Abstract
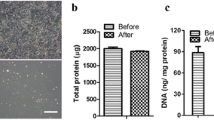
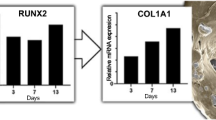
Seeding cells and scaffolds play pivotal roles in bone tissue engineering and regenerative medicine. Wharton’s jelly-derived mesenchymal stem cells (WJCs) from human umbilical cord represent attractive and promising seeding cells in tissue regeneration and engineering for treatment applications. This study was carried out to explore the biocompatibility of scaffolds to seeding cells in vitro. Rod-like nano-hydroxyapatite (RN-HA) and flake-like micro-hydroxyapatite (FM-HA) coatings were prepared on Mg-Zn-Ca alloy substrates using micro-arc oxidation and electrochemical deposition. WJCs were utilized to investigate the cellular biocompatibility of Mg-Zn-Ca alloys after different surface modifications by observing the cell adhesion, morphology, proliferation, and osteoblastic differentiation. The in vitro results indicated that the RN-HA coating group was more suitable for cell proliferation and cell osteoblastic differentiation than the FM-HA group, demonstrating better biocompatibility. Our results suggested that the RN-HA coating on Mg-Zn-Ca alloy substrates might be of great potential in bone tissue engineering.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Can A, Karahuseyinoglu S. Concise review: human umbilical cord stroma with regard to the source of fetus-derived stem cells. Stem Cells, 2007, 25: 2886–2895
Troyer DL, Weiss ML. Wharton’s jelly-derived cells are a primitive stromal cell population. Stem Cells, 2008, 26: 591–599
Witte F, Hort N, Vogt C, Cohen S, Kainer KU, Willumeit R, Feyerabend F. Degradable biomaterials based on magnesium corrosion. Curr Opin Solid St M, 2008, 12: 63–72
Zhang S, Zhang X, Zhao C, Li J, Song Y, Xie C, Tao H, Zhang Y, He Y, Jiang Y, Bian Y. Research on an Mg-Zn alloy as a degradable biomaterial. Acta Biomater, 2010, 6: 626–640
Zheng YF, Gu XN, Xi YL, Chai DL. In vitro degradation and cytotoxicity of Mg/Ca composites produced by powder metallurgy. Acta Biomater, 2010, 6: 1783–1791
Lowenstam HA, Weiner S. On biomineralization. Oxford: Oxford University Press, 1989
McConnell D. The crystal structure of bone. Clin Orthop, 1962, 23: 253–268
Huang Y, Qu Y, Yang B, Li W, Zhang B, Zhang X. In vivo biological responses of plasma sprayed hydroxyapatite coatings with an electric polarized treatment in alkaline solution. Mater Sci Eng C, 2009, 29: 2411–2416
Gao J, Guan S, Chen J, Wang L, Zhu S, Hu J, Ren Z. Fabrication and characterization of rod-like nano-hydroxyapatite on mao coating supported on Mg-Zn-Ca alloy. Appl Surf Sci, 2011, 257: 2231–2237
Gao J, Shi X, Yang B, Hou S, Meng E, Guan F, Guan S. Fabrication and characterization of bioactive composite coatings on Mg-Zn-Ca alloy by MAO/sol-gel. J Mater Sci: Mater in Med, 2011, 22: 1681–1687
Dorozhkin SV. Nanosized and nanocrystalline calcium orthopho-sphates. Acta Biomater, 2010, 6: 715–734
Bose S, Dasgupta S, Tarafder S, Bandyopadhyay A. Microwave-processed nanocrystalline hydroxyapatite: Simultaneous enhancement of mechanical and biological properties. Acta Biomater, 2010, 6: 3782–3790
Lu YP, Chen YM, Li ST, Wang JH. Surface nanocrystallization of hydroxyapatite coating. Acta Biomater, 2008, 4: 1865–1872
Wang J, Shaw LL. Nanocrystalline hydroxyapatite with simultaneous enhancements in hardness and toughness. Biomaterials, 2009, 30: 6565–6572
Li J, Yin Y, Yao F, Zhang L, Yao K. Effect of nano-and micro-hydroxyapatite/chitosan-gelatin network film on human gastric cancer cells. Mater Lett, 2008, 62: 3220–3223
Roy M, Bandyopadhyay A, Bose S. Induction plasma sprayed nano hydroxyapatite coatings on titanium for orthopaedic and dental implants. Surf Coat Technol, 2011, 205: 2785–2792
