Abstract
Increasing evidence suggests that bacteria are capable of creating specific redox conditions which are visible as species-specific continuous redox potential (E h) measurements. It has been demonstrated that continuous measurements of E h are valuable for tracking bacterial metabolic activities of bacterial species in liquid cultures. However, it remains uncertain whether this phenomenon is widespread among bacterial species and whether E h measurements reflect similar mechanisms in more complex systems such as soils and sediments. The purpose of this study is therefore to evaluate whether bacteria that naturally occur in sediments have the capacity to control E h and assess the relative partitioning of biological processes involved in E h in natural sediments. To this end, continuous E h measurements are linked to growth of bacteria in liquid cultures and bacterial metabolic activity in aquatic sediment microcosms containing the bioturbator Tubifex spp., in which we evaluate bacterial partitioning in microcosms treated with the bacteriocide formalin. The tested bacterial species (Micrococcus luteus, Paracoccus pantotrophus and Aminobacter aminovarans) appeared to have specific stable E h signals during linear-exponential growth phase, suggesting that these species are capable of exerting an extracellular control on E h measurements, thereby supporting the notion that species-specific E h signals may be widespread among bacterial species. Formalin treatment reduced temporal variability of E h in sediment microcosms. This outcome suggests that bacterial metabolism and inherent relative contributions of members of bacterial community principally determine development of E h in sediment systems and that quantitation of sediment electrochemical properties may offer a potential indicator that characterizes bacterial processes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Increasing evidence suggests that bacteria are capable of creating redox conditions which are visible as species-specific continuous E h measurements (Brasca et al. 2007; Reichart et al. 2007; Michelon et al. 2010; Ignatova et al. 2010; Tachon et al. 2010). It has been demonstrated that continuous measurements of E h are valuable for tracking bacterial metabolic activities in liquid cultures (e.g. Escalante-Minakata et al. 2009; Rosu et al. 2010), in which mixed bacterial assemblages may generate a composite redox potential where fluctuations reflect changes in relative metabolic activities of the different species within these assemblages (Escalante-Minakata et al. 2009; Rosu et al. 2010). This suggests the presence of an apparent redox niche, which is supported by the early observation of redox-taxis in a number of bacterial species (e.g. Grishanin et al. 1991; Bespalov et al. 1996). This phenomenon has been demonstrated for a limited number of bacterial species, but may be more widespread than previously anticipated (Rabaey et al. 2007; Reichart et al. 2007).
In more complex systems such as soils and sediments, measurements of E h are generally linked to concentrations of terminal electron acceptors involved in bacterial respiration (e.g. oxygen, nitrate, manganese et cetera). In this, E h is formally defined by the Nernst equation and thereby carries an erroneous assumption that redox reactions in a system are reversible, complete and in equilibrium, while many reactions in nature are neither complete nor reversible. This approach does not consider the energetically flexible bacterial metabolites that serve as redox intermediates, while it has recently been demonstrated that bacterial-derived extracellular metabolites can dominate redox measurements under culture conditions (Xie et al. 2010). Numerous studies were not able to couple redox potential and concentrations of terminal electron acceptors in both soils and sediments, and uncertainties exist over the extent to which reactions participating in E h are biologically mediated (Smayda 1990; Roden et al. 2004; Naudet and Revil 2005). It thus remains uncertain whether measurements of E h in sediments accordingly reflect bacterial metabolism, suggesting that the relative contribution of bacteria in E h measurements in sediment systems requires further evaluation.
The aim of this study is therefore to evaluate whether bacteria that naturally occur in sediments have the capacity to create species-specific E h conditions and assess the relative partitioning of biological processes involved in E h in sediments. To this end, continuous E h measurements are linked to growth of bacteria in liquid cultures and bacterial metabolic activity in aquatic sediment microcosms.
