Abstract
Introduction
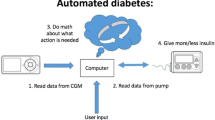
The advanced hybrid closed loop (AHCL) MiniMed™ 780G system changes basal insulin delivery every 5 min and auto bolus in response to sensor glucose values. We assessed the performance of the AHCL system in real-world settings for individuals with type 1 diabetes (T1DM) as well as user and clinician perspectives and satisfaction.
Methods
We held two peer group discussions: one having adults with T1DM/parents of children and adolescents with T1DM to understand their experiences with the AHCL system and another with healthcare providers (HCPs). Responses from the discussions were analyzed and categorized into themes by two independent researchers, with any inconsistencies resolved by consensus. We also analyzed data from the system uploaded to CareLink personal software. Glycemic outcomes, including time in range (TIR), time below range (TBR), time above range (TAR), mean sensor glucose (SG) levels, glucose management indicator (GMI), sensor use, and percentage of time spent in AHCL, were determined.
Results
The peer group discussions revealed numerous key themes and issues for each group, such as the significance of setting reasonable expectations, carbohydrate counting and bolus dosing, technical difficulties, and overall user experience. The users (n = 25; T1DM; 17 female; age 13.8 ± 7.49 years; A1C 6.54 ± 0.45%; duration of diabetes 6 ± 6.78 years) were very satisfied with the system. Most users experienced consistent blood glucose values with very few hypoglycemic episodes. However, there were a few limitations reported, such as hyperglycemic episodes caused by inaccuracies in carb counting, issues with sensor connectivity, and cannula blockages or kinking for those using insulin Fiasp. Users achieved a mean GMI of 6.4 ± 0.26%, TIR of 83.0 ± 8.12%, TBR (54–70 mg/dL) of 2.0 ± 0.81%, TBR* (< 54 mg/dL) of 0%. All of the users achieved a TIR of > 70%.
Conclusion
The use of the AHCL system in T1DM resulted in robust glycemic control, minimizing hypoglycemia. Providing training to both users and HCPs can help them use the system effectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Why carry out the study? |
To date, insulin pumps required lot of manual interventions but the average time in range remained suboptimal. |
780G is the first ever advanced device with both basal and bolus automation. |
The real-world experience could be different from the data from clinical trials. |
User experience will provide not only the merits but also the demerits of a new technology. |
What was learned from the study? |
Users met recommended goals for glucose management indicator (GMI), time in range (TIR), and times below range (TBR) (both TBR < 70 and TBR < 54). |
All users (100%) achieved the recommended GMI goal of < 7.0% and TIR goal of > 70%. |
Most users had consistent blood glucose values with minimal hypoglycemic episodes. |
Limitations reported included hyperglycemic episodes from inaccuracies in carb counting, sensor connectivity issues, and cannula blockages or kinking for insulin Fiasp users. |
Training for users and healthcare providers is essential for device familiarity and effective use. |
Introduction
The early implementation of continuous glucose monitoring (CGM), continuous subcutaneous insulin infusion (CSII), or automated insulin delivery (AID) in diabetes treatment can be advantageous, based on the requirements and preferences of the person and/or caregiver [1]. The integrated use of CGM and insulin pump therapy, together with algorithms for control, known as the “closed loop” or “artificial pancreas” system, has significantly improved the management of type 1 diabetes mellitus (T1DM) [2]. This system monitors glucose levels in real time and automatically adjusts insulin delivery on the basis of algorithms, allowing for a more personalized and dynamic approach to insulin delivery. In addition to improved glucose control, this approach reduces the frequency and severity of hypoglycemic episodes, and increases the overall quality of life for people with T1DM.
Current commercially available AID systems adjust basal insulin delivery in real time. Some of the advanced systems also deliver correction doses. Although insulin delivery in closed loop systems aims to be fully automated in the future, currently used advanced hybrid closed loop (AHCL) systems still require manual input of carbohydrate consumption to calculate the mealtime insulin doses, and any changes in the physical activity must be manually communicated to the system [1]. Numerous studies involving a range of systems with diverse algorithms, insulin pumps, and sensors have been conducted in both adult and pediatric populations [3,4,5,6,7,8,9]. Studies have shown that AID systems may lead to a decrease in A1C levels and an improvement in time in range (TIR) [10,11,12,13]. The use of AID systems is based on the individual’s preference and the ability of the person with diabetes and/or the caregiver to safely and efficiently use the technology.
