Abstract
Dry evergreen montane forests in Ethiopia are severely threatened. The status of species composition and structure of forest vegetation are important indicators to understand the trends of threats on local plant communities. In the present study, we examined the floristic composition and structure of the Kibate Forest, Wonchi Highland, Ethiopia along environmental gradients. Sixty-six (30 m × 30 m) plots were established every 100 m interval along altitudinal gradients (2811‒3073 m a.s.l.) in five transect lines for vegetation and environmental data collection. In total, 125 vascular plant species belonging to 104 genera and 52 families were identified. Eighteen species (14%) were endemic to Ethiopia and Eritrea. The two most dominant families, Asteraceae (29 species) and Lamiaceae (eight species) accounted for 30% of the total number of species. The highest number of species (54%) was herbs. Four major community types (viz., Olinia rochetiana-Myrsine melanophloeos, Ilex mitis-Galiniera saxifraga, Erica arborea-Protea gaguedi, and Hagenia abyssinica-Juniperus procera) were identified. The highest species richness, evenness, diversity, and importance value index were in community types 2 and 4. About 82% of the species and all endemic taxa except five were recorded in these two community types. The most dominant woody species were O. rochetiana, E. arborea, Olea europaea subsp. cuspidata, Myrica salicifolia, I. mitis var. mitis, and H. abyssinica with different patterns of population structure. The results show that there was a weak correlation between species richness and altitude. Our findings confirm that environmental variables both with interactions (such as altitude) and without interactions (such as livestock grazing) significantly (p < 0.05) affect species richness. Anthropogenic activities and overgrazing by livestock appear to be the main threat in community types 2 and 3. Urgent management practices and conservation measures such as prohibiting forest clearing and overgrazing and planting indigenous trees through community participation should be considered in community types that are rich in endemic species but are highly threatened.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forests are a valuable natural resource that can provide several ecosystem services, including carbon storage, soil preservation, nutrient cycling, and habitat to diverse species, and provide important socio-economic and cultural benefits (van Breugel et al. 2011; Kent 2012; RBG Kew 2016). Ethiopia is a hotspot of biodiversity due to its great variety of forest types (EBI 2014). However, dry evergreen montane forests are severely threatened due to the expansion of agriculture, overgrazing by livestock, and ineffective management (Getahun 2018). Accordingly, the current status of species composition and structure of these forests are important indicators to understanding the trends of threats on local plant communities (Lykke 1998; Chiarucci et al. 2011; Kent 2012; Longa et al. 2020). Plant species richness and abundance distribution vary along wide altitudinal gradients, either in a monotonic or hump-shaped pattern (Stevens 1992; Rahbek 1995). The decrease in species richness with increasing altitude seems to be a general pattern (Rahbek 1995; Didita et al. 2010; Dibaba et al. 2014; Berhanu et al. 2016; Pandey et al. 2016; Girma et al. 2018). According to Rahbek (1995), the variation of species richness with elevation might be connected to reduced temperatures and precipitation, area, and mountain slopes and the assumed corresponding reduction in productivity. However, the highest community diversity and species richness per hectare were reported at relatively high altitudes (2359‒3078 m a.s.l.) (Meragiaw et al. 2018). Similar results have been reported elsewhere in Ethiopia (Mekonnen 2009; Adugna 2010) and for tree species in China (Zhang et al. 2016). However, other studies conducted in the southwestern Ethiopian highlands showed that species richness did not show a strong response to wide altitudinal gradients (1000‒3000 m a.s.l.) (Desalegn and Beierkuhnlein 2010). These studies indicate a lack of consensus about the relationship between species richness and altitude.
According to Jamil (2012) and Dorji et al. (2014), the relationships of species richness and environmental variables are stronger in the interaction between variables than in variables without interaction. Generally, the interaction between physical environmental variables such as altitude, slope, and anthropogenic factors, including human disturbance and grazing by livestock, could control and shape floristic composition and diversity (Stevens1992; Rahbek 1995; Hadera 2000; Soromessa et al. 2004; Jamil 2012; Lee et al. 2012; Zhang et al. 2016; Girma et al. 2018; Meragiaw et al. 2018). With its diverse climatic conditions and wide range of ecological types, Ethiopia is a major center of plant diversity and endemism (Kelbessa et al. 1992; WCMC 1992; Vivero et al. 2006; Friis et al. 2010). Accordingly, the Flora of Ethiopia and Eritrea is diverse, with about 6500 to 7000 higher plant species, of which about 12% are endemic (WCMC 1992; Hedberg et al. 2009). However, many rare species are increasingly threatened by habitat loss and degradation (Vivero et al. 2006).
According to the International Union for Conservation of Nature (IUCN) Red List, about 21% of global plant species are currently threatened with extinction. In the tropics, the destruction of forests due to ever-increasing anthropogenic influences was identified as the main threat to 85% of threatened species (IUCN 2015; RBG Kew 2016). Likewise, the remnant natural forest cover of Ethiopia has been decreasing over time due to various forms of anthropogenic- induced pressures (Didita et al. 2010; van Breugel et al. 2011; Gashaw et al. 2015). Expansion of agricultural land, overgrazing by livestock, and selective tree felling for charcoal, fire-wood, and construction are some of the anthropogenic factors (WCMC 1992; Hadera 2000; Desalegn and Beierkuhlein 2010; Meragiaw et al. 2018). Generally, many researchers agree that disturbances to natural vegetation is a complex interaction of both natural and anthropogenic factors that govern the distribution of species abundance (Desalegn and Beierkuhlein 2010; Sunil et al. 2017; Meragiaw et al. 2018).
Understanding the patterns of plant species distribution and factors influencing them is a fundamental precondition for effective conservation of biodiversity (Magurran 2004; Lee et al. 2012; Sosef et al. 2017). Despite the present high rate of deforestation in Ethiopia (Moges et al. 2010), the remnant forests are still considered rich, both in species diversity and endemism (Kelbessa et al. 1992; Vivero et al. 2006; Hedberg et al. 2009; Friis et al. 2010; Meragiaw et al. 2018). A study on the remnant forest patches of Ethiopia is thus urgently needed as baseline information for ensuring sustainable use of natural vegetation, its conservation, and ecological management practices. However, baseline information on the status of floristic composition and structure is still lacking in some dry evergreen forests in general and for the Kibate Forest vegetation in particular. Investigation of the floristic composition and structure of the Kibate Forest along environmental gradients was conducted to address the following questions: (1) What are the main plant community types and do they vary in composition and diversity, i.e., in species richness, evenness, endemism, and dominance? (2) What does the vegetation structure look like? (3) How do anthropogenic and physical environmental gradients affect species richness? and, (4) What are the major threats to the Forest? To answer these questions, the following specific objectives of this study were formulated. These were to: (1) identify the main plant community types by species richness, evenness, endemism, diversity, and similarity (Sorensen’s) and comparison among the community types; (2) examine the vegetation structure using frequency, importance value index, population distribution pattern, and species dominance; (3) assess the relationship between species richness with environmental and anthropogenic factors, without the interaction of variables and with interactions between variables; and, (4) investigate the major threats to the Kibate Forest and suggest possible management practices.
