Abstract
Introduction
Choroidal thickness (CT) plays an important role in the pathogenesis of various ocular diseases, including neovascular age-related macular degeneration (nAMD). Previous studies evaluated the CT variations after anti-vascular endothelial growth factor (VEGF) injections in patients with nAMD, but the results are still controversial. The present study aimed to evaluate the CT at different times (15, 30, 60, 90, and 365 days) after intravitreal aflibercept injections and its correlation with the baseline CT in treatment-naïve patients with nAMD. Secondly, the study evaluated the correlation between CT variation at 365 days and the number of intravitreal injections received.
Methods
This was a prospective, open-label, single-arm pilot study. Twenty-one treatment-naïve nAMD eyes were enrolled. The study population underwent three monthly aflibercept injections (loading phase) and additional injections as needed (pro re nata regimen). A complete ophthalmological examination, including optical coherence tomography (OCT) was performed at each visit. CT was measured manually by two independent observers. All patients were evaluated at baseline and at 15, 30, 60, 90, and 365 days after the first intravitreal injection.
Results
CT showed a statistically significant reduction at days 15, 90, and 365 in comparison to baseline. However, the major reduction of CT was observed at day 15 and in eyes with a thicker choroid at baseline. No significant correlation between CT variation and the number of injections performed was found.
Conclusion
Our findings contribute to clarifying the role of aflibercept injections in choroidal vasculature, confirming its effect after the first 2 weeks. Moreover, CT can be considered as a potential biomarker, as it reflects the pharmacological effect of anti-VEGF drugs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Why carry out this study? |
Age-related macular degeneration (AMD) is the major cause of vision impairment and loss in elderly people, but thanks to anti-vascular endothelial growth factor (VEGF) therapy, blindness associated with neovascular AMD has been markedly decreased. |
Choroidal thickness is a valid parameter to assess during anti-VEGF therapy administration since it shows variations in relation to anti-VEGF injections. However, the results on this topic are still controversial. |
We evaluated choroidal thickness in treatment-naïve patients with nAMD receiving aflibercept injections in order to better investigate its effect on the choroidal vasculature. |
What was learned from the study? |
Choroidal thickness showed a reduction in comparison to baseline at 15, 90, and 365 days after the first injection in treatment-naïve patients with nAMD receiving three monthly aflibercept injections followed by a pro re nata regimen. |
The major reduction was observed at 15 days from the first injection. Similarly, literature data suggest a maximum reduction of the macular neovascularization (MNV) 2 weeks after the anti-VEGF injection. |
This finding might be attributable to a reduction of the choroidal vascular permeability and, thus, to a decrease in MNV activity. Moreover, a greater reduction was found in patients with a thicker choroid at baseline. |
Our results confirm the effect of aflibercept on eye vasculature after the first weeks and open up new horizons to tailored treatments for nAMD based on choroidal thickness assessment. |
Introduction
As a result of longer life expectancy and general ageing of populations, age-related macular degeneration (AMD) is a major cause of vision impairment and blindness in elderly people and its prevalence has markedly increased within the past 2 decades [1]. Although the dry form is more common among the patients affected, the neovascular form has a more rapid and aggressive development, with a significant impact on quality of life.
With the introduction of anti-vascular endothelial growth factor (VEGF) therapy in 2006, the proportion of visually impaired eyes with neovascular AMD (nAMD) dropped and recent population-based data have suggested that legal AMD-related blindness has been reduced by 50% in some countries [1]. Medications such as aflibercept represent the most effective treatment for acute nAMD and help to prevent the abnormal growth of new immature blood vessels, which often leads to leakage of fluid and/or blood into the subretinal space, the neural retina itself, or the subretinal pigment epithelium (RPE) space.
Accordingly with these advances in the management, AMD imaging has encountered an important evolution with optical coherence tomography (OCT) and, more recently, with OCT-angiography (OCT-A). OCT-A is a non-invasive technique able to detect macular neovascularization (MNV) with the same sensitivity of dye angiography.
