Abstract
The field of mycology has grown from an underappreciated subset of botany, to a valuable, modern scientific discipline. As this field of study has grown, there have been significant contributions to science, technology, and industry, highlighting the value of fungi in the modern era. This paper looks at the current research, along with the existing limitations, and suggests future areas where scientists can focus their efforts, in the field mycology. We show how fungi have become important emerging diseases in medical mycology. We discuss current trends and the potential of fungi in drug and novel compound discovery. We explore the current trends in phylogenomics, its potential, and outcomes and address the question of how phylogenomics can be applied in fungal ecology. In addition, the trends in functional genomics studies of fungi are discussed with their importance in unravelling the intricate mechanisms underlying fungal behaviour, interactions, and adaptations, paving the way for a comprehensive understanding of fungal biology. We look at the current research in building materials, how they can be used as carbon sinks, and how fungi can be used in biocircular economies. The numbers of fungi have always been of great interest and have often been written about and estimates have varied greatly. Thus, we discuss current trends and future research needs in order to obtain more reliable estimates. We address the aspects of machine learning (AI) and how it can be used in mycological research. Plant pathogens are affecting food production systems on a global scale, and as such, we look at the current trends and future research needed in this area, particularly in disease detection. We look at the latest data from High Throughput Sequencing studies and question if we are still gaining new knowledge at the same rate as before. A review of current trends in nanotechnology is provided and its future potential is addressed. The importance of Arbuscular Mycorrhizal Fungi is addressed and future trends are acknowledged. Fungal databases are becoming more and more important, and we therefore provide a review of the current major databases. Edible and medicinal fungi have a huge potential as food and medicines, especially in Asia and their prospects are discussed. Lifestyle changes in fungi (e.g., from endophytes, to pathogens, and/or saprobes) are also extremely important and a current research trend and are therefore addressed in this special issue of Fungal Diversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Studies on fungi were previously frowned upon. Alison Pouliot (2023) wrote in The Guardian “Fungi have endured a long history of neglect and disdain. In 1887 the British mycologist William Hay commented that he who studied fungi “must boldly face a good deal of scorn … and is actually regarded as a sort of idiot among the lower orders”. A few years earlier the nature writer Margaret Plues observed how the stranger “blinded by conventionalities” sneered at those seeking fungi. Unlike birders who look upwards for their charismatic avian delights, fungus hunters glance downwards, for what the “father of modern taxonomy”, Swedish biologist Carl Linnaeus, referred to as “thievish and voracious beggars”.” This has certainly changed with studies on fungi now being looked at much more positively, especially with their importance in human, animal and plant disease (Hyde et al. 2018; Mapook et al. 2022; Niego et al. 2023a, b), potential in novel compound discovery (Hyde et al. 2019; Mapook et al. 2022), value in industrial applications (Hyde et al. 2019) and potential in the biocircular economy (Kirchherr et al. 2017; Meyer et al. 2016, 2020).
The effect of metrics on mycology
An often-overlooked topic is the effect of Metrics on the study of Mycology. We can only speak from our experience, but it would be interesting to see how this affected other mycologists. In the 1990s the Web of Science started to influence university assessments (1996 onwards) and promotions. At the time the highest impact mycology journals, Mycologia and Mycological Research, had impact factors of 1–2, while some ecological journals had impact factors of 3–4. Therefore, the peer pressure led some of us to turn towards ecological research, so we published several papers at the end of the Century with an ecological slant (e.g., Hyde and Lee 1998; Taylor et al. 1999; Luo et al. 2004; Tao et al. 2008). At the turn of the century, molecular research was becoming important and journals specialising in this topic also had higher impact factors of 3–4. So again, the trend was to do molecular research and publish in these high impact journals (e.g., Liew et al. 2000, 2002; Guo et al. 2001).
During the early 2000s the ecological and molecular trends continued, whereas after 2010 some mycological journals overtook the ecological journals, so ecological studies in mycology became less important. As time progressed, the impact factors of some mainstream mycological journals (Fungal Diversity, Persoonia, Studies in Mycology) increased and these became the desired journals to publish in. The focus of these journals was mainly diversity, taxonomy, and phylogeny and thus these topics became popular. Thus, our research also focused on this area and we published many important papers during this period (e.g., Shearer et al. 2010; Hyde et al. 2013, 2014a, b). More recently it has become important to publish in Quartile 1 journals and Fungal Biology Reviews, Fungal Diversity, IMAfungus, the Journal of Fungi, Mycosphere and Studies in Mycology have become desirable journals. The future is hard to predict, phylogenomics and functional genomics have become hot topics, however in mycology this is not matched by the high-impact journals where such papers are published, but this may change. At least for the time being biodiversity, taxonomy and phylogenetics will remain important topics as papers can be published in high-impact journals.
The effect of Web of Science metrics on books has been extremely detrimental. During the mid stages of the careers of some of us published more than 20 books. At the time books were needed for promotions. As for now, there is very little kudos from publishing books, as they have no impact factor and scientific books make little profit. This is why very few comprehensive monographs are presently published.
In conclusion, there is no doubt that the Web of Science has had a profound influence on the research and publications in mycology. Whether one considers this a good or bad thing will depend very much on the researcher's interests.
The golden era of mycology
We the senior authors of this paper began their careers in the 1980s, the number of mycologists was declining and places such as CBS, IMI, Kew, and British Universities were shedding these specialists at an alarming rate. In this period, it was hugely difficult to obtain funding and as stated above, the impact factor of mycological journals was low. Thus, it was almost impossible for a young Ph.D. to obtain a university position. Fast forward to the present day, the emerging economies of Asia and South America are recruiting large numbers of mycologists. Even The Royal Botanical Gardens, Kew, has realised the importance of mycology and created several new positions for mycologists. It has become much easier to obtain funding and the impact factors of some mycology journals are some of the highest in specialised science. Thus, it is the golden era of mycology and this should last for some time to come because of the applied potential of the fungi (Hyde et al. 2019).
Below we provide notes on 15 important research trends and discuss their current research, limitations, and future expectations. We use these trends to indicate the expected research in mycology in the future.
Current trends, limitations, and future research in emerging diseases and their control
As an applied area of research that is directly linked to human health, medical mycology is probably one of the most rapidly evolving subdisciplines. Fundamental changes are taking place in all aspects: in the fungi and their evolution, in the hosts and their immunity, in the environmental conditions of human lifestyles, and at a global scale in climate and sustainability.
From a fungal perspective, hosts have become available with dramatically altered susceptibility to infection due to immunological alterations, constitutional diseases, or as a side effect of medical intervention. The second half of the twentieth century is marked by the emergence of patient populations that were immunocompromised in view of the treatment of life-threatening or chronic inflammatory and systemic diseases, while also organ transplants became standard therapy. In the USA, an estimated 2.7% of the population may have evidence of immunosuppression (Harpaz et al. 2016), and this number is growing due to increasing life expectancy and eldering demography in large parts of the world. Immunosuppression generally aims for a temporary or local decrease of innate cellular immune responses. Inevitably, this highly successful intervention has a downside in the emergence of a wide diversity of environmental fungi as opportunistic pathogens (de Hoog et al. 2020), many of which were previously unknown to medical mycology. The ability of these fungi to adapt to the compromised conditions within the debilitated host and potentially tolerate human endothermic conditions are the key factors in the pathogenic processes of environmental fungi (Köhler et al. 2014). At the scale of the individual patient, management and control of fungal disease is dependent on the level of diagnostics, understanding of pathogenic processes, knowledge of fungus-host interaction, and available therapy. This enables precision medicine focused on defined patient groups (Märtson et al. 2021), with antifungal stewardship leading to the design of personalised, tailored therapy preventing inappropriate use of prophylaxis (Singh et al. 2018).
More epidemics are expected
Microbial epidemics have always battered humanity and are expected to increase in frequency. The early pandemics, like plague and cholera, mostly originated from polluted water, as urban hygiene was at a low level until the nineteenth century, but the majority of the recent pandemics are zoonotic, and the intervals between the epidemics are decreasing. We recently witnessed the HIV, MERS, SARS, and COVID-19 pandemics that originated from primates, camels, and possibly from bats and intermediate hosts. Among novel threats are avian and swine flu which already are associated with human infection, having reached stage 2 (in a scale of 5; Piret and Boivin 2021) in adaptation to humans. Large-scale animal breeding, global trade of exotic animals, increased population density, and intercontinental travel enhance the spread, spill-over, and adaptation of microbial pathogens. In fungi, these often concern multiple infections from a single source, e.g., with Sporothrix schenckii from plant material (Dooley et al. 1997; Govender et al. 2015) or Verruconis gallopava from contaminated straw in a chicken coop (Blalock et al. 1973). Others are promoted by viral infections damaging the human cellular immune system. The HIV pandemic led to pronounced infections of true pathogens (Carpouron et al. 2022) in Cryptococcus, Sporothrix, Talaromyces, and Histoplasma. The COVID-19 pandemic enhanced the infection of opportunists in Rhizopus (Singh et al. 2021; Nehara et al. 2021) and Aspergillus (Calderón-Parra et al. 2022). Serious problems are looming in the monoculture of animal farming, where large numbers of animals are packed within limited space. The agent of goat Q-fever, Coxiella burnetii and that of bird flu, H7N9 Influenza-A are already becoming endemic in Europe, and both can be transmitted to humans with serious consequences. Unexpected fungi may follow in their slipstream. As a result, bioindustry plants today are close to implementing similar biosafety measures as microbiological laboratories.
Changes in lifestyle follow a global trend. In the past, endemic infectious diseases and regional epidemics were associated with agricultural labour on the field. Traumatic eye and subcutaneous infections from plant material and dermatophytoses acquired from farm animals were prevalent. Today, the farmer is placed at a distance from infectious agents by modern technology. In addition, rural populations diminish worldwide as a result of urbanization. Diseases like chromoblastomycosis by black fungi or cattle-borne Trichophyton verrucosum infection are disappearing. They are replaced by infections particularly involving from pet animals (Zhan et al. 2015). For example, 44.5% of U.S. households own dogs, and 29% cats (www.forbes.com). The spectrum of animals held is becoming wider, with novel agents from exotic animals appearing, such as the emergence of Trichophyton erinacei from the African pygmy hedgehog (Hsieh et al. 2010).
An interdisciplinary One-health approach is mandatory to prevent unforeseen and expected consequences of intensive agriculture. Evaluation of emerging infectious diseases can be done in a broader sense e.g. by reducing monoculture, regardless of the specific etiologic pathogen, and comparative genomics will allow further investigation of genetic and biological characteristics, especially in terms of adaptation to new habitats. Population translocation is accelerated by human-made climate change (Kimutai et al. 2022), as large areas are likely to become uninhabitable (https://www.un.org/africarenewal/). Human populations will spread disease at an unprecedented pace, conversely, in the wealthy part of the world, global travel is likely to increase. Also, fungal biodiversity patterns and mycobiomes are changing under the pressure of changing environmental conditions (Větrovský et al. 2019; Case et al. 2022), making it harder to predict epidemic expansions. Future pandemics will be very hard to control.
Problematic therapy and future solutions
Fungi are expected to be the infectious health problem of the future, because infections are chronic and recalcitrant, often with poor host response despite in vitro susceptibility (Vinh et al. 2023; Berman et al. 2020). The emerging resistance to commonly used antifungals in some of the major opportunists is a global concern in medical mycology. In addition, the discordance between overall treatment outcome and low levels of clinical resistance may be attributable to antifungal drug tolerance (Berman et al. 2020). Acquired antifungal resistance (Fisher et al. 2022) is a probable result of similar compounds being used in agriculture (Barber et al. 2020). In regions with intense farming, Aspergillus fumigatus comprises up to 20% of azole-resistant strains in natural populations (Chowdhary et al. 2013). The dermatophyte Trichophyton indotineae is often resistant to terbinafine, which is the main antifungal used by the public (Singh et al. 2018). In several orders of the fungal kingdom, such as Hypocreales, Microascales, and Mucorales, resistance is not acquired, but intrinsically present, even in the absence of previous exposure to antifungals (Caramalho et al. 2017). Also, some yeasts, among which is the emerging hospital agent Candida auris have reduced susceptibility, causing problems comparable to those of MRSA. Despite advances in antifungal therapy, there are few drug classes. New generations of antifungals are being developed with novel targets, such as echinocandins and orotomides, which are promising but generally are effective in only a limited number of species, or luliconazole which can only be applied topically. Currently, several promising antifungal agents undergoing clinical investigation, each with unique mechanisms of action. These include novamycin, an antifungal peptide that disrupts the plasma membrane, leading to cell lysis. Another agent, olorofim, belongs to the orotomide class of drugs and inhibits pyrimidine biosynthesis by reversibly inhibiting mitochondrial dihydroorotate dehydrogenase. Additionally, fosmanogepix, a GPI inhibitor, hinders the activity of Gwt1, preventing GPI anchoring. These agents exemplify novel drug targets and showcase their potential in the field of antifungal drug development. There are multiple bottlenecks to the successful development of antifungal drugs due to the unique characteristics of fungal cells. These challenges include the need to establish high-throughput screening methods, optimize target specificity and bioavailability, and minimize host toxicity to ensure efficacy. In silico approaches have the potential to expedite the early stages of drug development; however, their effectiveness relies on the availability of high-resolution protein structures and a strong mechanistic understanding of signaling pathways and the impact of therapeutic intervention on fungal physiology (Velazhahan et al. 2023).
Novel types of therapy have been applied to some of the classical, severe, and previously untreatable diseases that ultimately led to the death of the patient. In the past these were ascribed to supposedly uncommon, highly virulent fungi (Rajendran et al. 2003); particularly the mutilating diseases by black fungi (Bonifaz et al. 2013) and Mucor irregularis (Kang et al. 2014) have been mysterious. Today, these fungi are understood as being opportunists in patients with inherited immune disorders (Lanternier et al. 2015). Mutations in the essential dectin-signalling CARD9 protein increase susceptibility to particular fungal infections (Song et al. 2020). These primary immunodeficiencies include several hundreds of single-gene inborn errors of immunity and occur in close to 1% of the human population (Quinn et al. 2022). Similar to the increase in allergic diseases in the developed world, this problem is likely to grow in the future. Classical antifungal therapy is only temporarily successful in these patients due to emerging resistance. Immunotherapy or stem cell transplantation seems to provide a successful cure (Grumach et al. 2015) and may open new ways of therapy in chronic infections. For pathogens that are controlled by adaptive cellular immunity, the development of preventative tools such as vaccines might be an option (Oliveira et al. 2021).
The need for reliable diagnostics
Medical mycology comprises 778 species in 250 genera (www.atlasclinicalfungi.org, dd 01–04–2023), and this number is increasing nearly every month. General knowledge of the great majority of these species is scant, while a very limited number of prevalent clinical taxa have been thoroughly investigated. The recent emergence of novel agents, such as Candida auris and Sporothrix brasiliensis, demonstrates severe gaps in understanding sources and routes of infection, potential pathology, immune response, and therapy. Numerous taxa are described on the basis of just a few isolates, neglecting intraspecific variability. Without an understanding of clonality, recombination, uniparental sex, and horizontal gene transfer, it is difficult to develop reliable diagnostics for hospital routines (Song et al. 2023). Diversity studies and taxonomy are fundamental but need a stronger integration with the sciences that follow (de Hoog et al. 2023). The focus on molecular phylogeny overshadows the comprehension of the practical significance of the described entities for applied sciences and patient care. Phylogeny provides insight into adaptive trends between species, and nomenclature opens the doors toward existing literature. However, phylogeny-based rearrangement of classical genera leads to fragmentation, such that the coherence between pathogens is lost again. For example, the 52 medically relevant candida-like species are now dispersed over 19 genera and this process is continuing. This underlines the responsibility of the taxonomist to provide a workable system.
One limitation in controlling emerging diseases is the lack of rapid diagnostic tools and surveillance systems. Timely detection and identification of pathogens are essential for implementing effective control measures. Diagnostic methods are reliable only when based on sound taxonomy. The future of diagnostics may include multi-microbial tools that can provide a comprehensive view of microbial diversity and detect bacteria, fungi, and viruses simultaneously in clinical samples. Currently, protein- and DNA-based methods are available such as PCR, gene and genome sequencing, metagenomics, and MALDI-ToF, but these are not suitable for point-of-care diagnosis (POD), particularly in resource-limited settings or during outbreaks. Based on advanced knowledge, simplified techniques can be developed that are applicable in a wide range of settings and require less human implementation (Prattes et al. 2016) and are therefore particularly useful in developing regions (Osaigbovo and Bongomin 2021). An artificial intelligence approach has been evaluated for the autodetection of fungal hyphae from a microscopic image and seems promising (Koo et al. 2021). Additionally, microfluidic approaches such as lab-on-chip (Richter et al. 2022) can be utilized to promote fast detection of host/microbial biomarkers.
Overall, developing and improving diagnostic techniques and surveillance capabilities should be a priority. Future research should focus on enhancing our understanding of the ecological factors contributing to disease emergence. Exploring the complex interactions between pathogens, hosts, and the environment can help identify high-risk areas and predict disease outbreaks. Furthermore, strengthening international collaborations, data sharing, and communication networks is essential for a coordinated global response to emerging fungal infections.
Current trends, limitations, and future research in novel compound and drug discovery
Role of fungi in classical natural product-based drug discovery
Natural products have historically been one of the most important sources for human therapeutics, and they remain indispensable for the discovery and development of new drugs (Atanasov et al. 2021; Newman and Cragg 2020). As summarized by Bills and Gloer (2016), several of the most important pharmaceuticals and agrochemicals are derived from fungi. Aside from the blockbuster cardiovascular drugs like statins and the immunomodulating compounds like cyclosporine and fingolimod (Mapook et al. 2022; Niego et al. 2023b), the beta-lactams are certainly the best-known class of fungi-derived drugs (Fig. 1).
Since the discovery of penicillin, scientists have turned to fungi to find cures for mankind's diseases. Out of this arose the blooming age of antibiotics in the late 1940s which lasted until the 1960s (Karwehl and Stadler 2016; da Cunha et al. 2019), followed by a period of neglectful thinking that such drugs would always be available and last for eternity. This has given rise to resistance against these drugs and has slowly become a mass problem (Mitra et al. 2022). Roughly 60 years later, the focus has now turned on having to find new drugs predominantly against superbugs, strains of, e.g. bacteria, which are resistant to multiple classes of antibiotics, if not all, as in the case of some recalcitrant strains of Mycobacterium tuberculosis (Gygli et al. 2017). By far the most dangerous bacterial pathogens, however, are the organisms that are classified in the ESKAPE panel: Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa und Enterobacter spp. often become multi-resistant and are responsible for an increasing number of deaths (Miethke et al. 2021).
The main research focus in the medical field over the last four decades has shifted to different topics, such as finding chemotherapeutics for cancerous diseases (a field, in which natural products also are playing a major role), but in particular for diabetes and other metabolic disorders, cardiovascular drugs and other indications in which higher revenues can be expected. One of the reasons for this shift was the fact that the development costs for drugs have exploded, and regardless of the indication, the development of novel drugs from scratch may now cost up to a billion USD (Wouters et al. 2020). Especially because of the extremely high attrition rates, it became less fashionable to develop totally novel molecules from natural sources to drugs, and the antibiotics and other anti-infective sectors in which the natural products are particularly strong have been heavily affected by this trend. (Atanasov et al. 2021).
With this focal shift, many biomedical research branches are now devoted to drug development for these and other types of diseases of affluence. Therefore, the major contributor to new FDA-approved drugs over the past 30 years has been “biologicals” (macromolecular medicinal products of fermentative origin) (Rader 2008; Newman and Cragg 2020; Butler et al. 2023). Its most prominent representative is of course recombinant human insulin (Falcetta et al. 2022). Generally, their use is currently focused primarily on oncology, rheumatology, gastroenterology, and cardiology (Walsh and Walsh 2022).
Thus, the main problem remains: finding new drugs against superbugs and bringing them to the market as quickly as possible, and counteracting their resistance. Therefore, classical drug discovery research, which is based on natural product chemistry, has been given new attention in the last few years. Natural products have their fair share in the approval of new drugs by the FDA, while in contrast, the approval of new synthetic drugs has almost disappeared (Newman and Cragg 2020; Butler et al. 2023), the proportion of fungal natural products among the newly approved drugs is relatively small.
Yet, at the same time, the last approved new compound class of antibiotics, pleuromutilins, that came onto the market are derived from fungi (Paukner and Riedl 2017; Mapook et al. 2022). With the diverse range of natural products fungi have to offer, they remain one of the most promising sources for new lead structures. In addition, science has dedicated itself to the development of new dosage forms, particularly nanotechnology, which is a cutting-edge research topic at present. One of the most promising fungal metabolites with biomedical use in this regard is chitinosan, a fungal chitin-derivative under investigation as a drug delivery system, as hemostatic, and as medical material for wound healing (Nawawi et al. 2020). This would make natural products more bioavailable and thus a lot more interesting for use as drugs.
One big problem of the pharmaceutical industry is that all large companies have given up or outsourced their capacities for natural product research and development. Not only for the mycological field, but the search for novel compounds from nature also requires the availability of interdisciplinary resources and expertise, ranging from classical microbiology via know-how in biotechnology to analytical and preparative natural product chemistry (Fig. 2). Once a given company has abandoned these R&D activities, which had historically been built up over several decades, it may take decades to get back the know-how.
Overview of current state of fungal drug research (created with www.BioRender.com)
On the other hand, there is hope. A large number of bioactivity screening systems are nowadays available for newly discovered natural products, which often only require small amounts compared to the past. This does not mean that already known natural products cannot be tested in these new assays as well, and stand out as hits. One such example would be rubiginosin C (Quang et al. 2004), which has been known from Hypoxylon rubiginosum for almost 20 years, yet only recently stood out to be a highly promising biofilm inhibitor of pathogenic Candida spp. (Zeng et al. 2023).
Just as bioactivity assays need less and less amount of material, classic natural products drug discovery has changed in this regard. New technologies in the field of structure elucidation, mainly nuclear magnetic resonance spectroscopy (NMR) and mass spectrometry (MS) allow for structure elucidation of smaller and smaller amounts. One look into the respective literature shows that natural product isolation and characterization of less than 1 mg has become the norm, with a trend to even lower amounts (< 0.5 mg), such as five unprecedented diketopiperazines from the endophytic fungus Batnamyces globulariicola, cultured from the medicinal plant Globularia alypum, native to Algeria (Noumeur et al. 2020). This opens up drug discovery in completely new territories, which have previously been disclosed. Not only geographical territories, where in recent times fungal natural products research has had and is focusing predominantly on tropical, understudied regions (Sandargo et al. 2019), but also specialized ecological niches, can thus now be investigated for potential drug producers.
A challenge here can be the adherence to all legal procedures, such as the Nagoya protocol (Heinrich and Hesketh 2019). However, the greater limitation in all these cases lies with the cultivation conditions, which in turn can lead to challenges in the reproduction of interesting drug candidates and makes an upscaling of these more difficult, as well as potentially elaborate isolation procedures. After all, a complex upstream process (USP) followed by a lavish downstream process (DSP) leads to a high cost of goods, which makes a drug candidate unappealing (Suresh and Basu 2008).
A frequent solution to this problem is the fermentation of an easier-to-produce precursor molecule which is then chemically modified in further steps, called “semisynthesis”. A majority of fungal drugs on the market have at least one step of semisynthesis, not only to increase production, but largely to improve bioavailability (like micafungin, an antifungal agent semi-synthesized from fermentative echinocandin; Hashimoto 2009), or to improve its (selective) bioactivity (like ceftaroline fosamil (teflaro® / Zinforo®) the last generation semisynthetic cephem to enter the market with improved selectivity for multi-resistant Gram-positive bacteria, Newman and Cragg 2020). In some cases, semisynthesis is also used to lower the actual bioactivity, such as in the case of irofulven, a lesser toxic derivative of illudin S (Chaverra-Muñoz and Hüttel 2022). These chemical modifications, a part of a vast field of medicinal chemistry, are becoming increasingly important and it is impossible to imagine a future without them. The best example of the success of a semisynthetic approach based on fungal metabolites is probably the cephalosporins, where five generations of compound families were synthesized over several decades, resulting in substantial improvements in efficacy against human pathogens (Lin and Kück 2022).
However, the now-established way of producing non-synthesizable natural substances more cost-effectively is synthetic biology, which introduces the BGCs into a new, easier-to-cultivate host via heterologous production or can even allow for total biosynthesis of novel natural products. As shown for strobilurins (Nofiani et al. 2018) and pleuromutilins (Alberti et al. 2017a, b) it has even been possible to transfer the BGC from Basidiomycota into workhorses like Aspergillus (flavus var.) or yzae that belong to the Ascomycota. It has even been possible to synthesize natural products using enzymes, as recently demonstrated for psilocybin by Fricke et al. (2017). The mechanisms of many biosynthetic pathways, including those that encode for rather rare and complicated metabolite classes, have been elucidated over the past years (Feng et al. 2020; Tian et al. 2020; Schotte et al. 2020).
Current trends and future potential
When the first genomes of filamentous fungi became available, the large number of biosynthetic gene clusters in these fungi surprised the researchers, as the number of clusters by far succeeded the diversity of SMs known from these fungi during previous natural product isolation studies. Since then, genome mining has been established as an alternative way for the discovery of new secondary metabolites. Its common workflow is the identification of biosynthetic gene clusters (BGCs), the heterologous expression or activation of interesting BGCs, followed by metabolite characterization. Examples (Fig. 3) yielded in novel carbon skeletons include the discovery of new meroterpenoids from a gene cluster encoding polyketide synthase, prenyltransferase, terpenoid cyclase, and other tailoring enzymes (Zhang et al. 2018); the discovery of flavunoidine, whose gene cluster contains two terpene gene clusters and a nonribosomal peptide synthetase (Yee et al. 2020) and the sesterterpenes schultriene and nigtetraene discovered by heterologous expression of fungal bifunctional terpene syntases (Jiang et al. 2022). Arbusulic acid was isolated besides other compounds after epigenetic activation of the endophytic fungus Calcarisporium arbuscula (Mao et al. 2015), whereas the production of tripyridone in Aspergillus nidulans was induced by co-cultivation with filamentous Streptomycetes bacteria producing antifungal glycopeptide antibiotics (Gerke et al. 2022).
Precondition for the genome mining is the availability of full genome information to be mined. Although fewer fungal genomes have been sequenced compared to the bacterial kingdom, more than 1000 fungal genomes have already been interpreted for secondary metabolite production (Robey et al. 2021). However, most of those studies focused on well-studied genera such as Aspergillus, Fusarium, and Penicillium.
Thus, it is important to expand sequencing efforts to less studied, nevertheless very prolific groups of fungi. Based on high-quality genome sequences for 13 representative species of Hypoxylaceae, Kuhnert et al. (2021) surveyed the diversity of biosynthetic pathways and found 783 biosynthetic pathways across the 14 studied species, the majority of which were arranged in biosynthetic gene clusters. Of the 375 gene cluster families (GCF) found, only ten GCFs were conserved across all of these fungi, indicating that a high degree of speciation is accompanied by changes in secondary metabolism. Besides giving insights in the genetic background that drives the production and diversification of secondary metabolites, this and similar studies will help to systematically access the secondary metabolites of prolific fungal secondary metabolite producers.
A drawback of the genome mining approaches is that often compounds with no obvious bioactivity are isolated. Thus, target-directed genome mining efforts try to increase the odds of finding bioactive compounds. Because antimicrobial producers must be self-resistant to their own metabolites, fungi often encode resistance genes to avoid self-toxicity in the BGCs of corresponding bioactive compounds. FunARTS, the Fungal bioActive compound Resistant Target Seeker, is a recent exploration engine to specifically mine for these resistance genes (Yilmaz et al. 2023).
So far, genome mining has largely focused on unknown metabolites from predicted BGCs with known core enzymes, like PKS, NRPS, and terpene synthases. However, the most spectacular findings are expected in the search for unknown secondary metabolites with so far unidentified types of core enzymes, the so-called “unknown-unknowns”. Yee et al. (2023) identified a new arginine-containing cyclodepsipeptide synthase, which is responsible for a highly modified cyclo-arginine-tyrosine dipeptide. However, since the scaffold of this compound was already known from derivatives, it remains to be seen if this method can meet the high expectations.
Metabolomics is a recently developed, complementary research field. The identification of known compounds, a process known as dereplication, is readily possible with HPLC–MS/MS data from crude extracts if reference data are available. Thus, the integration of genomics and metabolomics has the potential to revolutionize fungal natural product research (Hautbergue et al. 2018). Beyond metabolomics, the integration of transcriptomics and proteomics data may provide an additional layer of information for pathway analysis (“multi-omics”). However, it is very important to reduce the complexity of the vast amount of data in a sensible way (Rinschen et al. 2019). Of particular interest is the systematic connection of pathway data to biological activities. However, it is, of course, not possible to assess the biological effects of single components in mixtures like extracts unless the respective compounds can be isolated to purity. The same holds true for their unambiguous identification, for which techniques like 2D-NMR spectroscopy, crystallization, or derivatization remain indispensable.
Furthermore, synthetic biology, designing and constructing biological modules, biological systems, and biological machines or re-design of existing biological systems for useful purposes, will greatly help to access the chemical diversity uncovered by genome mining (Skellam et al. 2019; Keller 2019). The combination of genome mining with a synthetic biology-based method of heterologous biosynthesis is a promising approach to rationally access NPs with novel structures and biological activities. As an example, this method was used to explore the biological activity-related chemical space of fungal decalin-containing diterpenoid pyrones (Tsukada et al. 2020). Even though these compounds do not have very prominent and selective biological activities, the latter study shows the feasibility of the approach to expand the chemical space. Meroterpenoids (Matsuda and Abe 2016), NRPS-PKS hybrids (Boettger and Hertweck 2013), and other compounds of mixed biosynthetic origin are increasingly being studied, also because their BGC can be made out in the genomes rather easily.
Finally, yet importantly, the application of artificial intelligence (AI) to natural product drug discovery has grown tremendously in recent years, due to ever-increasing computing power, extensive storage, and accessible software (Saldivar-Gonzalez et al. 2022). Thus, we expect the rational application of AI to assist in discovering bioactive natural products and capturing the molecular “patterns” of these privileged structures for combinatorial design or target selectivity. Main help will most likely be the prediction of chemical structures from microbial genomes and automation of the natural product dereplication process, the identification of NP substructures, computer-assisted structure elucidation, and virtual screening approaches. Additionally, the prediction of biological functions of the NPs will be afforded by the recent advances in machine learning having leveraged to accelerate the accurate atomic-resolution structure prediction of proteins (Lin et al. 2023a, b).
Conclusion
Above we have summarized the classical development of natural product-based drug discovery with fungi as well as various modern options that recently became available via the –OMICS technologies. Curiously, the options to quickly improve the production and activities of natural compounds nowadays appear higher than ever before, while the lack of relevant expertise in the pharmaceutical industry steadily contributes to the innovation gap regarding novel drugs that can be brought to the market. A further increase of public funding and incentives for the industry to re-establish capacities and know-how for natural product-based drugs would be highly desirable, especially with respect to indications like anti-infectives, where the greatest need is going to arise in the future.
Current trends, limitations, and future research in fungal classification and phylogenomics
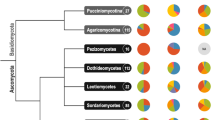
The term "phylogenomics" was introduced by Jonathan Eisen in 1998 for improving functional predictions for uncharacterized genes and to study the evolution of gene families in the genome-scale phylogenetic analysis (Eisen 1998). Compared with multi-locus phylogenetic analysis, genomes contain more species evolution information, and a larger dataset can decrease sampling errors. Reconstruction of phylogeny based on genome-scale data can more realistically reflect the evolutionary relationship between taxa (Virendra and Somnath 2009). In the last 20 years, advances in next-generation sequencing technologies and bioinformatics have made the genomic era now a reality. Phylogenomics has since become a widely used approach in evolutionary biology and genomics (Guttmacher and Collins 2003; McGuire et al. 2020; Eisen and Fraser 2003; Virendra and Somnath 2009). There has been a rapid increase of fungal genome data in public databases, such as NCBI database (https://www.ncbi.nlm.nih.gov/datasets/genome/?taxon=4751) and 1000 Fungal Genomes Project (https://mycocosm.jgi.doe.gov/mycocosm/home/1000-fungal-genomes). Researchers are therefore increasingly using genomic data to infer the evolutionary relationships among fungi, and have obtained remarkable progress in fungal taxonomy. A number of long-standing taxonomic questions have been resolved, and at the same time, the accuracy of fungal classification and the understanding the of fungal evolution have been improved greatly (Zhang et al. 2017). Pizarro et al. (2018) utilized a genome-scale dataset of 2556 single-copy protein-coding genes to reconstruct a phylogeny of the most diverse group of lichen-forming fungi. This study strongly supported the monophyly of major clades and resolved previously unresolved relationships in the family Parmeliaceae. Based on morphological characters and two-locus phylogenetic analysis, the important plant-pathogenic genus Pythium sensu lato was split into five genera: Pythium, Elongisporangium, Globisporangium, Ovatisporangium, and Pilasporangium (Uzuhashi et al. 2010). This revision however, was not generally accepted by the scientific community due to the lack of phylogenetic support until a phylogenomic study of Pythium was conducted by Nguyen et al. (2022).
In higher-level taxonomic studies, phylogenomic analysis helps in resolving several problematic lineages, including the grouping of microsporidia + Rozella with Cryptomycota (James et al. 2013), the formal phylogenetic classification of splitting zygomycete taxa into Mucoromycota and Zoopagomycota (Spatafora et al. 2016), and the systematic placement of the xerotolerant mold Wallemia as the earliest diverging group in Agaricomycotina (Padamsee et al. 2012). In addition, Li et al. (2021b) discovered that ~ 85% of fungal taxonomy ranks used in the dataset were broadly consistent with both genome sequence divergence and divergence times at higher taxonomic levels, suggesting the effectiveness of using the divergence time approach to rank taxonomic lineages.
Limitations
While phylogenomics has brought powerful analytic methods to fungal taxonomy, there are some limitations that should be considered. Current phylogenomic studies suffer from limited taxon sampling, which can lead to incomplete or inaccurate phylogenetic reconstructions. The phylogenomic study by Chen et al. (2023a) included all available genomes of Sordariomycetes from the public database. The final dataset only comprised 156 genera, 50 families, 17 orders, and five subclasses, which is much lower than the number of existing taxa (1619 genera, 184 families, 46 orders, and seven subclasses) summarised in the latest outline (Wijayawardene et al. 2022a, b). Most genomes in the public database are from pathogenic or biotechnologically useful fungi, with whole genome sequencing was conducted by non-fungal specialists for non-taxonomic purposes. This led to poor sampling in many groups, which may be underrepresented in genomic databases. Furthermore, because most genomes are not from ex-type or representative strains, there are no morphological characterizations for these genomes. This impedes the combination of phylogenomic analysis and morphological studies. In fungal species delineation, there have been several attempts to differentiate species using genome data; however, this kind of application has been hampered by the lack of clear and universal criteria (Sepúlveda et al. 2017; Gostinčar 2020; Matute and Sepúlveda 2019). The cost of whole genome sequencing has been decreasing due to advancements in sequencing technologies. However, it remains relatively more expensive as compared to Sanger sequencing, which is used in multigene phylogenetic analysis. Furthermore, whole genome sequencing instruments are not available in underdeveloped countries. The subsequent requirements of extensive computational resources, sophisticated bioinformatic skills and poor collaborations between bioinformaticians and taxonomists also limit the promotion of phylogenomics in fungal taxonomy to some extent.
Future
The main limitation of phylogenomics in fungal taxonomy is an unbalanced genomic sampling. As more fungal genomes are sequenced, including those from understudied and diverse fungal lineages, phylogenomic studies can benefit from increased genomic sampling. This will enable a more comprehensive understanding of fungal diversity and evolution, filling in gaps in the fungal tree of life and improving the resolution of phylogenetic relationships. The vast majority of fungal taxonomic groups do however, not have the ability and conditions to perform phylogenomics analysis Therefore, the use of morphology and multi-locus analysis will be the main methodologies used in fungal taxonomy. Phylogenomic approaches will be supplements to solve some taxonomic problems. The fungal taxonomy community should support and advocate for the implementation of phylogenomics into taxonomy. It is hoped that phylogenomics will provide a more comprehensive and accurate understanding of fungal diversity and evolution in the future.
Current trends, limitations, and future research in genomics and ecology of fungal plant pathogens
A brief history of the origin and evolution of fungi
Some studies have shown that the ability of fungi to colonise plants is ancient, suggesting a correlation between streptophyte algae (embryophytes and their closest green algal relatives) and the lineages of fungi Ascomycota, Basidiomycota, and Chytridiomycota around 1 billion years ago (Jones et al. 2015). The application of genome analysis and comparative studies has supported this hypothesis. For instance, the presence of pectinases and cellulases through millions of years of evolutionary history suggests that most of the ancestors of fungi evolved using plant-based nutrition (Berbee et al. 2017). Moreover, as algae evolved into land plants, and as the polysaccharides in their cell walls differentiated, fungal enzymes accompanied this evolution process (Lange et al. 2019). In this regard, fungi developed strategies to invade complex substrates, making them the principal degraders of biomass, and efficient saprotrophs and pathogens able to infect a wide range of hosts (Bucher et al. 2004; Berbee et al. 2017; Howlett et al. 2015). In natural ecosystems, the interaction between plants and their pathogens drives a co‑evolutionary dynamic (Howlett et al. 2015). However, in managed ecosystems, as crops evolve through artificial selection (e.g., selection of desired traits), pathogens need to rapidly evolve and adapt to new environmental conditions and niches leading to the increase of virulent phenotypes (Möller and Stukenbrock 2017). Consequently, the awareness of mycologists and plant pathologists to study fungal pathogens and to evaluate their evolutionary relationships based on genome data has been considerable in recent studies (Ball et al. 2020; Garcia et al. 2021; Priest et al. 2020).
What does genomics offer?
Genome analysis is pivotal to understanding the mechanisms underlying the infection processes (Schikora-Tamarit and Gabaldón 2022) and adoption patterns involved in fungal lifestyles and ecological niches (Gonçalves et al. 2022; Janusz et al. 2017). Additionally, genome data provides information on gene families and their functional potential for virulence and host infection (Ball et al. 2020). This is of paramount importance for the plant pathology sector to understand the biology of diseases, to improve diagnostic methods, and ultimately to manage and/or prevent disease outbreaks (Aylward et al. 2017; Weisberg et al. 2021). Therefore, current efforts on fungal genome sequencing have been made to cover the above-mentioned purposes by unveiling:
-
(1)
carbohydrate-active enzymes responsible for the degradation of plant cell walls, showing the ability of fungi to penetrate and colonise plant tissues.
-
(2)
biosynthetic gene clusters for the discovery of bioactive compounds with pharmaceutical, biomedicine, and agricultural applications.
-
(3)
transmembrane transporters of ions, sugars, and molecules that contribute to fungal virulence.
-
(4)
pathogenicity/virulence genes and candidate effectors that manipulate the host immune defense.
-
(5)
candidate genes involved in evolutionary processes that shape fungal pathogens' adaptation to different environments (e.g., increasing temperatures, drought, high levels of salinity).
-
(6)
the role of secreted effectors on fungal virulence and lifestyle switching.
-
(7)
genes related to morphological, physiological, and reproduction among different isolates.
-
(8)
genetic basis for multi-omics analyses to offer a complete overview on plant-pathogen interactions.
-
(9)
genomic variants linked to virulence through the analysis of single nucleotide polymorphisms (SNPs) (Constantin et al. 2021; Garcia et al. 2021; Gonçalves et al. 2022; Grandaubert et al. 2019; Xu 2020).
Current limitations on genome analysis
The important roles of fungi and their ubiquity and plurivorous nature, coupled with the advances in next-generation sequencing (NGS) have driven researchers to sequence numerous fungal genomes (Möller and Stukenbrock 2017; Priest et al. 2020). This has led to a greater availability of fungal genomes in public databases (e.g., NCBI, JGI Genome Portal) which significantly expanded our knowledge of the infection processes, fungal ecology, and genome evolution (Aylward et al. 2017; Xu 2020). However, there is a limited understanding of the impact of genome architectures on the upsurge of pathogenicity changes and the adaptation to changing environments mainly due to:
-
(1)
increasing numbers of fungal genomes published as genome announcements lacking functional gene annotations.
-
(2)
interpretation of genomes based mostly on low-quality assemblies, weak annotations, and homology-based predictions.
-
(3)
shortage of sufficient funding to obtain whole genome sequencing and to access publicly available databases and web surfaces for data analysis.
-
(4)
wrongly identified fungal species and misidentification of the submitted genomes in databases which introduce errors and deceive further analysis.
-
(5)
requirement of experts in data sciences and plant pathology for a correct and careful interpretation of data.
These limitations are challenging for researchers impairing the achievement of high-quality and large-scale comparative analysis (Gabaldón 2020; Stavrou et al. 2018).
The close relationship between bioinformaticians and basic mycology
Mycology is integrative and requires experts with diverse skills in fungal biology, bioinformatics, and molecular biology (Hibbett et al. 2013). Despite the efforts to understand the biology and ecology of fungi, mycologists and plant pathologists need to establish a close collaboration with bioinformaticians. Combining their expertise can be beneficial to deepen our knowledge of fungi and their role in biological processes and lead to several advantages (Aylward et al. 2017; Gautam et al. 2022; Roth et al. 2023; Wijayawardene et al. 2022a, b):
-
(1)
Bioinformaticians can assist mycologists in analysing the genomes of several fungal species, thus providing valuable insights into their evolution, physiology, and potential applications.
-
(2)
By analysing the gene expression (transcriptomics) and protein profiles (proteomics) of fungi, bioinformaticians can help mycologists understand how genes are regulated and the biological functions of proteins.
-
(3)
Comparing the genomes of different fungi can assist in the identification of common features and differences between species. This can shed light on the evolutionary relationships and the genetic basis of unique properties of certain fungi.
-
(4)
In environmental mycology, metagenomics allows for the analysis of all the genetic material present in a sample. In this regard, bioinformaticians can handle the vast amounts of data generated from such studies and help in the identification of different fungal species.
-
(5)
Bioinformatics tools can aid in predicting the functions of genes and proteins in fungi, even for species where experimental validation may be challenging or time-consuming.
-
(6)
Understanding the genetic factors involved in fungal pathogenicity can lead to the discovery of potential drug targets. Bioinformatic analysis can assist in identifying crucial genes and pathways related to fungal virulence.
-
(7)
Bioinformaticians can facilitate the sharing of large datasets and help establish collaborative efforts between different research groups and institutions in the field of mycology.
-
(8)
Bioinformaticians can create software tools and databases tailored specifically for mycological research, making it easier for mycologists to analyse and interpret their data.
Overall, the collaboration between bioinformaticians and basic mycologists can accelerate research, increase the accuracy of analyses, and provide a better understanding of fungi (Roth et al. 2023). As technologies and computational methods continue to advance, this partnership will play an increasingly critical role in advancing mycological research and its applications in various fields, including medicine, agriculture, and industry (Weisberg et al. 2021; Wijayawardene et al. 2022a, b).
Future perspectives in untangling genome architectures
Despite the above-mentioned challenges, further efforts should be made to continue pursuing knowledge on the mechanisms of virulence, processes of genome evolution, environmental adaptations, and speciation events (Aylward et al. 2017; Ball et al. 2020). This information can be achieved through the analysis of genome compartments that are enriched in effectors and transposable elements, responsible for genomic plasticity and the evolution of new virulence phenotypes (Möller and Stukenbrock 2017). Therefore, comparative whole genome analysis between pathogenic and non-pathogenic isolates will contribute to.
-
(1)
identifying those compartments and determining their roles in the rapid evolution of fungi.
-
(2)
monitoring the population dynamics of pathogens.
-
(3)
revealing fungal ecological adaptations.
-
(4)
understanding evolutionary dynamics (Ball et al. 2020; Grandaubert et al. 2019).
Moreover, bearing in mind that fungi can adopt different lifestyles (e.g., endophytes, pathogens, saprotrophs), genome analysis should be considered in future studies for a deep understanding of the molecular traits and mechanisms that determine lifestyle switches (Constantin et al. 2021; Hilário and Gonçalves 2023). As a single reference genome cannot reflect the genetic diversity of a species, it is also vital to increase the number of sequenced genomes of closely related and divergent isolates within a given species to assess evolutionary relationships (Gabaldón 2020; Xia et al. 2022). High-quality genomes are required to obtain more accurate and correct information from a fungal genome and its functional annotation. In this regard, it has been suggested that plant pathology programs must provide their students with more opportunities to deepen their skills in data sciences (Xu 2020). This will surely help them to gain experience and expertise in working with genomic datasets, thus improving the quality of the published data (Weisberg et al. 2021).
The integration of all these approaches provided by genomics will certainly enable new perspectives in untangling fungal-pathogen interactions, shed light on the biology, ecology, and evolution of pathogens, and ultimately in identifying diagnostic markers for agricultural settings (Hilário and Gonçalves 2023; Weisberg et al. 2021; Xia et al. 2022).
Current trends, limitations and future research in functional genomics
Functional genomics is an interdisciplinary field that integrates molecular biology and cell biology, utilizing the massive amount of data generated by genomic and transcriptomic projects. This has the aim of deciphering how genes and their regulatory elements work together to carry out biological processes and determine the functions of specific genes in various biological systems (Smit et al. 2017). These functional genomic approaches to fungi enable researchers to shed light on the molecular mechanisms underlying fungal biology, focusing on their growth, development, metabolism, and response to environmental stimuli (Chethana et al. 2020; Peng et al. 2022; Huang et al. 2023). The significance of fungal functional genomics lies in its ability to elucidate gene function, paving the way for advancements in various fields, including medicine, agriculture, and biotechnology (Huberman 2021). Furthermore, identifying these genes and proteins that are essential for critical processes like fungal survival and pathogenicity provides the knowledge required to develop targeted therapies and antifungal drugs that disrupt vital processes in pathogenic fungi impacting plants, animals, and humans, improving the treatment of fungal infections and reducing the emergence of drug resistance (Sanz et al. 2017; Segal et al. 2018; Chethana et al. 2020; Bruno et al. 2021; Peng et al. 2022; Huang et al. 2023). Moreover, functional genomics provides insights into evolutionary relationships, gene family expansions or contractions, and the acquisition of novel traits that are important to understand the processes that shaped fungal diversity and adaptation to different ecological niches (Kim et al. 2014; Liu et al. 2017; Ball et al. 2020; Garcia et al. 2021; Priest et al. 2020).