The MiniMed™ 780G system utilizes an AHCL algorithm that delivers basal insulin automatically every 5 min and provides adjustable blood glucose targets of 100, 110, or 120 mg/dL (or 5.5, 6.1, or 6.7 mmol/L) with automatic correction bolus delivery up to every 5 min, if the algorithm determines that it is needed. For optimal glycemic results, the MiniMed™ 780G system requires user-initiated meal announcements. The device improves blood glucose levels by automatically correcting for inaccuracies in carbohydrate estimation and late or missed meal boluses up to every 5 min while accommodating daily glucose variability without user intervention [14].
Data was collected from 4120 people with T1DM who used the MiniMed™ 780G system in a real-world setting, and it was demonstrated that the device could effectively control blood glucose levels while maintaining safety from hypoglycemia. This suggested that the device could provide achievable glycemic control in a practical, everyday setting [2]. A key study evaluating the safety and effectiveness of the AHCL system was carried out in adolescents and adults, which showed that an AHCL reduced A1C from 7.5% to 7.0%, TIR increased from 68.8% to 74.5%, and time below range (TBR) reduced from 3.3% to 2.3% [15]. When the MiniMed™ 780G system is set with optimal settings, which include a 100 mg/dL glucose target and active insulin time (AIT) of 2 h, users were able to achieve a TIR of 78.8 ± 5.5%. In a separate randomized controlled trial of the AHCL system conducted in children, adolescents, and adults, the proportion of users achieving a TIR greater than 70% increased from 12% at baseline to 51% when using the AHCL system [16].
Studies conducted on users of the MiniMed™ 780G system have demonstrated improved outcomes, such as a reduction in the glucose management indicator (GMI), a surrogate of HbA1c, and an increase in TIR compared to baseline or before closed loop initiation [17]. These improvements were also associated with a decrease in the time spent in hypoglycemia. Similar findings have been observed in other investigations of closed loop therapies, including various trials and pediatric studies of AID systems, where these therapies have led to improved glycemic control without an increase or with a decrease in hypoglycemia [16, 18, 19].
The aim of the study was to evaluate both the user and clinician perspectives and satisfaction with the MiniMed™ 780G system for diabetes management. In addition, the study also aimed to evaluate the real-world performance of the system for people with T1DM.
Methods
We conducted two separate peer group discussions, (1) of adults with T1DM/parents of children and adolescents with T1DM to understand the experience and uncover the reasons behind the participants’ satisfaction or dissatisfaction with the MiniMed™ 780G and (2) of healthcare providers (HCPs) to analyze their observations regarding the system.
Adults with T1DM and parents of children or adolescents with T1DM were identified by the multidisciplinary team in the study center and were approached to discuss the study during their regular outpatient visits. With consenting adults or parents/caretakers, the peer group discussion was conducted on January 1, 2023. The peer group discussion of HCPs (doctors, nurses, dietitians, and diabetes educators) was conducted on January 10, 2023. The discussion included all the HCPs involved in 780G users follow-up. The responses from the peer group discussion were analyzed to establish relationships and uncover key themes. The discussion and interpretation were categorized into various themes by two independent researchers manually. Inconsistencies were resolved by consensus.
We also analyzed the MiniMed™ 780G system data uploaded to CareLink personal software from September 15, 2022 to February 1, 2023 by individuals who participated in the peer group discussion and who provided consent for their data to be aggregated. Users with at least 10 days of sensor glucose data after AHCL were included in the analysis. This is in line with previous publications and provides a level of consistency in the analysis [20]. Glycemic outcomes including the mean percentage of TIR (70–180 mg/dL), TBR* (< 54 mg/dL), TBR (54–70 mg/dL), time above range (TAR) (180–250 mg/dL), and TAR* (> 250 mg/dL) were determined. The mean sensor glucose (SG) levels and GMI were also assessed, as well as the sensor use, and the percentage of time spent in AHCL. Descriptive analysis using mean and standard deviation for continuous variables and proportion (%) for categorical variables was done.
The study was approved by the institutional ethics committee of the center [IEC/JDC/722/2022]. All participants and/or their parents/guardians signed informed consent documents.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2005. Participants or parents/caregivers gave written consent to participate, collect, and publish the data. All the participants or parents/caregivers gave written consent to publish the verbatim responses in the study. The responses were rechecked with the participants or parents/caregivers. All the HCPs participated in the study also gave written consent to publish the verbatim responses in the study. HCPs rechecked and confirmed the responses.
