Abstract
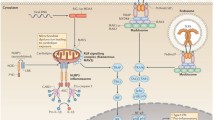
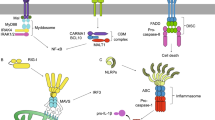
Innate immunity offers the first line of defense against infections and other types of danger such as tumorigenesis. Its discovery provides tremendous therapeutic opportunities for numerous human diseases. Delving into the structural basis of signal transduction by innate immune receptors, our lab has recently helped to establish the new paradigm in which innate immune receptors transduce ligand-binding signals through formation of higher-order assemblies containing intracellular adapters, signaling enzymes and their substrates. These large signalosome assemblies may be visible under light microscopy as punctate structures in the µm scale, connecting to the underlying molecular structures in the nm scale. They drive proximity-induced enzyme activation, and provide a mechanism for signaling amplification by nucleated polymerization. These supramolecular signaling complexes also open new questions on their cellular organization and mode of regulation, pose challenges to our methodology, and afford valuable implications in drug discovery against these medically important pathways.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Coley WB. The treatment of malignant tumors by repeated inoculations of erysipelas: with a report of ten original cases. Am J Med Sci, 1893, 105: 487–511
Janeway CA Jr. Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb Symp Quant Biol, 1989, 54 Pt 1: 1–13
Poltorak A, He X, Smirnova I, Liu MY, Huffel CV, Du X, Birdwell D, Alejos E, Silva M, Galanos C, Freudenberg M, Ricciardi-Castagnoli P, Layton B, Beutler B. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science, 1998, 282: 2085–2088
Medzhitov R, Preston-Hurlburt P, Janeway CA Jr. A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature, 1997, 388: 394–397
Medzhitov R, Janeway C Jr. Innate immune recognition: mechanisms and pathways. Immunol Rev, 2000, 173: 89–97
Medzhitov R, Janeway CA Jr. Innate immune recognition and control of adaptive immune responses. Semin Immunol, 1998, 10: 351–353
Iwasaki A, Medzhitov R. Regulation of adaptive immunity by the innate immune system. Science, 2011, 327: 291–295
Medzhitov R, Janeway CA. An ancient system of host defense. Curr Opin Immunol, 1998, 10: 12–15
Yin Q, Fu TM, Li J, Wu H. Structural biology of innate immunity. Ann Rev Immunol, 2015, 33: 393–416
Netea MG, Wijmenga C, O’Neill LA. Genetic variation in Toll-like receptors and disease susceptibility. Nat Immunol, 2012, 13: 535–542
Song DH, Lee JO. Sensing of microbial molecular patterns by Toll-like receptors. Immunol Rev, 2012, 250: 216–229
Rawling DC, Pyle AM. Parts, assembly and operation of the RIG-I family of motors. Curr Opin Struct Biol, 2014, 25: 25–33
Mason DR, Beck PL, Muruve DA. Nucleotide-binding oligomerization domain-like receptors and inflammasomes in the pathogenesis of non-microbial inflammation and diseases. J Innate Immun, 2012, 4: 16–30
Vance RE. The NAIP/NLRC4 inflammasomes. Curr Opin Immunol, 2015, 32: 84–89
Barber GN. Innate immune DNA sensing pathways: STING, AIMII and the regulation of interferon production and inflammatory responses. Curr Opin Immunol, 2011, 23: 10–20
Loo YM, Gale M Jr. Immune signaling by RIG-I-like receptors. Immunity, 2011, 34: 680–692
Roberts TL, Idris A, Dunn JA, Kelly GM, Burnton CM, Hodgson S, Hardy LL, Garceau V, Sweet MJ, Ross IL, Hume DA, Stacey KJ. HIN-200 proteins regulate caspase activation in response to foreign cytoplasmic DNA. Science, 2009, 323: 1057–1060
Henao-Mejia J, Elinav E, Strowig T, Flavell RA. Inflammasomes: far beyond inflammation. Nat Immunol, 2012, 13: 321–324
