Abstract
Introduction
LY2963016 insulin glargine (LY IGlar) and Lantus® (IGlar), both with identical primary amino acid sequences, were compared in two phase 3 studies for intrapatient blood glucose variability.
Methods
ELEMENT-1 was a 52-week study in patients with type 1 diabetes (T1D), which included Japanese patients, and ELEMENT-2 was a 24-week study in non-Japanese patients with type 2 diabetes (T2D). In ELEMENT-1, 535 patients with T1D were evaluable (268 LY IGlar and 267 IGlar). Of these, 100 were Japanese patients (49 LY IGlar and 51 IGlar). In ELEMENT-2, 756 patients with T2D were evaluable (376 LY IGlar and 380 IGlar). We evaluated and compared intrapatient blood glucose variability of LY IGlar and IGlar in these studies from three different perspectives: intrapatient between-day fasting blood glucose variability, intrapatient between-day daily mean blood glucose variability, and intrapatient within-day blood glucose variability.
Results
Overall, evaluations of all three indices showed that intrapatient blood glucose variability was similar between LY IGlar and IGlar throughout the study periods both in the overall populations of patients with T1D and T2D and also in the subgroup of Japanese patients with T1D.
Conclusion
Intrapatient blood glucose variability between LY IGlar and IGlar was shown to be similar in patients with T1D or T2D.
Clinical Trial Registration
NCT01421147 (ELEMENT-1) and NCT01421459 (ELEMENT-2).
Funding
Eli Lilly and Company (Indianapolis, IN, USA); Boehringer-Ingelheim (Ridgefield, CT, USA); Eli Lilly Japan K.K. (Kobe, Japan) and Nippon Boehringer Ingelheim Co., Ltd. (Tokyo, Japan).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
LY2963016 insulin glargine (LY IGlar) is the first biosimilar insulin to receive marketing authorization in the European Union [1] and Japan [2] and was authorized as the first follow-on insulin in the USA [3]. LY IGlar has an identical primary amino acid sequence and the same pharmaceutical form and strength as Lantus® insulin glargine (IGlar; recombinant DNA origin; Sanofi-Aventis, Paris, France) [4]. LY IGlar and IGlar have been shown to have similar pharmacokinetic (PK) and pharmacodynamic (PD) properties [5, 6]. Randomized phase 3 clinical trials in patients with type 1 diabetes (T1D; ELEMENT-1) or type 2 diabetes (T2D; ELEMENT-2) further demonstrated that LY IGlar and IGlar have similar efficacy, safety, and immunogenicity profiles [7,8,9]. In both ELEMENT-1 and ELEMENT-2, the change in glycated hemoglobin (HbA1c) from baseline to the 24-week endpoint was used to demonstrate the equivalence of efficacy between LY IGlar and IGlar [7, 8].
To show similarity of the two products, it is important to make comparisons from both inter- and intrasubject perspectives [10]. For LY IGlar and IGlar, similarity in both inter- and intrasubject PK/PD parameters has been shown in three biopharmaceutical studies conducted in healthy subjects [6]. Reports from ELEMENT-1 and ELEMENT-2 showed similarity of glycemic control between LY IGlar and IGlar based on HbA1c [7, 8], which is a good indicator of interpatient (i.e., population average) glycemic control but is not an appropriate indicator of intrapatient glycemic control [11,12,13]. Patients with the same HbA1c value may have small or large fluctuations in blood glucose profiles over time, as HbA1c does not reflect daily or between-day variations in blood glucose levels. Therefore, previously reported comparisons of LY IGlar and IGlar based on HbA1c have shown that they are similar in glycemic control from an interpatient perspective, but considering the limitations of HbA1c in reflecting blood glucose variability, the intrapatient perspective needs to be further assessed. Additionally, differences between Asians and Caucasians in, for example, genetic susceptibility to diabetes and underlying pathophysiology, body mass index, diet, insulin dose requirements, and postprandial hyperglycemia, have been suggested/reported [14, 15]. Given these differences, blood glucose variability data in such a subpopulation is of clinical interest not only in Japan but also in other East Asian countries. The ELEMENT-1 study included a Japanese subpopulation as an East Asian subpopulation and thus allowed for a separate analysis. Accordingly, the aim of this study was to evaluate and compare the intrapatient variability of daily blood glucose values in patients treated with LY IGlar and IGlar in the overall population of patients with T1D and in the subgroup of Japanese patients in ELEMENT-1, and in the overall population of patients with T2D in ELEMENT-2.
