Abstract
Objective
Positron emission tomography (PET)/computed tomography (CT) using 68Ga-labeled 1,4,7,10-tetraazacyclododecane-N,N′,N″,N‴-tetraacetic acid-d-Phe1-Tyr3-octreotide (DOTATOC) has been used to detect neuroendocrine tumors (NETs). The purpose of this study was to investigate the clinical efficacy of DOTATOC-PET/CT for detecting clinically suspected NETs when conventional imaging modalities were negative or inconclusive, in terms of additional value.
Methods
A total of 46 patients were analyzed retrospectively. Among them, 14 patients underwent a DOTATOC-PET/CT scan for detecting unknown primary tumors after histopathological confirmation of a NET at metastatic sites (group A): 7 patients for detecting metastasis or recurrence after surgery for NET because of their high hormone levels but with no recurrence detected by other imaging modalities (group B); the remaining 25 patients for detecting suspected NETs because their hormone levels were high with no history of histopathologically proven NET (group C). Additional information was assessed, according to each situation.
Results
In group A, unknown primary tumors were suspected by DOTATOC-PET/CT in 8 of 14 patients (gastrointestinal/pancreatic NET in 7 patients, prostatic cancer in 1 patient), but prostatic cancer was not confirmed by histopathology (i.e., false positive). In group B, DOTATOC-PET/CT depicted lesions in six of seven patients, including nodal metastasis (n = 5) and liver metastasis (n = 1). In group C, DOTATOC-PET/CT did not demonstrate any abnormal foci except in one case of pancreatic NET. Additional information was obtained in 50, 86, and 4 % of cases, in groups A, B, and C, respectively.
Conclusions
DOTATOC-PET/CT was useful for detecting NETs, especially when recurrence or metastases were suspected because of high hormone levels after surgery for a NET. It is unlikely, however, that additional information can be acquired in patients with no history of NET simply based on high hormone levels.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Positron emission tomography computed tomography using 18F-fluorodeoxyglucose (FDG-PET/CT) has been widely accepted for clinical tumor imaging [1]. Primary or metastatic lesions are depicted as a hypermetabolic area of glucose in many cancers, which is helpful for staging, restaging, and monitoring the response to therapy. Despite dramatic spread of FDG-PET/CT use during the past decade, neuroendocrine tumors (NETs), which commonly express somatostatin receptors, are often not FDG-avid, for which conventional FDG-PET/CT does not always provide useful information for therapeutic management. For these tumors, 111In-octreotide has been used in Europe and the United States for somatostatin receptor scintigraphy [2, 3], but it is not routinely available in Japan.
68Ga-labeled octreotide, including 68Ga-labeled 1,4,7,10-tetraazacyclododecane-N,N′,N″,N‴-tetraacetic acid-d-Phe1-Tyr3-octreotide (DOTATOC), has been used as a PET tracer for somatostatin receptor imaging mainly in Europe [4–6]. Compared with 111In-labeled compounds, more promising results have been reported, especially for detecting lung or bone metastasis [7]. There have already been a few articles demonstrating the clinical efficacy of DOTATOC-PET/CT for unknown primary NETs [8, 9]. The data are limited, however, and the value of PET/CT with 68Ga-labeled compounds clinically has not been investigated in Japan, although there has been one article that elucidated characteristics of NETs quantitatively with 111In-labeled octreotide investigation [10]. Because it has been reported that there are large differences in the features of gastroenteropancreatic NETs in Japan and Western countries [11], the clinical efficacy of DOTATOC-PET/CT should be evaluated in a Japanese population.
The aim of this study was to evaluate the clinical usefulness of PET/CT using 68Ga-DOTATOC in NET patients with suspected unknown primary or recurrent tumors in each situation when other conventional imaging modalities were negative or inconclusive, from a view point of additional information.