Rathinam VA, Vanaja SK, Fitzgerald KA. Regulation of inflammasome signaling. Nat Immunol, 2012, 13: 333–332
Ting JP, Duncan JA, Lei Y. How the noninflammasome NLRs function in the innate immune system. Science, 2010, 327: 286–290
Sun L, Wu J, Du F, Chen X, Chen ZJ. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science, 2013, 339: 786–791
Thome M. CARMA1, BCL-10 and MALT1 in lymphocyte development and activation. Nat Rev Immunol, 2004, 4: 348–359
Blonska M, Lin X. CARMA1-mediated NF-kappaB and JNK activation in lymphocytes. Immunol Rev, 2009, 228: 199–211
Lin SC, Lo YC, Wu H. Helical assembly in the MyD88-IRAK4-IRAK2 complex in TLR/IL-1R signalling. Nature, 2010, 465: 885–890
Tartaglia LA, Ayres TM, Wong GH, Goeddel DV. A novel domain within the 55 kd TNF receptor signals cell death. Cell, 1993, 74: 845–853
Ferrao R, Wu H. Helical assembly in the death domain (DD) superfamily. Curr Opin Struct Biol, 2012, 22: 241–247
Park HH, Logette E, Raunser S, Cuenin S, Walz T, Tschopp J, Wu H. Death domain assembly mechanism revealed by crystal structure of the oligomeric PIDDosome core complex. Cell, 2007, 128: 533–546
Wang L, Yang JK, Kabaleeswaran V, Rice AJ, Cruz AC, Park AY, Yin Q, Damko E, Jang SB, Raunser S, Robinson CV, Siegel RM, Walz T, Wu H. The Fas-FADD death domain complex structure reveals the basis of DISC assembly and disease mutations. Nat Struct Mol Biol, 2010, 17: 1324–1329
Hou F, Sun L, Zheng H, Skaug B, Jiang QX, Chen ZJ. MAVS forms functional prion-like aggregates to activate and propagate antiviral innate immune response. Cell, 2011, 146: 448–461
Qiao Q, Yang C, Zheng C, Fontan L, David L, Yu X, Bracken C, Rosen M, Melnick A, Egelman EH, Wu H. Structural architecture of the CARMA1/Bcl10/MALT1 signalosome: nucleation-induced filamentous assembly. Mol Cell, 2013, 51: 766–779
Lu A, Magupalli VG, Ruan J, Yin Q, Atianand MK, Vos MR, Schroder GF, Fitzgerald KA, Wu H, Egelman EH. Unified polymerization mechanism for the assembly of ASC-dependent inflammasomes. Cell, 2014, 156: 1193–1206
Wu B, Peisley A, Tetrault D, Li Z, Egelman EH, Magor KE, Walz T, Penczek PA, Hur S. Molecular imprinting as a signal-activation mechanism of the viral RNA sensor RIG-I. Mol Cell, 2014, 55: 511–523
Xu H, He X, Zheng H, Huang LJ, Hou F, Yu Z, de la Cruz MJ, Borkowski B, Zhang X, Chen ZJ, Jiang QX. Structural basis for the prion-like MAVS filaments in antiviral innate immunity. eLife, 2014, 3: e01489
Wu H. Higher-order assemblies in a new paradigm of signal transduction. Cell, 2013, 153: 287–292
Li J, McQuade T, Siemer AB, Napetschnig J, Moriwaki K, Hsiao YS, Damko E, Moquin D, Walz T, McDermott A, Chan FK, Wu H. The RIP1/RIP3 necrosome forms a functional amyloid signaling complex required for programmed necrosis. Cell, 2012, 150: 339–350
Lenz G, Davis RE, Ngo VN, Lam L, George TC, Wright GW, Dave SS, Zhao H, Xu W, Rosenwald A, Ott G, Muller-Hermelink HK, Gascoyne RD, Connors JM, Rimsza LM, Campo E, Jaffe ES, Delabie J, Smeland EB, Fisher RI, Chan WC, Staudt LM. Oncogenic CARD11 mutations in human diffuse large B cell lymphoma. Science, 2008, 319: 1676–1679
Cai X, Chen J, Xu H, Liu S, Jiang QX, Halfmann R, Chen ZJ. Prion-like polymerization underlies signal transduction in antiviral immune defense and inflammasome activation. Cell, 2014, 156: 1207–1222
Granier S, Kobilka B. A new era of GPCR structural and chemical biology. Nat Chem Biol, 2012, 8: 670–673
Tay S, Hughey JJ, Lee TK, Lipniacki T, Quake SR, Covert MW. Single-cell NF-kappaB dynamics reveal digital activation and analogue information processing. Nature, 2010, 466: 267–271
