Abstract
Introduction
Glucagon-like peptide-1 (GLP-1) receptor agonists represent highly efficacious treatment options for type 2 diabetes. Liraglutide was amongst the first authorised for use in 2010, but once-weekly semaglutide represents the most efficacious GLP-1 analogue currently available for type 2 diabetes. The aim of the present analysis was therefore to evaluate the long-term cost-effectiveness of once-weekly semaglutide 1 mg versus liraglutide 1.8 mg with a lowered acquisition cost in the UK, as potentially lower cost liraglutide formulations may soon be developed.
Methods
Outcomes were projected over patients’ lifetimes using the IQVIA Core Diabetes Model (v9.0). Baseline cohort characteristics were sourced from SUSTAIN 2, with changes in HbA1c, blood pressure and body mass index applied from a network meta-analysis, in which SUSTAIN 2 was used to inform the semaglutide arm. Modelled patients received semaglutide or liraglutide for 3 years, after which treatment was intensified to basal insulin. Costs were accounted from a healthcare payer perspective and expressed in 2021 pounds sterling (GBP). The acquisition cost of liraglutide was reduced by 33% compared with the currently marketed formulation.
Results
Life expectancy and quality-adjusted life expectancy were projected to improve with once-weekly semaglutide 1 mg, by 0.05 years and 0.06 quality-adjusted life years, respectively, versus liraglutide 1.8 mg. Clinical benefits were due to a reduced incidence of diabetes-related complications with semaglutide. Direct costs were estimated to be GBP 280 lower with semaglutide, entirely because of avoidance of diabetes-related complications versus liraglutide. Semaglutide 1 mg was therefore considered dominant versus liraglutide 1.8 mg, even with the liraglutide price reduced by 33%.
Conclusion
Once-weekly semaglutide 1 mg is likely to represent a dominant treatment option versus liraglutide 1.8 mg for the treatment of type 2 diabetes in the UK, even with the liraglutide price reduced by 33%.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Once-weekly semaglutide 1 mg represents one of the most efficacious treatments for type 2 diabetes and has been associated with greater improvements in blood glucose levels and body weight versus liraglutide 1.8 mg in a published network meta-analysis, factors associated with a reduced risk of long-term diabetes-related complications |
Liraglutide was one of the first glucagon-like peptide-1 receptor agonists authorised for use in 2010 and potentially lower cost formulations may soon be developed |
The present study aimed to evaluate the long-term cost-effectiveness of once-weekly semaglutide 1 mg versus liraglutide 1.8 mg with a lowered acquisition cost in the UK |
What was learned from the study? |
Once-weekly semaglutide 1 mg was associated with a reduced incidence of long-term diabetes-related complications, improvements in life expectancy and quality-adjusted life expectancy of 0.05 years and 0.06 quality-adjusted life years, respectively, and cost savings of GBP 280 versus liraglutide 1.8 mg |
Semaglutide 1 mg was considered a dominant treatment option versus liraglutide 1.8 mg, even with the liraglutide price reduced by 33% |
Introduction
Type 2 diabetes is associated with a substantial clinical and economic burden in the UK. In 2021, diabetes was estimated to affect almost 4 million people in the UK, with approximately GBP 23 billion spent on treating the disease [1]. Therefore, intense pressure remains on UK healthcare systems to offer clinically effective and cost-effective medications. The majority of healthcare expenditure associated with diabetes is due to long-term complications, the incidence of which can be influenced by changes in glycated haemoglobin (HbA1c), as well as other physiological parameters such as blood pressure and body weight [2,3,4,5,6,7,8,9,10]. Indeed, guidelines published by the National Institute for Health and Care Excellence (NICE) in the UK indicate an HbA1c target of 6.5% (48 mmol/mol) for people with type 2 diabetes managed with lifestyle modifications and one antidiabetic agent, and a target of 7.0% (53 mmol/mol) for people with type 2 diabetes not adequately controlled by a single glucose-lowering drug, as well as a weight loss target of 5–10% [11]. Medications that can improve these outcomes over the short term can therefore offer benefits to people with diabetes and to healthcare payers by reducing the incidence and associated costs of long-term complications.
Amongst modern therapy options for type 2 diabetes, glucagon-like peptide-1 (GLP-1) receptor agonists represent one of the most efficacious treatment classes, offering clinically important reductions in blood glucose levels and body weight alongside a low risk of hypoglycaemia [12]. Once-daily liraglutide was one of the first approved GLP-1 receptor agonists in the UK (after exenatide) [13]. However, in the years since the approval of liraglutide, many other GLP-1 receptor agonists have been developed, and once-weekly semaglutide represents arguably the most effective GLP-1 receptor agonist currently available [12, 14, 15]. Indeed, once-weekly semaglutide was associated with significant cardiovascular benefits versus placebo in the SUSTAIN 6 trial, and significantly improved HbA1c, systolic blood pressure and body weight versus liraglutide 1.8 mg in a published network meta-analysis (NMA) in people with inadequate glycaemic control on 1–2 oral antidiabetic medications (OADs) [12, 16]. This correlates with head-to-head trial data from SUSTAIN 10, which indicated that once-weekly semaglutide 1 mg was associated with clinical benefits versus liraglutide 1.2 mg [17]. A subsequent long-term cost-effectiveness analysis indicated that once-weekly semaglutide 1 mg was dominant (associated with clinical benefits and cost savings) versus liraglutide 1.2 mg in the UK, but cost-conscious healthcare payers may require evidence that once-weekly semaglutide remains cost-effective versus the more efficacious dose of liraglutide, particularly given the potential development of lower cost liraglutide formulations after the expected patent expiry in 2023 [18, 19].
