Abstract
Chromium (Cr) ion is regarded as a particularly hazardous contaminant due to its high toxicity and potential carcinogens. The Cr6+ ions were removed in the current work using a new fabricated low-cost adsorbent called Pea pods biochar-NH2 (PBN). PBN was characterized using BET, BJH, SEM, FTIR, TGA, DSC, XRD, XPS and EDX. The surface area and the pore size of PBN were decreased due to the amination process on the pea pods’ biochar-H2O2 (PBO). Optimized condition of different parameters of Cr6+ ions adsorption by PBN was observed at the pH 1.5, contact time (180 min), starting concentration (100 mg/L), and 0.1 g of PBN. The maximal adsorption capacity (Qm) of PBN is 384.62 mg/g. Different error models were used to test the isotherm models’ results. The mechanism of the adsorption was proposed based on the XPS analysis. The adsorption process had an R2 value of 0.999 as the best fit with the Freundlich isotherm (FIM) and pseudo-second-order kinetic (PSOM) models. These obtained data explored that the generated PBN can be a novel, cost-effective material for the adsorption of Cr6+ ions from an aqueous solution.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The concurrent increasing trend of human needs and subsequent technological advancements, expansion of urban and transportation facilities, the rapid growth of agricultural and manufacturing activities, and over-exploitation of environmental components are a few instances among other human-induced activities responsible for distressing water contamination (Aguilera et al. 2021; Ogamba et al. 2021; Bhagat et al. 2021). Thousands of previous studies have reported that global water has been severely polluted by organic and inorganic materials containing heavy metals (HMs). The concentration of HMs in the ecosystem is reported to be higher than the permissible limit (Ogamba et al. 2021; Bhagat et al. 2021). Heavy metals tend to accumulate in a living organism, and these are non-biodegradable. The previous studies also revealed that the toxicity of metals in the environment carriage severe impacts on public health and subsequent stimulus to the malfunction of the ecosystem (Kumar 2020). The higher concentration of highly toxic HMs, including arsenic, chromium, mercury, lead, and nickel, having severe toxic effects have been reported. The permissible limit of arsenic, chromium mercury, lead and copper are 0.01, 0.10, 0.002, 0.015, and 1.3 mg/L, respectively, by USEPA (Kumar 2020; Al-Makishah et al. 2020). The various human-induced activities such as; landfilling, using pesticides in agriculture, mine drainage, infiltration of untreated industrial effluents, dumping of municipal effluents, excessive uses of solvents, radioactive wastes and chemical wastes are directly or indirectly responsible for increasing the HMs in the aquatic environment (Barakat et al. 2019). The bioaccumulation of HMs is accountable for adverse impacts on living organisms (Kumar et al. 2020). The chronic health hazards include cancer, coronary disease, liver damage, neurological and cardiovascular disease, damage of the central nervous system, and sensory disturbances due to some toxic HMs such as; mercury, lead, antimony, chromium, arsenic, silver, cadmium, and aluminum etc. (Chai 2021). Chromium is one of the metals from animals, rocks, plants, soil, and atmosphere expatriate volcanoes. They are diverse oxidation forms such as Cr0, Cr2+, Cr3+ and Cr6+ ions. Chromium is one of the metals reported for applied in different industrial applications like textile, electroplating, dyeing, tanning of leather, electroplating, wood preservatives, metal processing, pigment and paints, canning and steel fabrication industry (El-Nemr et al. 2020a; He and Li 2020; Raj and Maiti 2020; Syeda and Yap 2022). The various adverse effect of chromium on human health has been reported, including mutagenic, carcinogenic, epigastric pain, nausea, vomiting, teratogenic, producing lung tumors and severe diarrhea (Al-Makishah et al. 2020). The IARC (International Agency for Research on Cancer) has categorized Cr6+ as a class-1 (one) human carcinogenic metal. Hence, it is of great concern to eliminate the Cr6+ from contaminated water (Syeda and Yap 2022). Therefore, different technologies like; physical, chemical, physio-chemical and biological water treatment methods are used for the adsorption of HMs containing chromium from wastewater (Karimi-Maleh 2021; Lu et al. 2021; Gu et al. 2021; Zhu 2022; Bao et al. 2021; Huang et al. 2021; Zhu, et al. 2021; Jahan et al. 2021). A substantial number of processes of this expertise have been using ion exchange (Alyuz and Veli 2009), chemical precipitation (Crini et al. 2018), adsorption (Kumar 2020; Martinson and Reddy 2009), reverse osmosis (Olmez 2009), ultrafiltration (Xiang et al. 2016), nanofiltration (Lata and Samadder 2016), coagulation-flocculation (Abulbasher et al. 2002; Michael 1991), electrodialysis (Figoli et al. 2010), electrochemical process (Bojic et al. 2009) and flotation (Duan et al. 2010). Among these, adsorption, membrane filtration and ion exchange have been frequently used for HMs decontamination from water. Most conventional methods are less efficient due to the resistance and persistence of HMs in wastewater (Raj and Maiti 2020; Abu Taleb et al. 2020; Dey et al. 2019). Although, for maintaining the cell functions of living organisms, some metals with low concentrations, like; copper, iron, manganese and zinc, are needed, while high concentrations are poisonous (Kumar 2020; Abu Taleb et al. 2020). Therefore, finding environmentally friendly and cost-effecting water remediation methods is demanding. The adsorption process is a popular technique with easy operation, economically convenient, wide selectivity, significant pollutants recovery, efficiency and reusability (Sadrzadeh et al. 2009; Kabdasli et al. 2009; Zamboulis et al. 2011; Barakat 2011). Thousands of adsorbents materials are applied in the adsorption process (Fu and Wang 2011), including alumina (Al2O3) (Barakat et al. 2019), biochar (El-Nemr et al. 2020a), activated carbon (Syeda and Yap 2022), graphene oxide (Kumar 2020), titanium dioxide (TiO2) (Karimi-Maleh 2021), kaolin clay (Lu et al. 2021) and silica (Kumar 2020; Abu Taleb et al. 2020). Recent trends in the adsorption process are to modify the composite adsorbent because of their exclusive characteristics, for example, enlarged surface area, small size, functional groups available with sensitive sites and regeneration facilities (He and Li 2020; Raj and Maiti 2020). A part from other composite adsorbents, bio-based composite adsorbents were reported as excellent alternatives due to their eco-friendly nature, low cost, and naturally abundant. The biological sources of adsorbent are mainly from agricultural byproducts such as pods, rice husks, seed shells of sunflowers, eggshells, etc., having stable, nontoxic, environmentally friendly, hydrophilic nature and biodegradable behaviors (Malik et al. 2016; Cheng, et al. 2021; Wang 2020; Gupta et al. 2021; Rodriguez-Narvaez et al. 2017; Eniola et al. 2019; Orooji et al. 2021). The biochar having structured carbon matrix materials are recently used for heavy metal decontamination, including chromate ions from wastewater (El-Nemr et al. 2020a). The biochar can mainly be produced from biomass by applying pyrolysis treatment without oxygen at low temperatures (˂ 700 °C). The biochar taking extended surface area, extremely porous and plentiful surface functional groups including OH, COOH, C=O, etc. (El-Nemr et al. 2020a) were considered excellent supporting materials for modifying bio-based composite for surface adsorbent of heavy metals including Cr6+ ion (Zhang et al. 2019). The biochar-based composite materials were found as excellent adsorption capacity of metal ions, especially Cr6+ ions adsorption, due to their active surface properties as well as excellent electrostatic interface flanked by the functional groups of materials protonation and the negatively charged chromate ions (Rafique et al. 2021). In the last several years, biochar has been synthesized with numerous biomass sources, including pyrolysis of hardwood, brewer’s draff and corn straw, etc., for the abundance of surface functional groups and subsequent enhancement of removal capacity of heavy metals (Murad et al. 2022). Several past kinds of research directed that using biochar for decontaminating HMs depends on the sources of raw materials used for the pyrolysis of biomass and modified biochar materials (El-Nemr et al. 2022). Therefore, evaluating modified biochar with functional materials is a great concern of scientists for effective use in environmental remediation, especially HMs decontaminating from contaminated water environments. One of the most often used reducing agents is aniline, along with substances like Triethylenetetramine, Na2SO3 and NH3H2O (El-Nemr et al. 2020a; El-Nemr et al. 2022). However, choosing each reducing agent can depend on reaction condition, contaminant type and desired reduction performance. The rationale of employing ammonia to modify biochar for Cr6+ ion adsorption removal is to increase its surface area and pore volume, add new functional groups, and improve its stability in order to increase its adsorption capacity. This strategy for wastewater treatment is innovative, promising, green and environmentally by using agriculture waste (Pea pods) with potential sustainable applications across numerous sectors. Many studies deal with the removal of Cr6+ ion through adsorption. However, to the author’s knowledge, the modification of sulphonated biochar PB by adding ammonia (PBN) is used for the first time for Cr6+ ion removal.