Materials and methods
Redox potential development in culture media
In order to test whether organotrophic bacteria that naturally occur in aquatic sediments have species-specific effects on redox potential, the taxonomically and metabolically distinct bacterial species Micrococcus luteus (strictly aerobic), Paracoccus pantotrophus (facultative aerobic, denitifier) and Aminobacter aminovorans (strictly aerobic) (obtained from the Netherlands Culture Collection of Bacteria, NCCB, originally sampled from aquatic sediments) were cultured in triplicate in brain heart broth (MERCK) and peptonised milk nutrient (Sigma) (ratio 100:15) with continuous stirring. Bacterial growth was monitored by measuring the absorbance at 600 nm (Shimadzu 1601-UV), and continuous redox potential development was measured every minute using permanently installed redox microelectrodes and dataloggers described recently (Vorenhout et al. 2011). After 40 h, formalin was added (final concentration: 2 %) in order to disrupt bacterial activity and monitoring of E h development continued.
Redox potential in aquatic sediments
Species-specificity of E h conditions in subsurface sediment layers (1–2 mm depth) was evaluated (measured every minute) in sediment microcosms (Ø 25 mm) consisting of 17.5 g quartz. As organic matter, we used 3 g nettle powder (Urtica dioica) since this plant is easily degradable and often dominates the riparian zones of temperate aquatic systems. Treatments included inoculums of single bacterial species, in which sterile microcosms were used as negative control.
In order to test the relative portioning of bacteria-mediated control on redox potential measurements, we re-considered E h in relation to bacterial activity in microcosms as described recently (Hunting and Van der Geest 2011). In brief, organic rich sediment was collected from Lake Drontermeer, The Netherlands (52°58′N, 5°50′E) for natural bacterial communities and organic matter. The collected sediment was frozen in order to kill fauna and thawed to allow for bacterial re-growth. Natural sediments were subsequently mixed with clean river sand (ignited quartz; grain size 0.5–1.0 mm; ratio 1:5). This mixture was homogenized and oxygenated prior to experimentation. Experiments were performed in a circular polyvinyl chloride core which allows for continuous monitoring of sediment without disturbance. The core (110 mm diameter, 250 mm height) has 7 gastight connectors for bacterial probes alongside the redox electrodes containing a Nuclepore polycarbonate membrane. The membrane acts as semi-permeable membrane (mesh size 5 μm) to sample porewater containing bacterial cells while retaining particles and floccules. Redox potential (E h) was measured at various depths (0, 10, 20, 30, 40, 90 mm; every 15 min.) using permanently installed platinum electrodes and a calomel reference electrode (Vorenhout et al. 2004). Measurements of parameters in cores commenced immediately after homogenization, and continued toward the end of the experimental runs at fixed time-intervals over a period of 4–5 days. Sediments were allowed to reach steady state conditions (24–48 h) and were subsequently irrigated and reworked by the introduction of the aquatic oligochaete Tubifex spp. (approx. 500 individuals/m2). Bacterial activity was determined by sampling pore water (400 μL) alongside the platinum electrodes on a daily basis at fixed times and immediately assayed for bacterial activity as described below. The relative participation of bacterial activity on E h development was assessed in bulk sediment mixed with dilute formaldehyde (final concentration 0.46 ± 0.1 %) in order to stop bacterial activity, while preventing contribution of the formaldehyde/methanol redox couple. Sediment was spread over 0.5 m2 trays to minimize depth (1–2 mm) for 48 h in a flow cabinet prior to experimentation to allow evaporation of uncomplexed formalin. Dilute formaldehyde was used as this appears the most suitable treatment to inhibit long term bacterial activity with minimal increase of dissolved nutrients (Tuominen et al. 1994) and preliminary runs revealed that Tubifex spp. could survive formalin treated conditions for at least 48 h.