As fungi play essential roles in agriculture, both as beneficial symbionts and devastating plant pathogens (Hyde et al. 2019), functional genomics research also provides insights into fungal-plant interactions, including symbiosis, disease resistance, and nutrient uptake (Seo et al. 2015). Understanding these interactions can lead to the development of strategies for crop protection, enhancing plant health, and improving agricultural productivity (Bhardwaj et al. 2014; Yang et al. 2023). Hyde et al. (2019) discussed the immense biotechnological potential of fungi to produce a wide range of enzymes, bioactive compounds, and secondary metabolites for industrial processes or pharmaceutical compounds. This knowledge can be harnessed to optimize fungal strains for biotechnological applications and develop new bioproducts. Overall, fungal functional genomics is essential for expanding our knowledge of fungal biology, enhancing food security, boosting human health, and spurring innovation across various industries (Wijayawardene et al. 2023a, b). By uncovering the functions of fungal genes and their interactions, this field contributes to the development of new strategies, therapies, and biotechnological applications with broad societal impacts.
Current trends in fungal functional genomic studies
The advent of genomics research has revolutionized our understanding of fungi by generating an unprecedented volume of data, serving as the foundation for functional research in this diverse group of organisms. The sequencing of fungal genomes has produced enormous datasets that provide valuable insights into the genetic makeup of various fungal species (Noble and Andrianopoulos 2013; Ma et al. 2014; Yan et al. 2018; Grandaubert et al. 2019; Constantin et al. 2021; Garcia et al. 2021; Gonçalves et al. 2022; Greener et al. 2022). Integrating this massive data with other "omics'' approaches, such as transcriptomics and proteomics, offers a holistic perspective of fungal functional genomics, paving the way for groundbreaking discoveries and innovative applications in industries such as agriculture, medicine, and biotechnology (Muller et al. 2013; Abram 2015; Grandaubert et al. 2019; Chethana et al. 2020; Xu 2020; Constantin et al. 2021; Garcia et al. 2021; Gonçalves et al. 2022; Peng et al. 2022; Huang et al. 2023). In recent years, several prominent trends have emerged in fungal functional genomics. These trends encompass a range of techniques and approaches that are reshaping our understanding of fungal biology and offering new avenues for applications in various fields.
A prominent trend in functional genomic research is the integration of multi-omics datasets, such as genomics, transcriptomics, proteomics, and metabolomics, into functional research, providing a deeper understanding of the functional elements within fungal genomes and their relationships to fungal biology (Chethana et al. 2020; Presley et al. 2020; Constantin et al. 2021; Garcia et al. 2021; Gonçalves et al. 2022; Li et al. 2022a, b, c; Peng et al. 2022; Huang et al. 2023). Such research has the potential to uncover novel insights into the biology, evolution, and biotechnological potential of fungi by correlating genomic information with gene expression profiles, protein interactions, and metabolic pathways. For instance, Presley et al. (2020) integrated functional research with transcriptomics (comparative RNA-Seq) and proteomics to identify various interaction methods and the individual proteins mediating these relationships among two model brown rot fungi of softwood timber. Additionally, the integration of metabolomics and functional genomics in the study by Li et al. (2022a, b, c) elucidated the metabolic adaptations of Trichoderma reesei under cellulase-inducing circumstances, revealing key regulatory mechanisms for efficient cellulase synthesis. Furthermore, comparative genomics research combined with functional genomic research in fungi facilitates the identification of conserved genes, orthologs, regulatory elements, and lineage-specific adaptations, and provides insights into the evolutionary innovations and specialised functions that contribute to the diversity of fungal lifestyles, including pathogenicity, symbiosis, and environmental adaptation (Muller et al. 2013; Abram 2015; Yan et al. 2018; Grandaubert et al. 2019; Xu 2020; Constantin et al. 2021; Garcia et al. 2021; Gonçalves et al. 2022). For example, comparative analysis of the transcriptome and genome data of Aspergillus species identified genes responsible for carbon utilization, secondary metabolism, and stress response, laying the foundation for exploiting them for biotechnological and medical applications (Terabayashi et al. 2010; de Vries et al. 2017). Furthermore, functional studies, in conjunction with comparative analysis, revealed the evolution of the fungal chitin synthase (CHS) gene family, as well as its relationship to fungal morphogenesis and adaptability to ecological niches (Liu et al. 2017). Large-scale comparative analysis of 135 genomes of mycorrhizal species revealed their complex symbiotic traits through gene duplications and diversifications and provided evidence for convergent evolution, and identified key genetic components involved in the nutrient exchange between mycorrhizal fungi and their plant hosts (Miyauchi et al. 2020). Similar research can aid in understanding the origin and development of plant-fungal relationships and their impact on ecosystem function.
Another emerging trend in the study of pathogenic fungi is using functional genomic techniques in conjunction with transcriptomics, proteomics, and gene editing to uncover the genetic mechanisms underlying their pathogenicity and host interactions. These studies lay the groundwork for identifying virulence factors, comprehending host–pathogen interactions, and developing tailored antifungal therapies (Chethana et al. 2020; Chakraborty et al. 2021; Duan et al. 2021; Kowalski et al. 2021; Peng et al. 2022; Przybyla and Gilbert 2022; Yu et al. 2022; Huang et al. 2023). A recent study used a combination of functional genomics and solid-state NMR spectroscopy to identify key genes involved in the cell wall assembly and remodeling of the fungal cell wall architecture of Neurospora crassa, and provided new insights into the spatial arrangement and interactions of cell wall components, improving our understanding of the structural response of fungal pathogens to stresses and revealing potential targets for antifungal therapies (Chakraborty et al. 2021). Advances in genome editing tools, such as CRISPR-Cas9, have opened up new paths for functional genomics research in fungi, enabling precise modifications to fungal genomes and accelerating the advances in fungal biology and pathogenicity (Kowalski et al. 2021; Duan et al. 2021; Wang et al. 2023a, b, c). Kowalski et al. (2021) used CRISPR-Cas9 to discover the critical role of its gene CZF1 in Candida glabrata virulence and biofilm formation, highlighting its potential as a therapeutic target, while Duan et al. (2021) used a similar approach to identify the function of FgHOG1, in regulating stress responses and virulence in Fusarium graminearum. Characterizing these genes is crucial, as they can be utilized in developing green super crops that exhibit superior productivity and resilience to abiotic/biotic stresses using functional genomics and multi-omics technologies, holding great potential for addressing global food security challenges and reducing the environmental impact of agriculture (Varshney et al. 2019; Yu et al. 2022).
Limitations on functional genomic studies
Several constraints limit the progress of functional genomics research and our understanding of fungal biology and interactions. In general, fungal genomes are complex, with varied genome sizes, repeated sequences, and high levels of genetic plasticity (Noble and Andrianopoulos 2013; Möller and Stukenbrock 2017). One major challenge for conducting functional genomic research is that the functions of a significant portion of fungal genes remain unknown or hypothetical, making it challenging to interpret complex regulatory networks and pathways (Bouhired et al. 2007; Schäpe et al. 2019). While advanced genetic manipulation tools are available in many fungal species, these genetic tools and techniques are lacking in certain fungal species, hindering the efficient, targeted manipulation and functional analysis of specific genes (Huang and Cook 2022; Wang et al. 2023a, b, c). Furthermore, functional genomic research has focused primarily on well-studied model fungi (Magee et al. 2003; Lee and Dighton 2013), leaving many fungal species of ecological or industrial value with a scarcity of data. Gene and functional redundancy is often associated with fungal genomes (Noble and Andrianopoulos 2013; Herzog et al. 2020), affecting and restricting the success of functional genomics studies, as knocking out a single gene might not result in a noticeable phenotype due to the presence of redundant or compensatory mechanisms that contribute to similar or overlapping functions (El-Brolosy and Stainier 2017). Additionally, functional genomic studies often rely on phenotypic assays, which can be complex and time-consuming for characters that are difficult to quantify or visualize. These assays may not fully capture interactions in natural fungal habitats (Franco-Duarte et al. 2019), limiting their applicability to fungi. Environmental factors, developmental phases, and interactions with other organisms impact fungal gene functions (Rangel et al. 2015; Momin and Webb 2021; Lin et al. 2023a, b). A further limitation of functional genomics research is that it often focuses on specific circumstances or time points, which may not capture the entire range of fungal gene functions. Addressing and overcoming these limitations requires continuous advancements in genetic manipulation techniques, improvements in genome annotations, and the development of standardized methodologies, leading to a more thorough and complete understanding of fungal biology and interactions.
Future perspectives for functional genomic research in fungi
The future of functional genomic research in fungi holds great promise. Advancements in high-throughput sequencing technologies, including single-cell genomics and long-read sequencing integrated with multi-omics approaches, facilitate the comprehensive characterization of fungal genomes and transcriptomes, thereby providing a more holistic understanding of fungal biology (Lorrain et al. 2019; Li et al. 2021a, b; Tedersoo et al. 2021a, b; Massart et al. 2022). The future of functional research should be expanded to a broader array of fungal taxa, including non-model fungi and those with agricultural, biotechnological, economical, and ecological relevance to overcome one of the previously mentioned limitations (Swift et al. 2019). In addition, future research should delve into the genetic basis of symbiotic relationships, pathogenicity, and host-microbe interactions (Bosch et al. 2019; Fiorilli et al. 2020). Understanding the molecular mechanisms underlying these interactions will affect agriculture, human health, and ecosystem dynamics.
The widespread application of cutting-edge gene editing tools such as CRISPR-Cas9, pathway engineering, and synthetic gene circuits will be increasingly applied to functional fungal research (García-Granados et al. 2019; Xia et al. 2019; Otero-Muras and Carbonell 2021). This research will facilitate the design and engineering of fungi to produce valuable compounds, bioremediation, and other biotechnological applications (Arun et al. 2023; Ghosh et al. 2023). Further research is needed to improve these tools to achieve more efficient delivery methods, increased precision, and the ability to target specific genomic loci, thereby facilitating the identification of key genes and regulatory elements for synthetic biology applications with greater precision and scalability (Porto et al. 2020). Furthermore, rapidly advancing technologies such as single-cell genomics, transcriptomics, and automated, high-throughput phenotyping tools will be applied to fungal functional genomics research for studying cellular processes and gene expression at the single-cell level and enable understanding of the contributions of specific cell populations to fungal physiology and development (Maviane-Macia et al. 2019; Wösten 2019; Jansen et al. 2021; Seto et al. 2023). The automated, high-throughput phenotyping tools are specifically used to simultaneously investigate multiple phenotypic traits across a large fungal population, thereby accelerating research progress (Maviane-Macia et al. 2019; Jansen et al. 2021). Automated imaging, robotic systems, and machine learning algorithms assess the functions of genetic modifications and aid in identifying novel gene functions, characterizing gene networks, and discovering phenotypes associated with specific genetic variants (Shariff et al. 2010; Wainaina and Taherzadeh 2022). Moreover, future functional genomic research should increasingly be integrated into environmental metagenomics studies to investigate the gene expression patterns, metabolic adaptations, and functional traits of fungi in diverse environmental conditions, thereby contributing to our understanding of fungal ecology, ecosystem dynamics, and the impact of environmental changes on fungal communities (Gómez-Silva et al. 2019; Zhao et al. 2023).
In addition to those mentioned above, functional research integrated with multiple omics data sets (genomics, transcriptomics, proteomics, metabolomics, and epigenomics) to provide a system-level understanding of fungal biology and its responses to environment will continue to be a major trend in the future as well (Gómez-Silva et al. 2019; Swift et al. 2019; Li et al. 2021a, b; Wijayawardene et al. 2023a, b; Zhao et al. 2023). Therefore, data integration methods and computational analysis tools require advancements to successfully uncover intricate regulatory networks, metabolic pathways, and molecular interactions within fungal cells and communities (Wijayawardene et al. 2023a, b). Functional annotation tools and methods require further improvements to ensure the accuracy and reliability of gene function predictions in fungal genomes. This can be achieved by establishing improved, reliable annotation pipelines by integrating experimental data, comparative genomics, and functional assays. The future of fungal functional genomics relies on collaborative research efforts and data sharing among research communities. Therefore, establishing a centralized database, repositories, and platforms for sharing functional genomics data will be of utmost importance (Byrd et al. 2020; Schatz et al. 2022) as they can accelerate discoveries and enable the broader scientific community to access and utilize the wealth of information generated through functional genomics studies.
Current trends, limitations, and future research in fungi and the biocircular economy
Current trends
Fungi are versatile organisms. They can produce a wide range of products, from food and feed to chemicals and fuels (Lübeck and Lübeck 2022; Copetti 2019; Hyde et al. 2019; Meyer et al. 2021; Vandelook et al. 2021; Strong et al. 2022). They can also break down organic and inorganic materials and compounds, making them a potential waste management and pollution control tool (Vaksmaa et al. 2023; Dashtban et al. 2010; Harms et al. 2011; Deshmukh et al. 2016). As a result, fungi are increasingly seen as critical players in the biocircular economy, an economic system that aims to minimise waste and maximise the reuse of resources (Kirchherr et al. 2017). With their ability to grow on various organic feedstocks and be functionalised into a range of diverse material types, fungi have the potential to revolutionise the way we think about and approach sustainability.
Fungi decompose, meaning they break down organic matter into smaller molecules that other organisms can reuse (Osono 2007; Holden et al. 2013, Niego et al. 2023a, b), a vital process for the ecosystem's functioning. This ability makes them a potential tool for waste management and pollution control. Fungi can be crucial in utilising farm waste and producing fertiliser or food. Agricultural waste, such as crop straw and livestock manure, can be treated through composting or aerobic fermentation to produce organic fertiliser. This process can help reduce dependence on chemical fertilisers and improve the application rate of organic fertiliser in soil (Mengqi et al. 2021).
Additionally, agro-industrial wastes can be used as raw materials for producing biofuels, enzymes, vitamins, antioxidants, animal feed, antibiotics, and other chemicals through solid-state fermentation using fungi and other microorganisms (Sadh et al. 2018). Varma et al. (2015) showed that the decomposition rate of agricultural waste increased when inoculated with a white-rot fungus, while Aspergillus niger was efficient in producing cellulases and agro-waste materials (Jasani et al. 2016). Inoculating Trametes versicolor and Fomes fomentarius on the compost of an organic fraction of municipal solid waste led to a higher degrading ratio and increase of enzymatic activities (Voběrková et al. 2017).
Fungi can produce high-value food and feed products like mushrooms, Quorn, and tempeh (Amara and El-Baky 2023). These products are often considered more sustainable than traditional animal-based products, requiring less land and water to produce (Boland et al. 2013). Some species or isolates can synthesise chemicals like enzymes and antibiotics (e.g., Jakubczyk and Dussart 2020; Sanchez and Demain 2017; Khan et al. 2014; Conrado et al. 2022; De Silva et al. 2012, 2013). These products can be used in different industries, including the food, pharmaceutical, and energy sectors. Others can produce several materials, such as biopolymers, biocomposites, and mycelium-based foams, that can be used in various applications, including packaging, construction, and medical devices (e.g., Sydor et al. 2022; Yang et al. 2021; Manan et al. 2021; Alemu et al. 2022; Vandelook et al. 2021; Bitting et al. 2022; Biala and Ostermann 2022).
Fungal biotechnology also produces biomaterials that can replace petroleum-based ones in various industries, including food, packaging, textile, leather, and automotive (Cerimi et al. 2019; Raman et al. 2022; Meyer et al. 2020). For example, the production of lipase by strains of Aspergillus niger, used in several industries, including food, pharmaceutical, and cosmetics, has been studied and optimised (Colla et al. 2016; Alabdalall et al. 2020). A new and more efficient bioprocess has been developed to produce bioethanol from agricultural waste (starch of avocado seeds) by using a natural strain of Saccharomyces cerevisiae) (Caballero-Sanchez et al. 2023). A new mycelium-based biodegradable and recyclable foam that could be used in many applications, such as packaging and insulation, has been developed (Gandia et al. 2021; Karana et al. 2018; Yang et al. 2009). This could lead to the construction of healthier buildings made of components that are grown instead of manufactured and can be triggered to biodegrade at the end of their life.
Fungi, especially mycelium, are increasingly recognized for their potential in carbon sequestration and as a source of bio-based materials, keeping climate-warming carbon dioxide out of the atmosphere. Indeed, using biodegradable building materials can contribute significantly to carbon sequestration. Bio-based materials integrate various mitigation techniques, including low embodied energy and carbon, affordability, recyclability, utilisation of locally sourced materials, and the ability to repurpose waste and byproducts (Alemu et al. 2022). Biological materials offer indirect benefits in reducing organic waste. This is because the raw materials used to produce microbial-based materials are often locally available organic wastes. This promotes recycling and waste reduction, contributes to local economies, and reduces the carbon footprint associated with transporting materials. According to Kumarappan et al. (2018), using biological materials in construction could reduce carbon emissions by nearly 800 million tons annually.
Fungi have also been found to break down contaminants such as polyaromatic hydrocarbons, heavy metals, herbicides, pesticides, cyanotoxins, pharmaceuticals, antibiotics, phthalates, dyes, and detergents (Akhtara and Mannana 2020). In this process, fungi produce enzymes that can break down specific types of waste. For example, fungi can produce enzymes that can break down plastics (e.g., Andler and Goddard 2018; Okal et al. 2023; Ekanayaka et al. 2022; Temporiti et al. 2022; Ren et al. 2021), wood, and other materials (e.g., Orth et al. 1993; Hammel et al. 1997; Azelee et al. 2020; Goodell et al. 2020; Beltrán-Flores et al. 2022; Geethanjali et al. 2020). These enzymes can then treat waste materials in various settings, such as industrial wastewater treatment plants and landfills, and grow them directly on the substrate. In this way, fungi can also be used to produce biogas from different organic materials, including food waste, agricultural waste, and sewage sludge. They can break down these materials and produce methane, the main component of biogas (Dollhofer et al. 2015; Kazda et al. 2014; Kovács et al. 2022). In addition to their natural ability to break down waste materials, scientists are also developing new biotechnological solutions to harness the power of fungi for waste disposal and ecosystem restoration (Kulshreshtha et al. 2014; Akhtar and Mannan 2020; Akpasi et al. 2023).
Limitations
Even though fungi have great potential and diverse applications, several downsides must be addressed. One of the critical areas of research in fungi and the biocircular economy is the development of new fungal strains and bioprocesses to produce high-value products (Tiwari and Dufossé 2023). The development of even more sustainable and cost-effective ways to make these products, will significantly impact the biocircular economy. Developing new fungal strains and bioprocesses for producing high-value products is however, complex and challenging. For example, there is a need for further research and development to optimise the use of fungi as sources for novel compounds and as cell factories for the large-scale manufacture of bio-based products (Lübeck and Lübeck 2022; Meyer et al. 2016; Vandelook et al. 2021).
Additionally, controlling fungal pathogenicity is a significant challenge that needs to be addressed to improve the efficiency and safety of using fungi in biotechnology (Meyer et al. 2016). Also, their slow growth rate, the intrinsic characteristics of a species and the ecological conditions it is exposed to (Gostinčar et al. 2022), and sensitivity to environmental conditions (Bakar et al. 2020) are constraints to their utilisation on the industrial scale. Research should address these limitations by developing or selecting faster-growing and more efficient fungal strains, optimising bioprocesses for producing high-value products from fungi and developing new methods for their use.
There is also the potential for fungi to produce harmful metabolites such as toxins (Bennett and Klich 2003). This includes mycotoxins such as aflatoxins, ochratoxins, and trichothecenes that can cause a variety of health problems, including food poisoning, liver damage, and cancer (World Health Organization 2023; Bennett and Klich 2003; Omotayo et al. 2019; El-Sayed et al. 2022), alkaloids such as psilocybin, psilocin, and ergot alkaloids that can have a variety of effects on humans, including hallucinogenic, stimulant, and sedative effects (Plazas and Faraone 2023); Polyketides including penicillin, griseofulvin, and lovastatin that can act antibiotic, antifungal, and anti-inflammatory effects on humans (Prescott et al. 2023; Conrado et al. 2022). However, several steps can be taken to mitigate this risk of exposure to harmful metabolites produced by fungi, such as growing fungi in controlled environments, testing fungi for the presence of harmful metabolites, and processing fungi in ways that can inactivate toxic metabolites.
Furthermore, while mycelium-based materials have excellent properties and ecological advantages, challenges remain to overcome. One of the primary challenges is their variable structural strength depending on the substrate, strain, incubation time, and fabrication process (Alemu et al. 2022), which means they cannot support much weight and restricts their use in specific applications, making it challenging to produce consistent, high-quality materials. While the raw materials for growing mycelium are inexpensive, the initial costs associated with mass production and distribution, typical of industrial fabrication, are high. Furthermore, mycelium, like any living organism, can be unpredictable, which could lead to inconsistencies in the final product.
Another challenge is scaling up the production of mycelium biomaterials to a level that can significantly contribute to global carbon sequestration. Also, the longevity of mycelium materials in different environments is not fully understood, and if these materials degrade quickly, they may release stored carbon back into the atmosphere. It becomes evident that the lack of established standards or regulations for using these materials in many applications, could slow their adoption. Besides, as with any new technology, public perception and acceptance will play a role in the widespread adoption of mycelium materials. These challenges underscore the need for further research and development in this field.
Future research
Fungi play a significant role in the biocircular economy by providing sustainable alternatives to traditional products and materials. Despite many challenges, fungal biotechnology has a growing trend towards producing fungal-based biomaterials that can contribute to a more sustainable and circular economy (Meyer et al. 2016; Kržišnik et al. 2023; Delvendahl et al. 2023; Wikandari et al. 2022). However, some challenges and limitations must be addressed, such as the slow growth rate of some fungi, their sensitivity to environmental conditions, and the potential for some fungi to produce harmful metabolites.
Future research in fungi and the biocircular economy will likely focus on overcoming these limitations and developing new and innovative ways to use fungi to create a more sustainable future. Developing new methods to drive more sustainable ways to deal with waste materials using fungi is a crucial area of research in the biocircular economy. This may include developing more robust fungal strains, optimising bioprocesses for producing high-value products and developing new methods for degrading and recycling waste materials using fungi. Developing new fungi-based materials with improved properties, studying the potential of fungi to produce harmful metabolites, and developing strategies to mitigate this risk are also important. By addressing the challenges and unlocking the full potential of fungi, we can create new opportunities to develop bio-based products.
Several key factors underscore the necessity for research on mycelium-based composites. Firstly, A deeper understanding of the properties of mycelium materials and how to manipulate them is required. Secondly, research can aid in devising more efficient and cost-effective production techniques. Thirdly, the absence of established standards or regulations for using mycelium materials in many applications necessitates research to help formulate these standards and regulations, thereby facilitating the adoption of these materials. Furthermore, mycelium-based materials hold potential in various applications, from construction to carbon sequestration. Research can assist in exploring these applications and optimising the materials for each use. Lastly, more research is imperative to comprehend the environmental impact of mycelium materials, such as their longevity and potential for carbon sequestration. This research needs to underscore the potential of mycelium-based composites and emphasize the importance of continued investigation in this field.
Finally, selecting highly promising strains is also an active area of investigation. Researchers are working on large-scale phenotyping of fungal strains to evaluate their potential for degrading non-natural, industrial compounds (Navarro et al. 2021). Additionally, using innovative approaches, such as genetic engineering for enzymes, fuels, and chemicals from lignocellulose biomass is important (Madhavan et al. 2022). These efforts aim to unlock the full potential of fungi as sources for novel compounds and as cell factories for the large-scale manufacture of bio-based products.
Current trends, limitations, and future research in the quest for species numbers
The number of fungal species is a hotly debated topic among mycologists (Hawksworth et al. 1991; Hawksworth and Lücking 2017; Hyde et al. 2020a, b). The estimated number is currently higher than the known species (Phukhamsakda et al. 2022). A large number of novel taxa have been introduced in recent years (Fig. 4). In this section, we discuss current trends in introducing and estimating species numbers. How do these methods and approaches result in novel species. Do the current papers dealing with the topic, overestimate the species number? We address future direction needs in the quest for the species number?
Number of new species introduced from 2000 to 2023. (Numbers extracted from Index Fungorum.org (https://www.indexfungorum.org/names/Names.asp)
Current trends
Novel fungi, are they an untapped potential?
Introducing novel species is common practice, but what is the point? Besides the need to understand fungal diversity worldwide, taxa also have a huge potential in biotechnology (Sandargo et al. 2019; Hyde et al. 2020a, b; Niego et al. 2023a, b) and therefore it is essential to search for and describe novel taxa. Figure 5 illustrates the number of novel species introduced in the last 22 years. Although there have been yearly fluctuations, since the turn of the century, the number of new species that have been described has increased from 1264 (in 2000) to 2734 (in 2022), peaking at 3216 in 2020. The figures for 2021 and 2022 were slightly lower (2772, 2734), probably due to the restrictions due to Covid-19. Similarly, the number of new genera described has increased from 98 in 2000, tripling to 319 in 2020, with a peak of 340 in 2015. Again, figures for 2021 and 2022 were lower (206, 233).
Number of new genera introduced from 2000 to 2023. (Numbers extracted from Index Fungorum.org (https://www.indexfungorum.org/names/Names.asp)
The increase in the number of novel fungi described yearly might be as a result of expanding collection sites or increased studies in certain countries (e.g., China, Thailand) and advancement of DNA techniques which have become standard practice (Cheek et al. 2020). Based on the ratio of plants to fungi, Hawksworth (1991) estimated there could be around 1.5 million fungal species, of which only about 10% had been described. However, based on high-throughput sequencing (HTS), it is estimated that the number of fungal species could be as high as 11.7 to 13.2 million (Blackwell 2011; Baldrian et al. 2021). However, HTS results in a large number of unidentified taxa and technically compromised sequences (Nilsson et al. 2014), thus estimations based on HTS might be an overestimation.
In 2022, a study series was published to estimate the global numbers of fungi. In this series, Wijayawardene et al. (2022a, b) reported that around 30,000 anamorphic (or asexual) species belonging to 3800 genera have been described, while Senanayake et al. (2022) estimated 1.37 to 2.56 million teleomorphic (sexual) species of which 83,000 have been described. The number of basidiomycetes species is estimated to be between 1.4 and 4.2 million (He et al. 2022), whereas single-celled yeast species are estimated to be 20,000 (Boekhout et al. 2021). New yeast species have been introduced at a very rapid rate, particularly due to an extensive research contribution from Asia, in which China has yielded many taxonomic novelties (Boekhout et al. 2021). Several studies have focused on estimating the number of species based on ecological niches. Mora et al. (2011) predicted that there could be over 50,000 (0.005 million) marine species. On the other hand, Sarma (2019) showed that the number of marine species increased from 530 in 2009 to 1112 species by 2015. Jones et al. (2019) listed 1257 marine species, which included 943 ascomycetes. Thus, the number of species of from marine environments is increasing gradually. However, we are far behind in ocean exploration as compared to that in terrestrial habitats. Research on freshwater fungi has also greatly increased and numerous new species have been described, particularly from China and Thailand (Calabon et al. 2023; Yang et al. 2023). Dong et al. (2020) introduced nine new genera of freshwater Dothideomycetes and 33 new species, whereas Luo et al. (2019) introduced two new families, three new genera, and 47 new species belonging to Sordariomycetes.
Hyde et al. (2020a, b) considered that the newly introduced species curve had not reached asymptote, based on studies in Asia. Mycology was well-established in Australia, Europe, New Zealand, South Africa and the USA, whereas in the past two decades it has been increasing in Asia and South America (Hyde et al. 2020a, b). The earliest studies on fungal taxonomy were by European mycologists (Wijesinghe et al. 2023). However, based on the State of the World’s Plants and Fungi (Royal Botanic Gardens (Kew; Antonelli et al. 2020), the proportion of fungi introduced since 2019 from Europe is lower (23%) as compared to the Asia (41%).
The current introduction of novel species and genera introduced from 2020 to 2022 is likely to continue to increase due to several factors. (1) There are no longer travel restrictions and forays will become commonplace. (2) Annually published collections such as in the Fungal diversity notes (Boonmee et al. 2021; Jayawardena et al. 2022), Fungal planet description sheets (Crous et al. 2022; Tan et al. 2022) and Mycosphere notes (Thambugala et al. 2018; Manawasinghe et al. 2022) which encourage collections of novel taxa and have higher citation scores as compared to the average of other papers in mycology. Some open-access journals are also encouraging papers comprising novel species with designated special issues as this is beneficial for improving journal citation indexes.
What is a fungal species?
In order to estimate fungal numbers, it is crucial to understand what a species is. The definition of a genus and species has received much attention (Liu et al. 2016; Chethana et al. 2021; Lücking et al. 2021). Publications have focused on providing recommendations to introduce new species. Aime et al. (2021) provide a basis to accurately introduce a new species or a name. Chethana et al. (2021) provided overall recommendations for introducing new species. Recommendations for establishing species for specific groups of fungi include namely lower fungi (Voigt et al. 2021), ascomycetes (Maharachchimbura et al. 2021), plant pathogens (Jayawardene et al. 2021; Manawasinghe et al. 2021), and yeasts (Boekhout et al. 2022). In all cases, a polyphasic approach has been recommended, which includes the combination of morphology, phylogeny, ecology, chemistry, and any other useful evidence. It is not clear that these recommendations have been followed in recent publications. Introducing new taxa based on only one strain, only sequence variations, missing required gene regions, invalid species names, and poor taxon sampling (Kularathnage et al. 2023; Tang et al. 2023; Yasanthika et al. 2023; Zhu et al. 2023) are some examples.
With the increased availability of molecular data, the phylogenetic species concept has resulted in a number species becoming cryptic such as in pestalotiod genera (Maharachchikumbura et al. 2014a, b), Diaporthe (Udayanga et al. 2014) and Colletotrichum (Jayawardena et al. 2022). When introducing new species, it is essential to establish a complete sampling of representative taxon in the phylogenetic analysis. For example, the Diaporthe eres species complex is highly diverse and D. eres is polyphyletic (Udayanga et al. 2014). Therefore, it is necessary to add additional strains as well as the type species when introducing a new species in this complex. Diaporthe rosicola (Wanasinghe et al. 2018) and D. mahothocarpi (as mahothocarpus in Gao et al. 2015) were introduced without adding adequate strains. A combined multigene (ITS, tub2, calmodulin (cal), and tef1) analysis, D. rosicola and D. mahothocarpus clustered together with D. eres with an increased number of strains D. eres (Manawasinghe et al. 2019) suggesting that both D. rosicola and D. mahothocarpus are genotypes of D. eres.
Fungi are one of the most diverse groups of organisms, and therefore it is necessary to take into account population biology when introducing novel taxa. Inter-species diversity is based on the host or geographical regions. Bhunjun et al. (2022) predicted that specious genera could contain more species than expected. The limiting factor in defining a species in a specious genus however, depends on the ability to determine the amount of variation that can be accepted in a single species. For example, Colletotrichum has 14 species complexes and 248 accepted species, of which C. gloeosporioides is one of the most diverse and widely distributed species (Jayawardena et al. 2022). Colletotrichum gloeosporioides has 12% and 88% genetic differentiation between and within host populations, respectively, based on the genetic diversity of the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) region (Liu et al. 2023). Therefore, to define a new species which is closely related to C. gloeosporioides, we assume that it should exceed this genetic variation. Therefore, within-species diversity that must be concisely dealt with for most of the asexual and specious genera. In the present era of the phylogenetic species concept, morphology tends to have become less important. Most novel introductions of species provide excellent descriptions. However, environmental factors may affect characters such as spore length and width. Thus, describing a novel species using a single strain may be questionable. Therefore, when introducing a new species, it is recommended to use more than one strain (Manawasinghe et al. 2021). The use of the single strain has the risk of introducing a new genotype with sequencing errors. There are numerous examples of this. Pseudopithomyces kunmingensis was introduced by Karun and Hyde [as 'kunmingnensis'], in Hyde et al. (2017) using a single strain. Abeywickrama et al. (2023) found no significant nucleotide differences in GAPDH and tef1-α sequence data between P. chartarum and P. kunmingensis. Thus, they synonymised Pseudopithomyces kunmingensis under P. chartarum, based on morphological similarities and phylogenetic analysis. Zhang et al. (2021a, b) synonymised recently published Botryosphaeria species; B. auasmontanum, B. minutispermatia, B. quercus, B. sinensis, B. wangensis and B. qinlingensis under B. dothidea based on sequence similarities where all these species share 99% or over similarities with the type sequence of B. dothidea. Zhang et al. (2023a, b) reduced C. pandanicola (Tibpromma et al. 2018) to C. siamense as there are only few variations in four gene regions (ITS four bp, gapdh six bp, chs-1 four bp, act two bp, and tub2 one bp).
There is no universal method to characterise a new species and species concepts and approaches in mycology will always be subjective. However, it is necessary to standarise sequence-based novel species introduction. Otherwise rather than describing distinct novel taxa we may separate a single taxon into several species.
Limitations
There are many under-studied geographic regions (e.g., Africa, South America) and more research effort is needed in these areas (Antonelli et al. 2020). Contributions to the fungi in North America and Europe have, however, decreased in recent years as compared to those in Asia (Hyde et al. 2020a, b). One possible reason for this could be the lack resources allocated for basic mycological research (Rambold et al. 2013). African rainforests have a huge plant biodiversity, which is subjected to climate change, forest expansion, deforestation and human interactions (Mahli et al. 2013) and therefore the study of their fungi is important before they become extinct.
The multifarious regulatory measures adopted by individual nations greatly restrict access and exploration of biological hotspots for those interested in sampling and studying their fungal biodiversity. Ironically, most such biodiversity hotspots are based in tropical and subtropical regions (Kumar et al. 2017; Deharveng and Bedos 2019) and fall within the category of developing countries. As the name suggests, these countries have limited financial and physical resources. Therefore, the economic policies of these developing nations cannot afford biodiversity explorations. This situation gets more complex when these countries are signatory to the Convention on Biological Diversity (CBD), Access and Benefit-sharing (ABS) rules and other bureaucratic barriers preventing joint research or international collaborations. One example is the status of mycology in Sri Lanka. Even though mycology in Asia has a rapid growth, Sri Lanka, with its most prominent biodiversity, lacks sufficient studies due to limited resources and stringent biodiversity policies (Wijayawardene et al. 2023a, b).
Despite being poorly studied ecological niches, studies of fungi in terrestrial habitats have been greater as compared to aquatic and marine habitats (Jones et al. 2019). The fungi associated with freshwater streams, mangroves, karst regions, and rocks are some habitats that have recently been explored (Osorio et al. 2017; Norphanphoun et al. 2019; Calabon et al. 2023; Yang et al. 2023). Mangrove fungi represent the second-largest ecological group of marine fungi (Deshmukh et al. 2018). Several novel taxa were introduced in early studies on marine fungi (Hyde and Jones 1988; Hyde et al. 1992). The majority of these taxa lacked sequence data, and need to be recollected, sequenced and published as new collections. Studies on rock fungi are less common, despite their diversity, probably because these species being slow-growing, poor competitors, widespread in nature, and occurring commonly (Coleine et al. 2021). Entomopathogenic fungi are well-researched due to their potential as biocontrol agents, yet compared to phytopathogenic fungi, the introduction of novel species associated with ants and arthropods is comparatively low. A recent study by Zhang et al. (2023a, b) introduced five novel nematode-trapping fungi associated with soil and freshwater sediment samples. Thus, it reflects expanding studies on fungi associated with arthropods, nematodes and other animals that have a high potential to result in novel taxa.
Fungal lifestyles are diverse and have been discussed in detail. Yet, several lifestyles have been either poorly studied or overlooked. Endophytes are one of the most studied fungal lifestyles (Purahong and Hyde 2011) yet remain far behind in numbers of new taxa because not many species were sequenced. Concurrently, the HTS approach presupposes very high fungal numbers within the same community (Dissanayake et al. 2018; Jayawardena et al. 2018; Abeywickrama et al. 2023). It has been estimated that number of endophytes be approximately one million (Sun and Guo 2012) however, current isolation techniques and the inability to culture them on artificial media have become limiting factors to finding new species.
Future research needs
Fungi have an amazing potential in resolving future global needs such as renewable energy, human nutrition, health, and sustainable agricultural. Fundamental research in mycology is a separate discipline to zoology and botany (Christensen 1989; Rambold et al. 2013) and should be treated as such (Rambold et al. 2013). Though fungi play a crucial role in ecosystem stability, the involvement of mycologists in ecological and diversity studies is less when compared to botanists and zoologists (Schmit and Mueller 2007; Rambold et al. 2013). This is the case even when dealing with topical studies such as climate change (Tibpromma et al. 2021). Fungi need to be appreciated as a key component in ecosystem stability, and their contribution should never be underestimated. Simultaneously mycologists need to diversify and integrate their research into subdisciplines, such as biotechnology, biochemistry, ecology, genetics, natural products and nutritional science, and pathology.
Over the last two decades, DNA sequencing, high-throughput technology and molecular methods have advanced significantly (Nilsson et al. 2019a, b; Mapook et al. 2022). It is important to verify species in genera such as Colletotrichum, Diaporthe, Fusarium and Pestalotiopsis as they an important phytopathogens and may also produce a number of metabolites (Manawasinghe et al. 2019; Matio Kemkuignou et al. 2022).
The use of polyphasic approaches and other techniques to justify the introduction of novel species has been stipulated (Chethana et al. 2021). However, in a situation where the answer to ‘What is a fungal species?’ changes constantly, introducing a new species will continue to be a challenge. At present, most studies rely on the phylogenetic species concept combined with morphology. Poor taxon sampling and biased referencing must be avoided. However, taxonomy is subjective and open to critical evaluations.
The quest for fungal numbers should be expanded to understudied regions and habitats including Africa, Australia, South America, and some Asian countries. Furthermore, a concerted effort must be in place for future collections to adequately cover all the underexplored niches like forest ecosystems, deep sea, marine, and extreme conditions, including caves, hot springs, and rock-associated fungi. The necessity to explore fungal species from extreme environments is important because that is where the potential lies for the development of new secondary metabolites due to the adaptive nature of fungi to extreme environmental conditions (Gostinčar et al. 2022; Yu et al. 2023).
Fungal species number estimations require further research (Hyde et al. 2020a, b). Even though the actual number of species might be lower or higher than the present estimates, it is obvious that there are a numerous understudied and unexplored regions with hidden mycota. Without a doubt, these novel species will contribute to the development of agriculture, industrial biotechnology, medicine, and disease management.
Current trends, limitations, and future research in machine learning in mycology
As a subfield of artificial intelligence, machine learning is normally categorised into two primary types: unsupervised and supervised learning (Mahesh 2020). Unsupervised learning algorithms recognize hidden patterns, structures, or relationships within the given observations where the input data does not have corresponding output labels or target values. K-means clustering and principal component analysis are the most typical unsupervised learning methods to find elusive patterns in high-dimensionality datasets (Ding and He 2004). In some sense, phylogenetic trees can be treated as outcomes of unsupervised algorithms that the algorithm does not have prior knowledge of the tree topology or relationships between the sequences. Instead, it seeks to find the most likely tree that best explains the observed sequence data without relying on predefined classifications. While supervised learning algorithms rely on well-constructed training datasets with input data and the corresponding outputs, develop a predicting model by learning the hidden associated patterns, and use the model to predict the outputs of new input instances. In the era of data explosion, a large number of biological data are being generated by advanced analytic instruments, and machine learning algorithms have been a widely used analytical method to extract information and reveal hidden patterns from overwhelming amounts of data or complex data (Ma et al. 2014; Greener et al. 2022). Some image-recognition-based fungal classification and disease diagnosis models have been developed for fungi to identify fungal species or detect specific morphological features. Zahan et al. (2021) developed a deep learning-based approach to classify the edible, inedible, and poisonous mushroom types. Also, they developed a model to detect mushroom diseases (Zahan et al. 2022). Mikhail et al. (Genaev et al. 2020) developed a model to recognize wheat rust diseases and can differentiate between leaf rust and stem rust. In genomics, some machine learning algorithms were used in well-known programs, such as AUGUSTUS, to de novo annotate gene models in new sequenced fungal and other eukaryotic genomes, and effectorP to predict fungal effector protein candidates from entire secreted protein datasets (Sperschneider and Dodds 2022). Machine learning algorithms are being used to predict the ecological roles and functional traits of fungi based on genomic traits (Fijarczyk et al. 2022; Chen et al. 2023b), which can help in the early identification of devastating fungal species with potential high pathogenicity to humans or plants. There have been some applications of machine learning algorithms in mining secondary metabolites (Aghdam and Brown 2021; Skinnider et al. 2016, 2020).
Limitations
In mycology, the limited availability of comprehensive and reliable datasets, particularly for rare or understudied fungi, can be challenging. Machine learning models by image recognition to identify fungal species have been developed only for economic macrofungi, whereas researchers cannot obtain enough morphological images for training the predictive model for most wild macrofungi. In the study of microfungi, it is not easy to obtain the image data because we cannot directly observe the useful diagnostic characteristics without the assistance of optical microscopes. Highly similar characteristics in the morphology of some plant pathogens make image recognition useless in identifying microfungi. Machine learning algorithms, especially deep learning, often act as "black boxes" with limited interpretability (Azodi et al. 2020). However, mycologists need to understand the specific biological mechanisms and identification processes.
Future
For image recognition in macrofungi, a shared database of image data checked by taxonomists should be developed for obtaining more diverse and reliable morphological images of some rare fungi species. Integration of multi-omics data, such as genomic, transcriptomic, proteomic, and metabolomic data, can provide a more comprehensive understanding of fungal biology. Machine learning models can uncover the hidden association between the genomic traits and the capacity of producing beneficial secondary metabolites, which avoids duplicated screening and expands screening populations in genome mining, helping to quickly target the fungal strains that are candidates for producing candidate drugs (Fungal genomes scoured for drugs 2018). Collaborations between mycologists and data scientists and the availability of high-reliable data and advanced computational techniques will drive further progress in this field.
Current trends, limitations, and future research in detecting plant pathogens
Plant pathology is the field of study of plant diseases and disease-causing agents, across a diverse range of natural and man-made environments. They cause substantial yield losses in several economically important crops, resulting in economic and social adversity (Venbrux et al. 2023). Plant pathogens can be microfungi, macrofungi, and fungus-like organisms (Hyde et al. 2014a, b; Jayawardena et al. 2019). Human practices such as monoculture farming and global trade affect the spread of plant pathogens and the emergence/reemergence of diseases (Gomdola et al. 2022). Accurate estimates of disease incidence, severity, and the negative effects of the disease on quality are very important. Hence, the early detection and identification of pathogens are important to reduce the associated agricultural and social losses. These pathogens have been identified and are based mainly on morphology and molecular analyses (Senanayake et al. 2020; Dissanayake et al. 2020). Several techniques are available to detect plant pathogens including culture-based, PCR-based, sequencing-based, and immunology-based techniques. The evolutionary processes of these pathogenic fungi were often fuelled by genetic variation in pathogen populations (Manawasinghe et al. 2021). Based on multi-loci analyses, a better understanding of pathogenic fungi has been obtained. Many cryptic species, in genera such as Colletotrichum, and Diaporthe were identified (Talhinas and Baroncelli 2021; Norphanphoun et al. 2022).
Non-invasive optical and spectral detection methods such as imaging efficiently detect plant diseases (Venbrux et al. 2023). It has been shown that stressed or diseased plants produce a different spectral signature compared to that of healthy plants (Zubler and Yoon 2020). According to Singh et al. (2021), spectral analysis can be applied at different scales, ranging from taking high-resolution images from a single leaf to an entire field. In large cultivation areas, imaging can help to detect a ‘hot spot’ that is experiencing biotic stress. Fusarium virguliforme, the causative agent of sudden death syndrome in soybean can be detected via satellite imaging (Raza et al. 2020). This technique provides real-time detection and detects biotic stress without any sample collection.
Cultivation-based methods are used to isolate and grow the pathogen on a selective or semi-selective medium (Senanayake et al. 2020). Isolates obtained via this method need to be confirmed by morphological, molecular, and biochemical assays (Ferone et al. 2020). Different types of PCR-based diagnostic methods have been used to identify fungal phytopathogens (Table 1). For a better resolution of the pathogens, analytical profile index, microplates, matrix-assisted laser desorption/ionisation, and fatty acid profiling (Chen et al. 2020; Ferone et al. 2020). Fatty acid profile analysis allows taxonomic identification of the pathogen up to the species level and is cost-effective as well as a rapid method in species identification (Lacey et al. 2021). In taxonomic identification, DNA barcoding also plays an important role (Hyde et al. 2014a, b; Chethana et al. 2021). In recent years various molecular techniques such as Amplified Fragment Length Polymorphism (AFLP), Inter Simple Sequence Repeats (ISSR), Random Amplified Polymorphic DNAs (RAPD), Simple Sequence Repeats (SSR), Sequence-Related Amplified Polymorphism (SRAP), and Inter Primer Binding Site (iPBS) amplification techniques have been successfully used in plant pathogen identification (Cannon et al. 2012; Longya et al. 2020). Genealogical Concordance Phylogenetic Species Recognition is used to differentiate species that lack distinguished morphological characteristics when enough independent samples per lineage are provided (Jayawardena et al. 2021a, b). Bhunjun et al. (2020; 2021) highlighted the importance of using different molecular approaches such as Automatic Barcode Gap Discovery (ABGD), General mixed Yule‑coalescent, and Objective clustering in plant pathogen identification. Fusarium head blight caused by Fusarium graminearum can be identified by a CRISPR-Cas12a-based dual recognition technique developed to detect this pathogen as low as 1 fg/μL of total DNA (). For the detection of Botrytis cinerea and Didymella bryoniae, two DNA targets were used in the development of a reusable microfluidic bioassay. Thermal denaturation of DNA was then performed to regenerate the oligonucleotide sequence and a quick assessment of multiple target detection using the laser-induced fluorescence detection technique was done in less than 10 min (Qu et al. 2017). Loop-mediated isothermal amplification (LAMP) assays have been used recently in plant pathogen detection. A quick, sensitive, and focused LAMP-based test has been used to detect Sclerotinia sclerotiorum (Grabicoski et al. 2020). Wang et al. (2020) mentioned that highly specific LAMP primers for Fusarium proliferatum are appropriate for the TEF-1 region. The use of LAMP assay for quick detection of F. proliferatum causing ear and kernel rot in maize was also described by Wang et al. (2020). Karimi et al. (2019) described the detection of Colletotrichum nymphaeae infection in asymptomatic strawberry plants based on LAMP assays.