The aim of the present study was therefore to assess the long-term cost-effectiveness of once-weekly semaglutide 1 mg versus liraglutide 1.8 mg, with a lower liraglutide price applied, in people with type 2 diabetes with inadequate glycaemic control on 1–2 OADs in the UK.
Methods
Modelling Approach
The IQVIA Core Diabetes Model (version 9.0) was used to project clinical and cost outcomes over patients’ lifetimes (up to 50 years) [20]. The structure of the model, as well as its assumptions, features and capabilities, has been previously published [21]. Two validation studies for the model have also been performed [22, 23]. Model outputs relevant to the present study include life expectancy (measured in years), quality-adjusted life expectancy [measured in quality-adjusted life years (QALYs)], direct costs, incremental cost-effectiveness ratios (ICERs), cumulative incidence and time to onset of diabetes-related complications, and cost-effectiveness scatterplots and acceptability curves.
Future clinical and cost outcomes were discounted at 3.5% per annum, in line with guidance published by NICE [24]. The base case and deterministic sensitivity analyses were performed using a first-order Monte Carlo approach, with probabilistic sensitivity analysis (PSA) capturing second-order uncertainty. Cardiovascular outcomes were predicted with the UKPDS 68 risk equations in the base case analysis, in line with recommendations published by the model proprietors [22]. UK-specific life tables published by the World Health Organisation were used to inform background mortality [25].
Baseline Cohort Characteristics and Changes in Physiological Parameters
The SUSTAIN 2 clinical trial was used as a basis for baseline cohort characteristics, as this was used to inform the once-weekly semaglutide arm of the NMA (Table S1) [26]. Analyses using the IQVIA Core Diabetes Model require a large amount of specific data, including values for 19 baseline risk factors and the separate proportions of patients with history of 20 different complications. Real-world evidence publications often do not capture this level of data, and the SUSTAIN 2 trial was therefore seen as an ideal source, as data for each required input could be extracted (with the exception of alcohol and tobacco consumption, which were set to UK-specific values) [27, 28]. The mean (standard deviation) age of the cohort was 55.1 (10.0) years, with mean duration of diabetes of 7.0 (5.1) years, mean HbA1c of 8.1 (0.9)% [65.0 (10.2) mmol/mol] and mean body mass index (BMI) of 32.5 (6.2) kg/m2. These characteristics were previously used in a published cost-effectiveness analysis of once-weekly semaglutide in Spain [29].
Changes in physiological parameters, comprising HbA1c, systolic and diastolic blood pressure, and body weight (converted to BMI using the mean height from SUSTAIN 2) were sourced from the published NMA (Table S2) [16]. Changes in HbA1c, BMI, and systolic blood pressure were significantly greater with once-weekly semaglutide 1 mg versus liraglutide 1.8 mg. Changes in other physiological parameters and adverse event rates included in the IQVIA Core Diabetes Model, including serum lipids and hypoglycaemic event rates, were set to zero in both treatment arms to ensure that assumptions in these parameters did not drive cost-effectiveness outcomes.
Treatment Intensification and Long-Term Parameter Progression
A simple treatment algorithm was used to evaluate cost-effectiveness in the base case analysis. Modelled patients were assumed to receive once-weekly semaglutide or liraglutide for 3 years before discontinuing treatment and intensifying to basal insulin therapy with insulin Abasaglar® (a biosimilar insulin glargine available for use in the UK since 2015), which they received for the remainder of their lifetimes [30]. This approach was designed to avoid modelling assumptions unduly impacting the conclusions of the analysis and was in line with previously published analyses of GLP-1 receptor agonists and data from general practice in Europe, which reported a mean duration of treatment with GLP-1 receptor agonists of 2.4 years (rounded up to 3 years, as treatment switching in the IQVIA Core Diabetes Model can only occur at the end of an annual cycle) [31,32,33,34]. Alternative approaches to treatment switching were explored in sensitivity analyses.
Following application of changes in physiological parameters in the first year of the analysis, HbA1c and BMI were assumed to remain constant while patients received semaglutide or liraglutide. After 3 years, on intensification to basal insulin, differences were abolished by bringing HbA1c to 7.0% (53 mmol/mol) and returning BMI to baseline in both arms. This yielded a fair cost-effectiveness analysis, with differences in HbA1c and BMI maintained only while there was a difference in costs. Moreover, data from longer cardiovascular outcomes trials (CVOTs) of semaglutide (SUSTAIN 6) and liraglutide (LEADER) have shown that changes from baseline in HbA1c and body weight are generally sustained as long as patients are receiving therapies [35, 36]. Blood pressure and serum lipids were assumed to follow the natural progression algorithms built into the IQVIA Core Diabetes Model (as described by Palmer et al.) [21]. Following intensification to basal insulin, rates of 408 and 10 events per 100 person-years were applied for non-severe and severe hypoglycaemia, respectively, based on data from the UK Hypoglycaemia Study Group [37].
