Abstract
Methamphetamine (METH) addiction is a biopsychosocial disorder that is accompanied by multiple relapses even after prolonged abstinence, suggesting the possibilities of long-lasting maladaptive epigenetic changes in the brain. Here, we show that METH administration produced time-dependent increases in the expression of corticotropin-releasing hormone (Crh/Crf), arginine vasopressin (Avp), and cocaine- and amphetamine-regulated transcript prepropeptide (Cartpt) mRNAs in the rat nucleus accumbens (NAc). Chromatin immunoprecipitation (ChIP) assays revealed that METH increased the abundance of phosphorylated CREB (pCREB) at the promoter of Cartpt but not at Avp or Crh DNA sequences. In contrast, METH produced DNA hypomethylation at sites near the Crh transcription start site (TSS) and at intragenic Avp sequences. METH also increased DNA hydroxymethylation at the Crh TSS and at intragenic Avp sites. In addition, METH increased the protein expression of ten-eleven-translocation enzymes that catalyze DNA hydroxymethylation. Importantly, METH increased TET1 binding at the Crh promoter and increased TET3 binding at Avp intragenic regions. We further tested the role of TET enzymes in METH-induced changes in gene expression by using the TET inhibitor, 1,5-isoquinolinediol (IQD), and found that IQD blocked METH-induced increases in Crh and Avp mRNA expression. Together, these results indicate that METH produced changes in neuropeptide transcription by both activation of the cAMP/CREB pathway and stimulation of TET-dependent DNA hydroxymethylation. These results provide molecular evidence for epigenetic controls of METH-induced changes in the expression of neuropeptides.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Methamphetamine (METH) is an addictive psychostimulant whose abuse is widespread throughout the world. Similar to other psychostimulants, its rewarding properties are due to activation of the reward pathway that projects from the ventral tegmental area (VTA) to diverse brain structures including the nucleus accumbens (NAc) [1, 2]. Perturbations in this pathway may be involved in promoting relapses [3]. Importantly, because some neurotransmitters may be regulators of various addiction stages that include initiation, escalation, and recurrent relapses [4], pharmacological manipulations of these brain systems may offer opportunities to treat addiction [5, 6]. In fact, addiction appears to stimulate diverse pathways that may serve as a coincident signal to promote compulsive drug-taking behaviors [7, 8]. Some of these pathways may use neuropeptides that are known to respond to stress, as recently described in a microarray paper that documented METH-induced increases in the expression of Crh/Crf, Avp, and Cartpt in the NAc [8]. These peptides are known participants in stress responses in several brain regions [9, 10]. Indeed, the NAc is known to be activated by stress [11], possibly through the regulation of CRF since CRF-containing cell bodies are located in that structure [12, 13].
Exposure to psychostimulants has now been shown to result in epigenetic modifications in several regions of the brain [8, 14]. These epigenetic events include covalent histone modifications, DNA methylation, and expression of non-coding RNAs [15, 16]. Although most of these studies have investigated the effects of cocaine, a few reports have begun to identify a role for epigenetic mechanisms in METH-induced changes in the brain. For example, Ikegami et al. (2010) [17] showed that METH increased methylation of histone H3 at lysine 4 (H3K4me3) in the NAc. METH also produced differential effects on histone acetylation in the NAc, decreased histone deacetylase HDAC1 protein levels, and increased HDAC2 expression [18]. METH-induced conditioned place preference (CPP) increased histone H3 di- and trimethylation at lysine 4 (H3K4me2 and me3) but decreased H3K27me2 abundance [19]. Moreover, METH increased the expression of the DNA methyltransferase, DNMT1 [20, 21]. Interestingly, parental METH exposure produced DNA hypermethylation at 70 loci but DNA hypomethylation at 39 loci in the hippocampus of F1 mice [22]. More recently, we have shown that abstinence from METH SA is associated with increased hydroxymethylation in a subgroup of rats [23]. Taken together, these observations support the notion that METH may significantly alter biochemical pathways and behaviors via diverse epigenetic mechanisms including DNA methylation/demethylation processes.
DNA hypermethylation of cytosines adjacent to guanines (CpG sites) is thought to be responsible for transcription repression while DNA hypomethylation is more often associated with increased gene expression [24]. Demethylation is generated by the oxidation of 5-methylcytosine (5mC) to 5-hydroxymethylcytosine (5hmC) by ten-eleven translocation proteins [25] and by TET-dependent generation of 5-formylcytosine and carboxylcytosine followed by action of thymine-DNA glycosylase and base excision repair (BER) [26]. 5hmC is highly enriched in the adult brain [27] and appears to play a crucial role in the establishment of epigenetic states that promote gene expression and rapid behavioral adaptation [28].
As mentioned above, studies focusing on epigenetic neural adaptations in the NAc are important because that structure is an important link in the pathways that subsume drug-induced behavioral phenomena [29,30,31] including relapse [32]. Here, we report that METH increased Crh and Avp mRNA levels via increasing DNA hydroxymethylation around the Crh TSS and in Avp intragenic regions in the NAc. We found, in addition, that Crh TSS hydroxymethylation is mediated by TET1 while Avp hydroxymethylation is secondary to the actions of TET3. In contrast, METH-induced changes in Cartpt expression are mediated by increased binding of phosphorylated CREB (pCREB) at the Cartpt promoter. Together, these results support the notion that METH can produce a diversity of epigenetic changes in the NAc and identify DNA hydroxymethylation as a potentially important target for the development of anti-addictive drugs.
Methods and Materials
Animals and Drug Treatment
All animal treatments and procedures were approved by the National Institute of Drug Abuse Animal Care and Use Committee according to the Guide for the Care and Use of Laboratory Animals (ISBN 0-309-05377-3). Male Sprague-Dawley rats, 8–10 weeks old (Charles River Labs, Raleigh), weighing 250–300 g were housed in a humidity- and temperature-controlled (22.2 ± 0.2 °C) room with free access to food and water. The rats were group-housed (two rats per cage) with the regular light/dark schedule. All animals acclimated to the facility for 1 week. Following habituation, rats were randomly assigned to two groups (12 rats per group for each time point) and received intraperitoneal (i.p.) injections of saline or METH (10 mg/kg). This is a non-blinded study. To study the time-course effect of the METH injection, animals were euthanized at 1, 2, 3, or 4 weeks after the saline or drug injection. The euthanization procedure was completed at a consistent time of the day. NAc tissues were dissected and immediately frozen to be used in RT-qPCR, western blot analysis, methylcytosine DNA immunoprecipitation (MeDIP), and hydroxyMeDIP (hMeDIP) assays. For ChIP, MeDIP, and hMeDIP experiments, the NAc tissues were processed as described below.
For co-treatment with TET inhibitor, rats received an initial 1,5-isoquinolinediol (IQD) (3 mg/kg dissolved in DMSO, Santacruz) i.p. injection 4 h prior to either saline or to METH injections on day 1. Subsequently, IQD was injected on days 2, 4, and 6 days after the METH injection. During subsequent weeks (2nd, 3rd, and 4th week), two injections of IQD were given per week. The administered IQD dose was according to the published literature [33, 34]. This dose schedule resulted in four experimental groups (eight rats in each group): saline/saline (control); saline/METH; IQD (3 mg/kg)/saline (IQD + saline); IQD (3 mg/kg)/METH (IQD + METH).