This work mainly focused on modifying a highly efficient biochar composite adsorbent fabricated with pea Pisum sativum pods to absorb hexavalent chromium (Cr6+) from water. The synthesized materials were effusively categorized by using structural, morphological, spectra and chemical methods applying well-known material-characterizing tools, including Scanning Electron Microscopy (SEM), Fourier Transform Infrared Spectrophotometer (FTIR), Energy Dispersive Spectroscopy (EDX), X-ray photoelectron spectroscopy (XPS), Thermal gravimetrical analysis (TGA), and X-ray diffraction (XRD). The essential new factor in this work is that the prepared PBN biochar was the only one among the three prepared biochars (PB, PBO, and PBN) that showed high adsorption efficiency of Cr6+. Therefore the adsorption of Cr6+ ion by PBN was investigated using different factors that affect the adsorption process, including reaction time, initial concentration, the effect of interfering ions, and temperature. Different well knows kinetic (KMs) and isotherm models (IMs) were evaluated to determine the Cr6+ ion adsorption mechanism onto PBN biochar composite. The mechanistic insight and mathematical modeling also investigated the interaction between selected contaminated ions and fabricated adsorbents.
Materials and methods
Materials and equipment
The biomass applied for biochar fabrication is pea Pisum sativum pods, which are obtained from the local area in Alexandria, Egypt. It was cleaned well with water and dried at 105 °C for 20 h, and the dried materials were crushed to a fine powder (El-Nemr et al. 2020a). H2SO4 (99%) and NH4OH (25%) were purchased from Sigma Aldrich, Germany. By dissolving 2.8289 g of K2Cr2O7 in 1000 mL of distilled water (DW), 1.0 g/L (1000 mg/L) of stock solution of Cr6+ ions was produced (El-Nemr et al. 2022). This typical solution was then applied to prepare further working solutions using the dilution procedure. The stock solution was used to create Cr6+ ions with starting concentrations fluctuating from 0 to 100 mg/L, which were then analyzed to display the standard curve (El-Nemr et al. 2022). FTIR spectroscopy (using platinum ATR) model V-100 coupled to Bruker VERTEX70, Germany, was used to investigate the kinds of functional groups present on the biochars’ surface.
The adsorption–desorption isotherm of N2 gas on samples was measured at N2 gas boiling point. he samples’ BET surface area (SBET) quantities were obtained by N2 adsorption at 77 K using BELSORP–Mini II, BEL Japan, Inc. (Gregg and Sing 1982; Rouquerol et al. 1999). Examination of the isotherm was supported by the BET plot to calculate the monolayer volume (Vm) (cm3 (STP) g–1), the surface area (SBET) (m2/g), the total volume of pores (p/p0) (cm3/g), energy constant (C) and mean diameter of pores (nm). Also the micropore surface area (Smi) and micropore volume (Vmi), as well as the mesopore surface area (Smes) and mesopore volume (Vmes) of samples, were measured by the t-plot and Barrett-Joyner-Halenda (BJH) methods, respectively, following the BELSORP-Mini II software. The size distribution of pores is measured by applying BJH desorption isotherm method (Barrett et al. 1951).
Using an EDX and SEM (QUANTA 250), elemental analysis was achieved on the surface morphology of the biochar samples. The SDT650-Simultaneous Thermal Analyzer equipment was used to perform thermal analyses between 25 and 1000 °C with a 5 °C per minute as a ramping temperature (El-Nemr et al. 2022; El Nemr et al. 2017; Ragab et al. 2022). Elemental analysis was performed using a Thermo Fisher Scientific K-Alpha XPS with a pass energy of 50 eV at a base pressure of ~ 10−9 mbar.
Fabrication of Pea pods biochar (PBN)
100 g of the crushed pea Pisum sativum pods were cooked for 4 h in a refluxed system with 400 mL of an 80% H2SO4 solution (El-Nemr et al. 2020a; El-Nemr et al. 2023; El-Nemr et al. 2022). The reaction mixture containing biochar was filtered out, repeatedly cleaned with DW until the pH of the washing water became neutral, and then washed with ethanol. The reaction mixture was then ice-cooled. The prepared biochar was dried for 12 h at 105 °C. The subsequently formed biochar sample was nominated as PB (El-Nemr et al. 2023). The prepared PB (50 g) was reacted with H2O2 in DW for 4 h to increase the possibility of reaction with NH4OH via oxidation with H2O2 (El-Nemr et al. 2020b). The H2O2-modified biochar (PBO) was filtered, repeatedly cleaned with DW and ethanol, and dried at 105 °C overnight. In a fume hood, 25 g of biochar PBO were cooked for 4 h using a refluxed system in 120 mL of NH4OH. The reaction mixture was cooled to room temperature and filtered to produce PBN. The recovered PBN was repeatedly cleaned with DW, followed by ethanol, and then dried at 105 °C for 20 h to provide 24.5 g of PBN.
Batch adsorption experiment
A batch experiment adsorption was used to assess the adsorption capacity, thermodynamic, and kinetic properties of PBN (El-Nemr et al. 2020a, 2023; El-Nemr et al. 2022). A series of flasks were agitated at 200 rpm for 180 min with different initial concentrations of Cr6+ ions solution and PBN doses (0.5–2.5 g/L) (El- Nemr et al. 2022). 0.1 M HCl or NaOH solution was applied to bring the pH of the solution up to the necessary levels. Throughout the adsorption equilibrium studies, the solution pH was kept constant. Following separating around 0.1 mL of the solution from the absorbent, the concentration of Cr6+ ion was measured at periodic intervals. The Cr6+ ion concentration was measured by spectrophotometer (λmax 545 nm) via color complex formation using 1,5–diphenyl carbazide as reagent (El-Nemr et al. 2022). The adsorbent capacities of adsorption at time t at interval times were calculated via Eq. (1):
where qt is the adsorption capacity of the PBN at time t (mg/g); C0 is the Cr6+ (mg/L) beginning concentration; Ct is the remaining Cr6+ ions concentration over a period of time t (mg/L); V (L) is the Cr6+ ions solution volume, and W (g) is the PBN mass.
The impact of pH on Cr6+ ion adsorption was considered by using 0.1 g of the PBN to 100 mL of 200 mg/L of Cr6+ ion solutions with starting pH values (1.5–9.5). The pH values were adjusted with diluted 0.1 M HCl or NaOH solutions. The mixture was then agitated at 200 rpm at room temperature (rt), and samples were taken periodically to determine the concentration of Cr6+ ions.
The impact of PBN dose on Cr6+ ions adsorption and the isotherm investigation was performed by different concentrations of Cr6+ solutions (50–300 mg/L), and PBN dose (0.5–2.5 g/L) were agitating at 200 rpm at room temperature and measured at intermission times for analysis of Cr6+ ions remaining concentration. The data from all adsorption studies are reported as mean values, and each adsorption experiment was carried out in triplicate.
Equilibrium and kinetics investigations
A monolayer adsorption with uniform energies is assumed by the Langmuir model (LIM), and there is no transmigration of PBN in the surface plane. The LIM linear equation can be expressed as Eq. (2) (El Nemr et al. 2010):
where Qm (mg/g) reproduces monolayer maximum capacity; Ka (L/mg) is constant, showing the adsorption equilibrium sorption energy.
Equation (3) displays the linear Freundlich isotherm model (FIM form (El Nemr et al. 2010):
In this equation, KF is the adsorption capacity constant, and 1/n denotes the adsorption or surface heterogeneity intensity, increasing heterogeneity as the value approaches zero. Equation (4) provides the Freundlich model’s maximal adsorption capacity (QmF, mg/g):
Equation (5) represents the Temkin isotherm model’s (TIM) linear form (El Nemr et al. 2010).
where β = (RT)/b, T (Kelvin) is the absolute temperature, and R (8.314 J/mol K) is the universal gas constant. The constant b is correlated to the adsorption heat (El Nemr et al. 2010).