Methods
Study Design
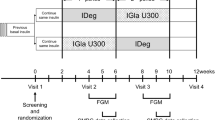
In this report, data from ELEMENT-1 and ELEMENT-2 were analyzed. Detailed study methods and results for the primary endpoints for both studies have been reported [7, 8] as well as those for the subgroup analysis of Japanese patients in ELEMENT-1 [16]. ELEMENT-1 was a 52-week open-label study of patients with T1D being treated with basal and bolus insulin; it included a 24-week treatment period and a 28-week extension period [7]. ELEMENT-2 was a 24-week double-blind study of patients with T2D who were insulin-naive or previously treated with IGlar [8]. In both studies, patients were randomized to LY IGlar or IGlar treatment. The initial dose was equivalent to their prestudy insulin dose, except that patients with T2D who were insulin-naive started with 10 units of LY IGlar or IGlar. The LY IGlar or IGlar dose was titrated on the basis of daily blood glucose levels [7, 8]. Both of these studies were conducted in accordance with the International Conference on Harmonization Guidelines for Good Clinical Practice and the Declaration of Helsinki [17], and all patients provided written informed consent [7, 8]. This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Outcomes
Intrapatient blood glucose variability with LY IGlar and IGlar was assessed and compared using three measures of variability derived from 7-point self-monitored blood glucose (SMBG) profiles (premeal for each meal, postmeal for breakfast and lunch, bedtime, and 03:00 h), which were collected three times in the 2-week period before each clinic visit. Patients visited the clinic at weeks 0 (baseline), 2, 6, 12, 18, 24, 30, 36, 44, and 52 in ELEMENT-1 and at weeks 0 (baseline), 2, 12, and 24 in ELEMENT-2.
The three measures of variability analyzed were intrapatient between-day FBG variability, intrapatient between-day daily mean blood glucose variability, and intrapatient within-day blood glucose variability. Intrapatient between-day FBG variability for each visit was calculated as the standard deviation (SD) of the three morning premeal blood glucose values. Intrapatient between-day daily mean blood glucose variability for each visit was calculated as the SD of the three daily means of the 7-point SMBG profiles. Intrapatient within-day blood glucose variability for each visit was calculated as the mean of the three SDs of the 7-point SMBG profiles.
Statistical Analysis
The analysis population was comprised of all randomized patients who took at least one dose of study drug. If the measurement for a visit was missing, the previous non-missing measurement was analyzed using last observation carried forward (LOCF) methodology. HbA1c is shown in the NGSP value as recommended by the Japan Diabetes Society [18]. Each measurement was analyzed using an analysis of covariance model with country, time of basal insulin injection (daytime, evening/bedtime), and treatment as fixed effects, and baseline HbA1c as covariate. The analyses described in this manuscript were prespecified in the study protocol and/or statistical analysis plan.
Results
In ELEMENT-1, 535 patients with T1D were included in the analysis (268 LY IGlar and 267 IGlar). Of these, 100 were Japanese patients (49 LY IGlar and 51 IGlar). In ELEMENT-2, 756 patients with T2D were included in the analysis (376 LY IGlar and 380 IGlar). No Japanese patients were included in ELEMENT-2.
Least-squares (LS) means of intrapatient between-day FBG variability in both treatment groups ranged from 36.3 to 46.7 mg/dL in the overall population of patients with T1D, and from 33.2 to 56.3 mg/dL in the subgroup of Japanese patients. There were no statistically significant differences at any visit or at endpoint (LOCF) in patients with T1D, except for two visits (weeks 6 and 52) in the overall population, where the values were significantly lower in the LY IGlar group compared with the IGlar group (Fig. 1a, b). In the population with T2D, LS means of intrapatient between-day FBG variability in both treatment groups ranged from 13.7 to 18.4 mg/dL, and no statistically significant differences were observed at any visit or at endpoint (LOCF) (Fig. 1c).
Intrapatient between-day FBG variability. a All patients with T1D. b Japanese patients with T1D. c Patients with T2D. Intrapatient between-day FBG variability calculated using standard deviation of FBG from SMBG (standard deviation of 3 values per visit) (bar graph; left axis). To display dispersion, mean FBG is also shown (line graph; right axis). Treatment comparisons at each visit were analyzed using ANCOVA: *p < 0.05 LY IGlar vs IGlar. For a and b, data from weeks 2, 18, 30, 44, and week 24 (LOCF) are not shown. No statistical significance was observed at these visits or endpoints. ANCOVA analysis of covariance, BL baseline, FBG fasting blood glucose, IGlar Lantus insulin glargine, LOCF last observation carried forward, LS least-squares, LY IGlar LY2963016 insulin glargine, N number of patients with data, SMBG self-monitored blood glucose, T1D type 1 diabetes, T2D type 2 diabetes
LS means of intrapatient between-day daily mean blood glucose variability in both treatment groups ranged from 15.6 to 21.5 mg/dL in the overall population with T1D and from 22.1 to 30.8 mg/dL in the subgroup of Japanese patients. There were no statistically significant differences between treatment groups at any visit or at endpoint (LOCF) in patients with T1D (Fig. 2a, b). In the population with T2D, LS means of intrapatient between-day daily mean blood glucose variability in both treatment groups ranged from 9.8 to 14.0 mg/dL, and no statistically significant differences were observed at any visit or at endpoint (LOCF) (Fig. 2c).