Quantitative PCR Analysis of mRNA Levels
Total RNA was isolated from one NAc hemisphere using RNeasy Mini kit (Qiagen) (n = 8–12 rats per group). Unpooled total RNA (0.5 μg) isolated from NAc samples was reverse-transcribed with oligo dT primers using Advantage RT-for-PCR kit (Clontech). RT-qPCR was performed essentially as described previously [18, 35] with Roche LightCycler 480 II (Roche Diagnostics) using iQ SYBR Green Supermix (BioRad). For all RT-qPCR experiments, individual data were normalized using the corresponding 18S mRNA level. The results are reported as fold changes calculated as the ratios of normalized gene expression data for METH-treated groups (at various time points) in comparison to the control group (SS). Primers for Crh (forward sequence 5′-CCT CGC AGA ACA GTG-3′ and reverse sequence 5′-CCT CAG AAG GTG GAA GGT G-3′), Crhr1 (forward sequence-5′-GTC CCT GAC CAG CAA TGT TT-3′ and reverse sequence-5′-CGG AGT TTG GTC ATG AGG AT-3′), Crhr2 (forward sequence-5′-AAG GTC CTA GGA GTG ATC CGA TT-3′ and reverse sequence-5′-GGA GCC ACC AGA GAG TGC AG-3′), Avp (forward sequence-5′-GAG TGT CGA GAG GGT TT-3′ and reverse sequence-5′-GGC GAT GGC TCA GTA GA-3′), Avpr1a (forward sequence-5′-CGT GGG TCC CTT TCA TA-3′ and reverse sequence-5′-GAA ATT CTC ATC CCA GAC T-3′), Avpr1b (forward sequence-5′-GGC CTA CAT CAC CTG GA-3′ and reverse sequence-5′-GCA GAA GAT GAG GAC TTA T-3′), and Cartpt (forward sequence-5′-TGG GAA GAA GAG GGA CT-3′ and reverse sequence-5′-TAA TTT GCA CAT GCT TCC A-3′) were synthesized at the Synthesis and Sequencing Facility of Johns Hopkins University (Baltimore, MD).
Immunoblot Analysis
Western blot analyses were carried out from NAc protein lysates (n = 6 rats per group). Samples were homogenized separately in 10% w/v of ice-cold 10 mM HEPES buffer (pH 7.4) containing 10 mM KCl, 1.5 mM MgCl2, 1%-Igepal CA-630 supplemented with a Roche protease inhibitor cocktail tablet (Roche Diagnostics). The homogenate was centrifuged for 5 min at 1400 × g to pellet nuclear fraction. The nuclear fraction was re-suspended in a nuclear lysis buffer (20 mM HEPES, 25% glycerol, 840 mM NaCl, 1.5 mM MgCl2, 0.4 mM EDTA). The supernatant was the cytosolic fraction. Protein concentrations of the nuclear fractions were determined by a BCA assay (ThermoFisher Scientific) and were then denatured with sample buffer (62.5 mM Tris-HCl, 10% glycerol, 2% SDS, 0.1% bromophenol blue, and 50 mM DTT) at 100 °C for 5 min and subjected to SDS-PAGE. Proteins were electrophoretically transferred to Hybond-PVDF membrane (Amersham). Subsequently, the membranes were incubated overnight at 4 °C with specific antibodies against CREB and pCREB (Cell Signaling, #9197 and #9191), DNMT1 (Abcam, ab54759), DNMT3A, DNMT3B (Cell Signaling, #3598 and #2161), TET1, TET2, and TET3 (Santa Cruz, sc163443, sc136926, and sc139186). After incubation, the blots were washed with TBS containing 0.1% Tween-20. The membranes were then incubated with HRP-conjugated anti-rabbit, anti-mouse, or anti-goat secondary antibody for 1 h at room temperature. To confirm equal protein loading, the blots were reprobed with α-tubulin antibody (1:4000, 2 h at room temperature; Sigma). ECL plus chemiluminescent reagents (GE Healthcare) were used to detect protein expression. Signal intensity was measured with Carestream Molecular Imaging software. Experiments were done twice.
Chromatin Immunoprecipitation Assay
NAc tissue was rapidly removed from rat brains (n = 8–10 rats per group), minced to ~ 1-mm-sized pieces, and immediately cross-linked in 1% formaldehyde for 15 min at room temperature. The cross-linking reaction was stopped by adding glycine to a final concentration of 0.125 M. The tissue was washed five times in cold PBS containing a Roche protease inhibitor cocktail tablet (Roche Diagnostics) and 1 mM PMSF (Sigma). Tissues were then rapidly frozen on dry ice. The fixed NAc tissues were suspended in SDS lysis buffer (Millipore) containing the Roche protease inhibitor cocktail (Roche Diagnostics) and 1 mM PMSF and homogenized twice for 10 s. Each sample was transferred to TPX plastic tube (Diagenode) and sonicated (15 cycles, 30″ ON and 30″ OFF) using the Diagenode Bioruptor device. Afterwards, fragmentation was checked by gel analysis to confirm a sheared range of 300–600 bp. Dynabeads (Life Technologies) were incubated with 5 μg of ChIP antibodies directed against pCREB (Cell Signaling, #9191), TET1 (ActiveMotif, #61443), TET2 (Millipore, MABE462), and TET3 (Santa Cruz, sc139186). As a control, NAc tissues were also incubated with 5 μg nonimmune rabbit IgG (Millipore, 12–370) overnight at 4 °C.
Equal amounts of chromatin lysate (60 μg) were diluted with ChIP dilution buffer (Millipore) to a final volume of 1.5 mL, and 100 μL of the pre-immunoprecipitated lysate was saved as “input” for later normalization. The chromatin lysate was then immunoprecipitated with appropriate antibodies overnight at 4 °C. The beads were sequentially washed once with low salt, high salt, LiCl, and Tris-EDTA according to the manufacturer’s instructions. The DNA-protein complexes were then eluted from the beads with 500 μL of NaHCO3/SDS elution buffer. DNA and proteins were dissociated at 65 °C for 4 h under high-salt conditions, followed by RNase A treatment for 30 min at 37 °C and proteinase K treatment for 1 h at 55 °C. The DNA was then extracted with phenol/chloroform, precipitated with ethanol, and finally resuspended in 80 μL of 10 mM Tris pH 8.0.
qPCR was performed with ChIP specific primers for Crh, Avp, and Cartpt (see Table S1 for details), using Roche LightCycler (Roche Diagnostics) and with iQ SYBR Green (BioRad) monitoring. All PCR reactions were performed in duplicate and included negative controls (no DNA). LightCycler software was used to calculate standard curves calculated using serial dilutions (100–0.1 ng) of input genomic DNA.