To speed up the identification of plant pathogens and allow their identification in the field, a number of serological methods have been developed, mainly based on the enzyme-linked immunosorbent assays (ELISA), which can be considered an immunological method (Luchi et al. 2020). Different immunoassay methods have been used to visualise the binding of a specific antibody to its related antigen (Miller and Martin 1988). As ELISA needs a laboratory facility, a simple paper-based dip-stick assay, namely lateral flow devices (LFD) to detect the presence or absence of a target analyte in a liquid sample was developed. This method is widespread as it allows rapid in-field detection of plant pathogens in a few minutes (Boonham et al. 2014; Tomlinson et al. 2010). Quantum dots offer a unique optical property known as fluorescence resonance energy transfer between two reactive molecules (Sanzari et al. 2019). This property is exploited by QT-FRET immunoassays for visual identification of pathogen infections such as Aspergillus amstelodami (Safarpour et al. 2012).
Digital agriculture is an innovative approach to farming that uses modern technologies to enhance agricultural practices. High-resolution remote sensing and data analytics are being utilised to monitor and predict disease outbreaks. This digital agriculture technology allows for timely and targeted interventions, reducing the need for blanket applications of pesticides. Biosensors comprise devices that consist of a biorecognition element combined with a physicochemical transducer that generates a measurable signal upon the binding of the target analyte with the biorecognition element (Bridle and Desmulliez 2021). Biosensors are promising tools for point-of-care applications, as they are generally low-cost, easy to use, and can provide fast results (Bridle and Desmulliez 2021).
Studies of the population genetics of fungi and fungi-like pathogens are essential to identify and clarify the disease epidemiology as well as to devise management strategies (Atallah and Subbarao 2012). Genome sequencing of fungal pathogens has provided information on extensive variation in genome structure and composition between species, especially between individuals of the same species (Eschenbrenner et al. 2020). Hundreds of fungal pathogen genomes are now available and analysing these genomes provide information about the genes that are responsible for causing the diseases (Plissonneau et al. 2017). A study done by Zhang et al. (2023a, b) revealed that the comparative genome of Colletotrichum species from different lineages revealed that expanded gene families encoding CAZymes are thought to be one of the likely explanations for the widespread and polyphagous nature of species in the C. acutatum, C. boninense and C. gloeosporioides species complexes. Liu et al. (2022) used 94 Colletotrichum species based on 1893 single-copy orthologous genes to provide the most comprehensive genome tree. Datasets from population genomics built on NGS can be used to identify variations including single nucleotide polymorphisms (SNPs), insertions and deletions (INDELS), and structural variations (Potgieter et al. 2020). Next-Generation Sequencing (NGS) is another popular current trend in the identification of phytopathogenic fungi. Calonectria pseudonaviculata was detected by using NGS (Malapi-Wight et al. 2016). Puccinia striiformis f. sp. tritici is the causative agent of the wheat yellow stripe. In order to identify the population of this emerging pathogen field pathogenomics was done using RNA-Seq-based NGS of pathogen-infested wheat leaves (Hubbard et al. 2015). It was shown that there is a dramatic shift in the pathogenic population in the UK, probably due to an introduction of a different set of emerging and exotic pathogen lineages. Molecular diagnostics for the cucurbit downy mildew pathogen Pseudoperonospora cubensis, was conducted via RNA and DNA-based NGS approaches. Based on the comparative genomic analyses based on RNA-Seq of closely related species of Pseudoperonospora humuli, seven specific regions were identified in P. cubensis (Withers et al. 2016).
A hybrid and hierarchical de novo association strategy was used to sequence the genome of Monilinia fructicola (Mfrc123), the brown rot pathogen, through a combination of Illumina short-read NGS and Pacific Biosciences (PacBio) long-read third-generation sequencing platforms (Angelini et al. 2019). The genome of the coffee rust fungus Hemileia vastarix was sequenced using PacBio RS II and Illumina HiSeq platforms (Porto et al. 2019).
Limitations
The major limitation in plant pathology is the emergence and resurgence of pathogens. New plant pathogens or more aggressive strains evolve due to climate changes, global trade, and human activities (Olsen et al. 2011). Identifying these novel pathogens and managing their threats is a significant challenge for plant pathologists (Zeilinger et al. 2016). Karnal bunt is a fungal disease of wheat, durum, and rye triticale caused by Tilletia indica. This disease first emerged in Karnal, India during 1931 and was initially restricted to South Asia and Iraq (Singh et al. 1989). It became of global importance following its discovery in Mexico in 1972, and USA in 1996 (Bonde et al. 1997). The disease decreases seed viability and flour quality and wheat consisting of 3% or greater bunted kernels is considered unfit for human consumption (Bansal et al. 1984; Brennan et al. 1992). The causative agent of chestnut blight is Cryphonectria parasitica and it was restricted to East Asia. Japanese chestnut (Castanea crenata) which is resistant to this disease was introduced from Japan to North America during the late nineteenth century. The causative agent has been moved with imported chestnut plants and infected to American chestnut and has killed most mature American chestnut (C. dentate) trees within their natural range (Anagnostakis 2001). Anderson et al. (2004) listed many emerging and resurgent cases of plant pathogens worldwide.
Morphological observations and interpretations in pathogen identification can be rather difficult and are often based on the interpretative skills and experience of the analyst (Rajapaksha et al. 2019). Not all pathogens can be cultivated in an artificial environment (Jayawardena et al. 2018), leading to reliance on sequence data obtained directly from the infected material, which has proven to be difficult. With the use of DNA sequence data, mycologists came up with different genes that can be used to give a better resolution in species identification. However, there is no standard as to which gene(s) should be analyzed or how much sequence divergence is needed, or what statistical support is required at both the individual locus and the combined concatenated sequence levels to determine whether different strains belong to different species (Lücking et al. 2020). This has resulted in dubious species identification.
Another limitation is that some taxonomic studies rely on relatively few samples (one strain) when introducing phytopathogens. However, further analyses of closely related DNA sequences based on ‘phylogenetic or genotypic cluster species’ with a larger sample size, may reveal an abundance of recombination (Liu et al. 2016; Zhang et al. 2023a, b). The best examples of this are Colletotrichum siamense and Diaporthe eres. Separate species designations are mainly due to the inadequate sampling size (Liu et al. 2016). In the use of the GCPSR concept, the resulting trees are only as informative as the specific loci chosen for sequencing and the alignment used as input data (Taylor et al. 1999). If the sequences that are used are wrong, the result will also be wrong leading to misidentification of species. The major limitation in NGS is the time consumption during assembly and analysis of large amounts of sequence data (Espindola et al. 2015) as well as low yield and/or integrity, stability and impurities (Cortés-Maldonado et al. 2020). Also, expertise in bioinformatics and mycology are necessary for NGS analysis and are mandatory to avoid any misinterpretations.
Plant diseases can be caused by multiple fungal genera that affect diverse hosts with different tissue specificities involving a range of symptoms (Bhunjun et al. 2021). In order to confirm whether a species is actually the disease-causing agent, a pathogenicity assay is needed. There are limitations from selecting inoculation methods (wounded, non-wounded, spore suspension, mycelium plug) to analysing the disease symptoms (disease charts) (Bhunjun et al. 2021). Determining whether a fungus is a true pathogen needs more studies. Some species have been introduced or reported as associated with a disease when Kochs’ postulates cannot be implemented. In the case of Colletotrichum, a pathogenic genus, pathogenicity assay data are available for only one-third of its species. Disease epidemiology of fungal genera or species is not well-studied or identified. Hence, this makes it difficult to identify how the diseases are spread and when to apply the control measures.
Due to the changes in climate and weather patterns, global trade, and human activities, many new fungal pathogens are emerging, or more aggressive strains are re-emerging (Gomdola et al. 2022). Identifying and managing these novel threats is a significant challenge for plant pathologists. While imaging techniques can detect biotic stress in the plant before visual symptoms appear, the technique lacks capability to identify specific pathogens. More novel approaches for determining, characterising, and monitoring fungal pathogens are required as traditional methods are time-consuming and have other limitations.
Future
Incorporating a new disease paradigm such as pathobiome can provide more information about the disease epidemiology. The pathobiome concept, has been invoked in cases where the disease is believed to result from interactions between a set of organisms (including eukaryotic, microbial and viral communities) within the plant and its biotic environment, leading to the deterioration of host health status is needed (Collinge et al. 2022). Although the concept of pathobiome is relatively new in plant pathology, several studies have reported on diseases caused by multi-species “disease complexes” (Mazzola and Freilich 2017). Examples of diseases caused by multi-species complexes include tomato pith necrosis, soft rot in broccoli, and young grapevine decline. These diseases are caused by multiple bacterial or fungal species that interact synergistically to impact disease development. It is suggested that the pathobiome concept be applied to postharvest diseases, providing a more comprehensive perspective on disease development involving intricate assemblages of microorganisms (Droby et al. 2022).
The developments in omics approaches in plant disease ecology have been particularly important as fungi can be spread around the world via globalisation changing the composition and ecology of habitats. Invasive pathogens with a broad range of hosts can cause chaotic results in ecosystems. Shifting temperatures and the frequency and duration of weather conditions over time result in phenomena such as the rapid evolution of microbial pathogens or environmental stress which can weaken plant hosts. Omics are primarily aimed to enhance the understanding of plant-pathogen interactions at the molecular level (Crandall et al. 2020). A multi-omics approach allows for a detailed account of plant-microbial interactions and can eventually allow us to build predictive models for how microbes and plants will respond to stress under environmental changes (Santini et al. 2015; Gilbert et al. 2010; Rizzo et al. 2002). These technologies will identify new targets for disease control and improve the development of resistant plant varieties (Filgueiras et al. 2019). Pangenomics can be used to identify virulence processes in a rapidly evolving fungal plant pathogen (Chen et al. 2023a).
Climate-resilient plant pathology is studying the impact of climate change on plant diseases and developing adaptive strategies to combat disease outbreaks under changing climatic conditions (Velásquez et al. 2018). Disease forecasting tools are not used abundantly at present. This can help the farmers to identify when a disease is going to occur as well as the best time to use fungicides (El Jarroudi et al. 2017; Maddalena et al. 2023).
Current trends, limitations, and future research in HTS
The study of fungal diversity is a rapidly growing field, with researchers worldwide working to understand their distribution in diverse ecosystems. The development of high-throughput sequencing (HTS) technology and the application of meta-approaches, such as microbiome sequencing, has revolutionised how researchers analyse and interpret fungal diversity (Gutleben et al. 2018). High-throughput sequencing is based on the sequencing of the internal transcribed spacer (ITS) region (as the primary barcode), followed by the small (SSU) and large subunit ribosomal ribonucleic acid (LSU) and their combinations (Taberlet et al. 2012; Nilsson et al. 2019a, b; Tedersoo et al. 2020a, b; Semenov 2021). The sequencing generates multiple sequences from the same samples, which have output as operational taxonomic units (OTUs) (Nilsson et al. 2019a, b; Tedersoo et al. 2020a, b). Runnel et al. (2022) demonstrated the effectiveness of long-read HTS, specifically PacBio HTS, for the taxonomic identification of fungal specimens. Their study showed the advantages of long-read HTS over the traditional Illumina sequencing (Sanger method), including higher success rates and the ability to detect gene polymorphism, taxonomic delimitation, and ecological and population-level studies.
High-throughput sequencing can provide comprehensive insights into taxonomic and functional diversity of fungal communities. High-throughput sequencing techniques have enabled the identification and characterisation of diverse fungal species in various environments such as soil, plants, and the human body. It can recover DNA sequences in extreme environments (Tedersoo et al. 2020a, b; Rämä et al. 2017; Ogaki et al. 2021a, b) where cultivation is difficult or impracticable (Semenov 2021). These methods have also shed light on fungal microbiome diversity, composition, and functional potential (D’Hondt et al. 2021; Fan et al. 2023). In the most recent studies, HTS has significantly impacted estimating the number of fungi (Wu et al. 2019; Baldrian et al. 2022a, b). Põlme et al. (2020) introduced the user-friendly database FungalTraits, a stand-alone spreadsheet dataset covering 17 lifestyle-related traits of fungal and Stramenopila genera, and the endemicity of soil taxa (Tedersoo et al. 2022). The GlobalFungi (https://globalfungi.com/, Větrovský et al. 2020) and the Global Soil Mycobiome consortium dataset (Tedersoo et al. 2021a, b) have attempted to gather information on soil fungal diversity and construct a curated HTS sequence database, respectively. Other studies have focused on fungi to answer specific questions. For example, Baldrian et al. (2022a, b) estimated the number of fungi; Sun et al. (2019) used different HTS platforms for measuring Fungi and Oomycetes.
Several examples have highlighted the potential of HTS in fungal studies. In clinical settings, it has been used for the early identification of infection in culture-negative and food-born pathogens. In ecology, it has been used to study fungi in several environments, such as aerial (Mbareche et al. 2019), aquatic (Hassett et 2017; Lepère et al. 2019; Souza et al. 2021; Garmendia et al. 2021) and soil (e.g., Young et al. 2016; Tedersoo et al. 2020a, b, 2021a, b, 2022; Yasanthika et al. 2022; Wydro et al. 2022). It has also been used in plant pathogen detection and surveillance (Bérubé et al. 2018; Piombo et al. 2021).
Limitations
While high-throughput sequencing has revolutionised microbiome research, it is not without its limitations. HTS methods have challenges and possible sources of error, which require careful consideration when using and interpreting HTS data and outcomes. One major challenge is the high rate of erroneous base calls produced by HTS technologies, such as Illumina sequencing machines, which have errors at approximately 0.1-1E10−2 per base sequenced (Lou et al. 2013). This error rate can present a profound barrier in contexts where rare genetic variants are sought. Methods are also unable to differentiate living and dead cells or organisms, amplifying inactive DNA (Tuininga et al. 2009). This can lead to inaccurate estimations of microbiome structures and, consequently, functional capacities (Dlott et al. 2015; Carini et al. 2020; Nagler et al. 2021). This effect would be most noticeable if artefact DNA is abundant and if the taxa represented in the relic DNA pool do not accurately reflect the taxa present as living cells (Carini et al. 2020).
Additionally, during the analysis stage, the absence of reference databases and the amplification of selected barcodes, results in challenges in assigning OTUs at lower taxonomic ranks, resulting in erroneous or unclassified taxa (Hongsanan et al. 2018; Wu et al. 2019). Comparisons between datasets such as the Global Fungi dataset (Větrovský et al. 2020) and GSMc (Tedersoo et al. 2021a, b) have shown significant levels of contradictions in the richness of certain fungal groups due to different sampling strategies and analytical biases such as the use of different primers for metabarcoding and lack of properly annotated reference sequences (Tedersoo et al. 2021a, b).
High-throughput sequencing requires storage facilities, computational power, and specialised personnel (Rincon-Florez et al. 2013; Sota et al. 2014; Nilsson et al. 2019a, b; Wu et al. 2019), which are limited to a few research institutions or specialised companies. However, the successful implementation of high-throughput sequencing extends beyond these resources. It requires a close collaboration between bioinformaticians and mycologists. This interdisciplinary collaboration is crucial for meaningful results. Bioinformaticians bring their expertise in managing and interpreting large datasets and help design experiments and analyse data in ways that answer the specific questions which mycologists are interested in, while mycologists provide the necessary biological context and understanding of the studied organisms and guide bioinformaticians in understanding the biological significance of their findings.
HTS analysis lacks standardised methods for sample collection, DNA extraction, library preparation, and data analysis. The variability introduced by these differences can hinder the comparability of findings across studies. Standardisation of protocols and methodologies is necessary to ensure reproducibility and facilitate meta-analyses (Li et al. 2022a, b, c). Accurately identifying and classifying fungal species using HTS data is another limitation in mycobiome research. Fungal taxonomy is complex and constantly evolving, and reference databases for fungal sequences are often incomplete or inadequate. Additional techniques or curated databases may be required to accurately assign taxonomy to fungal sequences (D’Hondt et al. 2021). To overcome these limitations and shape future research, several promising directions have emerged. Integrating multi-omics data, such as metagenomics, metatranscriptomics, and metabolomics, can provide a holistic understanding of microbial community dynamics (Muller et al. 2013; Abram 2015). Establishing standardised protocols for sample collection, storage, and data analysis is crucial to ensure the reliability and comparability of results (Bella et al. 2013; Wang et al. 2023a, b, c). Moreover, exploring microbiome-based therapeutics and the role of microbiomes in environmental sustainability, including soil health and sustainable agriculture, holds promise for addressing global challenges (Tedersoo et al. 2017).
Future research
Notwithstanding these limitations, HTS remains a powerful tool for microbiome research, providing comprehensive insights into the taxonomic and functional diversity of microbial communities. Advancements in DNA and RNA sequencing technologies have enabled the integrative study of fungal communities, including their taxonomic profiles and functional and ecological attributes. This allows for a better interpretation of communities and the ability to address questions related to ecosystem functioning, including intra- and interkingdom interactions. However, standardisation, taxonomy challenges, and multi-omics data integration remain critical focus areas in mycobiome research. Ongoing advancements in HTS technology, such as long-read sequencing and data analysis, help to address some of these limitations and improve the accuracy and reliability of HTS-generated data. As HTS data accumulate, it becomes increasingly important to use those data to explore new research questions, hypotheses, and theories.
Fungal metabarcoding is an interdisciplinary and reproducible research strategy that requires expertise in mycology, ecology, Earth sciences, bioinformatics, statistics, and laboratory and analytical procedures. Indeed, while HTS does present technical challenges, it also opens up a world of opportunities for interdisciplinary collaboration. This collaboration is beneficial and essential to fully harness the potential of HTS in mycology and other biological fields. The synergy between bioinformatics and mycology can lead to what were previously impossible breakthroughs within a single discipline. It allows for a more comprehensive understanding of the data, leading to more accurate and meaningful results. By working together, bioinformaticians and mycologists can unlock new insights and push the boundaries of what is possible in mycology. Mycology must ask significant scientific questions and target large-scale patterns and processes. Producing reproducible results by following applicable standards and protocols, providing ample detail on data processing and analysis, and making all relevant data freely and openly available is crucial. Failure to do so will maintain the view that fungi matter only to mycologists, a belief that has haunted mycology for far too long.
Fungal nanotechnology: fungi-based nanoparticles, current applications, challenges and prospects for future research
Nanotechnology is a rapidly advancing field that involves manipulating and controlling substances at the nanoscale, which is about 10–9 m in size (Bayda et al. 2020). It encompasses a wide range of scientific disciplines, i.e., biology and engineering, to understand and harness the unique properties of materials at the atomic and molecular levels (Paramasivam et al. 2021). This field holds tremendous potential for various industries, including electronics, medicine, energy, and manufacturing, by enabling the development of novel materials and devices with enhanced properties and functionalities (Green et al. 2015; Peer et al. 2007; Meyers et al. 2006). In nanotechnology, nanoparticles have become an integral part of our daily lives, often working behind the scenes to enhance product functionality and user experience. A prevalent example is in the field of cosmetics, where nanoparticles, particularly of titanium dioxide and zinc oxide, are employed in sunscreens to offer broad-spectrum UV protection without leaving a white residue on the skin (Smijs and Pavel 2011). In medicine, nanoparticles have shown promise in targeted drug delivery, allowing for increased drug efficiency and reduced side effects (Yusuf et al. 2023). The electronics industry harnesses the unique electronic, optical, and mechanical properties of nanoparticles to develop improved displays, batteries, and memory storage devices (Malik et al. 2023). Nanoparticles also enhance the protective nature of some textiles, filling them with water-repellent, stain-resistant, or even antimicrobial properties (Saleem and Zaidi 2020). In the environmental sector, nanoparticles aid in water purification processes by binding to and removing contaminants (Kumar 2023). Thus, from personal care products to high-tech gadgets and environmental solutions, nanoparticles play an essential role in driving advancements and refining everyday experiences.
The demand for clean, non-toxic, and environmentally friendly approaches, commonly referred to as "green chemistry", in nanoparticle synthesis and assembly is increasing. This has prompted researchers to explore biological systems for inspiration (Mukherjee et al. 2001). Recently, the utilization of microorganisms and plants for synthesising metal nanoparticles (MtNPs) has gained recognition as an efficient and sustainable method, enabling further exploration of microorganisms as “nanofactories”. Microorganisms, cultivated on a large scale, play a crucial role as nanofactories due to their ability to accumulate and detoxify heavy metals. This capability is attributed to the presence of diverse reductase enzymes that facilitate the reduction of metal salts into MtNPs.
Fungi, as a diverse group of organisms, have gained significant attention in recent years for their potential application in various fields, including nanotechnology. Moreover, fungi present a convenient option for cultivation as nanofactories, allowing for the production of nanoparticles with precise control over their size and morphology (Gade et al. 2008; Ahluwalia et al. 2014; Azmath et al. 2016; Khan et al. 2017, Guilger-Casagrande de Lima 2019). The utilization of fungi in nanoparticle synthesis represents a promising and sustainable approach within the rapidly evolving field of nanotechnology, with applications in areas such as medicine, agriculture, and environmental remediation (Guilger-Casagrande de Lima 2019; Moond et al. 2022; Sharma et al. 2023). Myconanotechnology, as it is commonly known, is thus defined as the interface between nanotechnology and mycology (Hanafy 2018; Sousa et al. 2020; Adebayo et al. 2021). In this section of the article, our aim is to discuss the fungi-based nanoparticles, current applications, limitations, and potential future prospects in this captivating field of research.
Biosynthesis of nanoparticles by fungi
Fungi have become a valuable addition to the microorganisms used in nanoparticle production. They are effective candidates for synthesizing metal nanoparticles, both intracellularly and extracellularly. In intracellular synthesis, fungi serve as nanofactories for the production of metal nanoparticles within their cellular structures. This process entails the uptake and accumulation of metal ions by fungal cells, subsequently reducing these ions to generate nanoparticles. Conversely, extracellular synthesis involves the utilization of fungal biological systems to generate metal nanoparticles outside the confines of the cells. This mechanism is facilitated through the secretion of enzymes or biomolecules by fungi, which act as reducing agents to convert metal ions into nanoparticles within the surrounding environment. Fungal-synthesized nanoparticles have excellent dispersion and stability. Fungi offer advantages such as the presence of specific enzymes, ease of handling in the lab, scalability, and cost-effectiveness (Castro-Longoria et al. 2012; Siddiqi and Husen 2016; Guilger-Casagrande and Lima 2019; Bourzama et al. 2021). Fungi can produce various metal nanoparticles, including silver, gold, platinum, palladium, copper, iron, selenium and tellurium nanoparticles.
Silver nanoparticles
The utilization of fungi for the biological synthesis of silver nanoparticles (AgNPs) has garnered significant attention in scientific research over the past two decades. Notably, more than 120 fungal species from diverse taxa, including Ascomycota, have demonstrated the ability to produce nanosilver (Loshchinina et al. 2023). Basidiomycota, in particular, have attracted considerable interest as bio-objects for nanoparticle fabrication, as highlighted by Loshchinina et al. (2023). Their unique properties and potential applications make the mycosynthesis of AgNPs a compelling area of investigation in the field of nanotechnology. These nanoparticles have been found to possess remarkable antibacterial, antifungal, anticancer, antioxidant, and larvicidal activities, among other beneficial properties (Khan et al. 2018; Ratan et al. 2020). Notably, the same nanoparticles have been shown to exhibit a diverse range of biological activities, underscoring their versatility and potential for various applications (Rafique et al. 2017a, b; Razak et al. 2021). The broad spectrum of therapeutic and functional attributes displayed by mycosynthesized AgNPs reinforces their significance in fields such as medicine, biotechnology, and environmental science.
Gold nanoparticles
Gold nanoparticles (AuNPs) have found widespread application in diverse processes, ranging from chemical and biological sensing to bio-imaging, nonlinear optics, catalysis, targeted drug delivery, and gene delivery (Elahi and Baghersad 2018). Additionally, they exhibit antimicrobial and antioxidant properties, making them valuable in cancer and infectious disease therapy (Ahmed et al. 2016). The biological synthesis of AuNPs by fungi has also been extensively investigated, with various species demonstrating the ability to form these nanoparticles. Notably, these AuNPs show promise as dual-modal (chemo-photothermal) therapeutic agents for anticancer applications. It was observed that synthesized gold nanospheres (10–50 nm) effectively inhibited the growth of clinically significant Gram-positive and Gram-negative bacteria, as well as pathogenic fungi (Loshchinina et al. 2023). These findings highlight the potential of mycosynthesized AuNPs as versatile and effective therapeutic candidates.
Platinum nanoparticles
Platinum nanoparticles (PtNPs) have catalytic, magnetic, and optical properties, as well as antimicrobial, antioxidant, and anticancer properties (Jeyaraj et al. 2019; Fahmy et al. 2020). While the mycosynthesis of PtNPs has been less well-studied compared to silver and gold nanoparticles, some Ascomycota species have been found to have the ability to form PtNPs. In a study by Borse et al. (2015), PtNPs synthesized by Saccharomyces boulardii showed anticancer activity.
Palladium nanoparticles
The synthesized PdNPs exhibit unique physicochemical properties, such as excellent catalytic activity and enhanced stability. These properties make them suitable for various applications, such as the development of novel photothermal, photoacoustic, antimicrobial, and antitumor agents, gene/drug carriers, prodrug activators, and biosensors (Phan et al. 2019). The mycosynthesis of palladium nanoparticles (PdNPs) in fungal cultures has received limited attention, with only a few publications reporting its occurrence in Agaricus bisporus, Inonotus obliquus, and Saccharomyces cerevisiae (Mohana et al. 2020, Gil et al. 2018, Sriramulu et al. 2018, Saitoh et al. 2020).
Copper nanoparticles
Biosynthesized copper nanoparticles (CuNPs) possess a range of beneficial properties, including antibacterial, antifungal, antiviral, and anticancer activities. These nanoparticles have potential applications in targeted drug delivery, cosmetics, catalysis, microelectronics, gas sensors, high-temperature superconductors, solar cells, bactericide agents, wound dressings, biopesticides, bioremediation, biodegradation, and energy storage (Rafique et al. 2017a, b; Al-Hakkani 2020; Chaerun et al. 2022). However, the mycosynthesis of CuNPs remains an area of limited research, with species such as Agaricus, Aspergillus, Fusarium, Hypocrea, Shizophyllum, Stereum, and Trichoderma reported to be capable of producing CuNPs (Loshchinina et al. 2023).
Iron nanoparticles
Iron nanoparticles (FeNPs) and iron-based nanomaterials play a crucial role in addressing environmental pollution through their ability to degrade organic dyes, remove heavy metals, and treat wastewater. In addition, these nanomaterials hold promise in biomedicine as antimicrobial agents (Saif et al. 2016; Pasinszki and Krebsz 2020). The production of FeNPs has been primarily investigated in Ascomycota micromycetes, with species such as Alternaria, Aspergillus, Fusarium, Penicillium, Pleurotus, Rhizopus, and Trichoderma being reported as capable of synthesizing FeNPs (Loshchinina et al. 2023).
Selenium nanoparticles
Selenium nanoparticles (SeNPs) have garnered significant attention due to their reduced toxicity compared to inorganic and organic selenium compounds, as well as their biocompatibility, bioavailability, and biomedical properties. Nano-selenium demonstrates remarkable antimicrobial, anticancer, antidiabetic, antiparasitic, and antioxidant activities (Bisht et al. 2022). SeNPs find applications in targeted drug delivery, bioremediation, nanobiosensors, food supplements, and various other fields (Shoeibi et al. 2017). Selenium nanoparticles (SeNPs) synthesized through mycosynthesis using culture liquid of Aspergillus flavus and Candida albicans have demonstrated potent antifungal activity. These nanoparticles exhibit the ability to inhibit fungal growth at lower concentrations compared to conventional antifungal drugs (Bafghi et al. 2021). The biological synthesis of SeNPs has been observed in a substantial number of fungal species, with Aspergillus, Penicillium, and Trichoderma being among the most extensively studied genera in this regard (Loshchinina et al. 2023).
Tellurium nanoparticles
These nanoparticles possess a wide range of properties, including photoconductivity, thermoconductivity, piezoelectricity, non-linear optical behavior, antioxidant activity, antimicrobial activity, anticancer effects, immunomodulation, and cytotoxicity. Moreover, TeNPs show potential for applications in drug delivery, bioremediation, and biorecovery, making them an area of significant interest (Zambonino et al. 2021). However, the formation of TeNPs in fungi remains relatively understudied. Thus far, tellurium nanospheres have been successfully obtained from species such as Aspergillus welwitschiae, Aureobasidium pullulans, Mortierella humilis, Penicillium chrysogenum, Phanerochaete chrysosporium, Phoma glomerata, and Trichoderma harzianum (Loshchinina et al. 2023).
Current trends in fungal nanobiotechnology
Current trends in myconanotechnology applications highlight the significant potential of this field across various sectors. The unique properties and capabilities of fungi make them valuable tools in medicine, agriculture, environmental remediation, and materials science. Recent developments in fungal nanoparticle synthesis and manipulation have paved the way for novel applications and outcomes. Over the years, significant advancements have been made in the synthesis and manipulation of fungal-derived nanoparticles, leading to innovative applications and results. The combination of fungal biology and nanotechnology holds great promise for addressing societal challenges and revolutionizing industries.
Medicine and biomedical applications
Fungal nanotechnology has shown great promise in the field of medicine. Fungal-derived nanoparticles have been utilized in drug delivery systems, diagnostics, and therapeutics. For example, the antifungal properties of silver nanoparticles synthesised by fungi have been explored for the treatment of fungal infections (Gajbhiye et al. 2009; Liang et al. 2022). These nanoparticles exhibit enhanced efficacy and reduced toxicity compared to traditional antifungal drugs (Bafghi et al. 2021). Furthermore, fungal nanocarriers have been developed to deliver drugs to specific target sites, improving drug bioavailability and reducing side effects (Bafghi et al. 2021). Fungal nanoparticles have also been used in biosensing and imaging applications (Ahmed et al. 2016, Elahi and Baghersad 2018, Kalimuthu et al. 2020). Their unique optical and magnetic properties make them suitable for developing biosensors, imaging agents, and contrast agents for various imaging modalities, including magnetic resonance imaging (MRI) and fluorescence imaging. Fungal nanotechnology holds great potential in early disease detection, personalized medicine, and targeted therapy (Rai et al. 2019; Mota et al. 2023).
Agriculture and crop improvement
Fungal nanotechnology has found applications in agriculture and crop improvement. Fungal nanoparticles can be used as biofertilizers, biopesticides, and growth promoters (Mishra and Kumar 2009; Prasad et al. 2014; Ponmurugan et al. 2016). For example, nanoparticles synthesized from fungal extracts have been shown to enhance plant growth, improve nutrient uptake, and increase crop yield (Tripathi et al. 2017; Khalifa and Hasaneen 2018). These nanoparticles can also act as carriers for delivering nutrients, pesticides, and genetic materials to plants, enabling more efficient and targeted delivery (Kumar et al. 2017; Tripathi et al. 2017; Cao et al. 2018). Additionally, fungal nanotechnology has the potential to address challenges in plant disease management (Wang et al. 2021). Fungal nanoparticles can inhibit the growth of plant pathogens and suppress the development of diseases (Abbacia et al. 2014; Adebayo et al. 2021). They can also be used in the development of nanosensors for the early detection of plant diseases, allowing for timely interventions and reduced crop losses.
Environmental remediation
Fungal nanotechnology offers promising solutions for environmental remediation and pollution control. Fungi have the ability to synthesize nanoparticles with unique properties that can be employed in the removal of pollutants from soil, water, and air. Fungal nanoparticles can act as efficient adsorbents, catalysts, or photocatalysts for the degradation of organic pollutants, heavy metal ions, and even radioactive contaminants (Viswanath et al. 2008, 2014; Bahrulolum et al. 2021). These nanoparticles can be used in water treatment, wastewater purification, and soil remediation processes, contributing to sustainable environmental management.
Industrial science
The industrial application of fungal nanobiotechnology holds significant potential for various sectors. Fungal nanoparticles can be utilized in the development of advanced materials, such as nanocomposites, coatings, and membranes, with improved properties and performance (Muñoz et al. 2006; Tsivileva et al. 2021). These materials find applications in the automotive, aerospace, electronics, and packaging industries. Fungal nanobiotechnology also offers opportunities for the production of high-value chemicals, enzymes, and biofuels through the use of fungal bioreactors (Williamson et al. 1998, Polizeli et al. 2005, Azin et al. 2007, Shraddha et al. 2011, Uday et al. 2016, Elegbede and Lateef 2018). The ability of fungi to synthesize nanoparticles with unique properties, combined with their scalability and sustainability, makes them attractive candidates for industrial processes.
Current challenges in fungal nanobiotechnology
Since fungi-based nanotechnology is developing, it faces several limitations and challenges that need to be overcome for successful implementation. One of the major hurdles is the limited exploration of fungal diversity suitable for nanotechnology applications. The availability of fungal species with the ability to produce nanoparticles and possess desired properties is restricted, hampering the discovery of potential fungal candidates (Adebayo et al. 2021). Another challenge lies in the incomplete understanding of fungal nanoparticle synthesis mechanisms (Guilger-Casagrande de Lima 2019). The complex interplay between fungal cells and the surrounding environment during nanoparticle synthesis is a subject of ongoing research. Gaining a deeper understanding of these mechanisms is crucial to optimize and control the synthesis process.
Scalability and reproducibility pose significant challenges when transitioning from laboratory-scale to industrial-scale production of fungal nanoparticles (Ganeshan et al. 2021). Maintaining consistent and reproducible synthesis conditions, ensuring the quality and desired properties of the nanoparticles, and developing scalable and reliable production methods become increasingly complex at larger scales. Therefore, it is necessary to focus on the development of robust and efficient production processes to enable the practical implementation of fungi-based nanotechnology.
Precise control over nanoparticle properties is another limitation that needs to be addressed. Factors such as size, shape, composition, and surface characteristics significantly influence the functionality of nanoparticles (Loshchinina et al. 2023). However, achieving precise control over these properties is challenging in fungi-based nanotechnology, limiting the customization and optimization of fungal nanoparticles for specific applications. Further research and innovation are required to enhance control over these properties and enable tailored nanoparticle design.
Biocompatibility and toxicity concerns are critical considerations, particularly in applications involving medicine and environmental remediation (Deng et al. 2018; Dasari et al. 2014). A thorough assessment of the impact of fungal nanoparticles on living organisms and ecosystems is necessary to ensure their safe use. Understanding the long-term effects and potential risks associated with exposure to fungal nanoparticles is an ongoing challenge that requires comprehensive research and evaluation.
The regulatory landscape for fungi-based nanotechnology is evolving, with a lack of clear guidelines and regulations specific to fungal nanoparticles. Addressing potential safety concerns, establishing standardized protocols, and developing appropriate regulations are essential to ensure responsible and ethical implementation (Arnaldi and Muratorio 2013; Loshchinina et al. 2023). Collaboration between researchers, industry stakeholders, and regulatory bodies is crucial to develop a robust regulatory framework that ensures the safe and effective use of fungal nanoparticles.
The cost-effectiveness and commercial viability are significant factors for the widespread adoption of fungi-based nanotechnology. Large-scale production processes need to be developed to reduce production costs and make fungal nanoparticles economically competitive with other nanomaterials (Singh et al. 2016). Additionally, optimizing the scalability and stability of fungal nanoparticle synthesis methods is necessary to meet the demands of industrial applications.
Way forward in fungal nanobiotechnology
Fungal nanotechnology is an emerging field with significant potential for advancement. Overcoming the limitations and challenges in fungi-based nanotechnology requires interdisciplinary research, collaboration, and innovation. Expanding the exploration of fungal diversity, gaining a deeper understanding of synthesis mechanisms, developing scalable and reproducible production methods, enhancing control over nanoparticle properties, addressing biocompatibility and toxicity concerns, establishing regulatory frameworks, and optimizing cost-effectiveness and commercial viability are crucial steps towards harnessing the full potential of fungi-based nanotechnology in various sectors.
Researchers are actively exploring different fungal species and refining synthesis methods to improve the efficiency, scalability, and reproducibility of producing fungal nanoparticles. Additionally, there is a growing focus on unravelling the intricate mechanisms involved in fungal nanoparticle synthesis, enabling better control over their size, shape, and surface properties. Moving forward, fungal nanotechnology offers promising applications in personalized medicine, with the development of targeted therapies and diagnostic tools customized to individual patients. It also has the potential to revolutionize sustainable agriculture practices by reducing reliance on chemical inputs and enhancing crop productivity. Furthermore, the application of fungal nanoparticles in environmental remediation presents an opportunity to address pressing challenges in pollution control and waste management.
To fully harness the potential of fungal nanotechnology, efforts should be dedicated to the comprehensive exploration and study of a wider range of fungal species. This can be achieved through biodiversity surveys, screening programs, and collaborations between researchers and fungal collections. Further research is needed to enhance our understanding of the mechanisms underlying fungal nanoparticle synthesis, utilizing interdisciplinary collaborations and advanced techniques such as genomics, transcriptomics, and proteomics.
The scalability and reproducibility of production methods are crucial considerations, necessitating optimization of growth conditions, synthesis parameters, and downstream processing steps. Collaboration between academia and industry is key to bridging the gap between laboratory-scale research and industrial-scale production. Investment in advanced characterization techniques, such as electron microscopy and spectroscopic methods, can provide valuable insights into the morphology, composition, and surface properties of fungal nanoparticles. To achieve better control over nanoparticle properties, research efforts should focus on studying the influence of fungal species, growth conditions, and synthesis parameters. Strategies such as genetic engineering, surface modification, and controlled synthesis techniques can enable precise control over desired properties. Rigorous safety assessment protocols and risk management strategies are essential to evaluate the toxicity and biocompatibility of fungal nanoparticles and ensure their responsible and safe implementation. Collaboration between researchers, industry stakeholders, and regulatory bodies is vital for developing a robust regulatory framework for fungi-based nanotechnology. Clear guidelines and regulations should be established through active engagement with the scientific community. Additionally, research should aim to enhance cost-effectiveness and commercial viability by optimizing production processes and exploring alternative substrates.
Moreover, fungal nanotechnology offers the capability to create innovative materials with distinctive structures and functionalities. Fungal mycelium, the vegetative component of fungi, has been harnessed as a sustainable and biodegradable material for the production of bioplastics, construction materials, and even 3D-printed objects (Holt et al. 2012; Haneef et al. 2017; Gantenbein et al. 2023). This field holds significant promise in the advancement of tailored materials with specific properties to cater to diverse applications.
Current trends, limitations and future research in AMF
Arbuscular Mycorrhizal Fungi (AMF) form symbiotic associations with plant roots that provides benefits to both parties. Most currently described AMF taxa belong to the phylum Glomeromycota (or subphylum Glomeromycotina)(Wijayawardene et al. 2020). Within this phylum, there are five orders, 16 families and 49 genera. Here is a summary of current trends, limitations, and future research in AMF.
Current trends
Through their symbiotic relationship with plants, AM fungi receive up to 20% of the total photosynthetic C fixed by the plant (Smith et al. 1997) with an estimated global average of 6.2% (Hawkins et al., 2018). Plants can transfer photosynthate C via arbuscular fungal structures in the root cortex to external hyphae in soil within hours (Terrer et al. 2016). As exchange, plants receive from their mycorrhizal fungal symbionts mineral nutrients and a higher capacity for water uptake. Arbuscular mycorrhizal symbiosis also enhances host plant protection against biotic stress. Recent developments in the field of mineral nutrients (particularly phosphorus and nitrogen) uptake by AM plants via their mycorrhizal symbionts was covered by several review papers (Kuyper and Jansa 2023; Xie et al. 2022), therefore we do not aim to comprehensive overview of this topic in this chapter.
Besides the ongoing research on AM physiology, significant attention is given to the ecological aspects of AM symbiosis. By interacting with approx. 75% of vascular plants, AM fungi play important role in all terrestrial ecosystems having large effects on vegetation and ecosystem processes (Brundrett and Tedersoo 2018). Therefore, response of AMF to global change and their role in mitigating its adverse effects have gained much interest. The climatic niche optima and width differ among the species of AM fungi. While Acaulosporaceae has a realized niche optima in low temperature conditions, Gigasporaceae has a realised niche optima in high temperature and high precipitation conditions (Davison et al. 2021). The width of the AM fungal temperature niche appears to be limiting, likely narrower than in other fungal guilds (Davison et al. 2021; Vetrovsky et al. 2019). These findings indicate that changes of MAT and MAP can particularly affect the composition of AM fungal communities. Climate change factors such as warming and precipitation changes can indeed affect AMF abundance and community composition directly (Cotton et al. 2015). While the increase of CO2 and temperature does not seem to affect AMF profoundly, drought and nitrogen deposition often decrease AM biomass in soils and all of the above factors can bring about changes in AMF community composition (Baldrian et al. 2022a, b). A study by Jia et al. (2021) has shown that mycorrhizal fungi can maintain plant community stability by mitigating the negative effects of nitrogen deposition in Central Asia. Another study by Mei et al. (2019) also showed that AMF can alleviate phosphorus limitation by reducing plant N:P ratios under warming and nitrogen addition in meadow soils.
There is increasing research focusing on how AMF perform their ecological function at a commmunity level and how AMF communities respond to climate changes (Xu et al. 2022). Based on a grassland extreme drought experiment in Inner Mongolia, research by Fu et al. (2022) found that the robustness of the AM fungal community response increased with drought intensity. The use of effective AMF communities may stand better chances to mitigate some drawbacks under field conditions. In the forest ecosystems of the temperate zone, the increase of nitrogen deposition with warming leads to the transition from forests dominated by ectomycorrhiza-forming trees to those forming arbuscular mycorrhiza, best described from the continental USA (Baldrian et al. 2023). Since the rates of microbial processes in AM forest soils including N cycling and C storage differs from ECM forests (Averill and Hawkes 2016), this transition has a far-reaching importance for global biogeochemisty.
The interaction between AMF and other microorganisms also gains increasingly interest in recent years, especially the increasing research about microbiome of AM fungal hyphosphere (Zhang et al. 2022). AM fungal hyphae recruit distinct microbes into their hyphosphere, the narrow region of soil influenced by hyphal exudates (Zhang et al. 2022). Recent research have also found that natural and agricultural ecosystems, various bacteria can live in the AM fungal hyphosphere and the hyphosphere microbiome is unique compared with other microbial communities when compared with the bulk soil (Zhang et al. 2022). In addition, various functions of the hyphosphere microbiome were also reported and focused on. The hyphosphere microbiome has been believed to be able to mobilize soil P and interact with soil bacteria (Svenningsen et al. 2018).
In addition, there is an increased interest in the use of AMF in sustainable agriculture due to its potential to improve plant growth, nutrient uptake, and soil health. AMF can improve soil structure and quality, increase nutrient availability and uptake, enhance plant resistance to diseases and pests, and reduce water usage (Otun and Achilonu 2023). This makes them a valuable tool in sustainable agriculture and ecological restorations, especially in low-input systems. Vahter et al. (2020) indicated that co-introduction of native AM fungi and plants is an efficient means of initializing species-rich vegetation recovery in barren post-mining landscapes. The practical application of AMF in sustainable agriculture—especially as biofertilizers—has been also frequently addressed (Madawala 2021). For instance, Al-Karaki and Williams (2021) showed that AMF inoculation can affect the growth, nutrition, and physiological responses of soybean to water deficit. Many research articles have already reported the beneficial roles of AMF protecting their hosts against fungal pathogens (Krzyzaniak et al. 2021). Overall, the current trend in the use of AMF in sustainable agriculture is to enhance plant growth and yield while reducing the environmental impact of agriculture.
Limitations
Limited understanding of the mechanisms behind the symbiotic association between AMF and plant roots, which hinders the optimization of AMF use in agriculture or other application. Though in recent years, the whole genome of several AM fungal species and isolates have been sequenced (i.e. Diversispora epigaea (Sun et al. 2019), Rhizophagus clarus (Kobayashi et al. 2018)), which are helpful to explore the diverse functions of AM fungi. But the mechanism underlying these effects needs more exploration (Shi et al. 2023). However, the limited understanding of the cellular and molecular basis of forming AM symbiosis can also hinder the applications of AMF.
Another limitation of AM research seems to be the difficulty in transforming the success stories of AMF under glasshouse conditions to real farmer fields (Helander et al. 2018). Challenges in mass production and application of AMF in agriculture due to the need for living inoculum and the difficulty in maintaining fungal viability during storage and transportation. The outcomes of mycorrhizal symbiosis on plant health and nutrient cycles are affected by many diverse biotic and abiotic environmental factors. The effects of environmental factors, such as soil type, pH, and climate, on the effectiveness of AMF remain poorly understood (Powell and Rillig 2018).
For the exploration of the relative importance of AMF in ecosystem processes, the main limitation is represented by the absence of a reliable molecular marker that is common to AMF and other fungi. The recent progress at least has led to the design of molecular barcoding markers that are able to capture well the composition of the AMF communities through sequencing of long stretches of rDNA (Kolarikova et al. 2021) Also the complexity of AMF genomes (Miyauchi et al. 2020) represents a grand challenge for the use of genomics, metagenomics and metatranscriptomics as a tool to explore AMF biology and ecology.
