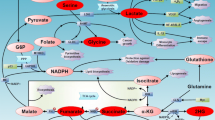
Abstract
Altered metabolism in cancer was first discovered by Otto Warburg early last century. Although the Warburg Effect has been widely used in tumor detection, relatively little progress had been made in mechanistic understanding of cancer metabolism in the subsequent eight decades. Genetic studies have recently identified mutations in human cancer targeting multiple enzymes involved in intermediate metabolism. One emerging mechanism common to these mutant enzymes is the accumulation of a metabolite that alters the epigenetic control.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Warburg O. On the origin of cancer cells. Science, 1956, 123: 309–314
Warburg O. Über den stoffwechsel der carcinomzelle. J Mol Med, 1925, 4: 534–536
Avril N, Dose J, Jänicke F, et al. Metabolic characterization of breast tumors with positron emission tomography using f-18 fluorodeoxyglucose. J Clin Oncol, 1996, 14: 1848–1857
de Santis M, Becherer A, Bokemeyer C, et al. 2-18fluoro-deoxy-d-glucose positron emission tomography is a reliable predictor for viable tumor in postchemotherapy seminoma: An update of the prospective multicentric sempet trial. J Clin Oncol, 2004, 22: 1034–1039
Oermann E K, Wu J, Guan K L, et al. Alterations of metabolic genes and metabolites in cancer. Semin Cell Dev Biol, 2012, 23: 370–380
Hsu P P, Sabatini D M. Cancer cell metabolism: Warburg and beyond. Cell, 2008, 134: 703–707
Vander Heiden M G, Cantley L C, Thompson C B. Understanding the warburg effect: The metabolic requirements of cell proliferation. Sci Signal, 2009, 324: 1029
Wise D R, Thompson C B. Glutamine addiction: A new therapeutic target in cancer. Trends Biochem Sci, 2010, 35: 427–433
Daye D, Wellen K E. Metabolic reprogramming in cancer: Unraveling the role of glutamine in tumorigenesis. Semin Cell Dev Biol, 2012, 23: 362–369
Le A, Lane A N, Hamaker M, et al. Glucose-independent glutamine metabolism via TCA cycling for proliferation and survival in B cells. Cell Metab, 2012, 15: 110–121
Metallo C M, Gameiro P A, Bell E L, et al. Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia. Nature, 2011, 481: 380–384
Mullen A R, Wheaton W W, Jin E S, et al. Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature, 2011, 481: 385–388
Wise D R, Ward P S, Shay J E, et al. Hypoxia promotes isocitrate dehydrogenase-dependent carboxylation of α-ketoglutarate to citrate to support cell growth and viability. Proc Natl Acad Sci USA, 2011, 108: 19611–19616
Fuchs B C, Bode B P. Amino acid transporters ASCT2 and LAT1 in cancer: Partners in crime? Semin Cancer Biol, 2005, 15: 254–266
Fuchs B C, Finger R E, Onan M C, et al. ASCT2 silencing regulates mammalian target-of-rapamycin growth and survival signaling in human hepatoma cells. Am J Physiol Cell Physiol, 2007, 293: C55–C63
Hu W, Zhang C, Wu R, et al. Glutaminase 2, a novel p53 target gene regulating energy metabolism and antioxidant function. Sci Signal, 2010, 107: 7455
Suzuki S, Tanaka T, Poyurovsky M V, et al. Phosphate-activated glutaminase (GLS2), a p53-inducible regulator of glutamine metabolism and reactive oxygen species. Sci Signal, 2010, 107: 7461
Hao H X, Khalimonchuk O, Schraders M, et al. SDH5, a gene required for flavination of succinate dehydrogenase, is mutated in paraganglioma. Science, 2009, 325: 1139–1142
Baysal B E, Ferrell R E, Willett-Brozick J E, et al. Mutations in SDHD, a mitochondrial complex II gene, in hereditary paraganglioma. Science, 2000, 287: 848–851
