Abstract
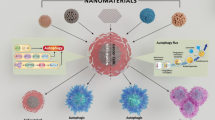
The biosafety issue of nanoscale materials is getting more and more attention with their increasing manufacture and application. In the research of cellular effects and underlying mechanisms related to toxicity of nanomaterials, most emphasis were placed on processes such as apoptosis, metabolic inhibition and oxidative stress. Recent evidence suggests that autophagy is part of the biological effects by nanomaterials and various kinds of nanomaterials are capable of disturbing the autophagic process. This review will highlight the importance of autophagy as an emerging mechanism of nanomaterial toxicity and the implication in the therapy of autophagy-related diseases. We summarize current research status of interaction between nanomaterials and autophagic pathways. It is of note that nanomaterials can either induce or block autophagy, which result in similar phenotype but completely different biological consequence. It is therefore important to perform comprehensive analysis of the whole autophagic flux in the future research.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lundqvist M, Stigler J, Elia G, et al. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. P Natl Acad Sci USA, 2008, 105: 14265–14270
Chomoucka J, Drbohlavova J, Huska D, et al. Magnetic nanoparticles and targeted drug delivering. Pharmacol Res, 2010, 62: 144–149
Hafeli U O, Riffle J S, Harris-Shekhawat L, et al. Cell uptake and in vitro toxicity of magnetic nanoparticles suitable for drug delivery. Mol Pharmaceut, 2009, 6: 1417–1428
Petrenko V A. Evolution of phage display: From bioactive peptides to bioselective nanomaterials. Expert Opin Drug Del, 2008, 5: 825–836
Aitken R, Chaudhry M, Boxall A, et al. Manufacture and use of nanomaterials: Current status in the UK and global trends. Occup Med-Oxford, 2006, 56: 300–306
Helland A, Wick P, Koehler A, et al. Reviewing the environmental and human health knowledge base of carbon nanotubes, Environ Health Persp, 2007, 15: 1125–1131
Marambio-Jones C, Hoek E M. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J Nanopart Res, 2010, 12: 1531–1551
Thomas K, Sayre P. Research strategies for safety evaluation of nanomaterials, Part I: Evaluating the human health implications of exposure to nanoscale materials, J Nanopart Res, 2005, 87: 316–321
Xia T, Kovochich M, Brant J, et al. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett, 2006, 6: 1794–1807
Li N, Venkatesan M I, Miguel A, et al. Induction of heme oxygenase-1 expression in macrophages by diesel exhaust particle chemicals and quinones via the antioxidant-responsive element. 1 expression in macrophages by diesel exhaust particle chemicals and quinones via the antioxidant-responsive element. J Immunol, 2000, 165: 3393–3401
Braydich-Stolle L K, Schaeublin N M, Murdock R C, et al. Crystal structure mediates mode of cell death in TiO2 nanotoxicity. J Nanopart Res, 2009, 11: 1361–1374
Donaldson K, Stone V, Tran C, et al. Nanotoxicology. Occup Environ Med, 2004, 61: 727–728
Stern S T, Johnson D N. Role for nanomaterial-autophagy interaction in neurodegenerative disease. Autophagy, 2008, 4: 1097–1100
Stern S, Adiseshaiah P, Crist R. Autophagy and lysosomal dysfunction as emerging mechanisms of nanomaterial toxicity. Part Fibre Toxicol, 2012, 9: 20