Analytical techniques
Dissolved oxygen concentrations were monitored using an oxygen micro-sensor (Unisense OX25) connected to a picoammeter (Unisense, PA2000). Bacterial activity was analyzed by dehydrogenase enzyme activity via the reduction of 2-(p-iodophenyl)-3-(p-nitrophenyl)-5-phenyl tetrazolium chloride (INT) to formazan (INTF), since INT reduction proved an accurate assay of electron transfer system activities (ETSA) under both anaerobic and aerobic conditions (Smith and Mcfeters 1997; Hunting et al. 2010). Bacterial cells were simultaneously sampled alongside the platinum electrodes (400 μL porewater sample). The samples were vortexed and incubated at 20 °C for 30 min. Bacterial cell integrity was subsequently disrupted by 5 min sonication at room temperature (Branson, 1510). An aqueous INT solution (200 μL; 6.2 × 10−2 M) was added and the samples were mixed and incubated for 1 h at 20 °C under dark room conditions. Enzymatic activity was stopped by adding 500 μL of acetone and measured spectrophotometrically at 490 nm (Shimadzu, 1601-UV). An INT formazan standard was used for calibration, in which two protons equal the transfer of a single electron. Controls treated with formalin were included to correct for abiotic reduction of INT.
Results and discussion
Monospecific cultures and microcosms
Continuous redox potential (E h) development of the individual bacterial species and their corresponding growth are presented in Fig. 1a–d. E h becomes stable during continuous and exponential growth phase, but becomes variable at the onset of stationary bacterial growth (Fig 1a–d, left panel).
Development of redox potential E h during growth of the organotrophic bacteria a M. luteus, b P. panthothrophus, c A. aminovorans in culture conditions. Development of E h can be compared to bacterial growth (d). One of the replicates is shown for clarity. It is evident that E h measurements become stable during continuous and exponential growth of bacteria, and becomes variable during stationary growth. Dagger symbol indicates addition of formalin resulting in stabilization of E h measurements, thereby reflecting relative partitioning of bacteria-mediated processes in measurements of E h
Redox values of individual species during exponential growth and the corresponding standard deviation of the replicates are shown in Table 1. The redox measurements were similar in both liquid cultures and mono-specific sediment microcosms, although sediment microcosm appeared more variable among replicates compared to liquid cultures. However, the reported variations correspond well with species-specific E h variations as described previously (Brasca et al. 2007). Subsequent addition of formalin in order to stop bacterial metabolism at the progressive stage of bacterial growth reduced variability in E h measurements (Fig 1a–c, right panel). This outcome demonstrates the capacity of the tested bacteria to mediate E h measurements.
Sediment microcosms with natural bacterial communities
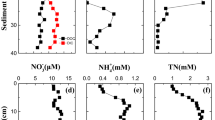
Bacterial activity was generally most pronounced at 1 cm depth and often decreased with increasing depth in deeper layers of the sediment, yet occasionally peaked at a depth of 4 or 9 cm. Incubation of sediment with dilute formaldehyde decreased the bacterial activity to below the limits of detection (Fig. 2b). At the end of the experiment, bacterial activity was detected at the surface layers of the sediment microcosms (Fig. 2b), coinciding with reducing activity at the surface layer (Fig. 4b).
Oxygen generally penetrated to 10 mm depth with highest concentrations at the sediment surface (237 lM/L 235 -1), although rapidly decreasing after a few millimeters (Fig. 3a). Addition of Tubifex spp. at 48 h resulted in a delayed increased penetration of dissolved oxygen (Fig. 3a). Treatments with dilute formaldehyde did not differ, as oxygen penetration depth also increased after the addition of Tubifex spp. (Fig. 3b).
Redox potential (E h) ranged from −470 to 340 mV in depth and showed some variation, especially after the introduction of Tubifex spp. (Fig. 4a). E h in sterilized sediments ranged from −80 to 10 and showed minimal variation over depth which was constant over time (Fig. 4b). At the end of the experimental period, reducing activity was visible at the surface layers (Fig. 4b). Overall, these results suggest a dominant partitioning of bacterial activity in the dynamics of E h (cf. Figs. 2 and 4), and that measurements of E h did not co-evolve with oxygen measurements (cf. Figs. 3 and 4).