Gimm O, Armanios M, Dziema H, et al. Somatic and occult germline mutations in SDHD, a mitochondrial complex II gene, in nonfamilial pheochromocytoma. Cancer Res, 2000, 60: 6822–6825
McWhinney S R, Pasini B, Stratakis C A. Familial gastrointestinal stromal tumors and germ-line mutations. N Engl J Med, 2007, 357: 1054–1056
Ricketts C, Woodward E R, Killick P, et al. Germline SDHB mutations and familial renal cell carcinoma. J Nat Cancer Inst, 2008, 100: 1260–1262
Bardella C, Pollard P J, Tomlinson I. SDH mutations in cancer. Biochim Biophys Acta Bioenerg, 2011, 1807: 1432–1443
Timmers H J, Kozupa A, Eisenhofer G, et al. Clinical presentations, biochemical phenotypes, and genotype-phenotype correlations in patients with succinate dehydrogenase subunit B-associated pheochromocytomas and paragangliomas. J Clin Endocrinol Metab, 2007, 92: 779–786
Gimenez-Roqueplo A P, Favier J, Rustin P, et al. Mutations in the SDHB gene are associated with extra-adrenal and/or malignant phaeochromocytomas. Cancer Res, 2003, 63: 5615–5621
Amar L, Baudin E, Burnichon N, et al. Succinate dehydrogenase B gene mutations predict survival in patients with malignant pheochromocytomas or paragangliomas. J Clin Endocrinol Metab, 2007, 92: 3822–3828
Pollard P, Briere J, Alam N, et al. Accumulation of Krebs cycle intermediates and over-expression of hif1α in tumours which result from germline FH and SDH mutations. Hum Mol Genet, 2005, 14: 2231–2239
Xiao M, Yang H, Xu W, et al. Inhibition of α-kg-dependent histone and DNA demethylases by fumarate and succinate that are accumulated in mutations of FH and SDH tumor suppressors. Genes Dev, 2012, 26: 1326–1338
Tomlinson I P, Alam N A, Rowan A J, et al. Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nat Genet, 2002, 30: 406–410
Campione E, Terrinoni A, Orlandi A, et al. Cerebral cavernomas in a family with multiple cutaneous and uterine leiomyomas associated with a new mutation in the fumarate hydratase gene. J Invest Dermatol, 2007, 127: 2271–2273
Carvajal-Carmona L G, Alam N A, Pollard P J, et al. Adult leydig cell tumors of the testis caused by germline fumarate hydratase mutations. J Clin Endocrinol Metab, 2006, 91: 3071–3075
Ylisaukko-oja S K, Cybulski C, Lehtonen R, et al. Germline fumarate hydratase mutations in patients with ovarian mucinous cystadenoma. Eur J Hum Genet, 2006, 14: 880–883
Yang H, Ye D, Guan K L, et al. IDH1 and IDH2 mutations in tumorigenesis: Mechanistic insights and clinical perspectives. Clin Cancer Res, 2012, 18: 5562–5571
Parsons D W, Jones S, Zhang X, et al. An integrated genomic analysis of human glioblastoma multiforme. Science, 2008, 321: 1807–1812
Mardis E R, Ding L, Dooling D J, et al. Recurring mutations found by sequencing an acute myeloid leukemia genome. N Engl J Med, 2009, 361: 1058–1066
Balss J, Meyer J, Mueller W, et al. Analysis of the IDH1 codon 132 mutation in brain tumors. Acta Neuropathol, 2008, 116: 597–602
Bleeker F E, Lamba S, Leenstra S, et al. IDH1 mutations at residue p.R132 (idh1r132) occur frequently in high-grade gliomas but not in other solid tumors. Hum Mutat, 2009, 30: 7–11
Hartmann C, Meyer J, Balss J, et al. Type and frequency of IDH1 and IDH2 mutations are related to astrocytic and oligodendroglial differentiation and age: A study of 1010 diffuse gliomas. Acta Neuropathol, 2009, 118: 469–474
Watanabe T, Nobusawa S, Kleihues P, et al. IDH1 mutations are early events in the development of astrocytomas and oligodendrogliomas. Am J Pathol, 2009, 174: 1149–1153