Li W, Guan S, Chen J, Hu J, Chen S, Wang L, Zhu S. Preparation and in vitro degradation of the composite coating with high adhesion strength on biodegradable Mg-Zn-Ca alloy. Mater Charact, 2011, 62: 1158–1165
Matsumoto T, Okazaki M, Inoue M, Yamaguchi S, Kusunose T, Toyonaga T, Hamada Y, Takahashi J. Hydroxyapatite particles as a controlled release carrier of protein. Biomaterials, 2004, 25: 3807–3812
Okada S, Ito H, Nagai A, Komotori J, Imai H. Adhesion of osteoblast-like cells on nanostructured hydroxyapatite. Acta Biomater, 2010, 6: 591–597
Kandori K, Saito M, Saito H, Yasukawa A, Ishikawa T. Adsorption of protein on non-stoichiometric calcium-strontium hydroxyapatite. Colloid Surface A, 1995, 94: 225–230
Sharpe J, Sammons R, Marquis P. Effect of pH on protein adsorption to hydroxyapatite and tricalcium phosphate ceramics. Biomaterials, 1997, 18: 471–476
Kang MI, Lee WY, Oh KW, Han JH, Song KH, Cha BY, Lee KW, Son HY, Kang SK, Kim CC. The short-term changes of bone mineral metabolism following bone marrow transplantation. Bone, 2000, 26: 275–279
Marom R, Shur I, Solomon R, Benayahu D. Characterization of adhesion and differentiation markers of osteogenic marrow stromal cells. J Cell Physiol, 2005, 202: 41–48
Stucki U, Schmid J, Hammerle CF, Lang NP. Temporal and local appearance of alkaline phosphatase activity in early stages of guided bone regeneration. A descriptive histochemical study in humans. Clin Oral Implant Res, 2001, 12: 121–127
Elias KL, Price RL, Webster TJ. Enhanced functions of osteoblasts on nanometer diameter carbon fibers. Biomaterials, 2002, 23: 3279–3287
Webster TJ, Ergun C, Doremus RH, Siegel RW, Bizios R. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials, 2000, 21: 1803–1810
Aubin JE. Advances in the osteoblast lineage. Biochem Cell Biol, 1998, 76: 899–910
Franceschi RT. The developmental control of osteoblast-specific gene expression: Role of specific transcription factors and the extracellular matrix environment. Crit Rev in Oral Biol Med, 1999, 10: 40–57
Knabe C, Berger G, Gildenhaar R, Klar F, Zreiqat H. The modulation of osteogenesis in vitro by calcium titanium phosphate coatings. Biomaterials, 2004, 25: 4911–4919
Morinobu M, Ishijima M, Rittling SR, Tsuji K, Yamamoto H, Nifuji A, Denhardt DT, Noda M. Osteopontin expression in osteoblasts and osteocytes during bone formation under mechanical stress in the calvarial suture in vivo. J Bone Miner Res, 2003, 18: 1706–1715
Satomura K, Krebsbach P, Bianco P, Gehron Robey P. Osteogenic imprinting upstream of marrow stromal cell differentiation. J Cell Biochem, 2000, 78: 391–403
Sodek J, Zhang Q, Goldberg H, Domenicucci C, Kasugai S, Wrana J, Shapiro H, Chen J. Non-collagenous bone proteins and their role in substrate-induced bioactivity. In: Davies JE, eds. The Bone-Biomaterial Interface. Toronto: University of Toronto Press, 1991. 97–110
Frank O, Heim M, Jakob M, Barbero A, Schafer D, Bendik I, Dick W, Heberer M, Martin I. Real-time quantitative RT-PCR analysis of human bone marrow stromal cells during osteogenic differentiation in vitro. J Cell Biochem, 2002, 85: 737–746
Vitolo MI, Anglin IE, Mahoney WM Jr., Renoud KJ, Gartenhaus RB, Bachman KE, Passaniti A. The RUNX2 transcription factor cooperates with the YES-associated protein, YAP65, to promote cell transformation. Cancer Biol Ther, 2007, 6: 856–863
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at springerlink.fh-diploma.de
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Guan, F., Ma, S., Shi, X. et al. Biocompatibility of nano-hydroxyapatite/Mg-Zn-Ca alloy composite scaffolds to human umbilical cord mesenchymal stem cells from Wharton’s jelly in vitro. Sci. China Life Sci. 57, 181–187 (2014). https://doi.org/10.1007/s11427-014-4610-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4610-9