Regarding the choroid, evidence in the literature suggests that choroidal thickness plays an important role in the physiology of the eye and in the pathogenesis of various ocular diseases, including AMD. Previous studies evaluated variations in the choroidal thickness after the loading phase (three monthly injections) and after 1 year, or even more, of individualized regimens (treat and extend, T&E; pro re nata, PRN) in treatment-naïve patients with nAMD, but results are still controversial and potential changes at 15 days from the first aflibercept injection have not been previously investigated. Considering that several OCT-A studies [2,3,4] have reported a maximum reduction of the MNV flow area and vessel density 2 weeks after the anti-VEGF injection, we hypothesized a similar behavior also for the choroid. Thus, we analyzed choroidal thickness at 15, 30, 60, 90, and 365 days from the first aflibercept injection, in order to investigate the effect of aflibercept on choroidal vasculature.
Methods
The current study was approved by the Ethics Committee/Institutional Review Board of the Catholic University under protocol number 1189 with the acronym TWINS PLUS. The study was designed as a prospective, open-label, pilot, single-arm study. Twenty-one eyes of 21 Caucasian patients [female (F)/male (M) 10:11] affected by nAMD and naïve to treatment were enrolled. The mean age was 82.1 years ± 5.9.
The inclusion criteria were the following:
-
1.
Male or female over 50 years old, who has signed informed consent before any treatment was performed.
-
2.
Clinical and instrumental diagnosis of neovascular AMD with active MNV.
-
3.
Eyes affected by nAMD with no history of intravitreal injections prior to the study (treatment-naϊve).
-
4.
MNV or related sequelae (i.e., pigment epithelial detachment (PED), subretinal or subretinal pigment epithelial hemorrhage, blockade of fluorescence, macular edema, subretinal or sub-RPE or intraretinal fluid) which involves the center of the fovea.
The exclusion criteria were the following:
-
1.
Any concomitant ocular disorders in the studied eye that might have confounded the interpretation of the results.
-
2.
Compromised visual acuity (VA) or conditions requiring medical or surgical interventions, including cataracts, retinal vascular occlusion, retinal detachment, macular holes, and MNV due to any cause other than AMD (e.g., ocular histoplasmosis and pathologic myopia).
-
3.
Any use of eye drops, such as anti-glaucomatous or anti-inflammatory drugs, for ocular diseases; any intraocular surgery in the studied eye within 3 months prior to enrollment; previous vitrectomy.
-
4.
History of current myocardial infarction or cerebral vascular diseases, any drugs that possibly interfered with choroidal thickness, such as sildenafil and acetazolamide.
-
5.
The inability to comply with study procedures.
The primary endpoint was to evaluate the choroidal thicknesses at different times (15, 30, 60, 90, and 365 days) following intravitreal aflibercept injections and their correlation with the baseline choroidal thickness in patients with nAMD naïve to treatment.
The secondary endpoint was to evaluate the correlation between choroidal thickness variation at 365 days and the number of intravitreal injections received.
Before starting the anti-VEGF treatment, a baseline visit was performed to select the eligible patients. All the included patients were treated with three monthly consecutive aflibercept injections (loading phase) and additional intravitreal injections, given reactively in an OCT-guided PRN protocol. Choroidal thickness evaluations were assessed at baseline and at 15, 30, 60, 90, and 356 days after the first aflibercept injection. In consideration of the choroidal circadian rhythm, we chose to perform the OCT assessment between 10 and 12 a.m. for each patient. The aflibercept injections were performed immediately after the OCT scans at baseline and at 30 and 60 days. The OCT device was a Spectralis HRA + OCT (Heidelberg Engineering, Heidelberg, Germany) with enhanced depth imaging (EDI).