Patients and methods
Patients
A total of 46 consecutive patients (male/female ratio 12:34; mean age 56 years, range 27–80 years) who underwent DOTATOC-PET/CT at our institution were analyzed. The patients were divided into three groups according to their scanning records. In group A, PET/CT was performed after metastatic NETs were confirmed histopathologically, but primary tumors had not been identified by other imaging modalities. In group B, DOTATOC-PET/CT scan was performed to evaluate suspected recurrent lesions because of a high level of gastrin (n = 5), insulin (n = 1), or chromogranin A (n = 1) after they underwent curative surgery for histologically proven NETs. Conventional imaging tests were negative. In group C, NETs had been suspected based on laboratory data without definitive localization of primary sites by conventional imaging modalities. In this group, patients had been suspected of having NET because of high levels of gastrin (n = 9), adrenocorticotropic hormone (ACTH) (n = 8), insulin (n = 5), 5-hydroxyindoleacetic acid (n = 2), or chromogranin A (n = 1). We checked all patients who had not taken a long-acting octreotide analogue within 1 month before their DOTATOC-PET/CT scan. The ethics committee of our institution approved the study, and written informed consent was obtained from all patients.
PET scanning
PET/CT scanning was performed using a combined PET/CT scanner (Discovery ST Elite; GE Healthcare, Waukesha, WI, USA). This system integrates a PET scanner with multidetector-row CT (16 detectors) and permits acquisition of co-registered CT and PET images during a single examination. For DOTATOC-PET/CT scanning, approximately 130 MBq of 68Ga-DOTATOC was injected intravenously, and whole-body PET/CT scanning was performed approximately 60 min after injection. Just before the PET/CT scanning, patients were requested to void the bladder. Initially, starting at the level of the thigh, the low-dose CT scans were acquired during shallow breathing, and scanning included the area from the upper thigh to the skull. Immediately after CT, a PET emission scan was acquired with an acquisition time of 3 min per bed position. For one patient in group A and two patients in group C, diagnostic CT scans were also performed 30 and 90 s after intravenous administration of 100 mL iopromide (Iopamiron 300; Nihon Beyer, Osaka, Japan) with an injection rate of 3 mL/s. The total acquisition time was 20–30 min. Unenhanced CT data were used for attenuation correction, and images were reconstructed using a commercial three-dimensional iterative reconstruction algorithm called VUE Point Plus (matrix 128 × 128, interval 3.27 mm, 2 iterations, 14 subsets).
Image analysis
PET/CT images were interpreted visually by at least two board-certified radiologists/nuclear medicine physicians based on consensus. Focal moderate-to-intense uptake was regarded as abnormal, but accumulations of DOTATOC in some organs (e.g., pituitary gland, adrenal gland, accessory spleen) that were considered physiological [12] were excluded. As a quantitative analysis, standardized uptake values (SUVs) were calculated for each lesion if patients had abnormal foci, but quantitative values were not used for diagnostic criteria in this investigation. The findings were compared with the final diagnoses obtained by histopathological confirmation or clinical follow-up for at least 6 months.
Results
Patients’ profiles and PET/CT findings are summarized in Table 1.
Group A
There were 14 patients who had been suspected of having a primary NET because of pathologically proven liver metastasis (n = 9), nodal metastasis (n = 3), or bone metastasis (n = 2). In four of nine patients with metastatic liver tumors, DOTATOC-PET/CT demonstrated positive findings, such as a suspected primary tumor in the duodenum (n = 2), jejunum (n = 1), and pancreatic tail (n = 1) with the maximum SUV (SUVmax) ranging from 2.8 to 19.7. One case in which a duodenal submucosal tumor was confirmed to be a primary NET is shown in Fig. 1. DOTATOC-PET/CT showed no abnormal findings for the remaining five patients. In three patients with nodal metastasis, DOTATOC-PET/CT revealed abnormal uptake in the duodenum (n = 1) and jejunum (n = 2). For two patients with bone metastasis, DOTATOC-PET/CT was negative in one patient but showed intense focal uptake in the prostate, suggesting prostatic cancer. In the other patient, the uptake was confirmed by biopsy to be due to benign prostatic hypertrophy. The primary site remained unknown. Thus, a final diagnosis of a gastroenteropancreatic NET was obtained in 7 of 14 patients (50 %) histopathologically (n = 3) and clinically (n = 4).