The Dubinin–Radushkevich model (DRIM) (El Nemr et al. 2010) assesses the actual porosity-free energy and characteristics of adsorption. The dose of DRIM does not undertake a homogeneous surface or constant adsorption potential. The DRIM can be written as Eq. (6), where β (mmol2/J2) is a coefficient corresponding to the adsorption mean free energy of, qm (mmol/g) is the maximum adsorption capacity, and ε (J/mmol) is the Polanyi potential, which is written as: ε = RT(1 + 1/Ce).
Pseudo-first-order model (PFOM) is commonly stated as a linear Eq. (7) (El Nemr et al. 2010):
where qe (mg/g) and qt (mg/g) are the equilibrium adsorption capacity and adsorption capacity at time t, respectively, k1 (L/min) is the rate constant of PFOM adsorption. The plot of t versus log (qe − qt) should give a linear relationship from which predicted qe and k1 could be calculated from the linear plot intercept and slope, respectively.
Equation (8) can be used to provide the linear pseudo-second-order model (PSOM) (El Nemr et al. 2010):
where k2 (g/mg min) is the PSOM adsorption rate constant used to measure the beginning sorption rate, h, by Eq. (9).
Values of k2 and qe can be calculated from the plots’ intercept and slope of t/qt versus t, respectively.
Equation (10) typically describes the linear Elovich kinetic model (EM) (El Nemr et al. 2010):
where α (mg/g min) and β (g/mg) are the beginning adsorption rate and the desorption constant. Plot of ln (t) versus qt should give a linear relationship with an intercept of (1/β) × ln (αβ) and a slope of (1/β).
Equation (11) is utilized to study the potential of the intra-particular diffusion kinetic model (IPDM) (El Nemr et al. 2010):
As the value of intercept C increases, the fighting to the external mass transfer increases. The regression line slope in the plot of qt against t1/2 can be used to directly calculate Kdif, the IPDM rate constant. Different error function analyses were used to investigate the IMs of the experimental adsorption results (El Nemr et al. 2010).
Author statement for the use of plants. In this study, Experimental research and field studies on plant material (Pea Pisum sativum pods), including the collection of plant waste material, complies with relevant institutional, national, and international guidelines and legislation.
Results and discussion
Features of PBN
FTIR investigation
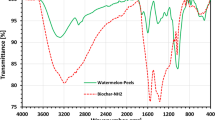
The generated PBN sample was subjected to FTIR analysis to classify the functional groups and determine how the chemical alteration affected the existence of new functional groups. Figures 1 and 2 show the FTIR curve of three different Pea biochars and raw pea pods. In pea pods, PB, and PBO biochars, the strong peak at 3225–3379 cm–1 indicates the O–H stretching vibration, while the broad adsorption band about 3168 cm–1 is revealing of the presence of the -OH group of glucose and the –NH of the amino group in PBN (Fig. 2). This new peak’s appearance recommended that the –NH2 group had been added to the structure of the biochar. Pea pods and three Pea pod biochars included the band at 2920–2926 cm–1, recognized as the –CH stretching vibration. The –COOH group, which was present in PB and PBO biochars but entirely vanished in pea pods and PBN, can be recognized as the C=O stretching that caused the adsorption band at 1702 cm–1. However, PBO-treated biochar had greater strength at 1702 cm–1 when compared to PB-treated biochar, suggesting that H2O2 treatment may have strengthened the C=O group. The 1621 cm–1 band is practically present in pea pods, PB, and PBO biochars and suggests the C=O stretching vibration of β-ketone. Pea pods also displayed a significant peak at 1621 cm–1, contrasting with PB and PBO biochars, indicating that sulfuric acid and H2O2 may have had less impact on the C=O functional group than raw pea pods. Only at 1560 cm–1 in PBN biochar did the N–H stretching vibration in secondary aromatic amine or fatty amine arise, showing that NH4OH alteration increases the N–H group in PBN. The C–O group was weak and exclusively manifested in pea pods, PB, and PBO, as shown by the peak at 1415–1438 cm–1. The stretching vibration of the –N=C=O group, or mostly allocated to C–N stretching, is thought to be in charge of the adsorption peak at 1380 cm–1. Following the chemical treatment with NH4OH, these additional peaks demonstrated the presence of nitrogen groups on the surface of pea pod biochar. Compared to pea pods and PBN, PB and PBO exhibit an increase in the C–O–C asymmetric stretching group due to oxygenated carbon chains peaking at 1240–1157 cm–1. The band at 1030–1035 cm–1 represents the C–O–H group that occurred in three biochars, while it is stronger in raw pea pods. Additionally, there was a clear distinction between pea pods and the two types of biochar in the band strength range of 1030–1035 cm–1, showing that the NH4OH alteration significantly impacted the C–O–H group of biochar.
Properties of PBN surface
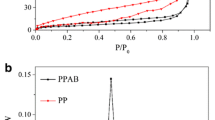
To determine how the reagents H2O2 and NH4OH affected the surface characterization, the N2 adsorption–desorption investigation of the biochar made from pea pods was conducted. The BET and BJH methods were applied to calculate the textural qualities. The specific surface area (SBET) of biochars calculated by BET decayed as PB (23.12 m2/g) > PBO (14.15 m2/g) > PBN (11.82 m2/g). The change notably decreased the meso-surface area of PBO and PBN. The monolayer volume values of PB, PBO, and PBN biochars were 4.91, 3.14, and 2.41 cm3(STP)/g, respectively. This showed a similar trend with surface area. 0.024, 0.021, and 0.020 cm3/g, respectively, were the total volume values of the biochars made from PB, PBO, and PBN. The mesopores in PB, PBO, and PBN biochars had mean sizes of 4.55, 8.12, and 8.66 nm, respectively. According to this outcome, PBO and PBN, particularly PBN biochar, expanded their pore sizes during the alteration procedure. As PB (21.41 m2/g), PBO (16.15 m2/g), and PBN (14.11 m2/g) decreased in mesosurface area, biochars’ mesosurface area decreased. A significant change was that the PB biochar’s surface area was decreased. 0.030, 0.028, and 0.025 cm3/g, respectively, were the mesopore volume values for PB, PBO, and PBN biochars. Mesopore distribution peak values for PBO and PBN biochars were 1.22 and 1.66 nm, respectively, compared to 1.22 for PB biochar. Due to pore obstruction by the additional groups, the surface area and total volume of pores in biochar reduced after chemical treatment. While the PBO and PBN had been blocked by the –OH and –NH2 groups, the unmodified biochar had many empty pores available for N2 molecules to adsorb. The amine modification did, however, increase the mean pore diameter in PBN biochars, which may have been caused by the amine groups filling in tiny pores.
Morphological investigation
SEM analyses of PB, PBO and PBN biochars are shown in Fig. 3a, b, and c, respectively. The prepared PB biochar’s porous structure is clearly depicted in Fig. 3a, with the various size pores indicating a large surface area. The PBO biochar image demonstrates that after being exposed to H2O2, the pores did not distort (Fig. 3b). The surface analysis of PBN biochar is presented in Fig. 3c, demonstrating that the amino group-producing active sites occluded the majority of the caves and pores.
Elements in biochars
EDX analysis was applied to determine the elements in the PB, PBO, and PBN samples. The EDX analysis results of prepared biochars are shown in Table 1. Before H2O2 and NH4OH reagents modified PB biochar, the data in Table 1 showed the deficiency of N2. The EDX analysis of PBN revealed that nitrogen comprised roughly 19.56% of the sample weight. The carbon and oxygen contents in PBN were 61.52 and 17.76% of the total sample weight, respectively.
Thermal investigation of prepared biochars
Figure 4a shows that the degradation of Pea pods (raw material) and PB occurs in three phases, while that of PBO and PBN biochars occurs in just two. With weight losses of around 4.041, 8.776, 3.948, and 6.132% for pea pods, PB, PBO, and PBN biochars, respectively. The first stage, which proceeds at temperatures from 50 to 150 °C, includes the samples’ loss of surface-bound water and moisture. Nearly 63.68% of the weight was lost at 150–350, 20.53% at 150–275, 37.79% at 150–1000 °C, and 34.97% at 150–1000 °C for pea pods, PB, PBO, and PBN, respectively, in the 2nd mass loss stage. In the 3rd mass loss step, with approximately the mass loss of 5.014% at 350–1000 °C and 25.70% at 275–1000 °C for pea pods and PB, respectively. The percentages of 27.27, 45.10, 58.26, and 58.98% were achieved, demonstrating greater stability for oxidized and amine-modified biochar than raw pea pods and PB. At the same time, the mass remained in the order of pea pods < PB < PBO < PBN.