Future research
New technologies such as next-generation sequencing (NGS) provide insight into how AMF interact with indigenous counterparts and to screen beneficial microbial candidates with specific functional traits. In the grassland soil, mycorrhizal fungi alter root exudation to cultivate a beneficial microbiome for plant growth (Xu et al. 2023).
Investigating the genetic and molecular mechanisms involved in the symbiotic association between AMF and plant roots to better understand the factors that determine AMF effectiveness. It has been reported that these compounds, known as strigolactones, induce hyphal branching in an arbuscular mycorrhizal (AM) fungus, and subsequent work by Besserer et al. (2006) showed that a synthetic strigolactone stimulated spore germination in two other species of AM fungi. The roles of strigolactones or other root exudes in the formation of AMF in plant roots need further research (Kuyper 2023).
Beside the root exudes, the identification of genes and gene products that are responsible for controlling the growth and development of AMF under stressful conditions also need more focus (Begum et al. 2019).
One of the key areas of research is focused on the molecular signals that are exchanged between the plant and the AMF. Studies have shown that the exchange of signals involves a range of molecular components, including receptor-like kinases, transcription factors, and small signaling molecules. For example, a study by Gomez-Roldan et al. (2008) identified a receptor-like kinase in the plant that is essential for the recognition of AMF and the initiation of symbiosis.
Another area of research is focused on identifying the genes involved in the symbiotic association. This has been facilitated by advances in high-throughput sequencing technologies, which have allowed researchers to identify candidate genes involved in the symbiotic association. For example, a study by Tisserant et al. (2013) identified several genes in the AMF that are involved in nutrient exchange with the plant.
Furthermore, researchers are also exploring the role of epigenetic mechanisms in the symbiotic association. Epigenetic modifications, such as DNA methylation and histone modifications, can play a crucial role in regulating gene expression and the development of the symbiotic association between the plant and the AMF. For example, a study by Zouari et al. (2014) showed that DNA methylation is involved in the regulation of genes related to nutrient transport in the symbiotic association.
Exploring the potential of AMF to mitigate the effects of climate change on plant growth and nutrient uptake in different soil types and environmental conditions is also essential, especially in those ecosystems that are so far understudied, namely tropical AM forests (Baldrian et al. 2023). Soil microplastic (MP) pollution has emerged as a main factor of global change. A recent study by Moreno‐Jiménez et al. (2022) has shown that the use of AMF to sustainably manage agricultural ecosystems contaminated with MP, buffering and counteracting the effects of MP.
AMF can enhance plant nutrient uptake under different soil conditions, including nutrient-poor soils and soils with imbalanced nutrient ratios. This is because AMF can access nutrients that are not available to the plant through their extensive mycelium network, which can reach into deeper soil layers (Lehmann et al., 2015). AMF can also facilitate the uptake of essential nutrients such as nitrogen, phosphorus, and micronutrients, which can be limiting in some soils. The identification of genes and gene products that are responsible for controlling the growth and development of AMF under stressful conditions also need more focus from the researchers (Begum et al. 2019).
Besides members of the phylum Glomeromycota, there is a growing body of research suggesting that also other fungal groups may act as arbuscular mycorrhizal symbionts. Specifically, fine endophytes, Glomus tenue, traditionally considered to be AM fungus are related to Mucoromycotina, not Glomeromycota (Orchard et al. 2017). Recent study provides that these Mucoromycotina ‘fine root endophytes’ are nutritional mutualists with flowering plants (Hoysted et al. 2023). Understanding the similarities and differences in character of the symbioses between plants and fungi from Glomeromycotina and Mucoromycotina may shed more light on co-evolution of land plants and fungi (Field et al. 2019).
Further better understanding of AM fungal ecology should be stimulated by a recently compiled GlobalAMFungi database, which collates a comprehensive collection of published data on the composition of AM fungal communities (Větrovský et al. 2023). This open source and open access database contains almost 50 million observations of AM fungal amplicon DNA sequences across almost 8,500 samples with geographical locations and additional metadata.
One of the important challenges for better understanding of AM ecology represents independent development of AM fungal taxonomy and AM fungal ecology. While studies aiming to describe AM fungal community composition have so far largely relied on sequencing the SSU rRNA region ((Opik et al. 2014) but see e.g., Kohout et al. 2014; van Geel; (Schlaeppi et al. 2016), AM fungal morphospecies are molecularly described using different marker. Moreover, only a subset of AM fungal species is available in pure culture collections, due to the obligatory symbiotic lifestyle (e.g., INVAM (Sturmer et al. 2021)). Therefore, morphologically described AM fungal species corresponds to a subset of the AM fungal molecular diversity (Öpik et al. 2014). Although this schism dates back more than a decade, there are only limited attempts to at least into some extant interconnect these two field by sequencing longer DNA molecules, spanning all commonly utilized barcoding regions of AM fungi (de Souza et al. 2018; Kolarikova et al. 2021; Kruger et al. 2012). However, AM fungal phylogroups that are identified only on the basis of their DNA are categorised differently by studies using different AM fungal barcoding regions, which limits the direct comparability of such data.
Current trends, limitations and future research in data repositories
In the realm of biological sciences, the study of fungi has gained remarkable momentum, uncovering their intricate roles in ecosystems, medicine, agriculture, and industry. As our understanding of fungal diversity and significance expands, so does the need for robust data management and dissemination platforms. Data repository is basically termed as data archive or library. Data repositories dedicated to fungi have emerged as crucial hubs for storing, sharing, and analyzing a wealth of fungal-related information. In recent years, the world of data repositories for fungi has undergone notable transformations, aligning with broader advancements in technology and research practices, such as genomic data, taxonomy, and phylogeny, open access and collaboration, and visualization and data integration tools.
Current trends
An increased number of online data repositories has become a significant and convenient resource for all genomic information for fungal variety research and development. An online open data repository has made fungal research more accessible, faster, and more systematic. High-quality and functional genomic data and DNA sequencing technologies have led to an explosion of genomic data for various fungal species. Data repositories are increasingly focused on curating and hosting high-quality fungal genome sequences, along with associated metadata. Detailed metadata and annotations for fungal sequences were becoming increasingly important. Repositories were working to improve the quality and completeness of metadata to enhance the usability and interpretability of the stored data. The advancement of molecular phylogenetics in fungi involved creating and maintaining comprehensive phylogenetic databases that helped in understanding the evolutionary relationships among different fungal species. New species identification requires phylogenetic analysis and comparison among a large number of closer species, which is possible only with the availability of a large number of DNA sequence datasets and is solved with the availability of this facility (Nilsson et al. 2006).
Several efforts have been made to provide information on fungi collection and compilation, but systematic and online databases were only developed a few years ago. These tools enabled the scientific community and academic sector to study the diversity and evolution of fungi. Ullah et al. (2022) have compiled a collection of 76 fungal databases based on scientifically published literature. Research centers, universities, private companies, etc. are more interested in developing these kinds of websites, and the number is growing. With the opening of a broad spectrum of fungal applications, today’s generation of researchers is intensively engaged in research, including the discovery of new species and secondary metabolites and their applications in medicine and biomaterials. Mycobank, GenBank, Index Fungorum, Fungal Names, Species Fungorum, FacesofFungi, e-fungi, Global Biodiversity Information Facility (GBIF), etc. are some examples of data repositories covering fungal nomenclature and details (Table 2).
Limitations
Despite many benefits, using online data repositories is crucial and challenging to study fungal biogeography as the current depositories have some limitations. Many data repositories might have inconsistent or incomplete data entries, which can hinder the reliability and usefulness of the information available. Ensuring high data quality and completeness requires continuous efforts and strict data curation processes. The molecular evolutionary study is never-ending and primary research is based on the availability of DNA sequences. Also, advancements in science and technology will scrutinize problems in the latest online database system. Taxonomic identification’s accuracy relies more on the quality of data available in online repositories (Smith et al. 2016). With the addition of new species or fungal sequences, there is a proportionately increased probability of errors like duplication, incorrect naming, and incomplete species details. These issues have not been solved completely and must be cured on time. The scientific community has an important topic of discussion on whether to have single or multiple databases.
The research on fungal diversity has not covered the entire geography, creating complications in using repository data (Phillips et al. 2009; Troia and McManamay 2016; Hao et al. 2021). A uniform and systematic study of their geographic origin and evolutionary evidence can minimize art factual patterns about their common characteristics. Data repositories not only cannot provide all features but also require laboratory facilities to manage all the locally produced experimental data (Garwood 2006). The description of all fungal species should be scientifically understood and be uniform in every part of the world. For this, the global standard has to be established and operated accordingly. The Nagoya Protocol, agreed by 117 countries, ensures benefits that arise from the use of genetic resources of respective countries (Ryan et al. 2019); however, there are a considerable number of informally collected fungal specimens in countries with poor infrastructure and experts, which have to be analyzed and merged into the online data repositories. Additionally, ensuring the accuracy, consistency, and standardization of fungal data remains a challenge. Differences in data formats, metadata, and annotation practices can hinder data integration and analysis. We propose that the fungal taxonomic challenges are continually evolving, and there can be discrepancies and uncertainties in the classification and naming of fungal species. This can complicate data retrieval and interpretation.
Future
The potential future research directions and trends in the field of data repositories for fungi might emphasize greater community involvement, enabling researchers, citizen scientists, and enthusiasts to contribute data, share observations, and collectively build a more comprehensive understanding of fungi. Fungal research on macro-ecological and biogeographic patterns is possible only with an accurate, reliable, and complete database, which can be made with the currently used databases. According to Wang et al. (2023a, b, c), “One fungus, one name” is the easiest, correct, and most effective standard for fungal nomenclature. Combined morphological and phylogenetic analysis of fungal species avoids duplication and misinterpretation of the species worldwide, thereby demanding a global standard. Some researchers have practised and published on data cleansing in data repositories (Hao et al. 2021). All the authorised data depositories can be cleaned for higher accuracy and reliability of the information in modern research. The biogeographic regionalization function requires details about species, including geographic coordinates and collection dates. The problem of species duplication and incorrect naming in databases is a significant concern that must be addressed.
In addition, the DNA barcoding technique, a general sequencing approach, is highly used to identify fungal species, and has to be applied uniformly to all the specimens (Herbert and Gregory 2005). With the availability of an increased number of sequences, evolutionary relationships at a higher level of resolution can be performed (Gabaldon 2020). For this, large-scale scientific research has to be conducted effectively at every level and form of life, like marine, freshwater, terrestrial, desert, snow-covered regions (Polar Regions and snow-capped mountains), living body associations, and so on. Furthermore, most fungal genetic resource collections are generally kept in industrialized countries, whereas least developed or developing countries are rich in resources and diversity without appropriate infrastructure on a global scale (Ryan et al. 2019). Fungal research and infrastructure in the least developed countries are lacking, and future steps can be taken to strengthen them around the globe. The worldwide biodiversity reports and depositories reflect this discrepancy. Collaboration among developed and underdeveloped countries and international culture collections in fungal specimen registration, taxonomic study, compilation in data repositories, and publication is the prime remedy for overall progress and development in fungal biodiversity research. The global scientific community has a huge scope to study a vast fungus diversity to upgrade present lead online data repositories.
Future repositories will emphasize greater community involvement, enabling researchers, citizen scientists, and enthusiasts to contribute data, share observations, and collectively build a more comprehensive understanding of fungi. The development of interactive data visualization and exploration will be used as user-friendly interfaces, and interactive visualization tools could be developed within repositories, making it easier for researchers to explore and analyze complex fungal datasets. Future repositories may focus on predictive modeling using machine learning and AI techniques. Researchers might use these models to predict fungal behavior, interactions, and responses to environmental changes. Through enhanced functional annotations, researchers could focus on exploring biological pathways and both ecological and environmental.
Current trends, limitations and future research in biocontrol
Biocontrol, also known as biological control, is a strategy for managing pests in an eco-friendly way and making a sustainable environment without any adverse impact on humans, plants, and animals. The use of beneficial microorganisms, such as fungi, bacteria, and viruses, as biocontrol agents against plant pathogens has become more prevalent. This approach harnesses the natural antagonistic interactions between beneficial microorganisms and plant pathogens to suppress disease development.
Current trends
The research on biocontrol of plant diseases, including postharvest diseases of fruits and vegetables, has undergone significant development over the past four decades. Biocontrol research was initiated in response to concerns regarding the potential carcinogenicity of numerous synthetic fungicides that were being utilised, and the anticipated withdrawal or prohibition of various classes of chemical fungicides. In fact, this has remained a driving force behind postharvest biocontrol research, as many governmental regulatory agencies have imposed increasingly stringent limits on the number of residues allowed on fruits and vegetables (Droby et al. 2016). Numerous studies have identified and screened various types of microorganisms from soil and other substrates and environments, which exhibit varying degrees of control efficacy against different plant diseases (Abdelhai et al. 2019; Ahima et al. 2019). Some of these microorganisms have been successfully applied in agricultural production, resulting in significant social and economic benefits (Hyde et al. 2019).
Due to the necessity of enhancing biocontrol activity through multiple strategies, the scope of biocontrol has been expanded to encompass natural products derived from plants or animals as well as compounds produced by other organisms that can be utilised for pathogen control (Li et al. 2022b). Integrated pest management (IPM) is gaining traction as a sustainable approach to managing plant diseases and pests. It involves combining various control methods, such as biological control, cultural practices, and judicious use of pesticides, to minimise the impact on the environment and human health while effectively controlling plant pathogens. Nanotechnology in plant pathology such as nanoparticles and nanoformulations is being explored for their potential in delivering biocontrol agents or plant defense elicitors more effectively. Nanotechnology offers precise and targeted delivery, increasing the efficiency of disease control and minimizing environmental impacts. High-resolution remote sensing and data analytics are being utilized to monitor and predict disease outbreaks. This digital agriculture technology allows for timely and targeted interventions, reducing the need for blanket applications of pesticides. Besides the strategy for plant disease control, numerous studies have been conducted on the mechanisms of action. Tjamos et al. (2004) demonstrated that Paenibacillus alvei K165 released chitinolytic compounds in an LB growth medium supplemented with glycol chitin, which suggests the potential of K165 to inhibit the growth of Botrytis cinerea causing grapevine bunch rot through extracellular chitinases.
With the advance in technology, the use of next generation sequencing technology in combination with universal primer sets, has provided insights into the global microbial community present on plant microbiome. For example, Abdelfattah et al. (2021) conducted a global analysis of the microbiome of ‘Royal Gala’ apples and investigated the composition and diversity of microbial communities present in various fruit tissues at harvest. The fungal and bacterial communities associated with apple fruit exhibit variations in composition and structure, which are significantly influenced by geographical location. This can be utilized to develop novel approaches for managing fruit quality and safety, as well as reducing losses caused by the establishment and proliferation of postharvest pathogens. Li et al. (2022b) provided current and future trends in the biocontrol of postharvest disease and discussed the use of marker-assisted selection (MAS) to identify markers for resistance to disease including the use of double-stranded RNA (dsRNA) to protect plants from phytopathogen.
Limitations
Biocontrol methods do not result in the accumulation of toxic residues within the environment. However, many biocontrol agents have a limited scope in eradicating diseases and pests as compared to chemicals as well as they are specific to certain diseases, pathogens, or environmental conditions. Therefore, the efficacy of microorganisms is restricted to certain types of diseases and pests with unique features, while other factors such as application techniques, duration of use, air quality, temperature, humidity levels, sunlight exposure, and storage conditions for each type of biological agent can also impact their effectiveness. The lack of broad-spectrum biocontrol agents poses a challenge in the search for effective biocontrol agents capable of controlling a wide range of plant pathogens. Ethical and regulatory considerations are also a limitation for studies and applications. The release of genetically modified organisms (GMOs) and other biocontrol agents may raise ethical concerns and regulatory challenges. Striking a balance between environmental safety and effective control is an ongoing issue.
Fungi develop resistance against fungicides and the overreliance on chemical fungicides has led to the development of resistance in many plant pathogens which is one of the major limitations in plant pathology. As a result, some pathogens have become more difficult to control using conventional chemical methods. Plasmopara viticola is an oomycete that causes Downy mildew. This is one of the major threats for grapevine production, due to the yield losses that are associated with severe disease epidemics (Gessler et al. 2011). Plasmopara viticola is an obligate parasite of grapevine (Vitis vinifera). Most V. vinifera cultivars are highly susceptible to the pathogen (Toffolatti et al. 2018a, 2020). This high susceptibility makes chemical control of the pathogen to ensure an adequate yield. Plasmopara viticola is a polycyclic pathogen, able to undergo numerous infection cycles during a single grapevine growing season. It overwinters as oospores in dead leaves on the vineyard floor and oospores produce a single macrosporangium with favourable weather conditions in spring. The zoospores infect the receptive grapevine tissues through stomata by rain or dew (Massi et al. 2021). Therefore, frequent fungicide applications are needed in vineyards located in areas with frequent rainfall and moderate temperatures during the grapevine growing season (Toffolatti et al. 2018b).
Future
Research into natural or synthetic compounds such as elicitors that can activate plant defenses against pathogens without causing harm to beneficial organisms could revolutionise disease control. Future research should prioritize the development of more environmentally friendly biological control methods. This includes exploring more efficient natural control agents and integrating diverse control strategies to decrease dependence on chemical pesticides, as well as mitigate environmental contamination and ecosystem degradation. Microbiome-based approaches are another future trend that will understand the plant microbiome and its influence on disease development. This can open new avenues for disease management. Manipulating the plant microbiome or introducing beneficial microbes could offer innovative biocontrol strategies (Afridi et al. 2022).
The development of biotechnology and gene editing technology is expected to provide new tools and methods for biological control. Gene editing techniques can be employed to enhance the attributes of natural predators or beneficial insects, fungi, and bacteria thereby enhancing their efficacy in pest and disease management. In addition, gene editing can also be used to develop crop varieties that are resistant to pests and diseases. For instance, CRISPR-Cas9 and Gene Editing hold promise for developing disease-resistant plant varieties. Further research in this area could lead to the development of more resilient crops (Rivero et al. 2022).
It is also important to seek more efficient methods to forecast and monitor the occurrence and distribution of pests and diseases. This will help agriculturalists and ecosystem managers in implementing strategies to mitigate economic losses and ecological harm during disease outbreaks. Further integrating precision agriculture techniques and artificial intelligence can enhance disease monitoring, prediction, and management at a finer scale (Talaviya et al. 2020). Research into the identification of virulent genes and their functions are needed to implement better control strategies (John et al. 2021). Future research should pay more attention to the service functions of biological control in ecosystems, with a particular focus on elucidating the fungal role in biodiversity maintenance and ecosystem stability (Liu et al. 2022). This will help us to better utilize ecosystem services for biological control while protecting and restoring ecosystem functions.
Current trends, limitations and future research in edible fungi and functional foods
Current trends in the introduction of new mushrooms to the table
Edible fungi are consumed as both food, and in traditional Chinese medicine as well as used as productive sources of various food products and chemicals (Alam et al. 2021). Ascomycetes, have played an important role in the production of food and medicinal compounds. According to Kirk et al. (2008) many ascomycetes producing substantial fruit bodies are edible. Many genera, such as Cordyceps, Morchella, Tuber and truffle-like fungi are used in medicine due to their bioactive substances (Wasser 2011; 2014). Ophiocordyceps sinensis has been used in Chinese medicine due to its biological activity (Lin and Li 2011). However, natural Ophiocordyceps sinensis yields have declined by over 90% over the past 25 years, leading to a rapid increase in price and reduced affordability (Feng et al. 2008). As a consequence of this scarcity, natural alternatives such as C. liangshanensis, C. militaris, and C. cicadicola have been promoted in markets (Yang et al. 2009). Morchella, is a popular, valuable and palatable mushroom which has been determined to be rich with bioactive components (Ali et al. 2011; Fu et al. 2013; Zhang et al. 2021a, b). Truffles and truffle-like fungi are also well-known revered taxa which are in high demand for both culinary as well potential use to offset climate change (Thomas and Büntgen 2019).
Mushrooms in basidiomycetes are also consumed, and their use is a significant part of folk medicine. The mushroom industry, including cultivation of mushrooms and key derivatives, has had a direct and observable benefit to human health, and as a result has grown rapidly (Lu et al. 2020). Parallel to this trend of industry growth and discovery, consumers of various nations have been increasingly exposed to using wild ingredients in novel gastronomic experiences, while considering sensory, cultural, social, and historical factors (Adapon 2008; Youssef and Spence 2021). Table 3 is a summary of how novel mushrooms are currently being used.
Current trends in edible fungi as meat replacements
There is a growing trend for consuming edible fungi for use as part of a sustainable and healthier diet for human muscle growth (Das et al. 2021). The majority of fungi used as food are macrofungi, or mushrooms, with fleshy fruiting bodies, distinct flavors and aromas, and strong nutritional qualities (Lesa et al. 2022). Importantly, mushrooms are largely made up of protein, and includes all nine of the essential amino acids that humans require but cannot synthesize (González et al. 2020; Yuwa-Amornpitak et al. 2020; Davila and Du 2023). Additionally, mushroom compounds contain antibacterial and antioxidant properties can extend the shelf life of meat products (Pérez Montes et al. 2021).
As a result of their high protein content and bioactive properties, consumers are more frequently usinge edible fungi as meat substitutes. Products of the mushroom group that are especially suitable for vegetarian diets and a good option for those who do not consume meat include Agaricus, Auricularia, Boletus, Flammulina, Grifola, Hericium, Lentinus, Phallus, Pleurotus, Tremella, Tuber and Volvariella (Prasad et al. 2015; Li et al. 2021a, b). Furthermore, in the development of more sustainable meat products, mushrooms have been used as an addition or supplement to a diet including meats (Table 4). In the contemporary world, consumers perceive meat substitutes as ethical, nutritious, and eco-friendly alternatives. There is, however, a need for further changes in consumer ideas, perceptions, as well as understanding and more clean-label healthy meat substitute innovations (Aschemann-Witzel et al. 2020). Furthermore, many studies are developing healthy meat replacements, and it remains necessary to demonstrate whether edible fungi can be used as a viable replacement for meat consumption.
Current trends in edible fungi as dietary supplement products and/or functional foods
Edible fungi are regarded as high-quality food products with nutritional and functional properties (Al-Obaidi et al. 2021). While fungi have been consumed by humans for centuries, their status as a key food with potential benefit to many aspects of human health has increased in recent years (Assemie and Abaya 2022). With the discovery of novel benefits of edible fungi, the global market for functional mushroom food is expected to continue to increase from the 2022 market value of USD 15.3 billion (Globe Newswire 2022). Fungal products have traditionally been used as functional foods, nutritional supplements, and sources of bioactive chemicals for a variety of medical purposes (Hyde et al. 2019). Recent research has shown that as next-generation ingredients in healthy food (Das et al. 2021), they can enhance health and immunity with their proteins, polysaccharides, terpenes and lipids. Critically, these components have been shown to have anticancer, antidiabetic, antihypercholesterolemia, anti-inflammatory, antiviral and immunomodulatory effects (Hamza et al. 2023).
In addition to medicinal qualities and use in muscle-building diets, edible fungi are suitable for the formation of low-caloric functional meals due to their low-fat content, high-quality proteins, dietary fiber and presence of bioactive compounds (Das et al. 2021). The edible fungi that are being most actively explored for use as functional foods include Agaricus bisporus, Hericium spp., Lentinus edodes, Monascus purpureus, Ophiocordyceps sinensis, and Pleurotus spp. (Gopalakrishnan et al. 2005; He 2018; Niego et al. 2021; Thongbai et al. 2015). The popularity of these edible mushrooms has resulted in increased attention, leading to breakthroughs in mushroom research and further promotion of various discoveries regarding their health-promoting benefits. (Kumar et al. 2021a, b).
Such research has led to unexpected discoveries including the benefits of edible fungi as a nutritional supplement (Bell et al. 2022). This research has not been ignored by the public, leading to an increase in their use as dietary supplements and functional foods. Furthermore, studies have suggested that the bioactive compounds found in mushrooms and other edible fungi can be extracted and used as supplements or adjuvant therapies for various health conditions (Valverde et al. 2015). The most optimal fungal supplements have been shown to provide critical daily advantages, such as improved brain function, a stronger immune system, greater energy, and better sleep (Healthline 2023; Lovering 2021).
Numerous edible fungi dietary supplements are on the market and are currently economically valuable (Bell et al. 2022). The inclusion of mushroom powder in food items increased consumer approval by improving taste and juiciness. In the aftermath of the SARS-CoV-2 (COVID-19) epidemic, people are focused on increasing their antioxidant activity through nutrition. Various bioactive substances from fungi, such as ergosterol, gallic acid, and cordycepin, have been shown to be important in preventing or lowering the severity of COVID-19 (Hamza et al. 2023). Therefore, using dried mushroom powder to develop added value coproducts is crucial (Hamza et al. 2023). Mushroom products are available in powder capsule, pill, and in liquid form. Due to their ease of use and extensive availability, capsules and tablets dominate the global market (Market Research Report 2023). The market value of functional fungi is promising and worth exploring.
Current trends in edible fungi are cosmeceuticals
The combination of cosmetics and pharmaceuticals is known as cosmeceuticals. Cosmeceuticals are topically applied products such as creams, lotions, and ointments that contain biologically active ingredients with benefits similar to prescription medications (Sharma 2011). According to Hyde et al. (2010), cosmetic products have substances that modify the skin’s biological function by providing nutrients and improving its appearance, radiance, texture, as well as having anti-aging activity. Natural ingredients are sought for the cosmetic industry due to their competitive efficacy and lower toxicity effects (Taofiq et al. 2016a, b). Many mushroom extracts have been reported with various bioactivities; see Table 5, 6 based on literature reviews (Taofiq et al. 2016a). The most common bioactivities were antityrosinase, anticollagenase, antihyaluronidase, and antielastase. However, more research may be needed to uncover further activity. Abd Razak et al. 2020 reported that the extract of four edible mushrooms, namely Pleurotus ostreatus, Ganoderma lucidum, Auricularia polytricha and Schizophyllum commune, were examined for antioxidant, antihyaluronidase and antityrosinase activity. Based on Hyde et al. (2010), some cosmetic products have extracts of Ophiocordyceps sinensis (Berk.) G.H. Sung, J.M. Sung, and Tremella fuciformis Berk. to increase the moisturizing effect. In the Center of Excellence for Fungal Research of Mae Fah Lung University, Chiang Rai, Thailand, The King’s Cordy Serum product is produced, which contains Cordyceps militaris (L.) Fr. extract and this extract has been shown to facilitate antiwrinkle effects and add moisture and antioxidants to the skin (Hyde et al. 2019).
The bamboo mushroom or Phallus indusiatus Vent. is an edible mushroom but can be eaten at the egg stage only (Habtemariam 2019). The bamboo mushroom has been studied and investigated regarding the cultivation of bamboo pulp, and the potential consumption of further products of the fungi (Thaisuchat et al. 2022). Bamboo mushroom extract has benefits for skin care; it has been developed into natural cosmetics in various forms (Razif and Fung 2021). Of note, extracts from the mucus cover of bamboo mushrooms were found to be rich in hyaluronic acid, the same substance which has been isolated from snail slime (Chumkhiao 2021). This extract has been shown to help the skin's nourishing properties, reduce irritation, and add moisture. Furthermore, gluconic acid of the bamboo mushroom extract helps exfoliate skin cells and strengthen collagen under the skin as well as moisturize the skin which helps reduce premature aging (Shivhare et al. 2013; Chumkhiao 2021). The skin care products of the extract of straw mushrooms (Volvariella sp.) are effective in reducing the pigmentation of melanin in the skin, making the skin appear radiant, reducing wrinkles, and adding moisture to the skin. Straw mushrooms contain polysaccharides that have the ability to add moisture to the skin and antioxidant properties (Sangthong et al. 2022). Wu et al. (2016) reported cosmetic products containing mushrooms and their function, various edible mushroom species, and substances that are now used in cosmetics or that are patented to be used in these products.
Limitations
Instead of using techniques that are damaging to the environment such as burning, mushrooms can be grown with relative ease using a variety of low value agricultural and forest by-products (Okuda 2022). As a result, mushroom production is proposed to be a lucrative industry, particularly in underdeveloped nations. Furthermore, mushroom cultivation is an example of cyclical agriculture that supports the sustainability of forests and the environment by preventing soil degradation (Kohsaka et al. 2021). Both the "bright" features of mushroom farming that promote sustainability and the "dark" ones that work against such impacts must be addressed. Unfortunately, despite its contribution to sustainability, there is a lack of worldwide awareness and stagnation in global uptake in mushroom cultivation (Elisashvili 2012). The explosive growth and expansion of the mushroom industry in China is a remarkable illustration of rural development fueled by bio-innovation and the dissemination of technology (Zhang et al. 2014). In addition to being a good example of a traditional recycling economy, sustainable agriculture, and forestry, it is also a great example of rural economic development and poverty relief (Zhang et al. 2014). Furthermore, it will be critical to exchange fundamental know-how and experience with facilities that deal with mushrooms (Okuda 2022).
The introduction of sustainability certification schemes is another effective strategy to highlight the environmental benefits of the mushroom industry (Okuda 2022). On the downside, the massive amount of mushroom spores produced by indoor cultivation endangers the health of growers, pollutes facilities, and erodes the natural genetic variety. Despite having both a "bright" and a "dark" side, the mushroom industry appears to be moving in the direction of greater environmental sustainability as a result of trends and new findings (Okuda 2022). Profits are only a small portion of the benefit to a nation's total mushroom production. Despite this, the working climate in this sector may not be sustainable due to the extreme competitiveness of prices. The working environment must also be taken care of if mushroom cultivation is to be sustainable. Although this type of issue could be overly generic, it should not be dismissed and should be acknowledged as a widespread difficulty in many sectors, not just mushroom farming. An efficient way to raise the perceived value of mushrooms internationally and ensure the profitability of mushroom production as a marketing strategy is to address sustainability issues associated with mushroom production as soon as possible (Okuda 2022). Additional enhancements in the production of currently cultivated species are also required, such as year-round cultivation of "seasonal" species and greater quality control. We can test a non-green (white agricultural) revolution in underdeveloped nations and the rest of the world using mushrooms, a special group of fungi. On a local, national, and international scale, mushrooms have the ability to significantly improve the socioeconomic and environmental conditions that affect human welfare (Chang and Wasser 2017a, b).
The health-promoting qualities of mushrooms are mainly attributable to their high content of fiber, particularly chitin and glucans (Stachowiak and Reguła, 2012; Jayachandran et al. 2017). The glucans, whether homo- or heteroglucans, play an important role in the structure of the fungal cell wall (Manzi and Pizzoferrato 2000; Ferreira et al. 2015; Vetter 2023). These are made up of sugar (mainly glucose) chains, and the linear or even branching structure is created by normal glycosidic linkages (Vetter 2023). The integrity of this triple helix structure and the unique configuration of β-glucans appear to be crucial for the biological actions of the molecule (Vetter 2023). Antitumor (anticarcinogenic) and immune-stimulating activities are two of glucans' key biological effects (Kothari et al. 2018; Wu et al. 2021a, b; Vetter 2023). Some glucan types may also have additional categories of benefits (such as anti-diabetic, antioxidant, etc.) linked with them (Vetter 2023). Moreover, the chitin molecule is made up of monomers of 1,4-N-acetyl-D-glucosamine chemically (Vetter 2023). All fungi in the Eukaryota kingdom typically have cell walls that contain chitin, which is an extremely resilient and water-insoluble component (Vetter and Siller 1991; Vetter 2007; 2023; Zhang et al. 2016). Because the needed chitinase activity in the digestive systems of animals and humans is quite low, the actual chitin content of a mushroom fruit body seems to be a significant determinant in its digestibility (Vetter 2023). However, there is another side to the significance of the chitin content in nutritional physiology: it contributes significantly to the dietary fiber fraction and, as a result, helps to meet the fiber needs of typical digestive processes (Vetter 2023).
Nevertheless, new initiatives are needed to elucidate the bioactive compounds yet to be identified in various mushrooms and their medicinal potential (Raghavendra et al. 2017). To promote preclinical and clinical research and ensure its safety, innovative toxicological studies are required (Elkhateeb 2020; Frost 2016; Jeitler et al. 2020). More research on mushrooms would be useful to find active components that contribute to functional food properties, health promotion, and disease prevention (Bell et al. 2022; Rousta et al. 2023). Although the study of medicinal mushrooms has recently rapidly advanced, more work remains. Regarding their pharmacological effects, many species are still unstudied or underappreciated. Identifying the compounds in the extracts, the metabolites responsible for their effects, their chemical characterization, and their mode of action are also of utmost importance (Venturella et al. 2021). The majority of studies that have been reported on the immunomodulatory actions were carried either in vivo or in vitro, which may not accurately reflect the function in the human body (Yin et al. 2021; Kozarski et al. 2023). Therefore, more research is still needed to understand the mechanism and use of in the body (Yin et al. 2021). Moreover, to maintain quality, it is essential to regulate, monitor and standardize the production of mushroom supplements along the supply chain, from cultivation to extraction and preparation of the commercial formulation (Venturella et al. 2021).
Future research
The current demand to maintain human well-being, and to focus on healthy lifestyles, is a key driver of fungi and fungus products research (Al-Obaidi et al. 2021; Hyde et al. 2019; Mapook et al. 2022).The medical, health, and beauty businesses have grown significantly due to the demand for herbal products, especially new drugs, dietary supplements, beverages, and cosmetics (Ekor 2014). Along with the increase in the aging society this has resulted in a preference for natural products. These help to build immunity and promote health. Global wellness institute predicts that in 2020, the market value of health and wellness products was worth US$ 4.4 trillion and is expected to increase to US$ 6 trillion by 2025 (Global wellness institute 2020). Therefore, research and development (R&D) of human welfare products from natural products such as plants, animals, microorganisms, and particularly fungi will be important.
Fungi are important producers of natural products (Alberti et al. 2017a, b; Sanchez and Demain 2017). However, only about 150,000 species of fungi and fungus-like taxa have been reported, although far more than 1.5 million species are estimated to exist (Gautam et al. 2022). Therefore, studies on the biodiversity of edible and other fungi and their uses are important as it should result in new mushrooms and mushroom products. Research to find new sources of fungi that produce novel medicinal compounds, studies on the genetic levels of biosynthetic gene expression to increase the efficiency of biological metabolites and research to verify health claims of consuming fungi, clinical trials, new innovations, and market research are areas for future study.
Mycoremediation of polluted soils—trends, limitations and future research
Fungal-based bioremediation, also known as mycoremediation, is defined as the degradation, removal, or isolation of environmental pollutants using fungi (Stamets 2005; Akhtar and Mannan 2020). Fungi have a broad range of enzymes capable of degrading complex inorganic and organic pollutants, making them ideal candidates for the remediation of contaminated soils. They have been successfully used for the removal of hydrocarbons, heavy metals, pesticides, industrial waste products, and even radioactive materials from soils (Singh et al. 2020). Mycoremediation holds great promise for further development, offering a broad range of cost-effective mechanisms for alleviating contaminated soil systems.
Current trends in this field include the use of genomics-based toolkits coupled with traditional culture methods for the screening of fungal species from different environments, with the aim of discovering fungi that have the capacity to be used in the bioremediation of contaminated soils (Park and Choi 2020; Akpasi et al. 2023). Fungi found growing in polluted soils or on complex substrates can be tested in the laboratory for their ability to break down, bio-transform, or hyperaccumulate various pollutants. An extension of this work is the use of nanoparticles derived from fungi which can be used for the targeted delivery of enzymes and the adsorption of contaminants in the soil, thus increasing the overall efficiency of the process (Alazaiza et al. 2021; Kumar et al. 2021a, b; Sharma et al. 2023). Another trend in mycoremediation is the investigation of fungi with the ability to hyperaccumulate heavy metals, and how to not only increase the scale and efficiency of hyperaccumulation but also the successful removal of pollutants after the fungi have assimilated the heavy metals (Hu et al. 2021; Borovička et al. 2022). In addition to the hyperaccumulation of heavy metals, the ability of certain fungal groups, such as white rot fungi, to remove xenobiotics from soils is of particular interest to restoration scientists as xenobiotics are finding their way into soils systems at alarming rates as a result of anthropogenic activities (Khatoon et al. 2021).
Limitations
Despite the positive applications of mycoremediation and the potential for these practices to effectively remove pollutants from soil systems, there are certain limitations that need to be addressed. Compared to bacterial-based bioremediation methods, fungi can be slow in degrading pollutants. Furthermore, the rate of fungal growth and the associated degradation of substrates is influenced by soil factors such as soil pH, temperature, moisture, and nutrient availability (Soares and Rousk 2019). These constraints will impact how effectively fungi can be used for bioremediation purposes. In addition, the large-scale use of fungi has not been widely tested and the lack of knowledge surrounding how to use fungi at scale for soil restoration efforts remains a key limitation in developing this technology (Chen et al. 2020). Lastly, in order to effectively use fungi for bioremediation purposes, there are numerous site-specific considerations that need to be addressed. Not only the type of pollutant, but also the environmental and biophysical conditions at each site will influence the success of the mycoremediation program.
Future
Mycoremediation of soil systems holds great promise for future applications, but it is currently limited in the scale of application due to knowledge gaps and constraints in our ability to effectively harness the full potential of the fungi involved. As such, there are numerous areas of research required to improve the mycoremediation process. At the field level, large-scale trials are needed to test how effectively fungi can be used for bioremediation in real-world scenarios. However, such trials should be performed in conjunction with laboratory-based research. An improved understanding of fungi and their life cycles will enable scientists to select and breed fungi for targeted use in specific environments or for certain types of pollutants. Network analyses of soil microorganisms can provide insight into how different microbes interact in the soil, allowing scientists to understand the synergistic behavior between fungi and other soil organisms, such as bacteria, and archaea (Matchado et al. 2021). This will enable the development of broader bioremediation strategies, potentially employing a range of organisms and thus enhancing the overall efficiency of the bioremediation process.
The genetic modification of fungi to enhance their capacity for pollutant degradation will be a key area of research in the future, including the upregulation of existing enzymes or the integration of enzymes from other organisms (Stein et al. 2018; Kumar et al. 2020). This work should be coupled with omics-based research. A deeper understanding of genomics, proteomics, transcriptomics, and metabolomics will allow scientists to better understand the bioremediation processes and, thus, potentially manipulate the genes and metabolic pathways involved.
Therefore, a multidisciplinary approach, combining taxonomists, molecular biologists and field practitioners, is required if we are to benefit from the use of fungi in large-scale bioremediation programs. The vast array of pollutants found in soils and the impact these have on soil biodiversity, agricultural productivity, and ecosystem health requires direct action on our behalf if soils are to be successfully restored. Fungi offer great potential to help in the restoration process, and future research programs should contribute to making mycoremediation a realistic strategy for use at the landscape level.
Conclusion and appraisal of papers of Special Issue
In this paper we have reviewed 16 topics that we consider as important current trends in fungal biology research. A further topic, not covered in this paper are changes in the lifestyles of fungi as it is the subject of this special issue. Fungi can occupy numerous life modes and their abilities to produce enzymes with multivarious functions outside the fungal mycelial allow them to cross the different lifestyle barriers. Many fungi are thought to have evolved from endophytic ancestors and this is the theme of the paper by Bhunjun and coauthors. Endophytes live symptomless within plant hosts. When the plant is stressed, they may become opportunistic pathogens or at leaf senescence they become active saprobes using their armoury of enzymes to degrade the dying plant cells. Chen and coauthors use genomics to show how opportunistic pathogens of Botryosphaeriaceae evolved from saprobic or endophytic ancestors. Necrotic pathogens may also kill the host and then digest the dead cells using enzymes also commonly found in endophytes and saprobes (Bucher et al. 2004, Shao et al. 2021). With their ability to degrade chitin and keratin, fungi can infect other fungi cells walls, living and dead insects, human and animal skin and hair. In this special Quan and coauthors examine the evolution of human pathogens in Chaetothyriales, Tang and coauthors the evolution of hair pathogens, Yu and coauthors discuss the fungal pathogens of cultivated mushrooms and Coleine and co-authors discussed the effect of harsh environments such as high and low temperatures, and industrial pollution on the lifestyle changes of black fungi based on comparative genomic analyses.
Data Availability
There are no datasets in this paper.
