Abstract
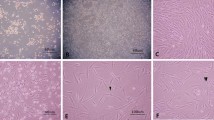
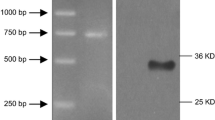
Under certain experimental conditions, bone marrow mesenchymal stem cells (MSCs) express neuronal phenotypes and neuronal markers, which suggests that they could be used to treat various neurological diseases. In the present study, MSCs were isolated from adult rat bone marrow, cultivated, and evaluated for neurotrophin expression profiles, as well as the potential to differentiate into functional neuronal-like cells in vitro. MSCs from passage 5 were pre-induced with DMEM/F12 medium containing 10% fetal bovine serum (FBS) and 10 ng/mL bFGF (fibroblast growth factor-2). Subsequently, a chemical inductor containing Dimethyl Sulphoxide (DMSO), Butylated Hydroxyanisole (BHA) and forskolin were used to induce neural expression of MSCs. Expression patterns of nestin, NF-200, and GFAP at time points before and after induction were detected by immunofluorescence. Nerve Growth Factor (NGF), brain-derived neurotrophic factor (BDNF) expressions in MSCs were evaluated by RT-PCR. The whole-cell patch clamp technique was utilized to elucidate the electrical behavior of MSC before and after 24-h differentiation induction. Immunofluorescence analysis revealed that MSCs expressed nestin (57.1% ± 6.9%), but not NF-200 or GFAP. Following neural induction, the cells exhibited a neuronal-like appearance. Nestin and NF-200 expression was positive in the neuronal-like cells, but GFAP expression was negative. After 6-, 12- and 24-h induction, the ratio of nestin-positive cells was 96.5% ± 1.9%, 88.1% ± 5.4%, and 33.5% ± 5.4%. NF-200 positive cells were 90.1% ± 2.9%, 97.5% ± 1.3%, and 98.1% ± 1.6%, respectively. However, prior to induction, MSCs already expressed NGF and BDNF. With a stimulus impulse of 40 mV, the density of the transient outward K current was (9.95 ± 4.85) pA/pF (n = 9) and (328.50 ± 30.62) pA/pF (n = 9) before and after induction, and the density of transient calcium ion currents was (−0.059 ± 0.027) pA/pF (n = 7) and (−6.66 ± 0.50) pA/pF (n = 7), respectively. Transient outward potassium currents and calcium ions currents gradually increased following induction. In addition, MSCs isolated from bone marrow exhibited characteristics of neuronal progenitor cells and expressed neurotrophins. These cells exhibited the capacity to differentiate into functional neuronal-like cells in vitro. These results suggested that MSCs express high levels of nestin and could be utilized for therapeutic strategies to treat nervous system diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Grove J E, Bruscia E, Krause D S. Plasticity of bone marrow-derived stem cells. Stem Cells, 2004, 22: 487–500
Yuan J, Yu J X, Huang B, et al. Induction of corneal epithelial progenitors from bone-marrow mesenchymal stem cells of rhesus monkeys in vitro. Chinese Sci Bull, 2007, 52: 2216–2225
Lu Z Z, Wu Z Z, Zhang Q W et al. Notch signaling stimulates osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. Chinese Sci Bull, 2004, 49: 815–818
Deng W, Obrocka M, Fischer I, et al. In vitro differentiation of human marrow stromal cells into early progenitors of neural cells by conditions that increase intracellular cyclic AMP. Biochem Biophys Res Commun, 2001, 282: 148–152
Kopen G C, Prockop D J, Phinney D G. Marrow stromal cells migrate throughout forebrain and cerebellum, and they differentiate into astrocytes after injection into neonatal mouse brains. Proc Natl Acad Sci USA, 1999, 96: 10711–10716
Woodbury D, Schwarz E J, Prockop D J, et al. Adult rat and human bone marrow stromal cells differentiate into neurons. J Neurosci Res, 2000, 61: 364–370
Kim B J, Seo J H, Bubien J K, et al. Differentiation of adult bone marrow stem cells into neuroprogenitor cells in vitro. Neuroreport, 2002, 13: 1185–1188
Dezawa M, Kanno H, Hoshino M, et al. Specific induction of neuronal cells from bone marrow stromal cells and application for autologous transplantation. J Clin Invest, 2004, 113: 1701–1710
Brazelton T R, Rossi F M V, Keshet G I, et al. From marrow to brain: expression of neuronal phenotypes in adult mice. Science, 2000, 290: 1775–1779
Li Y, Chen J, Chopp M. Adult bone marrow transplantation after stroke in adultrats. Cell Transplant, 2001, 10: 31–40
Mahmood A, Lu D, Li Y, et al. Intracranial bone marrow transplantation after traumatic brain injury improving functional outcome in adult rats. J Neurosurg, 2001, 94: 589–595
Hofstetter C P, Schwarz E J, Hess D, et al. Marrow stromal cells form guiding strands in the injured spinal cord and promote recovery. Proc Natl Acad Sci USA, 2002, 99: 2199–2204
Mezey E, Chandross K J, Harta G, et al. Turning blood into brain: Cells bearing neuronal antigens generated in vivo from bone marrow. Science, 2000, 290: 1779–1782