Methylated DNA Immunoprecipitation and Hydroxymethylated DNA Immunoprecipitation
Genomic DNA was isolated from NAc tissue (n = 10 rats per group) by overnight proteinase k treatment, phenol-chloroform extraction, ethanol precipitation, and RNase digestion. Subsequently, 300 μL fractions of DNA (20 μg) were sheared by ultrasonic treatment using the Diagenode Bioruptor (12 cycles, 30 s “ON”, 30 s “OFF”) to obtain a fragment size between ~ 200–600 bp. After denaturation (10 min at 95 °C), 5 μg DNA was then immunoprecipitated overnight at 4 °C using 5 μL of mouse monoclonal anti-5mC antibody (Millipore, MABE146) for MeDIP assay or 5 μL of rabbit polyclonal anti-5hmC antibody (ActiveMotif, #39769) for hMeDIP assay in a final volume of 500 μL IP buffer (10 mM sodium phosphate (pH 7.0), 140 mM NaCl, 0.05% Triton X-100). We incubated the mixture with 80 μL of Dynabeads (Life Technologies) overnight at 4 °C and washed it three times with 700 μL of IP buffer. We then treated the beads with proteinase K for 3 h at 50 °C and recovered the methylated or hydroxymethylated DNA by phenol-chloroform extraction followed by ethanol precipitation. Sheared “input” DNA samples were collected prior to immunoprecipitation for subsequent comparison with immunoprecipitated DNA.
qPCR on MeDIP and hMeDIP Samples
We carried out qPCR reactions with 20 ng of input DNA and immunoprecipitated methylated or hydroxymethylated DNA. For qPCR reactions, we used the iQ SYBR Green PCR master mix (BioRad) and Roche thermal cycler (Roche Diagnostics). The primer sequences and location are shown in Table S1. Reactions were done in duplicates and standard curves were calculated on serial dilutions (100–0.1 ng) of input genomic DNA. To evaluate the relative enrichment of target sequences after MeDIP or hMeDIP, we calculated the ratios of the signals in the immunoprecipitated DNA versus input DNA.
Statistical Analysis
All the quantitative data are presented as mean + SEM. For time-course data comparing control and METH-treated groups at various time points, as well as for the experiment involving IQD (TET inhibitor co-treatment) and one-way ANOVA followed by Fisher’s PLSD, post hoc analysis was used to calculate significance for MeDIP, hMeDIP, and ChIP experiments, the data were assessed by unpaired Student’s t test (Stat View version 4.02). For all experiments, the null hypothesis was rejected at p < 0.05.
Results
METH Causes Long-Lasting Increases in Gene Expression of Crh, Avp, and Cartpt Neuropeptides
Injection of METH (10 mg/kg) produced substantial changes in gene expression in rats euthanized 1 month later, with 338 genes being upregulated and 165 being downregulated (Fig. 1a). Among these genes were stress-related Crh, Avp, and Cartpt that showed increased expression [36] (Fig. 1b). To measure potential METH-induced time-dependent changes in Crh, Avp, and Cartpt mRNA levels, we injected the rats with either saline or METH and euthanized the animals at 1, 2, 3, or 4 weeks after the saline or METH injection. Figure 1 shows the time course of Crh (Fig. 1c), Avp (Fig. 1d), and Cartpt (Fig. 1e) expression after the drug injection. METH caused time-dependent increases in the mRNA levels of all three genes. The increases in the expression of Crh [F(4,47) = 4.837, p = 0.0024] and Cartpt injection [F(4,51) = 2.727, p = 0.0392] were apparent after 2 weeks and remained elevated for the duration of the study (Fig. 1c and e). METH-induced increases in Avp expression were significant at 3 and 4 weeks after the injection [F(4,44) = 4.577, p = 0.0035] (Fig. 1d). We also measured mRNA levels of Crh and Avp receptors. Supplemental Fig. S1 shows these results. Crhr1 [F(4,48) = 4.134, p = 0.0059], Crhr2 [F(4,42) = 4.892, p = 0.0025], and Avpr1a [F(4,45) = 4.213, p = 0.0056] mRNA levels showed significant increases whereas Avpr1b [F(4,53) = 2.571, p = 0.0483] showed significant decrease at 1 and 4 weeks after METH injection (Fig. S1).
a Rats were treated with saline or METH (10 mg/kg), and microarray analyses (n = 6 rats per group) were performed 30 days later. The microarray data has been deposited in the NCBI database: GEO accession number GSE46717. b List of stress-related neuropeptide genes whose mRNA was significantly increased (greater than + 1.8-fold, p < 0.01) include Avp (10.8-fold), Avpr1a (8.12-fold), Cartpt (5.65-fold), and Crh (4-fold). METH produces time-dependent increased expression of c Crh, d Avp, and e Cartpt. Total RNA was extracted from the NAc (n = 8–12 rats per group), and quantitative PCR for Crh, Avp, and Cartpt was carried out as described in the text. The relative amounts of mRNA were normalized to 18S and quantified. Values represent means ± SEM of fold changes relative to the controls. Analysis of variance (ANOVA) followed by Fisher’s PLSD (protected least significance difference) was used to determine statistical significance. Key to statistics: *p < 0.05; **p < 0.01; and ***p < 0.001, in comparison to the control group
METH Increases Enrichment of pCREB on Cartpt Gene Promoter
Psychostimulant-induced changes in gene expression are mediated, in part, by a dopamine-cAMP-CREB (Cyclic AMP Response Element-Binding) pathway that involves CREB phosphorylation [37, 38]. CREB is also involved in regulating stress-induced gene expression [7]. We thus wondered if the injection of METH might increase phosphorylated CREB (pCREB) in a time-dependent fashion. To test this idea, we used specific antibodies to measure CREB and pCREB protein abundance and found no changes in CREB protein levels (Fig. 2a) but time-dependent increases in pCREB abundance in NAc nuclear fractions (Fig. 2b). The initial increase in pCREB abundance was apparent by 1 week (~ 1.7-fold) and reached 2.5-fold after 4 weeks injection [F(4,20) = 7.019, p = 0.0011]. We next tested if METH might increase pCREB recruitment onto Crh, Avp, and Cartpt CRE DNA sequences by performing ChIP-qPCR assays. Figure 2c illustrates that METH increased pCREB binding on the Cartpt gene promoter region but not on Crh or Avp CRE DNA sequences (see Table S1 for primer details).
METH increased CREB phosphorylation and pCREB abundance at the Cartpt gene promoter. Western blot analyses (n = 6 rats per group) revealed no METH-induced changes in a CREB protein levels but significant time-dependent increases in the abundance of b pCREB. Representative photomicrographs show results of three samples per time point. For quantification, the signal intensity was normalized to α-tubulin. Values represent means ± SEM of fold changes relative to the controls. Statistical analysis was performed as in Fig. 1. Key to statistics: *p < 0.05; **p < 0.01; and ***p < 0.001 in comparison to the control group. For ChIP assays, we used animals euthanized at the 30-day time points. ChIP assays were performed using an anti-pCREB (cell signaling), and quantitative PCR was performed as described in the text using specific ChIP primers directed at Crh, Avp, or Cartpt c promoter regions. d Primers used for PCR following ChIP using antibodies against pCREB are pictured in dotted arrows and were designed so that resulting PCR amplicons contain the corresponding CRE site. CRE site sequence: TGACGTCA. In Avp promoter, the CRE site presents only half of the binding sequence. CRE half-site sequence: CGTCA (see Table S1 for primer sequences). Values represent means ± SEM of fold changes relative to the controls. Unpaired Student’s t test was used for statistical analyses. Key to statistics: *p < 0.05; **p < 0.01; and ***p < 0.001 in comparison to the control group
DNA Methylation and Hydroxymethylation Regulate METH-Induced Expression of Crh and Avp
Because pCREB did not appear to play a role in Crh and Avp mRNA expression, we wondered if METH-induced DNA hypomethylation might be involved in the upregulation of these mRNAs. Analysis of METH-induced effects on DNA methylation status of the Crh and Avp genes by using MeDIP-qPCR assays revealed significant METH-induced DNA hypomethylation at the Crh promoter region [t = 2.209, p = 0.049] (Fig. 3a) and at a CpG-rich region within the Avp gene body [t = 2.615, p = 0.024] (Fig. 3b). However, when we measured the protein expression of DNA methyltransferases (DNMT1, DNMT3A, and DNMT3B) [39], we found that METH increased the protein expression of DNMT1 [F(4,20) = 35.897, p < 0.0001] but not the expression of DNMT3A and DNMT3B (Fig. 4a–c). Although these results are consistent with previous findings showing METH-induced increases in DNMT1 [20, 21], they suggested that the METH-induced DNA hypomethylation was most likely not secondary to decreased DNMT expression but probably to TET-induced DNA hydroxymethylation. TET proteins convert 5mC to 5hmC and 5fC (5-formyl cytosine) and 5caC (5-carboxyl cytosine) [25]. DNA hydroxymethylation is also known to be a stable epigenetic mark that regulates gene expression [40, 41]. We thus tested the possibility that METH might cause increased DNA hydroxymethylation in the Nac. We indeed detected drug-induced increased hMeDIP enrichment at the Crh TSS [t = − 5.618, p < 0.0001] and the Avp intragenic region [t = − 3.509, p = 0.0038] (Fig. 3c and d).