References
Abbacia A, Azzouz N, Bouznit Y (2014) A new copper doped montmorillonite modified carbon paste electrode for propineb detection. Appl Clay Sci 90:130–134
Abd Razak DL, Jamaluddin A, Abd Rashid NY, Sani NA, Abdul Manan M (2020) Assessment of cosmeceutical potentials of selected mushroom fruitbody extracts through evaluation of antioxidant, anti-hyaluronidase and anti-tyrosinase activity. Multidiscip Sci J 3:329–342
Abdelfattah A, Freilich S, Bartuv R, Zhimo VY, Kumar A, Biasi A, Salim S, Feygenberg O, Burchard E, Dardick C, Liu J, Khan A, Ellouze W, Ali S, Spadaro D, Torres R, Teixido N, Ozkaya O, Buehlmann A, Vero S, Mondino P, Berg G, Wisniewski M, Droby S (2021) Global analysis of the apple fruit microbiome: are all apples the same? Environ Microbiol 23:6038–6055
Abdelhai MH, Awad FN, Yang Q, Mahunu GK, Godana EA, Zhang H (2019) Enhancement the biocontrol efficacy of Sporidiobolus pararoseus Y16 against apple blue mold decay by glycine betaine and its mechanism. Biol Control 139:104079
Abeywickrama PD, Qian N, Jayawardena RS, Li Y (2023) Endophytic fungi in green manure crops; friends or foe? Mycosphere 14:1–106. https://doi.org/10.5943/mycosphere/14/1/1
Abram F (2015) Systems-based approaches to unravel multi-species microbial community functioning. CSBJ 3:24–32
Adapon J (2008) Culinary Art and Anthropology. Berg Oxford UK
Adebayo EA, Azeez MA, Alao MB, Oke AM, Aina DA (2021) Fungi as veritable tool in current advances in nanobiotechnology. Heliyon 7:e08480. https://doi.org/10.1016/j.heliyon.2021.e08480
Afridi MS, Javed MA, Ali S, De Medeiros FHV, Ali B, Salam A, Marc RA, Alkhalifah DHM, Selim S, Santoyo G (2022) New opportunities in plant microbiome engineering for increasing agricultural sustainability under stressful conditions. Front Plant Sci 13:899464
Agerer R, Rambold G (2021) DEEMY—an information system for characterization and determination of ectomycorrhizae. http://www.deemy.de. Accessed 13 Sept 2023
Aghdam SA, Brown AMV (2021) Deep learning approaches for natural product discovery from plant endophytic microbiomes. Environ Microbiome 16:6. https://doi.org/10.1186/s40793-021-00375-0
Ahima J, Zhang X, Yang Q, Zhao L, Tibiru AM, Zhang H (2019) Biocontrol activity of Rhodotorula mucilaginosa combined with salicylic acid against Penicillium digitatum infection in oranges. Biol Control 135:23–32
Ahluwalia V, Kumar J, Sisodia R, Shakil NA, Walia S (2014) Green synthesis of silver nanoparticles by Trichoderma harzianum and their bioefficacy evaluation against Staphylococcus aureus and Klebsiella pneumoinae. Ind Crops Prod 55:202–206. https://doi.org/10.1016/j.indcrop.2014.01.026
Ahmed S, Annu Ikram S, Yudha SS (2016) Biosynthesis of gold nanoparticles: a green approach. J Photochem Photobiol B 161:141–153
Ahrendt SR, Mondo SJ, Haridas S, Grigoriev IV (2022) MycoCosm, the JGI’s fungal genome portal for comparative genomic and multiomics data analyses. In: Microbial environmental genomics (MEG), New York, pp 271−291
Aime MC, Miller AN, Aoki T, Bensch K, Cai L, Crous PW, Hawksworth DL, Hyde KD, Kirk PM, Lücking R, May TW, Malosso E, Redhead SA, Rossman AY, Stadler M, Thines M, Yurkov AM, Zhang N, Schoch CL (2021) How to publish a new fungal species, or name, version 3.0 IMA Fungus 12:1–15
Akhtar N, Mannan MA (2020) Mycoremediation: expunging environmental pollutants. Biotech Rep 26:e00452. https://doi.org/10.1016/j.btre.2020.e00452
Akpasi SO, Anekwe IMS, Tetteh EK, Amune UO, Shoyiga HO, Mahlangu TP, Kiambi SL (2023) Mycoremediation as a potentially promising technology: current status and prospects—a review. Appl Sci 13:4978. https://doi.org/10.3390/app13084978
Alabdalall AH, ALanazi NA, Aldakeel SA, AbdulAzeez S, Borgio JF (2020) Molecular, physiological, and biochemical characterization of extracellular lipase production by Aspergillus niger using submerged fermentation. PeerJ. https://peerj.com/articles/9425/
Alam A, Agrawal K, Verma P (2021) Fungi and its by-products in food industry: an unexplored. https://doi.org/10.1007/978-981-16-1947-2_5
Alam N, Ki NY, Lee JS, Cho HJ, Lee TS (2012) Consequence of the antioxidant. Activities and tyrosinase inhibitory effects of various extract from the fruiting bodies of Pleurotus ferulae. Saudi J Biol Sci 19:111–118
Alazaiza MY, Albahnasawi A, Ali GA, Bashir MJ, Copty NK, Amr SSA, Al Maskari T (2021) Recent advances of nanoremediation technologies for soil and groundwater remediation: a review. Water 13:2186. https://doi.org/10.3390/w13162186
Alberti F, Foster GD, Bailey AM (2017a) Natural products from filamentous fungi and production by heterologous expression. Appl Microbiol Biotechnol 101:493–500. https://doi.org/10.1007/s00253-016-8034-2
Alberti F, Khairudin K, Venegas ER, Davies JA, Hayes PM, Willis CL, Bailey AM, Foster GD (2017b) Heterologous expression reveals the biosynthesis of the antibiotic pleuromutilin and generates bioactive semi-synthetic derivatives. Nat Comm 8:1831
Alemu D, Tafesse M, Mondal AK (2022) Mycelium-based composite: the future sustainable biomaterial. Int J Biomater 2022:1–12
Al-Hakkani MF (2020) Biogenic copper nanoparticles and their applications: a review. SN Applied Sciences 2:505
Ali H, Sannai J, Sher H, Rashid A (2011) Ethnobotanical profile of some plant resources in Malam Jabba valley of Swat, Pakistan. J Med Plant Res 5:4676–4687
Al-Karaki GN, Williams MJAPP (2021) Mycorrhizal mixtures affect the growth, nutrition, and physiological responses of soybean to water deficit 43:75
Al-Obaidi JR, Jambari NN, Ahmad-Kamil EI (2021) Mycopharmaceuticals and nutraceuticals: promising agents to improve human well-being and life quality. J Fungus 7:7. https://doi.org/10.3390/jof7070503
Amara AA, El-Baky NA (2023) Fungi as a source of edible proteins and animal feed. J Fungi 9:73
Anagnostakis SL (2001) American chestnut sprout survival with biological control of the chestnut-blight fungus population. Ecol Manag 152:225–233
Anderson PK, Cunningham AA, Patel NG, Morales FJ, Epstein PR, Daszak P (2004) Emerging infectious diseases of plants: pathogen pollution, climate change and agrotechnology drivers. Trends Ecol Evol 19:535–544
Andler SM, Goddard JM (2018) Transforming food waste: how immobilized enzymes can valorize waste streams into revenue streams. NPJ Sci Food 19:1–11
Angelini RMDM, Romanazzi G, Pollastro S, Rotolo C, Faretra F, Landi L (2019) New high-quality draft genome of the brown rot fungal pathogen Monilinia fructicola. Genome Biol Evol 11:2850–2855
Antonelli A, Smith RJ, Fry C, Simmonds MS (2020) State of the World’s Plants and Fungi (Doctoral dissertation, Royal Botanic Gardens (Kew) Sfumato Foundation
Arnaldi S, Muratorio A (2013) Nanotechnology, uncertainty and regulation. A guest editorial. Nano Ethics 7:173–175
Arun KB, Madhavan A, Tarafdar A, Sirohi R, Anoopkumar AN, Kuriakose LL, Awasthi MK, Binod P, Varjani S, Sindhu R (2023) Filamentous fungi for pharmaceutical compounds degradation in the environment. A sustainable approach. Environ Technol Innov 31:103182
Aschemann-Witzel J, Gantriis RF, Fraga P, Perez-Cueto FJA (2020) Plant-based food and protein trend from a business perspective: markets, consumers, and the challenges and opportunities in the future. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2020.1793730
Assemie A, Abaya G (2022) The Effect of Edible Mushroom on Health and Their Biochemistry. International J Microbiol 2022:8744788. https://doi.org/10.1155/2022/8744788
Atallah ZK, Subbarao KV (2012) Population biology of fungal plant pathogens. Plant fungal pathogens: methods and protocols pp 333–363
Atanasov AG, Zotchev SB, Dirsch VM, Supuran CT (2021) Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov 20:200-216
Averill C, Hawkes CV (2016) Ectomycorrhizal fungi slow soil carbon cycling. Ecol 19:937–947
Aylward J, Steenkamp ET, Dreyer LL, Roets F, Wingfield BD, Wingfield MJ (2017) A plant pathology perspective of fungal genome sequencing. IMA Fungus 8:1–15
Azelee NIW, Manas NHA, Dailin DJ, Ramli ANM, Shaarani SM (2020) Biological treatment of agro-industrial waste. In: Valorisation of agro-industrial residues–volume I: Biological Approaches, Eds., ZA Zakaria, R Boopathy, JR Dib, pp 59–79
Azin M, Moravej R, Zareh D (2007) Production of xylanase by Trichoderma longibrachiatum on a mixture of wheat bran and wheat straw: optimization of culture condition by Taguchi method. Enzyme Microb Technol 40: 801–805
Azmath P, Baker S, Rakshith D, Satish S (2016) Mycosynthesis of silver nanoparticles bearing antibacterial activity. Saudi Pharm J 24:140–146
Azodi CB, Tang J, Shiu SH (2020) Opening the Black Box: interpretable machine learning for geneticists. Trends Genet 36:442–455. https://doi.org/10.1016/j.tig.2020.03.005
Bae JT, Sim GS, Lee DH, Lee BC, Pyo HB, Choe TB, Yun JW (2005) Production of exopolysaccharide from mycelial culture of Grifola frondosa and its inhibitory effect on matrix metalloproteinase-1 expression in UV-irradiated human dermal fibroblasts. FEMS Microbiol 251: 347–354
Bafghi MH, Darroudi M, Zargar M, Zarrinfar H, Nazari R (2021) Biosynthesis of selenium nanoparticles by Aspergillus flavus and Candida albicans for antifungal applications. Micro Nano Lett 16:656–669
Bahrulolum H, Nooraei S, Javanshir N, Tarrahimofrad H, Mirbagheri VS, Easton AJ, Ahmadian G (2021) Green synthesis of metal nanoparticles using microorganisms and their application in the agrifood sector. J Nanobiotechnol 19:86. https://doi.org/10.1186/s12951-021-00834-
Bakar NA, Karsani SA, Alias SA (2020) Fungal survival under temperature stress: A proteomic perspective. PeerJ 8:e10423. https://doi.org/10.7717/peerj.10423
Baldrian P, Větrovský T, Lepinay C, Kohout P (2021) High-throughput sequencing view on the magnitude of global fungal diversity. Fungal Divers 19:1–9
Baldrian P, Bell-Dereske L, Lepinay C, Vetrovsky T, Kohout P (2022a) Fungal communities in soils under global change. Stud Mycol 1–24
Baldrian P, Větrovský T, Lepinay C, Kohout P (2022b) High-throughput sequencing view on the magnitude of global fungal diversity. Fungal Divers 114:539–547
Baldrian P, Lopez-Mondejar R, Kohout P (2023) Forest microbiome and global change. Nat Rev Microbiol 21:487–501
Ball B, Langille M, Geddes-McAlister J (2020) Fun(gi)omics: advanced and diverse technologies to explore emerging fungal pathogens and define mechanisms of antifungal resistance. MBio 11:e01020–20
Banerjee DK, Das AK, Banerjee R, Pateiro M, Nanda PK, Gadekar YP, Biswas S, McClements DJ, Lorenzo JM (2020) Application of enoki mushroom (Flammulina velutipes) stem wastes as functional ingredients in goat meat nuggets. Foods 9:432. https://doi.org/10.3390/foods9040432
Bansal R, Singh DV, Joshi LM (1984) Effect of karnal bunt pathogen [Neovossia indica (Mitra) Mundkur] on weight and viability of wheat seed. Indian J Agric Sci 54:663–666
Barber AE, Riedel J, Sae-Ong T, Kang L, Brabetz W, Panagiotou G, Deising HB, Kurzai O (2020) Effects of agricultural fungicide use on Aspergillus fumigatus abundance, antifungal susceptibility, and population structure. mBio 11:e02213–20
Basidiochecklist (2023) http://www.basidiochecklist.info. Accessed 27 Aug 2023
Bayda S, Adeel M, Tuccinardi T, Cordani M, Rizzolio F (2020) The history of nanoscience and nanotechnology: from chemical–physical applications to nanomedicine. Molecules 25:112. https://doi.org/10.3390/molecules25010112
Begum N, Qin C, Ahanger MA, Raza S, Khan MI, Ashraf M, Ahmed N, Zhang L (2019) Role of arbuscular mycorrhizal fungi in plant growth regulation: implications in abiotic stress tolerance. Front Plant Sci 10:1068
Bell V, Silva CRPG, Guina J, Fernandes TH (2022) Mushrooms as future generation healthy foods. Front Nutr 9:1050099. https://doi.org/10.3389/fnut.2022.1050099
Beltrán-Flores E, Pla-Ferriol M, Martínez-Alonso M, Gaju N, Blánquez P, Sarrà M (2022) Fungal bioremediation of agricultural wastewater in a long-term treatment: biomass stabilization by immobilization strategy. J Hazard Mater 439:129614
Bennett J, Klich M (2003) Chotoxins. Clin. Microbiol. Rev 16:497–516
Benson DA, Clark K, Karsch-Mizrachi I, Lipman DJ, Ostell J, Sayers EW (2015) GenBank. Nucleic Acids Res 43:30–35
Berbee ML, James TY, Strullu-Derrien C (2017) Early diverging fungi: diversity and impact at the dawn of terrestrial life. Annu Rev Microbiol 71:41–60
Berman J, Krysan DJ (2020) Drug resistance and tolerance in fungi. Nat Rev Microbiol. 18:319-331
Bérubé JA, Gagné PN, Ponchart JP, Tremblay ÉD, Bilodeau GJ (2018) Detection of Diplodia corticola spores in Ontario and Québec based on High Throughput Sequencing (HTS) methods. Canadian Plant Pathol J 40:378-386
Besserer A, Puech-Pagès V, Kiefer P, Gomez-Roldan V, Jauneau A, Roy S, Portais JC, Roux C, Bécard G, Séjalon-Delmas N (2006) Strigolactones stimulate arbuscular mycorrhizal fungi by activating mitochondria. PLosS Biol 4:1239-1247
Bhardwaj D, Ansari MW, Sahoo RK, Tuteja N (2014) Biofertilizers function as key player in sustainable agriculture by improving soil fertility, plant tolerance and crop productivity. Microbial Cell Factories 13:1
Bhat RG, Browne GT (2010) Specific detection of Phytophthora cactorum in diseased strawberry plants using nested polymerase chain reaction. Plant Pathol J 59:121–129
Bhunjun CS, Dong Y, Jayawardena RS, Jeewon R, Phukhamsakda C, Bundhun D, Hyde KD, Sheng J (2020) A polyphasic approach to delineate species in Bipolaris. Fungal Divers 102:225–256
Bhunjun CS, Phukhamsakda C, Jayawardena RS, Jeewon R, Promputtha I, Hyde KD (2021) Investigating species boundaries in Colletotrichum. Fungal Divers 107:107–127
Bhunjun CS, Niskanen T, Suwannarach N, Wannathes N, Chen YJ, McKenzie EHC, Maharachchikumbura SSN, Buyck B, Zhao CL, Fan YG, Zhang JY, Dissanayake AJ, Marasinghe DS, Jayawardena RS, Kumla J, Padamsee M, Chen YY, Liimatainen K, Ammirati JF, Phukhamsakda C, Liu JK, Phonrob W, Randrianjohany E, Hongsanan S, Lumyong S (2022) The numbers of fungi: are the most speciose genera truly diverse?. Fungal Divers 114: 387-462
Biala E, Ostermann M (2022) Mycostructures—growth-driven fabrication processes for architectural elements from mycelium composites. Archit Struct Constr 2:509–519
Bills GF, Gloer JB (2016) Biologically active secondary metabolites from the fungi. Microbiol Spectrum 4:1–32
Bisht N, Phalswal P, Khanna PK (2022) Selenium nanoparticles: a review on synthesis and biomedical applications. Mater Adv 3:1415–1431
Bitting S, Derme T, Lee J, Van Mele T, Dillenburger B, Block P (2022) Challenges and opportunities in scaling up architectural applications of mycelium-based materials with digital fabrication. Biomimetics 7:1–22
Blackwell M (2011) The Fungi: 1, 2, 3… 5.1 million species?. Am J Bot 98:426-438
Blalock HG, Georg LK, Durieux WT (1973) Encephalitis in turkey poults due to Dactylaria (Diplorhinotrichum) gallopava, a case report and its experimental reproduction. Avian Dis 17:179-204
Bilska K, Kulik T, Ostrowska-Kołodziejczak A, Buśko M, Pasquali M, Beyer M, Baturo-Cieśniewska A, Juda M, Załuski D, Treder K, Denekas J (2018) Development of a highly sensitive FcMito qPCR assay for the quantification of the toxigenic fungal plant pathogen Fusarium culmorum. Toxins 10:211
Boekhout T, Aime MC, Begerow D, Gabaldón T, Heitman J, Kemler M, Khayhan K, Lachance MA, Louis EJ, Sun S, Vu D, Yurkov A (2021) The evolving species concepts used for yeasts: from phenotypes and genomes to speciation networks. Fungal Divers 109:27–55
Boekhout T, Amend AS, El Baidouri F, Gabaldón T, Geml J, Mittelbach M, Robert V, Tan CS, Turchetti B, Vu D, Wang QM, Yurkov A (2022) Trends in yeast diversity discovery. Fungal Divers 114:491–537
Boettger D, Hertweck C (2013) Molecular diversity sculpted by fungal PKS–NRPS hybrids. Chem Bio Chem14:28-42
Boland MJ, Rae AN, Vereijken JM, Meuwissen MP, Fischer AR, van Boekel MA, Rutherfurd SM, Gruppen H, Moughan PJ, Hendriks WH (2013) The future supply of animal-derived protein for human consumption. Trends Food Sci Technol 29:62–73
Bonde MR, Peterson GL, Schaad NW, Smilanick JL (1997) Karnal bunt of wheat. Plant Dis 81:1370-1377
Bonifaz A, Davoudi MM, de Hoog GS, Padilla-Desgarennes C, Vázquez-González D, Navarrete G, Meis JF, Badali H (2013) Severe disseminated phaeohyphomycosis in an immunocompetent patient caused by Veronaea botryosa. Mycopathologia 175:497–503
Boonham N, Kreuze J, Winter S, van der Vlugt R, Bergervoet J, Tomlinson J, Mumford R. (2014) Methods in virus diagnostics: from ELISA to next generation sequencing. Virus Res 186:20-31
Boonmee S, Wanasinghe DN, Calabon MS, Huanraluek N, Chandrasiri SKU, Jones GEB, Rossi W, Leonardi M, Singh SK, Rana S, Singh PN, Maurya DK, Lagashetti AC, Choudhary D, Dai YC, Zhao CL, Mu YH, Yuan SH, He SH, Phookamsak R, Jiang HB, Martín MP, Dueñas M, Telleria MT, Hyde KD (2021) Fungal diversity notes 1387–1511: taxonomic and phylogenetic contributions on genera and species of fungal taxa. Fungal Divers 111:1–335
Borovička J, Braeuer S, Walenta M, Hršelová H, Leonhardt T, Sácký J, Goessler W (2022) A new mushroom hyperaccumulator: cadmium and arsenic in the ectomycorrhizal basidiomycete Thelephora enicillata. Sci Total Environ 826:154227. https://doi.org/10.1016/j.scitotenv.2022.154227
Borse V, Kaler A, Banerjee UC (2015) Microbial synthesis of platinum nanoparticles and evaluation of their anticancer activity. IJETEE 11:26–31
Bosch TC, Guillemin K, McFall‐Ngai M (2019) Evolutionary “experiments” in symbiosis: the study of model animals provides insights into the mechanisms underlying the diversity of host–microbe interactions. Bio Essays 41:1800256
Bouhired S, Weber M, Kempf-Sontag A, Keller NP, Hoffmeister D (2007) Accurate prediction of the Aspergillus nidulans terrequinone gene cluster boundaries using the transcriptional regulator LaeA. Fungal Genet Biol 44:1134-1145
Bourzama G, Ouled-Haddar H, Marrouche M, Aliouat A (2021) Iron uptake by fungi isolated from arcelor mittal-annaba-in the northeast of Algeria. Braz J Poult Sci 23. https://doi.org/10.1590/1806-9061-2020-1321
Brennan JP, Warham EJ, Byerlee D, Hernandez‐Estrada J (1992) Evaluating the economic impact of quality‐reducing, seed‐borne diseases: lessons from Karnal bunt of wheat. Agric Econom 6:345-352
Brewer MT, Turner AN, Brannen PM, Cline WO and Richardson EA (2014) Exobasidium maculosum, a new species causing leaf and fruit spots on blueberry in the southeastern USA and its relationship with other Exobasidium spp. parasitic to blueberry and cranberry. Mycologia 106:415-423
Bridle H, Desmulliez M (2021) Chapter 7–biosensors for the detection of waterborne pathogens. In: Bridle H (ed) Waterborne pathogens, 2nd edn. Academic Press, New York, pp 189–235. https://doi.org/10.1016/B978-0-444-64319-3.00007-1
Brundrett MC, Tedersoo L (2018) Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol 220:1108-1115
Bruno M, Dewi IM, Matzaraki V, Ter Horst R, Pekmezovic M, Rösler B, Groh L, Röring RJ, Kumar V, Li Y, Carvalho A (2021) Comparative host transcriptome in response to pathogenic fungi identifies common and species-specific transcriptional antifungal host response pathways. Comput Struct Biotechnol J 19:647–663
Bucher VVC, Hyde KD, Pointing SB, Reddy CA (2004) Production of wood decay enzymes, mass loss and lignin solubilization in wood by marine ascomycetes and their anamorphs. Fungal Divers 15:1–14
Butler MS, Henderson IR, Capon RJ, Blaskovich MAT (2023) Antibiotics in the clinical pipeline as of December 2022. J Antibiot 76:431–473. https://doi.org/10.1038/s41429-023-00629-8
Byrd JB, Greene AC, Prasad DV, Jiang X, Greene CS (2020) Responsible, practical genomic data sharing that accelerates research. Nat Rev Genet 21:615–629
Caballero-Sanchez L, Lázaro-Mixteco PE, Vargas-Tah A, Castro-Montoya AJ (2023) Pilot-scale bioethanol production from the starch of avocado seeds using a combination of dilute acid-based hydrolysis and alcoholic fermentation by Saccharomyces cerevisiae. Microb Cell Factories 22:1–10
CABI Culture Collection (2023). https://www.cabi.org/products-and-services/bioscience-services/microorganism-supply-services/. Accessed 13 Sept 2023
Calabon MS, Hyde KD, Jones EBG, Bao DF, Bhunjun CS, Phukhamsakda C, Shen HW, Gentekaki E, Al Sharie AH, Barros J, Chandrasiri KSU, Hu DM, Hurdeal VG, Rossi W, Valle LG, Zhang H, Figueroa M, Raja HA, Seena S, Song HY, Dong W, El-Elimat T, Leonardi M, Li Y, Li YJ, Luo ZL, Ritter CD, Strongman DB, Wei MJ, Balasuriya A (2023) Freshwater fungal biology. Mycosphere 14:195–413
Calderón-Parra J, Mills-Sanchez P, Moreno-Torres V, Tejado-Bravo S, Romero-Sánchez I, Balandin-Moreno B, Calvo-Salvador M, Portero-Azorín F, García-Masedo S, Muñez-Rubio E, Ramos-Martinez A, Fernández-Cruz A (2022) COVID-19-associated pulmonary aspergillosis (CAPA): risk factors and development of a predictive score for critically ill COVID-19 patients. Mycoses 65:541–550
Cannon PF, Damm U, Johnston PR, Weir BS (2012) Colletotrichum: current status and future directions. Stud Mycol 73:181–213
Cao L, Zhou Z, Niu S, Cao C, Li X, Shan Y (2018) Positive-charge functionalized mesoporous silica nanoparticles as nanocarriers for controlled 2, 4-dichlorophenoxy acetic acid sodium salt release. J Agric Food Chem 66:6594–6603
Caramalho R, Tyndall JDA, Monk BC, Larentis T, Lass-Flörl C, Lackner M (2017) Intrinsic short-tailed azole resistance in mucormycetes is due to an evolutionary conserved amino acid substitution of the lanosterol 14α-demethylase. Sci Rep7:1–10
Carini P, Delgado-Baquerizo M, Hinckley ELS, Holland‐Moritz H, Brewer TE, Rue G, Fierer N (2020) Effects of spatial variability and relic DNA removal on the detection of temporal dynamics in soil microbial communities. MBio 11:10-1128
Carnielli-Queiroz L, Fernandes PMB, Fernandes AAR, Ventura JA (2019) A rapid and reliable method for molecular detection of Fusarium guttiforme, the etiological agent of pineapple fusariosis. Braz Arc Biol Technol 62: e19180591
Carpouron JE, de Hoog GS, Gentekaki E, Hyde KD (2022) Emerging animal-associated fungal diseases. J Fungi 8:611. https://doi.org/10.3390/jof8060611
Case NT, Berman J, Blehert DS, Cramer RA, Cuomo C, Currie CR, Ene IV, Fisher MC, Fritz-Laylin LK, Gerstein AC, Glass NL, Gow NAR, Gurr SJ, Hittinger CT, Hohl TM, Iliev ID, James TY, Jin H, Klein BS, Kronstad JW, Lorch JM, McGovern V, Mitchell AP, Segre JA, Shapiro RS, Sheppard DC, Sil A, Stajich JE, Stukenbrock EE, Taylor JW, Thompson D, Wright GD, Heitman J, Cowen LE (2022) The future of fungi:threats and opportunities. G3 (Bethesda) 12:jkac224. https://doi.org/10.1093/g3journal/jkac224
Castro-Longoria E, Moreno-Velásquez SD, Vilchis-Nestor AR, Arenas-Berumen E, Avalos-Borja M (2012) Production of platinum nanoparticles and nanoaggregates using Neurospora crassa. J Microbiol Biotechnol 22:1000–1004
CBS Fungus Collection (2023) https://wi.knaw.nl. Accessed 2 Sept 2023
Cerimi K, Akkaya KC, Pohl C, Schmidt B, Neubauer P (2019) Fungi as source for new bio-based materials: a patent review. Fungal Biol 6:1–10
Cha MH, Heo JY, Lee C, Lo YM, Moon B (2014) Quality and sensory characterization of white jelly mushroom (Tremella Fuciformis) as a meat substitute in pork patty formulation. J Food Process Preserv 38:2014-2019. https://doi.org/10.1111/jfpp.12178
Chaerun SK, Prabowo BA, Winarko R (2022) Bionanotechnology: the formation of copper nanoparticles assisted by biological agents and their applications as antimicrobial and antiviral agents. Environ Nanotechnol Monit Manag 18:100703
Chakraborty A, Fernando LD, Fang W, Widanage MCD, Wei P, Jin C, Fontaine T, Latgé JP, Wang T (2021) A molecular vision of fungal cell wall organization by functional genomics and solid-state NMR. Nat Commun 12:6346
Chang ST, Wasser SP (2017a) The cultivation and environmental impact of mushrooms. In: Oxford research encyclopedia of environmental science
Chang ST, Wasser SP (2017b) The cultivation and environmental impact of mushrooms. Printed from the oxford Research Encyclopaedia, Environmental Science (c) Oxford University Press, Oxford, pp 43
Chaverra-Muñoz L, Hüttel S (2022) Optimization of the production process for the anticancer lead compound illudin M: process development in stirred tank bioreactors. Microb Cell Fact 21:145. https://doi.org/10.1186/S12934-022-01870-W
Cheek M, Lughadha NE, Kirk P, Lindon H (2020) New scientific discoveries: plants and fungi. Plants, People, Planet 2:371–388
Chen W, Jiao S, Li Q, Du N (2020) Dispersal limitation relative to environmental filtering governs the vertical small‐scale assembly of soil microbiomes during restoration. J Appl Ecol 57:402–412. https://doi.org/10.1111/1365-2664.13533
Chen YP, Su PW, Hyde KD, Maharachchikumbura SSN (2023a) Phylogenomics and diversification of Sordariomycetes. Mycosphere 14:414–451. https://doi.org/10.5943/mycosphere/14/1/5
Chen YP, Su PW, Stadler M, Xiang R, Hyde KD, Tian WH, Maharachchikumbura SSN (2023b) Beyond observation: genomic traits and machine learning algorithms for predicting fungal lifestyles. Mycosphere 14(1):1530–1563. https://doi.org/10.5943/mycosphere/14/1/17
Cherry JM, Adler C, Ball C, Chervitz SA, Dwight SS, Hester ET, Jia Y, Juvik G, Roe TY, Schroeder M, Weng S, Botstein D (1998) SGD: saccharomyces genome database. Nucleic Acid Res 26:73–79
Chethana KWT, Peng JB, Xing QK, Zhang W, Hyde KD, Zhao W, Yan J (2020) LtEPG1, a secretory endopolygalacturonase protein, regulates the virulence in Lasiodiplodia theobromae on Vitis vinifera. Phytopathology 110:1727–1736
Chethana KWT, Manawasinghe IS, Hurdeal VG, Bhunjun CS (2021) What are fungal species and how to delineate them?. Fungal Divers 109:1–25
Chien CC, Tsai ML, Chen CC, Chang SJ, Tseng CH (2008) Effects on tyrosinase activity by the extracts of Ganoderma lucidum and related mushrooms. Mycopathologia 166:117–120
Chowdhary A, Kathuria S, Xu J, Meis JF (2013) Emergence of azole-resistant Aspergillus fumigatus strains due to agricultural azole use creates an increasing threat to human health. PLoS Pathogens 9(10): e1003633
Christensen M (1989) A view of fungal ecology. Mycologia 81:1–19
Chumkhiao S (2021) Comparative research report on the active ingredient content in the oviparous agar of bamboo mushrooms, Thai and Chinese cultivars, short and long skirts (white). Received research grants from Suranaree University of Technology (In Thai)
Coleine C, Stajich JE, de Los Ríos A, Selbmann L (2021) Beyond the extremes: rocks as ultimate refuge for fungi in drylands. Mycologia 113:108-133
Colla LM, Primaz AL, Benedetti S, Loss RA, Lima MD, Reinehr CO, Bertolin TE, Costa JAV (2016) Surface response methodology for the optimization of lipase production under submerged fermentation by filamentous fungi. Braz J Microbiol 47:461–467
Collinge DB, Jensen B, Jørgensen HJ (2022) Fungal endophytes in plants and their relationship to plant disease. Curr Opin Microbiol 69:102177
Conrado R, Gomes TC, Roque GSC, De Souza AO (2022) Overview of bioactive fungal secondary metabolites: cytotoxic and antimicrobial compounds. Antibiotics 11:1604
Constantin ME, Fokkens L, De Sain M, Takken FL, Rep M (2021) Number of candidate effector genes in accessory genomes differentiates pathogenic from endophytic Fusarium oxysporum strains. Front Plant Sci 12:761740
Copetti MV (2019) Fungi as industrial producers of food ingredients. Curr Opin Food Sci 25:52–56
Cornell M, Alam I, Soanes D, Wong H, Rattray M, Hubbard SJ, Talbot N, Lings BJ (2015) e-Fungi:an e-science infrastructure for comparative functional genomics in fungal species. In: Proceedings of 4th UK e-science all hands meeting AHM, pp 20–22
Cortés-Maldonado L, Marcial-Quino J, Gómez-Manzo S, Fierro F, Tomasini A (2020) A method for the extraction of high quality fungal RNA suitable for RNAseq. J Microbiol Methods 170:10585
Cotton TEA, Fitter AH, Miller RM, Dumbrell AJ, Helgason T (2015) Fungi in the future: interannual variation and effects of atmospheric change on arbuscular mycorrhizal fungal communities. New Phytol 205:1598-1607
Crandall SG, Gold KM, Jiménez-Gasco MDM, Filgueiras CC, Willett DS (2020) A multi-omics approach to solving problems in plant disease ecology. PLoS ONE 15:e0237975. https://doi.org/10.1371/journal.pone.023797
Crous PW, Gams W, Stalpers JA, Robert V, Stegehuis G (2004) Mycobank:an online initiative to launch mycology into the 21st century. Stud Mycol 50:19–22
Crous PW, Boers J, Holdom D, Steinrucken TV, Tan YP, Vitelli JS, Shivas RG, Barrett M, Boxshall AG, Broadbridge J, Larsson E, Lebel T, Pinruan U, Sommai S, Alvarado P, Bonito G, Decock CA, De la Peña-Lastra S, Delgado G, Houbraken J, Maciá-Vicente JG, Raja HA, Rigueiro-Rodríguez A, Rodríguez A, Wingfield MJ, Adams SJ, Akulov A, AL-Hidmi T, Antonín V, Arauzo S, Arenas F, Armada F, Aylward J, Bellanger JM, Berraf-Tebbal A, Bidaud A, Boccardo F, Cabero J, Calledda, F, Corriol G, Crane JL, Dearnaley JDW, Dima B, Dovana F, Eichmeier A, Esteve-Raventós F, Fine M, Ganzert L, García D, Torres-Garcia D, Gené J, Gutiérrez A, Iglesias P, Istel Ł, Jangsantear P, Jansen GM, Jeppson M, Karun NC, Karich A, Khamsuntorn P, Kokkonen K, Kolarík M, Kubátová A, Labuda R, Lagashetti AC, Lifshitz N, Linde C, Loizides M, Luangsa-ard JJ, Lueangjaroenkit P, Mahadevakumar S, Mahamedi AE, Malloch DW, Marincowitz S, Mateos A, Moreau PA, Miller AN, Molia A, Morte A, Nebesářová J, Nigrone E, Nuthan BR, Oberlies NH, Pepori AL, Rämä T, Rapley D, Reschke K, Robicheau BM, Roets F, Roux J, Saavedra M, Sakolrak B, Santini A, Ševčíková H, Singh PN, Singh SK, Somrithipol S, Spetik M, Sridhar KR, Starink-Willemse M, Taylor VA, van Iperen AL, Vauras J, Walker AK, Wingfield BD, Yarden O, Cooke AW, Manners AG, Pegg KG, Groenewald JZ (2022) Fungal planet description sheets: 1383–1435. Pers Mol Phylogen Evol 48:261–371
Cyberliber (2023). http://www.cybertruffle.org.uk. Accessed 27 Aug 2023
da Cunha BR, Fonseca LP, Calado CRC (2019) Antibiotic discovery: where have we come from, where do we go? Antibiotics 8:45. https://doi.org/10.3390/antibiotics8020045
Dandapat S, Sinha MP (2015) Antioxidant and anti-Inflammatory activity of Pleurotus tuber-regium (Rumph. ex Fr.) singer. Adv Biol Res 9:140–145
Das AK, Nanda PK, Dandapat P, Bandyopadhyay S, Gullón P, Sivaraman GK, McClements DJ, Gullón B, Lorenzo JM (2021) Edible mushrooms as functional ingredients for development of healthier and more sustainable muscle foods: a flexitarian approach. Molecules 26:2463. https://doi.org/10.3390/molecules26092463
Dasari T, Deng H, McShan D, Yu H (2014) Nanosilver-based antibacterial agents for food safety, food poisoning: outbreaks, bacterial sources and adverse health effects. NOVA Science Publishers, Hauppauge, pp 35–62
Dashtban M, Schraft H, Syed TA, Qin W (2010) Fungal biodegradation and enzymatic modification of lignin. Int J Biochem Mol Biol 1:36
Davila M, Du X (2023) Primary exploration of mushroom protein hydrolysis and cooking impact on the protein amino acid profiles of Agaricus bisporus and Lentinula edodes mushrooms. Int J Gastron Food Sci 32:100710. https://doi.org/10.1016/j.ijgfs.2023.100710
Davison J, Moora M, Semchenko M, Adenan SB, Ahmed T, Akhmetzhanova AA (2021) Temperature and pH define the realised niche space of arbuscular mycorrhizal fungi. New Phytol 231:763–776
Deharveng L, Bedos A (2019) Biodiversity in the tropics. In: Encyclopedia of caves. Academic Press, New York, pp 146–162
De Hoog GS, Guarro J, Gené J, Ahmed S, Al-Hatmi AMS, Figueras MJ, Vitale RG (2020) Atlas of clinical fungi, 4th ed. Foundation Atlas of Clinical Fungi, Hilversum p 1599
De Hoog GS, Walsh TJ, Kwon-Chung KJ, Borman A, Vitale RG, Ahmed SA (2023) A conceptual framework and recommendations for nomenclatural stability in medically important fungi: a proposed global consensus guideline for fungal name change supported by ISHAM, ASM, ECMM, MSGERC, MMSA, FDLC, CLSI, EUCAST, EFISG/ESCMID, and CAP* J Clin Microbiol (in press)
Delvendahl N, Dienel HL, Meyer V, Langen N, Zimmermann J, Schlecht M (2023) Narratives of fungal-based materials for a new bioeconomy era. Innovation 36:96–106
Deng H, Zhang Y, Yu H (2018) Nanoparticles considered as mixtures for toxicological research. J Environ Sci Health C 36:1–20
Depotter JR, Rodriguez-Moreno L, Thomma BP, Wood TA (2017) The emerging British Verticillium longisporum population consists of aggressive Brassica pathogens. Phytopathology 107:1399-1405
Deshmukh R, Khardenavis AA, Purohit HJ (2016) Diverse metabolic capacities of fungi for bioremediation. Indian J Microbiol 56:247–264
Deshmukh SK, Gupta MK, Prakash V, Reddy MS (2018) Mangrove-associated fungi: a novel source of potential anticancer compounds. J Fungi 4:101
De Silva DD, Rapior S, Hyde KD, Bahkali AH (2012) Medicinal mushrooms in prevention and control of diabetes mellitus. Fungal Divers 56:1–29
De Silva DD, Rapior S, Sudarman E, Stadler M, Xu J, Alias AS, Hyde KD (2013) Bioactive metabolites from macrofungi: ethnopharmacology, biological activities and chemistry. Fungal Divers 62:1–40
de Souza FA, da Silva IR, Barreto MBBD, Oehl F, Goto BT, Maia LC (2018) Racocetra crispa (Glomeromycotina) delimited by integrative evidence based on morphology, long continuous nuclear rDNA sequencing and phylogeny. Mycol Prog 17:999-1011
de Vries RP, Riley R, Wiebenga A (2017) Comparative genomics reveals high biological diversity and specific adaptations in the industrially and medically important fungal genus Aspergillus. Genome Biol 18: 28. https://doi.org/10.1186/s13059-017-1151-0
D’hondt K, Kostic T, McDowell R, Eudes F, Singh BK, Sarkar S, Markakis M, Schelkle B, Maguin E, Sessitsch A (2021) Microbiome innovations for a sustainable future. Nat Microbiol 6:138–42
Di Bella JM, Bao Y, Gloor GB, Burton JP (2013) High throughput sequencing methods and analysis for microbiome research. J Microbiol Methods 95:401–14
Ding C, He X (2004) K-means clustering via principal component analysis. In: Proceedings of the twenty-first international conference on Machine learning, pp 29. https://doi.org/10.1145/1015330.1015408
Discomycetes (2023). https://discomycetes.org. Accessed 26 June 2023
Dissanayake AJ, Purahong W, Wubet T, Hyde KD (2018) Direct comparison of culture-dependent and culture-independent molecular approaches reveal the diversity of fungal endophytic communities in stems of grapevine (Vitis vinifera). Fungal Divers 90:85-107
Dissanayake AJ, Bhunjun CS, Maharachchikumbura SSN, Liu JK (2020) Applied aspects of methods to infer phylogenetic relationships amongst fungi. Mycosphere 11:2652–2676
Dlott G, Maul JE, Buyer J, Yarwood S (2015) Microbial rRNA: rDNA gene ratios may be unexpectedly low due to extracellular DNA preservation in soils. J Microbiol Methods 115:112–120
Dollhofer V, Podmirseg SM, Callaghan TM, Griffith GW, Fliegerová K (2015) Anaerobic fungi and their potential for biogas production. Biogas Sci Technol 151:41–61
Dong W, Wang B, Hyde KD, McKenzie EHC, Raja HA, Tanaka K, Abdel-Wahab MA, Abdel-Aziz FA, Doilom M, Phookamsak R, Hongsanan S, Wanasinghe DN, Yu XD, Wang GN, Yang H, Tang J, Thambugala KM, Tian Q, Luo ZL, Yang JB, Miller AN, Fournier J, Boonmee S, Hu DM, Nalumpang S, Zhang H (2020) Freshwater dothideomycetes. Fungal Divers 105:319–575
Dooley DP, Bostic PS, Beckius ML (1997) Spook house sporotrichosis: a point-source outbreak of sporotrichosis associated with hay bale props in a halloween haunted house. Arch Intern Med 157:1885-1887
Droby S, Romanazzi G, Tonutti P (2016) Alternative approaches to synthetic fungicides to manage postharvest decay of fruit and vegetables: needs and purposes of a special issue. Postharvest Biol Technol 122:1–2
Droby S, Zhimo VY, Wisniewski M, Freilich S (2022) The pathobiome concept applied to postharvest pathology and its implication on biocontrol strategies. Postharvest Biol Technol 189:111911
Duan Y, Qiao Y, Wang Y, Zhao M, Wang Z, Huang L, Kang Z (2021) Functional analysis of the FgHOG1 gene involved in stress responses and virulence in Fusarium graminearum using CRISPR-Cas9. Front Microbiol 12:748459
Eisen JA (1998) Phylogenomics: improving functional predictions for uncharacterized genes by evolutionary analysis. Genome Res 8:163–167. https://doi.org/10.1101/gr.8.3.163
Eisen JA, Fraser CM (2003) Phylogenomics: intersection of evolution and genomics. Science 300:1706–1707. https://doi.org/10.1126/science.1086292
Ekanayaka AH, Tibpromma S, Dai D, Xu R, Suwannarach N, Stephenson SL, Dao C, Karunarathna SC (2022) A review of the fungi that degrade plastic. J Fungi 8:772
Ekor M (2014) The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Neurol. https://doi.org/10.3389/fphar.2013.00177
Elahi N, Kamali M, Baghersad MH (2018) Recent biomedical applications of gold nanoparticles: a review. Talanta 184:537–556
El-Brolosy MA, Stainier DY (2017) Genetic compensation: a phenomenon in search of mechanisms. PLoS Gen 13:1006780
Elegbede JA, Lateef A (2018) Valorization of corn-cob by fungal isolates for production of xylanase in submerged and solid-state fermentation media and potential biotechnological applications. Waste Biomass Valor 9:1273–1287
Elisashvili VI (2012) Submerged cultivation of medicinal mushrooms: bioprocesses and products. Int J Med Mushrooms 14:211–239
El Jarroudi M, Kouadio L, El Jarroudi M, Junk J, Bock C, Diouf AA, Delfosse P (2017) Improving fungal disease forecasts in winter wheat: a critical role of intra-day variations of meteorological conditions in the development of Septoria leaf blotch. Field Crops Res 213:12–20
Elkhateeb WA (2020) What medicinal mushroom can do? Chem Res J 5(1):106–118
El-Sayed ESR, Hazaa MA, Shebl MM, Amer MM, Mahmoud SR Khattab AA (2022) Bioprospecting endophytic fungi for bioactive metabolites and use of irradiation to improve their bioactivities. AMB Express 12:46
Eschenbrenner CJ, Feurtey A, Stukenbrock EH (2020) Population genomics of fungal plant pathogens and the analyses of rapidly evolving genome compartments. Stat Popul Genomics, pp 337–355
Espindola A, Schneider W, Hoyt PR, Marek SM, Garzon C (2015) A new approach for detecting fungal and oomycete plant pathogens in next generation sequencing metagenome data utilising electronic probes. Int J Data Mining Bioinform 12:115-128
Faganello FDS, CARRER R, Dias VD, MORELLO RMSC, CUNHA MGD (2017) Molecular diagnosis of Guignardia citricarpa in asymptomatic sweet orange tissue. Revista Brasileira de Fruticultura 39:e518
Fahmy SA, Preis E, Bakowsky U, Azzazy HMES (2020) Platinum nanoparticles: green synthesis and biomedical applications. Molecules 25:4981
Falcetta P, Aragona M, Bertolotto A, Bianchi C, Campi F, Garofolo M, Prato SD (2022) Insulin discovery: a pivotal point in medical history. Metabolism 127:154941. https://doi.org/10.1016/j.metabol.2021.154941
Fan K, Chu H, Eldridge DJ, Gaitan JJ (2023) Soil biodiversity supports the delivery of multiple ecosystem functions in urban greenspaces. Nat Ecol Evol 7:113–26
Faria CB, Abe CA, da Silva CN, Tessmann DJ, Barbosa-Tessmann IP (2012) New PCR assays for the identification of Fusarium verticillioides, Fusarium subglutinans, and other species of the Gibberella fujikuroi complex. Int J Mol Sci 13:115–132
Feng K, Yang YQ, Li SPR (2008) In: Li SP, Wang YT (eds) Pharmacological activity-based quality control of Chinese herbs. Nova Science Publisher, New York, pp 155–78
Feng J, Surup F, Hauser M, Miller A, Wennrich JP, Stadler M, Cox RJ, Kuhnert E (2020) Biosynthesis of oxygenated brasilane terpene glycosides involves a promiscuous N-acetylglucosamine transferase. Chem Comm 56:12419-12422
Ferone M, Gowen A, Fanning S, Scannell AG (2020). Microbial detection and identification methods: bench top assays to omics approaches. Compr Rev Food Sci Food Saf 19:3106-3129
Ferreira IC, Heleno SA, Reis FS, Stojkovic D, Queiroz MJR, Vasconcelos MH, Sokovic M (2015) Chemical features of Ganoderma polysaccharides with antioxidant, antitumor and antimicrobial activities. Phytochemistry 114: 38–55
Field KJ, Bidartondo MI, Rimington WR, Hoysted GA, Beerling DJ, Cameron DD (2019) Functional complementarity of ancient plant-fungal mutualisms: contrasting nitrogen, phosphorus and carbon exchanges between Mucoromycotina and Glomeromycotina fungal symbionts of liverworts. New Phytol 223:908–921
Fijarczyk A, Hessenauer P, Hamelin RC, Landry CR (2022) Lifestyles shape genome size and gene content in fungal pathogens. bioRxiv 19:1–57. https://doi.org/10.1101/2022.08.24.505148
Filgueiras R, Mantovani EC, Althoff D, Fernandes Filho EI, Cunha FF (2019) Crop NDVI monitoring based on sentinel 1. Remote Sens 11(12):1441. https://doi.org/10.3390/rs11121441
Fiorilli V, Catoni M, Lanfranco L, Zabet NR (2020) Interactions of plants with bacteria and fungi: molecular and epigenetic plasticity of the host. Front Plant Sci 11:274
