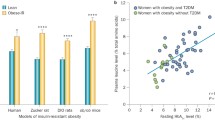
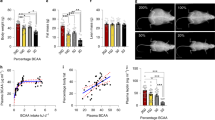
Abstract
Amino acids are fundamental nutrients required for protein synthesis. The branched chain amino acids (BCAAs) leucine, isoleucine, and valine are the most abundant of the essential amino acids. BCAAs have recently been recognized as having functions in processes other than simple nutrition. For example, metabolic diseases are characterized by higher levels of circulating BCAAs. Moreover, supplementation with or deficiency in BCAAs is closely related to the regulation of metabolic homeostasis. Indeed, leucine deprivation induces increased lipolysis and thermogenesis, which result in fat loss, as well as suppressed lipogenesis and enhanced insulin sensitivity in the liver. Accumulating evidence has indicated that several amino acid sensors, including GCN2, ATF4, mTOR, and AMPK, play pivotal roles in the regulation of lipid metabolism, glucose metabolism, and energy homeostasis. Furthermore, the hypothalamus is critical for sensing amino acid levels and mediates the metabolic adaptation of the body upon limitation of essential amino acids (EAAs) through regulating expression of the S6K1, MC4R, and CRH. In this review, we highlight recent studies investigating the cellular mechanisms linking amino acids, amino acid sensors, metabolic regulation, and metabolic diseases. Amino acid sensing and metabolic regulation have become research hotspots in the metabolic field.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wasan K M, Looije N A. Emerging pharmacological approaches to the treatment of obesity. J Pharm Pharm Sci, 2005, 8: 259–271
Wild S, Roglic G, Green A, et al. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care, 2004, 27: 1047–1053
Jia W P, Wang C, Jiang S, et al. Characteristics of obesity and its related disorders in China. Biomed Environ Sci, 2010, 23: 4–11
Yang W, Lu J, Weng J, et al. Prevalence of diabetes among men and women in China. N Engl J Med, 2010, 362: 1090–1101
Fan J G, Farrell G C. Epidemiology of non-alcoholic fatty liver disease in China. J Hepatol, 2009, 50: 204–210
Hofbauer K G. Molecular pathways to obesity. Int J Obes Relat Metab Disord, 2002, 26(Suppl 2): S18–27
Maejima Y, Iwasaki Y, Yamahara Y, et al. Peripheral oxytocin treatment ameliorates obesity by reducing food intake and visceral fat mass. Aging (Albany NY), 2011, 3: 1169–1177
Bruhat A, Jousse C, Fafournoux P. Amino acid limitation regulates gene expression. Proc Nutr Soc, 1999, 58: 625–632
Parker B, Noakes M, Luscombe N, et al. Effect of a high-protein, high-monounsaturated fat weight loss diet on glycemic control and lipid levels in type 2 diabetes. Diabetes Care, 2002, 25: 425–430
Layman D K, Boileau R A, Erickson D J, et al. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J Nutr, 2003, 133: 411–417
Layman D K, Walker D A. Potential importance of leucine in treatment of obesity and the metabolic syndrome. J Nutr, 2006, 136: 319S–323S
Felig P, Marliss E, Cahill G F Jr. Plasma amino acid levels and insulin secretion in obesity. N Engl J Med, 1969, 281: 811–816
Newgard C B, An J, Bain J R, et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab, 2009, 9: 311–326
Zhang Y, Guo K, LeBlanc R E, et al. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes, 2007, 56: 1647–1654
Nairizi A, She P, Vary T C, et al. Leucine supplementation of drinking water does not alter susceptibility to diet-induced obesity in mice. J Nutr, 2009, 139: 715–719
Guo F, Cavener D R. The GCN2 eIF2alpha kinase regulates fatty-acid homeostasis in the liver during deprivation of an essential amino acid. Cell Metab, 2007, 5: 103–114