Results
The peer group discussions uncovered many major themes and concerns for each group. Separate peer group discussions provided unique perspectives and experiences of each group. Six HCPs and 25 users/parents of the users shared their experiences and observations.
Healthcare Providers’ Perspective and Experience
The peer group discussion among HCPs (n = 6; one doctor, two diabetes nurses, two diabetes educators, and one dietitian) revealed the importance of pre-initiation training for successful use of the MiniMed™ 780G AHCL system. HCPs recommended setting realistic expectations for the system, attending educational sessions, and discussing glucose targets with patients to help them understand how the system works and what goals they should aim for. Carbohydrate counting could be challenging for some users, so HCPs suggested providing food charts, teaching patients how to read food labels, and encouraging them to keep a food diary. They also emphasized the importance of regular interaction with the system and understanding its capabilities and limitations.
The overall experience of the healthcare professionals was positive, with reports of improved patient outcomes such as fewer hypoglycemic events and improved quality of life. However, fake carb entries were identified as a common problem possibly due to anxiety around rapidly rising glucose levels. AHCL was seen to be effective in reducing the frequency of hypo- and hyperglycemia. Some HCPs suggested the need for a wider target range of glucose levels. Overall, the healthcare professionals reported that their patients were happier and enjoying a better quality of life (Table 1).
Overall User Experience
The users from our center (n = 25; T1DM; 17 female; age 13.8 ± 7.49 years; A1C 6.54 ± 0.45%; duration of diabetes 6 ± 6.78 years; 6 insulin pump naive) were very satisfied with the 780G AHCL system. Overall, the MiniMed™ 780G advanced hybrid closed loop insulin delivery system received positive feedback from the users in terms of achieving consistent blood glucose levels, compensating for highs and lows, and improving glucose control during the night. The users reported feeling much more relaxed and having “hypo-free” nights. Participants except a few appreciated the alerts and reminders provided by the system that helped them manage their blood glucose levels within the target range. The system was found to be easy to use once learned, and the fact that it was waterproof was considered a great feature.
However, some participants reported difficulties with adjusting for post-meal hyperglycemia and the need to enter a “fake” carb entry. One participant reported that the autocorrection bolus took a lot of time, resulting in high blood glucose levels. The size of the pump and tubing made it difficult for some children to participate in outdoor sports, and some participants felt that the system was not suitable for elderly or individuals who might have difficulty understanding the steps involved in using the system.
Some technical issues were also reported, such as connectivity issues with the sensor, cannula blockage kinking for those using insulin Fiasp, and too many alarms. Participants also mentioned that the target range was inflexible, and some reported pain and marks from the insertion of the infusion set. Nonetheless, some participants reported learning through their mistakes and becoming proficient in using the system over time (Table 2).
Overall Performance of the MiniMed™ 780G System Based on Data from CareLink
A total of 27 users uploaded data into CareLink personal software within the observation period (4 months: 54–121 days), of whom 25 provided consent for their data to be aggregated. Users achieved a mean GMI of 6.4 ± 0.26% (interquartile range [IQR] 6.2–6.7%), TIR of 83.0 ± 8.12%, TBR (54–70 mg/dL) of 2.0 ± 0.81%, TBR* (< 54 mg/dL) of 0%, and TAR (180–250 mg/dL) of 13.5 ± 6.60% and TAR* (> 250 mg/dL) of 3.0 ± 1.41%. The median (IQR) TIR achieved was 84% (80–91%) (Table 3, Fig. 1). All of the users achieved a TIR of > 70%. The glucose sensor was in use for a mean of 88.5 ± 8.18% of the time and the users were in AHCL for a mean of 96 ± 3.65% of the time.
Discussion
Usability is a critical aspect of technology, especially in the context of medical devices like the MiniMed™ 780G, which play a crucial role in the management of T1DM and insulin-dependent T2DM. Evaluating the usability of the device can help to identify any areas of concern, such as user interface design, ease of use, or device functionality, and inform improvements that can be made to enhance the overall user experience. The psychosocial impacts of hybrid closed loop systems like the MiniMed™ 780G can be significant for people with T1DM and their care providers. Positive user experiences can improve quality of life, increase confidence in managing the disease, and reduce the burden of diabetes-related stress and anxiety [21].