Hussain S, Al-Nsour F, Rice A B, et al. Cerium dioxide nano-particles induce apoptosis and autophagy in human peripheral blood monocytes. ACS Nano, 2012, 6: 5820–5829
Sohaebuddin S K, Thevenot P T, Baker D, et al. Nanomaterial cytotoxicity is composition, size, and cell type dependent. Part Fibre Toxicol, 2010, 7: 22
Zhang Q, Yang W, Man N, et al. Autophagy-mediated chemosensitization in cancer cells by fullerene C60 nanocrystal. Autophagy, 2009, 5: 1107–1117
He C, Klionsky D J. Regulation mechanisms and signaling pathways of autophagy. Annu Rev Genet, 2009, 43: 67
Mizushima N. Autophagy in protein and organelle turnover. Cold Spring Harb Symp Quant Biol, 2011, 76: 397–402
De Stefano D, Carnuccio R, Maiuri M C. Nanomaterials toxicity and cell death modalities. J Drug Deliv, 2012, 2012: 1–12
Mizushima N, Yoshimori T, Levine B. Methods in mammalian autophagy research. Cell, 2010, 140: 313–326
Zabirnyk O, Yezhelyev M, Seleverstov O. Nanoparticles as a novel class of autophagy activators. Autophagy, 2007, 3: 278–281
Li C, Liu H, Sun Y, et al. PAMAM nanoparticles promote acute lung injury by inducing autophagic cell death through the Akt-TSC2-mTOR signaling pathway. J Mol Cell Biol, 2009, 1: 37–45
Verma N K, Conroy J, Lyons P E, et al. Autophagy induction by silver nanowires: A new aspect in the biocompatibility assessment of nanocomposite thin films. Toxicol Appl Pharm, 2012, 264: 451–461
Sun T T, Yan Y W, Zhao Y, et al. Copper oxide nanoparticles induce autophagic cell death in A549 cells. PLoS One, 2012, 7: 1–7
De Duve C. The lysosome. Sci Am, 1963, 208: 64
Shintani T, Klionsky D J. Autophagy in health and disease: A double-edged sword. Science, 2004, 306: 990–995
Rabinowitz J D, White E. Autophagy and metabolism. Science, 2010, 330: 1344–1348
Degenhardt K, Mathew R, Beaudoin B, et al. Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell, 2006, 10: 51–64
Yu L, Strandberg L, Lenardo M J. The selectivity of autophagy and its role in cell death and survival. Autophagy, 2008, 4: 567–573
Khan M I, Mohammad A, Patil G, et al. Induction of ROS, mitochondrial damage and autophagy in lung epithelial cancer cells by iron oxide nanoparticles. Biomaterials, 2012, 33: 1477–1488
Bianco A, Kostarelos K, Prato M. Applications of carbon nanotubes in drug delivery. Curr Opin Chem Biol, 2005, 9: 674
Lin Y, Taylor S, Li H, et al. Advances toward bioapplications of carbon nanotubes. J Mater Chem, 2004, 14: 527–541
Wong S, Kam N, Dai H. Single walled carbon nanotubes for transport and delivery of biological cargos. Phys Status Solidi B, 2006, 243: 3561–3566
Bianco A, Kostarelos K, Prato M. Opportunities and challenges of carbon-based nanomaterials for cancer therapy. Expert Opin Drug Del, 2008, 5: 331–342
Yamawaki H, Iwai N. Cytotoxicity of water-soluble fullerene in vascular endothelial cells. Am J Physiol-Cell Ph, 2006, 290: C1495–C1502
Harhaji L, Isakovic A, Raicevic N, et al. Multiple mechanisms underlying the anticancer action of nanocrystalline fullerene. Eur J Pharmacol, 2007, 568: 89–98
Zhang Q, Yang W, Man N, et al. Autophagy-mediated chemosensitization in cancer cells by fullerene C60 nanocrystal. Autophagy, 2009, 5: 1107–1117
Wei P F, Zhang L, Lu Y, et al. C60(Nd) nanoparticles enhance chemotherapeutic susceptibility of cancer cells by modulation of autophagy. Nanotechnology, 2010, 21: 1–12