Cota D, Proulx K, Smith K A, et al. Hypothalamic mTOR signaling regulates food intake. Science, 2006, 312: 927–930
Cheng Y, Meng Q, Wang C, et al. Leucine deprivation decreases fat mass by stimulation of lipolysis in white adipose tissue and upregulation of uncoupling protein 1 (UCP1) in brown adipose tissue. Diabetes, 2010, 59: 17–25
Cheng Y, Zhang Q, Meng Q, et al. Leucine deprivation stimulates fat loss via increasing CRH expression in the hypothalamus and activating the sympathetic nervous system. Mol Endocrinol, 2011, 25: 1624–1635
Elmquist J K, Coppari R, Balthasar N, et al. Identifying hypothalamic pathways controlling food intake, body weight, and glucose homeostasis. J Comp Neurol, 2005, 493: 63–71
Cypess A M, Lehman S, Williams G, et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med, 2009, 360: 1509–1517
Du Y, Meng Q, Zhang Q, et al. Isoleucine or valine deprivation stimulates fat loss via increasing energy expenditure and regulating lipid metabolism in WAT. Amino Acids, 2012, 43: 725–734
She P, Reid T M, Bronson S K, et al. Disruption of BCATm in mice leads to increased energy expenditure associated with the activation of a futile protein turnover cycle. Cell Metab, 2007, 6: 181–194
Wang T J, Larson M G, Vasan R S, et al. Metabolite profiles and the risk of developing diabetes. Nat Med, 2011, 17: 448–453
Muto Y, Sato S, Watanabe A, et al. Effects of oral branched-chain amino acid granules on event-free survival in patients with liver cirrhosis. Clin Gastroenterol Hepatol, 2005, 3: 705–713
Tremblay F, Krebs M, Dombrowski L, et al. Overactivation of S6 kinase 1 as a cause of human insulin resistance during increased amino acid availability. Diabetes, 2005, 54: 2674–2684
Xiao F, Huang Z, Li H, et al. Leucine deprivation increases hepatic insulin sensitivity via GCN2/mTOR/S6K1 and AMPK pathways. Diabetes, 2011, 60: 746–756
Iwanaka N, Egawa T, Satoubu N, et al. Leucine modulates contraction- and insulin-stimulated glucose transport and upstream signaling events in rat skeletal muscle. J Appl Physiol, 2010, 108: 274–282
Higuchi N, Kato M, Miyazaki M, et al. Potential role of branched-chain amino acids in glucose metabolism through the accelerated induction of the glucose-sensing apparatus in the liver. J Cell Biochem, 2011, 112: 30–38
Meijer A J, Sauerwein H P. Amino acid-dependent signal transduction and insulin sensitivity. Curr Opin Clin Nutr Metab Care, 1999, 2: 207–211
Wek S A, Zhu S, Wek R C. The histidyl-tRNA synthetase-related sequence in the eIF-2 alpha protein kinase GCN2 interacts with tRNA and is required for activation in response to starvation for different amino acids. Mol Cell Biol, 1995, 15: 4497–4506
Kilberg M S, Pan Y X, Chen H, et al. Nutritional control of gene expression: How mammalian cells respond to amino acid limitation. Annu Rev Nutr, 2005, 25: 59–85
Karpinski B A, Morle G D, Huggenvik J, et al. Molecular cloning of human CREB-2: An ATF/CREB transcription factor that can negatively regulate transcription from the cAMP response element. Proc Natl Acad Sci USA, 1992, 89: 4820–4824
Vallejo M, Ron D, Miller C P, et al. C/ATF, a member of the activating transcription factor family of DNA-binding proteins, dimerizes with CAAT/enhancer-binding proteins and directs their binding to cAMP response elements. Proc Natl Acad Sci USA, 1993, 90: 4679–4683
Carraro V, Maurin A C, Lambert-Langlais S, et al. Amino acid availability controls TRB3 transcription in liver through the GCN2/eIF2alpha/ATF4 pathway. PLoS One, 2010, 5: e15716