Materials and methods
Description of the study area
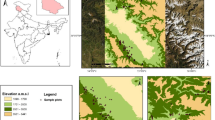
The study was conducted in the Kibate Forest 155 km southwest of Addis Ababa in Wonchi District, characterized by the Wonchi Crater Lake. Wonchi is found in the southwest Shewa Zone of Oromia Regional State (Fig. 1). Kibate Forest is a small forest patch that covers 450.2 ha, situated in the Gibe watershed of the Shewa floristic region at 8° 46′ 30.1908′'‒8° 47′ 36.7584′' N and 37° 51′ 53.8524′'‒37° 54′ 19.0152′' E. Altitudes range between 2800 and 3387 m a.s.l. The vegetation type is dry evergreen montane forest, widely intersected by areas of cultivation (Friis et al. 2010). In addition, Kibate Forest includes a small portion of sub-afroalpine vegetation at its peak and Walga River riparian vegetation that connects to the Wonchi Crater Lake (Meragiaw et al. 2018). The local dominant soil types are luvisols with volcanic ash as parent material (Mengistu 2006).
Primary climate data of the nearest station, Woliso-Giyon (2058 m a.s.l.), was collected from the National Meteorology Agency of Ethiopia. The study area has a unimodal rainfall distribution of an annual 1160 mm. The long rainy season stretches from March to the end of September. The highest rainfall is recorded in July and August, while a short dry season runs from October to February with little rainfall. Mean annual temperature is 19.3 °C with 1 °C and 28.0 °C mean minimum and maximum, respectively.
Data collection and analysis.
Sampling design
A reconnaissance survey was carried out in the Kibate Forest from December 2017 to January 2018 to collect information on the accessibility of sites and to determine sampling methods. Following the reconnaissance survey, five transect lines were laid out systematically along the edges of Wonchi Crater Lake, forest track/road, two small forest valleys with streams, and along the Walga stream. For the systematic sample of the five transects, the altitudinal ranges (2800‒3100 m a.s.l.) were divided into three classes with a 100 m a.s.l. interval, point position recorded using Gramin GPS 72 with an accuracy of 5 to 10 m under normal conditions, extending within 15 m under the dense forest canopy. Similarly, slopes (20‒70%) of each plot were recorded using a clinometer (percent scale). Vegetation and environmental data collection began at the lower edge of the forest and continued to the heights along the altitudinal gradient in all transects to include the representative floristic composition in each elevation class. Sixty-six 30 m × 30 m plots were established every 100 m. The number of plots in each elevation class was determined by the length of transect and varied from nine plots, (following the small stream in the middle of the forest), to 18 (along the bank of the Walga stream).
Vegetation data collection
In each plot, cover-abundance percentages were estimated for all vascular plant species following the Braun-Blanquet scale and then converted by van der Maarel ordinal transformation (van der Maarel 1979). For population structure analysis, diameter at breast height (DBH) of each woody species with DBH ≥ 2.5 cm and height ≥ 2.5 m were recorded (Woldu et al. 1999) using a diameter tape and Suunto clinometers, respectively. Woody species with DBH ≤ 2.5 cm were counted. Vascular plant specimens were collected with the help of local guides, pressed, dried, and brought to the National Herbarium of Ethiopia (ETH) at Addis Ababa University. They were then identified as per herbarium procedures, confirmed by a taxonomic expert, and deposited at the ETH. In addition, the status of endemic species and their distribution in the flora were cross-checked with previous research (Vivero et al. 2006; Hedberg et al. 2009; Meragiaw et al. 2016; IUCN 2019).
Environmental data collection:
Altitude and geographic coordinates were recorded for each of the 66 plots using GPS. In addition, ecological disturbances such as grazing intensity and human disturbances (felling of trees for firewood, charcoal production, and burning of vegetation for expansion of agricultural land) were noted, based on visual inspection. The status of human interference was estimated through the etic or cross-cultural approach following Hadera (2000), and subjective scales were designated as 0 = nil; 1 = low; 2 = moderate, and 3 = heavy. Similarly, intensity of livestock grazing was estimated following Woldu and Backeus (1991) as 0 = nil, 1 = slight, 2 = moderate, 3 = heavy, and 4 = destructive.
Plant community type analysis:
Vegetation data were analyzed using R Statistical Program following Crawley (2013), Oksanen et al. (2016), and Woldu (2017). Four community types were identified using a hierarchical clustering strategy of one minus similarity ratio as resemblance index with the ward's method. A synoptic table analysis was produced to determine the characteristic species within each of the 66 plots. Two characteristic species with high synoptic cover-abundance values (mean frequency x mean cover-abundance) were used to name the plant community following Kent (2012).
Vegetation structure analysis:
All recorded species and transformed abundance scores were used as input data for the similarity matrix. The vegetation structure was described using frequency distribution, density, DBH, height, and importance value index (IVI) for each species. The IVI was calculated by summing relative density, frequency, and dominance following Phillips (1959) and Kent (2012) to determine vegetation structure and the dominant forest species.
Analysis of population structure was categorized into four distribution patterns on the basis of stem density/ha across five DBH classes (Class1 = 2.5‒15.0; Class 2 = 15.1‒30.0; Class 3 = 30.1‒45.0; Class 4 = 45.1‒60.0; and Class 5 ≥ 60.1 cm). Basal area (BA), DBH, and heights were calculated following Aldred and Alemdag (1988).
Community diversity analysis:
Shannon–Wiener indices of diversity (H), species richness, and Shannon's equitability or evenness (J) were used to describe community diversity in the Kibate Forest using R Statistical package (vegan) (Woldu 2017), based on the original formula of Shannon and Wiener (1949). Similarly, Sorensen’s similarity (Ss) coefficient evaluated the similarity between community types (Sorenson 1948; Kent 2012). Species richness is often used in conservation studies to determine the healthy functioning of natural ecosystems. The index is not diversity by itself, rather it can be converted into the effective number of species (true diversity) that allows a comparison of biodiversity with other community types. The conversion of H (diversity) into true diversity (D) followed Jost (2007).