Cost and Utility Inputs
The analysis took a healthcare payer perspective in the UK and expressed costs in 2021 pounds sterling (GBP). Costs captured medications, consumables and management costs (sourced from list prices published in the Monthly Index of Medical Specialities [MIMS]) as well as the costs of treating diabetes-related complications (sourced from a published systematic literature review) [38, 39]. Costs of treating diabetes-related complications that were based on annually published sources were updated to the most recent versions available, while costs from non-annually updated sources were inflated where necessary to 2021 GBP using the Health and Community Health Services Index published by the Personal Social Services Research Unit (PSSRU; Table S3) [40,41,42,43,44,45,46,47]. The cost of liraglutide 1.8 mg was initially reduced by 33%, thereby matching the current daily price of the more commonly used 1.2 mg dose of liraglutide (and both doses of once-weekly semaglutide) in the UK. As generic GLP-1 receptor agonists are yet to be marketed, any price reduction applied for liraglutide would be entirely speculative. It was therefore assumed that any initial price reduction associated with a generic formulation would bring liraglutide 1.8 mg (currently the most costly GLP-1 receptor agonist in the UK) in line with the price of the 1.2 mg dose and other GLP-1 receptor agonists. Sensitivity analysis tested a further reduction in the liraglutide price.
Resource use from the semaglutide arm of SUSTAIN 2 was used to inform annual costs of treatment, with 100% of patients receiving concomitant metformin and 4.9% of patients receiving concomitant pioglitazone for the duration of the analysis. No needles were required for once-weekly semaglutide administration, as these are included in the semaglutide box, but one needle per day was required for liraglutide and basal insulin therapy. It was assumed that no self-monitoring of blood glucose testing was required with semaglutide and liraglutide, but one test per day was assumed to be required following intensification to basal insulin.
A 2014 review by Beaudet et al. was used to inform health-state utilities and event-based disutilities in the model, which represent the default utilities in the IQVIA Core Diabetes Model and met the criteria of the NICE reference case (Table S4) [48]. The only exceptions were hypoglycaemia disutilities, which were sourced from a 2014 publication by Evans et al. (published after the literature searches by Beaudet et al. had been completed) [49].
Key Drivers of Clinical Benefits
The key drivers of clinical outcomes were assessed by performing separate analyses that applied the differences in HbA1c, blood pressure and BMI in the semaglutide arm in turn, with all other parameters set to the values observed in the liraglutide arm. This allowed the impact of each physiological parameter to be assessed.
Sensitivity Analyses
Modelling the long-term consequences of diabetes from short-term clinical data is inherently associated with uncertainty. Sensitivity analyses were therefore performed to assess the robustness of the base case findings. These included: shortening the time horizon to 35, 20 and 10 years; applying clinical and cost discount rates of 0% and 6% in separate analyses; maintaining treatment effects in BMI for the duration of the analysis in both arms; applying the upper and lower 95% confidence intervals of the estimated treatment differences in HbA1c and BMI in turn in separate analyses; assuming treatment switching after 2, 5 or 10 years in separate analyses; applying the UKPDS progression equation for HbA1c and assuming treatment switching when HbA1c exceeded 7.5% (58 mmol/mol); applying the UKPDS 82 risk equations to predict clinical outcomes; increasing and decreasing the direct costs of treating diabetes-related complications by 10%; reducing the liraglutide price by a further 20% (representing a 47% reduction on the original Victoza 1.8 mg price); applying an alternative disutility for BMI, giving greater utility weight to weight changes; and probabilistic sensitivity analysis (PSA) [50].
Compliance with Ethics Guidelines
The SUSTAIN 2 trial was done in compliance with the International Conference on Harmonisation Good Clinical Practice Guideline and the Declaration of Helsinki and was approved by local ethics committees. Obtained written informed consent from all participants was included in the trial.
Results
Base Case Analysis
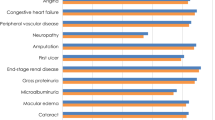
Long-term projections in patients with inadequate glycaemic control on 1–2 OADs indicated that once-weekly semaglutide 1 mg was associated with improvements in discounted life expectancy of 0.05 years and discounted quality-adjusted life expectancy of 0.06 QALYs versus liraglutide 1.8 mg (Table 1). Benefits in both life expectancy and quality of life with once-weekly semaglutide were due to a reduced cumulative incidence and delayed time to onset of diabetes-related complications (both micro- and macrovascular) over the long term (Fig. 1).
Direct costs were estimated to be GBP 280 lower with once-weekly semaglutide 1 mg versus liraglutide 1.8 mg over patients’ lifetimes (Fig. 2). Once-weekly semaglutide was associated with similar treatment costs to liraglutide 1.8 mg because of the comparable daily acquisition costs, with cost savings driven by avoidance of diabetes-related complications. Most notably, once-weekly semaglutide was associated with mean cost savings of GBP 77 per patient due to avoided ulcer, amputation and neuropathy complications and GBP 96 per patient due to avoided cardiovascular complications.
Projections of long-term outcomes indicated that both life expectancy and quality-adjusted life expectancy were improved with once-weekly semaglutide 1 mg compared with liraglutide 1.8 mg at a reduced cost from a healthcare payer perspective (Table 1). Once-weekly semaglutide 1 mg was therefore considered dominant versus liraglutide 1.8 mg for the treatment of type 2 diabetes in the UK, with a 33% reduction applied to the liraglutide price.