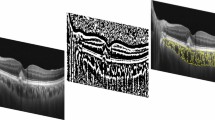
The choroidal thickness was manually measured with the caliper function on horizontal B-scans at seven points from the posterior edge of the RPE to the choroid–sclera junction at 500-μm intervals up to 1.500 μm temporal and nasal to the fovea, according to the technique of Coscas et al. [5]. An example of the measurement technique is illustrated in Fig. 1.
Standard dilated ophthalmic examinations of both eyes were performed at each visit using a slit lamp and indirect stereo-ophthalmoscope. The pupils were dilated with eye drops (e.g., tropicamide) as necessary.
The measurements were performed twice by two independent observers trained in retinal-imaging analysis, and the average was used as a single entry for the statistical analysis.
Statistical Analysis
The statistical analysis was performed using the software package R (4.0.3 release). Choroidal thicknesses at different times were analyzed through a visual inspection of the qq-plot, followed by a Shapiro–Wilk test. Choroidal thicknesses were measured by two distinct operators and their results were first compared using a Wilcoxon test for paired samples, then the agreement between the two operators was investigated by using a Bland–Altman analysis and a Passing–Blablok regression. Subsequent analysis was carried out using the average between the two operators.
The variations in choroidal thickness at baseline and 15, 30, 60, 90, and 365 days were analyzed with a repeated-measures analysis of variance (ANOVA) test, followed by post hoc analysis with a Bonferroni test. Choroidal thickness change as a function of the intravitreal injections performed was investigated with a one-way ANOVA test.
The relationship between continuous variables was investigated using a simple linear model. The normality of the residues was checked with a Shapiro–Wilk test and through a visual inspection of the residue plot. The best regression line was reported together with 95% confidence and prediction bands. Correlation between continuous variables was assessed using Pearson’s coefficient r. The ggplot2 package was used to visualize data. Linear regressions with the corresponding confidence bands and ANOVA analysis were performed with the R function lm and aov, respectively.
Results
Demographic data and mean choroidal thicknesses at baseline are summarized in Table 1.
The first independent observer measured the following mean choroidal thicknesses: 190.51, 167.88, 173.13, 171.08, 171.21, and 165.56 μm at baseline and at 15, 30, 60, 90, and 365 days after the first intravitreal injection, respectively. The second independent observer measured the following mean choroidal thicknesses: 181.15, 159.54, 169.17, 170.51, 163.14, and 161.93 μm at the same timepoints.
The agreement between the measurements performed by the two operators was investigated by the Bland–Altman analysis and the Passing–Blablok regression. The Bland–Altman analysis suggested the presence of a positive and statistically significant bias of 5.65 µm (95% CI 1.81–9.49) between the two operators. The Passing–Blablok regression provided a partial confirmation of Bland–Altman results regarding the existence of a positive bias, but the analysis showed that its extent is not clinically relevant in the thickness range considered. Therefore, the two operators can be considered equivalent. For this reason, we chose to conduct the analysis with the choroidal thickness values obtained from the mean measurements of the two operators.
A quantitative comparison of the results at different times was performed using the one-way ANOVA test for repeated measures which considers that the measurements were registered on the same set of patients. The current analysis suggested the presence of statistically significant differences between choroidal thicknesses at different times (p = 0.00051), although the percentage of variance explained by the time variable was only a few percentage units. A post hoc analysis carried out with the Bonferroni correction highlighted the presence of statistically significant differences between baseline and 15 days, between baseline and 90 days, and finally between baseline and 365 days. Figure 2 displays the mean choroidal thickness for each patient at different times (baseline, 15, 30, 60, 90, and 365 days). At all timepoints, data appear strongly scattered as a result of the inter-individual variability among the subjects recruited. Although the analysis is not conclusive, it suggests that aflibercept treatment may result in choroidal thickness reduction, as shown in Fig. 3.