Yan H, Parsons D W, Jin G, et al. IDH1 and IDH2 mutations in gliomas. N Engl J Med, 2009, 360: 765–773
Chou W C, Hou H A, Chen C Y, et al. Distinct clinical and biologic characteristics in adult acute myeloid leukemia bearing the isocitrate dehydrogenase 1 mutation. Blood, 2010, 115: 2749–2754
Dang L, Jin S, Su S M. IDH mutations in glioma and acute myeloid leukemia. Trends Mol Med, 2010, 16: 387–397
Gravendeel L A M, Kloosterhof N K, Bralten L B C, et al. Segregation of non-p.R132h mutations in IDH1 in distinct molecular subtypes of glioma. Hum Mutat, 2010, 31: E1186–E1199
Gross S, Cairns R A, Minden M D, et al. Cancer-associated metabolite 2-hydroxyglutarate accumulates in acute myelogenous leukemia with isocitrate dehydrogenase 1 and 2 mutations. J Exp Med, 2010, 207: 339–344
Ho P A, Alonzo T A, Kopecky K J, et al. Molecular alterations of the IDH1 gene in AML: A children’s oncology group and southwest oncology group study. Leukemia, 2010, 24: 909–913
Tefferi A, Lasho T L, Abdel-Wahab O, et al. IDH1 and IDH2 mutation studies in 1473 patients with chronic-, fibrotic- or blast-phase essential thrombocythemia, polycythemia vera or myelofibrosis. Leukemia, 2010, 24: 1302–1309
Wagner K, Damm F, Gohring G, et al. Impact of IDH1 R132 mutations and an IDH1 single nucleotide polymorphism in cytogenetically normal acute myeloid leukemia: SNP rs11554137 is an adverse prognostic factor. J Clin Oncol, 2010, 28: 2356–2364
Ward P S, Patel J, Wise D R, et al. The common feature of leukemia-associated IDH1 and IDH2 mutations is a neomorphic enzyme activity converting α-ketoglutarate to 2-hydroxyglutarate. Cancer Cell, 2010, 17: 225–234
Amary M F, Bacsi K, Maggiani F, et al. IDH1 and IDH2 mutations are frequent events in central chondrosarcoma and central and periosteal chondromas but not in other mesenchymal tumours. J Pathol, 2011, 224: 334–343
Amary M F, Damato S, Halai D, et al. Ollier disease and maffucci syndrome are caused by somatic mosaic mutations of IDH1 and IDH2. Nat Genet, 2011, 43: 1262–1265
Pansuriya T C, van Eijk R, d’Adamo P, et al. Somatic mosaic IDH1 and IDH2 mutations are associated with enchondroma and spindle cell hemangioma in ollier disease and maffucci syndrome. Nat Genet, 2011, 43: 1256–1261
Borger D R, Tanabe K K, Fan K C, et al. Frequent mutation of isocitrate dehydrogenase (IDH)1 and IDH2 in cholangiocarcinoma identified through broad-based tumor genotyping. Oncologist, 2011, 17: 72–79
Wang P, Dong Q, Zhang C, et al. Mutations in isocitrate dehydrogenase 1 and 2 occur frequently in intrahepatic cholangiocarcinomas and share hypermethylation targets with glioblastomas. Oncogene, 2013, 32: 3091–3100
Hemerly J P, Bastos A U, Cerutti J M. Identification of several novel non-p.R132 IDH1 variants in thyroid carcinomas. Eur J Endocrinol, 2010, 163: 747–755
Murugan A K, Bojdani E, Xing M. Identification and functional characterization of isocitrate dehydrogenase 1 (IDH1) mutations in thyroid cancer. Biochem Biophys Res Commun, 2010, 393: 555–559
Kang M R, Kim M S, Oh J E, et al. Mutational analysis of IDH1 codon 132 in glioblastomas and other common cancers. Int J Cancer, 2009, 125: 353–355
Gaal J, Burnichon N, Korpershoek E, et al. Isocitrate dehydrogenase mutations are rare in pheochromocytomas and paragangliomas. J Clin Endocrinol Metab, 2010, 95: 1274–1278
Pusch S S F, Meyer J, Mittelbronn M, et al. Glioma IDH1 mutation patterns off the beaten track. Neuropathol Appl Neurobiol, 2011, 37: 428–430