Figure 4b illustrations the DTA of pea pods, PB, PBO and PBN samples. The DTA curvature of the Pea pods (black) sample shows two peaks at the temperature of flow (Tf), 76.67 and 342.45 °C, while the DTA curve of the PB (green) sample shows two bands at a lower Tf (75.95 and 213.84 °C), indicating that the Pea pods may have higher stability than PB sample. However, as seen from the DTA curves, the Pea pods (black) and the PB (green) samples exhibit two distinct degradation peaks during pyrolysis. Figure 4b’s decomposition bands exhibited two bands that persisted after being treated with H2SO4, indicating that the amount of the pea pods’ (black) breakdown was reduced due to the acid treatment. The DTA curvatures of the PBO and PBN samples revealed two well-resolved deterioration peaks at a temperature of flow (Tf) (76.60, 400.97 °C) and (76.73, 408.76 °C), respectively, and onset points at (56.17, 271.13 °C) and (56.23, 253.11 °C), respectively. This demonstrated that altering PB samples with H2O2 and amine boosted their stability. However, these findings also demonstrated that the modification reagent’s application significantly impacts the PB products’ degree of crystallinity.
Materials can be compared using thermal transitions and differential scanning calorimetry (DSC). Raw pea pods and the biochars PB, PBO, and PBN are shown in the DSC in Fig. 4c. The crystallization of H2O molecules was the primary cause of the low temperatures at which all samples began to crystallize. The DSC of pea pod raw material identified a temperature range between 348.29 and 758.59 °C. The DSC revealed a crystallization temperature of 219.84 °C for PB. The biochars PBO and PBN, respectively, do not show any more phase changes.
X-ray photoelectron spectroscopy (XPS)
XPS was used to qualitatively analyze the functional groups on the surface of the prepared PBN (Zhang et al. 2022; Alegre et al. 2022). The high-resolution XPS data of S2p, C1s, and O1s were used to study the sulfur, carbon, and oxygen bonding states. Figure 5a shows the broad full XPS spectra of PBN. As seen from the graphs, characteristic peaks of S2p, C1s, O1s, N1s are found in PPB, witnessing that S and N have been successfully retained on PBN. The peaks located in 171.22, 287.97, 402.73, and 535.03 eV correspond with S2P, C1s, N1s and O1s, respectively.
Analysis of the high-resolution S2p scan (Fig. 5b) was indicative of 2 S species in the PBN sample: one S2p3/2 peak at the binding energy (BE) of 167.2 eV for C–SO2–C (sulfone) or C–SO3H (sulfonate), and another S2p3/2 peak at the binding energy of 170.09 eV ascribed to high-valence sulfur of sulfate or sulfate-like species. Most of the S was present in the Sulfate or sulfonate functional group(s) (~ 64.1 at.%) (Zhang et al. 2022). These peaks agree with those reported in the literature for the S2p3/2 binding energy of organic sulfur compounds (Zhang et al. 2022; Alegre et al. 2022).
Figure 5c shows that C1s exhibits three peaks by curve fitting of the C1s spectrum. The C1s spectrum can be deconvoluted into three peaks at 284.83 (9.23%), 287.23 (79.69%), and 290.57 eV (11.08%), assigned to C=C/C–C/C–H bonds, C=O/C=N bonds, and –O/C=O bonds, respectively (Zhang et al. 2022; Alegre et al. 2022; Liu et al. 2020; Kim et al. 2014; Glenis et al. 1999; Chen et al. 2018; Xing et al. 2016). The N1s XPS spectra of PBN could be deconvoluted into two types of N-containing compounds, and results are depicted in Fig. 2d. The peaks of N1s located at 398.77 (pyridinic N) 2.83% and at 402.07 (graphitic N) 97.17% (Xing et al. 2016; Yang et al. 2019), respectively in PBN. The O1s XPS spectrum of the PBN exhibits two peaks at 533.08 and 532.54 eV, corresponding to (C–O) and (H2O) (Fig. 3e) (Zhang et al. 2022; Alegre et al. 2022; Liu et al. 2020; Kim et al. 2014; Glenis et al. 1999; Chen et al. 2018; Xing et al. 2016; Yang et al. 2019; Kao et al. 2018).
Adsorption of Cr6+ ions on Pea pods-biochar-NH2 (PBN)
Impact of pH
According to Zhanqi et al. (2007), the solution pH impacts the solubility of metal ion speciation and the potential charges of active sites on the materials (Mohan et al. 2015). As a result, the effectiveness of removing Cr6+ ions from PBN was examined at various pH levels between 1.5 and 9.5. These outcomes demonstrated a high PBN removal efficiency for Cr6+ ions at a low pH (pH < 4). According to Fig. 6a, the most significant removal efficiency for PBN was 95.79% at pH 1.5 at room temperature, a starting concentration of 100 mg/L and 0.1 g/L of biochar. As a result, it was determined from Fig. 6b that the measured capacity of adsorption was 95.79 mg/g at pH 1.5 and 0.1 g/L of PBN. A similar result was previously seen when Cr6+ ions were adsorbed on coffee charcoal treated with polyphenol-formaldehyde and polyphenol-acetaldehyde (Mulani et al. 2013; Liang et al. 2020). Furthermore, according to Li et al. (2014), an increase in pH results in greater electrostatic attraction, which boosts adsorption effectiveness. Similar outcomes were observed when Cr6+ ion was adsorbed using nanoparticles or biochar made from sugar beet tailings, magnetic eucalyptus leaf residue, or sugarcane harvest biochar at lower pH levels (Wang et al. 2014; Yuan et al. 2009; Dong et al. 2011; Xiao et al. 2018). Additionally, it has been discovered in an earlier study that Cr6+ ions can easily be converted to Cr3+ ions at lower pH values (pH < 3); this was another essential explanation for the highest Cr6+ ions elimination at pH 1.5. (Verma et al. 2006; Choudhary et al. 2017). PBN has essential functional groups that promote the best Cr6+ ion adsorption onto biochar surfaces. Given that PBN’s surface area is larger than that of raw biochar, it also plays a substantial role in the adsorption of Cr6+ ions from polluted water.
Time of contact influence
As residence/time of contact in the aqueous solution increases, the efficacy of Cr6+ ions adsorption employing PBN also increases. As reported in Fig. 7, the influence of contact time on Cr6+ ion adsorption was investigated using a Cr6+ ion concentration of 50–300 mg/L and a fixed PBN dose of 1 g/L using PBN at pH 1.5. With removal efficiencies for PBN ranging between 70 and 100%, Cr6+ ions adsorption equilibrium was attained in roughly 180 min. After 180 min of contact, it is presented that the removal of Cr6+ ions continues, albeit minimally when the contact time is raised to 24 h. As a result of the availability of free active sites, the maximal concentration of Cr6+ ions was adsorbed onto PBN surfaces after 180 min, according to these data. At ambient temperature, Cr6+ ions have been seen to slowly adsorb on PBN for pH 1.5. Because of the electrostatic interaction between the Cr6+ ions and PBN surface in the water solution, it was shown that the adsorption efficiency increased less with respect to residence duration.
Influence of beginning Cr6+ ions concentration
Studying the impact of the removal efficiency on the initial concentration of Cr6+ ions is crucial. Thus, at different initial concentrations of the Cr6+ ions, the impact of the starting concentration on an adsorption ability was seen. At Cr6+ ion concentrations of 50, 100, 150, 200, and 300 mg/L, the impact of the initial concentrations of Cr6+ adsorption was investigated. The elimination and adsorption capacity (qe, mg/g) was increased quickly with an increase in the starting concentration of Cr6+ ions. The adsorbents’ type and availability as functional groups at a given moment are related to the starting concentration parameter. With 0.5–3.0 g/L of PBN at a pH of 1.5, the elimination of the Cr6+ ions from the solution was studied. Figure 8 presents experimental result points for the adsorption of Cr6+ ions by PBN at various concentrations and illustrates how the starting concentration affects the removal of Cr6+ ions. For PBN, the maximum uptake capacity was between 100 and 418 mg/g at 300 mg/L. When starting concentrations were increased from 50 to 300 mg/L, the adsorbent’s ability for adsorption increased. During the adsorption procedure, Cr6+ molecules must first pass through the boundary layer. They must diffuse from the boundary layer onto the adsorbent’s surface, and finally, they must diffuse into an adsorbent with a porous structure. This procedure will require a considerable amount of contact time. With an increase in the starting concentration of Cr6+ ions, the adsorption increased as a result of an increase in the concentration gradient that drives the force for the mass transfer. Because many heavy metal molecules were present at the initial concentration in a given area of the surface, surface saturation was dependent on concentration. This is the case due to the mass transfer of the Cr6+ ions from the liquefied phase to the solid phase is mostly driven by the initial concentration. Thus, the lower adsorption would result from the larger initial concentration (Yin et al. 2019; Pandey and Mishra 2011).