Intrapatient between-day daily mean BG variability. a All patients with T1D. b Japanese patients with T1D. c Patients with T2D. Intrapatient between-day daily mean BG variability calculated using standard deviation of daily mean BG from 7-point SMBG profiles (standard deviation of 3 values per visit) (bar graph; left axis). To display dispersion, daily mean BG is also shown (line graph; right axis). Treatment comparisons at each visit were analyzed using ANCOVA; not significant at all visits. For a and b, data from weeks 2, 18, 30, 44, and week 24 (LOCF) are not shown. No statistical significance was observed at these visits or endpoints. ANCOVA analysis of covariance, BG blood glucose, BL baseline, IGlar Lantus insulin glargine, LOCF last observation carried forward, LS least-squares, LY IGlar LY2963016 insulin glargine, N number of patients with data, SMBG self-monitored blood glucose, T1D type 1 diabetes, T2D type 2 diabetes
LS means of intrapatient within-day blood glucose variability in both treatment groups ranged from 44.1 to 56.7 mg/dL in the overall population with T1D and from 49.1 to 64.8 mg/dL in the subgroup of Japanese patients. There were no statistically significant differences between treatment groups at any visit or at endpoint (LOCF) in patients with T1D (Fig. 3a, b). In the population with T2D, LS means of intrapatient within-day blood glucose variability in both treatment groups ranged from 32.1 to 40.0 mg/dL, and no statistically significant differences were observed at any visit or at endpoint (LOCF) (Fig. 3c).
Intrapatient within-day BG variability. a All patients with T1D. b Japanese patients with T1D. c Patients with T2D. Intrapatient within-day daily mean BG variability calculated using mean of daily BG standard deviations from 7-point SMBG profiles (mean of 3 values per visit) (bar graph; left axis). To display dispersion, daily mean BG is also shown (line graph; right axis). Treatment comparisons at each visit were analyzed using ANCOVA; not significant at all visits. For a and b, data from weeks 2, 18, 30, 44, and week 24 (LOCF) are not shown. No statistical significance was observed at these visits or endpoints. ANCOVA analysis of covariance, BG blood glucose, BL baseline, LOCF last observation carried forward, LS least-squares, N number of patients with data, SMBG self-monitored blood glucose, T1D type 1 diabetes, T2D type 2 diabetes
Discussion
This is the first report of comparisons of intrapatient blood glucose variability between LY IGlar and IGlar using data from the ELEMENT-1 and ELEMENT-2 studies. In this analysis, intrapatient blood glucose variability was assessed and compared using three measures: intrapatient between-day FBG variability, intrapatient between-day daily mean blood glucose variability, and intrapatient within-day blood glucose variability.
Intrapatient between-day FBG variability showed day-to-day variability of FBG, which was used as part of dose adjustment algorithms in both ELEMENT-1 and ELEMENT-2, as it is used in actual clinical settings. Intrapatient between-day daily mean blood glucose variability and intrapatient within-day blood glucose variability were calculated using all measurements from 7-point SMBG profiles to show day-to-day and within-day variability in blood glucose, respectively. All three measures of intrapatient blood glucose variability after treatment with LY IGlar were similar to those with IGlar in the overall populations of patients with T1D and T2D and in the subgroup of Japanese patients with T1D.
In the LY IGlar development program, phase 1 studies with healthy volunteers showed similarities of both inter- and intrasubject PK and PD between LY IGlar and IGlar in an experimental setting using euglycemic clamps [6]. Similarity of average interpatient glycemic control (i.e., HbA1c) with LY IGlar and IGlar has also been reported in ELEMENT-1 and ELEMENT-2 [7, 8]; however, average interpatient HbA1c values cannot be used to appropriately estimate intrapatient glycemic control [11,12,13]. Assessment of intrapatient blood glucose variability of LY IGlar and IGlar using data from ELEMENT-1 and ELEMENT-2 (conducted in settings more similar to that of regular clinical practice than the biopharmaceutical studies mentioned above) indicated that intrapatient glycemic control was also similar between LY IGlar and IGlar. In combination with the similarity in interpatient average glycemic control reported previously, comprehensive similarity in glycemic control has been demonstrated for LY IGlar and IGlar in patients with T1D or T2D.