This user experience and clinician perspective study provides insights into the users as well as HCPs’ perceptions of the MiniMed™ 780G AHCL system. Understanding both the user and HCP perspectives on the MiniMed™ 780G is important for evaluating the overall impact and effectiveness of the device in the management of T1DM. A high level of satisfaction was generally expressed by the users as well as HCPs. AID has the potential to successfully manage T1D with no fear of hypoglycemia. Initial research on the pump revealed a general improvement in user satisfaction based on the set quality of life indicators, including reduced anxiety and worry, as well as improved physical abilities like greater mobility and decreased pain [22].
Some users appreciated the reduced requirement for manual input while in auto mode, but a larger number indicated that they felt there was actually an increased need for their involvement, including frequent alerts. Alarm fatigue occurs when users receive too many alerts or alarms from their CGM system, leading to desensitization and ignoring of alarms [23]. This can be a major concern because ignored alarms can lead to missed high or low glucose events, which can result in serious health consequences. Alarm fatigue is often seen during post-meal hyperglycemia when blood glucose levels are high after eating a meal. This is because high blood glucose levels often trigger multiple alarms, leading to repetitive alerts and alarms. Over time, users may become desensitized to the alarms and ignore them, leading to missed opportunities for corrective action. To address alarm fatigue, it is important for users to work closely with their HCPs to adjust alarm settings and create personalized alarm plans. In addition, it is important for users to educate themselves on the causes and management of post-meal hyperglycemia, including the use of mealtime insulin doses and adjustments to lifestyle factors such as physical activity and diet.
While the majority of the users believed that the system helped them to maintain better glucose control and consistent blood glucose levels, others reported that the pump caused their blood glucose to be higher than their normal levels and the insulin doses given were quite minimal.
The MiniMed™ 780G system, despite utilizing advanced algorithms for basal insulin delivery, still requires users to manually input the amount of carbohydrates consumed before meals and snacks. However, the system includes an automatic high/low correction feature that can manage unannounced snacks by delivering additional insulin as needed, based on the current glucose level, insulin on board, and other relevant factors. Accurately counting carbohydrates can be one of the biggest challenges of using the MiniMed™ 780G AHCL or any other insulin delivery system that relies on carbohydrate counting. This is because the information available to the user, such as the carbohydrate content of food, can be inaccurate, and it can be difficult to determine the exact amount of carbohydrates in a meal or snack [24].
The correction dose of insulin presents another significant challenge. The correction dose in auto mode is solely controlled by the algorithm. When there is persistent hyperglycemia, the users switch out of auto mode and take correction doses using the bolus calculators. The discussion also found that many users input a quantity of carbohydrates without actually consuming any carbohydrates (fake carb entry/ghost carb), for the correction of persistent hyperglycemia. It is not uncommon for hypoglycemia to occur 2–3 h after this fake meal entry, and in such cases, the excess insulin may still cause hypoglycemia even if the automated basal delivery is reduced or stopped. This in turn leads to an increase in glycemic fluctuations and a decrease in TIR. The findings align with the feedback from users accessible on online platforms or public forums, suggesting that they resort to using alternative methods such as falsifying carbohydrate input or manually bolusing by exiting auto mode to address their concerns [25].
Real-world evidence can provide important insights into the effectiveness and safety of new therapies in real-world settings and can help to determine whether the results from small and highly structured clinical trials can be generalized to broader populations. Randomized controlled trials (RCTs) are considered the gold standard for evaluating the efficacy of a therapy. However, RCTs are often conducted in highly controlled environments with strict inclusion and exclusion criteria, which may not reflect the characteristics of users in the real world. Real-world data (RWD) can provide a more realistic picture of the outcomes of therapeutic interventions because it reflects the experiences of users in everyday clinical practice [26]. Discrepancies between clinical studies and real-world results are expected and have been reported in the literature as up to 27% [27].
The measurement of TIR is an important indicator for evaluating glucose control and patterns, with a strong correlation to A1C in multiple studies [28,29,30]. TBR and TAR are additional parameters that are useful for making insulin dose adjustments and reevaluating treatment plans [31, 32]. The American Diabetes Association (ADA) guidelines for 2023 recommend a target of achieving more than 70% TIR, with less than 4% TBR and less than 1% TBR < 54 mg/dL for non-pregnant adults. For those at high risk of hypoglycemia, a target of greater than 50% TIR with less than 1% TBR is recommended [33]. The primary goal for individuals with T1DM and type 2 diabetes (T2DM) is to achieve and maintain at least 70% TIR, with a minimum of TBR and time above range TAR and less than 4% (or 1 h per day) of TBR (< 70 mg/dL) [34, 35].