Key Drivers of Clinical Benefits
Testing for the key drivers of clinical benefits by applying the differences in HbA1c, blood pressure and BMI in turn indicated reductions in HbA1c as the key driver of improved quality-adjusted life expectancy for once-weekly semaglutide 1 mg versus liraglutide 1.8 mg, with improvements of 0.04 QALYs when only this difference between the treatment arms was applied. Differences in BMI and blood pressure made smaller contributions to the overall clinical benefit with semaglutide, with quality-adjusted life expectancy improvements of 0.03 and 0.02 QALYs, respectively, versus liraglutide when only these differences were applied.
Sensitivity Analyses
Sensitivity analyses tested a variety of the input parameters and assumptions used in the base case analysis, and these showed that results were robust (Table 2). Once-weekly semaglutide 1 mg remained dominant versus liraglutide 1.8 mg in all but two analyses. Use of the UKPDS progression equation for HbA1c and treatment switching at a 7.5% (58 mmol/mol) threshold led to improved clinical outcomes with semaglutide, but increased incremental costs due to patients receiving semaglutide and liraglutide for different lengths of time (4 years with semaglutide and 3 years with liraglutide). Once-weekly semaglutide was therefore associated with an ICER of GBP 1313 per QALY gained in this analysis. Reducing the liraglutide price by a further 20% (representing a 47% reduction on the original Victoza 1.8 mg price) led to increased costs with once-weekly semaglutide, but it remained cost-effective with an ICER of GBP 4365 per QALY gained versus liraglutide 1.8 mg. Changes in incremental outcomes in all other sensitivity analyses were minor and reinforced the conclusions of the base case analysis.
PSA, with sampling around baseline cohort characteristics, treatment effects and costs, showed similar mean incremental results to the base case analysis but with increased measures of variance (Fig. 3). Once-weekly semaglutide 1 mg was associated with mean improvements in quality-adjusted life expectancy of 0.06 QALYs and direct cost savings of GBP 187 versus liraglutide 1.8 mg and was therefore considered dominant in the PSA. Based on these outcomes, and a willingness-to-pay threshold of GBP 20,000 per QALY gained, the probability that once-weekly semaglutide would be cost-effective versus liraglutide 1.8 mg was estimated to be 66.1% (Fig. 4).
Discussion
Based on the results of a published NMA, the present study has demonstrated that once-weekly semaglutide 1 mg is likely to represent a dominant treatment option versus liraglutide 1.8 mg for the treatment of type 2 diabetes in the UK, even when the liraglutide price is reduced by 33%, and a cost-effective treatment option when the liraglutide price is reduced by 47% [6]. Greater reductions in HbA1c, blood pressure and BMI from the NMA with once-weekly semaglutide led to a reduced incidence and delayed time to onset of diabetes-related complications over the long term. While these differences were small, benefits were notably in favour of once-weekly semaglutide, with reductions in complications affecting both mortality and quality of life (such as cardiovascular complications) and solely quality of life (such as ophthalmic complications). These results should inform healthcare payers, physicians and people with type 2 diabetes in the UK when considering GLP-1 receptor agonists as potential treatment options.
Affordability of medications in the UK remains a key consideration for the NHS, with limited resources meaning cost-effective medications can still prove unaffordable due to their budget impact. As GLP-1 receptor agonists are a modern treatment class for type 2 diabetes, many are still under patent and potentially less costly formulations are yet to be manufactured. However, clinical evidence shows that GLP-1 receptor agonists have differences in efficacy and that there is a clear hierarchy in terms of effectiveness. Once-weekly semaglutide is arguably the most efficacious GLP-1 receptor agonist currently available, as it has shown clinical benefits versus fellow GLP-1 receptor agonists exenatide extended-release in SUSTAIN 3, dulaglutide in SUSTAIN 7, liraglutide 1.2 mg in SUSTAIN 10, and liraglutide 1.8 mg and lixisenatide in an NMA [15,16,17, 51, 52]. Previous cost-effectiveness analyses conducted in the UK setting have also indicated once-weekly semaglutide as a highly cost-effective medication versus dulaglutide and liraglutide 1.2 mg based on the SUSTAIN 7 and 10 clinical trials, and cost-effectiveness analyses in other European settings have demonstrated once-weekly semaglutide to be cost-effective versus GLP-1 receptor agonists exenatide and lixisenatide [18, 29, 31, 33, 53,54,55,56]. The present analysis adds to this evidence by demonstrating that once-weekly semaglutide is more effective and cost saving versus the most efficacious dose of liraglutide when the price of liraglutide is reduced by 33% in the base case analysis and highly cost-effective when the price is reduced by a further 20% (representing a 47% reduction on the original Victoza price) in a sensitivity analysis. While other treatment options exist for type 2 diabetes (including more radical surgical interventions), these comparisons were beyond the scope of the present study, from which results indicate once-weekly semaglutide as an attractive treatment option versus liraglutide.