METH caused DNA hypomethylation (a and b) and increased DNA hydroxymethylation (c and d) at CpG-rich sites near the promoter of Crh and at CpG-rich Avp intragenic sites. For MeDIP and hMeDIP assay, we used animals euthanized at the 30-day time point (n = 10 rats per group). Antibodies directed against either 5mC (Millipore) or 5hmC (ActivMotif) were used to precipitate methylated or hydroxymethylated DNA. Relative enrichment of 5mC or 5hmC was calculated using real-time PCR with specific primers directed at the promoter region of Crh and at the intragenic Avp CpG rich site (+ 1.8Kb). e Primers used for MeDIP and hMeDIP assay are pictured in dotted arrows (see Table S1 for primer sequences). Values represent means ± SEM of fold enrichment relative to controls. Statistical significance was determined by unpaired Student’s t test. Key to statistics: *p < 0.05; **p < 0.01 in comparison to the control group
METH causes time-dependent increased DNMT1 protein expression. Western blot analyses (n = 6 rats per group) showed significant time-dependent increases in the protein levels of a DNMT1 and no change in b DNMT3A and c DNMT3B. Representative photomicrographs show results of three samples per time point. For quantification, the signal intensity was normalized to α-tubulin. Values represent means ± SEM of fold changes relative to the controls. Statistical significance for the different time points was compared by analysis of variance followed by Fisher’s PLSD. Key to statistics: *p < 0.05; **p < 0.01; ***p < 0.001 vs. control group
TET Proteins Mediate METH-Induced Alterations in DNA Hydroxymethylation
To test if TET proteins were involved in DNA hydroxymethylation, we measured TET1, TET2, and TET3 protein levels in nuclear fractions obtained from the NAc. There were significant time-dependent increases in TET1 and TET3 protein levels in the NAc after METH, with TET1 reaching 3.8-fold [F(4,20) = 9.055, p = 0.0005] (Fig. 5a) and TET3 reaching 2.5-fold increases [F(4,20) = 27.245, p < 0.0001] (Fig. 5c). TET2 expression only reached 1.4-fold [F(4,20) = 2.429, p = 0.0815] increases (Fig. 5b).
TET1 and TET3 respectively regulate METH-induced DNA hydroxymethylation at Crh and Avp DNA sequences (n = 8–10 rats per group). Western blot analyses (n = 6 rats per group) revealed significant METH-induced time-dependent changes in the protein levels of TET1 (a) and TET3 (c). TET2 protein (b) levels were only increased at 4 weeks after the METH injection. Representative photomicrographs show results of three samples per group. For quantification, the signal intensity was normalized to α-tubulin. Values represent means ± SEM of fold changes relative to controls. ANOVA followed by Fisher’s PLSD was used to detect significance level. Key to statistics: *p < 0.05; **p < 0.01; ***p < 0.001, in comparison to the control group. METH increased enrichment of TET1 on the Crh gene promoter (d) but not on the Avp intragenic site (g). There was, however, increased TET3 binding at the Avp intragenic region but (i) not on the Crh promoter (f). TET2 binding was not affected by METH (e, h). Values represent means ± SEM of fold enrichment relative to the controls. Statistical significance was determined by unpaired Student’s t test. Key to statistics: *p < 0.05, in comparison to the control group. Co-treatment with TET protein inhibitor, 1,5-isoquinolinediol (IQD) (n = 8 rats per group), blocked the METH-induced increases in Crh (j) and Avp mRNA levels (k). Values represent means ± SEM of fold changes relative to the controls. Measurements of mRNA levels and statistical analysis are described in Fig. 1. Key to statistics: *p < 0.05 vs. control group
To further investigate the role of TET proteins in METH-induced DNA hydroxymethylation, we used ChIP-PCR with specific antibodies against the TET proteins to measure TET binding to Avp and Crh (Fig. 5d–i). We found significant METH-induced increased enrichment of TET1 on the Crh promoter sequence [t = − 2.500, p = 0.0245] (Fig. 5d) and increased enrichment of TET3 on Avp intragenic CpG-rich site [t = − 2.228, p = 0.0428] (Fig. 5i). There were neither significant changes in TET2 (Fig. 5e) and TET3 (Fig. 5f) binding at the Crh promoter nor in TET1 (Fig. 5g) and TET2 (Fig. 5h) binding at the Avp intragenic site.
So far, our observations had indicated that METH increased Crh and Avp expression by TET-dependent DNA hydroxymethylation. To further test the role of TET in this process, we used the TET inhibitor, IQD [34], that prevents DNA demethylation and the accumulation of TET-induced DNA intermediates [33]. IQD alone did not influence Crh and Avp expression. However, IQD co-treatment with the psychostimulant significantly blocked METH-induced increased Crh (Fig. 5j) and Avp (Fig. 5k) mRNA levels.
Discussion
The present study shows that an injection of METH produced long-lasting alterations in the expression of Crh, Avp, and Cartpt mRNAs. These neuropeptides are involved in a diversity of neuroendocrine responses to stressful events [5, 42]. These results are consistent with the observations that a single injection of amphetamine (AMPH) can cause hypersecretion of plasma adrenocorticotropin hormone (ACTH) and corticosterone measured at 1 and 3 weeks [43]. We show, in addition, that METH increased pCREB binding at the Cartpt promoter but not at Crh and Avp promoters. In contrast, METH caused DNA hydroxymethylation at a CpG-rich region in the promoter of the Crh gene and at a CpG-rich intragenic site of the Avp gene. DNA hydroxymethylation is known to be catalyzed by TET enzymes [25]. Consistent with this fact, we also found that METH increased TET1 binding at the Crh promoter and TET3 binding at the Avp intragenic site. Importantly, TET inhibition with the drug, IQD, blocked METH-induced Crh and Avp mRNA overexpression. Our findings are consistent with the demonstration that METH-induced gene transcription is regulated by diverse epigenetic mechanisms that include post-translational histone modifications and DNA methylation [18, 20, 22, 38, 44].