Fisher MC, Berman AIA, Bicanic T, Bignell EM, Bowyer P, Bromley M, Brüggemann R, Garber G, Cornely OA, Gurr SJ, Harrison TS, Kuijper E, Rhodes J, Sheppard DC, Warris A, White PL, Xu J, Zwaan B, Verweij PE (2022) Tackling the emerging threat of antifungal resistance to human health. Nat Rev Microbiol 20:557–571
Franco-Duarte R, Černáková L, Kadam S, Kaushik K (2019) Advances in chemical and biological methods to identify microorganisms from past to present. Microorganisms 7:130
Fricke J, Blei F, Hoffmeister D (2017) Enzymatic synthesis of psilocybin. Angew Chem Int Ed 56:1235212355
Frost M (2016) Three popular medicinal mushroom supplements: a review of human clinical trials. All Fac Publ 1609
Fu L, Wang Y, Wang J, Yang Y, Hao L (2013) Evaluation of the antioxidant activity of extracellular polysaccharides from Morchella esculenta. Food Funct 4:871–879
Fu W, Chen BD, Rillig MC, Jansa J, Ma W, Xu C (2022) Community response of arbuscular mycorrhizal fungi to extreme drought in a cold-temperate grassland. New Phytol 234:2003–2017
Fu Y, Zhang L, Cong M, Wan K, Jiang G, Dai S, Wang L, Liu X (2021) Application of Auricularia cornea as a pork fat replacement in cooked sausage. Coatings 11:14-32. https://doi.org/10.3390/COATINGS11111432
Fungal Growth database (2023) https://www.indexfungorum.org/. Accessed 12 Sept 2023
Fungal ITS Database Commons (2023) https://www.indexfungorum.org/. Accessed 12 Sept 2023
Fungal MLST (2023) https://mlst.mycologylab.org/. Accessed 12 Sept 2023
Gabaldón T (2020) Grand challenges in fungal genomics and evolution. Front Fungal Biol 1:594855
Gade AK, Bonde P, Ingle AP, Marcato PD, Durán N, Rai MK (2008) Exploitation of Aspergillus niger for synthesis of silver nanoparticles. J Biobased Mater Bioenergy 2:243–247. https://doi.org/10.1166/jbmb.2008.401
Gajbhiye M, Kesharwani J, Ingle A, Gade A, Rai M (2009) Fungus-mediated synthesis of silver nanoparticles and their activity against pathogenic fungi in combination with fluconazole. Nanomed Nanotechnol Biol Med 5:382–386
Gandia A, van den Brandhof JG, Appels FV, Jones MP (2021) Flexible fungal materials: shaping the future. Trends Biotechnol 39:1321–1331
Ganeshan S, Kim SH, Vujanovic V (2021) Scaling-up production of plant endophytes in bioreactors: concepts, challenges and perspectives. Bioresour Bioprocess 8:63. https://doi.org/10.1186/s40643-021-00417-y
Gantenbein S, Colucci E, Käch J, Trachsel E, Coulter FB, Rühs PA, Masania K, Studart AR (2023) Three-dimensional printing of mycelium hydrogels into living complex materials. Nat Mater 22:128–134. https://doi.org/10.1038/s41563-022-01429-5
Gao Y, Su Y, Sun W, Cai L (2015) Diaporthe species occurring on Lithocarpus glabra in China, with descriptions of five new species. Fungal Biol 119:295-309
Garcia JF, Lawrence DP, Morales-Cruz A, Travadon R, Minio A, Hernandez-Martinez R, Rolshausen PE, Baumgartner K, Cantu D (2021) Phylogenomics of plant associated Botryosphaeriaceae species. Front Microbiol 12:652802
García-Granados R, Lerma-Escalera JA, Morones-Ramírez JR (2019) Metabolic engineering and synthetic biology: synergies, future, and challenges. Front Bioeng Biotech 7:36
Garmendia G, Alvarez A, Villarreal R, Martínez-Silveira A, Wisniewski M, Vero S (2021) Fungal diversity in the coastal waters of King George Island (maritime Antarctica). World J Microbiol Biotechnol 37:1–12
Garwood K, Garwood C, Hedeler C, Griffiths T (2006) Model-driven user interfaces for bioinformatics data resources:regenerating the wheel as an alternative to reinventing it. BMC Bioinform 7:1–14
Gautam AK, Verma RK, Avasthi S, Sushma, Bohra Y, Devadatha B, Niranjan M, Suwannarach N (2022) Current insight into traditional and modern methods in fungal diversity estimates. J Fungi 8:226
Geethanjali PA, Gowtham HG, Jayashankar M (2020) Biodegradation potential of indigenous litter dwelling ligninolytic fungi on agricultural wastes. Bull Natl Res Centre 44:1–14
Genaev M, Ekaterina S, Afonnikov D (2020) Application of neural networks to image recognition of wheat rust diseases. In: 2020 cognitive sciences, genomics and bioinformatics (CSGB), pp 40–42. https://doi.org/10.1109/CSGB51356.2020.9214703
Gerke J, Köhler AM, Wennrich JP, Große V, Shao L, Heinrich AK, Bode HB, Chen W, Surup F, Braus GH (2022) Biosynthesis of antibacterial iron-chelating tropolones in Aspergillus nidulans as response to glycopeptide-producing Streptomycetes. Front Fungal Biol 2:777474. https://doi.org/10.3389/ffunb.2021.777474
Gessler C, Pertot I, Perazzolli M (2011) Plasmopara viticola: a review of knowledge on downy mildew of grapevine and effective disease management. Phytopathol Mediterr 50:3–44
Ghosh S, Rusyn I, Dmytruk OV, Dmytruk KV, Onyeaka H, Gryzenhiut M, Gafforov Y (2023) Filamentous fungi for sustainable remediation of pharmaceutical compounds, heavy metal and oil hydrocarbons. Front Bioeng Biotech 11:1106973
Gil YG, Kang S, Chae A, Kim YK, Min DH, Jang H (2018) Synthesis of porous Pd nanoparticles by therapeutic chaga extract for highly efficient tri-modal cancer treatment. Nanoscale 10:19810–19817
Gilbert GS, Parker IM (2010) Rapid evolution in a plant‐pathogen interaction and the consequences for introduced host species. Evol Appl 3:144-156
Globe Newswire (2022) Functional mushroom market size to exceed USD 23.3 billion by 2030 with 10.1% CAGR.In-depth analysis, global market share, size, industry trends & forecast 2023–2030. https://www.globenewswire.com/en/news-release/2023/04/20/2650858/0/en/Functional-Mushroom-Market-Size-to-Exceed-USD-23-3-Billion-by-2030-with-10-1-CAGR-In-depth-Analysis-Global-Market-Share-Size-Industry-Trends-Forecast-2023-2030.html#:~:text=“According to the latest research,approximately 10.1%25 during the forecast
Global wellness institute (2020) Wellness economy. https://globalwellnessinstitute.org/what-is-wellness/what-is-the-wellness-economy/ Accessed 19 Sep 2023
Gomdola D, Bhunjun CS, Hyde KD, Jeewon R, Pem D, Jayawardena RS (2022) Ten important forest fungal pathogens: a review on their emergence and biology. Mycosphere 13:612–671
Gomez-Roldan V, Fermas S, Brewer PB, Puech-Pagès V, Dun EA, Pillot J-P, Letisse F, Matusova R, Danoun S, Portais J-C (2008) Strigolactone inhibition of shoot branching. Nature 455(7210):189–194
Gómez-Silva B, Vilo-Muñoz C, Galetović A, Dong Q Castelan-Sanchez H, Davila-Ramos S, Cortes-Lopez NG, Martinez-Avila L, Dobson ADW, Batista-Garcia RA (2019) Metagenomics of Atacama lithobiontic extremophile life unveils highlights on fungal communities, biogeochemical cycles and carbohydrate-active enzymes. Microorganisms 7:619
Gonçalves MFM, Hilário S, Van de Peer Y, Esteves AC (2022) Genomic and metabolomic analyses of the marine fungus Emericellopsis cladophorae: insights into saltwater adaptability mechanisms and its biosynthetic potential. J Fungi 8:31
González A, Cruz M, Losoya C, Nobre C, Loredo A, Rodríguez R, Contreras J, Belmares R (2020) Edible mushrooms as a novel protein source for functional foods. Food Funct 11:1–15. https://doi.org/10.1039/d0fo01746a
Goodell B, Winandy JE, Morrell JJ (2020) Fungal degradation of wood: emerging data, new insights and changing perceptions. Coatings 10:1210
Gopalakrishnan C, Pawar RS, Bhutani KK (2005) Development of Agaricus bisporus as a nutraceutical of tomorrow. Acta Hortic 680:45–47. https://doi.org/10.17660/ActaHortic.2005.680.5
Gostinčar C (2020) Towards genomic criteria for delineating fungal species. J Fungi 6 (4):246
Gostinčar C, Zalar P, Gunde-Cimerman N (2022) No need for speed: slow development of fungi in extreme environments. Fungal Biol Rev 39:1–14
Govender NP, Maphanga TG, Zulu TG, Patel J, Walaza S, Jacobs C, Ebonwu JI, Ntuli S, Naicker SD, Thomas J (2015) An outbreak of lymphocutaneous sporotrichosis among mine-workers in South Africa. Plos Neglect Trop Dis. https://doi.org/10.1371/journal.pntd.0004096
Grabicoski EMG, Jaccoud-Filho DDS, Lee D, Henneberg L, Pileggi M (2020) Real-time quantitative and ion-metal indicator LAMP-based assays for rapid detection of Sclerotinia sclerotiorum. Plant Dis 104:1514–1526
Grandaubert J, Dutheil JY, Stukenbrock EH (2019) The genomic determinants of adaptive evolution in a fungal pathogen. Evol Lett 3:299–312
Grangeia C, Batista C, Raposo M, Barros L, Cadavez V, Ferreira ICFR (2010) Incorporation of Boletus edulis extract in beef burgers patties: preliminary evaluation of the antioxidant protective effects. In: 6th International conference on simulation and modelling in the food and bio-industry 2010
Green MA, Emery K, Hishikawa Y, Warta W, Dunlop ED (2015) Solar cell efficiency tables (Version 45). Prog Photovolt Res Appl 23:1–9. https://doi.org/10.1002/pip.2573
Greener JG, Kandathil SM, Moffat L, Jones DT (2022) A guide to machine learning for biologists. Nat Rev Mol Cell Biol 23:40–55. https://doi.org/10.1038/s41580-021-00407-0
Grumach AS, de Queiroz-Telles F, Migaud M, Lanternier F, Filho NR, Palma SM, Constantino-Silva RN, Casanova JL, Puel A (2015) A homozygous CARD9 mutation in a Brazilian patient with deep dermatophytosis. J Clin Immunol 35:486–490
Guilger-Casagrande M, Lima RD (2019) Synthesis of silver nanoparticles mediated by fungi:a review. Front Bioeng Biotechnol 7:287
Güldener U, Mannhaupt G, Münsterkötter M, Haase D, Oesterheld M, Stümpflen V, Adam G (2006) FGDB: a comprehensive fungal genome resource on the plant pathogen Fusarium graminearum. Nucleic Acids Res 34:456−458
Guo LD, Hyde KD, Liew ECY (2001) Detection and taxonomic placement of endophytic fungi within frond tissues of Livistona chinensis based on rDNA sequences. Mol Phylogenet Evol 20:1–13
Gutleben J, Chaib De Mares M, Van Elsas JD (2018) The multi-omics promise in context: from sequence to microbial isolate. Crit Rev Microbiol 44:212–229
Guttmacher AE, Collins FS (2003) Welcome to the genomic era. NEJM 349:996–998. https://doi.org/10.1056/NEJMe038132
Gygli SM, Borrell S, Trauner A, Gagneux S (2017) Antimicrobial resistance in Mycobacterium tuberculosis: mechanistic and evolutionary perspectives. FEMS Microbiol Rev 41:354–373. https://doi.org/10.1093/femsre/fux011
Habtemariam S (2019) The chemistry, pharmacology and therapeutic potential of the edible mushroom Dictyophora indusiata (Vent ex. Pers.) Fischer (Synn. Phallus indusiatus). Biomedicines 7:98
Hammel KE (1997) Fungal degradation of lignin. Driven Nat 33:45
Hamza A, Ghanekar S, Kumar SD (2023) Current trends in health-promoting potential and biomaterial applications of edible mushrooms for human wellness. Food Biosci 51: 102290. https://doi.org/10.1016/j.fbio.2022.102290
Hanafy MH (2018) Myconanotechnology in veterinary sector: status quo and future perspectives. Int J Vet Sci Med 6:270–273
Haneef M, Ceseracciu L, Canale C, Bayer IS, Heredia-Guerrero JA, Athanassiou A (2017) Advanced materials from fungal mycelium: fabrication and tuning of physical properties. Sci Rep 7:41292. https://doi.org/10.1038/srep41292
Hao T, Elith J, Guillera-Arroita G, Lahoz-Monfort JJ (2021) Enhancing repository fungal data for biogeographic analyses. Fungal Ecol 53:101097
Hapsari R, Elya B, Amin J (2012) Formulation and evaluation of antioxidant and tyrosinase inhibitory effect from gel containing the 70% ethanolic Pleurotus ostreatus extract. Int J Med Arom Plants 2:135–140
Harms H, Schlosser D, Wick LY (2011) Untapped potential: exploiting fungi in bioremediation of hazardous chemicals. Nat Rev Microbiol 9:177-192
Harpaz R, Dahl RM, Dooling KL (2016) Prevalence of immunosuppression among US adults, 2013. JAMA 316:2547-2548. https://doi.org/10.1001/jama.2016.16477
Hashimoto S (2009) Micafungin: a sulfated echinocandin. J Antibiot 62:27–35. https://doi.org/10.1038/ja.2008.3
Hassett BT, Ducluzeau ALL, Collins RE, Gradinger R (2017) Spatial distribution of aquatic marine fungi across the western Arctic and sub‐arctic. Environ Microbiol 19:475-484
Hautbergue T, Jamin EL, Debrauwer L, Puel O, Oswald IP (2018) From genomics to metabolomics, moving toward an integrated strategy for the discovery of fungal secondary metabolites. Nat Prod Rep 35:147-173. https://doi.org/10.1039/c7np00032d
Havis ND, Nyman M, Oxley SJP (2014) Evidence for seed transmission and symptomless growth of Ramularia collo‐cygni in barley (Hordeum vulgare). Plant Pathol 63:929–936
Hawkins HJ, Cargill RIM, Van Nuland ME, Hagen SC, Field KJ, Sheldrake M, Soudzilovakaia NA, Kiers ET (2018) Mycorrhizal mycelium as a global carbon pool. Curr Biol 33:560–573
Hawksworth DL (1991) The fungal dimension of biodiversity—magnitude, significance, and conservation. Mycol Res 95:641–655
Hawksworth DL, Lücking R (2017) Fungal diversity revisited: 2.2 to 3.8 million species. Microbiol Spectr 5:1–17
He J (2018) Harvest and trade of caterpillar mushroom (Ophiocordyceps sinensis) and the implications for sustainable use in the Tibet Region of Southwest China. J Ethnopharmacol 221:86–90. https://doi.org/10.1016/j.jep.2018.04.022
He MQ, Zhao RL, Liu DM, Denchev TT (2022) Species diversity of Basidiomycota. Fungal Divers 114:281–325
Healthline (2023) 6 Benefits of reishi mushroom (plus side effects and dosage). https://www.healthline.com/nutrition/reishi-mushroom-benefits
Hedeler C, Wong HM, Cornell MJ, Alam I, Soanes DM, Rattray M, Hubbard SJ, Talbot NJ, Oliver SG, Paton NW (2007) e-Fungi:a data resource for comparative analysis of fungal genomes. BMC Genomics 8:426
Heinrich M, Hesketh A (2019) 25 years after the `Rio Convention’––Lessons learned in the context of sustainable development and protecting indigenous and local knowledge. Phytomedicine 53:332–343. https://doi.org/10.1016/J.PHYMED.2018.04.061
Helander M, Saloniemi I, Omacini M, Druille M, Salminen JP, Saikkonen KJ (2018) Glyphosate decreases mycorrhizal colonization and affects plant-soil feedback. Sci Total Environ 642:285-291
Herbert PD, Gregory TR (2005) The promise of DNA barcoding for taxonomy. Syst Biol 54(5):852–859
Herzog S, Brinkmann H, Vences M, Fleißner A (2020) Evidence of repeated horizontal transfer of sterol C-5 desaturase encoding genes among Dikarya fungi. Mol Phy Evol 150:106850
Hibbett DS, Stajich JE, Spatafora JW (2013) Toward genome-enabled mycology. Mycologia 105:1339–1349
Hilário S, Gonçalves MFM (2023) Mechanisms underlying the pathogenic and endophytic lifestyles in Diaporthe: an omics-based approach. Horticulturae 9:423
Holden SR, Treseder KK (2013) A meta-analysis of soil microbial biomass responses to forest disturbances. Front Microbiol 4:163
Holt GA, McIntyre G, Flagg D, Bayer E, Wanjura JD, Pelletier MG (2012) Fungal mycelium and cotton plant materials in the manufacture of biodegradable molded packaging material: evaluation study of select blends of cotton byproducts. J Biobased Mater Bioenergy 6:431–439
Hongsanan A, Jeewon R, Purahong W, Xie N, Liu JK, Jayawardena RS, Ekanayaka AH, Dissanayake A, Raspe O, Hyde KD, Stadler M, Persoh D (2018) Can we use environmental DNA as holotypes? Fungal Divers 92:1–30
Howlett BJ, Lowe RG, Marcroft SJ, van de Wouw AP (2015) Evolution of virulence in fungal plant pathogens: exploiting fungal genomics to control plant disease. Mycologia 107:441–451
Hoysted GA, Field KJ, Sinanaj B, Bell CA, Bidartondo MI, Pressel S (2023) Direct nitrogen, phosphorus and carbon exchanges between Mucoromycotina 'fine root endophyte' fungi and a flowering plant in novel monoxenic cultures. New Phytol 238:70–79
Hsieh CW, Sun PL, Wu YH (2010) Trichophyton erinacei infection from a hedgehog: a case report from Taiwan. Mycopathologia 170:417–421
Hu Y, Mortimer PE, Hyde KD, Kakumyan P, Thongklang N (2021) Mushroom cultivation for soil amendment and bioremediation. Circ Agric Syst 1:1–14. https://doi.org/10.48130/CAS-2021-0011
Huang J, Cook DE (2022) The contribution of DNA repair pathways to genome editing and evolution in filamentous pathogens. FEMS Microbiol Rev 46:fuac035
Huang HC, Hsu TF, Chao HL, Chen CC, Chui SW, Cgang TM (2014) Inhibition of melanogenesis in murine melanoma cells by Agaricus brasiliensis methanol extract and anti-reactive oxygen species (ROS) activity. Afr J Microbiol Res 8:519–524
Huang C, Peng J, Zhang W, Chethana KWT (2023) LtGAPR1 is a novel secreted effector from Lasiodiplodia theobromae that interacts with NbPsQ2 to negatively regulate infection. J Fungi 9:188
Huanraluek N, Padaruth OD, Jayawardena RS, Li WJ, Hongsanan S, Jeewon R, Maharachchikumbura S, Senanayake I, Phukhamsakda C, Wijayawardene N, Shivas R, Goonasekara ID, Norphanphoun C, Pem D, Clabon M, Bjunjun CS, Hyde KD (2021). https://www.coelomycetes.org: databank that contributes for the classification, identification and nomenclature of Coelomycetes. Asian J Mycol 4:114–122
Hubbard A, Lewis CM, Yoshida K, Ramirez-Gonzalez RH, de Vallavieille-Pope C, Thomas J, Kamoun S, Bayles R, Uauy C, Saunders DG (2015) Field pathogenomics reveals the emergence of a diverse wheat yellow rust population. Genome Biol 16:1–15
Huberman LB (2021) Developing functional genomics platforms for fungi. Msystems 6:10–128
Husain H, Huda-Faujan N (2020) Potential application of grey oyster mushroom stems as halal meat replacer in imitation chicken nuggets. Food Res 4:179–186. https://doi.org/10.26656/fr.2017.4(S1).S18
Hyde KD, Jones EBG (1988) Marine mangrove fungi. Mar Ecol 9:15-33
Hyde KD (1992) Fungi from decaying intertidal fronds of Nypa fruticans, including three new genera and four new species. Bot J Linnean Soc 110:95–110
Hyde KD, Lee SY (1998) Ecology of mangrove fungi and their role in nutrient cycling: what gaps occur in our knowledge? Hydrobiologia 295:107–118
Hyde KD, Bahkali AH, Moslem MA (2010) Fungi—an unusual source for cosmetics. Fungal Divers 43:1–9
Hyde KD, Jones EBG, Liu JK, Ariyawansa H, Boehm E, Boonmee S, Braun U, Chomnunti P, Crous PW, Dai DQ, Diederich P, Dissanayake A, Doilom M, Doveri F, Hongsanan S, Jayawardena RS, Lawrey JD, Li YM, Liu YX, Lücking R, Monkai J, Muggia L, Nelsen MP, Pang KL, Phookamsak R, Senanayake IC, Shearer CA, Suetrong S, Tanaka K, Thambugala KM, Wijayawardene NN, Wikee S, Wu HX, Zhang Y, Aguirre-Hudson B, Alias SA, Aptroot A, Bahkali AH, Berezza JL, Bhat DJ, Camporesi E, Chukeatirote E, Gueidan C, Hawksworth DL, Hirayama K, de Hoog S, Kang JC, Knudsen K, Li WJ, Li XH, Liu ZY, Mapook A, McKenzie EHC, Miller AN, Mortimer PE, Phillips AJL, Raja HA,Scheuer C, Schumm F, Taylor JE, Tian Q, Tibpromma S, Wanasinghe DN, Wang Y, Xu JC, Yacharoen S, Yan JY, Zhang M (2013) Families of dothideomycetes. Fungal Divers 63:1–313
Hyde KD, Nilsson RH, Aisyah SA, Ariyawansa HA, Blair JE, Cai L, de Cock AWAM, Dissanayake AJ, Glockling SL, Goonasekara ID, Gorczak M, Hahn M, Jayawardena RS, van Kan JA, Laurence MH, Levesque AC, Li X, Liu JK, Maharachchikumbura SSN, Manamgoda DS, Martin FN, Mckenzie EHC, McTaggart AR, Mortimer PE, Zhou N (2014) One stop shop: backbones trees for important phytopathogenic genera: I. Fungal Divers 67:21–125
Hyde KD, Swe A, Zhang KQ (2014b) Nematode-trapping Fungi. In: Zhang K, Hyde KD (eds) Nematode-trapping fungi: fungal diversity research series. Mushroom Found. https://doi.org/10.1007/978-94-017-8730-7
Hyde KD, Norphanphoun C, Abreu VP, Bazzicalupo A, Chethana KWT, Clericuzio M, Monika C. Dayarathne MC, Dissanayake AJ, Ekanayaka AH, He MQ, Hongsanan S, Huang SK, Subashini C. Jayasiri SC, Jayawardena RS, Karunarathna A, Konta S, Kušan I, Lee H, Li JF, Lin CG, Liu NG, Lu YZ,Luo ZL, Manawasinghe IS, Mortimer PE (2017) Fungal diversity notes 603– 708: taxonomic and phylogenetic notes on genera and species. Fungal Divers 87:1–235
Hyde KD, Al-Hatmi AM, Andersen B, Boekhout T, Buzina W, Dawson Jr TL, Eastwood DC, Jones EBG, de Hoog S, Kang Y, Longcore JE, McKenzie EHC, Meis JF, Pinson-Gadais L, Rathnayaka AR, Richard-Forget F, Stadler M, Theelen B, Thongbai B, Tsui CKM (2018) The world’s ten most feared fungi. Fungal Divers 93:161–194
Hyde KD, Xu J, Rapior S, Jeewon R, Lumyong S, Niego AGT, Abeywickrama PD, Aluthmuhandiram JVS, Brahamanage RS, Brooks S, Chaiyasen A, Chethana KWT, Chomnunti P, Chepkirui C, Chuankid B, de Silva NI, Doilom M, Faulds C, Gentekaki E, Stadler M (2019) The amazing potential of fungi, 50 ways we can exploit fungi industrially. Fungal Divers 97:1–136. https://doi.org/10.1007/s13225-019-00430-9
Hyde KD, Chethana KWT, Jayawardena RS, Luangharn T, Calabon MS, Jones EBG, Hongsanani S, Lumyong S (2020a) The rise of mycology in Asia. ScienceAsia 46:1–11
Hyde KD, Jeewon R, Chen YJ, Bhunjun CS, Calabon MS, Jiang HB, Lin CG, Norphanphoun C, Sysouphanthong P, Pem D, Tibpromma S, Zhang Q, Doilom M, Jayawardena RS, Liu JK, Maharachchikumbura SSN, Phukhamsakda C, Phookamsak R, Al-Sadi AM, Naritsada Thongklang N, Wang Y, Gafforov Y, Jones EBG, Lumyong S (2020b) The numbers of fungi: is the descriptive curve flattening? Fungal Divers 103:219–27
Irinyi L, Serena C, Garcia-Hermoso D, Arabatzis M, Desnos-Ollivier M, Vu D, Cardinali G, Arthur I, Normand AC, Giraldo A, da Cunha KC, Sandoval-Denis M, Hendrickx M, Nishikaku AS, de Azevedo Melo AS, Merseguel KB, Khan A, Parente Rocha JA, Sampaio P, da Silva Briones MR, Ferreira RC, de Medeiros Muniz M, Castañón-Olivares LR, Estrada-Barcenas D, Cassagne C, Mary C, Duan SY, Kong F, Sun AY, Zeng X, Zhao ZT, Gantois N, Botterel F, Robbertse B, Schoch C, Gams W, Ellis D, Halliday C, Chen S, Sorrell TC, Piarroux R, Colombo AL, Pais C, de Hoog S, Zancopé-Oliveira RM, Taylor ML, Toriello C, de Almeida Soares CM, Delhaes L, Stubbe D, Dromer F, Ranque S, Guarro J, Cano-Lira JF, Robert V, Velegraki A, Meyer W (2015) International Society of Human and Animal Mycology (ISHAM)-ITS reference DNA barcoding database—the quality controlled standard tool for routine identification of human and animal pathogenic fungi. Med Mycol J 55:313–337
Jakubczyk D, Dussart F (2020) Selected fungal natural products with antimicrobial properties. Molecules 25:911
James TY, Pelin A, Bonen L, Ahrendt S, Sain D, Corradi N, Stajich JE (2013) Shared signatures of parasitism and phylogenomics unite Cryptomycota and Microsporidia. Curr Biol 23:1548–1553. https://doi.org/10.1016/j.cub.2013.06.057
Jansen R, Küsters K, Morschett H, Wiechert W, Oldiges M (2021) A fully automated pipeline for the dynamic at‐line morphology analysis of microscale Aspergillus cultivation. Fungal Biol Biotechnol 8:1–10
Janusz G, Pawlik A, Sulej J, Świderska-Burek U, Jarosz-Wilkolazka A, Paszcynski A (2017) Lignin degradation:microorganisms, enzymes involved, genomes analysis and evolution. FEMS Microbiol Rev 41:941–962
Jasani H, Umretiya N, Dharajiya D, Kapuria M, Shah S, Patel J (2016) Isolation, optimization and production of cellulase by Aspergillus niger from agricultural waste. J Pure Appl Microbiol 10:1159–1167
Jayachandran M, Xiao J, Xu B (2017) A critical review on health promoting benefits of edible mushrooms through gut microbiota. Int J Mol Sci 18(9):1934
Jayasiri SC, Hyde KD, Ariyawansa H, Bhat J (2005) The Faces of Fungi database:fungal names linked with morphology, phylogeny and human impacts. Fungal Divers 74:3–18
Jayawardena RS, Purahong W, Zhang W, Wubet TL, Li XH, Liu M, Zhao W, Hyde KD, Liu JH, Yan J (2018) Biodiversity of fungi on Vitis vinifera L. revealed by traditional and high-resolution culture-independent approaches. Fungal Divers 90:1–84
Jayawardena RS, Hyde KD, McKenzie EHC et al (2019) One stop shop III: taxonomic update with molecular phylogeny for important phytopathogenic genera: 51–75 (2019). Fungal Diversity 98:77–160. https://doi.org/10.1007/s13225-019-00433-6
Jayawardena RS, Bhunjun CS, Hyde KD, Gentekaki E (2021a) Colletotrichum: lifestyles, biology, morpho-species, species complexes and accepted species. Mycosphere 12: 519–669
Jayawardena RS, Hyde KD, de Farias ARG, Bhunjun CS (2021b) What is a species in fungal plant pathogens?. Fungal Divers 109:239-266
Jayawardena RS, Hyde KD, Wang S, Sun YR, Suwannarach N, Sysouphanthong P, Abdel-Wahab MA, Abdel-Aziz FA, Abeywickrama PD, Abreu VP, Armand A, Aptroot A, Bao DF, Begerow D, Bellanger JM, Bezerra JDP, Bundhun D, Calabon MS, Cao T, Cantillo T, Carvalho JLVR, Chaiwan N, Chen CC, Courtecuisse R, Wang Y (2022) Fungal diversity notes 1512–1610: taxonomic and phylogenetic contributions on genera and species of fungal taxa. Fungal Divers 117:1–272
Jeitler M, Michalsen A, Frings D, Hübner M, Fischer M, Koppold-Liebscher DA, Kessler CS (2020) Significance of medicinal mushrooms in integrative oncology: a narrative review. Front Pharmacol 11:580–656
Jeyaraj M, Gurunathan S, Qasim M, Kang MH, Kim JH (2019) A comprehensive review on the synthesis, characterization, and biomedical application of platinum nanoparticles. Nanomaterials 9:1719
Jia YY, Walder F, Wagg C, Feng G (2021) Mycorrhizal fungi maintain plant community stability by mitigating the negative effects of nitrogen deposition on subordinate species in Central Asia. J Veg Sci 32:e12944
Jiang L, Yang H, Zhang X, Li X, Lv K, Zhang W, Zhu G, Liu C, Wang Y, Hsiang T, Zhang L, Liu X (2022) Schultriene and nigtetraene:two sesterterpenes characterized from pathogenetic fungi via genome mining approach. Appl Microbiol Biotechnol 106:6047–6057. https://doi.org/10.1007/s00253-022-12125-4
John E, Singh KB, Oliver RP, Tan KC (2021) Transcription factor control of virulence in phytopathogenic fungi. Mol Plant Pathol 22:858–881
Jones EBG, Pang KL, Abdel-Wahab MA, Scholz B, Hyde KD, Boekhout T, Ebel R, Rateb ME, Henderson L, Sakayaroj J, Suetrong S, Dayarathne MC, Kumar V, Raghukumar S, Sridhar KR, Bahkali AHA, Gleason FH, Norphanphoun C (2019) An online resource for marine fungi. Fungal Divers 96:347–433
Jones EBG, Suetrong S, Sakayaroj J, Bahkali AH, Abdel-Wahab MA, Boekhout T, Pang KL (2015) Classification of marine Ascomycota, Basidiomycota, Blastocladiomycota and Chytridiomycota. Fungal Divers 73:1–72
Kalimuthu K, Cha BS, Kim S, Park KS (2020) Eco-friendly synthesis and biomedical applications of gold nanoparticles: a review. Microchem J 152:104296
Kang D, Jiang X, Wan H, Ran Y, Hao D, Zhang C (2014) Mucor irregularis infection around the inner canthus cured by amphotericin B: a case report and review of published literatures. Mycopathologia 178:129-133
Karana E, Blauwhoff D, Hultink EJ, Camere S (2018) When the material grows: A case study on designing (with) mycelium-based materials. Int J Des 12:119-136
Karimi K, Arzanlou M, Pertot I (2019) Development of novel species-specific primers for the specific identification of Colletotrichum nymphaeae based on conventional PCR and LAMP techniques. Eur J Plant Pathol 156:463–475
Karwehl S, Stadler M (2016) Exploitation of fungal biodiversity for discovery of novel antibiotics. Curr Top Microbiol Immunol 398:303–338. https://doi.org/10.1007/82_2016_496
Kazda M, Langer S, Bengelsdorf FR (2014) Fungi open new possibilities for anaerobic fermentation of organic residues. Energy Sustain Soc 4:1–9
Keller NP (2019) Fungal secondary metabolism: regulation, function and drug discovery. Nat Rev Microbiol 17:167-180
Matio Kemkuignou B, Schweizer L, Lambert C, Anoumedem EGM, Kouam SF, Stadler M, Marin-Felix Y, (2022) New polyketides from the liquid culture of Diaporthe breyniae sp. nov. (Diaporthales, Diaporthaceae). MycoKeys 90:85-118
Kersey PJ, Lawson D, Birney E, Derwent PS (2010) Ensembl genomes: extending Ensembl across the taxonomic space. Nucleic Acids Res 38:563–569
Khalifa NS, Hasaneen MN (2018) The effect of chitosan–PMAA–NPK nanofertilizer on Pisum sativum plants. 3 Biotech 8:193
Khan AA, Bacha N, Ahmad B, Lutfullah G, Farooq U, Cox RJ (2014) Fungi as chemical industries and genetic engineering for the production of biologically active secondary metabolites. Asian Pac J Trop Biomed 4:859–870
Khan AU, Malik N, Khan M, Cho MH, Khan MM (2018) Fungi-assisted silver nanoparticle synthesis and their applications. Bioprocess Biosyst Eng 41:1–20
Khan NT, Khan MJ, Jameel J, Jameel N, Rheman SUA (2017) An overview: biological organisms that serves as nanofactories for metallic nanoparticles synthesis and fungi being the most appropriate. Bioceram Dev Appl 7:101. https://doi.org/10.4172/2090-5025.1000101
Khatoon H, Rai JPN, Jillani A (2021) Role of fungi in bioremediation of contaminated soil. In: Sharma VK, Shah MP Kumar A (eds) Fungi bio-prospects in sustainable agriculture, environment and nano-technology. Elsevier, New York
Kim K, Choi B, Lee I, Lee H, Kwon S, Oh K, Kim AY (2011) Bioproduction of mushroom mycelium of Agaricus bisporus by commercial submerged fermentation for the production of meat analogue. J Sci Food Agric 91:1561–1568. https://doi.org/10.1002/jsfa.4348
Kim SY, Go KC, Song YS, Jeong YS (2014) Extract of the mycelium of T. matsutake inhibits elastase activity and TPA-induced MMP-1 expression in human fibroblasts. Int J Mol Med 34:1613–1621
Kim YJ (2007) Antimelanogenic and antioxidant properties of gallic acid. Biol Pharmaceut Bull 30:1052–1055
Kimutai J, New M, Wolski P, Otto F (2022) Attribution of the human influence on heavy rainfall associated with flooding events during the 2012, 2016, and 2018 March-April-May seasons in Kenya. Weather Climate Extremes 38:100529
Kirchherr J, Reike D, Hekkert M (2017) Conceptualizing the circular economy: an analysis of 114 definitions. Resour Conserv Recycl 127:221–232
Kirk PM, Cannon PF, David JC, Stalpers JA (2008) Ainsworth & Bisby’s dictionary of the fungi, 10th edn. CAB International, Wallingford
Kobayashi Y, Maeda T, Yamaguchi K, Kameoka H, Tanaka S, Ezawa T, Shigenobu S, Kawaguchi M (2018) The genome of Rhizophagus clarus HR1 reveals a common genetic basis for auxotrophy among arbuscular mycorrhizal fungi. Bmc Genomics 19:465
Köhler JR, Casadevall A, Perfect J (2014) The spectrum of fungi that infects humans. Cold Spring Harb Perspect Med 5:a019273. https://doi.org/10.1101/cshperspect.a019273
Kohsaka R, Ito K, Miyake Y, Uchiyama Y (2021) Cultural ecosystem services from the afforestation of rice terraces and farmland: Emerging services as an alternative to monoculturalization. Forest Ecol Manag 497:119481
Kolarikova Z, Slavikova R, Kruger C, Kruger M, Kohout P (2021) PacBio sequencing of Glomeromycota rDNA: a novel amplicon covering all widely used ribosomal barcoding regions and its applicability in taxonomy and ecology of arbuscular mycorrhizal fungi. New Phytol 231:490-499
Koo T, Kim MH, Jue MS (2021) Automated detection of superficial fungal infections from microscopic images through a regional convolutional neural network. PLoS ONE. 16:e0256290. https://doi.org/10.1371/journal.pone.0256290
Kopchinskiy A, Komoń M, Kubicek CP, Druzhinina IS (2005) TrichoBLAST: a multilocus database for Trichoderma and Hypocrea identifications. Mycol Res 109:658–660
Kothari D, Patel S, Kim SK (2018) Anticancer and other therapeutic relevance of mushroom polysaccharides: a holistic appraisal. Biomed Pharmacother 105:377-394
Kovács E, Szűcs C, Farkas A, Szuhaj M, Maróti G, Bagi Z, Rákhely G, Kovács KL (2022) Pretreatment of lignocellulosic biogas substrates by filamentous fungi. J Biotechnol 360:160-170
Kowalski CH, Kerkaert JD, Liu Z, Ge X, Schorr M (2021) Candida glabrata CZF1 is a potent transcriptional activator of key gluconeogenic genes. mBio 12:e03461–20
Kozarski M, Klaus A, van Griensven L, Jakovljevic D, Todorovic N, Wan WAAQI, Vunduk J (2023) Mushroom β-glucan and polyphenol formulations as natural immunity boosters and balancers: nature of the application. Food Sci Hum Well 12: 378-396
Kruger M, Kruger C, Walker C, Stockinger H, Schussler A (2012) Phylogenetic reference data for systematics and phylotaxonomy of arbuscular mycorrhizal fungi from phylum to species level. New Phytol 193:970–984
Kržišnik D, Gonçalves J (2023) Environmentally conscious technologies using fungi in a climate-changing world. Earth 4:69–77
Krzyzaniak Y, Magnin-Robert M, Randoux B, Fontaine J, Lounès-Hadj Sahraoui ALH (2021) Combined use of beneficial bacteria and arbuscular mycorrhizal fungi for the biocontrol of plant cryptogamic diseases: evidence, methodology, and limits. Symbiotic Soil Microorg 429–468
Kuhnert E, Navarro-Muñoz JC, Becker K, Stadler M; Collemare J; Cox RJ (2021) Secondary metabolite biosynthetic diversity in the fungal family Hypoxylaceae and Xylaria hypoxylon. Stud Mycol 99:100-118. https://doi.org/10.1016/j.simyco.2021.100118
Kularathnage ND, Tennakoon DS, Zhu X, Zhou J (2023) Reinstating dyfrolomyces and introducing Melomastia pyriformis sp. nov. (Pleurotremataceae, Dyfrolomycetales) from Guangdong Province,China. Curr Res Environ Appl Mycol 13:426–438
Kulshreshtha S, Mathur N, Bhatnagar P (2014) Mushroom as a product and their role in mycoremediation. AMB Express 4:1–7
Kumar A, Yadav AN, Mondal R, Kour D, Subrahmanyam G, Shabnam AA, Malyan SK (2021) Myco-remediation: a mechanistic understanding of contaminants alleviation from natural environment and future prospect. Chemosphere 284:131325. https://doi.org/10.1016/j.chemosphere.2021.131325
Kumar K, Mehra R, Guiné RPF, Lima MJ, Kumar N, Kaushik R, Ahmed N, Yadav AN, Kumar H (2021) Edible mushrooms: a comprehensive review on bioactive compounds with health benefits and processing aspects. Foods 10:2996. https://doi.org/10.3390/foods10122996
Kumar NS (2023) Smart and innovative nanotechnology applications for water purification. Hybrid Adv 3:100044. https://doi.org/10.1016/j.hybadv.2023.100044
Kumar RS, Singh D, Bose SK, Trivedi PK (2020) Biodegradation of environmental pollutant through pathways engineering and genetically modified organisms approaches. In: Microorganisms for sustainable environment and health. Elsevier, New York, pp 137–165
Kumar S, Kumar D, Dilbaghi N (2017) Preparation, characterization, and bio-efficacy evaluation of controlled release carbendazim-loaded polymeric nanoparticles. Environ Sci Pollut Res 24:926–937
Kumarappan N, SudharsanP HC (2018) A comparative analysis of bacteria made bio brick and conventional brick. Int J Chemtech Res 11:132–136
Kuyper TW, Jansa J (2023) Arbuscular mycorrhiza: advances and retreats in our understanding of the ecological functioning of the mother of all root symbioses. Plant Soil
Kuyper TWJP (2023) Soil. The smartest plant? 1–3
Lacey RF, Sullivan-Hill BA, Deslippe JR, Keyzers RA, Gerth ML (2021) The Fatty Acid Methyl Ester (FAME) profile of Phytophthora agathidicida and its potential use as diagnostic tool. FEMS Microbiol Lett 368:fnab113
Lange L, Pilgaard B, Herbst FA, Busk PK (2019) Origin of fungal biomass degrading enzymes: evolution, diversity, and function of enzymes of early lineage fungi. Fungal Biol Rev 33:82–97
Lanternier F, Barbati E, Meinzer U, Liu L, Pedergnana V, Migaud M, Héritier S, Chomton M, Frémond ML, Gonzales E, Galeotti C, Romana S, Jacquemin E, Angoulvant A, Bidault V, Canioni D, Lachenaud J, Mansouri D, Mahdaviani SA, Adimi P, Mansouri N, Jamshidi M, Bougnoux ME, Abel L, Lortholary O, Blanche S, Casanova JL, Picard C, Puel A (2015) Inherited CARD9 deficiency in 2 unrelated patients with invasive Exophiala infection. J Infect Dis 211:1241–1250
Lee K, Dighton J (2013) Advancement of functional genomics of a model species of Neurospora and its use for ecological genomics of soil fungi. In: Genomics of soil-and plant-associated fungi. Springer, Berlin, pp 29-44
Lehmann A, Rillig MCJSB (2015) Arbuscular mycorrhizal contribution to copper, manganese and iron nutrient concentrations in crops–a meta-analysis. Soil Biol Biochem 81:147–158
Lepère C, Domaizon I, Humbert JF, Jardillier L, Hugoni M, Debroas D (2019) Diversity, spatial distribution and activity of fungi in freshwater ecosystems. PeerJ 7: e6247
Lesa KN, Khandaker MU, Iqbal FMR, Sharma R, Islam F, Mitra S, Emran TB (2022) Nutritional value, medicinal importance, and health-promoting effects of dietary mushroom (pleurotus ostreatus). J Food Qual 2022. https://doi.org/10.1155/2022/2454180
Li GJ, Hyde KD, Zhao RL, Hongsanan S (2016) Fungal diversity notes 253–366: taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers 78:1–237
Li N, Cai Q, Miao Q, Song Z, Fang Y, Hu B (2021a) High-throughput metagenomics for identification of pathogens in the clinical settings. Small Methods 5: 2000792
Li YN, Steenwyk JL, Chang Y, Wang Y, James TY, Stajich JE, Spatafora JW, Groenewald M, Dunn CW, Hittinger CT, Shen XX, Rokas A (2021b) A genome-scale phylogeny of the kingdom Fungi. Curr Biol 31:1653–1665. https://doi.org/10.1016/j.cub.2021.01.074
Li Z, Yao G, Wu R, Gao L (2022a) Integration of metabolomics and functional genomics reveals the metabolic adaptations of Trichoderma reesei under cellulase-inducing conditions. Appl Microbiol Biotechnol 106:909–920
Li X, Zeng S, Wisniewski M, Droby S, Yu L, An F, Leng Y, Wang C, Li X, He M, Liao Q, Liu J, Wang Y, Sui Y (2022b) Current and future trends in the biocontrol of postharvest diseases. Crit Rev Food Sci Nutr 8:1–13
Li W, Cui J, Li J, Guo J, Huang T, Zhang J, Hu H (2022c) Analysis of the fungi community variation during rice storage through high throughput sequencing. Processes 10:754
Liang X, Zhang S, Gadd GM, McGrath J, Rooney DW, Zhao Q (2022) Fungal-derived selenium nanoparticles and their potential applications in electroless silver coatings for preventing pin-tract infections. Regen. Biomater 9:rbac013
Liew ECY, Aptroot A, Hyde KD (2000) Phylogenetic significance of the pseudoparaphyses in loculoascomycete taxonomy. Mol Phylogeny Evol 16: 392–402
Liew ECY, Aptroot A, Hyde KD (2002) An evaluation of the monophyly of Massarina based on ribosomal DNA sequences. Mycologia 94: 803–813
Lin BQ, Li SP (2011) Cordyceps as an herbal drug. In Benzie IFF, Wachtel-Galor S (eds) Herbal medicine: biomolecular and clinical aspects. CRC Press, Broken Sound Parkway, pp 73–106
Lin X, Kück U (2022) Cephalosporins as key lead generation betalactam antibiotics. Appl Microbiol Biotechnol 106:8007–8020. https://doi.org/10.1007/s00253-022-12272-8
Lin L, Zhang T, Xu J (2023a) Genetic and environmental factors influencing the production of select fungal colorants: Challenges and opportunities in industrial applications. J Fungi 9(5):585
Lin Z, Akin H, Rao R, Hie B, Zhu Z, Lu W, Smetanin N, Verkuil R, Kabeli O, Shmueli Y, Costa ADS, Fazel-Zarandi M, Sercu T, Candido S, River A (2023b) Evolutionary-scale prediction of atomic-level protein structure with a language model. Science 379:1123–1130. https://doi.org/10.1126/science.ade2574
Ling KS, Wechter WP, Somai BM, Walcott RR, Keinath AP (2010) An improved real-time PCR system for broad-spectrum detection of Didymella bryoniae, the causal agent of gummy stem blight of cucurbits. Seed Sci Technol 38:692–703. https://doi.org/10.15258/sst.2010.38.3.17
Liu B, Liang X, Kong J, Jiao C, Li H (2023) Population structure and genetic diversity of Colletotrichum gloeosporioides on Citrus in China. Agronomy 13:184
Liu NG, Ariyawansa HA, Hyde KD, Maharachchikumbura SSN, Zhao RL, Phillips AJL, Jayawardena RS, Thambugala KM, Dissanayake AJ, Wijayawardene NN, Liu JK, Liu ZY, Jeewon R, Jones EBG, Jumpathong J (2016) Perspectives into the value of genera, families and orders in classification. Mycosphere 7:1649–1668
Liu Q, Sun X, Wu W, Liu Z, Fang G, Yang P (2022) Agroecosystem services: a review of concepts, indicators, assessment methods and future research perspectives. Ecol Indic 142:109218
Liu R, Xu C, Zhang Q, Wang S, Fang W (2017) Evolution of the chitin synthase gene family correlates with fungal morphogenesis and adaption to ecological niches. Sci Rep 7:44527. https://doi.org/10.1038/srep4452
Longya A, Talumphai S, Jantasuriyarat C (2020) Morphological characterization and genetic diversity of rice blast fungus, Pyricularia oryzae, from Thailand using ISSR and SRAP markers. J Fungi 6:article38
Lorrain C, Gonçalves dos Santos KC, Germain H, Hecker A, Duplessis S (2019) Advances in understanding obligate biotrophy in rust fungi. New Phytol 222:1190-1206
Loshchinina EA, Vetchinkina EP, Kupryashina MA (2023) Diversity of biogenic nanoparticles obtained by the fungi-mediated synthesis: a review. Biomimetics 8:1. https://doi.org/10.3390/biomimetics8010001
Lou DI, Hussmann JA, McBee RM, Acevedo A, Andino R, Press WH, Sawyer SL (2013) High-throughput DNA sequencing errors are reduced by orders of magnitude using circle sequencing. Proc Natl Acad Sci 110:19872–7
Lovering N (2021) Health benefits of lion’s mane. https://www.forbes.com/health/body/health-benefits-of-lions-mane/
Lu H, Lou H, Hu J, Liu Z, Chen Q (2020) Macrofungi: a review of cultivation strategies, bioactivity, and application of mushrooms. Compr Rev Food Sci Food Saf 19:2333–2356
Lübeck M, Lübeck PS (2022) Fungal cell factories for efficient and sustainable production of proteins and peptides. Microorganisms 10:753