The present analysis did not capture data from CVOTs of semaglutide and liraglutide, which could be seen as a limitation. Once-weekly semaglutide was associated with significant reductions in nonfatal stroke versus placebo in SUSTAIN 6, while liraglutide was associated with significant reductions in both cardiovascular and all-cause mortality versus placebo in LEADER [12]. However, these outcomes are not easily incorporated into current models for type 2 diabetes, which rely on progression and risk equations developed long before modern diabetes therapies were available [60]. A major pitfall is the potential double counting of benefits, as the mechanisms of action behind the outcomes observed in CVOTs are not currently known and could be in part due to changes in physiological parameters already applied in the analysis [57, 58]. Moreover, there is a risk of inappropriate application of hazard ratios derived from populations with high cardiovascular risk or prior cardiovascular disease in more general populations with type 2 diabetes [59, 60]. Differences between the patient populations evaluated in SUSTAIN 6 and LEADER must also be considered when comparing outcomes, which may not be generalisable across studies. An ideal solution to using these pertinent data in modelling analyses would be sharing of patient-level data from each CVOT and development of novel risk equations, although this would require significant encouragement and funding from health technology assessment bodies [60].
The recent update of the NICE clinical guidelines for type 2 diabetes in the UK have positioned GLP-1 receptor agonists as treatment options only when treatment with three oral antidiabetic medications is ineffective, and only in people with a BMI ≥ 35 kg/m2 or in people with a BMI < 35 kg/m2 for whom insulin therapy would have significant occupational implications or where weight loss would provide substantial benefits [11]. This is at odds with both guidelines published by the European Association for the Study of Diabetes, which recommend GLP-1 receptor agonists as a preferred second-line treatment option in patients with established cardiovascular disease or high cardiovascular risk and as a potential second-line therapy in populations with a compelling need to minimise hypoglycaemia or weight gain or promote weight loss, and results of previous health economic analysis in the UK and other European settings, which have indicated once-weekly semaglutide as a cost-effective treatment option versus oral agents, including dipeptidyl peptidase-4 (DPP-4) inhibitor sitagliptin and sodium-glucose cotransporter-2 (SGLT-2) inhibitor empagliflozin [12, 29, 61, 62]. Sub-group analyses have also demonstrated that once-weekly semaglutide remains consistently efficacious across populations with different baseline HbA1c and BMI values rather than just those with a high BMI at baseline [63,64,65]. Given these results, it could be argued that the BMI limits imposed by NICE on GLP-1 receptor agonists are too restrictive, and once-weekly semaglutide could represent a cost-effective treatment option in the full population of people with type 2 diabetes in the UK.
A limitation inherent to all long-term modelling studies of type 2 diabetes interventions was the reliance on projection of short-term data to long-term outcomes. However, this is an essential tenet of all long-term modelling studies and arguably provides the best available evidence for decision making in absence of long-term clinical trial data. Projecting outcomes over the entirety of patients’ lifetimes is also recommended by the American Diabetes Association in the computer modelling guidance for interventions for type 2 diabetes [20]. Throughout the analysis, every effort was made to limit uncertainty by utilising a published and validated model and by performing extensive sensitivity analyses around the key aspects of the study, which did not change the conclusion that once-weekly semaglutide was cost-effective.
Conclusion
Once-weekly semaglutide 1 mg likely represents a highly cost-effective treatment option versus liraglutide 1.8 mg for the treatment of people with type 2 diabetes in the UK, even with a reduced price applied for liraglutide.
References
International Diabetes Federation (IDF). Diabetes Atlas—10th Edition. 2021. https://diabetesatlas.org/data/en/country/209/gb.html. Accessed 27 Apr 2022.
UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837–53.
ADVANCE Collaborative Group, Patel A, MacMahon S, Chalmers J, Neal B, Billot L, Woodward M, Marre M, Cooper M, Glasziou P, Grobbee D, Hamet P, Harrap S, Heller S, Liu L, Mancia G, Mogensen CE, Pan C, Poulter N, Rodgers A, Williams B, Bompoint S, de Galan BE, Joshi R, Travert F. ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358:2560–72.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359:1577–89.
Stettler C, Allemann S, Jüni P, Cull CA, Holman RR, Egger M, Krähenbühl S, Diem P. Glycemic control and macrovascular disease in types 1 and 2 diabetes mellitus: meta-analysis of randomized trials. Am Heart J. 2006;152(1):27–38.
Ismail-Beigi F, Craven T, Banerji MA, Basile J, Calles J, Cohen RM, Cuddihy R, Cushman WC, Genuth S, Grimm RH Jr, Hamilton BP, Hoogwerf B, Karl D, Katz L, Krikorian A, O’Connor P, Pop-Busui R, Schubart U, Simmons D, Taylor H, Thomas A, Weiss D, Hramiak I, ACCORD trial group. Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet. 2010;376:419–30.
UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ. 1998;317(7160):703–13.
Cholesterol Treatment Trialists’ (CTT) Collaborators, Kearney PM, Blackwell L, Collins R, Keech A, Simes J, Peto R, Armitage J, Baigent C. Efficacy of cholesterol-lowering therapy in 18,686 people with diabetes in 14 randomised trials of statins: a meta-analysis. Lancet. 2008;371(9607):117–25.
Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med. 2008;358:580–91.
Griffin SJ, Borch-Johnsen K, Davies MJ, Khunti K, Rutten GE, Sandbæk A, Sharp SJ, Simmons RK, van den Donk M, Wareham NJ, Lauritzen T. Effect of early intensive multifactorial therapy on 5-year cardiovascular outcomes in individuals with type 2 diabetes detected by screening (ADDITION-Europe): a cluster-randomised trial. Lancet. 2011;378(9786):156–67.