Data also indicated a correlation between choroidal thickness decrease at 15 days and choroidal thickness prior to the treatment (Pearson correlation r = 0.445, p = 0.046). Data were analyzed using a linear regression model and the regression line [y = (− 5.4 ± 13.6) ns + (0.15 ± 0.07) x] is shown in Fig. 4 with confidence and prediction bands. According to the results of Pearson correlation, the regression line shows a positive and significant slope, meaning that thicker choroids at baseline were characterized by a greater reduction at 15 days. Thus, a thicker choroid may be predictive of a greater response to aflibercept. Although the regression analysis can be considered reliable (residuals distribution does not show significant deviations from normality), the prediction bands (dashed curves) are extremely large and therefore it is not possible to predict an outcome following the treatment.
The mean number of intravitreal injections performed over 365 days was 5 ± 1.4 and data showed no statistically significant correlation between choroidal thickness variation within the follow-up period and the number of injections performed.
Our results in terms of mean choroidal thickness at different times and number of injections for each patient of the study are summarized in Table 2.
All the eyes showed a reduction of the MNV activity by reduction in intraretinal cystic spaces and/or decrease in intraretinal hemorrhage after intravitreal injections. During the entire study, no signs or symptoms related to safety were detected, and all changes in concomitantly used medications were recorded. During the study period, no serious adverse events or adverse events related to the study treatments occurred.
Discussion
As one of the most highly vascular tissues in the body, the choroid is fundamental to maintain normal visual function and it also plays a key role in the pathophysiology of common chorioretinal diseases such as AMD [6, 7]. As the macular region has the highest metabolic activity of the retina, the underlying choroid has the greatest volume [8] and reflects the vascular status of the whole choroid. Thus, subfoveal choroidal thickness represents a valid parameter to assess choroidal health, especially in patients with nAMD during anti-VEGF therapy administration. Indeed, in these patients excessive choroidal thinning may be an undesired effect which can also lead to deterioration in the choriocapillaris–RPE complex and development of macular atrophy (MA) [9].
Aflibercept is designed to block both VEGF-A and placental growth factor which are involved in the development of MNV, but also in important physiological functions such as angiogenesis, microvascular permeability, and survival of vascular endothelial cells. For this reason, aflibercept may affect not only MNV but also choroidal vessels.
In the last few years, choroidal thickness changes during aflibercept administration have been investigated. Several research groups focused on short-term effects, reporting a significant reduction at 30, 60, and 90 days following aflibercept in treatment-naïve patients with nAMD [10,11,12]. A decrease in choroidal thickness was also found in pre-treated patients [13], but the effect was less marked than in treatment-naïve patients [11, 14]. Sariyeva Ismayılov et al. [14] suggested that this result could be attributed to higher amounts of VEGF and upregulated VEGF receptors in treatment-naïve patients.
Pellegrini et al. [15] found a significant reduction both in choroidal thickness and in choroidal vascularity index (CVI), calculated as the ratio of luminal area to the total choroidal area, after three monthly aflibercept injections in treatment-naïve nAMD. They proposed that this finding may be a consequence of VEGF suppression which leads to vasoconstriction and to a reduction in the choriocapillaris endothelial cell fenestrations. Another possibility is that choroidal changes after anti-VEGF treatment may be secondary to the suppression of the MNV activity and leakage.
Long-term effects of aflibercept injections in patients treated with individualized regimens (T&E or PRN regimens) after a loading dose of three monthly injections were described. Koizumi et al. [16] measured subfoveal choroidal thickness (SCT) variations at 3 and 12 months and found a decrease at 3 months which remained unchanged at 12 months, although there was some fluctuation in between treatments. Reductions in choroidal thickness at 12, 24, 36, and 48 months were assessed by other research groups [17,18,19,20,21].
To the best of our knowledge, choroidal thickness changes after 15 days from the first aflibercept injection had not yet been investigated. In the literature, there is evidence [2,3,4] of a cyclic variation of MNV in response to anti-VEGF therapy with a maximum reduction of the MNV 2 weeks after the first injection. Hypothesizing a parallel behavior between the MNV and the choroid, we decided to investigate choroidal thickness at 15 days from the aflibercept injection and we found that choroidal thickness showed a major decrease at this time. This reduction might be attributable to a reduction in choroidal vascular permeability and, thus, to a decrease in MNV activity.