The present observations further implicate a role of stress-related neuropeptides in the biochemical and molecular effects of drugs of abuse, at least, in the NAc. For example, the expression of Crh, Avp, and Cart is increased in various brain regions in response to a diversity of stressful events including administration of psychostimulants [42, 45,46,47]. Our observations of increased NAc Crh expression after the METH injection are consistent with previous studies that have reported increased Crh in several brain regions after withdrawal of several drugs of abuse [10, 48,49,50] including increased levels in the amygdala after METH withdrawal [51]. A potential role of Crh in METH-induced molecular and behavioral changes is supported by the fact that administration of CP-154-526, a Crh receptor 1 antagonist, interferes with METH-induced reinstatement of extinguished METH-seeking behavior [52]. In addition to Crh, we also found increases in Avp and Cartpt mRNA levels. Avp has previously been implicated in the acquisition of cocaine-seeking behavior [53] and cocaine withdrawal [54]. Cart peptide is also increased in reward-related brain subregions following administration of various psychostimulants [55,56,57]. Our results are also consistent with the report of increased Cartpt mRNA in the NAc of victims of cocaine overdose [58]. Increased Cartpt is thought to participate in the regulation of the biochemical effects of dopamine in the NAc [55,56,57]. This suggestion is supported by behavioral studies showing that injection of Cart peptide into the NAc reduces the rewarding effects of cocaine [59] and blunts AMPH-induced locomotor activity and sensitization [60, 61]. Although the behavioral effects of these neuropeptides have been well documented, the epigenetic mechanisms that control their expression remained to be clarified.
In this context, we first explore the potential role of CREB phosphorylation in the METH-induced increases in the expression of these genes because pCREB is known to regulate psychostimulant-induced gene expression [38, 62]. Other studies have also implicated pCREB in the regulation of Crh, Avp, and Cartpt expression [63,64,65]. Surprisingly, we found that pCREB binding was increased only on the Cartpt promoter, indicating that METH had increased Cartpt expression via the dopamine-PKA-pCREB pathway in a manner consistent with a direct dopaminergic input to Cart-peptide-containing neurons in the NAc [66]. In addition to CREB phosphorylation, recent studies have implicated DNA methylation in the regulation of Crh [67, 68] and of Avp [69] expression. The latter reports are consistent with our demonstration of METH-induced DNA hypomethylation on the Crh promoter and at the CpG-rich region within the Avp gene body. The drug-induced DNA hypomethylation is consistent with our observations of METH-induced increased DNA hydroxymethylation at the Crh and Avp DNA sequences (see Fig. 3). Importantly, DNA hydroxymethylation is thought to drive transcription of many genes [70, 71]. Hydroxymethylated DNA is very abundant in the brain where it coordinates transcriptional activity [27, 72]. Hydroxymethylcytosine (5hmC) is found in gene bodies and at intron-exon boundaries of synaptic plasticity-related genes [71, 73, 74]. Together, these facts suggest that this DNA marker may play an important role in the long-term effects of psychostimulant exposure including cocaine [75].
Our findings that METH increased TET1 binding on the Crh promoter and TET3 binding on the Avp intragenic region are of interest because they illustrate the possibility of differential binding of these enzymes at different loci of two genes that are co-regulated by METH. These observations are consistent with recent reports indicating that accumulation of 5hmC at promoter regions is driven by the actions of TET1 [76, 77] whereas intragenic of 5hmC is associated with the presence of TET3 [28, 78]. For example, overexpression of TET3 in mature olfactory neurons increased 5hmC levels in gene bodies that positively correlated with increased transcription [78]. Our pharmacological interventions with a TET inhibitor (IQD) that blocked the METH-induced changes in the expression of both Crh and Avp, suggesting this agent may be worth pursuing in animal models of addiction that includes stress-induced relapse to drug seeking. This statement is consistent with the report that IQD treatment can prevent impaired recognition memory measured in mice that have been exposed to a combination of stress and alcohol [79].
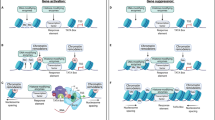
In summary, we have shown, for the first time, that TET-dependent DNA hydroxymethylation is the main determinant of Crh and Avp regulation in the rat NAc after injection of METH. Our results also specify TET1 and TET3 as the respective enzymes involved in the catalytic steps that produce DNA hydroxymethylation at Crh and Avp DNA sequences. These results shed light on the epigenetic mechanisms that link METH to neuropeptide expression in the rat NAc. The scheme in Fig. 6 illustrates these mechanisms. Furthermore, our demonstration that a TET inhibitor can block the transcriptional effects of METH indicates that such an approach may help to expand our armamentarium against METH addiction. This suggestion will need to be tested by assessing if these effects of METH can be expanded to self-administration models of addiction. The role of other stress-responsive systems including the extended amygdala will also need to be investigated in future experiments.
METH promotes diverse epigenetic modifications in the rat nucleus accumbens. The METH injection produced CREB phosphorylation and binding of pCREB on the CRE site on the Cartpt promoter and recruitment of the histone acetyl-transferase, CREB binding protein (CBP). This process leads to increased histone acetylation followed by increased Cartpt mRNA expression. On the other hand, METH increased TET1 binding at the Crh promoter and TET3 binding at an Avp intragenic site. The TET binding to these sequences promote DNA hydroxymethylation followed by increased Avp and Crh transcription. This statement is supported by the fact that the TET inhibitor, IQD, could prevent METH-induced increased Crh and Avp mRNA levels
References
Di Chiara G, Imperato A (1988) Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Natl Acad Sci U S A 85(14):5274–5278