Environmental data analysis:
In addition to hierarchical agglomerative clustering to identify community types, canonical correspondence analysis (CCA) was used to analyze the relationship between sample plots in community types and environmental factors, including altitude and anthropogenic influence. In addition, the relationship between species richness and abundance distribution with environmental variables (i.e., variables without interactions and interaction between variables) was tested using a multiple regression model significant at p < 0.05. Pearson’s correlation for the relationship between species richness and altitude was applied using a scatter plot graph.
Results
Floristic composition and diversity of Kibate Forest
Based on the floristic analysis, 125 vascular plants belonging to 104 genera in 52 families were found in 66 plots. A complete list of species is presented in Supplementary Material (Table S1). Few woody species (14%) were dominant but Agarista salicifolia (Comm. ex Lam.) Don., Erica arborea L., Hagenia abyssinica (Bruce) J.F.Gmelin, Ilex mitis (L.) Radlk. var.mitis, Juniperus procera Hochst. ex. Endl., Myrica salicifolia Hochst. ex A. Rich., Myrsine melanophloeos (L.) R. Br., Nuxia congesta R.Br. ex Fresen., Olea europaea L. subsp. cuspidata, and Olinia rochetiana A. Juss in the upper canopy, Conyza hypoleuca A. Rich., Hypericum revolutum Vahl., Myrsine africana L., Osyris quadripartita Decn., Rosa abyssinica Lendley., Rubus apetalus Poir, and Rumex nervosus Vahl. in the middle canopy and Thymus schimperi Ron. subsp. schimperi in the lower canopy accounted for more than half (66%) of the total cover-abundance values. The majority of families (92%) were angiosperms. Pteridophytes, consisting of three families represented by four species, and gymnosperms represented by only one family (Cupressaceae) were also recorded.
The species distribution patterns across the families were unequal. About 19% of the families were represented by more than two species. More than one-fourth (30%) of the species were recorded in the two most dominant families, Asteraceae and Lamiaceae, which were represented by 29 and eight species, respectively, Fabaceae and Poaceae by six species, Rosaceae and Urticaceae by five species, followed by Rubiaceae and Solanaceae, each by four species. Both Myrsinaceae and Scrophulariaceae were each represented by three species, and nine families (17.3%) by two species. The remaining 33 families (i.e., 63.5%) were each represented by one species.
Diversity and distribution of endemic taxa in the Kibate Forest
Eighteen species (14%) were endemic to Ethiopia and Eritrea. Fourteen were endemic to Ethiopia but the remaining were near-endemic, confined to both Ethiopia and Eritrea. All endemic species are widely distributed except Pelargonium wonchiense Vorster and M.G. Gilbert which is restricted to the study area (Shewa floristic region of Ethiopia). Table 1 shows the distribution of the species in plant families, the habit of each species, the IUCN threat level, and their distribution in the floristic regions of Ethiopia as described in the Flora of Ethiopia and Eritrea.
Plant growth forms in the Kibate Forest
The analysis of growth forms showed that herbaceous species accounted for more than half of the total number of species (54%), while shrub and tree species accounted for approximately 24% and 10%, respectively. Seven species (5% of the total) appeared as shrub or tree forms at various sites. In addition, four fern species, two lianas, and one epiphyte were identified. The herbaceous species, including all nonwoody monocotyledons and dicotyledons, belonged to the families Cyperaceae, Poaceae, Asteraceae, and Menispermaceae.
Classification of the vegetation data in the Kibate Forest
Classification was derived from the cover-abundance of the plot data consisting of 125 species collected in 66 plots. All these vegetation data, subjected to hierarchical agglomerative classification, produced four distinct community types (C1–C4) determined at a dissimilarity height of 1.2 of the dendrogram (Fig. 2). The adjacent branches of the dendrogram show proximity in slope and altitudinal ranges and hence exhibit corresponding similarity in species composition. The highest number of species was recorded in C2 and C4 while the least in C3. The first two are also rich in endemic taxa (Table 1).
The vertical axis of the dendrogram represents the distance or dissimilarity between labeled clusters, and the horizontal axis represents the sampled sites (plot 1‒66) clustering in four branches.
The community types were named after two characteristic species. Based on the mean frequency and mean cover-abundance values of the synoptic table, four community types were recognized. These are O. rochetiana-M. melanophloeos community type (C1), I. mitis-Galiniera saxifraga community type (C2), E. arborea-Protea gaguedi community type (C3), and H. abyssinica-J. procera community type (C4). The dominant species have a cover-abundance value of 2.0 and more, and characteristic species, ones confined in one community type with values greater than or equal to 1.0, are presented in the Supplementary Material (Table S2).
Distribution of community types in the Kibate Forest
Community type 1 O. rochetiana-M. melanophloeos: This community type is distributed between 2876 and 3021 m a.s.l. except for plot 1 located at 2838 m a.s.l. It is represented by 19 plots where 54 species are recorded. Eleven of thesesare unique to this community type; the other 43 are shared with other community types. Six species are shared only with C2. The 11 species exclusively associated with this community type include Andoropogon amethystinus Steud., Argyrolobium ramosissimum Bak., Brucea antidysenterica J. F. Mill., Carex steudneri Bock, Cineraria abyssinica Sch. Bip. ex A. Rich., Cyathula cylindrica Moq., Orobanche ramosa L., Primula verticillata subsp. simensis, Satureja punctata subsp. ovata, Senecio inornatus DC., and Solanum nigrum L. subsp. nigrum. A. salicifolia, M. africana, M. melanophloeos, N. congesta, O. europaea, O. rochetiana, and R. abyssinica are the main characteristic and dominant woody species in C1.
Community type 2 I. mitis-G. saxifraga: This community type is distributed in the wider altitudinal range from 2867 to 3044 m a.s.l.,showing some overlap in altitude with community type 1. It is represented by the highest number of species (73) in 18 plots. Of the total species, 54 are commonly shared with other community types, while 19 are found only in this community type. Species exclusiveto this community include Adenocarpus mannii (Hook. j) Hook. f., Echinops macrochaetus Fresen., Galium simense Fresen., Maytenus addat, Mentha x piperita L., Pilea johnstonii Oliv., Polygala abyssinica Fresen., Schefflera volkensii (Engl.) Harms, Senecio ochrocarpus, Silene burchellii DC. form D, Solanecio gigas, Stephania abyssinica (Dillon & A. Rich.) Walp., Solanum villosum Mill., Tanacetum cinarariifolium (Trev.) Sch. Bip., Urera hypselodendron A. Rich., Verbascum sinaiticum Benth., Vernonia leopoldi, Vernonia sp and Vernonia urticifolia A. Rich. G. saxifraga (Hochst.) Bridson, I. mitis var. mitis and R. apetalus are the dominant woody species in C2.