The study showed that users had a TIR of 83 ± 8.12%%, which is higher than the recommended goal of 70%. Additionally, the values for low TBR were also within the recommended limits, with 2 ± 0.81% TBR < 70 (recommended < 4%) and 0% TBR < 54 (recommended < 1%). The TAR > 180 and TAR > 250 values were 13.5 ± 6.60% and 3.3 ± 1.41%, respectively, which are below the recommended thresholds of < 25% and < 5%. Furthermore, the mean GMI was 6.4 ± 0.26%, which is lower than the recommended goal of < 7.0%. In terms of the percentage of users who achieved the recommended goals for GMI, TIR, and time below ranges (both TBR < 70 and TBR < 54), it was found that all users (100%) in the study achieved the recommended goal for GMI < 7.0%, as well as for TIR and TBR (both TBR < 70 and TBR < 54). The results are in line with previous clinical studies [2, 36].
By closely monitoring their glucose levels and regularly calibrating their system, users can gain a more accurate understanding of how their glucose levels are changing throughout the day and make more informed insulin dosing decisions. This can help them to achieve better glucose control and reduce the risk of hypo- and hyperglycemic events. Guardian Sensor 3, which is being currently used in the 780G AHCL system, has better accuracy than previous generation of sensors. To get the most out of the MiniMed™ 780G AHCL system, it is important for users to perform regular calibrations and glucose measurements. It is generally recommended that users calibrate their system every 12 h, in addition to regularly measuring their glucose levels with a fingerstick before making any insulin dosing decisions [37].
On the basis of the results of the study, we speculate that the use of the autocorrection bolus, along with lower glycemic targets, played a crucial role in achieving improved overall glycemic control among the users. Setting lower glycemic targets may have encouraged users to be more vigilant about their blood glucose levels and take necessary actions to maintain them within the desired range. But in a developing country like India, the market is non-reimbursable and the technology remains inaccessible to 90% of the T1D population which limits its widespread use.
The study, however, had a small sample size and was conducted at a single center. Though the data gathered from group discussions were self-reported, it was validated by observing actual user behavior and report. Additionally, the pump had only been available for approximately 5 months at the time of the study, and the participants who were using it were relatively new to the system. It would be valuable to conduct a follow-up assessment after 1 year to ascertain any changes in perception with longer user experience.
Conclusion
The use of the MiniMed™ 780G system in a real-world setting among people with T1DM yielded robust data on achievable glycemic control, while also maintaining safety from hypoglycemia. The achievement of consistent TIR levels above 80% has been a challenge for insulin pumps and other delivery systems. The MiniMed™ 780G insulin pump has become the first device to successfully achieve this threshold, surpassing the performance of all other insulin pumps and delivery systems. However, proper training is essential for the successful and continued use of new medical devices like the MiniMed™ 780G. By providing training to both users and HCPs, they can become familiar with the device and its features, understand how to use it effectively, and be able to troubleshoot any issues that may arise. The perspective of both users and HCPs can be used for the development of training programs and support materials for the MiniMed™ 780G, and ensure that users and HCPs have the necessary knowledge and skills to use the device effectively.
References
ElSayed NA, Aleppo G, Aroda VR, et al. 7. Diabetes technology: standards of care in diabetes—2023. Diabetes Care. 2023;46(Supplement_1):S111–27.
Silva JD, Lepore G, Battelino T, et al. Real-world performance of the MiniMed™ 780G system: first report of outcomes from 4120 users. Diabetes Technol Ther. 2022;24(2):113–9.
Ekhlaspour L, Forlenza GP, Chernavvsky D, et al. Closed loop control in adolescents and children during winter sports: use of the Tandem Control-IQ AP system. Pediatr Diabetes. 2019;20(6):759–68.
Buckingham BA, Christiansen MP, Forlenza GP, et al. Performance of the Omnipod personalized model predictive control algorithm with meal bolus challenges in adults with type 1 diabetes. Diabetes Technol Ther. 2018;20(9):585–95.
Renard E, Tubiana-Rufi N, Bonnemaison-Gilbert E, et al. Closed-loop driven by control-to-range algorithm outperforms threshold-low-glucose-suspend insulin delivery on glucose control albeit not on nocturnal hypoglycemia in prepubertal patients with type 1 diabetes in a supervised hotel setting. Diabetes Obes Metab. 2019;21(1):183–7.