Zhao S, Lin Y, Xu W, et al. Glioma-derived mutations in IDH1 dominantly inhibit IDH1 catalytic activity and induce HIF-1α. Science’s STKE, 2009, 324: 261
Dang L, White D W, Gross S, et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature, 2009, 462: 739–744
Figueroa M E, Abdel-Wahab O, Lu C, et al. Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell, 2010, 18: 553–567
Ward P, Cross J, Lu C, et al. Identification of additional IDH mutations associated with oncometabolite R(−)-2-hydroxyglutarate production. Oncogene, 2012, 31: 2491–2498
Xu W, Yang H, Liu Y, et al. Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of α-ketoglutarate-dependent dioxygenases. Cancer Cell, 2011, 19: 17–30
Jalbert L, Elkhaled A, Phillips J, et al. Presence of 2-hydroxyglutarate in IDH1 mutated low-grade glioma using ex vivo proton hr-mas spectroscopy. Proc Intl Soc Mag Reson Med, 2011, 19: 183
Kalinina J C A, Wang L, Yu Q, et al. Detection of “oncometabolite” 2-hydroxyglutarate by magnetic resonance analysis as a biomarker of IDH1/2 mutations in glioma. J Mol Med (Berl), 2012, 90: 1161–1171
Hutton Jr J J, Kaplan A, Udenfriend S. Conversion of the amino acid sequence gly-pro-pro in protein to gly-pro-hyp by collagen proline hydroxylase. Arch Biochem Biophys, 1967, 121: 384–391
Rose N R, McDonough M A, King O N F, et al. Inhibition of 2-oxoglutarate dependent oxygenases. Chem Soc Rev, 2011, 40: 4364
Pollard P, Loenarz C, Mole D, et al. Regulation of Jumonji-domain-containing histone demethylases by hypoxia-inducible factor (HIF)-1alpha. Biochem J, 2008, 416: 387–394
Hausinger R P. FeII/alpha-ketoglutarate-dependent hydroxylases and related enzymes. Crit Rev Biochem Mol Biol, 2004, 39: 21–68
Loenarz C, Schofield C J. Expanding chemical biology of 2-oxoglutarate oxygenases. Nat Chem Biol, 2008, 4: 152–156
Tuderman L, Myllylä R, Kivirikko K I. Mechanism of the prolyl hydroxylase reaction. Eur J Biochem, 1977, 80: 341–348
Letouzé E, Martinelli C, Loriot C, et al. SDH mutations establish a hypermethylator phenotype in paraganglioma. Cancer Cell, 2013, 23: 739–752
Killian J K, Kim S Y, Miettinen M, et al. Succinate dehydrogenase mutation underlies global epigenomic divergence in gastrointestinal stromal tumor. Cancer Discovery, 2013, 3: 648–657
Mason E F, Hornick J L. Succinate dehydrogenase deficiency is associated with decreased 5-hydroxymethylcytosine production in gastrointestinal stromal tumors: Implications for mechanisms of tumorigenesis. Modern Pathol, 2013, doi: 10.1038/modpathol.2013.86
Chowdhury R, Yeoh K K, Tian Y M, et al. The oncometabolite 2-hydroxyglutarate inhibits histone lysine demethylases. EMBO Rep, 2011, 12: 463–469
Lu C, Ward P S, Kapoor G S, et al. IDH mutation impairs histone demethylation and results in a block to cell differentiation. Nature, 2012, 483: 474–478
Duncan C G, Barwick B G, Jin G, et al. A heterozygous IDH1R132H/WT mutation induces genome-wide alterations in DNA methylation. Genome Res, 2012, 22: 2339–2355
Sasaki M, Knobbe C B, Munger J C, et al. IDH1 (R132H) mutation increases murine haematopoietic progenitors and alters epigenetics. Nature, 2012, 488: 656–659
Kriaucionis S, Heintz N. The nuclear DNA base 5-hydroxyme-thylcytosine is present in purkinje neurons and the brain. Science, 2009, 324: 929–930
Tahiliani M, Koh K P, Shen Y, et al. Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by mll partner TET1. Science, 2009, 324: 930–935