In the subsequent measurements (30, 60, 90, and 365 days), choroidal thickness showed a statistically significant reduction at 90 and 365 days, albeit the reduction was not as significant as at day 15. Indeed, the maximum reduction of choroidal thickness was observed at day 15, with a progressive return to values closer to baseline (even if the pre-treatment choroidal thickness was never reached). However, similarly to previous reports, we found a slight fluctuation of choroidal thickness in between treatments [16].
It is interesting to note that at day 30 choroidal thickness already showed higher values compared to day 15. A similar trend was observed in MNV vessels with a reproliferation after 30 days from the injection[2,3,4] because of arteriogenesis, which is not VEGF dependent like angiogenesis [22].
The analysis of the results was probably complicated by the non-homogeneous time intervals of the measurements and by the different number of injections performed over 356 days. The non-homogeneous distribution of the evaluations did not allow us to assess if a transient effect of aflibercept exists. Indeed, choroidal thickness could reduce immediately after aflibercept injection and then return to values previous to the treatment within, for example, a month. This hypothesis would help explain the heterogeneity of the results, as well as the greater effect observed at 15 days. This aspect could be particularly relevant and deserves a study based on more frequent monitoring in the interval between subsequent treatments.
Regarding long-term effects (365 days), our results are in agreement with the literature evidence. However, previously published reports analyzed the choroidal response to aflibercept in the Asian population [17,18,19,20,21]. The current study described the results in a sample of Caucasians.
According to other research groups, the effect of aflibercept on the choroid is controversial. Barakat et al. [23] reported no significant change in SCT at 12 and 24 months in treatment-naïve patients with nAMD. Similarly, Kumar et al. [24] observed that choroidal thickness did not change significantly with aflibercept injections in treatment-naïve patients and that there was no relationship between choroidal thickness and other OCT biomarkers or VA.
Moreover, the response of the choroid could be related to the type of anti-VEGF used. In a previous study [25] we found no significant reduction in choroidal thickness in treatment-naïve patients with nAMD receiving ranibizumab. Other reports showed choroidal thickness reductions in patients with nAMD treated with ranibizumab [26,27,28], but according to some authors [12, 13, 29] aflibercept seems to have a more significant effect on choroidal thickness compared to ranibizumab. Although there are several reports regarding the effects of aflibercept and ranibizumab on the choroid, choroidal thickness changes after intravitreal injection of bevacizumab have not been studied to a great extent. Sizmaz et al. [30] found that neither ranibizumab nor bevacizumab caused significant change in choroidal thickness at 1 month post-injection, while Ünlü et al. [31] reported a significant subfoveal choroidal thickness reduction after intravitreal injection of bevacizumab with a peak after the first week.
Since choroidal thinning could lead to MA, these findings may raise some concern about the possible increased risk of progression to MA in patients treated with aflibercept rather than with other anti-VEGF medications. However, the RIVAL study did not show any statistically significant differences between aflibercept and ranibizumab in determining MA in patients with nAMD treated over 24 months [32]. Although results in the literature are still inconclusive, the risk of MA in patients with nAMD treated with anti-VEGF therapies seems to be associated with the specific features of the MNV rather than with the treatment regimen or the anti-VEGF medication [33,34,35]. On the basis of the currently available data, what is certain is that undertreatment rather than overtreatment would be a greater risk for visual loss in patients with MNV [33, 36,37,38].