National Institute for Health and Care Excellence (NICE). Type 2 diabetes in adults: management (NG28). 2022. https://www.nice.org.uk/guidance/ng28/resources/visual-summary-full-version-choosing-medicines-for-firstline-and-further-treatment-pdf-10956472093. Accessed 13 May 2022.
Buse JB, Wexler DJ, Tsapas A, et al. 2019 Update to: Management of Hyperglycemia in Type 2 Diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2019; dci190066.
National Institute for Health and Care Excellence (NICE). Liraglutide for the treatment of type 2 diabetes mellitus (TA203). 2010. https://www.nice.org.uk/guidance/ta203. Accessed 27 Mar 2023.
European Medicines Agency (EMA). Ozempic (semaglutide). 2018. https://www.ema.europa.eu/en/medicines/human/EPAR/ozempic. Accessed 30 May 2022.
Trujillo JM, Nuffer W, Smith BA. GLP-1 receptor agonists: an updated review of head-to-head clinical studies. Ther Adv Endocrinol Metab. 2021;12:1–15.
Nuhoho S, Gupta J, Hansen BB, Fletcher-Louis M, Dang-Tan T, Paine A. Orally administered semaglutide versus GLP-1 RAs in patients with type 2 diabetes previously receiving 1–2 oral antidiabetics: systematic review and network meta-analysis. Diabetes Ther. 2019;10(6):2183–99.
Capehorn MS, Catarig AM, Furberg JK, et al. Efficacy and safety of once-weekly semaglutide 1.0mg vs once-daily liraglutide 1.2mg as add-on to 1–3 oral antidiabetic drugs in subjects with type 2 diabetes (SUSTAIN 10). Diabetes Metab. 2020;46(2):100–9.
Johansen P, Chubb B, Hunt B, Malkin SJP, Sandberg A, Capehorn M. Evaluating the long-term cost-effectiveness of once-weekly semaglutide versus once-daily liraglutide for the treatment of type 2 diabetes in the UK. Adv Ther. 2020;37(5):2427–41.
Novo Nordisk. Annual report 2021. 2022. https://www.novonordisk.com/content/dam/nncorp/global/en/investors/irmaterial/annual_report/2022/novo-nordisk-annual-report-2021.pdf. Accessed 16 Mar 2023.
American Diabetes Association Consensus Panel. Guidelines for computer modeling of diabetes and its complications. Diabetes Care. 2004;27:2262–5.
Palmer AJ, Roze S, Valentine WJ, Minshall ME, Foos V, Lurati FM, et al. The CORE Diabetes Model: projecting long-term clinical outcomes, costs and cost-effectiveness of interventions in diabetes mellitus (types 1 and 2) to support clinical and reimbursement decision-making. Curr Med Res Opin. 2004;20(Suppl 1):S5-26.
Palmer AJ, Roze S, Valentine WJ, Minshall ME, Foos V, Lurati FM, et al. Validation of the CORE Diabetes Model against epidemiological and clinical studies. Curr Med Res Opin. 2004;20(Suppl 1):S27-40.
McEwan P, Foos V, Palmer JL, Lamotte M, Lloyd A, Grant D. Validation of the IMS CORE diabetes model. Value Health. 2014;17:714–24.
National Institute for Health and Care Excellence. The guidelines manual: Process and methods (PMG6). https://www.nice.org.uk/process/pmg6/resources/the-guidelines-manual-pdf-2007970804933. Accessed 10 Dec 2021.
World Health Organisation. Global Health Observatory data repository: Life tables by country (United Kingdom). 2020. https://apps.who.int/gho/data/view.main.LT62260?lang=en. Accessed 10 Dec 2021.
Ahrén B, Masmiquel L, Kumar H, Sargin M, Karsbøl JD, Jacobsen SH, Chow F. Efficacy and safety of once-weekly semaglutide versus once-daily sitagliptin as an add-on to metformin, thiazolidinediones, or both, in patients with type 2 diabetes (SUSTAIN 2): a 56-week, double-blind, phase 3a, randomised trial. Lancet Diabetes Endocrinol. 2017;5(5):341–54.
Office for National Statistics. Adult Smoking Habits in Great Britain. 2019. https://www.ons.gov.uk/file?uri=/peoplepopulationandcommunity/healthandsocialcare/drugusealcoholandsmoking/datasets/adultsmokinghabitsingreatbritain/2019/adultsmokinghabitsingreatbritain2019final.xls. Accessed 10 Dec 2021.
World Health Organisation. Global Alcohol Report. 2014. http://www.who.int/substance_abuse/publications/global_alcohol_report/profiles/gbr.pdf. Accessed 10 Dec 2021.
Martín V, Vidal J, Malkin SJP, Hallén N, Hunt B. Evaluation of the long-term cost-effectiveness of once-weekly semaglutide versus dulaglutide and sitagliptin in the Spanish setting. Adv Ther. 2020;37(10):4427–45.
National Institute for Health and Care Excellence (NICE). Diabetes mellitus type 1 and type 2: insulin glargine biosimilar (Abasaglar) (ESNM64). 2015. https://www.nice.org.uk/advice/esnm64/chapter/key-points-from-the-evidence. Accessed 27 Mar 2023.