Effect of sterilization on measurements of redox potential (E h). Continuous E h measurements in a untreated sediments and b sediments treated with dilute (0.48 %) formalin. Arrows indicate addition of Tubifex spp. Absence of fluctuation in b reflects bacterial partitioning in E h fluctuations in a. One replicate is shown for clarity
Implications
This study considered high resolution temporal redox potential measurements in relation to bacterial growth and redox activity in liquid culture conditions and sediment systems. In liquid cultures, redox potential measurements slowly developed during the initial stages (lag phase) of bacterial growth and became stable during the continuous and exponential growth phase of the tested organotrophic bacterial species, suggesting that bacteria extracellularly mediated E h measurements during continuous growth and utilization of the available substrate. This corroborates redox development described for other bacterial species (e.g. Brasca et al. 2007; Reichart et al. 2007; Tachon et al. 2010). At the onset of the stationary growth phase, redox potential measurements became more variable. The stationary growth phase is typically characterized by a balance between bacterial lysis and synthesis (Roszak and Colwell 1987), and therefore fluctuations in E h likely reflect changes in culture media caused by the release of redox active compounds during bacterial lysis. This becomes evident when the bacteriocide formalin is added to the cultures, as this disrupted bacterial metabolism, synthesis and lysis, resulting in stable signals of E h for a significant period. During this period, E h differed between the different bacterial species. A mechanistic understanding of this observation remains uncertain and is subject for future research. Although we only used a limited number of (facultative) aerobic bacteria, these findings seem to suggest that bacteria that naturally occur in aquatic environment actively control redox conditions. This supports the notion that this phenomenon may be more widespread than previously anticipated (Reichart et al. 2007).
Electrical properties of bacterial communities are often attributed to extracellular electron shuttling by bacteria (e.g. Rabaey et al. 2007), and we suspect that a similar mechanism is responsible for the observed redox conditions in our study. A variety of mechanisms have been identified for extracellular electron shuttling and production of extracellular redox mediators in microbial communities. These include membrane bound pili, cytochromes and electron transport systems, and also the secretion of electron transport system related redox mediators such as cytochrome b/c, flavins and quinones in the extracellular matrix that either remain soluble or bind to electrodes (e.g. Rabaey et al. 2007; Lovley 2008; Marsili et al. 2008; Tachon et al. 2010; Juang et al. 2011). These compounds are produced in order to dispose respiratory electrons in the absence of terminal electron acceptors in the immediate environment, allowing electron shuttling to distant terminal electron acceptors (Rabaey et al. 2007; Xie et al. 2010). Since a number of redox mediators exist, it is difficult to predict which mechanisms would be potentially involved in our test systems, and this remains a central area for future research.
Bacteria were capable of exerting similar effects on redox potential in sediment microcosms as compared to liquid cultures. This suggests that bacteria are capable of creating apparent redox niches, irrespective of the medium. This result corroborates earlier findings of species-specific redox conditions and redox niche preferences as observed by the redox-tactic behavior of motile bacteria (e.g. Grishanin et al. 1991; Bespalov et al. 1996), but more importantly highlights the importance of redox niche construction. These phenomena are currently overlooked within microbial ecological theory and experimental design of studies manipulating bacterial assemblages.
In our natural sediment microcosms, bacterial activity and E h evolved in a parallel fashion. The addition of the sediment reworking oligocheate Tubifex spp. resulted in an increased oxygen penetration depth, increased redox potentials and bacterial activities in deeper layers of the sediment, as previously reported by (Davis 1974). Reduction of bacterial activity with dilute formalin washed out temporal fluctuation in E h, including the momentum of sediment reworking by Tubifex spp., while oxygen penetration depth increased after the addition of Tubifex spp. This suggests that fluctuations in E h measurements in untreated cores were principally mediated by shifts in the relative metabolic activity of members of the bacterial community. It has been demonstrated that microbial processes play a significant role in the electrolytic and interfacial geoelectrical properties of sediments (e.g. Atekwana et al. 2004). Previous reports on the contribution of bacterial activity on E h measurements, however, remain contradictive, as changes in sediment E h have been previously been attributed to both biotic and abiotic influences (e.g. Smayda 1990; Roden et al. 2004; Naudet and Revil 2005). Measurements of E h depend on concentrations of redox couples, but the contribution of redox couples to E h highly depends on electrode affinity, exchange density and kinetics at the surface of electrodes, in which E h measurements may be dominated by a single redox couple (Peiffer et al. 1992). Although oxygen is often considered to be a dominant redox active compound in the interpretation of E h, an increased penetration depth of oxygen did not result in shifts in E h. This corroborates the outcome of other studies that evaluated E h in relation to removal of oxygen, suggesting that the reduction of oxidizing compounds other than oxygen can be important (Grenthe et al. 1992; Tachon et al. 2010). Taken together, measurements of E h in complex natural systems seem to reflect the metabolic activity of resident bacterial communities rather than concentrations of terminal electron acceptors in our microcosms. This complements previous studies that linked E h to bacterial activity and community structure (Bertics and Ziebis 2009; Hunting and Van der Geest 2011; Hunting et al. 2012). Although it cannot be ruled out that E h measurements may still be overruled by redox couples unrelated to bacterial activity, quantitation of the electrochemical properties of the sediment may offer a potential indicator of bacterial processes in sediment systems.