Conclusion
The results of the current study increase our understanding of the pharmacological effect of aflibercept on the choroid, opening to the possibility of tailored treatments for patients with nAMD. Early choroidal response to aflibercept injections confirms its prompt efficacy on eye vasculature already from the first weeks. Moreover, choroidal thickness could potentially reflect disease activity, becoming a potentially useful biomarker to monitor the disease and to prevent disease recurrence. However, the current study has several limitations, such as the limited number of cases and the manual assessment of choroidal thickness. Furthermore, choroidal thickness evaluation could be supported by the evaluation of CVI, an emerging imaging tool which provides additional insights into choroidal angio-architecture with less variability. Unfortunately, a complete and good visualization of the entire choroid is necessary for both automated and semiautomated CVI analysis, hence artifacts due to OCT signal attenuation and projection represent an important limitation [39]. Further studies with automated software could be useful to obtain a more objective evaluation of the choroid and to support our findings elucidating their clinical significance.
References
Jonas JB, Cheung CMG, Panda-Jonas S. Updates on the epidemiology of age-related macular degeneration. Asia Pac J Ophthalmol (Phila). 2017. https://doi.org/10.22608/apo.2017251.
Marques JP, Costa JF, Marques M, Cachulo ML, Figueira J, Silva R. Sequential morphological changes in the CNV net after intravitreal anti-VEGF evaluated with OCT angiography. Ophthalmic Res. 2016;55(3):145–51.
Lumbroso B, Rispoli M, Savastano MC. Longitudinal optical coherence tomography-angiography study of type 2 naive choroidal neovascularization early response after treatment. Retina. 2015;35(11):2242–51.
Huang D, Jia Y, Rispoli M, Tan O, Lumbroso B. Optical coherence tomography angiography of time course of choroidal neovascularization in response to anti-angiogenic treatment. Retina. 2015;35(11):2260–4.
Coscas G, Zhou Q, Coscas F, et al. Choroid thickness measurement with RTVue optical coherence tomography in emmetropic eyes, mildly myopic eyes, and highly myopic eyes. Eur J Ophthalmol. 2012;22(6):992–1000.
Harris A, Chung HS, Ciulla TA, Kagemann L. Progress in measurement of ocular blood flow and relevance to our understanding of glaucoma and age-related macular degeneration. Prog Retin Eye Res. 1999;18(5):669–87.
Ciulla TA, Harris A, Chung HS, et al. Color Doppler imaging discloses reduced ocular blood flow velocities in nonexudative age-related macular degeneration. Am J Ophthalmol. 1999;128(1):75–80.
Manjunath V, Goren J, Fujimoto JG, Duker JS. Analysis of choroidal thickness in age-related macular degeneration using spectral-domain optical coherence tomography. Am J Ophthalmol. 2011;152(4):663–8.
Lipecz A, Miller L, Kovacs I, et al. Microvascular contributions to age-related macular degeneration (AMD): from mechanisms of choriocapillaris aging to novel interventions. GeroScience. 2019;41(6):813–45.
Koizumi H, Kano M, Yamamoto A, et al. Short-term changes in choroidal thickness after aflibercept therapy for neovascular age-related macular degeneration. Am J Ophthalmol. 2015;159(4):627-633.e1.
Mazaraki K, Fassnacht-Riederle H, Blum R, Becker M, Michels S. Change in choroidal thickness after intravitreal aflibercept in pretreated and treatment-naive eyes for neovascular age-related macular degeneration. Br J Ophthalmol. 2015;99(10):1341–4.
Yun C, Oh J, Ahn J, et al. Comparison of intravitreal aflibercept and ranibizumab injections on subfoveal and peripapillary choroidal thickness in eyes with neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2016;254(9):1693–702.
Gharbiya M, Cruciani F, Mariotti C, Grandinetti F, Marenco M, Cacace V. Choroidal thickness changes after intravitreal antivascular endothelial growth factor therapy for age-related macular degeneration: ranibizumab versus aflibercept. J Ocul Pharmacol Ther. 2015;31(6):357–62.
Sariyeva Ismayılov A, Esen E, Sızmaz S, Demircan AN. Aflibercept therapy in eyes with neovascular age-related macular degeneration and its effect on choroidal thickness. Clin Exp Optom. 2019;102(6):617–20.