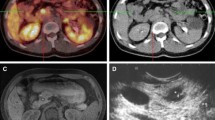
A 74-year-old woman had a metastatic liver tumor. A maximum intensity projection DOTATOC-PET image (a) and a DOTATOC-PET/CT transaxial fused image (b) are shown. A liver tumor was resected prior to DOTATOC-PET/CT, with the histopathologic confirmation of a neuroendocrine tumor (NET). A primary site had not been identified by conventional imaging modalities, including enhanced CT, MRI, and FDG-PET/CT. DOTATOC-PET/CT revealed intense uptake corresponding to the duodenal wall (arrows). Biopsy was performed, and NET G2 was confirmed histopathologically
Group B
Seven patients in group B underwent surgery for a NET. They had undergone DOTATOC-PET/CT. Except for one patient with a high insulin level, DOTATOC-PET/CT depicted 10 lesions in six patients (seven nodal metastases in five patients, three liver metastases in one patient). The SUVmax of these lesions ranged from 7.9 to 70.1. Two patients had histopathological confirmation after surgery, whereas the remaining four patients were under follow-up with no surgical treatment. Thus, DOTATOC-PET/CT provided additional information in six of seven patients (86 %). A representative case of nodal metastasis is shown in Fig. 2.
A 37-year-old man had a history of surgical resection of a gastrinoma. DOTATOC-PET with a maximum intensity projection image (a) and CT, DOTATOC-PET, and DOTATOC-PET/CT transaxial images (b) are demonstrated. During the follow-up period, the patient’s serum gastrin level increased, but recurrence had not been identified on CT. Focal intense DOTATOC uptake corresponds to a subcentimeter node (arrows), which was confirmed as a nodal metastasis during surgery
Group C
A total of 25 patients who had been suspected of having NETs underwent DOTATOC-PET/CT. One patient with suspected ACTH-producing tumors underwent DOTATOC-PET/CT, which identified a pancreatic NET with SUVmax of 68.5 (Fig. 3), followed by surgical confirmation. For the remaining 24 patients, DOTATOC-PET/CT did not provide any clinically relevant additional information. The detection rate was significantly lower than in other groups (Fisher’s exact test, p < 0.01).
A 51-year-old woman had a suspected ectopic ACTH-producing tumor. CT, DOTATOC-PET, and DOTATOC-PET/CT transaxial images are shown. Focal intense uptake is observed in the pancreatic tail (arrow), suggesting a pancreatic NET that had not been identified by unenhanced CT. Surgery was performed, supplying pathological confirmation of NET G2
Discussion
We demonstrated the clinical usefulness of 68Ga-DOTATOC-PET/CT when a primary or metastatic lesion had been suspected, according to three situations. Additional information would be expected especially when hormone levels are high after surgery for histologically proven NETs, as is often the case with FDG-PET/CT being performed for restaging FDG-avid tumors. Its clinical value was limited, however, when hormone levels were high in patients without a history of NETs. According to meta-analyses [13, 14], the diagnostic performance of DOTATOC-PET/CT has been excellent, but it is thought to be influenced by clinical scenarios in which DOTATOC-PET/CT scans are performed.
Articles have been published that are limited to cases in which NETs had been confirmed histopathologically without identifying a primary tumor (corresponding to our group A) [8, 9]. Prasad et al. [8] reported that DOTANOC-PET/CT identified the primary tumor in 35 of 59 patients (59 %) with documented NETs but an unknown primary. In their report, the primary sites were mainly in the pancreas (n = 16), followed by the small intestine (n = 14). Naswa et al. [9] demonstrated that DOTANOC-PET/CT found 12 of 20 primary tumors (60 %) in NET patients with unknown primary tumors. The primary sites were mainly in the midgut (four in the duodenum, four in the ileum, one in the colon). In our series, DOTATOC-PET/CT revealed primary sites in half of the patients, which is similar to the data from previous reports. Regarding detected primary sites, our findings were consistent with the results by Naswa et al. Pancreatic lesions were well evaluated by contrast-enhanced CT and magnetic resonance imaging at our institution. Therefore, it was a small number of cases in which the primary sites were located in the pancreas in this investigation.