Luchi N, Ioos R, Santini A (2020) Fast and reliable molecular methods to detect fungal pathogens in woody plants. Appl Microbiol Biotechnol 104:2453–2468
Lücking R, Aime MC, Robbertse B, Miller AN, Ariyawansa HA, Aoki T, Cardinali G, Crous PW, Druzhinina IS, Geiser DM, Hawksworth DL (2020) Unambiguous identification of fungi: where do we stand and how accurate and precise is fungal DNA barcoding? IMA Fungus 11:1–32
Lücking R, Leavitt SD, Hawksworth DL (2021) Species in lichen-forming fungi: balancing between conceptual and practical considerations, and between phenotype and phylogenomics. Fungal Divers 109:99–154
Luo J, Yin JF, Cai L, Zhang KQ, Hyde, KD (2004) Freshwater fungi in Lake Dianchi, a heavily polluted lake in Yunnan, China. Fungal Divers 16:93–112
Luo ZL, Hyde KD, Liu JK, Maharachchikumbura SS (2019) Freshwater sordariomycetes. Fungal Divers 99:451–660
Ma C, Zhang HH, Wang X (2014) Machine learning for big data analytics in plants. Trends Plant Sci 19:798–808. https://doi.org/10.1016/j.tplants.2014.08.004
Mabey Gilsenan J, Cooley J, Bowyer P (2012) CA DRE: the central aspergillus data repository. Nucleic Acids Res 40:660–666
Mabey JE, Anderson MJ, Giles PF, Miller CJ (2004) CA DRE:the Central Aspergillus Data REpository. Nucleic Acids Res 32:401–405
Madawala HJB (2021) Arbuscular mycorrhizal fungi as biofertilizers: current trends, challenges, and future prospects. Biofertilizers 2021:83–93
Maddalena G, Marone Fassolo E, Bianco PA, Toffolatti SL (2023) Disease forecasting for the rational management of grapevine mildews in the Chianti Bio-District (Tuscany). Plants 12:285
Madhavan A, Arun KB, Sindhu R, Jose AA, Pugazhendhi A, Binod P, Sirohi R, Reshmy R, Awasthi MK (2022) Engineering interventions in industrial filamentous fungal cell factories for biomass valorization. Bioresour Technol 344:126209
Magee PT, Gale C, Berman J, Davis D (2003) Molecular genetic and genomic approaches to the study of medically important fungi. Infection and immunity 71:2299–2309
Maharachchikumbura SS, Chen Y, Ariyawansa HA, Hyde KD et al (2021) Integrative approaches for species delimitation in Ascomycota. Fungal Divers 109:155-179
Mahé S, Duhamel M, Le Calvez T, Guillot L, Sarbu L, Bretaudeau A, Vandenkoornhuyse P (2012) PHYMYCO-DB: a curated database for analyses of fungal diversity and evolution. PLOS ONE 7(9):e43117. https://doi.org/10.1371/journal.pone.0043117
Mahesh B (2020) Machine learning algorithms-a review. Int J Sci Res 9:381–386. https://doi.org/10.21275/ART20203995
Malapi-Wight M, Salgado-Salazar C, Demers JE, Clement DL, Rane KK, Crouch JA (2016) Sarcococca blight: Use of whole-genome sequencing for fungal plant disease diagnosis. Plant Disease 100:1093–1100
Malhi Y, Adu-Bredu S, Asare RA, Lewis SL, Mayaux P (2013) African rainforests: past, present and future. Philos Trans R Soc B 368:20120312
Malik S, Muhammad K, Waheed Y (2023) Nanotechnology: a revolution in modern industry. Molecules 28(2):661. https://doi.org/10.3390/molecules28020661
Manan S, Ullah MW, Ul-Islam M, Atta OM,Yang G (2021) Synthesis and applications of fungal mycelium-based advanced functional materials. J Bioresour Bioprod 6:1–10
Manawasinghe IS, Dissanayake AJ, Li X, Liu M (2019) High genetic diversity and species complexity of Diaporthe associated with grapevine dieback in China. Front Microbiol 10:19-36
Manawasinghe IS, Phillips AJ, Xu J, Balasuriya A (2021) Defining a species in fungal plant pathology: beyond the species level. Fungal Divers 109:267-282
Manawasinghe IS, Zhang YX, Liao CF, Xiong YR (2022) Mycosphere notes 345–386. Mycosphere, 13:454–557
Manzi P, Pizzoferrato L (2000) Beta-glucans in edible mushrooms. Food Chem 68:315-318
Mao XM, Xu W, Li D, Yin WB, Chooi YH, Li YQ, Tang Y, Hu Y (2015) Epigenetic genome mining of an endophytic fungus leads to the pleiotropic biosynthesis of natural products. Angew Chem Int Ed 54:7592–7596. https://doi.org/10.1002/anie.201502452
Mapook A, Hyde KD, Hassan K, Matio Kemkuignou B et al (2022) Ten decadal advances in fungal biology leading towards human well-being. Fungal Divers 116:547–614
Market Research Report (2023) Global medicinal market overview. https://www.marketresearchfuture.com/reports/medicinal-mushroom-extract-market-4737
Märtson AG, Alffenaar JWC, Brüggemann RJ, Hope W (2021) Precision therapy for invasive fungal diseases. J Fungi (Basel) 8(1):18. https://doi.org/10.3390/jof8010018
Massart S, Adams I, Al Rwahnih M, Baeyen S (2022) Guidelines for the reliable use of high throughput sequencing technologies to detect plant pathogens and pests. Peer Community J. https://doi.org/10.24072/pcjournal.181
Massi F, Torriani SF, Borghi L, Toffolatti SL (2021) Fungicide resistance evolution and detection in plant pathogens: plasmopara viticola as a case study. Microorganisms 9:119
Matchado MS, Lauber M, Reitmeier S, Kacprowski T, Baumbach J, Haller D, List M (2021) Network analysis methods for studying microbial communities: a mini review. Computational and structural biotechnology journal 19:2687-2698. https://doi.org/10.1016/j.csbj.2021.05.001
Matsuda Y, Abe I (2016) Biosynthesis of fungal meroterpenoids. Nat Prod Rep 33:26–53
Matute DR, Sepúlveda VE (2019) Fungal species boundaries in the genomics era. Fungal Genet Biol 131:103249. https://doi.org/10.1016/j.fgb.2019.103249
Maviane-Macia F, Ribeyre C, Buendia L, Gaston M (2019) Experimental system and image analysis software for high throughput phenotyping of mycorrhizal growth response in Brachypodium distachyon. bioRxiv 779330
Mazzola M, Freilich S (2017) Prospects for biological soilborne disease control: application of indigenous versus synthetic microbiomes. Phytopathology 107:256–263
Mbareche H, Veillette M, Bilodeau GJ, Duchaine C (2019) Fungal aerosols at dairy farms using molecular and culture techniques. Sci Total Environ 653:253–263
McCluskey K, Wiest A, Plamann M (2010) The Fungal Genetics Stock Center: a repository for 50 years of fungal genetics research. J Biosci 35:119−126
McGuire AL, Gabriel S, Tishkoff SA, Wonkam A, Chakravarti A, Furlong EEM, Treutlein B, Meissner A, Chang HY, López-Bigas N, Segal E, Kim JS (2020) The road ahead in genetics and genomics. Nat Rev Genet 21:581–596. https://doi.org/10.1038/s41576-020-0272-6
Meghvansi MK, Khan MH, Gupta R, Veer V (2013) Identification of a new species of Cercospora causing leaf spot disease in Capsicum assamicum in northeastern India. Res Microbiol. 164(9):894–902. https://doi.org/10.1016/j.resmic.2013.08.003
Mei LL, Yang X, Zhang SQ, Zhang T, Guo JX (2019) Arbuscular mycorrhizal fungi alleviate phosphorus limitation by reducing plant N:P ratios under warming and nitrogen addition in a temperate meadow ecosystem. Sci Total Environ 686:1129-1139
Meng TX, Furuta S, Fukamizu S, Yamamoto R (2011) Evaluation of biological activities of extracts from the fruiting body of Pleurotus citrinopileatus for skin cosmetics. J Wood Sci 57:452–458
Mengqi Z, Shi A, Ajmal M, Ye L, Awais M (2021). Comprehensive review on agricultural waste utilization and high-temperature fermentation and composting. Biomass Convers Biorefin 13:5445–5468
Meyer V, Andersen MR, Brakhage AA, Braus GH, Caddick MX, Cairns TC, de Vries RP, Haarmann T, Hansen K, Hertz-Fowler C, Krappmann S (2016) Current challenges of research on filamentous fungi in relation to human welfare and a sustainable bio-economy: a white paper. Fungal Biol Biotechnol 3:1–17
Meyer V, Basenko EY, Benz JP, Braus GH, Caddick MX, Csukai M, De Vries RP, Endy D, Frisvad JC, Gunde-Cimerman N, Haarmann T (2020) Growing a circular economy with fungal biotechnology: a white paper. Fungal Biol Biotechnol 7:1–23
Meyer V, Cairns T, Barthel L, King R, Kunz P, Schmideder S, Müller H, Briesen H, Dinius A, Krull R (2021) Understanding and controlling filamentous growth of fungal cell factories: novel tools and opportunities for targeted morphology engineering. Fungal Biol Biotechnol 8:8
Meyers MA, Mishra A, Benson DJ (2006) Mechanical properties of nanocrystalline materials. Progress in Materials Science 51:427–556
Michalecka M, Bryk H, Poniatowska A, Puławska J (2016) Identification of Neofabraea species causing bull’s eye rot of apple in Poland and their direct detection in apple fruit using multiplex PCR. Plant Pathol 65:643–654
Miethke M, Pieroni M, Weber T, Brönstrup M et al (2021) Towards the sustainable discovery and development of new antibiotics. Nat Rev Chem 5:726–749
Miller AN, Bates ST (2017) The mycology collections portal (mycoportal). IMA Fungus 8:65–66
Miller SA, Martin RR (1988) Molecular diagnosis of plant diseases. Ann Rev Phytopathol 26:409–432
Minter DW (2011) The Ascomycota and the sampled red list index scheme. Fungal Conserv 1:45–53
Minter DW (2015) Cybertruffle: an online resource for mycology. Descr Taxon 84: 96
Mishra VK, Kumar A (2009) Impact of metal nanoparticles on the plant growth promoting rhizobacteria. Dig J Nanomater Biostruct 4:587–592
Mitra S, Sultana SA, Prova SR, Uddin TM, Islam F, Das R (2022) Investigating forthcoming strategies to tackle deadly superbugs: current status and future vision. Expert Rev Anti Infect Ther 20:1309–1332. https://doi.org/10.1080/14787210.2022.2122442
Miyake M, Yamamoto S, Sano O, Fujii M, Kohno K, Ushio S, Iwaki K, Fukuda S (2010) Inhibitory effects of 2-amino-3H-phenoxazin-3one on the melanogenesis of murine B16 melanoma cell line. Biosci Biotechnol Biochem 74:753–758
Miyauchi S, Kiss E, Kuo A, Drula E, Kohler A, Sánchez-García M, Morin E, Andreopoulos B, Barry KW, Bonito G, Buée M (2020) Large-scale genome sequencing of mycorrhizal fungi provides insights into the early evolution of symbiotic traits. Nat Commun 11:5125
Mohana S, Sumathi S (2020) Multi-functional biological effects of palladium nanoparticles synthesized using Agaricus bisporus. J Clust Sci 31:391–400
Molina-Castillo S, Espinoza-Ortega A, Thomé-Ortiz H, Moctezuma-Pérez S (2023) Gastronomic diversity of wild edible mushrooms in the Mexican cuisine. Int J Gastronom Food Sci 31:100652
Möller M, Stukenbrock EH (2017) Evolution and genome architecture in fungal plant pathogens. Nat Rev Microbiol 15: 756–771
Momin M, Webb G (2021) The environmental effects on virulence factors and the antifungal susceptibility of Cryptococcus neoformans. Int J Mol Sci 22:6302
Moond M, Singh S, Sangwan S, Devi R, Beniwal R (2022) Green aynthesis and applications of silver nanoparticles: a systematic review. AATCC J Res 9:272–285. https://doi.org/10.1177/24723444221119847
Moreno‐Jiménez E, Leifheit EF, Plaza C, Feng L, Bergmann J, Wulf A (2022) Effects of microplastics on crop nutrition in fertile soils and interaction with arbuscular mycorrhizal fungi. J Sustain Agric Environ 1:66–72
Mota IFL, de Lima Ls, Santana B. de M,Gobbo G de AM, Bicca JVML, Azevedo JRM, Veras LG, Taveira R de AA, Pinheiro GB, Mortari MR (2023) Alzheimer’s disease: innovative therapeutic approaches based on peptides and nanoparticles. Neuroscientist 29:78–96. https://doi.org/10.1177/10738584211016409
Mora C, Tittensor DP, Adl S, Simpson AG (2011) How many species are there on Earth and in the ocean?. PLoS Biol 9:e1001127
Mukherjee P, Ahmad A, Mandal D, Senapati S, Sainkar SR, Khan MI, Parishcha R, Ajaykumar PV, Alam M, Kumar R, Sastry M (2001) Fungus-mediated synthesis of silver nanoparticles and their immobilization in the mycelial matrix: a novel biological approach to nanoparticle synthesis. Nano Lett 1:515–519
Muller EE, Glaab E, May P (2013) Condensing the omics fog of microbial communities. Trends Microbiol 21:325–333
Muñoz AHS, Kubachka K, Wrobel K, Corona JFG, Yathavakilla SKV, Caruso JA, Wrobel K (2006) Se-enriched mycelia of pleurotus ostreatus: distribution of selenium in cell walls and cell membranes/cytosol. J Agric Food Chem 54:3440–3444
Nagler M, Podmirseg SM, Mayr M, Ascher‐Jenull J, Insam H (2021) The masking effect of extracellular DNA and robustness of intracellular DNA in anaerobic digester NGS studies: a discriminatory study of the total DNA pool. Mol Ecol 30:438-450
Navarro D, Chaduli D, Taussac S, Lesage-Meessen L, Grisel S, Haon M, Callac P, Courtecuisse R, Decock C, Dupont J, Richard-Forget F (2021) Large-scale phenotyping of 1,000 fungal strains for the degradation of non-natural, industrial compounds. Commun Biol 4:871
Nawawi WM, Jones M, Murphy RJ, Lee KY, Kontturi E, Bismarck A (2020) Nanomaterials derived from fungal sources—is it the new hype? Biomacromolecules 21:30–55. https://doi.org/10.1021/acs.biomac.9b01141
Ndayihanzamaso P, Karangwa P, Mostert D, Mahuku G, Blomme G, Beed F (2020) The development of a multiplex PCR assay for the detection of Fusarium oxysporum f. sp. cubense lineage VI strains in East and Central Africa. Eur. J. Plant Pathol. 158:495–509
Nehara HR, Puri I, Singhal VIS, Bishnoi BR, Sirohi P (2021) Rhinocerebral mucormycosis in COVID-19 patient with diabetes a deadly trio:case series from the north-western part of India. Indian J Med Microbiol 39:380-383
Newman DJ, Cragg GM (2020) Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J Nat Prod 83:770–803. https://doi.org/10.1021/acs.jnatprod.9b01285
Nguyen HDT, Dodge A, Dadej K, Rintoul TL, Ponomareva E, Martin FN, de Cock AWAM, Lévesque CA, Redhead SA, Spies CFJ (2022) Whole genome sequencing and phylogenomic analysis show support for the splitting of genus Pythium. Mycologia 114:501–515. https://doi.org/10.1080/00275514.2022.2045116
Niego AGT, Rapior S, Thongklang N, Raspé O et al (2021) Macrofungi as a nutraceutical source: Promising bioactive compounds and market value. J Fungi 7:397. https://doi.org/10.3390/jof7050397
Niego AGT, Lambert C, Mortimer P, Thongklang N, Rapior S, Grosse M, Schrey H, Charria-Giron E, Walker A, Hyde KD, Stadler M (2023a) The contribution of fungi to the global economy. Fungal Divers 1–43
Niego AGT, Rapior S, Thongklang N, Raspé O, Hyde KD, Mortimer P (2023b) Reviewing the contributions of macrofungi to forest ecosystem processes and services. Fungal Biol Rev 44:100294
Nilsson RH, Ryberg M, Kristiansson E, Abarenkov K (2006) Taxonomic reliability of DNA sequences in public sequence databases:a fungal perspective. PLoS ONE 1:e59
Nilsson RH, Hyde KD, Pawlowska J, Ryberg M (2014) Improving ITS sequence data for identification of plant pathogenic fungi. Fungal Divers 67:11–19
Nilsson RH, Anslan S, Bahram M (2019a) Mycobiome diversity:high-throughput sequencing and identification of fungi. Nat Rev Microbiol 17:95–109
Nilsson RH, Larsson KH, Taylor AF, Bengtsson-Palme J, Jeppesen TS, Schigel D, Kennedy P, Picard K, Glöckner FO, Tedersoo L, Saar I, Kõljalg U, Abarenkov K (2019b) The UNITE database for molecular identification of fungi: handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res 47:259–264
Noble LM, Andrianopoulos A (2013) Fungal genes in context: genome architecture reflects regulatory complexity and function. Genome Biol Evol 5:1336–1352
Nofiani R, de Mattos-Shipley K, Lebe KE, Han LC, Iqbal Z, Bailey AM, Willis CL, Simpson TJ, Cox RJ (2018) Strobilurin biosynthesis in basidiomycete fungi. Nat Commun 9(1):3940
Norphanphoun C, Jayawardena RS, Chen Y, Wen TC (2019) Morphological and phylogenetic characterization of novel pestalotioid species associated with mangroves in Thailand. Mycosphere, 10:531–578
Norphanphoun C, Gentekaki E, Hongsanan S, Jayawardena R, Senanayake IC, Manawasinghe IS, Abeywickrama PD, Bhunjun CS, Hyde KD (2022) Diaporthe: formalizing the species-group concept. Mycosphere 13:752–819
Noumeur SR, Teponno RB, Helaly SE (2020) Diketopiperazines from Batnamyces globulariicola, gen. & sp. nov. (Chaetomiaceae), a fungus associated with roots of the medicinal plant Globularia alypum in Algeria. Mycol Prog 19:589–603. https://doi.org/10.1007/s11557-020-01581-9
Ogaki MB, Pinto OH, Vieira R (2021a) Fungi present in Antarctic deep-sea sediments assessed using DNA metabarcoding. Microb Ecol 1:1–8
Ogaki MB, Câmara PE, Pinto OH (2021b) Diversity of fungal DNA in lake sediments on Vega Island, north-east Antarctic Peninsula assessed using DNA metabarcoding. Extremophiles 25:257–65
Okal EJ, Heng G, Magige EA, Khan S, Wu S, Ge Z, Zhang T, Mortimer PE, Xu J (2023) Insights into the mechanisms involved in the fungal degradation of plastics. Ecotoxicol Environ Saf 262:115202
Okuda Y (2022) Sustainability perspectives for future continuity of mushroom productiont: the bright and dark sides. Front Sustain Food Syst 6:1026508
Oliveira LVN, Wang R, Specht CA, Levitz SM (2021) Vaccines for human fungal diseases: close but still a long way to go. npj Vaccines 6:33
Olsen L, Choffnes ER, Relman DA, Rapporteurs LP (2011) Fungal diseases: an emerging threat to human, animal, and plant health: workshop summary. forum on microbial threats. The National Academic Press, Washington
Omotayo OP, Omotayo AO, Mwanza M, Babalola OO (2019) Prevalence of mycotoxins and their consequences on human health. Toxicol Res 35:1–7
Opik M, Davison J, Moora M, Zobel M (2014) DNA-based detection and identification of Glomeromycota: the virtual taxonomy of environmental sequences. Botany 92:135-147
Orchard S, Hilton S, Bending GD, Dickie IA, Standish RJ, Gleeson DB (2017) Fine endophytes (Glomus tenue) are related to Mucoromycotina, not Glomeromycota. New Phytol 213:481–486
Orth AB, Royse DJ, Tien MING (1993) Ubiquity of lignin-degrading peroxidases among various wood-degrading fungi. Appl Environ Microbiol 59:4017-4023
Osaigbovo II, Bongomin F (2021) Point of care tests for invasive fungal infections: a blueprint for increasing availability in Africa. Therap Adv Infect Dis Jan-Dec:8. https://doi.org/10.1177/20499361211034266
Osono T (2007) Ecology of ligninolytic fungi associated with leaf litter decomposition. Ecol Res 22:955–974
Osorio JA, Crous CJ, De Beer ZW, Wingfield MJ, Roux J (2017) Endophytic Botryosphaeriaceae, including five new species, associated with mangrove trees in South Africa. Fungal Biol 121:361–393
Otero-Muras I, Carbonell P (2021) Automated engineering of synthetic metabolic pathways for efficient biomanufacturing. Metab Engin 63:61–80
Otun S, Achilonu I (2023) Recent advances in plant: arbuscular mycorrhizal fungi associations and their application to cassava crops
Padamsee M, Kumar TKA, Riley R, Binder M, Boyd A, Calvo AM, Furukawa K, Hesse C, Hohmann S, James TY, LaButti K, Lapidus A, Lindquist E, Lucas S, Miller K, Shantappa S, Grigoriev IV, Hibbett DS, McLaughlin DJ, Spatafora JW, Aime MC (2012) The genome of the xerotolerant mold Wallemia sebi reveals adaptations to osmotic stress and suggests cryptic sexual reproduction. Fungal Genet Biol 49:217–226. https://doi.org/10.1016/j.fgb.2012.01.007
Paramasivam G, Palem VV, Sundaram T, Sundaram V, Kishore SC, Bellucci S (2021) Nanomaterials: synthesis and applications in theranostics. Nanomaterials (Basel) 11:3228. https://doi.org/10.3390/nano11123228
Park H, Choi IG (2020) Genomic and transcriptomic perspectives on mycoremediation of polycyclic aromatic hydrocarbons. Appl Microbiol Biotechnol 104:6919–6928. https://doi.org/10.1007/s00253-020-10746-1
Park KM, Kwon KM, Lee SH (2015) Evaluation of the antioxidant activities and tyrosinase inhibitory property from mycelium culture extracts. Environ Based Compl Alt Med 2015:1–7
Pasinszki T, Krebsz M (2020) Synthesis and application of zero-valent iron nanoparticles in water treatment, environmental remediation, catalysis, and their biological effects. Nanomaterials 10:917
Paukner S, Riedl R (2017) Pleuromutilins: potent drugs for resistant bugs-mode of action and resistance. Cold Spring Harbor Perspect Med 7:a027110. https://doi.org/10.1101/cshperspect.a027110
Peer D, Karp J, Hong S, Farokhzad OC, Margalit M, Langer R (2007) Nanocarriers as an emerging platform for cancer therapy. Nature Nanotech 2:751–760. https://doi.org/10.1038/nnano.2007.387
Pem D, Hongsanan S, Doilom M, Tibpromma S, Wanasinghe DN, Dong W, Liu NG, Phookamsak R, Phillips AJL, Jeewon R, Hyde KD (2019). https://www.dothideomycetes.org: an online taxonomic resource for the identification, dothideomycetes. Asian J Mycol 2:287–297
Peng J, Aluthmuhandiram JVS, Chethana KWT, Zhang Q, Xing Q, Wang H, Liu M, Zhang W, Li X, Yan J (2022) An NmrA-Like protein, Lws1, is important for pathogenesis in the woody plant pathogen Lasiodiplodia theobromae. Plants 11:2197
Pépin N, Punja ZK, Joly DL (2018) Occurrence of powdery mildew caused by Golovinomyces cichoracearum sensu lato on Cannabis sativa in Canada. Plant Dis 102: 2644
Pérez Montes A, Rangel-Vargas E, Lorenzo JM, Romero L, Santos EM (2021) Edible mushrooms as a novel trend in the development of healthier meat products. Curr Opin Food Sci 37:118-134. https://doi.org/10.1016/j.cofs.2020.10.004
Phan TTV, Huynh TC, Manivasagan P, Mondal S, Oh J (2019) An up-to-date review on biomedical applications of palladium nanoparticles. Nanomaterials 10:66
Phillips SJ, Dudik M, Elith J, Graham CH, Lehmann A, Leathwick J, Ferrier S (2009) Sample selection bias and presence-only distribution models: implications for background and pseudo-absence data. Ecol Appl 19:181–197. https://doi.org/10.1890/07-2153.1
Phukhamsakda C, Nilsson RH, Bhunjun CS, de Farias ARG, Sun YR, Wijesinghe SN, Raza M, Bao DF, Lu L, Tibpromma S, Dong W, Tennakoon DS, Tian XG, Xiong YR, Karunarathna SC, Cai L, Luo ZL, Wang Y, Manawasinghe IS, Camporesi E, Kirk PM, Promputtha I, Kuo CH, Su HY, Doilom M, Li Y, Fu YP, Hyde KD (2022) The numbers of fungi: contributions from traditional taxonomic studies and challenges of metabarcoding. Fungal Divers 114:327–386
Piombo E, Abdelfattah A, Droby S, Wisniewski M, Spadaro D, Schena L (2021) Metagenomics approaches for the detection and surveillance of emerging and recurrent plant pathogens. Microorganisms 9:188
Piret J, Boivin G (2021) Pandemics throughout history. Front Microbiol 11:631736. https://doi.org/10.3389/fmicb.2020.631736
Pizarro D, Divakar PK, Grewe F, Leavitt SD, Huang JP, Dal Grande F, Schmitt I, Wedin M, Crespo A, Lumbsch HT (2018) Phylogenomic analysis of 2556 single-copy protein-coding genes resolves most evolutionary relationships for the major clades in the most diverse group of lichen-forming fungi. Fungal Divers 92:31–41. https://doi.org/10.1007/s13225-018-0407-7
Plazas E, Faraone N (2023) Indole alkaloids from psychoactive mushrooms: chemical and pharmacological potential as psychotherapeutic agents. Biomedicines 11:461
Plissonneau C, Benevenuto J, Mohd-Assaad N, Fouché S, Hartmann FE, Croll D (2017) Using population and comparative genomics to understand the genetic basis of effector-driven fungal pathogen evolution. Front Plant Sci 8:119
Polizeli MLTM, Rizzatti ACS, Monti R, Terenzi HF, Jorge JA, Amorim DS (2005) Xylanases from fungi: properties and industrial applications. Appl Microbiol Biotechnol 67:577–591
Põlme S, Abarenkov K, Nilsson HR (2020) FungalTraits: a user-friendly traits database of fungi and fungus-like stramenopiles. Fungal Divers 105:1–6
Ponmurugan P, Manjukarunambika K, Elango V, Gnanamangai BM (2016) Antifungal activity of biosynthesised copper nanoparticles evaluated against red root-rot disease in tea plants. J Exp Nanosci 11:1019–31
Porto BN, Caixeta ET, Mathioni SM, Vidigal PMP, Zambolim L, Zambolim EM, Donofrio N, Polson SW, Maia TA, Chen C, Adetunji M (2019) Genome sequencing and transcript analysis of Hemileia vastatrix reveal expression dynamics of candidate effectors dependent on host compatibility. PLoS ONE 14:e0215598
Porto EM, Komor AC, Slaymaker IM, Yeo GW (2020) Base editing: advances and therapeutic opportunities. Nat Rev Drug Discov 19:839–859
Potgieter L, Feurtey A, Dutheil JY, Stukenbrock EH (2020) On variant discovery in genomes of fungal plant pathogens. Front Microbiol 11:626
Pouliot A (2023) The Guardian 20 March
Powell JR, Rillig MC (2018) Biodiversity of arbuscular mycorrhizal fungi and ecosystem function. New Phytol 220:1059-1075
Prasad R, Kumar V, Prasad KS (2014) Nanotechnology in sustainable agriculture: present concerns and future aspects. Afr J Biotechnol 13:705–713
Prasad S, Rathore H, Sharma S, Yadav AS (2015) Medicinal mushrooms as a source of novel functional food. IJFS 4, 221–225
Prasannath K, Galea VJ, Akinsanmi OA (2020) Characterisation of leaf spots caused by Neopestalotiopsis clavispora and Colletotrichum siamense in macadamia in Australia. Eur J Plant Pathol 156:1219–1225
Prattes J, Heldt S, Eigl S, Hoenigl M (2016) Point of care testing for the diagnosis of fungal infections: are we there yet? Curr Fungal Infect Rep 10:43–50. https://doi.org/10.1007/s12281-016-0254-5
Prescott TA, Hill R, Mas-Claret E, Gaya E, Burns E (2023) Fungal drug discovery for chronic disease: history, new discoveries and new approaches. Biomolecules 13: 986
Presley GN, Zhang J, Purvine SO, Schilling JS (2020) Functional genomics, transcriptomics, and proteomics reveal distinct combat strategies between lineages of wood-degrading fungi with redundant wood decay mechanisms. Front Microbiol 11:1646
Priest SJ, Yadav V, Heitman J (2020) Advances in understanding the evolution of fungal genome architecture. F1000Research 9:776
Przybyla L, Gilbert LA (2022) A new era in functional genomics screens. Nat Rev Genet 23:89–103
Purahong W, Hyde KD (2011) Effects of fungal endophytes on grass and non-grass litter decomposition rates. Fungal Divers 47:1–7
Qu X, Li M, Zhang H, Lin C, Wang F, Xiao M, Zhou Y, Shi J, Aldalbahi A, Pei H, Chen H, Li L (2017) Real-time continuous identification of greenhouse plant pathogens based on recyclable microfluidic bioassay system. ACS Appl Mater Interfaces 9:31568–31575
Quang DN, Hashimoto T, Stadler M, Asakawa Y (2004) New azaphilones from the inedible mushroom Hypoxylon rubiginosum. J Nat Prod 67:1152–1155. https://doi.org/10.1021/np040063l
Quinn J, Modell V, Orange JS, Modell F (2022) Growth in diagnosis and treatment of primary immunodeficiency within the global Jeffrey Modell Centers Network. Allergy Asthma Clin Immunol 18:19. https://doi.org/10.1186/s13223-022-00662-6
Rader RA (2008) (Re) defining biopharmaceutical. Nat Biotechnol 26:743–751. https://doi.org/10.1038/nbt0708-743
Rafique M, Sadaf I, Rafique MS, Tahir MB (2017a) A review on green synthesis of silver nanoparticles and their applications. Artif. Cells Nanomed. Biotechnol 45:1272–1291
Rafique M, Shaikh AJ, Rasheed R, Tahir MB, Bakhat HF, Rafique MS, Rabbani F (2017b) A review on synthesis, characterization and applications of copper nanoparticles using green method. NANO: Brief Rep Rev 12:1750043
Raghavendra VB, Venkitasamy C, Pan Z, Nayak C (2017) Functional foods from mushroom. In: Microbial functional foods and nutraceuticals, pp 65–91
Rai M, Yadav A, Ingle AP, Reshetilov A, Blanco-Prieto MJ, Feitosa CM (2019) Neurodegenerative diseases: the real problem and nanobiotechnological solutions. In: Rai M, Yadav A (eds) Nanobiotechnology in neurodegenerative diseases. Springer, New York pp 1–17
Raj M, Jeeva ML, Nath VS, Sankar S, Vidhyadharan P, Archana PV, Hedge V (2013) A highly sensitive nested-PCR method using a single closed tube for the detection of Colletotrichum gloeosporioides causing greater yam anthracnose. J Root Crops 39:163–167
Rajapaksha P, Elbourne A, Gangadoo S, Brown R, Cozzolino D, Chapman J (2019) A review of methods for the detection of pathogenic microorganisms. Analyst 144:396–411
Rajendran C, Khaitan BK, Mittal R, Ramam M, Bhardwaj M, Datta KK (2003) Phaeohyphomycosis caused by Exophiala spinifera in India. Med Mycol 41:437-441
Rämä T, Hassett BT, Bubnova E (2017) Arctic marine fungi:from filaments and flagella to operational taxonomic units and beyond. Bot Mar 60:433–52
Raman J, Kim DS, Kim HS, Oh DS, Shin HJ (2022) Mycofabrication of mycelium-based leather from brown-rot fungi. J Fungi 8:317
Rambold G, Stadler M, Begerow D (2013) Mycology should be recognized as a field in biology at eye level with other major disciplines–a memorandum. Mycological Progress 12:455-463
Rangel DEN, Braga GUL, Fernandes ÉKK, Keyser CA, Hallsworth JE, Roberts DW (2015) Stress tolerance and virulence of insect-pathogenic fungi are determined by environmental conditions during conidial formation. Curr Genet 61:383–404. https://doi.org/10.1007/s00294-015-0477-y
Rangel-Vargas E, Rodriguez JA, Domínguez R, Lorenzo JM, Sosa ME, Andrés SC, Rosmini M, Pérez-Alvarez JA, Teixeira A, Santos EM (2021) Edible mushrooms as a natural source of food ingredient/additive replacer. Foods 10:11. https://doi.org/10.3390/foods10112687
Ratan ZA, Haidere MF, Nurunnabi M, Shahriar SM, Ahammad AJS, Shim YY, Reaney MJT, Cho JY (2020) Green chemistry synthesis of silver nanoparticles and their potential anticancer effects. Cancers 12:855
Ratnasingham S, Hebert PD (2007) BOLD: The Barcode of Life Data System (http://www.barcodinglife.org). Molecular ecology notes 7:355−364
Raza MM, Harding C, Liebman M, Leandro LF (2020) Exploring the potential of high-resolution satellite imagery for the detection of soybean sudden death syndrome. Remote Sens 12:1213
Razak NHA, Zawawi NA, Chundawat TS (2021) Brief review on bioresources green synthesis of silver nanoparticles. J Adv Res Mater Sci 76:1–10
Razif MF, Fung SY (2021) Cosmeceuticals from mushrooms. In Advances in macrofungi. CRC Press, Boca Raton, pp 230–251
Ren GC, Pang AM, Gao Y, Wu SX, Ge ZQ, Zhang TF, Wanasinghe DN, Khan S, Mortimer PE, Xu JC, Gui H (2021) Polyurethane-degrading fungi from soils contaminated with rocket propellant and their ability to decompose alkyne terminated polybutadiene with urethane. Stud Fungi 6:224–239
Richter F, Bindschedler S, Calonne-Salmon M, Declerck S, Junier P, Stanley CE (2022) Fungi-on-a-chip: microfluidic platforms for single-cell studies on fungi. FEMS Microbiol Rev 46(6):039. https://doi.org/10.1093/femsre/fuac039
Rincon-Florez VA, Carvalhais LC, Schenk PM (2013) Culture-independent molecular tools for soil and rhizosphere microbiology. Diversity 5:581–612
Rinschen MM, Ivanisevic J, Giera M, Siuzdak G (2019) Identification of bioactive metabolites using activity metabolomics. Nat Rev Mol Cell Biol 20:353–367
Rivero RM, Mittler R, Blumwald E, Zandalinas SI (2022) Developing climate‐resilient crops: improving plant tolerance to stress combination. Plant J 109:373–389
Rizzo D, Garbelotto M, Davidson J, Slaughter G, Koike S (2002) Phytophthora ramorum as the cause of extensive mortality of Quercus spp. and Lithocarpus densiflorus in California. Plant Disease 86:205–214
Robert V, Stegehuis G, Stalpers J (2005) The MycoBank engine and related databases. https://www.MycoBank.org/
Robert V, Vu D, Amor AB, van de Wiele N (2013) MycoBank gearing up for new horizons. IMA Fungus 4:371–379
Robey MT, Caesar LK, Drott MT, Keller NP, Kelleher NL (2021) An interpreted atlas of biosynthetic gene clusters from 1,000 fungal genomes. PNAS 118(19):e2020230118. https://doi.org/10.1073/pnas.2020230118
Roth MG, Westrick NM, Baldwin TT (2023) Fungal biotechnology: from yesterday to tomorrow. Front Fungal Biol 4: 1135263
Rousta N, Aslan M, Yesilcimen Akbas M, Ozcan F, Sar T, Taherzadeh MJ (2023) Effects of fungal based bioactive compounds on human health. Crit Rev Food Sci Nutr 1–24
Ruksiriwanich W, Sirithunyalug J, Boonpisuttinant K, Jantrawut P (2014) Potent in vitro collagen biosynthesis stimulating and antioxidant activities of edible mushroom Volvariella volvacea aqueous extract. Int J Pharm Pharmaceut Sci 2:406–412
Runnel K, Abarenkov K, Copoț O (2022) DNA barcoding of fungal specimens using PacBio long‐read high‐throughput sequencing. Mol Ecol Resour 22:2871–9
Ryan MJ, McCluskey K, Verkleij G, Robert V, Smith D (2019) Fungal biological resources to support international development: challenges and opportunities World J Microbiol Biotechnol 35:139
Sadh PK, Duhan S, Duhan JS (2018) Agro-industrial wastes and their utilization using solid state fermentation: a review. Bioresour Bioprocess 5:1–15
Safarpour H, Safarnejad MR, Tabatabaei M, Mohsenifar A, Rad F, Basirat M, Shahryari F, Hasanzadeh F (2012) Development of a quantum dots FRET-based biosensor for efficient detection of Polymyxa betae. Can J Plant Pathol 34:507–515
Saif S, Tahir A, Chen Y (2016) Green synthesis of iron nanoparticles and their environmental applications and implications. Nanomaterials 6:209
Saitoh N, Fujimori R, Yoshimura T, Tanaka H, Kondoh A, Nomura T, Konishi Y (2020) Microbial recovery of palladium by baker’s yeast through bioreductive deposition and biosorption. Hydrometallurgy 196:105413
Saldívar-González FI, Aldas-Bulos VD, Medina-Franco JL, Plisson F (2022) Natural product drug discovery in the artificial intelligence era. Chem Sci 13:1526-1546
Saleem H, Zaidi SJ (2020) Sustainable use of nanomaterials in textiles and their environmental impact. Materials (Basel) 13:5134. https://doi.org/10.3390/ma13225134
Samithsonian Libraries (2023). https://www.sil.si.edu/DigitalCollections/tl-2/search.cfm
Sanchez S, Demain AL (2017) Bioactive products from fungi. Food Bioact. https://doi.org/10.1007/978-3-319-51639-4_3
Sandargo B, Chepkirui C, Cheng T, Chaverra-Muñoz L, Thongbai B, Stadler M, Hüttel S (2019) Biological and chemical diversity go hand in hand: basidiomycota as source of new pharmaceuticals and agrochemicals. Biotechnol Adv. https://doi.org/10.1016/j.biotechadv.2019.01.011
Sangthong S, Pintathong P, Pongsua P, Jirarat A, Chaiwut P (2022) Polysaccharides from Volvariella volvacea mushroom: extraction, biological activities and cosmetic efficacy. J Fungi 8(6):572
Santini A, Ghelardini L (2015) Plant pathogen evolution and climate change. CAB Rev 10:1–35
Santiago FH, Moreno JP, Cázares BX, Suárez JJA, Trejo EO, de Oca GMM, Aguilar ID (2016) Traditional knowledge and use of wild mushrooms by Mixtecs or Ñuu savi, the people of the rain, from Southeastern Mexico. J Ethnobiol Ethnomed 12:1–22
Sanz A, García R, Rodríguez-Peña J, Arroyo J (2017) The CWI pathway: Regulation of the transcriptional adaptive response to cell wall stress in yeast. J Fungi 4:1–12. https://doi.org/10.3390/jof4010001
Sanzari I, Leone A, Ambrosone A (2019) Nanotechnology in plant science: to make a long story short. Front Bioeng Biotechnol 7:120
Sarma VV (2019) Marine fungal diversity: present status and future perspectives. Microbial Divers Ecosyst Sustain Biotechnol Appl 267–291
Schäpe P, Kwon MJ, Baumann B, Gutschmann B (2019) Updating genome annotation for the microbial cell factory Aspergillus niger using gene co-expression networks. Nuc Acids Res 47:559–569
Schatz MC, Philippakis AA, Afgan E, Banks E (2022) Inverting the model of genomics data sharing with the NHGRI Genomic Data Science Analysis, Visualization, and Informatics Lab-space. Cell Genome 2(1):100085
Schikora-Tamarit MÀ, Gabaldón T (2022) Using genomics to understand the mechanisms of virulence and drug resistance in fungal pathogens. Biochem Soc Trans 50:1259–1268
Schlaeppi K, Bender SF, Mascher F, Russo G, Patrignani A, Camenzind T (2016) High-resolution community profiling of arbuscular mycorrhizal fungi. New Phytol 212:780–791
Schmit JP, Mueller GM (2007) An estimate of the lower limit of global fungal diversity. Biodivers Conserv 16:99-111
Schotte C, Li L, Wibberg D, Kalinowski J, Cox RJ (2020) Synthetic biology driven biosynthesis of unnatural tropolone sesquiterpenoids. Angew Chem Int Ed 59:23870-23878
Segal E, Gritsenko V, Levitan A, Yadav B, Dror N, Steenwyk J, Silberberg Y, Mielich K, Rokas A, Gow N, Kunze R, Sharan R, Berman J (2018) Gene essentiality analyzed by in vivo transposon mutagenesis and machine learning in a stable haploid isolate of Candida albicans. mBio 9:e02048–18. https://doi.org/10.1128/mBio.02048-18
Semenov MV (2021) Metabarcoding and metagenomics in soil ecology research:achievements, challenges, and prospects. Biol Bull Rev 11:40–53
Senanayake IC, Pem D, Rathnayaka AR, Wijesinghe SN, Tibpromma S, Wanasinghe DN, Phookamsak R, Kularathnage ND, Gomdola D, Harishchandra D, Dissanayake LS, Xiang XM, Ekanayaka AH, McKenzie EHC, Hyde KD, Zhang HX, Xie N (2022) Predicting global numbers of teleomorphic ascomycetes. Fungal Divers 114:237-278
Senanayake IC, Rathnayaka AR, Marasinghe DS, Calabon MS, Gentekaki E, Lee HB, Hurdeal VG, Pem D, Dissanayake LS, Wijesinghe SN, Bundhun D, Nguyen TT, Goonasekara ID, Abeywickrama PD, Bhunjun CS, Jayawardena RS, Wanasinghe DN, Jeewon R, Bhat DJ, Xiang MM (2020) Morphological approaches in studying fungi: collection, examination, isolation, sporulation and preservation. Mycosphere 11:2678–2754
Seo E, Choi DC (2015) Functional studies of transcription factors involved in plant defenses in the genomics era. Briefings Funct Genomics 14:260–267. https://doi.org/10.1093/bfgp/elv011
Sepúlveda VE, Márquez R, Turissini DA, Goldman WE, Matute DR (2017) Genome sequences reveal cryptic speciation in the human pathogen Histoplasma capsulatum. mBio 8:e01339–01317. https://doi.org/10.1128/mBio.01339-17
Seto K, Simmons DR, Quandt CA, Frenken T, Dirks AC, Clemons RA, McKindles KM, MxKay RML, James TY (2023) A combined microscopy and single-cell sequencing approach reveals the ecology, morphology, and phylogeny of uncultured lineages of zoosporic fungi. Mbio 14:1–25
Shao D, Smith DL, Kabbage M, Roth MG (2021) Effectors of plant necrotic fungi. Front Plant Sci 12:687713. https://doi.org/10.3389/fpls.2021.687713
Shariff A, Kangas J, Coelho LP, Quinn S, Murphy RF (2010) Automated image analysis for high-content screening and analysis. J Biomol Screen 15:726–734
Sharma B, Tiwari S, Kumawat KC, Cardinale M (2023) Nano-biofertilizers as bio-emerging strategies for sustainable agriculture development: potentiality and their limitations. Sci Total Environ 860:160476. https://doi.org/10.1016/j.scitotenv.2022.160476
Sharma P (2011) Cosmeceuticals:regulatory scenario in US, Europe & India Int J Pharm Technol 3:1512–1535
Shearer CA, Raja HA, Miller AN, Nelson P, Tanaka K, Hirayama K, Marvanova L, Hyde KD, Zhang Y (2010) The molecular phylogeny of freshwater dothideomycetes. Stud Mycol 64:145–153
Shi JC, Wang XL, Wang E (2023) Mycorrhizal symbiosis in plant growth and stress adaptation: from genes to ecosystems. Annu Rev Plant Biol 74:569–607
Shivhare SC, Malviya KG, Malviya KK, Jain V (2013) A review: Natural skin lighting and nourishing agents. Res J Top Cosmet Sci 4:21–25
Shoeibi S, Mozdziak P, Golkar-Narenji A (2017) Biogenesis of selenium nanoparticles using green chemistry. Top Curr Chem 375:88
Shraddha Shekher R, Sehgal S, Kamthania M, Kumar A (2011) Laccase: microbial sources, production, purification, and potential biotechnological applications. Enzym Res 2011, 217861. https://doi.org/10.4061/2011/217861
Siddiqi KS, Husen A (2016) Fabrication of metal nanoparticles from fungi and metal salts: scope and application. Nanoscale Res Lett 11:1–15
Sikdar P, Okubara P, Mazzola M, Xiao CL (2014) Development of PCR assays for diagnosis and detection of the pathogens Phacidiopycnis washingtonensis and Sphaeropsis pyriputrescens in apple fruit. Plant Disease 98:241–246
Singh DV, Agarwal R, Shrestha JK, Thapa BR, Dubin HJ (1989) First report of Neovossia indica on wheat in Nepal. Plant Dis 73:277
Singh P, Kim YJ, Zhang D, Yang DC (2016) Biological synthesis of nanoparticles from plants and microorganisms. Trends Biotechnol 34:588–599. https://doi.org/10.1016/j.tibtech.2016.02.006
Singh A, Masih A, Khurana A, Singh PK, Gupta M, Hagen F, Meis JM, Chowdhary A (2018) High terbinafine resistance in Trichophyton interdigitale isolates in Delhi, India harbouring mutations in the squalene epoxidase gene. Mycoses 61:477–484
Singh RK, Tripathi R, Ranjan A, Srivastava AK (2020) Fungi as potential candidates for bioremediation. In: Sigh P, Kumar A, Borthakur A (eds) Abatement of environmental pollutants: trends and strategies. Elsevier, New York
Singh D, Kaur H, Kajal NC (2021) Covid-19 and rhino-orbital mucormycosis—a case report. Int J Pul & Res Sci 5:1–4
Skellam E (2019) Strategies for engineering natural product biosynthesis in fungi. Trends Biotechnol 37:416-427. https://doi.org/10.1016/j.tibtech.2018.09.003
Skinnider MA, Johnston CW, Edgar RE, Dejong CA, Merwin NJ, Rees PN, Magarvey NA (2016) Genomic charting of ribosomally synthesized natural product chemical space facilitates targeted mining. PNAS 113:6343–6351. https://doi.org/10.1073/pnas.1609014113
Skinnider MA, Johnston CW, Gunabalasingam M, Merwin NJ, Kieliszek AM, MacLellan RJ, Li H, Ranieri MRM, Webster ALH, Cao MPT, Pfeifle A, Spencer N, To QH, Wallace DP, Dejong CA, Magarvey NA (2020) Comprehensive prediction of secondary metabolite structure and biological activity from microbial genome sequences. Nat Commun 11:6058. https://doi.org/10.1038/s41467-020-19986-1
Smijs TG, Pavel S (2011) Titanium dioxide and zinc oxide nanoparticles in sunscreens: focus on their safety and effectiveness. Nanotechnol Sci Appl 4:95–112. https://doi.org/10.2147/NSA.S19419