Pellegrini M, Bernabei F, Mercanti A, et al. Short-term choroidal vascular changes after aflibercept therapy for neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2021;259(4):911–8.
Koizumi H, Kano M, Yamamoto A, et al. Subfoveal choroidal thickness during aflibercept therapy for neovascular age-related macular degeneration: twelve-month results. Ophthalmology. 2016;123(3):617–24.
Itagaki K, Sekiryu T, Kasai A, Sugano Y, Ogasawara M, Saito M. Three-year outcome of aflibercept treatment for Japanese patients with neovascular age-related macular degeneration. BMC Ophthalmol. 2020;20(1):276.
Tsunekawa Y, Kataoka K, Asai K, Ito Y, Terasaki H. Four-year outcome of aflibercept administration using a treat-and-extend regimen in eyes with recurrent neovascular age-related macular degeneration. Jpn J Ophthalmol. 2021;65(1):69–76.
Maruko I, Ogasawara M, Yamamoto A, et al. Two-year outcomes of treat-and-extend intravitreal aflibercept for exudative age-related macular degeneration: a prospective study. Ophthalmol Retina. 2020;4(8):767–76.
Matsumoto H, Morimoto M, Mimura K, Ito A, Akiyama H. Treat-and-extend regimen with aflibercept for neovascular age-related macular degeneration: efficacy and macular atrophy development. Ophthalmol Retina. 2018;2(5):462–8.
Ito A, Matsumoto H, Morimoto M, Mimura K, Akiyama H. Two-year outcomes of a treat-and-extend regimen using intravitreal aflibercept injections for typical age-related macular degeneration. Ophthalmologica. 2017;238(4):236–42.
Schierling W, Troidl K, Troidl C, Schmitz-Rixen T, Schaper W, Eitenmüller IK. The role of angiogenic growth factors in arteriogenesis. J Vasc Res. 2009;46(4):365–74.
Barakat A, Rufin V, Tran THC. Two year outcome in treatment-naive patients with neovascular age-related macular degeneration (nAMD) using an individualized regimen of Aflibercept. J Fr Ophtalmol. 2018;41(7):603–10.
Kumar JB, Wai KM, Ehlers JP, Singh RP, Rachitskaya AV. Subfoveal choroidal thickness as a prognostic factor in exudative age-related macular degeneration. Br J Ophthalmol. 2019;103(7):918–21.
Minnella AM, Federici M, Falsini B, et al. Choroidal thickness changes after intravitreal ranibizumab for exudative age-related macular degeneration. BioDrugs. 2016;30(4):353–9.
Yamazaki T, Koizumi H, Yamagishi T, Kinoshita S. Subfoveal choroidal thickness after ranibizumab therapy for neovascular age-related macular degeneration: 12-month results. Ophthalmology. 2012;119(8):1621–7.
Cao X-S, Peng X-Y, You Q-S, Zhang Y-P, Jonas JB. Subfoveal choroidal thickness change after intravitreal ranibizumab for idiopathic choroidal neovascularization. Retina. 2014;34(8):1554–9.
Razavi S, Souied EH, Darvizeh F, Querques G. Assessment of choroidal topographic changes by swept-source optical coherence tomography after intravitreal ranibizumab for exudative age-related macular degeneration. Am J Ophthalmol. 2015;160(5):1006–13.
Kaya F. Change in choroidal thickness after intravitreal injection for treatment of neovascular age-related macular degeneration: ranibizumab versus aflibercept. J Fr Ophtalmol. 2017;40(10):832–8.
Sizmaz S, Kucukerdonmez C, Kal A, Pinarci EY, Canan H, Yilmaz G. Retinal and choroidal thickness changes after single anti-VEGF injection in neovascular age-related macular degeneration: ranibizumab vs bevacizumab. Eur J Ophthalmol. 2014;24(6):904–10.