Forlenza GP, Ekhlaspour L, Breton M, et al. Successful at-home use of the tandem control-IQ artificial pancreas system in young children during a randomized controlled trial. Diabetes Technol Ther. 2019;21(4):159–69.
Anderson SM, Buckingham BA, Breton MD, et al. Hybrid closed-loop control is safe and effective for people with type 1 diabetes who are at moderate to high risk for hypoglycemia. Diabetes Technol Ther. 2019;21(6):356–63.
Karageorgiou V, Papaioannou TG, Bellos I, et al. Effectiveness of artificial pancreas in the non-adult population: a systematic review and network meta-analysis. Metabolism. 2019;1(90):20–30.
Kesavadev J, Saboo B, Kar P, Sethi J. DIY artificial pancreas: a narrative of the first patient and the physicians’ experiences from India. Diabetes Metab Syndr. 2021;15(2):615–20.
Brown SA, Kovatchev BP, Raghinaru D, et al. Six-month randomized, multicenter trial of closed-loop control in type 1 diabetes. N Engl J Med. 2019;381(18):1707–17.
Sherr JL, Buckingham BA, Forlenza GP, et al. Safety and performance of the Omnipod hybrid closed-loop system in adults, adolescents, and children with type 1 diabetes over 5 days under free-living conditions. Diabetes Technol Ther. 2020;22(3):174–84.
Kovatchev B, Anderson SM, Raghinaru D, et al. Randomized controlled trial of mobile closed-loop control. Diabetes Care. 2020;43(3):607–15.
Biester T, Nir J, Remus K, et al. DREAM5: an open-label, randomized, cross-over study to evaluate the safety and efficacy of day and night closed-loop control by comparing the MD-Logic automated insulin delivery system to sensor-augmented pump therapy in patients with type 1 diabetes at home. Diabetes Obes Metab. 2019;21(4):822–8.
Pintaudi B, Gironi I, Nicosia R, et al. Minimed Medtronic 780G optimizes glucose control in patients with type 1 diabetes mellitus. Nutr Metab Cardiovasc Dis. 2022;32(7):1719–24.
Carlson AL, Sherr JL, Shulman DI, et al. Safety and glycemic outcomes during the MiniMed™ advanced hybrid closed-loop system pivotal trial in adolescents and adults with type 1 diabetes. Diabetes Technol Ther. 2022;24(3):178–89.
Collyns OJ, Meier RA, Betts ZL, et al. Improved glycemic outcomes with Medtronic MiniMed advanced hybrid closed-loop delivery: results from a randomized crossover trial comparing automated insulin delivery with predictive low glucose suspend in people with type 1 diabetes. Diabetes Care. 2021;44(4):969–75.
Beato-Víbora PI, Gallego-Gamero F, Lázaro-Martín L, Romero-Pérez MD, Arroyo-Díez FJ. Prospective analysis of the impact of commercialized hybrid closed-loop system on glycemic control, glycemic variability, and patient-related outcomes in children and adults: a focus on superiority over predictive low-glucose suspend technology. Diabetes Technol Ther. 2020;22(12):912–9.
Bergenstal RM, Nimri R, Beck RW, et al. A comparison of two hybrid closed-loop systems in adolescents and young adults with type 1 diabetes (FLAIR): a multicentre, randomized, crossover trial. Lancet. 2021;397(10270):208–19.
Breton MD, Kovatchev BP. One year real-world use of the control-IQ advanced hybrid closed-loop technology. Diabetes Technol Ther. 2021;23(9):601–8.
Bergenstal RM, Beck RW, Close KL, et al. Glucose management indicator (GMI): a new term for estimating A1C from continuous glucose monitoring. Diabetes Care. 2018;41(11):2275–80.
Farrington C. Psychosocial impacts of hybrid closed-loop systems in the management of diabetes: a review. Diabet Med. 2018;35(4):436–49.
Matejko B, Juza A, Kieć-Wilk B, et al. Transitioning of people with type 1 diabetes from multiple daily injections and self-monitoring of blood glucose directly to MiniMed 780G advanced hybrid closed-loop system: a two-center, randomized, controlled study. Diabetes Care. 2022;45(11):2628–35.