Kilberg M S, Shan J, Su N. ATF4-dependent transcription mediates signaling of amino acid limitation. Trends Endocrinol Metab, 2009, 20: 436–443
Averous J, Bruhat A, Jousse C, et al. Induction of CHOP expression by amino acid limitation requires both ATF4 expression and ATF2 phosphorylation. J Biol Chem, 2004, 279: 5288–5297
Harding H P, Novoa I, Zhang Y, et al. Regulated translation initiation controls stress-induced gene expression in mammalian cells. Mol Cell, 2000, 6: 1099–1108
Masuoka H C, Townes T M. Targeted disruption of the activating transcription factor 4 gene results in severe fetal anemia in mice. Blood, 2002, 99: 736–745
Mohamed H A, Yao W, Fioravante D, et al. cAMP-response elements in Aplysia creb1, creb2, and Ap-uch promoters: Implications for feedback loops modulating long term memory. J Biol Chem, 2005, 280: 27035–27043
Yang X, Matsuda K, Bialek P, et al. ATF4 is a substrate of RSK2 and an essential regulator of osteoblast biology; implication for Coffin-Lowry Syndrome. Cell, 2004, 117: 387–398
Cao J, Dai D L, Yao L, et al. Saturated fatty acid induction of endoplasmic reticulum stress and apoptosis in human liver cells via the PERK/ATF4/CHOP signaling pathway. Mol Cell Biochem, 2012, 364: 115–129
Ohoka N, Yoshii S, Hattori T, et al. TRB3, a novel ER stress-inducible gene, is induced via ATF4-CHOP pathway and is involved in cell death. EMBO J, 2005, 24: 1243–1255
Seo J, Fortuno E S, 3rd, Suh J M, et al. Atf4 regulates obesity, glucose homeostasis, and energy expenditure. Diabetes, 2009, 58: 2565–2573
Yoshizawa T, Hinoi E, Jung D Y, et al. The transcription factor ATF4 regulates glucose metabolism in mice through its expression in osteoblasts. J Clin Invest, 2009, 119: 2807–2817
Kode A, Mosialou I, Silva B C, et al. FoxO1 protein cooperates with ATF4 protein in osteoblasts to control glucose homeostasis. J Biol Chem, 2012, 287: 8757–8768
Wang C, Huang Z, Du Y, et al. ATF4 regulates lipid metabolism and thermogenesis. Cell Res, 2010, 20: 174–184
Li H, Meng Q, Xiao F, et al. ATF4 deficiency protects mice from high-carbohydrate-diet-induced liver steatosis. Biochem J, 2011, 438: 283–289
Wang C, Xia T, Du Y, et al. Effects of ATF4 on PGC1? expression in brown adipose tissue and metabolic responses to cold stress. Metabolism, 2012, doi:10.1016/j.metabol.2012.07.017
Wullschleger S, Loewith R, Hall M N. TOR signaling in growth and metabolism. Cell, 2006, 124: 471–484
Inoki K, Corradetti M N, Guan K L. Dysregulation of the TSC-mTOR pathway in human disease. Nat Genet, 2005, 37: 19–24
Lynch C J. Role of leucine in the regulation of mTOR by amino acids: Revelations from structure-activity studies. J Nutr, 2001, 131: 861S–865S
Harrington L S, Findlay G M, Gray A, et al. The TSC1-2 tumor suppressor controls insulin-PI3K signaling via regulation of IRS proteins. J Cell Biol, 2004, 166: 213–223
Carlson C J, White M F, Rondinone C M. Mammalian target of rapamycin regulates IRS-1 serine 307 phosphorylation. Biochem Biophys Res Commun, 2004, 316: 533–539
Ozes O N, Akca H, Mayo L D, et al. A phosphatidylinositol 3-kinase/Akt/mTOR pathway mediates and PTEN antagonizes tumor necrosis factor inhibition of insulin signaling through insulin receptor substrate-1. Proc Natl Acad Sci USA, 2001, 98: 4640–4645
Tremblay F, Brule S, Hee Um S, et al. Identification of IRS-1 Ser-1101 as a target of S6K1 in nutrient- and obesity-induced insulin resistance. Proc Natl Acad Sci USA, 2007, 104: 14056–14061
Draznin B. Molecular mechanisms of insulin resistance: Serine phosphorylation of insulin receptor substrate-1 and increased expression of p85alpha: The two sides of a coin. Diabetes, 2006, 55: 2392–2397