Xi ZX, Kleitz HK, Deng X, Ladenheim B, Peng XQ, Li X, Gardner EL, Stein EA et al (2009) A single high dose of methamphetamine increases cocaine self-administration by depletion of striatal dopamine in rats. Neuroscience 161(2):392–402. https://doi.org/10.1016/j.neuroscience.2009.03.060
Koob GF, Nestler EJ (1997) The neurobiology of drug addiction. J Neuropsychiatry Clin Neurosci 9(3):482–497
Koob GF, Buck CL, Cohen A, Edwards S, Park PE, Schlosburg JE, Schmeichel B, Vendruscolo LF et al (2014) Addiction as a stress surfeit disorder. Neuropharmacology 76 part B:370–382. https://doi.org/10.1016/j.neuropharm.2013.05.024
Bisagno V, Cadet JL (2014) Stress, sex, and addiction: potential roles of corticotropin-releasing factor, oxytocin, and arginine-vasopressin. Behav Pharmacol 25(5–6):445–457. https://doi.org/10.1097/FBP.0000000000000049
Logrip ML, Koob GF, Zorrilla EP (2011) Role of corticotropin-releasing factor in drug addiction: potential for pharmacological intervention. CNS Drugs 25(4):271–287. https://doi.org/10.2165/11587790-000000000-00000
Briand LA, Blendy JA (2010) Molecular and genetic substrates linking stress and addiction. Brain Res 1314C. https://doi.org/10.1016/j.brainres.2009.11.002
Cadet JL (2014) Epigenetics of stress, addiction, and resilience: therapeutic implications. Mol Neurobiol. https://doi.org/10.1007/s12035-014-9040-y
Koob GF (2010) The role of CRF and CRF-related peptides in the dark side of addiction. Brain Res 1314:3–14. https://doi.org/10.1016/j.brainres.2009.11.008
Zorrilla EP, Logrip ML, Koob GF (2014) Corticotropin releasing factor: a key role in the neurobiology of addiction. Front Neuroendocrinol 35(2):234–244. https://doi.org/10.1016/j.yfrne.2014.01.001
Pliakas AM, Carlson RR, Neve RL, Konradi C, Nestler EJ, Carlezon WA Jr (2001) Altered responsiveness to cocaine and increased immobility in the forced swim test associated with elevated cAMP response element-binding protein expression in nucleus accumbens. J Neurosci 21(18):7397–7403
Joseph SA, Knigge KM (1983) Corticotropin releasing factor: immunocytochemical localization in rat brain. Neurosci Lett 35(2):135–141
Merchenthaler I (1984) Corticotropin releasing factor (CRF)-like immunoreactivity in the rat central nervous system Extrahypothalamic distribution. Peptides 5(Suppl 1):53–69
Walker DM, Cates HM, Heller EA, Nestler EJ (2015) Regulation of chromatin states by drugs of abuse. Curr Opin Neurobiol 30:112–121. https://doi.org/10.1016/j.conb.2014.11.002
Krishnan J, Mishra RK (2014) Emerging trends of long non-coding RNAs in gene activation. FEBS J 281(1):34–45. https://doi.org/10.1111/febs.12578
Meng H, Cao Y, Qin J, Song X, Zhang Q, Shi Y, Cao L (2015) DNA methylation, its mediators and genome integrity. Int J Biol Sci 11(5):604–617. https://doi.org/10.7150/ijbs.11218
Ikegami D, Narita M, Imai S, Miyashita K, Tamura R, Narita M, Takagi S, Yokomizo A et al (2010) Epigenetic modulation at the CCR2 gene correlates with the maintenance of behavioral sensitization to methamphetamine. Addict Biol 15(3):358–361. https://doi.org/10.1111/j.1369-1600.2010.00219.x
Martin TA, Jayanthi S, McCoy MT, Brannock C, Ladenheim B, Garrett T, Lehrmann E, Becker KG et al (2012) Methamphetamine causes differential alterations in gene expression and patterns of histone acetylation/hypoacetylation in the rat nucleus accumbens. PLoS One 7(3):e34236. https://doi.org/10.1371/journal.pone.0034236
Aguilar-Valles A, Vaissière T, Griggs EM, Mikaelsson MA, Takács IF, Young EJ, Rumbaugh G, Miller CA (2014) Methamphetamine-associated memory is regulated by a writer and an eraser of permissive histone methylation. Biol Psychiatry 76(1):57–65. https://doi.org/10.1016/j.biopsych.2013.09.014
Jayanthi S, McCoy MT, Chen B, Britt JP, Kourrich S, Yau HJ, Ladenheim B, Krasnova IN et al (2014) Methamphetamine downregulates striatal glutamate receptors via diverse epigenetic mechanisms. Biol Psychiatry 76(1):47–56. https://doi.org/10.1016/j.biopsych.2013.09.034
Numachi Y, Shen H, Yoshida S, Fujiyama K, Toda S, Matsuoka H, Sora I, Sato M (2007) Methamphetamine alters expression of DNA methyltransferase 1 mRNA in rat brain. Neurosci Lett 414(3):213–217. https://doi.org/10.1016/j.neulet.2006.12.052
Itzhak Y, Ergui I, Young JI (2014) Long-term parental methamphetamine exposure of mice influences behavior and hippocampal DNA methylation of the offspring. Mol Psychiatry. https://doi.org/10.1038/mp.2014.7
Cadet JL, Brannock C, Krasnova IN, Jayanthi S, Ladenheim B, McCoy MT, Walther D, Godino A et al (2017) Genome-wide DNA hydroxymethylation identifies potassium channels in the nucleus accumbens as discriminators of methamphetamine addiction and abstinence. Mol Psychiatry 22(8):1196–1204. https://doi.org/10.1038/mp.2016.48
Illingworth RS, Bird AP (2009) CpG islands—‘a rough guide’. FEBS Lett 583(11):1713–1720. https://doi.org/10.1016/j.febslet.2009.04.012
Hackett JA, Sengupta R, Zylicz JJ, Murakami K, Lee C, Down TA, Surani MA (2013) Germline DNA demethylation dynamics and imprint erasure through 5-hydroxymethylcytosine. Science (New York, NY) 339(6118). https://doi.org/10.1126/science.1229277
Scourzic L, Mouly E, Bernard OA (2015) TET proteins and the control of cytosine demethylation in cancer. Genome Med 7(1). https://doi.org/10.1186/s13073-015-0134-6
Kriaucionis S, Heintz N (2009) The nuclear DNA base, 5-hydroxymethylcytosine is present in brain and enriched in Purkinje neurons. Science (New York, NY) 324(5929):929–930. https://doi.org/10.1126/science.1169786
Li X, Wei W, Zhao Q-Y, Widagdo J, Baker-Andresen D, Flavell CR, D’Alessio A, Zhang Y et al (2014) Neocortical Tet3-mediated accumulation of 5-hydroxymethylcytosine promotes rapid behavioral adaptation. PNAS 111(19):7120–7125. https://doi.org/10.1073/pnas.1318906111
Britt JP, Benaliouad F, McDevitt RA, Stuber GD, Wise RA, Bonci A (2012) Synaptic and behavioral profile of multiple glutamatergic inputs to the nucleus accumbens. Neuron 76(4):790–803. https://doi.org/10.1016/j.neuron.2012.09.040
Everitt BJ, Robbins TW (2016) Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol 67:23–50. https://doi.org/10.1146/annurev-psych-122414-033457
Stuber GD, Sparta DR, Stamatakis AM, van Leeuwen WA, Hardjoprajitno JE, Cho S, Tye KM, Kempadoo KA et al (2011) Excitatory transmission from the amygdala to nucleus accumbens facilitates reward seeking. Nature 475(7356):377–380. https://doi.org/10.1038/nature10194
Rothwell PE, Kourrich S, Thomas MJ (2011) Synaptic adaptations in the nucleus accumbens caused by experiences linked to relapse. Biol Psychiatry 69(11):1124–1126. https://doi.org/10.1016/j.biopsych.2010.12.028