Johnson-Lyles D N, Peifley K, Lockett S, et al. Fullerenol cytotoxicity in kidney cells is associated with cytoskeleton disruption, autophagic vacuole accumulation, and mitochondrial dysfunction. Toxicol Appl Pharm, 2010, 248: 249–258
Lee C M, Huang S T, Huang S H, et al. C60 fullerene-pentoxifylline dyad nanoparticles enhance autophagy to avoid cytotoxic effects caused by the β-amyloid peptide. Nanomedicine, 2011, 7: 107–114
Liu H L, Zhang Y L, Yang N, et al. A functionalized single-walled carbon nanotube-induced autophagic cell death in human lung cells through Akt-TSC2-mTOR signaling. Cell Death Dis, 2011, 2: 1–7
Markovic Z M, Ristic B Z, Arsikin K M, et al. Graphene quantum dots as autophagy-inducing photodynamic agents. Biomaterials, 2012, 33: 7084–7092
Chen G Y, Yang H J, Lu C H, et al. Simultaneous induction of autophagy and toll-like receptor signaling pathways by graphene oxide. Biomaterials, 2012, 33: 6559–6569
Sharma C S, Sarkar S, Periyakaruppan A, et al. Single-walled carbon nanotubes induces oxidative stress in rat lung epithelial cells. J Nanosci Nanotechno, 2007, 7: 2466–2472
Panessa-Warren B, Warren J, Wong S, et al. Biological cellular response to carbon nanoparticle toxicity. J Phys-Condens Mat, 2006, 18: S2185–S2201
Bottini M, Bruckner S, Nika K, et al. Multi-walled carbon nanotubes induce T lymphocyte apoptosis. Toxicol Lett, 2006, 160: 121–126
Lam C W, James J T, McCluskey R, et al. Pulmonary toxicity of single-wall carbon nanotubes in mice 7 and 90 days after intratracheal instillation. J Nanopart Res, 2004, 77: 126–134
Feng L, Liu Z. Graphene in biomedicine: Opportunities and challenges. Nanomedicine, 2011, 6: 317–324
Chen H, Müller M B, Gilmore K J, et al. Mechanically strong, electrically conductive, and biocompatible graphene paper. Adv Mater, 2008, 20: 3557–3561
Wu Z S, Ren W, Gao L, et al. Synthesis of graphene sheets with high electrical conductivity and good thermal stability by hydrogen arc discharge exfoliation. ACS Nano, 2009, 3: 411–417
Christensen I L, Sun Y P, Juzenas P. Carbon dots as antioxidants and prooxidants. J Biomed Nanotechnol, 2011, 7: 667–676
Agostinis P, Berg K, Cengel K A, et al. Photodynamic therapy of cancer: An update. Ca-Cancer J Clin, 2011, 61: 250–281
Ichimura Y, Kumanomidou T, Sou Y, et al. Structural basis for sorting mechanism of p62 in selective autophagy. J Biol Chem, 2008, 283: 22847–22857
AshaRani P V, Mun G L K, Hande M P, et al. Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano, 2008, 3: 279–290
Navarro E, Piccapietra F, Wagner B, et al. Toxicity of silver nanoparticles to chlamydomonas reinhardtii. Environ Sci Technol, 2008, 42: 8959–8964
Li J J, Hartono D, Ong C N, et al. Autophagy and oxidative stress associated with gold nanoparticles. Biomaterials, 2010, 31: 5996–6003
Ma X, Wu Y, Jin S, et al. Gold nanoparticles induce autophagosome accumulation through size-dependent nanoparticle uptake and lysosome impairment. ACS Nano, 2011, 5: 8629–8639
Wu Y N, Yang L X, Shi X Y, et al. The selective growth inhibition of oral cancer by iron core-gold shell nanoparticles through mitochondria-mediated autophagy. Biomaterials, 2011, 32: 4565–4573
Blanka H K, Catherine C B, Seher G A, et al. Induction of oxidative stress, lysosome activation and autophagy by nanoparticles in human brain-derived endothelial cells. Biochem J, 2012, 441: 813–821