Ünlü C, Erdogan G, Gunay BO, Kardes E, Akcay BIS, Ergin A. Subfoveal choroidal thickness changes after intravitreal bevacizumab injection for neovascular age-related macular degeneration and diabetic macular edema. Int Ophthalmol. 2017;37(1):147–58.
Gillies MC, Hunyor AP, Arnold JJ, et al. Macular atrophy in neovascular age-related macular degeneration: a randomized clinical trial comparing ranibizumab and aflibercept (RIVAL study). Ophthalmology. 2020;127(2):198–210.
Spooner KL, Fraser-Bell S, Cozzi M, et al. Macular atrophy incidence and progression in eyes with neovascular age-related macular degeneration treated with vascular endothelial growth factor inhibitors using a treat-and-extend or a pro re nata regimen: four-year results of the MANEX study. Ophthalmology. 2020;127(12):1663–73.
Gemenetzi M, Lotery AJ, Patel PJ. Risk of geographic atrophy in age-related macular degeneration patients treated with intravitreal anti-VEGF agents. Eye. 2017;31(1):1–9.
Daien V, Nguyen V, Essex RW, et al. Prevalence and characteristics of macular atrophy in eyes with neovascular age-related macular degeneration. A study from a long-term observational dataset: the fight retinal blindness! project. Br J Ophthalmol. 2020;104(8):1064–9.
Munk MR, Ceklic L, Ebneter A, Huf W, Wolf S, Zinkernagel MS. Macular atrophy in patients with long-term anti-VEGF treatment for neovascular age-related macular degeneration. Acta Ophthalmol. 2016;94(8):e757–64.
Grunwald JE, Daniel E, Huang J, et al. Risk of geographic atrophy in the comparison of age-related macular degeneration treatments trials. Ophthalmology. 2014;121(1):150–61.
Grunwald JE, Pistilli M, Ying G-S, Maguire MG, Daniel E, Martin DF. Growth of geographic atrophy in the comparison of age-related macular degeneration treatments trials. Ophthalmology. 2015;122(4):809–16.
Iovino C, Pellegrini M, Bernabei F, et al. Choroidal vascularity index: an in-depth analysis of this novel optical coherence tomography parameter. J Clin Med. 2020;9(2):595.
Acknowledgements
We would like to thank the ethical committee of the Catholic University Sacred Heart of Rome for their assistance in the realization of this project.
Funding
Open access funding provided by Università Politecnica delle Marche within the CRUI-CARE Agreement.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Chiara Centini, Maria Cristina Savastano, Gloria Gambini and Gabriele Ciasca. The first draft of the manuscript was written by Chiara Centini, Martina Maceroni, Angelo Maria Minnella and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Disclosures
No conflicts of interest exist for any author. The authors Angelo Maria Minnella, Chiara Centini, Gloria Gambini, Maria Cristina Savastano, Valeria Pagliei, Benedetto Falsini, Stanislao Rizzo, Gabriele Ciasca and Martina Maceroni declare they have no conflicts of interest directly related to this study.
Compliance with Ethics Guidelines
The study was approved by the Ethics Committee/Institutional Review Board of the Catholic University under protocol number 1189 with the acronym TWINS PLUS. This research adhered to the tenets of the Declaration of Helsinki and informed consent was obtained from all patients, after full and detailed explanation of the goals and procedures of the study. The study was performed in accordance with the current version of the Declaration of Helsinki [52nd World Medical Association (WMA) General Assembly, Edinburgh, Scotland, October 2000]. All patients provided written informed consent after detailed explanation about the aim of the study.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Minnella, A.M., Centini, C., Gambini, G. et al. Choroidal Thickness Changes After Intravitreal Aflibercept Injections in Treatment-Naïve Neovascular AMD. Adv Ther 39, 3248–3261 (2022). https://doi.org/10.1007/s12325-022-02129-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-022-02129-x