Impact of PBN dosage on Cr6+ ions adsorption
By adjusting the PBN dosage (0.5–3.0 g/L), starting Cr6+ ion concentration (50–300 mg/L), adsorption time (180 min), temperature (25 °C), and initial pH of 1.5, the impact of PBN dosage on the adsorption of Cr6+ ions by PBN was investigated. According to Fig. 9a, the adsorption efficiency of Cr6+ ions by PBN constantly rose as the amount of PBN adsorbent increased. However, this rise is barely noticeable at dosages of 50 and 75 mg/L, but it increases significantly at dosages of 100, 150, 200, and 300 mg/L, from 63 to 100%. At all PBN dosages (0.05–0.3 g/L) and all starting Cr6+ ion concentrations (50–300 mg/L) investigated, the removal of Cr6+ ions ranged between 63 and 100%. For all starting Cr6+ ions concentrations, the amount of Cr6+ ions absorbed at equilibrium (qe) decreases as the PBN dosage is increased. In addition, for initial Cr6+ ion concentrations of 50, 75, 100, 150, 200, and 300 mg/L, respectively, the amount of Cr6+ ions absorbed at equilibrium (qe) decreases from 92.91 to 16.57, 141.80 to 24.91, 140.36 to 33.20, 194.13 to 49.87, 252.78 to 66.54, and 418.41 to 99.72 mg/g with increasing the PBN amount from 0.5 to 3 g/L. As reported in Fig. 9a, b, the maximum (%) and least amount of removal process at equilibrium (qe) was carried out employing a dosage of 3 g/L of PBN.
Adsorption isotherms
The adsorbate (Cr6+ ions) diffusion with the adsorbent (PBN) is described by the adsorption isotherm. Non-linear monolayer adsorption to homogeneous surfaces without interfering with the absorbed particles is explained by the Langmuir isotherm model (LIM). The equilibrium condition explains the relationship between the solute absorbed by the absorbent surfaces and the beginning Cr6+ ions concentration in this model. However, the Freundlich isotherm model (FIM), which applies to nonlinear monolayer adsorption to heterogeneous surfaces and interacts with absorbed molecules, shows the heterogeneity of nonlinear adsorption of absorbents. The heat of adsorption is explained using the Temkin isotherm model (TIM) (i.e., sorbent–solute interactions). The homogeneous distribution of binding sites defines solute adsorption, and the heat of solute sorption decreases linearly with surface coverage. Moreover, the Dubinin–Radushkevich isotherm model (DRIM) distinguishes between the chemical sorption of the physical and metal ions in a water solution. It does this by describing how Gaussian energy is distributed onto heterogeneous adsorbent surfaces (Etnier et al. 1981).
Isotherm analyses were used to show the reciprocal interactions between Cr6+ ions and PBN, with the findings displayed in Fig. 10. When seen, as the original pollutant concentrations increased, the Cr6+ capture through PBN steadily increased. However, when the quantities of pollutants increased, the rivalry between Cr6+ ions and the restricted activity sites intensified, gradually increasing Cr6+ ion removal until equilibrium.
For the examined PBN adsorbent concentrations, the correlation coefficient values for the LIM were determined and are presented in Table 2 as R2 (0.176–0.985), Qm (128.21–384.62), and KL (0.04–0.80). As calculated by the LIM, the activated biochar’s adsorption capacity presupposes that adsorption occurred at homogeneous adsorption sites on the surface and that there are no interactions between the absorbed contaminants ions (Labidi et al. 2016).
The correlation coefficient R2 (0.705–0.952), Kf (65.34–153.99), and value of 1/n (0.270–1.428) for the FIM were determined and are shown in Table 2. Cr6+ ions are shown to have heterogeneous adsorption on the surface of PBN in the FIM.
The correlation coefficient R2 (0.572–0.975), AT (1.262–12.134), and βT (31.172–71.626) values for the TIM were determined and are shown in Table 2.
As demonstrated in Table 2, the DRIM analysis for Cr6+ ions revealed poor fitting R2 (0.529–0.904) with PBN. Table 2 also included the values for Qm (124.49–235.38) and E (476.73–2500.00) for various adsorbent doses. In general, FIM is regarded as the best-fitting Cr6+ ions adsorption on PBN. Additionally, 384.62 mg/g was the Qm of Cr6+ ions that could be removed with PBN.
Error function investigation for best-fit IM
Correlation coefficients (R2) for the FIM, TIM, LIM and DRIM were compared to the results of experimental equilibrium to choose the most suitable IM for the adsorption of Cr6+ ions on PBN adsorbent. Another technique for choosing the optimal IM given experimental result is to compare various error function values. Root mean square errors (RMS), Average percent errors (APE), hybrid error function (HYBRID), Chi-square error (X2), the sum of absolute errors (EABS), and Marquardt’s percent standard deviation (MPSD) are the main functions applied to identify the distribution of error between the estimated IMs and equilibrium values (Nemr et al. 2010; Li et al. 2022). Both correlation coefficients and error function terms clearly show that the FIM is the most appropriate (Table 3).
Adsorption kinetic investigation
The mechanism of Cr6+ ion adsorption onto PBN is explained using kinetic models. The PFOM is a kinetic model (KM) representing the adsorption kinetics under the impact of time of contact. The adsorption rate is closely related to the active sites present on the PBN surface, according to the PSOM. The system’s activation and deactivation energies, mass, and surface diffusion can all be predicted using the EIM. However, the internal adsorption of the absorbent in a water solution is interpreted by the IPDM. Through kinetic studies, the adsorption of PBN for Cr6+ ions was examined (Fig. 11). As shown, the removal of Cr6+ ions by PBN increased significantly in the first 30 min (Fig. 7), which was likely caused by the large number of adsorption sites that PBN provided. As the adsorption sites were filled, the equilibrium of Cr6+ ion binding to PBN was attained in 180 min. To quantitatively examine the binding process of Cr6+ ions on PBN, PFOM, PSOM, EM, and IPDM were used to represent the uptake effects (Li et al. 2021; Yin et al. 2020a; Yin et al. 2020b).
The R2 was used to evaluate the application of kinetic models, and a higher R2 value indicated better applicability. Adsorption kinetics generally provides a sense of the biochar’s adsorption capacity concerning time. Figure 11a illustrates the calculation of the rate constant, k1, and adsorption capacity at equilibrium (qe) from the linear graph of the values of Log (qe − qt) against time (t). By analyzing the PFOM linear form, the values of k1 (1.38–31.09), qe (0.30–108.12), and R2 (0.001–0.994) for the PFOM were determined. These values are displayed in Table 4.
The mass transfer dynamics serve as the foundation for the PSOM. The quantity of Cr6+ ions adsorbed at equilibrium (qe), as well as the PSOM constant, k2, may be calculated by graphing t/qe versus t, as seen in Fig. 11b. As presented in Table 4, the values of k2 (0.18–365.46), qe (16.64–384.62), and R2 (0.961–1.000) corresponded to the linear form of PSOM.
The intercept and slope of Fig. 11c were used for the EM’s α and β constants calculation, respectively, and the values measured are given in Table 6. For all PBN dosages, the EM assessed values are α (18–7.06 × 1074), β (0.045–7.184), and R2 (0.490–0.992).
If the lines shown in the curve of qt and t0.5 in Fig. 11d pass via the origin, according to Weber and Morris’ theory (Weber et al. 1963), the intraparticle diffusion step governs adsorption. However, when the drawn lines do not go through the origin, it is thought that film diffusion regulates the rate of the adsorption process (i.e., when the C value is high). For the adsorption of Cr6+ ions on PBN adsorbent at various dosages and varying beginning Cr6+ ions concentrations, the Webber-Morris adsorption line is shown in Fig. 11d. For the adsorption of Cr6+ ions to PBN, IPDM analysis determined Kdif, C, and R2 values as (0.059–6.948), (15.92–361.15), and (0.34–0.991), respectively, as shown in Table 5. The dynamics of Cr6+ ion sorption onto PBN were best suited by the PSOM (R2 = 1.000) of the four kinetic models. The results of this investigation are comparable to those from studies utilizing wastewater sludge for Cr6+ ion adsorption (Zhang et al. 2013).