Julia X Y, Li T H, Yu J, et al. Distinct biological effects of different nanoparticles commonly used in cosmetics and medicine coatings. Cell Biosci, 2011, 1: 19
Li H, Li Y, Jiao J, et al. Alpha-alumina nanoparticles induce efficient autophagy-dependent cross-presentation and potent antitumour response. Nat Nanotechnol, 2011, 6: 645–650
Chen L, Zhang B, Toborek M. Autophagy is involved in nanoalumina-induced cerebrovascular toxicity. Nanomed-Nanotechnol, 2013, 9: 212–221
Afeseh N H, Kanthasamy A, Gu Y, et al. Manganese nanoparticle activates mitochondrial dependent apoptotic signaling and autophagy in dopaminergic neuronal cells. Toxicol Appl Pharm, 2011, 256: 227–240
Wei M, Chen N, Li J, et al. Polyvalent immunostimulatory nanoagents with self-assembled CpG oligonucleotide-conjugated gold nanoparticles. Angew Chem Int Ed Engl, 2012, 51: 1202–1206
Wang B, Chen N, Wei Y, et al. Akt signaling-associated metabolic effects of dietary gold nanoparticles in Drosophila. Sci Rep, 2012, 2: 563
Khan M I, Mohammad A, Patil G, et al. Induction of ROS, mitochondrial damage and autophagy in lung epithelial cancer cells by iron oxide nanoparticles. Biomaterials, 2012, 33: 1477–1488
Bondy S C. The neurotoxicity of environmental aluminum is still an issue. Neurotoxicology, 2010, 31: 575–581
Oesterling E, Chopra N, Gavalas V, et al. Alumina nanoparticles induce expression of endothelial cell adhesion molecules. Toxicol Lett, 2008, 178: 160–166
Man N, Yu S H. Rare earth oxide nanocrystals as a new class of autophagy inducers. Autophagy, 2010, 6: 310–311
Chen Y, Yang L, Feng C, et al. Nano neodymium oxide induces massive vacuolization and autophagic cell death in non-small cell lung cancer NCI-H460 cells. Biochem Biophys Res Commun, 2005, 337: 52–60
Yu L, Lu Y, Man N, et al. Rare earth oxide nanocrystals induce autophagy in HeLa cells. Small, 2009, 5: 2784–2787
Zhang Y, Yu C, Huang G, et al. Nano rare-earth oxides induced size-dependent vacuolization: An independent pathway from autophagy. Int J Nanomedicine, 2010, 5: 601–609
Zhang Y, Zheng F, Yang T, et al. Tuning the autophagy-inducing activity of lanthanide-based nanocrystals through specific surface-coating peptides. Nat Mater, 2012, 11: 817–826
Hussain S, Garantziotis S. Interplay between apoptotic and autophagy pathways after exposure to cerium dioxide nanoparticles in human monocytes. Autophagy, 2013, 9: 101–103
He Y, Lu H T, Sai L M, et al. Microwave synthesis of water-Dispersed CdTe/CdS/ZnS core-shell-Shell quantum dots with excellent photostability and biocompatibility. Adv Mater, 2008, 20: 3416–3421
Peng C W, Li Y. Application of quantum dots-based biotechnology in cancer diagnosis: Current status and future perspectives. J Nanomater, 2010, 2010: 1–11
Stern S T, Zolnik B S, McLeland C B, et al. Induction of autophagy in porcine kidney cells by quantum dots: A common cellular response to nanomaterials? J Nanopart Res, 2008, 106: 140–152
Seleverstov O, Zabirnyk O, Zscharnack M, et al. Quantum dots for human mesenchymal stem cells labeling. A size-dependent autophagy activation. Nano Lett, 2006, 6: 2826–2832
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhong, W., Lü, M., Liu, L. et al. Autophagy as new emerging cellular effect of nanomaterials. Chin. Sci. Bull. 58, 4031–4038 (2013). https://doi.org/10.1007/s11434-013-6058-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-6058-x