Dhliwayo N, Sarras MP, Luczkowski E, Mason SM, Intine RV (2014) Parp inhibition prevents ten-eleven translocase enzyme activation and hyperglycemia-induced DNA demethylation. Diabetes 63(9):3069–3076. https://doi.org/10.2337/db13-1916
Lupachyk S, Shevalye H, Maksimchyk Y, Drel VR, Obrosova IG (2011) PARP inhibition alleviates diabetes-induced systemic oxidative stress and neural tissue 4-hydroxynonenal adduct accumulation: correlation with peripheral nerve function. Free Radic Biol Med 50(10):1400–1409. https://doi.org/10.1016/j.freeradbiomed.2011.01.037
Cadet JL, McCoy MT, Cai NS, Krasnova IN, Ladenheim B, Beauvais G, Wilson N, Wood W et al (2009) Methamphetamine preconditioning alters midbrain transcriptional responses to methamphetamine-induced injury in the rat striatum. PLoS One 4(11):e7812. https://doi.org/10.1371/journal.pone.0007812
Cadet JL, Brannock C, Ladenheim B, McCoy MT, Krasnova IN, Lehrmann E, Becker KG, Jayanthi S (2014) Enhanced upregulation of CRH mRNA expression in the nucleus accumbens of male rats after a second injection of methamphetamine given thirty days later. PLoS One 9(1):e84665. https://doi.org/10.1371/journal.pone.0084665
Konradi C, Cole RL, Heckers S, Hyman SE (1994) Amphetamine regulates gene expression in rat striatum via transcription factor CREB. J Neurosci 14(9):5623–5634
Krasnova IN, Chiflikyan M, Justinova Z, McCoy MT, Ladenheim B, Jayanthi S, Quintero C, Brannock C et al (2013) CREB phosphorylation regulates striatal transcriptional responses in the self-administration model of methamphetamine addiction in the rat. Neurobiol Dis 58:132–143. https://doi.org/10.1016/j.nbd.2013.05.009
Feng J, Fan G (2009) The role of DNA methylation in the central nervous system and neuropsychiatric disorders. Int Rev Neurobiol 89:67–84. https://doi.org/10.1016/S0074-7742(09)89004-1
Frauer C, Hoffmann T, Bultmann S, Casa V, Cardoso MC, Antes I, Leonhardt H (2011) Recognition of 5-hydroxymethylcytosine by the Uhrf1 SRA domain. PLoS One 6(6):e21306. https://doi.org/10.1371/journal.pone.0021306
Hashimoto H, Liu Y, Upadhyay AK, Chang Y, Howerton SB, Vertino PM, Zhang X, Cheng X (2012) Recognition and potential mechanisms for replication and erasure of cytosine hydroxymethylation. Nucleic Acids Res 40(11):4841–4849. https://doi.org/10.1093/nar/gks155
Rogge G, Jones D, Hubert GW, Lin Y, Kuhar MJ (2008) CART peptides: regulators of body weight, reward and other functions. Nat Rev Neurosci 9(10):747–758. https://doi.org/10.1038/nrn2493
Vanderschuren LJMJ, Schmidt ED, Vries TJD, Moorsel CAPV, Tilders FJH, Schoffelmeer ANM (1999) A single exposure to amphetamine is sufficient to induce long-term behavioral, neuroendocrine, and neurochemical sensitization in rats. J Neurosci 19(21):9579–9586
Cadet JL, Jayanthi S, McCoy MT, Ladenheim B, Saint-Preux F, Lehrmann E, De S, Becker KG et al (2013) Genome-wide profiling identifies a subset of methamphetamine (METH)-induced genes associated with METH-induced increased H4K5Ac binding in the rat striatum. BMC Genomics 14:545. https://doi.org/10.1186/1471-2164-14-545
Hubert GW, Jones DC, Moffett MC, Rogge G, Kuhar MJ (2008) CART peptides as modulators of dopamine and psychostimulants and interactions with the mesolimbic dopaminergic system. Biochem Pharmacol 75(1):57–62. https://doi.org/10.1016/j.bcp.2007.07.028
Naughton M, Dinan TG, Scott LV (2014) Chapter 5—corticotropin-releasing hormone and the hypothalamic–pituitary–adrenal axis in psychiatric disease. In: Eric Fliers MK, Johannes AR (eds) Handbook of clinical neurology, vol 124. Elsevier, Clinical Neuroendocrinology, pp. 69–91
Smith SM, Vaughan JM, Donaldson CJ, Rivier J, Li C, Chen A, Vale WW (2004) Cocaine- and amphetamine-regulated transcript activates the hypothalamic-pituitary-adrenal axis through a corticotropin-releasing factor receptor-dependent mechanism. Endocrinology 145(11):5202–5209. https://doi.org/10.1210/en.2004-0708
George O, Ghozland S, Azar MR, Cottone P, Zorrilla EP, Parsons LH, O'Dell LE, Richardson HN et al (2007) CRF–CRF1 system activation mediates withdrawal-induced increases in nicotine self-administration in nicotine-dependent rats. PNAS 104(43):17198–17203. https://doi.org/10.1073/pnas.0707585104
Sarnyai Z, Shaham Y, Heinrichs SC (2001) The role of corticotropin-releasing factor in drug addiction. Pharmacol Rev 53(2):209–244
Zorrilla EP, Valdez GR, Weiss F (2001) Changes in levels of regional CRF-like-immunoreactivity and plasma corticosterone during protracted drug withdrawal in dependent rats. Psychopharmacology 158(4):374–381. https://doi.org/10.1007/s002130100773
Nawata Y, Kitaichi K, Yamamoto T (2012) Increases of CRF in the amygdala are responsible for reinstatement of methamphetamine-seeking behavior induced by footshock. Pharmacol Biochem Behav 101(2):297–302. https://doi.org/10.1016/j.pbb.2012.01.003
Moffett MC, Goeders NE (2006) CP-154,526, a CRF type-1 receptor antagonist, attenuates the cue-and methamphetamine-induced reinstatement of extinguished methamphetamine-seeking behavior in rats. Psychopharmacology 190(2):171–180. https://doi.org/10.1007/s00213-006-0625-7
Rodríguez-Borrero E, Rivera-Escalera F, Candelas F, Montalvo J, Muñoz-Miranda WJ, Walker JR, Maldonado-Vlaar CS (2010) Arginine vasopressin gene expression changes within the nucleus accumbens during environment elicited cocaine-conditioned response in rats. Neuropharmacology 58(1):88–101. https://doi.org/10.1016/j.neuropharm.2009.06.040
Zhou Y, Litvin Y, Piras AP, Pfaff DW, Kreek MJ (2011) Persistent increase in hypothalamic arginine vasopressin gene expression during protracted withdrawal from chronic escalating-dose cocaine in rodents. Neuropsychopharmacology 36(10):2062–2075. https://doi.org/10.1038/npp.2011.97
Fagergren P, Hurd YL (1999) Mesolimbic gender differences in peptide CART mRNA expression: effects of cocaine. Neuroreport 10(16):3449–3452
Ogden CA, Rich ME, Schork NJ, Paulus MP, Geyer MA, Lohr JB, Kuczenski R, Niculescu AB (2004) Candidate genes, pathways and mechanisms for bipolar (manic–depressive) and related disorders: an expanded convergent functional genomics approach. Mol Psychiatry 9(11):1007–1029. https://doi.org/10.1038/sj.mp.4001547
Rogge GA, Jones DC, Green T, Nestler E, Kuhar MJ (2009) Regulation of CART peptide expression by CREB in the rat nucleus accumbens in vivo. Brain Res 1251:42–52. https://doi.org/10.1016/j.brainres.2008.11.011
Albertson DN, Pruetz B, Schmidt CJ, Kuhn DM, Kapatos G, Bannon MJ (2004) Gene expression profile of the nucleus accumbens of human cocaine abusers: evidence for dysregulation of myelin. J Neurochem 88(5):1211–1219. https://doi.org/10.1046/j.1471-4159.2003.02247.x