From Tables 4 and 5, a PSOM with high R2 values was used to illustrate how well Cr6+ ion eliminated PBN. According to the PSOM, the number of Cr6+ ions on the PBN surface controls the adsorption, and the rate-limiting process might be a chemical adsorption procedure (Qu et al. 2021).
Comparison of findings with those documented in the literature
The effectiveness of the adsorption of Cr6+ ions by various absorbents was compared with the PBN absorbent in the literature review summarized in Table 6 and revealed that the PBN absorbent was effective in adsorption Cr6+ ions.
Regeneration of PBN
Desorption studies of the Cr6+ ions from the loaded PBN were processed by 0.1 M NaOH and 0.1N HCl as the elution media to assess viability and PBN reusability as an absorbent. In this case, the desorption percentage decayed when the regeneration cycles were increased (Fig. 12). Eight consecutive adsorption/desorption cycles were applied using the regenerating PBN. The amount of adsorption remained constant throughout the cycles; however, after eight generations, it decreased by 14.9%. It could be used as a durable Cr6+ ions removal technique (Fig. 12).
Mechanism of Cr6+ ions adsorption by PBN
The mechanism proposed for the adsorption of the Cr6+ ions by PBN was explained in Fig. 13. After the dehydration of dry pea peels with 80% H2SO4 under reflux, many functional groups were formed on the adsorbent (PBS) like –SO3H and –COOH as reported in previous work (El-Nemr et al. 2020a, 2022, 2020b). Oxidation of PBS with 50% H2O2 (El-Nemr, et al. 2022) followed by reflux with 25% NH4OH solution produced PBN adsorbent with different functional groups on the surface, like C=N, NH2, O–H, and C–N groups. Also, from the XPS analysis of PBN (Fig. 5), graphitic-N and pyridinic-N are formed during biochar formation. The mechanism of the removal of Cr6+ ions in an acidic medium may be achieved via physical interaction due to electrostatic interaction between the positive hydrogen ions in the bulk solution and the nitrogen and oxygen functional groups on the PBN surface, then the surface charge became positive; subsequently, electrostatic interaction has occurred between the positively charged surface and the predominant chromium anionic species (HCrO4)–.
Conclusion
In this study, Pea pods biochar-NH2 (PBN) was created to examine the efficacy of Cr6+ ion adsorption from wastewater. It explored how PBN has been characterized and the impacts of pH, starting Cr6+ ions concentration, PBN dosages, and contact time on adsorption. The outcomes demonstrate that the amination method decreased the specific surface area of modified PBN and the size of pores. A significant amount of functional groups are generated because of the reaction with H2O2 and NH4OH solution, which aids in the mass adsorption of Cr6+ ions and creates a surface that can be used for Cr6+ ions adsorption. The equilibrium adsorption for Cr6+ ions gained an adsorption capacity of 384.62 mg/g by PBN at pH 1.5 for 100 ppm of Cr6+ ions concentration and room temperature. More than 70% of final adsorption was obtained at the first rapid stage, which is the most significant for the adsorption applications in the industry. Freundlich IM and PSOM have been selected as the best-fitted IM and KM for adsorption equilibrium analysis. According to the research, pea pod biochar-NH2 (PBN) is an excellent adsorbent option for the adsorption of Cr6+ ions from water.
Availability of data and material
The datasets used in this investigation are accessible for review upon request from the corresponding author of the paper.
References
Abu Taleb M et al (2020) Fabrication of SiO2/CuFe2O4/polyaniline composite: a highly efficient adsorbent for heavy metals removal from aquatic environment. Arab J Chem 13(10):7533–7543
Aguilera A et al (2021) Heavy metal pollution of street dust in the largest city of Mexico, sources and health risk assessment. Environ Monit Assess 193(4):193
Ahmad R, Hasan I (2017) Efficient remediation of an aquatic environment contaminated by Cr(VI) and 2, 4-dinitrophenol by XG-g-polyaniline@ZnO nanocomposite. J Chem Eng Data 62(5):1594–1607
Ahmad R, Hasan I, Mittal A (2017) Adsorption of Cr(VI) and Cd(II) on chitosan grafted polyaniline-OMMT nanocomposite: isotherms, kinetics and thermodynamics studies. Desalin Water Treat 58:144–153
Alegre C, Sebastián D, Lázaro MJ, Girolamo M, Aricò AS, Baglio V (2022) Influence of nitrogen and sulfur doping of carbon Xerogels on the performance and stability of counter electrodes in dye sensitized solar cells. Catalysts 12(3):264
Al-Makishah NH, Taleb MA, Barakat MA (2020) Arsenic bioaccumulation in arsenic-contaminated soil: a review. Chem Pap 74(9):2743–2757
Alyuz B, Veli S (2009) Kinetics and equilibrium studies for the removal of nickel and zinc from aqueous solutions by ion exchange resins. J Hazard Mater 167(1–3):482–488
Bao S et al (2021) Highly efficient and ultrafast removal of Cr(VI)in aqueous solution to ppb level by poly(allylamine hydrochloride) covalently cross-linked amino-modified graphene oxide. J Hazard Mater 409:124470
Barakat MA (2011) New trends in removing heavy metals from industrial wastewater. Arab J Chem 4(4):361–377
Barakat MA et al (2019) Novel Al(2)O(3)/GO/halloysite nanotube composite for sequestration of anionic and cationic dyes. RSC Adv 9(24):13916–13926
Barrett EP, Joyner LG, Halenda PP (1951) The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J Am Chem Soc 73(1):373–380
Bhagat SK, Tung TM, Yaseen ZM (2021) Heavy metal contamination prediction using ensemble model: case study of Bay sedimentation, Australia. J Hazard Mater 403:123492
Bojic A, Bojic D, Andjelkovic T (2009) Removal of Cu2+ and Zn2+ from model wastewaters by spontaneous reduction-coagulation process in flow conditions. J Hazard Mater 168(2–3):813–819
Chai WS et al (2021) A review on conventional and novel materials towards heavy metal adsorption in wastewater treatment application. J Clean Prod 296:126589
Chen H, Yu F, Wang G, Chen L, Dai B, Peng S (2018) Nitrogen and sulfur self-doped activated carbon directly derived from elm flower for high-performance supercapacitors. ACS Omega 3(4):4724–4732
Cheng Z et al (2021) Bottom-up ecofriendly strategy for construction of sustainable bacterial cellulose bioaerogel with multifunctional properties. Adv Mater Interfaces 8(7):2002101
Choudhary B et al (2017) Removal of hexavalent chromium upon interaction with biochar under acidic conditions: mechanistic insights and application. Environ Sci Pollut Res Int 24(20):16786–16797
Crini G et al (2018) Conventional and non-conventional adsorbents for wastewater treatment. Environ Chem Lett 17(1):195–213
Dey S, Bano F, Malik A (2019) Pharmaceuticals and personal care product (PPCP) contamination—a global discharge inventory. Pharmaceuticals and personal care products: waste management and treatment technology. Elsevier, pp 1–26
Dong X, Ma LQ, Li Y (2011) Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. J Hazard Mater 190(1–3):909–915
Duan J et al (2010) Synthesis of a novel flocculant on the basis of crosslinked Konjac glucomannan-graft-polyacrylamide-co-sodium xanthate and its application in removal of Cu2+ ion. Carbohyd Polym 80(2):436–441
El Nemr A (2007) Pomegranate husk as an adsorbent in the removal of toxic chromium from wastewater. Chem Ecol 23:409–425
El Nemr A (2009) Potential of pomegranate husk carbon for Cr(VI) removal from wastewater: kinetic and isotherm studies. J Hazard Mater 161:132–141
El Nemr A, El Sikaily A, Khaled A (2010) Modeling of adsorption isotherms of Methylene Blue onto rice husk activated carbon. Egypt J Aquat Res 36:403–425
El Nemr A, El-Sikaily A, Khaled A, Abdelwahab O (2015) Removal of toxic chromium from aqueous solution, wastewater and saline water by marine red alga Pterocladia capillacea and its activated carbon. Arab J Chem 8:105–117