Community type 3 E. arborea-P. gaguedi: This community type is distributed in the highest altitudinal ranges between 2955 and 3078 m a.s.l. except for plot 20 which lies at 2910 m a.s.l. It is represented by 16 plots where the fewest number of species (44) were recorded. Thirty-three species are commonly shared with other community types, while 11 are confined only in this community. Species unique to this community type include Bidens pachyloma, Campanula edulis Forssk, Epipactis africana Rendle, Euphoria platyphyllos L., Isodon schimperi (Vatke) J.K. Morton, Mikania capensis DC. Oldenlandia lancifolia (Schumach.) DC., Pittosporum viridiflorum Sims., Plectocephalus varians, Scabiosa columbaria L. and Senna multiglandulosa (Jacq.) Irwin & Barneby. E. arborea, M. salicifolia, O. quadripartita and P. gaguedi J. F. Gmel. are the dominant woody species.
Community type 4 H. abyssinica-J. procera: This community type is distributed at the lowest altitudes between 2811and 2899 m a.s.l. with exceptions for three plots located at high altitudes. These were plot 56 (2915 m a.s.l.), plot 42 (2988 m a.s.l.) and plot 47 (3060 m a.s.l.). This community had the highest number of species (73). Fifty species are commonly shared with other communities, while 23 were exclusiveto this community. Arundo donax K. Schum., Astragalus atropilosulus (Hoehst.) Bunge, Carex acutiformis Ehrb., C. steudneri, Cichorium intybus L., Crepis tenerrima (Sch. Bip. ex A. Rich.) R. E. Fries, Crotalaria sp., Distictis buccinatonia (DC.) A. Gentry, Droguetia iners (Forssk.) Schweinf, Festuca simensis Hochst. ex A. Rich., Girardinia bullosa (Steudel) Wedd., Haplocarpha schimperi (Sch.-Bip.) Beauv., Helichrysum formosissimum Sch.Bip.ex A.Rich., Mikaniopsis clematoides, Parochaetus communis D. Don., P. wonchiense, Pteris cretica L., Rhamnus staddo A. Rich., Sanicula elata Buch.-Ham. ex D. Don., Solanum marginatum L.f., Sida schimperiana Hochst. ex A. Rich., Streblochaete longiarista (A. Rich.) Pilg. and Urtica simensis are the species are found only in this community. H. abyssinica, H. revolutum, and J. procera, Jasminum stans were the dominant woody species.
Species richness, evenness, and diversity of community types in the Kibate Forest
Shannon–Wiener diversity index was computed for the four plant community types. Community true diversity decreased in the order of C2 > C4 > C1 > C3. Regarding species richness, the highest total number of species (73) were recorded in C2 and C4, followed by C1 and C3. The highest Shannon’s equitability (evenness) was in C2, whereas the least was in C1. Table 2 shows species richness, evenness, diversity index, true diversity, and maximum diversity values.
Similarity among community types in the Kibate Forest
Sorensen’s similarity coefficient (Ss) was computed to detect dissimilarities among the four community types. Similarity values in species composition ranged from 49 to 64%. The highest similarity was recorded between C1 and C2 (64%), followed by C2 and C4 (61%), C1 and C3 (54%), C1 and C4 (52%), C3 and C4 (50%). The least similarity was between C2 and C3 (49%).
Vegetation structures in the Kibate Forest
Frequency of plant species in the Kibate Forest
Based on species distribution over the 66 plots, the percentage frequency classes were grouped into six with nearly a 15% interval each. A: 1‒15, B: 16‒30, C: 31‒45, D: 46‒60, E: 61‒75, F: 76‒90. The results indicated that 97 (78%) species were in the lowest frequency class (A), followed by frequency class B with 12 (10%), D with 5 (4%), C and E with 4 (3%) each. The least frequency percentage (2%) was in the highest frequency class (F: 76‒90%). O. europaea subsp. cuspidata was the most frequent occurrence in 58 plots, followed by E. arborea in 54 plots, M. salicifolia in 50 plots, M. africana in 48 plots, O. rochetiana in 46 plots, O. quadripartita in 44 plots, R. abyssinica in 40 plots, N. congesta in 38 plots, H. abyssinica in 36 plots.
Importance value index of the four community types.
All plant species recorded were grouped into five classes based on their IVI values ranging from 1.0 to 30.5. Eighty-six species were in the IVI class ˂ 7, 14 species to 7‒14, 14 species to 15‒22, 8 species to 23–28; only 3 species had IVI values ˃ 28. Community type 2 had the highest importance value (29.8%), followed by C4 (29.0%), C1 (22.9%), and C3 (18.3%). Analysis of IVI revealed that the Kibate Forest was dominated by 20 woody species, accounting for 49% of the total values (979). Of the top 10 species, O. rochetiana was first (30.5), followed by E. arborea, O. europaea subsp. cuspidata, M. salicifolia, I. mitis var. mitis, H. abyssinica, N. congesta, J. procera, A. salicifolia, Maesa lanceolata Forssk. with IVI values ranging from 29.0 to 25.0 in that order.
Population distribution patterns in the Kibate Forest
The population distribution patterns were categorized in four classes represented by J. procera, E. arborea, H. abyssinica, and O. europaea subsp. cuspidata (Fig. 3a-d). The highest number of species was recorded in the first pattern (Fig. 3a), followed by the fourth (Fig. 3d).
Relationship between environment and species distribution in the Kibate Forest
The distribution pattern of species in the sample plots shows that human disturbance negatively affected the community types. Based on CCA analysis, human disturbance is associated more with C2 and C3 in both axes (CCA2 and CCA1). Altitude is more correlated with community type 2 which is distributed over a wide range of altitudes (Fig. 4). The direction and length of arrows show the degree of correlation between the sample plots (plant community types) and the variables.
Although there are small patches of forest and riparian vegetation at various altitudes, Kibate Forest extends from 2800 to 3100 m a.s.l. The largest number of plots (37) were laid out in the middle altitude (2901‒3000 m a.s.l.) where the larger portion of the forest is found, followed by the lowest altitude (2800‒2900 m a.s.l.) with 17 plots, while the least number of plots (12) were established at the highest altitude, above 3001 m a.s.l. Figure 5 shows a correlation of species richness per plot and altitudinal gradient of ‒0.13 which is an insignificant negative relationship. Further multiple regression analysis in Table 3 confirms that species richness and abundance distribution do not have a significant relationship with altitude alone (p = 0.19).