One male patient had a bone metastasis from a NET, which was confirmed histopathologically. Prostatic cancer was suspected as the primary tumor because of intense uptake in the prostate gland on DOTATOC-PET/CT. Histopathological evaluation of biopsy tissue, however, did not confirm a prostatic cancer. It may be because of a sampling error, but DOTATOC accumulation in the prostate was extensive, and eight specimens were acquired, covering almost the entire prostate. This case was therefore regarded as giving false-positive results. It is known that somatostatin receptors are expressed not only in normal prostate glands, but also in the presence of prostatic cancer or prostatic hypertrophy [15, 16]. It is thought that accumulation in the prostate would be a pitfall when interpreting DOTATOC-PET/CT images.
DOTATOC-PET/CT was useful when hormone levels were high after surgery was undertaken for NETs. This situation is similar to that of cases in which FDG-PET was conducted because of suspected recurrence or metastasis after surgery with a high level of tumor markers. As the pretest probability is high and other imaging modalities (e.g., conventional CT or FDG-PET/CT) are negative, it is reasonable that DOTATOC-PET/CT would be expected to yield relevant information. As demonstrated in Fig. 2, a subcentimeter node can be detected by CT, but it is difficult to distinguish it from a benign inflammatory node. In addition, NET tumors usually grow slowly. The high accumulation of DOTATOC in a subcentimeter node is helpful for detecting a local recurrence or metastasis after NET surgery.
On the other hand, without a history of NET, DOTATOC-PET/CT might not provide helpful information simply because hormone levels are high. Some reasons can be considered. First, primary sites may be too small to be detected by morphologic imaging modalities. If lesions are extremely small, uptake tends to be underestimated because of a partial volume effect with PET. Also, when the primary site is in the upper abdomen or alimentary tract, the uptake could be easily influenced by respiratory motion and peristalsis. Second, it is not guaranteed that there is a primary site because hyperfunctioning can cause high hormone levels (e.g., nesidioblastosis in suspected with an insulinoma or G-cell hyperplasia with a suspected gastrinoma). In addition, it has been reported that somatostatin receptor subtypes 2 and 5 are not well expressed in many insulinomas [17]. Thus, DOTATOC-PET/CT may have failed to depict the primary tumor.
One patient had been suspected of having an ACTH-producing tumor. Before DOTATOC-PET/CT scanning, the patient had undergone unenhanced CT, but enhanced CT performed after DOTATOC-PET/CT revealed a tumor in the pancreatic tail. Therefore, DOTATOC-PET/CT may be useful in such patients with high hormone levels if enhanced CT cannot be performed for some reason, including renal dysfunction.
There are some limitations in this investigation. First, the sample size is small. Also, histopathological confirmation was not obtained in all cases, so there may have been some false-positive cases despite the fact that DOTATOC-PET/CT provided additional information. It is not ethically acceptable to acquire histopathologic confirmation of every lesion. Because this study was retrospective, the imaging modalities before DOTATOC-PET/CT became available were varied. For this reason, there may be selection bias in our population. The scan times ranged from 57 to 82 min (average 65 min) after injection of 68Ga-DOTATOC. Based on the procedure guidelines, these scan times are considered normal after tracer administration [18], but the most optimal scanning time remains unknown. A different scanning time may alter the results.
Conclusion
Our data indicate that DOTATOC-PET/CT provided additional useful information, especially when recurrence or metastases were suspected because of high hormone levels after surgery for a primary NET. It did not yield any relevant information, however, in cases in which only the hormone levels were high.
References
Tagliabue L, Del Sole A. Appropriate use of positron emission tomography with [(18)F]fluorodeoxyglucose for staging of oncology patients. Eur J Intern Med. 2014;25:6–11.
Rambaldi PF, Cuccurullo V, Briganti V, Mansi L. The present and future role of (111)In pentetreotide in the PET era. Q J Nucl Med Mol Imaging. 2005;49:225–35.
Ambrosini V, Fani M, Fanti S, Forrer F, Maecke HR. Radiopeptide imaging and therapy in Europe. J Nucl Med. 2011;52(Suppl 2):42S–55S.
Gabriel M, Decristoforo C, Kendler D, Dobrozemsky G, Heute D, Uprimny C, et al. 68 Ga-DOTA-Tyr3-octreotide PET in neuroendocrine tumors: comparison with somatostatin receptor scintigraphy and CT. J Nucl Med. 2007;48:508–18.