This study aimed to test whether bacteria that naturally occur in aquatic sediments are capable of mediating redox potential, E h, and evaluate the relative contribution of bacteria-mediated activity on measurements of E h in natural sediments. Our results indicate that bacteria mediate redox conditions in both liquid cultures and sediment microcosms, and that fluctuations in E h can be minimized upon sterilization. This outcome illustrates the principal contribution of bacterial metabolism to redox potential measurements in more complex systems such as soils and sediments.
References
Atekwana EA, Werkema DD, Allen JP, Smart LA, Duris JW, Cassidy DP, Sauck WA, Rossbach S (2004) Evidence for microbial enhanced electrical conductivity in hydrocarbon-contaminated sediments. Geophys Res Lett 31:L23501
Bertics VJ, Ziebis W (2009) Biodiversity of benthic microbial communities in bioturbated coastal sediments is controlled by geochemical microniches. ISME J 3:1269–1285
Bespalov VA, Zhulin IB, Taylor BL (1996) Behavioral responses of Escherichia coli to changes in redox potential. Proc Natl Acad Sci USA 93:10084–10089
Brasca M, Morandi S, Lodi R, Tamburini A (2007) Redox potential to discriminate among species of lactic acid bacteria. J Appl Microbiol 103:1516–1524
Davis RB (1974) Tubificids alter profiles of redox potential and pH in profundal lake sediment. Limnol Oceanogr 19(2):342–346
Escalante-Minakata P, Ibarra-Junquera V, Rosu HC, De León-Rodriquez A, González-García R (2009) Online monitoring of Mezcal fermentation based on redox potential measurements. Bioprocess Biosyst Eng 32:47–52
Grenthe I, Stumm W, Laaksuharju M, Nilsson AC, Wikberg P (1992) Redox potentials and redox reactions in deep groundwater systems. Chem Geol 98:131–150
Grishanin RN, Chalmina II, Zhulin IB (1991) Behaviour of Azospirillum brasilense in a spatial gradient of oxygen and in a ‘redox’ gradient of an artificial electron acceptor. J Gen Microbiol 137:2781–2785
Hunting ER, van der Geest HG (2011) Predictability of bacterial activity and denitrification in aquatic sediments with continuous measurements of redox potential (E h). Int J Environ Sci Technol 8(3):553–560
Hunting ER, de Goeij JM, Asselman M, van Soest RWM, van der Geest HG (2010) Degradation of mangrove-derived organic matter in mangrove associated sponges. Bull Mar Sci 86:871–877
Hunting ER, Whatley MH, van der Geest HG, Mulder C, Kraak MHS, Breure AM, Admiraal W (2012) Invertebrate footprints on detritus processing, bacterial community structure and spatio-temporal redox profiles. Freshw Sci 31(3):724–732
Ignatova M, Prévost H, Leguerinel I, Guillou S (2010) Growth and reducing capacity of Listeria monocytogenes under different initial redox potential. J Appl Microbiol 108:256–265
Juang DF, Yang PC, Lee CH, Hsueh SC, Kuo TH (2011) Electrogenic capabilities of gram negative and gram positive bacteria in microbial fuel cell combined with biological wastewater treatment. Int J Environ Sci Technol 8(4):781–792
Lovley DR (2008) The microbe electric: conversion of organic matter to electricity. Curr Opin Biotechnol 19:564–571
Marsili E, Baron DB, Shikhare I, Coursolle D, Gralnick JA, Bond DR (2008) Shewanella secretes flavins that mediate extracellular electron transfer. Proc Natl Acad Sci USA 105:3968–3973