Woo M, Bacon O. Alarm fatigue. In: Making Healthcare Safer III: a critical analysis of existing and emerging patient safety practices. Agency for Healthcare Research and Quality (US). 2020. Accessed 10 Feb 2023
Reiterer F, Freckmann G, del Re L. Impact of carbohydrate counting errors on glycemic control in type 1 diabetes. IFAC-PapersOnLine. 2018;51(27):186–91.
A year of Medtronic 780G. https://www.pumpsandpricks.com/post/a-year-of-780g Accessed 10 Feb 2023.
Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35:1763–74.
Monti S, Grosso V, Todoerti M, Caporali R. Randomized controlled trials and real-world data: differences and similarities to untangle literature data. Rheumatology. 2018;57(Supplement_7):54–8.
Advani A. Positioning time in range in diabetes management. Diabetologia. 2020;63(2):242–52.
Avari P, Uduku C, George D, Herrero P, Reddy M, Oliver N. Differences for percentage times in glycemic range between continuous glucose monitoring and capillary blood glucose monitoring in adults with type 1 diabetes: analysis of the REPLACE-BG dataset. Diabetes Technol Ther. 2020;22(3):222–7.
Livingstone R, Boyle JG, Petrie JR. How tightly controlled do fluctuations in blood glucose levels need to be to reduce the risk of developing complications in people with type 1 diabetes? Diabet Med. 2020;37(4):513–21.
Mayeda L, Katz R, Ahmad I, et al. Glucose time in range and peripheral neuropathy in type 2 diabetes mellitus and chronic kidney disease. BMJ Open Diabetes Res Care. 2020;8(1):e000991.
Lu J, Ma X, Shen Y, et al. Time in range is associated with carotid intima-media thickness in type 2 diabetes. Diabetes Technol Ther. 2020;22(2):72–8.
ElSayed NA, Aleppo G, Aroda VR, et al. 6. Glycemic targets: standards of care in diabetes—2023. Diabetes Care. 2023;46(Supplement_1):S97–110.
Dovc K, Battelino T. Time in range centered diabetes care. Clin Pediatr Endocrinol. 2021;30(1):1.
Kesavadev J, Misra A, Saboo B, et al. Time-in-range and frequency of continuous glucose monitoring: recommendations for South Asia. Diabetes Metab Syndr. 2022;16(1):102345.
Beato-Víbora PI, Gallego-Gamero F, Ambrojo-López A, Gil-Poch E, Martín-Romo I, Arroyo-Díez FJ. Rapid improvement in time in range after the implementation of an advanced hybrid closed-loop system in adolescents and adults with type 1 diabetes. Diabetes Technol Ther. 2021;23(9):609–15.
Guardian Sensor 3. https://www.medtronic.com/us-en/healthcare-professionals/products/diabetes/continuous-glucose-monitoring-systems/guardian-sensor-3.html Accessed on 13 Feb 2023.
Acknowledgements
The authors express their gratitude to the participants and their families for their cooperation during the study and acknowledge their valuable contribution. In addition, we extend our thanks to all the HCPs who contributed to the content of the article.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author Contributions
Jothydev Kesavadev, Anjana Basanth and Gopika Krishnan contributed to the study design, analysis, and interpretation of data. Jothydev Kesavadev, Anjana Basanth, Gopika Krishnan, Arun Shankar, Geethu Sanal, Sunitha Jothydev critically reviewed, drafted, and edited the article and approved the final study for submission.
Disclosures
Jothydev Kesavadev, Anjana Basanth, Gopika Krishnan, Arun Shankar, Geethu Sanal, and Sunitha Jothydev have no personal, financial, commercial, or academic conflicts of interest to declare for the study.
Compliance with Ethical Guidelines
The study was approved by the institutional ethics committee of the center [IEC/JDC/722/2022]. All participants and/or their parents/guardians signed informed consent documents. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2005. Participants or parents/caregivers gave written consent to participate, collect, and publish the data. All the participants or parents/caregivers gave written consent to publish the verbatim responses in the study. The responses were rechecked with the participants or parents/caregivers. All the HCPs participated in the study also gave written consent to publish the verbatim responses in the study. HCPs rechecked confirmed the responses.
Data Availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Kesavadev, J., Basanth, A., Krishnan, G. et al. Real-World User and Clinician Perspective and Experience with MiniMed™ 780G Advanced Hybrid Closed Loop System. Diabetes Ther 14, 1319–1330 (2023). https://doi.org/10.1007/s13300-023-01427-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01427-z