Pederson T M, Kramer D L, Rondinone C M. Serine/threonine phosphorylation of IRS-1 triggers its degradation: Possible regulation by tyrosine phosphorylation. Diabetes, 2001, 50: 24–31
Um S H, Frigerio F, Watanabe M, et al. Absence of S6K1 protects against age- and diet-induced obesity while enhancing insulin sensitivity. Nature, 2004, 431: 200–205
Krebs M, Brunmair B, Brehm A, et al. The Mammalian target of rapamycin pathway regulates nutrient-sensitive glucose uptake in man. Diabetes, 2007, 56: 1600–1607
Hardie D G, Ross F A, Hawley S A. AMPK: A nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol, 2012, 13: 251–262
Hardie D G, Sakamoto K. AMPK: A key sensor of fuel and energy status in skeletal muscle. Physiology (Bethesda), 2006, 21: 48–60
Jakobsen S N, Hardie D G, Morrice N, et al. 5′-AMP-activated protein kinase phosphorylates IRS-1 on Ser-789 in mouse C2C12 myotubes in response to 5-aminoimidazole-4-carboxamide riboside. J Biol Chem, 2001, 276: 46912–46916
Inoki K, Zhu T, Guan K L. TSC2 mediates cellular energy response to control cell growth and survival. Cell, 2003, 115: 577–590
Zhang B B, Zhou G, Li C. AMPK: An emerging drug target for diabetes and the metabolic syndrome. Cell Metab, 2009, 9: 407–416
Ruderman N, Prentki M. AMP kinase and malonyl-CoA: Targets for therapy of the metabolic syndrome. Nat Rev Drug Discov, 2004, 3: 340–351
Long Y C, Zierath J R. AMP-activated protein kinase signaling in metabolic regulation. J Clin Invest, 2006, 116: 1776–1783
Leclerc I, Rutter G A. AMP-activated protein kinase: A new beta-cell glucose sensor? Regulation by amino acids and calcium ions. Diabetes, 2004, 53(Suppl 3): S67–74
Harper A E, Benevenga N J, Wohlhueter R M. Effects of ingestion of disproportionate amounts of amino acids. Physiol Rev, 1970, 50: 428–558
Gietzen D W, Hao S, Anthony T G. Mechanisms of food intake repression in indispensable amino acid deficiency. Annu Rev Nutr, 2007, 27: 63–78
Leung P M, Rogers Q R. Importance of prepyriform cortex in food-intake response of rats to amino acids. Am J Physiol, 1971, 221: 929–935
Hao S, Ross-Inta C M, Gietzen D W. The sensing of essential amino acid deficiency in the anterior piriform cortex, that requires the uncharged tRNA/GCN2 pathway, is sensitive to wortmannin but not rapamycin. Pharmacol Biochem Behav, 2010, 94: 333–340
Williams G, Bing C, Cai X J, et al. The hypothalamus and the control of energy homeostasis: Different circuits, different purposes. Physiol Behav, 2001, 74: 683–701
Tomaszuk A, Simpson C, Williams G. Neuropeptide Y, the hypothalamus and the regulation of energy homeostasis. Horm Res, 1996, 46: 53–58
Shimazu T. The hypothalamus and metabolic regulation, especially hypothalamic regulation of lipid metabolism and its disorder. Tanpakushitsu Kakusan Koso, 1984, 29: 1589–1599
Blouet C, Schwartz G J. Hypothalamic nutrient sensing in the control of energy homeostasis. Behav Brain Res, 2010, 209: 1–12
Cota D, Proulx K, Seeley R J. The role of CNS fuel sensing in energy and glucose regulation. Gastroenterology, 2007, 132: 2158–2168
Xia T, Cheng Y, Zhang Q, et al. S6K1 in the central nervous system regulates energy expenditure via MC4R/CRH pathways in response to deprivation of an essential amino acid. Diabetes, 2012, 61: 2461–2471
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Wang, C., Guo, F. Branched chain amino acids and metabolic regulation. Chin. Sci. Bull. 58, 1228–1235 (2013). https://doi.org/10.1007/s11434-013-5681-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-5681-x