Although altitude does not significantly affect species richness, it is interesting to see that the interactions between altitude with human disturbance, altitude with slope, and altitude with human disturbance and slope are significant (Table 3). This suggests that, as altitude increases, the effects of human disturbance and slope becomes less pronounced in some sites because of inaccessibility to livestock and people. On the other hand, the effect of disturbance at higher altitudes results in the reduction of large trees but the proliferation of herbaceous species in other sites.
Discussion
Floristic composition, community types, and diversity
Kibate Forest, which has a considerable taxonomic diversity (52 families and 104 genera) of vascular plant species (125) that contain 18 endemic species, is interesting both for its unique composition and its relation to environmental factors. However, it has a smaller number of woody species (52) than other similar sized forest patches elsewhere in Ethiopia (Kuma and Shibru 2015; Mekonen et al. 2015; Girma et al. 2018; Meragiaw et al. 2018). The degree of human and livestock disturbances, topographic, and climatic conditions could be considered responsible for the observed differences. In line with Desalegn and Beierkuhnlein (2010), altitude has not affected species richness per plot in the present study (Table 3). Regarding species richness and distribution across the plots, both the highest (29 in plot 53) and the least (5 in plot 13) number of species were found in community type 4 at the lowest altitude (2811‒2899 m a.s.l.).
The majority of species are herbs and their proliferation could be related to intensive anthropogenic effects, including selective felling of woody species for various uses (Meragiaw et al. 2016). The family Asteraceae had the highest number of species (29) in conformity with other similar floristic studies (Hundera et al. 2007; Meragiaw et al. 2016, 2018; Girma et al. 2018). It is the largest dicotyledonous family in Ethiopia and Eritrea with 440 species as a result of an adaptive rapid seed dispersal mechanism (Hedberg et al. 2009). Sorensen’s (Ss) similarity coefficients were also used to detect similarities between the four plant communities of the forest. The highest similarity coefficients (Ss = 0.64) between C1 and C2 could be because C2 covered the widest range of altitudes with some overlap with C1. This could create similar microclimatic conditions that favor the shared species in the two communities.The lowest similarity coefficients (Ss = 0.49) were observed between the C2 and C3. This could be due to all C3 plots were in the upper highland forest. The highest species richness, evenness, and diversity were recorded in C2, whereas the least species richness and diversity were in C3 but had the second-highest species evenness. However, both community types were subject to anthropogenic influences, particularly the selective felling of trees and overgrazing. Thus, altitude and slope variations may contribute to the differences between these two community types. In comparison with adjoining vegetation, the mean value of diversity of all community types of Kibate Forest (3.33) is greater than the Woynwuha natural forest (Mekonen et. al. 2015) but smaller than that of Walga riparian vegetation (3.55) that covers a wide range of altitudes (Meragiaw et al. 2018). In general, Kibate Forest shows different phytogeographical similarities with other forests in Ethiopia. The high number of similar species could be due to their geographical proximity, similar altitudes, and agroecological zones (Girma et al. 2018; Meragiaw et al. 2018). In contrast, the lowest number of similar species may arise from the wide variation in forest types, altitudinal ranges, climatic conditions, sample sizes, and degree of anthropogenic disturbances (Tegene et al. 2018; Atomsa and Dibbisa 2019).
Vegetation structure in the Kibate Forest
Seventy-eight percent of the species are in low-frequency classes (1, 2, and 3), indicating a high degree of floristic heterogeneity (Kent 2012; Dibaba et al. 2014). The low percent in classes 4 and 5may be related to the selective felling of large trees. Species that account for 74% of the density and basal area include J. procera, N. congesta, I. mitis var. mitis, O. europaea subsp. cuspidata, M. salicifolia, A. salicifolia, H. abyssinica, Buddleja polystachya Fresen. and M. melanophloeos. E. arborea and O. rochetiana were more frequent and were among the ten species which had higher IVI values. Similar studies elsewhere in Ethiopia also confirmed that species that have high basal area and IVI values could be considered as ecologically important woody species (Bekele 1994; Meragiaw et al. 2018).
Population structure is indicative of the stability and regeneration status of forest vegetation (Kent 2012). Four population patterns of forest structure have been documented in Kibate Forest. For the first pattern, similar studies elsewhere in Ethiopia also confirmed a J-shaped population distribution which had the highest density in DBH classes five and four and then decreased to the lowest DBH class, was produced by J. procera, A. salicifolia, M. salicifolia, G. saxifraga, Halleria lucida L., M. lanceolata, P. viridiflorum and P. gaguedi (Fig. 3a). Seed germination in the J-shaped structure, which leads to poor reproduction and an abnormal population structure, could be a serious issue (Bekele 1994; Meragiaw et al. 2018). A small number of individuals in lower diameter classes could be due to overgrazing and browsing of young seedlings. The second population pattern with high relative density in DBH classes 1 and 2 but absent in higher DBH classes was shown by Erica arborea, B. antidysenterica, and H. revolutum (Fig. 3b). This population pattern may be assumed to be an interrupted inverted J-shaped pattern, indicative of a recovering forest with many small stems and few large ones. The third population distribution pattern is close to a bell-shape and was illustrated by species such as M. melanophloeos, O. rochetiana, B. polystachya, H. abyssinica, I. mitis var. mitis, M. addat and S. volkensii with high density in the second and third DBH classes and no individuals at either ends (Fig. 3c). The fourth population pattern is bell-shaped and produced by two species, N. congesta and O. europaea subsp. cuspidata, where density increased with increasing DBH and the highest density was at DBH class 4 (Fig. 3d). The bell-shaped pattern could be attributed to several possibilities such as overgrazing at young stages for some species and/or slow seed germination due to a stony endocarp (e.g., O.europaea subsp. cuspidata) and/or selective felling of species with high value wood (Bekele 1994; Meragiaw et al. 2018).