Michelon D, Abraham S, Ebel B, De Conunck J, Husson F, Feron F, Gervais P, Cachon R (2010) Contribution of exofacial thiol groups in the reducing activity of Lactococcus lactis. FEBS J 227:2282–2290
Naudet V, Revil A (2005) A sandbox experiment to investigate bacteria-mediated redox processes on self-potential signals. Geophys Res Lett 32:L11405
Peiffer S, Klemm O, Pecher K, Hollerung R (1992) Redox measurements in aqueous solutions—a theoretical approach to data interpretation, based on electrode kinetics. J Contam Hydrol 10:1–18
Rabaey K, Rodrígeuz J, Blackall LJ, Keller J, Gross P, Batstone D, Verstraete W, Nealson KH (2007) Microbial ecology meets electrochemistry: electricity-driven and driving communities. ISME J 1:9–18
Reichart O, Szakmár K, Jozwiak Á, Felföldi J, Baranyai L (2007) Redox potential measurements as a rapid method for microbial testing and its validation for coliform determination. Int J Food Microbiol 114:143–148
Roden EE, Sobolev D, Glazer B, Luther GW III (2004) Potential for microscale bacterial Fe redox cycling at the aerobic–anaerobic interface. Geomicrobiol J 21:379–391
Rosu HC, Murguía JS, Ibarra-Junquera V (2010) Detection of mixed growth in the total biomass data by wavelet transforms. J Appl Res Technol 8(2):240–248
Roszak DB, Colwell RR (1987) Survival strategies of bacteria in the natural environment. Microbiol Rev 51(3):365–379
Smayda T (1990) The influence of lime and biological activity on sediment pH, redox and phosphorous dynamics. Hydrobiologia 192:191–203
Smith JJ, McFeters GA (1997) Mechanisms of INT (of 2-(4-iodophenyl)-3-(4-nitrophenyl)-5-phenyl tetrazolium chloride), and CTC (5-cyano-2,3-ditolyl tetrazolium chloride) reduction in Escherichia coli K-12. J Microbiol Methods 29:161–175
Tachon S, Brandsma JB, Yvon M (2010) NoxE NADH oxidase and the electron transport chain are responsible for the ability of Lactococcus lactis to decrease the redox potential of milk. Appl Environ Microbiol 76(5):1311–1319
Tuominen L, Kairesalo T, Hartikainen H (1994) Comparison of methods for inhibiting bacterial activity in sediment. Appl Environ Microbiol 60(9):3454–3457
Vorenhout M, Van der Geest HG, Van Marum D, Wattel K, Eijsackers HJP (2004) Automated and continuous redox potential measurements in soil. J Environ Qual 33:1562–1567
Vorenhout M, van der Geest HG, Hunting ER (2011) An improved datalogger and novel probes for continuous redox measurements in wetlands. Int J Environ Anal Chem 91:801–810
Xie X-H, Li EL, Tang ZK (2010) Sudden emergence of redox active Escherichia coli phenotype: cyclic voltammetric evidence of the overlapping pathways. Int J Electrochem Sci 5:1070–1081
Acknowledgments
The authors express their gratitude to the anonymous referees for their constructive comments on earlier drafts of this article.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Hunting, E.R., Kampfraath, A.A. Contribution of bacteria to redox potential (E h) measurements in sediments. Int. J. Environ. Sci. Technol. 10, 55–62 (2013). https://doi.org/10.1007/s13762-012-0080-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-012-0080-4