Ito S, D’Alessio A C, Taranova O V, et al. Role of Tet proteins in 5mC to 5hmC conversion, ES-cell self-renewal and inner cell mass specification. Nature, 2010, 466: 1129–1133
He Y F, Li B Z, Li Z, et al. Tet-mediated formation of 5-carbo-xylcytosine and its excision by TDG in mammalian DNA. Science, 2011, 333: 1303–1307
Ito S, Shen L, Dai Q, et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science, 2011, 333: 1300–1303
Noushmehr H, Weisenberger D J, Diefes K, et al. Identification of a CPG island methylator phenotype that defines a distinct subgroup of glioma. Cancer Cell, 2010, 17: 510–522
Turcan S, Rohle D, Goenka A, et al. IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature, 2012, 483: 479–483
Hewitson K S, Liénard B M, McDonough M A, et al. Structural and mechanistic studies on the inhibition of the hypoxia-inducible transcription factor hydroxylases by tricarboxylic acid cycle intermediates. J Biol Chem, 2007, 282: 3293–3301
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Dr. Guan KunLiang is a professor at the Department of Pharmacology, University of California San Diego (2007-present). He received his B.S. degree (1982) from Hangzhou University, China. As a fellow of the second class of CUSBEA, he attended Purdue University and received both his Ph.D. (1989) and postdoctoral training at Purdue University. Dr. Guan started as an assistant Professor at the University of Michigan (1992) and was promoted to full Professor in 2000. He was later appointed as the Halvor Christensen Professor (2003) at the University of Michigan. He received the MacArthur Fellowship (1998), Scherling-Plaugh Award (1999), and Distinguished Alumni Award (2006) at Purdue University, and was elected to Fellow of AAAS (2011). Dr. Guan studies signaling mechanisms of organ size control and tumorigenesis, with particular focus on the mTOR and Hippo pathways.
Dr. Xiong Yue is currently the William. R. Kenan Professor at the Lineberger Comprehensive Cancer Center in University of North Carolina at Chapel Hill. He obtained his B.S. degree in molecular biology from Fudan University in 1982. As a fellow of the CUSBEA class, he joined the University of Rochester as a predoctoral student and then began his postdoctoral training in Dr. David Beach’s lab at Cold Spring Harbor Laboratory. Dr. Xiong started his own lab in the University of North Carolina at Chapel Hill in 1993. His research mainly focuses on the mechanisms of cell proliferation and tumorigenesis. In 2006, Dr. Xiong Yue together with Dr. Guan KunLiang started the Molecular Cell Biology (MCB) lab in Fudan University to study metabolism alterations in human diseases. Dr. Xiong received the Pew Scholar in the Biomedical Sciences (1995), AACR Gertrude B. Elion Cancer Research Award (1999) and was elected as an AAAS fellow (2011). Dr. Yang Hui is a doctoral student at Fudan University and is supervised by Drs. Xiong and Guan. He graduated from Huazhong University of Science and Technology in 2008 and joined the MCB lab at Fudan University. Dr. Yang’s Ph.D. research mainly focuses on the dynamic regulation of epigenetic modifications by metabolites. He received the Ray Wu Prize in 2012.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Yang, H., Xiong, Y. & Guan, K. Metabolic alteration in tumorigenesis. Sci. China Life Sci. 56, 1067–1075 (2013). https://doi.org/10.1007/s11427-013-4549-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4549-2