Impacts of environmental variables on plant distribution in different community types
Altitude plays an important role in determining species diversity and abundance (Odland and Birks 1999; Mekonnen 2009; Adugna 2010; Didita et al. 2010; Dibaba et al. 2014; Girma et al. 2018; Meragiaw et al. 2018). In contrast to several findings, the distribution of species richness did not show a strong response to altitude alone in the present study. Desalegn and Beierkuhnlein (2010) arrived at conclusions similar to our study. Pandey et al. (2016) suggested, that for slope to have a significant impact on diversity and species richness, it needed to have a wide range along a long altitudinal gradient. In agreement with this study, a significant negative relationship between species richness and slope was reported by Sharma et al. (2009). Additionally, the factor with the most notable negative influence on vegetation, and hence on community types, appears to be disturbance (Fig. 4 and Table 3), but the level of intensity is marked at community types 2 and 3. Selective felling of trees, expansion of agriculture, and overgrazing grazing account for most of the disturbance to the vegetation in Kibate Forest. The positive significance of species richness with altitude and other factors (slope, human disturbance) could be due to an increase in herbaceous species as a result of gap formation when removal of a large woody species as described by Misganaw et al. (2016) and Woldu et al. (2020). At low elevations, anthropogenic disturbances contributed to the increase of plant diversity, especially shrubs and herbs in the understory (Zhang et al. 2016). Other researchers conclude that understory layers are more sensitive to changes in microenvironment (temperature, topographic heterogeneity, intensity of disturbance). Low to moderate disturbances can promote early successional species, and increase diversity and abundance in the herb layer (Odland and Birks 1999; Elliott et al. 2011).
Endemic species and conservation status of the natural vegetation
Kibate Forest has a large number of endemic species (18) which shows high ecological stability as proposed by the World Conservation and Monitoring Center (WCMC 1992). Endemicity is higher (18 species) than in the nearest riparian vegetation along the Walga river (Meragiaw et al. 2018). Among the plant species endemic to Ethiopia, Vivero et al. (2006) and IUCN (2019) have shown that M. addat is vulnerable to extinction, while Inula confertiflora and S. punctata are in a near-threatened category. However, Laggera tomentosa, S. gigas, V. leopoldi and T. schimperi subsp. schimperi are of least concern (Table 1). This suggests that special attention needs to be given to the community types containing species which are vulnerable and threatened. The threat level status of the rest of the 11 species is not provided. This requires further work to establish the threat level of many of the endemic species in the country. P. wonchiense, which carries the name of the study area, is confined in Shewa floristic region of the Flora of Ethiopia and Eritrea.
Knowledge of composition, diversity, and species distribution in plant community types should be used to design sustainable management programs of any forest ecosystem (Lykke 1998; Pandey et al. 2016; Girma et al. 2018). The green economy plan of Ethiopia includes improving forest cover on a large scale and promotes agroforestry at the household level around the study area and elsewhere. However, much attention is given to exotic and other species of immediate economic importance. This requires considerable attention on species that have multiple ecological and economic benefits and provide long-lasting ecosystem services. In line with this, agroforestry tree species, including indigenous ones, which are suitable for various agroecological and farming systems, could be promoted to meet both production and ecological objectives as well as to enhance species diversity and carbon sequestration (Meragiaw 2017).
A combination of rehabilitation and restoration techniques is available for conservation of natural vegetation (Hundera et al. 2007; Didita et al. 2010; Gashaw et al. 2015; Meragiaw et al. 2018). These may include reducing grazing pressures, prohibiting selective felling of trees, and limiting the expansion of agricultural land. However, a blanket recommendation of rehabilitation techniques and passive restoration strategies throughout the entire vegetation communities could be unmanageable, both technically and economically. Rehabilitation might be achieved through several steps (Chiarucci et al. 2011) prioritizing the most vulnerable parts of the forest such as community types 2 and 3 and those with the highest number of endemic species such as community type 2. Success of the rehabilitation efforts could serve as examples for conserving the rest of the forest. Duality and complexity of diversity have to be considered in designing conservation efforts. While high diversity seems to be desirable, even the distribution of a species in a community indicates a lack of dominance induced by anthropogenic influences. In this study, community types which had high diversity also had a high number of endemic species. If the objective of conservation focuses on species, then a moderate level of disturbance which promotes the proliferation of endemic species could be considered a possible option. If, however, rehabilitating the vegetation into an intact multistory forest is desired, then the sustainability of the endemic species could be compromised. Balancing the two contrasting options is a subject for further research.
Conclusions
This investigation of the vegetation ecology of Kibate Forest focused on taxonomic diversity, species richness, evenness, community types, and population structure. Although the forest is subject to considerable disturbances, it is floristically rich with 125 species, with more than 14% endemic to Ethiopia and Eritrea. The species composition is dominated by herbs and shrubs. The Angiosperm families Asteraceae and Lamiaceae make up 26% of the total plant species, with 20 and seven genera, respectively. The relatively high H average (3.33) and patterns of population structure are good indicators for harboring diverse plant species. The relationship between vegetation data and environmental factors shows that slope, livestock grazing, and human activities significantly affect species richness and distribution in the 66 plots. While altitude had no significant influence on species richness and abundance per plot, it positively affected interaction with slope and human activities. The most dominant woody species that showed different patterns of population structure were O. rochetiana, E. arborea, O. europaea subsp. cuspidata, M. salicifolia, I. mitis var. mitis, and H. abyssinica. From the structural analysis, the highest number of species in the J-shaped distribution pattern indicates the dominance of large individuals in higher classes than in lower classes, and results in the rare occurrence of small individuals. This is an implication of the existence of overgrazing by livestock in the area. Human activities and livestock grazing are the main threats in communities 2 and 3. Some of the endemic species are in near threatened status and vulnerable to extinction, while most are in the category of least concern. Among the community types identified, the highest species richness, evenness, diversity, and endemism occurred in community type 2, I. mitis-G. saxifraga in the mid-altitudes between 2867 to 3044 m a.s.l. More attention needs to be given to the middle altitudes of the forest to conserve the vulnerable and threatened endemic taxa. The finding of this study provides base-line information for conservation and management practices in community types rich in endemic species but are highly threatened.
References
Adugna F (2010) Impact assessment of dam construction on the floristic composition and diversity: a case of Gilgel Gibe III hydroelectric dam, Southwestern Ethiopia. M.Sc. Thesis. Addis Ababa University, Addis Ababa.
Aldred AH, Alemdag IS (1988) Guidelines for forest inventory. Information Report PI-X-77, Petawawa National Forestry Institute, Canadian Forestry Service, Chalk River, Ontario, p.134
Atomsa D, Dibbisa D (2019) Floristic composition and vegetation structure of Ades Forest, Oromia regional state, West Hararghe zone. Ethiopia Trop Plant Res 6(1):139–147
Bekele T (1994) Comprehensive summaries of Uppsala dissertations from the Faculty of Science and Technology: studies on remnant afromontane forest on the central Platea of Shewa, Ethiopia. Doctoral dissertation. Acata Universities, Uppsala.