Kagan JC, Magupalli VG, Wu H. Supramolecular organizing centres: location-specific higher-order signalling complexes that control innate immunity. Nat Rev Immunol, 2014, 14: 821–826
Kagan JC, Medzhitov R. Phosphoinositide-mediated adaptor recruitment controls Toll-like receptor signaling. Cell, 2006, 125: 943–955
Bonham KS, Orzalli MH, Hayashi K, Wolf AI, Glanemann C, Weninger W, Iwasaki A, Knipe DM, Kagan JC. A promiscuous lipid-binding protein diversifies the subcellular sites of toll-like receptor signal transduction. Cell, 2014, 156: 705–716
Seth RB, Sun L, Ea CK, Chen ZJ. Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-kappaB and IRF 3. Cell, 2005, 122: 669–682
Horner SM, Liu HM, Park HS, Briley J, Gale M Jr. Mitochondrial-associated endoplasmic reticulum membranes (MAM) form innate immune synapses and are targeted by hepatitis C virus. Proc Natl Acad Sci USA, 2011, 108: 14590–14595
Dixit E, Boulant S, Zhang Y, Lee AS, Odendall C, Shum B, Hacohen N, Chen ZJ, Whelan SP, Fransen M, Nibert ML, Superti-Furga G, Kagan JC. Peroxisomes are signaling platforms for antiviral innate immunity. Cell, 2010, 141: 668–681
Paul S, Kashyap AK, Jia W, He YW, Schaefer BC. Selective Autophagy of the Adaptor Protein Bcl10 Modulates T Cell Receptor Activation of NF-kappaB. Immunity, 2012
Rossman JS, Stoicheva NG, Langel FD, Patterson GH, Lippincott-Schwartz J, Schaefer BC. POLKADOTS are foci of functional interactions in T-Cell receptor-mediated signaling to NF-kappaB. Mol Biol Cell, 2006, 17: 2166–2176
Sahillioglu AC, Sumbul F, Ozoren N, Haliloglu T. Structural and dynamics aspects of ASC speck assembly. Structure, 2014, 22: 1722–1734
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at springerlink.fh-diploma.de
Wu Hao received her pre-medical training at Peking University from 1982 to 1985 and studied Medicine at Peking Union Medical College from 1985 to 1988. She obtained her Ph.D. degree in Biochemistry from Purdue University in 1992 and performed postdoctoral training at Columbia University. She became an Assistant Professor at Weill Cornell Medical College in 1997 and was promoted to Professor in 2003. In 2012, she moved to Harvard Medical School as Asa and Patricia Springer Professor of Biological Chemistry and Molecular Pharmacology, and as Senior Investigator in the Program in Cellular and Molecular Medicine at Boston Children’s Hospital. Dr. Wu Hao has received a number of honors, including Howard Hughes Medical Institute pre-doctoral fellowship, Aaron Diamond postdoctoral fellowship, Pew Scholar award, Rita Allen Scholar award, New York Mayor’s Award for Excellence in Science and Technology, Margaret Dayhoff Memorial Award from the Biophysical Society, and Purdue University Distinguished Science Alumni Award. She was elected to the National Academy of Sciences in 2015..
Qiao Qi completed his B.S. degree in the prestigious Yuanpei Program at Peking University and earned his Ph.D. degree with Dr. Xu Rui-Ming at Institute of Biophysics, Chinese Academy of Sciences. In 2012, he joined Dr. Wu Hao’s laboratory as a postdoctoral fellow. In his postdoctoral training, he focused on the molecular mechanism of signal transduction in innate and adaptive immune responses. In 2013, Dr. Qiao Qi was awarded the Cancer Research Institute postdoctoral fellowship to support his mechanistic studies on immunity and cancer.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Qiao, Q., Wu, H. Supramolecular organizing centers (SMOCs) as signaling machines in innate immune activation. Sci. China Life Sci. 58, 1067–1072 (2015). https://doi.org/10.1007/s11427-015-4951-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4951-z