Since blood glucose variability captures fluctuations in daily glucose profiles, it is also considered a predictor of hypoglycemia [19]. Even though HbA1c has been used as the primary measure of glycemic control, it only reflects long-term (over 1–2 months) average blood glucose levels. HbA1c cannot capture shorter-term fluctuations and is only weakly associated with the risk of severe hypoglycemia [20]. The results from this analysis further support the similarity of glycemic control achieved with LY IGlar and IGlar in both the short (as measured by blood glucose variability) and the long term (as measured by HbA1c).
One limitation of this analysis is that the blood glucose variability measures were derived from 7-point SMBG profiles, which only provide snapshots of glucose concentrations, unlike continuous glucose monitoring, which captures blood glucose levels over time [21]. Even though the studies were designed to collect data at the times when maximum and minimum blood glucose were likely to be observed, actual maximum and minimum values may not have been captured.
Conclusions
Intrapatient blood glucose variability was similar between LY IGlar and IGlar in patients with T1D or T2D. In addition, the intrapatient blood glucose variability measures were also similar between LY IGlar and IGlar in a subgroup of Japanese patients with T1D. This analysis of intrapatient blood glucose variability provides additional support for the similarity of LY IGlar and IGlar.
Abbreviations
- ANCOVA:
-
Analysis of covariance
- FBG:
-
Fasting blood glucose
- IGlar:
-
Lantus insulin glargine
- LOCF:
-
Last observation carried forward
- LS:
-
Least-squares
- LY IGlar:
-
LY2963016 insulin glargine
- PD:
-
Pharmacodynamics
- PK:
-
Pharmacokinetics
- SD:
-
Standard deviation
- SMBG:
-
Self-monitored blood glucose
- T1D:
-
Type 1 diabetes
- T2D:
-
Type 2 diabetes
References
Eli Lilly and Company. European Commission grants Lilly and Boehringer Ingelheim’s insulin glargine product marketing authorisation in Europe: first biosimilar insulin to receive regulatory approval in the EU. Indianapolis, IN: Eli Lilly and Company, Boehringer Ingelheim; 2014. http://lilly.mediaroom.com/index.php?s=9042&item=137348. Accessed 09 Aug 2017.
Pharmaceuticals and Medical Devices Agency. New drugs approved in FY 2014. http://www.pmda.go.jp/files/000206818.pdf. Accessed 09 Aug 2017.
US Food and Drug Administration. FDA approves Basaglar, the first “follow-on” insulin glargine product to treat diabetes. 2015. http://www.fda.gov/newsevents/newsroom/pressannouncements/ucm477734.htm. Accessed 09 Aug 2017.
European Medicines Agency. European public assessment report: Abasria. 2014. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/002835/WC500175383.pdf. Accessed 09 Aug 2017.
Heise T, Zhang X, Lam ECQ, et al. Duration of action of 2 insulin glargine products, LY2963016 and Lantus, in subjects with type I diabetes mellitus (T1DM). Diabetes. 2014;63(Suppl. 1):A228.
Linnebjerg H, Lam EC, Seger ME, et al. Comparison of the pharmacokinetics and pharmacodynamics of LY2963016 insulin glargine and EU- and US-approved versions of Lantus insulin glargine in healthy subjects: three randomized euglycemic clamp studies. Diabetes Care. 2015;38(12):2226–33.
Blevins TC, Dahl D, Rosenstock J, et al. Efficacy and safety of LY2963016 insulin glargine compared with insulin glargine (Lantus®) in patients with type 1 diabetes in a randomized controlled trial: the ELEMENT 1 study. Diabetes Obes Metab. 2015;17(8):726–33.
Rosenstock J, Hollander P, Bhargava A, et al. Similar efficacy and safety of LY2963016 insulin glargine and insulin glargine (Lantus®) in patients with type 2 diabetes who were insulin-naïve or previously treated with insulin glargine: a randomized, double-blind controlled trial (the ELEMENT 2 study). Diabetes Obes Metab. 2015;17(8):734–41.
Ilag LL, Deeg MA, Costigan T, et al. Evaluation of immunogenicity of LY2963016 insulin glargine compared with Lantus insulin glargine in patients with type 1 or type 2 diabetes mellitus. Diabetes Obes Metab. 2016;18(2):159–68.