El Nemr A, Ragab S, El Sikaily A (2017) Rapid synthesis of cellulose triacetate from cotton cellulose and its effect on specific surface area and particle size distribution. Iran Polym J 26(4):261–272. https://doi.org/10.1007/s13726-017-0516-2
El-Nemr MA et al (2020a) Ozone and ammonium hydroxide modification of biochar prepared from Pisum sativum peels improves the adsorption of copper(II) from an aqueous medium. Environ Process 7(3):973–1007
El-Nemr MA, Ismail IMA, Abdelmonem NM, Ragab S, El Nemr A (2020b) Ozone and ammonium hydroxide modification of biochar prepared from Pisum sativum peels improves the adsorption of copper(II) from an aqueous medium. Environ Process 7(2020):973–1007. https://doi.org/10.1007/s40710-020-00455-2
El-Nemr MA, Ismail IM, Abdelmonem NM, El Nemr A, Ragab S (2021) Amination of biochar derived from watermelon peel by triethylenetetramine and ammonium hydroxide for toxic chromium removal enhancement. Chin J Chem Eng 36:199–222
El-Nemr MA, El Nemr A, Hassaan MA, Ragab S, Tedone L, De Mastro G, Pantaleo A (2022) Microporous activated carbon prepared from Pisum sativum pods using different activation methods for adsorptive removal of acid orange 7 dye from water. Molecules 27:4840. https://doi.org/10.3390/molecules27154840
El-Nemr MA et al (2022) Biochar-SO prepared from pea peels by dehydration with sulfuric acid improves the adsorption of Cr6+ from water. Biomass Convers Bioref 2022:1–19
El-Nemr MA, Yılmaz M, Ragab S, El Nemr A (2022) Biochar-SO prepared from Pea peels by dehydration with sulfuric acid improves the adsorption of Cr6+ from water. Biomass Convers Bioref 20:01. https://doi.org/10.1007/s13399-022-02378-4
El-Nemr MA, Yılmaz M, Ragab S, Hassaan MA, El Nemr A (2023) Isotherm and kinetic studies of acid yellow 11 dye adsorption from wastewater using Pisum Sativum peels microporous activated carbon. Sci Rep 13(1):4268
Eniola JO, Kumar R, Barakat MA (2019) Adsorptive removal of antibiotics from water over natural and modified adsorbents. Environ Sci Pollut Res Int 26(34):34775–34788
Etnier CC et al (1981) A survey of sorption relationships for reactive solutes in soil. J Environ Qual 10(1):8–17
Fan LQ, Liu Q, Wan Y, Wang XD, Miao JX, Cai J, Chen W, Chen FH, Cheng L, Ji L (2019) Hexavalent chromium adsorption removal from aqueous solution by fe-modified biochar derived from rice straw. Appl Ecol Environ Res 17:15311–15327
Figoli A et al (2010) Influence of operating parameters on the arsenic removal by nanofiltration. Water Res 44(1):97–104
Fu F, Wang Q (2011) Removal of heavy metal ions from wastewaters: a review. J Environ Manag 92(3):407–418
Glenis S, Nelson A, Labes M (1999) Sulfur doped graphite prepared via arc discharge of carbon rods in the presence of thiophenes. J Appl Phys 86:4464–4466
Golestanifar HA, Haibati B, Amini H, Dehghani MH, Asadi A (2015) Removal of hexavalent chromium from aqueous solution by adsorption on γ-alumina nanoparticles. Environ Prot Eng 41(2):133–45
Gregg SJ, Sing KSW (1982) Adsorption surface area and porosity, 2nd edn. Academic Press, London
Gu W et al (2021) Integrative effect of citrate on Cr(VI) and total Cr removal using a sulfate-reducing bacteria consortium. Chemosphere 279:130437
Gupta G, Khan J, Singh NK (2021) Application and efficacy of low-cost adsorbents for metal removal from contaminated water: a review. Mater Today Proc 43:2958–2964
He X, Li P (2020) Surface water pollution in the middle Chinese loess plateau with special focus on hexavalent chromium (Cr6+): occurrence, sources and health risks. Expo Health 12(3):385–401
Huang Y et al (2021) Column study of enhanced Cr(VI) removal and removal mechanisms by Sporosarcina saromensis W5 assisted bio-permeable reactive barrier. J Hazard Mater 405:124115
Jahan K, Kumar N, Verma V (2021) Bacterial cellulose/PANi mat for Cr(VI) removal at acidic pH. J Appl Polym Sci 138(44):51309
Janiszewska D, Olchowski R, Nowicka A, Zborowska M, Marszałkiewicz K, Shams M, Giannakoudakis DA, Anastopoulos I, Barczak M (2021) Activated biochars derived from wood biomass liquefaction residues for effective removal of hazardous hexavalent chromium from aquatic environments. GCB Bioenergy 13:1247–1259
Junior ACG, Strey L, Lindino CA, Nacke H, Schwantes D, Seidel EP (2012) Applicability of the Pinus bark (Pinus elliottii) for the adsorption of toxic heavy metals from aqueous solutions. Acta Sci Technol 34:79–87
Kabdasli I et al (2009) Complexing agent and heavy metal removals from metal plating effluent by electrocoagulation with stainless steel electrodes. J Hazard Mater 165(1–3):838–845
Kao CW, Wu PT, Liao MY, Chung IJ, Yang KC, Tseng WYI, Yu J (2018) Magnetic nanoparticles conjugated with peptides derived from monocyte chemoattractant protein-1 as a tool for targeting atherosclerosis. Pharmaceutics 10(2):62
Karimi-Maleh H et al (2021) Recent advances in removal techniques of Cr(VI) toxic ion from aqueous solution: a comprehensive review. J Mol Liq 329:115062
Kim ND, Buchholz DB, Casillas G, José-Yacaman M, Chang RP (2014) Hierarchical design for fabricating cost-effective high performance supercapacitors. Adv Funct Mater 24:4186–4194
Kumar MPS, Phanikumar BR (2013) Response surface modelling of Cr6+ adsorption from aqueous solution by neem bark powder: Box-Behnken experimental approach. Environ Sci Pollut Res 20:1327–1343
Kumar R et al (2020) A recyclable multifunctional graphene oxide/SiO2@polyaniline microspheres composite for Cu(II) and Cr(VI) decontamination from wastewater. J Clean Prod 268:122290
Kumar R et al (2020) Experimental design and data on the adsorption and photocatalytic properties of boron nitride/cadmium aluminate composite for Cr(VI) and cefoxitin sodium antibiotic. Data Brief 28:105051
Labidi A et al (2016) Adsorption of copper on chitin-based materials: kinetic and thermodynamic studies. J Taiwan Inst Chem Eng 65:140–148
Lata S, Samadder SR (2016) Removal of arsenic from water using nano adsorbents and challenges: a review. J Environ Manag 166:387–406
Li K et al (2014) Removal of Cr(VI) from aqueous solutions using buckwheat (Fagopyrum esculentumMoench) hull through adsorption-reduction: affecting factors, isotherm, and mechanisms. CLEAN Soil Air Water 42(11):1549–1557
Li D et al (2021) Ammonia leaching mechanism and kinetics of LiCoO2 material from spent lithium-ion batteries. Chin Chem Lett 32(7):2333–2337
Li S, Zhang Z, Zhang C, Yi X, Hassaan MA, El Nemr A, Huang M (2022) Novel hydrophilic straw biochar for the adsorption of neonicotinoids: kinetics, thermodynamics, influencing factors, and reuse performance. Environ Sci Pollut Res 21:11. https://doi.org/10.1007/s11356-022-24131-0
Liang M et al (2020) Removal of aqueous Cr(VI) by magnetic biochar derived from bagasse. Sci Rep 10(1):21473
Liu YL, Cheng H, He YT (2020) Application and mechanism of sludge-based activated carbon for phenol and cyanide removal from bio-treated effluent of coking wastewater. Processes 8(1):82
Lu MM et al (2021) Response of microalgae Chlorella vulgaris to Cr stress and continuous Cr removal in a membrane photobioreactor. Chemosphere 262:128422
Malik DS, Jain CK, Yadav AK (2016) Removal of heavy metals from emerging cellulosic low-cost adsorbents: a review. Appl Water Sci 7(5):2113–2136
Martinson CA, Reddy KJ (2009) Adsorption of arsenic(III) and arsenic(V) by cupric oxide nanoparticles. J Coll Interface Sci 336(2):406–411
Michael P (1991) Introducing of groundwater. Chapaman Hall
Mittal A, Ahmad R, Hasan I (2016) Iron oxide-impregnated dextrin nanocomposite: synthesis and its application for the biosorption of Cr (VI) ions from aqueous solution. Desalin Water Treat 57(32):15133–15145
Mohan D et al (2015) Lead sorptive removal using magnetic and nonmagnetic fast pyrolysis energy cane biochars. J Coll Interface Sci 448:238–250
Mulani K et al (2013) Adsorption of chromium(VI) from aqueous solutions by coffee polyphenol-formaldehyde/acetaldehyde resins. J Polym 2013:1–11
Murad HA et al (2022) A remediation approach to chromium-contaminated water and soil using engineered biochar derived from peanut shell. Environ Res 204(Pt B):112125