Berhanu A, Woldu Z, Demissew S (2016) Elevation patterns of woody taxa richness in the evergreen Afromontane vegetation of Ethiopia. J For Res. https://doi.org/10.1007/s11676-016-0350-y
Chiarucci A, Bacaro G, Scheiner SM (2011) Old and new challenges in using species diversity for assessing biodiversity. Phil Trans R Soc B 366:2426–2437. https://doi.org/10.1098/rstb.2011.0065
Crawley MJ (2013) The R Book. Wiley, Chichester, pp 445–824
Desalegn W, Beierkuhnlein C (2010) Plant species and growth form richness along altitudinal gradients in the southwest Ethiopian highlands. J Veg Sci 21:617–626. https://doi.org/10.1111/j.16541103.2010.01177.x
Dibaba A, Soromessa T, Kelbessa E, Tilahun A (2014) Diversity, structure and regeneration status of the woodland and riverine vegetation of Sire Beggo in Gololcha District. Eastern Ethiopia Momona Ethiop J Sci 6(1):70–96
Didita M, Nemomissa S, Woldemariam T (2010) Floristic and structural analysis of the woodland vegetation around Dello Menna. Southeast Ethiopia J For Res 21(4):395–408. https://doi.org/10.1007/s11676-010-0089-9
Dorji T, Moe SR, Klein JA, Totland Ø (2014) Plant species richness, evenness, and composition along environmental gradients in an alpine meadow grazing ecosystem in Central Tibet. China Arct Antarct Alp Res 46(2):308–326. https://doi.org/10.1657/1938-4246-46.2.308
EBI (2014) Ethiopia’s fifth national report to the Convention on Biological Diversity. Ethiopian Biodiversity Institute, Addis Ababa. https://www.cbd.int/doc/world/et/et-nr-05-en.pdf
Elliott KJ, Harper CA, Collins B (2011) Herbaceous response to type and severity of disturbance. In: Greenberg CH, Collins BS, Thompson FR (eds) Sustaining young forest communities: ecology and management of early successional habitats in the central hardwood region, USA, Managing Forest Ecosystems. Springer, New York, pp 97–119
Friis I, Demissew S, Breugel VP (2010) Atlas of the potential vegetation of Ethiopia Copenhagen. Biol Skr 58:1–307
Gashaw T, Terefe H, Soromessa T, Ahmed S, Megersa T (2015) Riparian areas rehabilitation and restoration: an overview. Point J Agric Biotechnol Res 1(2):055–063
Getahun AA (2018) The status of ecosystem resources in Ethiopia: potentials, challenges and threats: review paper. J Biodivers Endanger Species 6:208. https://doi.org/10.4172/2332-2543.1000208
Girma Z, Chuyong G, Evangelista P, Mamo Y (2018) Vascular plant species composition, relative abundance, distribution, and threats in Arsi Mountains National Park. Ethiopia MT Res Dev 38(2):143–152. https://doi.org/10.1659/MRD-JOURNAL-D-17-00006.1
Hadera G (2000) A Study on the ecology and management of the Dessa forest in the northeastern escarpment of Ethiopia. M.Sc. Thesis. Addis Ababa University, Addis Ababa.
Hedberg I, Friis I, Person E (2009) General part and index to Vol 1–7. Flora of Ethiopia and Eritrea Vol 8. ETH, Addis Ababa, Ethiopia and Sweden.
Hundera K, Tamrat B, Ensermu K (2007) Floristic and phytogeographic synopsis of a dry Afromontane coniferous forest in Bale Mountains (Ethiopia): implications to biodiversity conservation. SINET: Ethiop J Sci 30:1–12
IUCN (2015) Conservation Successes Overshadowed by more Species Declines –IUCN Red List update. http://www.iucn.org/news_homepage/?21561/Conservationsuccesses-overshadowed-by-more-speciesdeclines--IUCN-Red-List-update
IUCN (2019) The IUCN Red List of Threatened Species. Version 2019-2. http://www.iucnredlist.org. [Downloaded on 18 July 2019].
Jamil T (2012) Models to relate species traits to environment: a hierarchical statistical approach. Docteral Dissertation, Wageningen University, Wageningen.
Jost L (2007) Partitioning diversity into independent alpha and beta components. Ecol 88(10):2427–2439
Kelbessa E, Demissew S, Woldu Z, Edwards S (1992) Some threatened endemic plants of Ethiopia. In: Hillman C, Edwards S, Asfaw Z (eds) The status of some plant resources in parts of tropical Africa. Botany 2000: East and Central Africa. NAPRECA Monograph Series no. 2. NAPRECA, Addis Ababa University, Addis Ababa, Ethiopia.
Kent M (2012) Vegetation description and data analysis: a practical approach, 2nd edn. Wiley, Chichester, p 438
Kew RBG (2016) The State of the World’s Plants Report -2016. Royal Botanic Gardens, Kew
Kuma M, Shibru S (2015) Floristic composition, vegetation structure, and regeneration status of woody plant species of Oda Forest of Humbo Carbon Project, Wolaita. Ethiopia J Bot. https://doi.org/10.1155/2015/963816
Lee C, Chun J, Song H, Cho H (2012) Altitudinal patterns of plant species richness on the Baekdudaegan Mountains, South Korea: mid-domain effect, area, climate, and Rapoport’s rule. Ecol Res. https://doi.org/10.1007/s11284-012-1001-1
Longa GD, Boscutti F, Marini L, Alberti G (2020) Coppicing and plant diversity in a lowland wood remnant in–North East Italy. Plant Biosyst–Int J Deal Aspects Plant Biol 154(2):173–180. https://doi.org/10.1080/11263504.2019.1578276
Lykke AM (1998) Assessment of species composition change in savanna vegetation by means of woody plants’ size class distributions and local information. Biodivers Conserv 7:1261–1275
Magurran AE (2004) Measuring biological diversity. Blackwell Science ltd, Malden, p 132
Mekonen T, Ayele B, Ashagrie Y (2015) Woody plant species diversity, structure and regeneration status of Woynwuha Natural Forest, North West Ethiopia. J Agric Environ Sci 1(2):90–113
Mekonnen A (2009) Impact of dam construction on plant species composition and diversity: the case of Koga irrigation dam, Northwestern Ethiopia. M.Sc. Thesis. Addis Ababa University, Addis Ababa.