US Food and Drug Administration. Guidance for industry statistical approaches to establishing bioequivalence. 2001. https://www.fda.gov/downloads/drugs/guidances/ucm070244.pdf. Accessed 15 Aug 2017.
Beck RW, Connor CG, Mullen DM, Wesley DM, Bergenstal RM. The fallacy of average: how using HbA(1c) alone to assess glycemic control can be misleading. Diabetes Care. 2017;40(8):994–9.
Kovatchev B, Cobelli C. Glucose variability: timing, risk analysis, and relationship to hypoglycemia in diabetes. Diabetes Care. 2016;39(4):502–10.
Smith-Palmer J, Brändle M, Trevisan R, Orsini Federici M, Liabat S, Valentine W. Assessment of the association between glycemic variability and diabetes-related complications in type 1 and type 2 diabetes. Diabetes Res Clin Pract. 2014;105(3):273–84.
Hu FB. Globalization of diabetes: the role of diet, lifestyle, and genes. Diabetes Care. 2011;34(6):1249–57.
Gu T, Hong T, Zhang P, et al. Insulin glargine combined with oral antidiabetic drugs for Asians with type 2 diabetes mellitus: a pooled analysis to identify predictors of dose and treatment response. Diabetes Ther. 2018;9(2):771–87.
Jinnouchi H, Chida D, Nishiyama H, Imaoka T. Similar efficacy and safety with LY2963016 (insulin glargine) and Lantus® (insulin glargine) in Japanese patients with type 1 diabetes mellitus: a subgroup analysis of the ELEMENT 1 study. Prog Med. 2015;35(9):1497–506 (in Japanese).
World Medical Association Declaration of Helsinki. Recommendations guiding physicians in biomedical research involving human subjects. JAMA. 1997;277(11):925–6.
Kashiwagi A, Kasuga M, Araki E, et al. International clinical harmonization of glycated hemoglobin in Japan: from Japan Diabetes Society to National Glycohemoglobin Standardization Program values. J Diabetes Investig. 2012;3(1):39–40.
DeVries JH. Glucose variability: where it is important and how to measure it. Diabetes. 2013;62(5):1405–8.
Kovatchev BP. Metrics for glycaemic control—from HbA(1c) to continuous glucose monitoring. Nat Rev Endocrinol. 2017;13(7):425–36.
Patton SR, Clements MA. Continuous glucose monitoring versus self-monitoring of blood glucose in children with type 1 diabetes—are there pros and cons for both? US Endocrinol. 2012;8(1):27–9.
Acknowledgements
Funding
The studies were funded by Eli Lilly and Company (Indianapolis, IN, USA) and Boehringer-Ingelheim (Ridgefield, CT, USA). Eli Lilly Japan K.K. (Kobe, Japan) and Nippon Boehringer Ingelheim Co., Ltd. (Tokyo, Japan) funded the article processing charges.
Medical Writing Assistance
The authors acknowledge the assistance of Michelle Carey, of Syneos Health, with the preparation of this article. This assistance was funded by Eli Lilly and Company (Indianapolis, IN, USA).
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, had full access to all of the data in this study, take complete responsibility for the integrity of the data and accuracy of the data analysis, and have given their approval for this version to be published.
Prior Presentation
This work was previously published as an abstract presented at the 60th Annual Meeting of the Japan Diabetes Society, Nagoya, Japan, May 2017 (III-P-168).
Disclosures
Hiroshi Nishiyama is an employee of Eli Lilly Japan K.K. and has company stock options. Tomotaka Shingaki is an employee of Eli Lilly Japan K.K. Yumi Suzuki is an employee of Eli Lilly Japan K.K. Liza L. Ilag is an employee of Eli Lilly and Company and has company stock options.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Data Availability
The data sets analyzed during the current study are not publicly available. Lilly provides access to the individual patient data from studies on approved medicines and indications as defined by the sponsor-specific information on http://clinicalstudydatarequest.com. Researchers need to have an approved research proposal submitted through http://clinicalstudydatarequest.com. Access to the data will be provided in a secure data sharing environment after signing a data sharing agreement.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced digital features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.6280895.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Nishiyama, H., Shingaki, T., Suzuki, Y. et al. Similar Intrapatient Blood Glucose Variability with LY2963016 and Lantus® Insulin Glargine in Patients with Type 1 (T1D) or Type 2 Diabetes, Including a Japanese T1D Subpopulation. Diabetes Ther 9, 1469–1476 (2018). https://doi.org/10.1007/s13300-018-0450-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-018-0450-0