Viljoen A, Hoxer CS, Johansen P, Malkin S, Hunt B, Bain SC. Evaluation of the long-term cost-effectiveness of once-weekly semaglutide versus dulaglutide for the treatment of type 2 diabetes mellitus in the UK. Diabetes Obes Metab. 2019;21(3):611–21.
Hunt B, Vega-Hernandez G, Valentine WJ, Kragh N. Evaluation of the long-term cost-effectiveness of liraglutide versus lixisenatide for treatment of type 2 diabetes mellitus in the UK setting. Diabetes Obes Metab. 2017;19(6):842–9.
Hunt B, Malkin SJP, Moes RGJ, Huisman EL, Vandebrouck T, Wolffenbuttel B. Once-weekly semaglutide for patients with type 2 diabetes: a cost-effectiveness analysis in the Netherlands. BMJ Open Diabetes Res Care. 2019;7: e000705.
Heap G. Decision Resources Group Report: Current Treatment Type 2 Diabetes Detailed, Expanded Analysis (EU5). https://decisionresourcesgroup.com/report/499707-biopharma-type-2-diabetes-current-treatment-detailed/. Accessed 12 Mar 2018.
Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834–44.
Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375(4):311–22.
UK Hypoglycaemia Study Group. Risk of hypoglycaemia in types 1 and 2 diabetes: effects of treatment modalities and their duration. Diabetologia. 2007;50(6):1140–7.
Monthly Index of Medical Specialities. https://www.mims.co.uk/. Accessed 10 May 2022.
Rtveladze K, Hoxer CS. Cost of type 2 diabetes management and associated complications in the United Kingdom. Value Health. 2017;20(9):A477.
NHS England. National Tariff Payment System 2022/23. 2022. https://www.england.nhs.uk/publication/national-tariff-payment-system-documents-annexes-and-supporting-documents/. Accessed 1 May 2022.
Alva ML, Gray A, Mihaylova B, Leal J, Holman RR. The impact of diabetes-related complications on healthcare costs: new results from the UKPDS (UKPDS 84). Diabetes Med. 2015;32(4):459–66.
Danese M, Gleeson M, Kutikova L, Griffiths R, Azough A, Khunti K, et al. Costs of cardiovascular (CV) events in the United Kingdom (UK) using real-world data. Value Health. 2015;18:A335–766.
Kent S, Schlackow I, Lozano-Kuhne J, Reith C, Emberson J, Haynes R, Gray A, Cass A, Baigent C, Landray MJ, Herrington W, Mihaylova B, SHARP Collaborative Group. What is the impact of chronic kidney disease stage and cardiovascular disease on the annual cost of hospital care in moderate-to-severe kidney disease? BMC Nephrol. 2015;16:65.
Chubb B, Tikkanen C. The cost of non-severe hypoglycaemia in Europe. Value Health. 2015;18(7):A611.
Hammer M, Lammert M, Mejias SM, Kern W, Frier BM. Costs of managing severe hypoglycaemia in three European countries. J Med Econ. 2009;12(4):281–90.
Ghatnekar O, Willis M, Persson U. Cost-effectiveness of treating deep diabetic foot ulcers with Promogran in four European countries. J Wound Care. 2002;11(2):70–4.
Curtis L, Burns A. Unit Costs of Health and Social Care 2021. Personal Social Services Research Unit, University of Kent, Canterbury. 2022. https://www.pssru.ac.uk/project-pages/unit-costs/unit-costs-of-health-and-social-care-2021/. Accessed 2 May 2022.
Beaudet A, Clegg J, Thuresson PO, Lloyd A, McEwan P. Review of utility values for economic modeling in type 2 diabetes. Value Health. 2014;17(4):462–70.
Evans M, Khunti K, Mamdani M, Galbo-Jorgensen CB, Gundgaard J, Bogelund M, Harris S. Health-related quality of life associated with daytime and nocturnal hypoglycaemic events: a time trade-off survey in five countries. Health Qual Life Outcomes. 2013;11(1):90.
Lee AJ, Morgan CL, Morrissey M, Wittrup-Jensen KU, Kennedy-Martin T, Currie CJ. Evaluation of the association between the EQ-5D (health-related utility) and body mass index (obesity) in hospital-treated people with Type 1 diabetes, Type 2 diabetes and with no diagnosed diabetes. Diabet Med. 2005;22(11):1482–6.
Ahmann AJ, Capehorn M, Charpentier G, et al. Efficacy and safety of once-weekly semaglutide versus exenatide ER in subjects with type 2 diabetes (SUSTAIN 3): a 56-week, open-label, randomized clinical trial. Diabetes Care. 2018;41(2):258–66.
Pratley RE, Aroda VR, Lingvay I, et al. Semaglutide versus dulaglutide once weekly in patients with type 2 diabetes (SUSTAIN 7): a randomised, open-label, phase 3b trial. Lancet Diabetes Endocrinol. 2018;6(4):275–86.
Malkin SJP, Russel-Szymczyk M, Psota M, Hlavinkova L, Hunt B. The management of type 2 diabetes with once-weekly semaglutide versus dulaglutide: a long-term cost-effectiveness analysis in Slovakia. Adv Ther. 2019;36(8):2034–51.