Ogamba EN, Charles EE, Izah SC (2021) Distributions, pollution evaluation and health risk of selected heavy metal in surface water of Taylor creek, Bayelsa State, Nigeria. Toxicol Environ Health Sci 13(2):109–121
Olmez T (2009) The optimization of Cr(VI) reduction and removal by electrocoagulation using response surface methodology. J Hazard Mater 162(2–3):1371–1378
Orooji Y et al (2021) Polysaccharide-based (nano)materials for Cr(VI) removal. Int J Biol Macromol 188:950–973
Pandey S, Mishra SB (2011) Organic-inorganic hybrid of chitosan/organoclay bionanocomposites for hexavalent chromium uptake. J Coll Interface Sci 361(2):509–520
Qu J et al (2021) KOH-activated porous biochar with high specific surface area for adsorptive removal of chromium(VI) and naphthalene from water: affecting factors, mechanisms and reusability exploration. J Hazard Mater 401:123292
Qu J et al (2022) Ball milling potassium ferrate activated biochar for efficient chromium and tetracycline decontamination: insights into activation and adsorption mechanisms. Bioresour Technol 360:127407
Rafique MI et al (2021) Immobilization and mitigation of chromium toxicity in aqueous solutions and tannery waste-contaminated soil using biochar and polymer-modified biochar. Chemosphere 266:129198
Ragab S, Eleryan A, El Nemr A (2022) Highly efficient esterification of cellulose using ferric perchlorate hexahydrate at room temperature. Sci Rep 12:5643. https://doi.org/10.1038/s41598-022-09669-w
Raj D, Maiti SK (2020) Sources, bioaccumulation, health risks and remediation of potentially toxic metal(loid)s (As, Cd, Cr, Pb and Hg): an epitomised review. Environ Monit Assess 192(2):108
Rodriguez-Narvaez OM et al (2017) Treatment technologies for emerging contaminants in water: a review. Chem Eng J 323:361–380
Rouquerol F, Rouquerol J, Sing KSW (1999) Adsorption by powders and porous solids. Academic Press, London
Sadrzadeh M et al (2009) Neural network modeling of Pb2+ removal from wastewater using electrodialysis. Chem Eng Process 48(8):1371–1381
Shahalam AM, Al-Harthy A, Al-Zawhry A (2002) Feed water pretreatment in RO systems: unit processes in the Middle East. Desalination 150:235–245
Sinha R et al (2022) Single-step synthesis of activated magnetic biochar derived from rice husk for hexavalent chromium adsorption: Equilibrium mechanism, kinetics, and thermodynamics analysis. Groundw Sustain Dev 18:100796
Sun X et al (2021) Adsorption of hexavalent chromium by sodium alginate fiber biochar loaded with lanthanum. Materials (Basel) 14(9):2224
Syeda HI, Yap PS (2022) A review on three-dimensional cellulose-based aerogels for the removal of heavy metals from water. Sci Total Environ 807(Pt 1):150606
Vasudevan M, Ajithkumar PS, Singh RP, Natarajan N (2016) Mass transfer kinetics using two-site interface model for removal of Cr(VI) from aqueous solution with cassava peel and rubber tree bark as adsorbents. Environ Eng Res 21:152–163
Verma A, Chakraborty S, Basu JK (2006) Adsorption study of hexavalent chromium using tamarind hull-based adsorbents. Sep Purif Technol 50(3):336–341
Vikrant Sarin KKP (2006) Removal of chromium from industrial waste by using eucalyptus bark. Bioresour Technol 97:15–20
Wang SY et al (2014) Combined performance of biochar sorption and magnetic separation processes for treatment of chromium-contained electroplating wastewater. Bioresour Technol 174:67–73
Wang D et al (2020) Fabrication and functionalization of biological graphene aerogel by reusing microorganism in activated sludge and ionic dyes. Chem Eng J 392:124823
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div 89:31–60
Xiang B et al (2016) Dithiocarbamate-modified starch derivatives with high heavy metal adsorption performance. Carbohydr Polym 136:30–37
Xiao R et al (2018) Enhanced sorption of hexavalent chromium [Cr(VI)] from aqueous solutions by diluted sulfuric acid-assisted MgO-coated biochar composite. Chemosphere 208:408–416
Xing Z, Ju Z, Zhao Y, Wan J, Zhu Y, Qiang Y, Qian Y (2016) One-pot hydrothermal synthesis of Nitrogen-doped graphene as high-performance anode materials for lithium ion batteries. Sci Rep 6(1):1–10
Xu S et al (2021) Synthesis, optical characterization, and adsorption of novel hexavalent chromium and total chromium sorbent: a fabrication of mulberry stem biochar/Mn-Fe binary oxide composite via response surface methodology. Front Environ Chem 2:692810
Yang R, Li KX, Lv CC, Cen BQ, Wang L, Liang BL (2019) The excellent performance of nitrogen-doped porous carbon nanowires modified activated carbon as air cathode catalyst for microbial fuel cells. J Solid State Electrochem 23(12):3437–3447. https://doi.org/10.1007/s10008-019-04403-5
Yin Y et al (2019) Adsorption of arsenic by activated charcoal coated zirconium-manganese nanocomposite: performance and mechanism. Coll Surf A 575:318–328
Yin P et al (2020a) One-step prepared prussian blue/porous carbon composite derives highly efficient Fe–N–C catalyst for oxygen reduction. Int J Hydrogen Energy 45(30):15100–15111
Yin Z et al (2020b) A novel magnetic biochar prepared by K(2)FeO(4)-promoted oxidative pyrolysis of pomelo peel for adsorption of hexavalent chromium. Bioresour Technol 300:122680
Yuan P et al (2009) Montmorillonite-supported magnetite nanoparticles for the removal of hexavalent chromium [Cr(VI)] from aqueous solutions. J Hazard Mater 166(2–3):821–829
Zamboulis D et al (2011) Metal ion separation and recovery from environmental sources using various flotation and sorption techniques. J Chem Technol Biotechnol 86(3):335–344
Zhang W et al (2013) Pb(II) and Cr(VI) sorption by biochars pyrolyzed from the municipal wastewater sludge under different heating conditions. Bioresour Technol 147:545–552
Zhang P et al (2019) Microwave-assisted chemical modification method for surface regulation of biochar and its application for estrogen removal. Process Saf Environ Prot 128:329–341
Zhang M, Sun R, Song G, Wu L, Ye H, Xu L, Parikh SJ, Nguyen T, Khan E, Vithanage M, Ok YS (2022) Enhanced removal of ammonium from water using sulfonated reed waste biochar-A lab-scale investigation. Environ Pollut 292:118412
Zhanqi G et al (2007) Microwave assisted rapid and complete degradation of atrazine using TiO(2) nanotube photocatalyst suspensions. J Hazard Mater 145(3):424–430
Zhu D et al (2021) Nano nickel embedded in N-doped CNTs-supported porous biochar for adsorption-reduction of hexavalent chromium. J Hazard Mater 416:125693
Zhu Y et al (2021) Insight into efficient removal of Cr(VI) by magnetite immobilized with Lysinibacillus sp. JLT12: mechanism and performance. Chemosphere 262:127901
Zhu X et al (2022) Machine learning exploration of the direct and indirect roles of Fe impregnation on Cr(VI) removal by engineered biochar. Chem Eng J 428:131967
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This work was partially supported by The Science, Technology and Innovation Funding Authority (STDF) of Egypt, project number SCSE-31235.
Author information
Authors and Affiliations
Contributions
MAE conceived the main idea of the paper and performed the experiment and theoretical calculation. MY wrote the original manuscript. SR and MAH helped prepare the characterization analyses and contributed various comments and ideas to help improve the overall quality of the research. BAM helped in writing the introduction. AEN designed the experiment, supervised the work, revised the manuscript and submitted it to the journal. All authors have reviewed and approved the paper in its published form.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participate
Consent to participate is not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Nemr, M.A., Yılmaz, M., Ragab, S. et al. Fabrication of Pea pods biochar-NH2 (PBN) for the adsorption of toxic Cr6+ ion from aqueous solution. Appl Water Sci 13, 194 (2023). https://doi.org/10.1007/s13201-023-02007-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-023-02007-z