Mengistu A (2006) Country pasture/forage resource profiles Ethiopia. FAO, Rome
Meragiaw M (2017) Role of agroforestry and plantation on climate change mitigation and carbon sequestration in Ethiopia. J Tree Sci 36(1):1–15. https://doi.org/10.5958/2455-7129.2017.00001.2
Meragiaw M, Asfaw Z, Argaw M (2016) The status of ethnobotanical knowledge of medicinal plants and the impacts of resettlement in Delanta, northwestern Wello, northern Ethiopia. Evid Based Complement Alternat Med 2016:1–24. https://doi.org/10.1155/2016/5060247
Meragiaw M, Woldu Z, Martinsen V, Singh BR (2018) Woody species composition and diversity of Riparian vegetation along the Walga River. Southwestern Ethiopia PLoS ONE 13(10):e0204733. https://doi.org/10.1371/journal.pone.0204733
Moges Y, Eshetu Z, Nune S (2010) Ethiopian Forest resources: current status and future management options in view of access to carbon finances. Forestry Research Center, EIAR/NBCU Project Office in Ethiopia, Addis Ababa, Ethiopia.
Odland A, Birks HJB (1999) The altitudinal gradient of vascular plant richness in Aurland, western Norway. Ecography 22:548–566
Oksanen J, Guillaume BF, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin, PR, O'Hara RB, Simpson GL, Solymos P, Stevens MHHS, Szoecs E, Wagner H (2016) vegan: community ecology package, R package version 2.4‒1. htps://cran. r-project.org.
Pandey KPI, Adhikari YP, Weber M (2016) Structure composition and diversity of forest along the altitudinal gradient in the Himalayas Nepal. Appl Ecol Environ Res; 14(2):235‒251. https://doi.org/10.15666/aeer/1402_235251
Phillips EA (1959) Methods of vegetation study. Holt, New York, NY, p 107
Rahbek C (1995) The elevational gradient of species richness: a uniform pattern? Ecography 18:200–205
Shannon CE, Wiener W (1949) The mathematical theory of communication. University of Illinois, Champaign
Sharma CM, Suyal S, Gairola S, Ghildiyal SK (2009) Species richness and diversity along an altitudinal gradient in moist temperate forest of Garhwal Himalaya. J Am Sci 5(5):119–128
Sorenson T (1948) A Method of establishing groups of equal amplitudes in plant sociology based on similarity of species content and its application to analyses of the vegetation on danish commons. Kongelige Danske Videnskabernes Selskab, Biologiske Skrifter 5:1–34
Soromessa T, Teketay D, Demissew S (2004) Ecological study of the vegetation in Gamo Gofa Zone. Southern Ethiopia Trop Ecol 45(2):209–221
Sosef MSM, Dauby G, Blach-Overgaard A, van der Burgt X, Catarino L, Damen T, Deblauwe V, Dessein S, Dransfield J, Droissart V, Duarte MC, Engledow H, Fadeur G, Figueira R, Gereau RE, Hardy OJ, Harris DJ, de Heij J, Janssens S, Klomberg Y, Ley AC, Mackinder BA, Meerts P, van de Poel JL, Sonké B, Stévart T, Stoffelen P, Svenning J-C, Sepulchre P, Zaiss R, Wieringa JJ, Couvreur TLP (2017) Exploring the floristic diversity of tropical Africa. BMC Biol 15(1):15. https://doi.org/10.1186/s12915-017-0356-8
Stevens GC (1992) The elevational gradient in altitudinal range: an extension of Rapoport's latitudinal rule to altitude. Am Nat 140: 893‒911. pmid:19426029.
Sunil C, Somashekar RK, Nagaraja BC (2017) Diversity and composition of riparian vegetation across forest and agroecosystem landscapes of river Cauvery, southern India. Trop Ecol 57(2):343–354
Tegene AS, Gamo FW, Cheche SS (2018) Woody vegetation composition, structure, and community types of Doshke Forest in Chencha. Int J Bio, Gamo Gofa Zone, Ethiopia. https://doi.org/10.1155/2018/4614790
van Breugel P, Kindt R, Lillesø J-PB, Bingham M, Demissew S, Dudley C, Friis I, Gachathi F, Kalema J, Mbago F, Minani V, Moshi HN, Mulumba JW, Namaganda M, Ndangalasi HJ, Ruffo CK, Vadaste M, Graudal LOV, Jamnadass RH (2011) Potential natural vegetation of Eastern Africa (Ethiopia, Kenya, Malawi, Rwanda, Tanzania, Uganda and Zambia) Vol. 6. An overview of the methods and material used to develop the map. Forest and Landscape Denmark working papers; 68/ 2011, p. 139.
van der Maarel E (1979) Transformation of cover abundance values in phytosociology and its effect on community. Vegetation 39:47–114
Vivero JL, Kelbessa E, Demessew S (2006) Taxonomyand ecology of African plants, their conservation and sustainable use. In: Ghazanfar SA, Beentje HJ (eds) Progress on the red list of Ethiopia and Eritrea conservation and biogeography of endemic of taxa. Royal Botanic Gardens, Kew, pp 761–778
World Conservation Monitoring Center (WCMC) (1992) Global biodiversity: status of the earth living recourses. Chapman and Hall, London, p 594
Woldu Z (2017) Comprehensive and ecological data analysis: basics, concepts and methods. Addis Ababa University, Addis Ababa, p 408
Woldu Z, Backeus I (1991) The shrub land vegetation in Western Shewa, Ethiopia and its possible recovery. J Veg Sci 2:173–180
Woldu Z, Fetene M, Abate A (1999) Vegetation under different tree species in Acacia woodland in the rift valley of Ethiopia. SINET: Ethiop J Sci 22(2):235–252
Woldu G, Solomon N, Hishe H, Gebrewahid H, Gebremedhin MA, Birhane E (2020) Topographic variables to determine the diversity of woody species in the exclosure of Northern Ethiopia. Heliyon 6(1):e03121
Zhang WX, Huang DZ, Wang RQ, Liu J, Du N (2016) Altitudinal patterns of species diversity and phylogenetic diversity across temperate mountain forests of Northern China. PLoS ONE 11(7):e0159995. https://doi.org/10.1371/journal.pone.0159995
Acknowledgements
The authors are grateful to Mr. Melaku Wondafrash from ETH for his assistance and confirmation of plant specimens. The primary climate data of the study area was from the National Meteorology Agency. We appreciate the people of Wonchi and field guides, especially Mr. Kassahun Kumessa, for providing pertinent field information.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: The work was supported by the Regional Capacity Building for Sustainable Natural Resource Management and Agricultural Improvement under Climate Change (CAPSNAC) Project of the Norwegian Program for Capacity Building in Higher Education and Research for Development (NORHED).
The online version is available at http://www.springerlink.com.
Corresponding editor: Tao Xu.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Meragiaw, M., Woldu, Z., Martinsen, V. et al. Floristic composition and structure of the Kibate Forest along environmental gradients in Wonchi, Southwestern Ethiopia. J. For. Res. 32, 2669–2682 (2021). https://doi.org/10.1007/s11676-021-01305-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-021-01305-z