Smit E, Souza T, Jennen D, Kleinjans J, Beucken T (2017) Identification of essential transcription factors for adequate DNA damage response after benzo(a)pyrene and aflatoxin B1 exposure by combining transcriptomics with functional genomics. Toxicology 390:74–82. https://doi.org/10.1016/j.tox.2017.09.002 j.tox.2017.09.002
Smith BE, Johnston MK, Lücking R (2016) From GenBank to GBIF: phylogeny-based predictive niche modeling tests accuracy of taxonomic identifications in large occurrence data repositories. PLoS ONE 11(3):e0151232
Smith SE, Read DJ, Harley JL (1997) Mycorrhizal symbiosis. Academic Press, San Diego
Soares M, Rousk J (2019) Microbial growth and carbon use efficiency in soil: links to fungal-bacterial dominance, SOC-quality and stoichiometry. Soil Biol Biochem 131:195-205. https://doi.org/10.1016/j.soilbio.2019.01.010
Song Y, Du M, Menezes da Silva N, Yang E, Vicente VA, de Hoog GS, Li RY (2020) Comparative analysis of clinical and environmental strains of Exophiala spinifera by long-reads sequencing and RNAseq reveal adaptive strategies. Front Microbiol 11:1880. https://doi.org/10.3389/fmicb.2020.01880
Song Y, Hu S, de Hoog GS, Liu X, Meng X, Xue R, van Diepeningen AD, Fokkens L, Li RY, Gao S (2023) Medical Fusarium: novel species, or uncertain identification? Mycosphere 14(1):2263–2283. https://doi.org/10.5943/mycosphere/14/1/27
Sota T, Kagata H, Ando Y (2014) Metagenomic approach yields insights into fungal diversity and functioning. In: Species diversity and community structure. Springer, Tokyo pp 1–23
Sousa F, Ferreira D, Reis S, Costa P (2020) Current insights on antifungal therapy: novel nanotechnology approaches for drug delivery systems and new drugs from natural sources. Pharmaceuticals 13:248
Souza LM, Ogaki MB, Câmara PE, Pinto OH, Convey P, Carvalho-Silva M, Rosa LH (2021) Assessment of fungal diversity present in lakes of Maritime Antarctica using DNA metabarcoding: a temporal microcosm experiment. Extremophiles 25:77–84
Spatafora JW, Chang Y, Benny GL, Lazarus K, Smith ME, Berbee ML, Bonito G, Corradi N, Grigoriev I, Gryganskyi A, James TY, O’Donnell K, Roberson RW, Taylor TN, Uehling J, Vilgalys R, White MM, Stajich JE (2016) A phylum-level phylogenetic classification of zygomycete fungi based on genome-scale data. Mycologia 108:1028–1046. https://doi.org/10.3852/16-042
Sperschneider J, Dodds PN (2022) EffectorP 3.0:prediction of apoplastic and cytoplasmic effectors in fungi and Oomycetes. Mol Plant Microbe Interact 35:146–156. https://doi.org/10.1094/mpmi-08-21-0201-r
Sriramulu M, Sumathi S (2018) Biosynthesis of palladium nanoparticles using Saccharomyces cerevisiae extract and its photocatalytic degradation behaviour. Adv Nat Sci Nanosci Nanotechnol 9:025018
Stachowiak B, Reguła J (2012) Health-promoting potential of edible macromycetes under special consideration of polysaccharides: a review. Eur Food Res Technol 234:369-380
Stajich JE, Harris T, Brunk BP, Brestelli J (2012) FungiDB:an integrated functional genomics database for fungi. Nucleic Acids Res 40:675–681
Stamets P (2005) Mycelium running: how mushrooms can help save the world. Ten Speed Press, Berkley
Stavrou AA, Mixão V, Boekhout T, Gabaldón T (2018) Misidentification of genome assemblies in public databases: the case of Naumovozyma dairenensis and proposal of a protocol to correct misidentifications. Yeast 35:425–429
Stein HP, Navajas-Pérez R, Aranda E (2018) Potential for CRISPR genetic engineering to increase xenobiotic degradation capacities in model fungi. Approach Bioremed 61–78
Strong PJ, Self R, Allikian K, Szewczyk E, Speight R, O’Hara I, Harrison MD (2022) Filamentous fungi for future functional food and feed. Curr Opin Biotechnol 76: 102729
Sturmer SL, Bever JD, Schultz PA, Bentivenga SP (2021) Celebrating INVAM: 35 years of the largest living culture collection of arbuscular mycorrhizal fungi. Mycorrhiza 31:117-126
Sun G, Liu J, Li G, Zhang X, Chen T, Chen J, Zhang H, Wang D, Sun F, Pan H (2015) Quick and accurate detection and quantification of Magnaporthe oryzae in rice using real-time quantitative polymerase chain reaction. Plant Disease 99:219–224
Sun X, Guo LD (2012) Endophytic fungal diversity: review of traditional and molecular techniques. Mycology, 3:65–76
Sun XP, Chen WB, Ivanov S, MacLean AM, Wight H, Ramaraj T (2019) Genome and evolution of the arbuscular mycorrhizal fungus Diversispora epigaea (formerly Glomus versiforme) and its bacterial endosymbionts. New Phytol 221:1556-1573
Suresh P, Basu, P (2008) Improving pharmaceutical product development and manufacturing: impact on cost of drug development and cost of goods sold of pharmaceuticals. J Pharm Innov 3:175-187. https://doi.org/10.1007/s12247-008-9043-1
Svenningsen NB, Watts-Williams SJ, Joner EJ, Battini F, Efthymiou A, Cruz-Paredes C (2018) Suppression of the activity of arbuscular mycorrhizal fungi by the soil microbiota. ISME J 12:1296-1307
Swift CL, Podolsky IA, Lankiewicz TS, Seppälä S, O'Malley MA (2019) Linking ‘omics’ to function unlocks the biotech potential of non-model fungi. Curr Opin Syst Biol 14:9-17
Sydor M, Cofta G, Doczekalska B, Bonenberg A (2022) Fungi in mycelium-based composites: usage and recommendations. Materials 15:6283
Taberlet P, Coissac E, Pompanon F (2012) Towards next‐generation biodiversity assessment using DNA metabarcoding. Mol Ecol 21:2045–2050
Talaviya T, Shah D, Patel N, Yagnik H, Shah M (2020) Implementation of artificial intelligence in agriculture for optimisation of irrigation and application of pesticides and herbicides. Artif Intell Agric 4:58–73
Talhinhas P, Baroncelli R (2021) Colletotrichum species and complexes: geographic distribution, host range and conservation status. Fungal Divers 110:109–198
Tan YP, Bishop-Hurley SL, Shivas RG, Cowan DA (2022) Fungal planet description sheets. Persoonia 1436–1477
Tang X, Lu YZ, Dissanayake LS, Goonasekara ID, Jayawardena RS (2023) Two new fungal genera (Diaporthales) found on Dipterocarpaceae in Thailand. Front Microbiol 14: 1169052
Tao G, Liu ZY, Hyde KD, Liu XZ, Yu ZN (2008) Whole rDNA analysis reveals novel and endophytic fungi in Bletilla ochracea (Orchidaceae). Fungal Divers 33:101–122
Taofiq O, González-Paramás AM, Martins A, Barreiro MF, Ferreira ICFR (2016a) Mushrooms extracts and compounds in cosmetics, cosmeceuticals and nutricosmetics—a review. Ind Crops Prod 90:38-48
Taofiq O, Heleno SA, Calhelha RC, Alves MJ, Barros L, Barreiro MF, González-Paramás AM, Ferreira IC (2016b) Development of mushroom-based cosmeceutical formulations with anti-inflammatory, anti-tyrosinase, antioxidant, and antibacterial properties. Molecules 21(10):1372
Taofiq O, Rodrigues F, Barros L, Barreiro MF, Ferreira IC, Oliveira MBP (2019) Mushroom ethanolic extracts as cosmeceuticals ingredients: Safety and ex vivo skin permeation studies. Food Chem Toxicol 127:228-236
Taylor JE, Hyde KD, Jines EBG (1999) Endophytic fungi associated with the temperate palm, Trachycarpus fortunei, within and outside its natural geographic range. New Phytol 142:335–346
Tedersoo L, Bahram M, Puusepp R (2017) Novel soil-inhabiting clades fill gaps in the fungal tree of life. Microbiome 5:42
Tedersoo L, Anslan S, Bahram M (2020a) Identifying the ‘unidentified’fungi: a global-scale long-read third-generation sequencing approach. Fungal Divers 103:273–293
Tedersoo L, Anslan S, Bahram M (2020b) Regional-scale in-depth analysis of soil fungal diversity reveals strong pH and plant species effects in Northern Europe. Front Microbiol 11:1953
Tedersoo L, Mikryukov V, Anslan S (2021a) The Global Soil Mycobiome consortium dataset for boosting fungal diversity research. Fungal Divers 111:573–588
Tedersoo L, Albertsen M, Anslan S, Callahan B (2021b) Perspectives and benefits of high-throughput long-read sequencing in microbial ecology. App Environ Microbiol 87:00626-21
Tedersoo L, Mikryukov V, Zizka A (2022) Global patterns in endemicity and vulnerability of soil fungi. Glob Chang Biol. 28:6696–710
Telenius A (2011) Biodiversity information goes public: GBIF at your service. Nordic J Bot 29:378−381
Temporiti MEE, Nicola L, Nielsen E, Tosi S (2022) Fungal enzymes involved in plastics biodegradation. Microorganisms 10:1180
Terabayashi Y, Sano M, Yamane N, Marui J, Tamano K, Sagara J, Dohmoto M, Oda K, Ohshima E, Tachibana K, Higa Y, Ohashi S, Koike H, Machida M (2010) Identification and characterization of genes responsible for biosynthesis of Kojic acid, an industrially important compound from Aspergillus oryzae. Fungal Genet Biol 47:953–961
Terrer C, Vicca S, Hungate BA, Phillips RP, Prentice IC (2016) Mycorrhizal association as a primary control of the CO2 fertilization effect. Science 353:72–74
Thaisuchat H, Karuehanon W, Boonkorn P, Meesumlee J, Malai S, Ruttanateerawichien K (2022) Bamboo waste recycling using Dictyophora indusiata mycelia cultivation. Int J Recycl Org Waste Agric 12:539–548
Thambugala KM, Wanasinghe DN, Phillips AJL, Camporesi E, Bulgakov TS, Phukhamsakda C, Ariyawansa HA, Goonasekara ID, Phookamsak R, Dissanayake A, Tennakoon DS, Tibpromma S, Chen YY, Liu ZY, Hyde KD (2018) Mycosphere notes 1–50: grass (Poaceae) inhabiting Dothideomycetes. Mycosphere 8:697–796
Thomas P, Büntgen U (2019) A risk assessment of Europe's black truffle sector under predicted climate change. Sci Total Environ 655:27−34
Thongbai B, Rapior S, Hyde KD, Wittstein K, Stadler M (2015) Hericium erinaceus, an amazing medicinal mushroom. Mycol Prog 14(10):91. https://doi.org/10.1007/s11557-015-1105-4
Tian DS, Kuhnert E, Ouazzani J, Wibberg D, Kalinowski J, Cox RJ (2020) The sporothriolides. A new biosynthetic family of fungal secondary metabolites. Chem Sci 11:12477-12484
Tibpromma S, Hyde KD, Bhat JD, Mortimer PE (2018) Identification of endophytic fungi from leaves of Pandanaceae based on their morphotypes and DNA sequence data from southern Thailand. MycoKeys 33:25
Tibpromma S, Dong Y, Ranjitkar S, Schaefer DA (2021) Climate-fungal pathogen modeling predicts loss of up to one-third of tea growing areas. Front Cell Infect Microbiol 11:610567
Tisserant E, Malbreil M, Kuo A, Kohler A, Symeonidi A, Balestrin, R, Charron P, Duensing N, Frei dit Frey N, Gianinazzi-Pearson V (2013) Genome of an arbuscular mycorrhizal fungus provides insight into the oldest plant symbiosis. Proc Natl Acad Sci 110(50):20117–20122
Tiwari P, Dufossé L (2023) Focus and insights into the synthetic biology-mediated chassis of economically important fungi for the production of high-value metabolites. Microorganisms 11:1141
Tjamos EC, Tsitsigiannis DI, Tjamos SE, Antoniou PP, Katinakis P (2004) Selection and screening of endorhizosphere bacteria from solarized soils as biocontrol agents against Verticillium dahliae of Solanaceous hosts. Eur J Plant Pathol 110:35–44
Toffolatti SL, De Lorenzis G, Brilli M, Moser M, Shariati V, Tavakol E, Maddalena G, Passera A, Casati P, Pindo M, Cestaro A (2020) Novel aspects on the interaction between grapevine and Plasmopara viticola: dual-rna-seq analysis highlights gene expression dynamics in the pathogen and the plant during the battle for infection. Genes (Basel) 11:261
Toffolatti SL, De Lorenzis G, Costa A, Maddalena G, Passera A, Bonza MC, Pindo M, Stefani E, Cestaro A, Casati P, Failla O (2018a) Unique resistance traits against downy mildew from the center of origin of grapevine (Vitis vinifera). Sci Rep 8: 12523
Toffolatti SL, Russo G, Campia P, Bianco PA, Borsa P, Coatti M, Torriani SF, Sierotzki H (2018b) A time-course investigation of resistance to the carboxylic acid amide mandipropamid in field populations of Plasmopara viticola treated with anti-resistance strategies. Pest Manag Sci 74:2822–2834
Tomlinson JA, Dickinson M, Hobden E, Robinson S, Giltrap PM, Boonham N (2010) A five-minute DNA extraction method for expedited detection of Phytophthora ramorum following pre-screening using Phytophthora spp. lateral flow devices. J Microbiol Methods 81:116–120
Torres-Cruz TJ, Whitaker BK, Proctor RH, Broders K (2022) FUSARIUM-ID v. 3.0:an updated, downloadable resource for Fusarium species identification. Plant Disease 106:1610–1616
Torres-Martínez BDM, Vargas-Sánchez RD, Torrescano-Urrutia GR, Esqueda M, Rodríguez-Carpena JG, Fernández-López J, Perez-Alvarez JA, Sánchez-Escalante A (2022) Pleurotus genus as a potential ingredient for meat products. Foods 11:779. https://doi.org/10.3390/foods11060779
Tripathi KM, Bhati A, Singh A, Sonker AK, Sarkar S, Sonkar SK (2017) Sustainable changes in the contents of metallic micronutrients in first generation gram seeds imposed by carbon nano-onions: life cycle seed to seed study. ACS Sustain Chem Eng 5:2906–2916
Troia MJ, McManamay RA (2016) Filling in the GAPS: evaluating completeness and coverage of open-access biodiversity databases in the United States. Ecol Evol 6:4654–4669
Trontin C, Bertaccini A, van de Bilt J, Bukovinszki T, Contaldo N, Cottyn B, De Vos P (2022) EPPO-Q-Bank-a tool for identification of plant quarantine pests. In: International plant health conference
Tsivileva O, Pozdnyakov A, Ivanova A (2021) Polymer Nanocomposites of Selenium Biofabricated Using Fungi. Molecules 26:3657. https://doi.org/10.3390/molecules26123657
Tsukada K, Shinki S, Kaneko A, Murakami K, Irie K, Murai M, Miyoshi H, Dan S, Kawaji K, Hayashi H, Kodama EN, Hori A, Salim E, Kuraishi T, Hirata N, Kanda Y, Asai T (2020) Synthetic biology based construction of biological activity-related library of fungal decalin-containing diterpenoid pyrones. Nat Commun 11:1830. https://doi.org/10.1038/s41467-020-15664-4
Tuininga AR, Miller JL, Morath SU, Daniels TJ, Falco RC, Marchese M, Sahabi S, Rosa D, Stafford KC (2009) Isolation of entomopathogenic fungi from soils and ixodes scapularis (acari:ixodidae) ticks:prevalence and methods. J Med Entomol 46:557–565
Uchida R, Ishikawa S, Tomoda H (2014) Inhibition of tyrosinase activity and melanine pigmentation by 2-hydroxytyrosol. Acta Pharm Sin B 4:141–145
Uday USP, Bandyopadhyay TK, Bhunia B (2016) Rapid development of xylanase assay conditions using Taguchi methodology. Bioengineered 7:424–431
Udayanga D, Castlebury LA, Rossman AY, Chukeatirote E (2014) Insights into the genus Diaporthe: phylogenetic species delimitation in the D. eres species complex. Fungal Divers 67:203–229
Ullah S, Rahman W, Ullah F, Jehan R (2022) FDBC: a comprehensive platform of the fungi databases collection. J Biol 11:1–6
US National Fungus Collections (2023). https://www.ars.usda.gov. Accessed 12 Sept 2023
Uzuhashi S, Kakishima M, Tojo M (2010) Phylogeny of the genus Pythium and description of new genera. Mycoscience 51:337–365. https://doi.org/10.1007/S10267-010-0046-7
Vahter T, Bueno CG, Davison J, Herodes K, Hiiesalu I, Kasari-Toussaint L (2020) Co-introduction of native mycorrhizal fungi and plant seeds accelerates restoration of post-mining landscapes. J Appl Ecol 57:1741–1751
Vaksmaa A, Guerrero-Cruz S, Ghosh P, Zeghal E, Hernando-Morales V, Niemann H (2023) Role of fungi in bioremediation of emerging pollutants. Front Mar Sci 10:1070905
Valverde ME, Hernández-Pérez T, Paredes-López O (2015) Edible mushrooms: improving human health and promoting quality life. Int J Microbiol 2015:1–14. https://doi.org/10.1155/2015/376387
Vandelook S, Elsacker E, Van Wylick A, De Laet L, Peeters E (2021) Current state and future prospects of pure mycelium materials. Fungal Biol Biotechnol 8:1–10
Varma VS, Ramu K, Kalamdhad AS (2015) Carbon decomposition by inoculating Phanerochaete chrysosporium during drum composting of agricultural waste. Environ Sci Pollut Res 22:7851–7858
Varshney RK, Ribaut JM, Buckler ES, Tuberosa R, Rafalski JA, Langridge P (2019) Can genomics boost productivity of orphan crops? Nat Biotechnol 37:713–718
Velazhahan V, McCann BL, Bignell E, Tate CG (2023) Developing novel antifungals: lessons from G protein-coupled receptors. Trends Pharmacol Sci 44:162–174
Velásquez AC, Castroverde CDM, He SY. (2018) Plant-pathogen warfare under changing climate conditions. Curr Biol 8:R619-R634
Venbrux M, Crauwels S, Rediers H (2023) Current and emerging trends in techniques for plant pathogen detection. Front Plant Sci 14:1120968
Venturella G, Ferraro V, Cirlincione F, Gargano ML (2021) Medicinal mushrooms: Bioactive compounds, use, and clinical trials. Int J Mol Sci 22(2):634. https://doi.org/10.3390/ijms22020634
Vetrovsky T, Kohout P, Kopecky M, Machac A, Man M, Bahnmann BD (2019) A meta-analysis of global fungal distribution reveals climate-driven patterns. Nat Commun 10:5142. https://doi.org/10.1038/s41467-019-13164-8
Větrovský T, Morais D, Kohout P, Lepinay C et al (2020) GlobalFungi, a global database of fungal occurrences from high-throughput-sequencing metabarcoding studies. Sci Data 7:228
Větrovský T, Kolaříková Z, Lepinay C, Awokunle Hollá S, Davison J, Gromyko A, Jelínková B, Kolařík M, Krüger M, Lejsková R, Michalčíková M, Michalová T, Moora M, Moravcová A, Moulíkov Š, Odriozola I, Öpik M, Pappová M, Piché-Choquette S, Popelářová A, Skřivánek J, Vlk L, Zobel M, Baldrian P, Kohout P (2023) GlobalAMFungi: a global database of arbuscular mycorrhizal fungal occurrences from high-throughput-sequencing metabarcoding studies. New Phytol. https://doi.org/10.1111/nph.19283
Vetter J, Siller I (1991) Chitin content of higher fungi. Zeitschrift für Lebensmittel-Untersuchung und Forschung 193:36-38
Vetter J (2007) Chitin content of cultivated mushrooms Agaricus bisporus, Pleurotus ostreatus and Lentinula edodes. Food Chem 102:6–9
Vetter J (2023) The mushroom glucans: molecules of high biological and medicinal importance. Foods 12:1009
Vinh DC (2023) Of mycelium and men: inherent human susceptibility to fungal diseases. Pathogens 12:456
Virendra SG, Somnath T (2009) Phylogenomics: evolution and genomics intersection. Int J Bioinform Res Appl 5:548–563. https://doi.org/10.1504/ijbra.2009.028682
Viswanath B, Chandra MS, Kumar KP, Rajasekhar Reddy B (2008) Production and purification of laccase from Stereumostrea and its ability to decolorize textile dyes. Dyn Biochem Process Biotechnol Mol Biol 2:19–25
Viswanath B, Rajesh B, Janardhan A, Kumar AP, Narasimha G (2014) Fungal laccases and their applications in bioremediation. Enzym Res 2014:163242. https://doi.org/10.1155/2014/163242
Voběrková S, Vaverková MD, Burešová A, Adamcová D, Vršanská M, Kynický J, Adam V (2017) Effect of inoculation with white-rot fungi and fungal consortium on the composting efficiency of municipal solid waste. Waste Manag 61: 157–16
Voigt K, James TY, Kirk PM, Santiago ALMDA, Waldman B, Griffith GW,Fu M, Radek R, Strassert JFH, Wurzbacher C, Jerônimo GH, Simmons DR, Seto K, Gentekaki E, Hurdeal VG, Hyde KD, Nguyen TTT, Lee HB (2021) Early-diverging fungal phyla: taxonomy, species concept, ecology, distribution, anthropogenic impact, and novel phylogenetic proposals. Fungal Divers 109:59–98
Wainaina S, Taherzadeh MJ (2022) Automation and artificial intelligence in filamentous fungi-based bioprocesses:A review. Biores Tech 128421
Walsh G, Walsh E (2022) Biopharmaceutical benchmarks 2022. Nat Biotechnol 40:1722–1760. https://doi.org/10.1038/s41587-022-01582-x
Wan Rosli WI, Solihah MA (2012) Effect on the addition of pleurotus Sajor-Caju (PSC) on physical and sensorial properties of beef patty. Int Food Res J 19:993–999
Wan Rosli WI, Solihah MA, Mohsin SSJ (2011) On the ability of oyster mushroom (Pleurotus sajor-caju) confering changes in proximate composition and sensory evaluation of chicken patty. Int Food Res J 18:1453–1469
Wanasinghe DN, Phukhamsakda C, Hyde KD, Jeewon R (2018) Fungal diversity notes 709–839: taxonomic and phylogenetic contributions to fungal taxa with an emphasis on fungi on Rosaceae. Fungal Divers 89:1–236
Wang Y, Zhu M, Zhang R, Yang H, Wang Y, Sun G, Jin S, Hsiang T (2009) Whole genome amplification of the rust Puccinia striiformis f. sp. tritici from single spores. J Microbiol Methods 77:229–234
Wang X, Zhou P, Cheng J, Chen Z, Liu X (2018) Use of straw mushrooms (Volvariella volvacea) for the enhancement of physicochemical, nutritional and sensory profiles of Cantonese sausages. Meat Sci 146:18-25. https://doi.org/10.1016/j.meatsci.2018.07.033
Wang L, Guo H, Liu X, Jiang G, Li C, Li X, Li Y (2019a) Roles of Lentinula edodes as the pork lean meat replacer in production of the sausage. Meat Science 156:44–51. https://doi.org/10.1016/j.meatsci.2019.05.016
Wang L, Li C, Ren L, Guo H, Li Y (2019b) Production of pork sausages using Pleurotus eryngii with different treatments as replacements for pork back fat. J Food Sci 84:3091–3098. https://doi.org/10.1111/1750-3841.14839
Wang Y, Wang C, Wang L, Zhang X, Yan J, Wang J, Wang M (2020) Development of loop-mediated isothermal amplification (LAMP) assay for rapid detection of Fusarium proliferatum causing ear and kernel rot on maize. Crop Protection 132:105142
Wang D, Xue B, Wang L, Zhang Y, Liu L, Zhou Y (2021) Fungus-mediated green synthesis of nano-silver using Aspergillus sydowii and its antifungal/antiproliferative activities. Sci Rep 11:1–9
Wang D, Jin S, Lu Q, Chen Y (2023a) Advances and challenges in CRISPR/Cas-based fungal genome engineering for secondary metabolite production: a review. J Fungi 9:362
Wang F, Wang K, Cai L, Zhao M, Kirk PM, Fan G, Sun Q, Li B, Wang S, Yu Z, Han D, Ma J, Wu L, Yao Y (2023b) Fungal names: a comprehensive nomenclatural repository and knowledge base for fungal taxonomy. Nucleic Acids Res 51:708–716
Wang Y, Zhang R, Pu Y, Wang D, Wang Y, Wu K (2023c) Sample collection, DNA extraction, and library construction protocols of the human microbiome studies in the international human phenome project. Phenomics 8:1–9
Wasser SP (2011) Current findings, future trends, and unsolved problems in studies of medicinal mushrooms. Applied Microbiology and Biotechnology 89:1323–1332
Wasser SP (2014) Medicinal mushroom science: current perspectives, advances, evidences, and challenges. Biomed J 37:345-356
Weisberg AJ, Grünwald NJ, Savory EA, Putnam ML (2021) Genomic approaches to plant-pathogen epidemiology and diagnostics. Annu Rev Phytopathol 59:311–332
Wijayawardene NN, Hyde KD, Al-Ani LKT, Tedersoo L, Haelewaters D, Rajeshkumar KC, Zhao RL, Aptroot A, Leontyev DV, Saxena RK, Tokarev YS, Dai DQ, Letcher PM, Stephenson SL, Ertz D, Lumbsch HT, Kukwa M, Issi IV, Madrid H, Phillips AJL, Selbmann L, Pfliegler WP, Horváth E, Bensch K, Kirk PM, Kolaříková K, Raja HA, Radek R, Papp V, Dima B, Ma J, Malosso E, Takamatsu S, Rambold G, Gannibal PB, Triebel D, Gautam AK, Avasthi S, Suetrong S, Timdal E, Fryar SC, Delgado G, Réblová M, Doilom M, Dolatabadi S, Pawłowska J, Humber RA, Kodsueb R, Sánchez-Castro I, Goto BT, Silva DKA, de Souza FA, Oehl F, da Silva GA, Silva IR, Błaszkowski J, Jobim K, Maia LC, Barbosa FR, Fiuza PO, Divakar PK, Shenoy BD, Castañeda-Ruiz RF, Somrithipol S, Lateef AA, Karunarathna SC, Tibpromma S, Mortimer PE, Wanasinghe DN, Phookamsak R, Xu J, Wang Y, Tian F, Alvarado P, Li DW, Kušan I, Matočec N, Maharachchikumbura SSN, Papizadeh M, Heredia G, Wartchow F, Bakhshi M, Boehm E, Youssef N, Hustad VP, Lawrey JD, Santiago ALCMA, Bezerra JDP, Souza-Motta CM, Firmino AL, Tian Q, Houbraken J, Hongsanan S, Tanaka K, Dissanayake AJ, Monteiro JS, Grossart HP, Suija A, Weerakoon G, Etayo J, Tsurykau A, Vázquez V, Mungai P, Damm U, Li QR, Zhang H, Boonmee S, Lu YZ, Becerra AG, Kendrick B, Brearley FQ, Motiejūnaitė J, Sharma B, Khare R, Gaikwad S, Wijesundara DSA, Tang LZ, He MQ, Flakus A, Rodriguez-Flakus P, Zhurbenko MP, McKenzie EHC, Stadler M, Bhat DJ, Liu JK, Raza M, Jeewon R, Nassonova ES, Prieto M, Jayalal RGU, Erdoğdu M, Yurkov A, Schnittler M, Shchepin ON, Novozhilov YK, Silva-Filho AGS, Liu P, Cavender JC, Kang Y, Mohammad S, Zhang LF, Xu RF, Li YM, Dayarathne MC, Ekanayaka AH, Wen TC, Deng CY, Pereira OL, Navathe S, Hawksworth DL, Fan XL, Dissanayake LS, Kuhnert E, Grossart HP, Thines M (2020) Outline of Fungi and fungus-like taxa. Mycosphere 11:1060-1456
Wijayawardene NN, Hyde K, Dai D, Sánchez-García M, Goto BT, Saxena RK, Erdoğdu M, Selçuk F, Rajeshkumar KC, Aptroot A, Błaszkowski J, Boonyuen N, da Silva GA, de Souza FA, Dong W, Ertz D, Haelewaters D, Jones EBG, Karunarathna SC, Kirk PM, Kukwa M, Kumla J, Leontyev DV, Lumbsch HT, Maharachchikumbura SSN, Marguno F, Martínez-Rodríguez P, Mešić A, Monteiro JS, Oehl F, Pawłowska J, Pem D, Pfliegler WP, Phillips AJL, Pošta A, He MQ, 55 Li JX, Raza M, Sruthi OP, Suetrong S, Suwannarach N, Tedersoo L, Thiyagaraja V, Tibpromma S, Tkalčec Z, Tokarev YS, Wanasinghe DN, Wijesundara DSA, Wimalaseana SDMK, Madrid H, Zhang GQ, Gao Y, Sánchez-Castro I, Tang LZ, Stadler M, Yurkov A, Thines M (2022a) Outline of Fungi and fungus-like taxa—2021. Mycosphere 13:53–453. https://doi.org/10.5943/mycosphere/13/1/2
Wijayawardene NN, Phillips AJ, Pereira DS, Dai DQ, Aptroot A, Monteiro JS, Druzhinina IS, Cai F, Fan X, Selbmann L, Coleine C, Castañeda-Ruiz RF, Kukwa M, Flakus A, Fiuza PO, Kirk PM, Kumar KCR, Arachchi ISI, Suwannarach N, Tang LZ, Boekhout T, Tan CS, Jayasinghe RPPK, Thines M (2022b) Forecasting the number of species of asexually reproducing fungi (Ascomycota and Basidiomycota). Fungal Divers 114:463–490
Wijayawardene NN, Boonyuen N, Ranaweera CB, de Zoysa HK (2023a) OMICS and other advanced technologies in mycological applications. J Fungi 9:688
Wijayawardene NN, Dai DQ, Premarathne BM, Wimalasena MK (2023b) Checklist, typification details, and nomenclature status of ascomycetous fungi originally described in Sri Lanka. Phytotaxa 611:1–105
Wijesinghe SN, Samarakoon MC, Camporesi E, Hyde KD, Jones EBG, Zucconi L, Perera RH, Boonmee S, de Silva NI, Rathnayaka AR, Pem D, Wanasinghe DN, Maharachchikumbura SSN, Wang Y (2023) Over the footprints of Italian mycology with emphasis on plant-associated Ascomycota. Curr Res Environ Appl 13:162–276
Wikandari R, Hasniah N, Taherzadeh MJ (2022) The role of filamentous fungi in advancing the development of a sustainable circular bioeconomy. Bioresource Technology 345:126531
Williamson PR, Wakamatsu K (1998) Ito S. Melanin biosynthesis in Cryptococcus
Withers S, Gongora-Castillo E, Gent D, Thomas A, Ojiambo PS, Quesada-Ocampo LM (2016) Using next-generation sequencing to develop molecular diagnostics for Pseudoperonospora cubensis, the cucurbit downy mildew pathogen. Phytopathology 106:1105–1116
Wong KM, Corradini MG, Autio W, Kinchla AJ (2019) Sodium reduction strategies through use of meat extenders (white button mushrooms vs. textured soy) in beef patties. Food Sci Nutr 7:506–518. https://doi.org/10.1002/fsn3.824
Wong KM, Decker EA, Autio WR, Toong K, DiStefano G, Kinchla AJ (2017) Utilizing mushrooms to reduce overall sodium in taco filling using physical and sensory evaluation. J Food Sci 82(10). https://doi.org/10.1111/1750-3841.13838
World Health Organization (WHO) (2023) Mycotoxins. https://www.who.int/news-room/fact-sheets/detail/mycotoxins. Accessed 5 Jul 2023
Wösten HA (2019) Filamentous fungi for the production of enzymes, chemicals and materials. Curr Opin Biotech 59:65–70
Wouters OJ, McKnee M, Luyten J (2020) Estimated research and development investment needed to bring a new medicine to market, 2009–2018 JAMA 323:844–853. https://doi.org/10.1001/jama.2020.1166
Wu B, Hussain M, Zhang W, Stadler M, Liu X, Xing M (2019) Current insights into fungal species diversity and perspective on naming the environmental DNA sequences of fungi Mycology 10:127–140
Wu L, Zhao J, Zhang X, Liu S, Zhao C (2021) Antitumor effect of soluble β-glucan as an immune stimulant. Int J Biol Macromol 179:116-124
Wu N, Dissanayake AJ, Manawasinghe IS, Rathnayaka AR, Liu JK, Phillip AJ, Promputtha I, Hyde KD (2021). https://botryosphaeriales.org/, an up-to-date classification and account of taxa of Botryosphaeriales. Database: 1–9. https://doi.org/10.1093/database/baab061
Wu Y, Choi MH, Li J, Yang H, Shin HJ (2016) Mushroom cosmetics: the present and future. Cosmetics 3:1–13
Wydro U (2022) Soil microbiome study based on DNA extraction: a review. Water 14(24):3999
Xia C, Qiu A, Wang M, Liu T, Chen W, Chen X (2022) Current status and future perspectives of genomics research in the rust fungi. Int J Mol Sci 23:9629
Xia PF, Ling H, Foo JL, Chang MW (2019) Synthetic genetic circuits for programmable biological functionalities. Biotec Adv 37(6):107393
Xie K, Ren YH, Chen AQ, Yang CF, Zheng QS, Chen J, Wang DS, Li YT, Hu SJ, Xu GH (2022) Plant nitrogen nutrition: the roles of arbuscular mycorrhizal fungi. J Plant Physiol 269:153591
Xu J (2020) Fungal species concepts in the genomics era. Genome 63:459–68
Xu XY, Qiu YP, Zhang KC, Yang F, Chen MF, Luo X (2022) Climate warming promotes deterministic assembly of arbuscular mycorrhizal fungal communities. Global Change Biology 28:1147-1161
Xu YJ, Chen Z, Li XY, Tan J, Liu F, Wu JP (2023) Mycorrhizal fungi alter root exudation to cultivate a beneficial microbiome for plant growth. Functional Ecology 37:664–675
Yan JY, Zhao WS, Chen Z, Xing QK, Zhang W, Chethana KWT, Xue MF, Xu JP, Phillips AJL, Wang Y, Liu JH, Liu M, Zhou Y, Jayawardena RS, Manawasinghe IS, Huang JB, Qiao GH, Fu CY, Guo FF, Dissanayake AJ, Peng YL, Hyde KD, Li XH (2018) Comparative genome and transcriptome analyses reveal adaptations to opportunistic infections in woody plant degrading pathogens of Botryosphaeriaceae. DNA Res 25:87–102
Yan ZF, Yang Y, Tian FH, Mao XX (2014) Inhibitory and acceleratory effects of Inonotus obliquus on tyrosinase activity and melanin formation in B16 melanoma cells. Ev Based Compl Alt Med 2014:1–11
Yang FC, Yang MJ, Cheng SH (2009) A novel method to enhance the mycelia production of Ganoderma lucidum in submerged cultures by polymer additives and agitation strategies. J Taiwan Inst Chem Eng 40:148-154
Yang J, Liu LL, Jones EG, Hyde KD (2023) Freshwater fungi from karst landscapes in China and Thailand. Fungal Divers 119:1–212
Yang K, Lee I, Nam S (2017) Development of a rapid detection method for Peronospora destructor using loop-mediated isothermal amplification (LAMP). HortScience 52:S413
Yang Y, Lu YT, Zeng K, Heinze T, Groth T, Zhang K (2021) Recent progress on cellulose‐based ionic compounds for biomaterials. Adv Mater 33:2000717
Yasanthika WAE, Wanasinghe DN, Mortimer PE, Monkai J, Farias ARG (2022) The importance of culture-based techniques in the genomic era for assessing the taxonomy and diversity of soil fungi. Mycosphere 13:724–751
Yasanthika WE, Tennakoon DS, De Farias ARG, Chethana, KT (2023) Isolation and characterization of novel Dothideomycetes species from forest soils in Chiang Rai and Krabi (Thailand): additions to the diversity of Curvularia and Verruconis. Phytotaxa 609:17–44
Yee DA, Kakule TB, Cheng W, Chen M, Chong CTY, Hai Y, Hang LF, Hung YS, Liu N, Ohashi M, Okorafor IC, Song Y, Tang M, Zhang Z, Tang Y (2020) Genome mining of alkaloidal terpenoids from a hybrid terpene and nonribosomal peptide biosynthetic pathway. J Am Chem Soc 142:710–714. https://doi.org/10.1021/jacs.9b13046
Yee DA, Niwa K, Perlatti B, Chen MB, Li YQ, Tang Y (2023) Genome mining for unknown–unknown natural products. Nat Chem Biol 19:633–640. https://doi.org/10.1038/s41589-022-01246-6
Yılmaz TM, Mungan MD, Berasategui A, Ziemert N (2023) FunARTS, the fungal bioActive compound Resistant Target Seeker, an exploration engine for target-directed genome mining in fungi, Nucl Acids Res 51:191–197. https://doi.org/10.1093/nar/gkad386
Yin Z, Liang Z, Li C, Wang J, Ma C, Kang W (2021) Immunomodulatory effects of polysaccharides from edible fungus: a review. Food Science and Human Wellness 10: 393–400
Yoon KN, Alam N, Lee JS, Lee KR (2011) Detection of phenolic compounds concentration and evaluation of antioxidant and antityrosinase activity of various extract from Lentinus edodes. World Appl Sci J 12:1851–1859
Young JM, Weyrich LS, Cooper A (2016) High‐throughput sequencing of trace quantities of soil provides reproducible and discriminative fungal DNA profiles. J Forensic Sci 61:478-484
Youssef J, Spence C (2021) Introducing diners to the range of experiences in creative Mexican cuisine, including the consumption of insects. Int J Gastron Food Sci 25:1–13
Yu S, Ali J, Zhou S, Ren G, Xie H, Xu J, Yu X, Zhou F, Peng S, Ma L, Yuan D (2022) From Green Super Rice to green agriculture: reaping the promise of functional genomics research. Mol Plant 15:9-26
Yu W, Pei R, Zhang Y, Tu Y, He B (2023) Light regulation of secondary metabolism in fungi. J Biol Eng 17:57
Yusuf A, Almotairy ARZ, Henidi H, Alshehri OY, Aldughaim MS (2023) Nanoparticles as drug delivery systems: a review of the implication of nanoparticles' physicochemical properties on responses in biological systems. Polymers (Basel) 15:1596. https://doi.org/10.3390/polym15071596
Yuwa-Amornpitak T, Butkhup L, Yeunyaw PN (2020) Amino acids and antioxidant activities of extracts from wild edible mushrooms from a community forest in the Nasrinual District, Maha Sarakham, Thailand. Food Sci Technol (Brazil) 40:18519. https://doi.org/10.1590/fst.18519
Zahan N, Hasan MZ, Malek MA, Reya SS (2021) A deep learning-based approach for edible, inedible and poisonous mushroom classification. In: 2021 international conference on information and communication technology for sustainable development (ICICT4SD), pp 440–444. https://doi.org/10.1109/ICICT4SD50815.2021.9396845
Zahan N, Hasan MZ, Uddin MS, Hossain S, Islam SF (2022) Chapter 10—a deep learning-based approach for mushroom diseases classification. In: Khan MA, Khan R, Ansari MA (eds) Application of machine learning in agriculture. Academic Press, New York, pp 191–212. https://doi.org/10.1016/B978-0-323-90550-3.00005-9
Zambonino MC, Quizhpe EM, Jaramillo FE, Rahman A, Santiago Vispo, N, Jeffryes C, Dahoumane SA (2021) Green synthesis of selenium and tellurium nanoparticles: current trends, biological properties and biomedical applications. Int J Mol Sci 22: 989
Zeilinger S, Gupta VK, Dahms TE, Silva RN, Singh HB, Upadhyay RS, Gomes EV, Tsui CK, Nayak SC (2016) Friends or foes? Emerging insights from fungal interactions with plants. FEMS Microbiol Rev 40:182–207
Zeng H, Stadler M, Abraham WR, Müsken M, Schrey H (2023) Inhibitory effects of the fungal pigment rubiginosin C on hyphal and biofilm formation in Candida albicans and Candida auris. J. Fungi 9:726. https://doi.org/10.3390/jof9070726
Zhan P, Li D, Wang C, Sun J, Geng C, Xiong Z, Seyedmousavi S, Liu W, de Hoog GS (2015) Epidemiological changes in tinea capitis over the sixty years of economic growth in China. Med Mycol J 53:691–698. https://doi.org/10.1093/mmy/myv057
Zhang F, Yang YQ, Zhou FP, Xiao W, Boonmee S, Yang XY (2023) Morphological and phylogenetic characterization of five novel nematode-trapping fungi (Orbiliomycetes) from Yunnan, China. J Fungus 9:735
Zhang L, Zhou JC, George TS, Limpens E, Feng G (2022) Arbuscular mycorrhizal fungi conducting the hyphosphere bacterial orchestra. Trend Plant Sci 27:402–411
Zhang N, Luo J, Bhattacharya D (2017) Chapter eight—advances in fungal phylogenomics and their impact on fungal systematics. In: Townsend JP, Wang Z (eds) Advances in genetics, vol 100 Academic Press, New York, pp 309–328. https://doi.org/10.1016/bs.adgen.2017.09.004
Zhang Q, Nizamani MM, Feng Y, Yang YQ, Jayawardena RS, Hyde KD, Wang Y, Li C (2023) Genome-scale and multi-gene phylogenetic analyses of Colletotrichum spp. host preference and associated with medicinal plants. Mycosphere 14:1–106
Zhang X, Wang TT, Xu QL, Xiong Y, Zhang L, Han H, Xu K, Guo WJ, Xu Q, Tan RX, Ge HM (2018) Genome mining and comparative biosynthesis of meroterpenoids from two phylogenetically distinct fungi. Angew Chem Int Ed 57:8184–8188. https://doi.org/10.1002/anie.201804317
Zhang Y, Geng W, Shen Y, Wang Y, Dai YC (2014) Edible mushroom cultivation for food security and rural development in China: bio-innovation, technological dissemination and marketing. Sustainability 6:2961–2973
Zhang Y, Mo M, Yang L, Mi F, Cao Y, Liu C, Tang X, Wang P, Xu J (2021) Exploring the species diversity of edible mushrooms in Yunnan, Southwestern China, by DNA barcoding. J Fungi 7:310. https://doi.org/10.3390/jof7040310
Zhang Y, Wang D, Chen Y, Liu T, Zhang S, Fan H, Liu H, Li Y (2021) Healthy function and high valued utilization of edible fungi. Food Sci Hum Wellness 10:408–420. https://doi.org/10.1016/j.fshw.2021.04.003
Zhang Z, Reponen T, Hershey GKK (2016) Fungal exposure and asthma: IgE and non-IgE-mediated mechanisms. Curr Allergy Asthma Rep 16:1–12
Zhao W, Chen Z, Yang X, Sheng L, Mao H, Zhu S (2023) Metagenomics reveal arbuscular mycorrhizal fungi altering functional gene expression of rhizosphere microbial community to enhance Iris tectorum's resistance to Cr stress. Sci Total Environ 895, 164970
Zhao YQ, Wu YH, Zhao XX, An MN, Chen JG (2014) Study on the TaqMan real-time PCR to the detection and quantification of Rhizoctonia solani AG-3 of tobacco target spot. Adv Mater Res 1010:80–83
Zhu YQ, Ma CY, Xue H, Piao CG, Li Y, Jiang N (2023) Two new species of Diaporthe (Diaporthaceae, Diaporthales) in China. MycoKeys 95:209–228
Zou Y, Yang C, Wang N, Zheng QW, Ye ZW, Wei T, Zhong JR, Guo LQ, Lin JF (2023) Development of Flammulina velutipes-based meat analogs with tunable physicochemical, structural, and sensory properties. Int J Food Eng 19:177–186
Zouari I, Salvioli A, Chialva M, Novero M, Miozzi L, Tenore GC, Bagnaresi P, Bonfante P (2014) From root to fruit: RNA-Seq analysis shows that arbuscular mycorrhizal symbiosis may affect tomato fruit metabolism. Bmc Genomics 15:1–19
Zubler AV, Yoon JY (2020) Proximal methods for plant stress detection using optical sensors and machine learning. Biosensors 10:1–27
Acknowledgements
This study is supported by the Major science and technology projects and key R&D plans/programs, Yunnan Province (202202AE090001), the open research project of “Cross-Cooperative Team” of the Germplasm Bank of Wild Species, Kunming Institute of Botany, Chinese Academy of Sciences (Grant No. 292019312511043), the Biodiversity Survey and Assessment Project of the Ministry of Ecology and Environment, PR China (2019HJ2096001006), the National Research Council of Thailand (NRCT) grant “Total fungal diversity in a given forest area with implications towards species numbers, chemical diversity and biotechnology” (Grant No. N42A650547). Gonkhom thanks the support from Research and researcher for industries (Grant No. PHD62I0018/2562). We also would like to thank Kunming Institute of Botany, Chinese Academy of Sciences, Flexible Talent Introduction Program—(E16441). The authors extend their appreciation to the International Scientific Partnership Program ISPP at King Saud University for funding this research work through ISPP-151. Micael F. M. Gonçalves thanks to the Portuguese Foundation for Science and Technology (FCT/MCTES) for the financial support to CESAM (UIDP/50017/2020 + UIDB/50017/2020 + LA/P/0094/2020). Petr Baldrian was supported by MEYS Czech Republic (Grant No. LUC23152).
Funding
Open access funding provided by FCT|FCCN (b-on). This study is supported by Major science and technology projects and key R&D plans/programs, Yunnan Province (202202AE090001). The open research project of “Cross-Cooperative Team” of the Germplasm Bank of Wild Species, Kunming Institute of Botany, Chinese Academy of Sciences (Grant No. 292019312511043). The Biodiversity Survey and Assessment Project of the Ministry of Ecology and Environment, PR China (2019HJ2096001006). The National Research Council of Thailand (NRCT) grant “Total fungal diversity in a given forest area with implications towards species numbers, chemical diversity and biotechnology” (Grant No. N42A650547). Didsanutda Gonkhom thanks the support from Research and researcher for industries (Grant No. PHD62I0018/2562). We thank Kunming Institute of Botany, Chinese Academy of Sciences, Flexible Talent Introduction Program—(E16441). The authors extend their appreciation to the International Scientific Partnership Program ISPP at King Saud University for funding this research work through ISPP-151.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Parts of the manuscript were written by all authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest, competing interests or relevant financial or non-financial interests to disclose.
Additional information
Handling Editor: Chitrabhanu Bhunjun.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hyde, K.D., Baldrian, P., Chen, Y. et al. Current trends, limitations and future research in the fungi?. Fungal Diversity 125, 1–71 (2024). https://doi.org/10.1007/s13225-023-00532-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13225-023-00532-5