Gæde P, Johansen P, Tikkanen CK, Pollock RF, Hunt B, Malkin SJP. Management of patients with type 2 diabetes with once-weekly semaglutide versus dulaglutide, exenatide ER, liraglutide and lixisenatide: a cost-effectiveness analysis in the Danish setting. Diabetes Ther. 2019;10(4):1297–317.
Malkin S, Russel-Szymczyk M, Liidemann G, Volke V, Hunt B. Once-weekly semaglutide versus once-daily liraglutide for the treatment of type 2 diabetes: a long-term cost-effectiveness analysis in Estonia. Diabetes Ther. 2019;10(1):159–76.
Carvalho D, Costa C, Hallén N, Baker-Knight J, Hunt B. A long-term cost-effectiveness analysis of treatments for type 2 diabetes in Portugal: Once-weekly semaglutide 1 mg versus once-daily empagliflozin 25 mg. Revista Portuguesa de Endocrinologia, Diabetes e Metabolismo. 2021;16(1–2):13–20.
Giugliano D, Chiodini P, Maiorino MI, Bellastella G, Esposito K. Cardiovascular outcome trials and major cardiovascular events: does glucose matter? A systematic review with meta-analysis. J Endocrinol Invest. 2019;42(10):1165–9.
Evans M, Berry S, Nazeri A, Malkin SJ, Ashley D, Hunt B, Bain SC. The challenges and pitfalls of incorporating evidence from cardiovascular outcomes trials in health economic modelling of type 2 diabetes. Diabetes Obes Metab. 2022;25:639–48.
Si L, Willis MS, Asseburg C, et al. Evaluating the ability of economic models of diabetes to simulate new cardiovascular outcomes trials: a report on the ninth mount hood diabetes challenge. Value Health. 2020;23(9):1163–70.
Willis M, Asseburg C, Nilsson A, Neslusan C. Challenges and opportunities associated with incorporating new evidence of drug-mediated cardioprotection in the economic modeling of type 2 diabetes: a literature review. Diabetes Ther. 2019;10:1753–69.
Gorgojo-Martínez JJ, Malkin SJP, Martín V, Hallén N, Hunt B. Assessing the cost-effectiveness of a once-weekly GLP-1 analogue versus an SGLT-2 inhibitor in the Spanish setting: Once-weekly semaglutide versus empagliflozin. J Med Econ. 2020;23(2):193–202.
Capehorn M, Hallén N, Baker-Knight J, Glah D, Hunt B. Evaluating the cost-effectiveness of once-weekly semaglutide 1 mg versus empagliflozin 25 mg for treatment of patients with type 2 diabetes in the UK setting. Diabetes Therapy. 2021;12(2):537–55.
Viljoen A, Blüher M, Chow F, le Roux C, Rosenstock J, Lausvig NL, Yildirim E, Lingvay I. Semaglutide reduces body weight vs. dulaglutide across baseline BMI subgroups in SUSTAIN 7. Diabetes. 2018;67(Supplement 1):1083-P.
Bain S, Araki E, Desouza C, Garg S, Rose L, Tsoukas G, Bergan EQ, Derving Karsbøl J, Devries JH. Semaglutide reduces HbA1c across baseline HbA1c subgroups across SUSTAIN 1–5 clinical trials. Diabetes. 2017;66(Supplement 1):1123‐P.
Leiter LA, Charpentier G, Chaykin L, Garvey WT, Warren ML, Karsbøl JD, Thielke D, Masmiquel L. Semaglutide reduces body weight across baseline BMI subgroups across SUSTAIN 1–5. diabetes. 2017;66(Supplement 1):1105‐P.
Acknowledgements
Funding
This study and the journal’s Rapid Service Fee were supported by funding from Novo Nordisk A/S. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing and Editorial Assistance
The authors thank Barrie Chubb at Novo Nordisk A/S for editorial support.
Author Contributions
All authors contributed to the study conception and design. Data collection and analysis were performed by Samuel JP Malkin and Barnaby Hunt. The manuscript was written by Samuel JP Malkin, with input from all authors. All authors read and approved the final manuscript.
Disclosures
Marc Evans has received consulting fees or honoraria from Novo Nordisk, NAPP, MSD, AstraZeneca, Sunovion, and Novartis; and lecture/speaker bureau fees from Novo Nordisk, NAPP, Mundipharma, AstraZeneca, MSD, Sunovion, and Novartis. Sasha Berry is an employee of Novo Nordisk Ltd. Samuel JP Malkin and Barnaby Hunt are employees of Ossian Health Economics and Communications, which received consulting fees from Novo Nordisk A/S to support preparation of the analysis. Abheet Sharma was an employee of Novo Nordisk during development of the manuscript, but has since left the company and is now employed outside Novo Nordisk at IQVIA.
Compliance with Ethics Guidelines
The SUSTAIN 2 trial was done in compliance with the International Conference on Harmonisation Good Clinical Practice Guideline and the Declaration of Helsinki and was approved by local ethics committees. Obtained written informed consent from all participants included in the trial.
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Evans, M., Berry, S., Malkin, S.J.P. et al. Evaluating the Long-Term Cost-Effectiveness of Once-Weekly Semaglutide 1 mg Versus Liraglutide 1.8 mg: A Health Economic Analysis in the UK. Diabetes Ther 14, 1005–1021 (2023). https://doi.org/10.1007/s13300-023-01408-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01408-2