Zhao L R, Duan W M, Reyes M, et al. Human bone marrow stem cells exhibit neural phenotypes and ameliorate neurological deficits after grafting into the ischemic brain of rats. Exp Neurol, 2002, 174: 11–20
Bertani N, Malatesta P, Volpi G, et al. Neurogenic potential of human mesenchymal stem cells revisited: Analysis by immunostaining, time-lapse video and microarray. J Cell Sci, 2005, 118: 3925–3936
Neuhuber B, Gallo G, Howard L, et al. Reevaluation of in vitro differentiation protocols for bone marrow stromal cells: Disruption of actin cytoskeleton induces rapid morphological changes and mimics neuronal phenotype. J Neurosci Res, 2004, 77: 192–204
Lu P, Blesch A, Tuszynski M H. Induction of bone marrow stromal cells to neurons: differentiation, transdifferentiation, or artifact? J Neurosci Res, 2004, 77: 174–191
Alvarez-Dolado M, Pardal R, Garcia-Verdugo J M, et al. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature, 2003, 425: 968–973
Coyne T M, Marcus A J, Woodbury D, et al. Marrow stromal cells transplanted to the adult brain are rejected by an inflammatory response and transfer donor labels to host neurons and glia. Stem Cells, 2006, 24: 2483–2492
Li Y, Chopp M, Chen J, et al. Intrastriatal transplantation of bone marrow nonhematopoietic cells improves functional recovery after stroke in adult mice. J Cereb Blood Flow MeTable, 2000, 20: 1311–1319
Deng Y B, Liu Y, Zhu W B, et al. The co-graft of human marrow stromal cells and embryo olfactory ensheating cells as a new approach to treat spinal cord injury in a rat model. Cytotherapy, 2008, 2: 1–14
Deng Y B, Liu X G, Liu Z G, et al. The effects of mesenchymal stem cells and controlling release GDNF on neurological structure and function after spinal cord injury in rhesus. Cytotherapy, 2006, 8: 210–214
Azizi S A, Stokes D, Augelli B J, et al. Engraftment and migration of human bone marrow stromal cells implanted in the brains of albino rats—similarities to astrocyte grafts. Proc Natl Acad Sci USA, 1998, 95: 3908–3913
Yaghoobi M M, Mowla S, Tarihi T, et al. Nucleostemin, a coordinator of self-renewal, is expressed in rat marrow stromal cells and turns off after induction of neural differentiation. Neurosci Lett, 2005, 390: 81–86
Nikpoor P, Mowla S J, Movahedin M, et al. CatSper gene expression in postnatal development of mouse testis and in subfertile men with deficient sperm motility. Hum Reprod, 2004, 19: 124–128
Hamill O P, Marty A, Neher E, et al. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch, 1981, 391: 85–100
Toma J G, Akhavan M, Fernandes K J, et al. Isolation of multipotent adult stem cells from the dermis of mammalian skin. Nat Cell Biol, 2001, 3: 778–784
Nunes M C, Roy N S, Keyoung H M, et al. Identification and isolation of multipotential neural progenitor cells from the subcortical white matter of the adult human brain. Nat Med, 2003, 9: 439–447
Yabe J T, Wang F S, Chylinski T, et al. Selective accumulation of the high molecular weight neurofilament subunit within the distal region of growing axonal neurites. Cell Motil Cytoskeleton, 2001, 50: 1–12
Chen X, Katakowski M, Li Y, et al. Human bone marrow stromal cell cultures conditioned by traumatic brain tissue extracts: Growth factor production. J Neurosci Res, 2002, 69: 687–691
Tondreau T, Lagneaux L, Dejeneffe M, et al. Bone marrow-derived mesenchymal stem cells already express specific neural proteins before any differentiation. Differentiation, 2004, 72: 319–326
Miles G B, Yohn D C, Wichterle H, et al. Functional properties of motoneurons derived from mouse embryonic stem cells. J Neurosci, 2004, 24: 7848–7858
Westerlund U, Moe M C, Varghese M, et al. Stem cells from the adult human brain develop into functional neurons in culture. Exp Cell Res, 2004, 289: 378–383
Jin K, Mao X O, Batteur S, et al. Induction of neuronal markers in bone marrow cells: Differential effects of growth factors and patterns of intracellular expression. Exp Neurol, 2003, 184: 78–89
Wislet-Gendebien S, Hans G, Leprince P, et al. Plasticity of cultured mesenchymal stem cells: switch from nestin-positive to excitable neuron-like phenotype. Stem Cells, 2005, 23: 392–402
Kehat I, Khimovich L, Caspi O, et al. Electromechanical integration of cardiomyocytes derived from human embryonic stem cells. Nat Biotechnol, 2004, 22: 1282–1289
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors contributed equally to this work
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Liu, Y., Ye, Z., Wang, Y. et al. Induction-dependent neural marker expression and electrophysiological characteristics of bone marrow mesenchymal stem cells that naturally express high levels of nestin. Chin. Sci. Bull. 56, 640–646 (2011). https://doi.org/10.1007/s11434-010-4310-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-010-4310-1