Poeppel TD, Binse I, Petersenn S, Lahner H, Schott M, Antoch G, et al. 68 Ga-DOTATOC versus 68 Ga-DOTATATE PET/CT in functional imaging of neuroendocrine tumors. J Nucl Med. 2011;52:1864–7.
Yang J, Kan Y, Ge BH, Yuan L, Li C, Zhao W. Diagnostic role of Gallium-68 DOTATOC and Gallium-68 DOTATATE PET in patients with neuroendocrine tumors: a meta-analysis. Acta Radiol. 2014;55:389–98.
Buchmann I, Henze M, Engelbrecht S, Eisenhut M, Runz A, Schäfer M, et al. Comparison of 68 Ga-DOTATOC PET and 111In-DTPAOC (Octreoscan) SPECT in patients with neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2007;34:1617–26.
Prasad V, Ambrosini V, Hommann M, Hoersch D, Fanti S, Baum RP. Detection of unknown primary neuroendocrine tumours (CUP-NET) using (68)Ga-DOTA-NOC receptor PET/CT. Eur J Nucl Med Mol Imaging. 2010;37:67–77.
Naswa N, Sharma P, Kumar A, Soundararajan R, Kumar R, Malhotra A, et al. 68Ga-DOTANOC PET/CT in patients with carcinoma of unknown primary of neuroendocrine origin. Clin Nucl Med. 2012;37:245–51.
Kubota K, Okasaki M, Minamimoto R, Miyata Y, Morooka M, Nakajima K, et al. Lesion-based analysis of (18)F-FDG uptake and (111)In-Pentetreotide uptake by neuroendocrine tumors. Ann Nucl Med. 2014;28:1004–10.
Ito T, Sasano H, Tanaka M, Osamura RY, Sasaki I, Kimura W, et al. Epidemiological study of gastroenteropancreatic neuroendocrine tumors in Japan. J Gastroenterol. 2010;45:234–43.
Kroiss A, Putzer D, Decristoforo C, Uprimny C, Warwitz B, Nilica B, et al. 68 Ga-DOTA-TOC uptake in neuroendocrine tumour and healthy tissue: differentiation of physiological uptake and pathological processes in PET/CT. Eur J Nucl Med Mol Imaging. 2013;40:514–23.
Treglia G, Castaldi P, Rindi G, Giordano A, Rufini V. Diagnostic performance of Gallium-68 somatostatin receptor PET and PET/CT in patients with thoracic and gastroenteropancreatic neuroendocrine tumours: a meta-analysis. Endocrine. 2012;42:80–7.
Geijer H, Breimer LH. Somatostatin receptor PET/CT in neuroendocrine tumours: update on systematic review and meta-analysis. Eur J Nucl Med Mol Imaging. 2013;40:1770–80.
Reubi JC, Waser B, Schaer JC, Markwalder R. Somatostatin receptors in human prostate and prostate cancer. J Clin Endocrinol Metab. 1995;80:2806–14.
Hansson J, Bjartell A, Gadaleanu V, Dizeyi N, Abrahamsson PA. Expression of somatostatin receptor subtypes 2 and 4 in human benign prostatic hyperplasia and prostatic cancer. Prostate. 2002;53:50–9.
Portela-Gomes GM, Stridsberg M, Grimelius L, Rorstad O, Janson ET. Differential expression of the five somatostatin receptor subtypes in human benign and malignant insulinomas—predominance of receptor subtype 4. Endocr Pathol. 2007;18:79–85.
Virgolini I, Ambrosini V, Bomanji JB, Baum RP, Fanti S, Gabriel M, et al. Procedure guidelines for PET/CT tumour imaging with 68 Ga-DOTA-conjugated peptides: 68 Ga-DOTA-TOC, 68 Ga-DOTA-NOC, 68 Ga-DOTA-TATE. Eur J Nucl Med Mol Imaging. 2010;37:2004–10.
Acknowledgments
We are grateful for support from a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan (25461816).
Conflict of interest
There are no potential conflicts of interest relevant to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nakamoto, Y., Sano, K., Ishimori, T. et al. Additional information gained by positron emission tomography with 68Ga-DOTATOC for suspected unknown primary or recurrent neuroendocrine tumors. Ann Nucl Med 29, 512–518 (2015). https://doi.org/10.1007/s12149-015-0973-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-015-0973-7