Jaworski JN, Hansen ST, Kuhar MJ, Mark GP (2008) Injection of CART (cocaine- and amphetamine-regulated transcript) peptide into the nucleus accumbens reduces cocaine self-administration in rats. Behav Brain Res 191(2):266–271. https://doi.org/10.1016/j.bbr.2008.03.039
Kim JH, Creekmore E, Vezina P (2003) Microinjection of CART peptide 55–102 into the nucleus accumbens blocks amphetamine-induced locomotion. Neuropeptides 37(6):369–373. https://doi.org/10.1016/j.npep.2003.10.001
Kim S, Yoon HS, Kim J-H (2007) CART peptide 55-102 microinjected into the nucleus accumbens inhibits the expression of behavioral sensitization by amphetamine. Regul Peptides 144(1–3):6–9. https://doi.org/10.1016/j.regpep.2007.07.003
Cornish JL, Hunt GE, Robins L, McGregor IS (2012) Regional c-Fos and FosB/ΔFosB expression associated with chronic methamphetamine self-administration and methamphetamine-seeking behavior in rats. Neuroscience 206:100–114. https://doi.org/10.1016/j.neuroscience.2012.01.004
Adler GK, Smas CM, Fiandaca M, Frim DM, Majzoub JA (1990) Regulated expression of the human corticotropin releasing hormone gene by cyclic AMP. Mol Cell Endocrinol 70(2):165–174
Dominguez G, Lakatos A, Kuhar MJ (2002) Characterization of the cocaine- and amphetamine-regulated transcript (CART) peptide gene promoter and its activation by a cyclic AMP-dependent signaling pathway in GH3 cells. J Neurochem 80(5):885–893. https://doi.org/10.1046/j.0022-3042.2002.00775.x
Greenwood M, Bordieri L, Greenwood MP, Melo MR, Colombari DSA, Colombari E, Paton JFR, Murphy D (2014) Transcription factor CREB3L1 regulates vasopressin gene expression in the rat hypothalamus. J Neurosci 34(11):3810–3820. https://doi.org/10.1523/jneurosci.4343-13.2014
Smith Y, Kieval J, Pr C, Mj K (1999) CART peptide-immunoreactive neurones in the nucleus accumbens in monkeys: ultrastructural analysis, colocalization studies, and synaptic interactions with dopaminergic afferents. J Comp Neurol 407(4):491–511. https://doi.org/10.1002/(sici)1096-9861(19990517)407:4<491::aid-cne3>3.0.co;2-0
Chen J, Evans AN, Liu Y, Honda M, Saavedra JM, Aguilera G (2012) Maternal deprivation in rats is associated with corticotrophin-releasing hormone (CRH) promoter hypomethylation and enhances CRH transcriptional responses to stress in adulthood. J Neuroendocrinol 24(7):1055–1064. https://doi.org/10.1111/j.1365-2826.2012.02306.x
Elliott E, Ezra-Nevo G, Regev L, Neufeld-Cohen A, Chen A (2010) Resilience to social stress coincides with functional DNA methylation of the Crf gene in adult mice. Nat Neurosci 13(11):1351–1353. https://doi.org/10.1038/nn.2642
Murgatroyd C, Patchev AV, Wu Y, Micale V, Bockmühl Y, Fischer D, Holsboer F, Wotjak CT et al (2009) Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat Neurosci 12(12):1559–1566. https://doi.org/10.1038/nn.2436
Hahn Maria A, Qiu R, Wu X, Li Arthur X, Zhang H, Wang J, Jui J, Jin S-G et al (2013) Dynamics of 5-hydroxymethylcytosine and chromatin marks in mammalian neurogenesis. Cell Rep 3(2):291–300. https://doi.org/10.1016/j.celrep.2013.01.011
Mellén M, Ayata P, Dewell S, Kriaucionis S, Heintz N (2012) MeCP2 binds to 5hmC enriched within active genes and accessible chromatin in the nervous system. Cell 151(7):1417–1430. https://doi.org/10.1016/j.cell.2012.11.022
Globisch D, Münzel M, Müller M, Michalakis S, Wagner M, Koch S, Brückl T, Biel M et al (2010) Tissue distribution of 5-hydroxymethylcytosine and search for active demethylation intermediates. PLoS One 5(12):e15367. https://doi.org/10.1371/journal.pone.0015367
Booth MJ, Branco MR, Ficz G, Oxley D, Krueger F, Reik W, Balasubramanian S (2012) Quantitative sequencing of 5-methylcytosine and 5-hydroxymethylcytosine at single-base resolution. Science 336(6083):934–937. https://doi.org/10.1126/science.1220671
Khare T, Pai S, Koncevicius K, Pal M, Kriukiene E, Liutkeviciute Z, Irimia M, Jia P et al (2012) 5-hmC in the brain is abundant in synaptic genes and shows differences at the exon-intron boundary. Nat Struct Mol Biol 19(10):1037–1043. https://doi.org/10.1038/nsmb.2372
Feng J, Shao N, Szulwach KE, Vialou V, Huynh J, Zhong C, Le T, Ferguson D et al (2015) Role of Tet1 and 5-hydroxymethylcytosine in cocaine action. Nat Neurosci 18(4):536–544. https://doi.org/10.1038/nn.3976
Kaas Garrett A, Zhong C, Eason Dawn E, Ross Daniel L, Vachhani Raj V, G-l M, King Jennifer R, Song H et al (2013) TET1 controls CNS 5-methylcytosine hydroxylation, active DNA demethylation, gene transcription, and memory formation. Neuron 79(6):1086–1093. https://doi.org/10.1016/j.neuron.2013.08.032
Xu Y, Wu F, Tan L, Kong L, Xiong L, Deng J, Barbera AJ, Zheng L et al (2011) Genome-wide regulation of 5hmC, 5mC, and gene expression by Tet1 hydroxylase in mouse embryonic stem cells. Mol Cell 42(4):451–464. https://doi.org/10.1016/j.molcel.2011.04.005
Colquitt BM, Allen WE, Barnea G, Lomvardas S (2013) Alteration of genic 5-hydroxymethylcytosine patterning in olfactory neurons correlates with changes in gene expression and cell identity. PNAS 110(36):14682–14687. https://doi.org/10.1073/pnas.1302759110
Pant R, Jangra A, Kwatra M, Singh T, Kushwah P, Bezbaruah BK, Gurjar SS, Phukan S (2017) Cognitive deficits induced by combined exposure of stress and alcohol mediated through oxidative stress-PARP pathway in the hippocampus. Neurosci Lett 653:208–214. https://doi.org/10.1016/j.neulet.2017.05.058
Acknowledgements
This work was supported by funds of the Intramural Research Program of the US Department of Health and Human Services/National Institutes of Health/National Institute on Drug Abuse.
Author information
Authors and Affiliations
Contributions
The study conception and design were created by JLC and SJ. SJ wrote the NIDA/IRP-ACUC protocol. SJ, BG, and MTM worked together in the acquisition of data including ChIP and DNA methylation assays. Data analysis and interpretation of data correlation were performed by SJ and BG. BL performed the animal injections and dissection. SJ and JLC prepared the manuscript. All authors contributed to critical revisions of the paper.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Jayanthi, S., Gonzalez, B., McCoy, M.T. et al. Methamphetamine Induces TET1- and TET3-Dependent DNA Hydroxymethylation of Crh and Avp Genes in the Rat Nucleus Accumbens. Mol Neurobiol 55, 5154–5166 (2018). https://doi.org/10.1007/s12035-017-0750-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0750-9