1962
Berkofsky-Fessler W, Nguyen T Q, Delmar P, et al. Preclinical biomarkers for a cyclin-dependent kinase inhibitor translate to candidate pharmacodynamic biomarkers in phase I patients. Mol Cancer Ther, 2009, 8: 2517–2525
Ergun A, Lawrence C A, Kohanski M A, et al. A network biology approach to prostate cancer. Mol Syst Biol, 2007, 3: 82
di Bernardo D, Thompson M J, Gardner T S, et al. Chemogenomic profiling on a genome-wide scale using reverse-engineered gene networks. Nat Biotechnol, 2005, 23: 377–383
Ding L, Getz G, Wheeler D A, et al. Somatic mutations affect key pathways in lung adenocarcinoma. Nature, 2008, 455: 1069–1075
Segal E, Friedman N, Koller D, et al. A module map showing conditional activity of expression modules in cancer. Nat Genet, 2004, 36: 1090–1098
Saijo T, Ishii G, Ochiai A, et al. Eg5 expression is closely correlated with the response of advanced non-small cell lung cancer to antimitotic agents combined with platinum chemotherapy. Lung Cancer, 2006, 54: 217–225
Okazaki Y, Furuno M, Kasukawa T, et al. Analysis of the mouse transcriptome based on functional annotation of 60770 full-length cDNAs. Nature, 2002, 420: 563–573
Metzker M L. Sequencing technologies—the next generation. Nat Rev Genet, 2010, 11: 31–46
Wang E T, Sandberg R, Luo S, et al. Alternative isoform regulation in human tissue transcriptomes. Nature, 2008, 456: 470–476
Mortazavi A, Williams B A, McCue K, et al. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods, 2008, 5: 621–628
Maher C A, Kumar-Sinha C, Cao X, et al. Transcriptome sequencing to detect gene fusions in cancer. Nature, 2009, 458: 97–101
Pan Q, Shai O, Lee L J, et al. Deep surveying of alternative splicing complexity in the human transcriptome by high-throughput sequencing. Nat Genet, 2008, 40: 1413–1415
Orom U A, Derrien T, Beringer M, et al. Long noncoding RNAs with enhancer-like function in human cells. Cell, 2010, 143: 46–58
Tripathi V, Ellis J D, Shen Z, et al. The nuclear-retained noncoding RNA MALAT1 regulates alternative splicing by modulating SR splicing factor phosphorylation. Mol Cell, 2010, 39: 925–938
Author information
Authors and Affiliations
Corresponding authors
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
Electronic supplementary material
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Sun, L., Luo, H., Liao, Q. et al. Systematic study of human long intergenic non-coding RNAs and their impact on cancer. Sci. China Life Sci. 56, 324–334 (2013). https://doi.org/10.1007/s11427-013-4460-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4460-x