Abstract
Introduction
This post hoc subset analysis of RESPOND evaluated the effectiveness of dimethyl fumarate (DMF) 240 mg twice daily in patients with relapsing multiple sclerosis (RMS) after suboptimal response to glatiramer acetate (GA; “first switch” patients), including patients with early MS (“early MS switch” patients).
Methods
Patients had discontinued GA due to suboptimal response and initiated DMF treatment within 60 days after enrollment. Relapse data were collected from medical records. First switch patients had had one prior approved MS therapy (GA) before initiating DMF treatment. Early MS switch patients were first switch patients with baseline Patient-Reported Expanded Disability Status Scale (PR-EDSS) score ≤ 3.5, ≤ 1 relapses in the past 1 year, or both.
Results
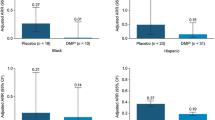
Among first switch patients (n = 231), the annualized relapse rate (ARR) was 0.48 (95% confidence interval [CI] 0.40–0.58) for 12 months before DMF initiation and 0.11 (95% CI 0.06–0.18) for 12 months after DMF initiation, a 78% decrease in ARR. Among early MS switch patients with baseline PR-EDSS score ≤ 3.5 (n = 120), ≤ 1 relapses in the prior year (n = 219), or both (n = 114), the ARRs (95% CIs) for 12 months before DMF initiation were 0.47 (0.37–0.59), 0.37 (0.32–0.44), and 0.39 (0.31–0.49), respectively; values for 12 months after DMF initiation were 0.06 (0.02–0.19), 0.09 (0.05–0.17), and 0.06 (0.02–0.20), respectively, an 87, 75, and 83% decrease in ARR. The proportion of patients relapse-free 12 months after DMF initiation versus 12 months before were 94 versus 59% in first switch patients, and 97 versus 58%, 94 versus 63%, and 97 versus 61% in early MS switch patients in the PR-EDSS score ≤ 3.5, ≤ 1 relapses in the prior year, or PR-EDSS score ≤ 3.5 and ≤ 1 relapses subgroups, respectively. After 12 months of DMF treatment, most patient-reported outcomes scores showed significant improvement.
Conclusions
DMF may be an effective treatment option in first switch and early MS switch patients with RMS who experience a suboptimal response to GA.
Trial Registration
ClinicalTrials.gov identifier: NCT01903291.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Dimethyl fumarate (DMF) has demonstrated sustained, significant efficacy in patients with relapsing–remitting multiple sclerosis (MS) in the pivotal Phase 3 trials DEFINE and CONFIRM, and in the ENDORSE long-term extension study. |
DMF may be effective in patients with early relapsing–remitting MS and suboptimal response to glatiramer acetate (GA); exploratory analyses suggest that early treatment with an appropriate therapy may lead to better outcomes. |
What did the study ask? |
This study evaluated the effectiveness of dimethyl fumarate (DMF) in patients whose only prior MS therapy was GA (“first switch” patients), including patients with early MS (“early MS switch” patients). |
What was learned from the study? |
In first switch/early MS patients, the annualized relapse rate was significantly lower 12 months after DMF initiation versus 12 months prior DMF initiation; after 12 months of DMF treatment, 94% of patients were relapse-free versus 59% before DMF initiation. |
Most patient-reported outcomes demonstrated statistically significant improvements after 12 months of DMF treatment. |
Digital Features
This article is published with digital features to facilitate understanding of the article. You can access the digital features on the article’s associated Figshare page. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13153934
Introduction
Multiple sclerosis (MS) is an immune-mediated inflammatory demyelinating disease of the central nervous system and a leading cause of neurological disability in young adults [1], occurring at least twice as commonly in women as in men [2].
Dimethyl fumarate (DMF; also known as gastro-resistant DMF) is an oral fumarate that has demonstrated sustained, significant efficacy in patients with relapsing–remitting MS in the DEFINE (ClinicalTrials.gov identifier: NCT00420212) and CONFIRM (NCT00451451) clinical trials and in the ENDORSE extension trial (NCT00835770) [3,4,5,6,7]. In exploratory analyses, clinical and magnetic resonance imaging outcomes in patients with early MS, including newly diagnosed patients and patients switching from injectable disease-modifying therapies, were consistent with those in the overall study populations [8, 9], suggesting that early treatment with an appropriate therapy may lead to better clinical outcomes. As of 30 June 2020, over 475,000 patients have been treated with DMF worldwide, representing more than 950,000 patient-years of exposure. Of these, 6335 patients (14,241 patient-years) were treated in clinical trials.
Real-world clinical evidence on efficacy, safety, and biomarkers can be informative for patient care decisions. Utilizing propensity score methods, a real-world study of 2236 patients from the Danish Multiple Sclerosis Registry found that DMF had a lower annualized relapse rate (ARR), higher relapse-free survival, and lower discontinuation due to disease breakthrough compared with teriflunomide [10]. A comparative effectiveness study from the German NeuroTransData Registry found that DMF had greater effectiveness compared with teriflunomide, interferons, and glatiramer acetate (GA) as well as similar effectiveness compared with fingolimod (FTY), results consistent with an aggregate of real-world studies [11]. In a US claims-based, propensity score–matched study of 3906 patients with MS switching from injectable to oral therapies, DMF had significantly greater reductions in ARR compared with teriflunomide and similar effectiveness to FTY [12]. Other studies have observed greater efficacy with FTY versus DMF in some outcome measures [13, 14]. Interestingly, a recent systematic literature review and network meta-analysis of real-world studies published since 2010 found DMF to be significantly more effective than GA, interferon betas, and teriflunomide, and comparable to FTY in reducing ARR and delaying time to first relapse [15].
Recently, biomarkers have shown correlation with disease severity and prognosis. For example, a prospective clinical trial in 52 treatment-naïve patients with relapsing MS (RMS) found that neurofilament light, a biomarker that has shown correlation with disease severity and prognosis, was decreased by 73% in cerebrospinal fluid, 69% in serum, and 55% in plasma after 1 year of DMF therapy [16]. In a prospective study, protective humoral immunity has been shown to be maintained and immunoglobulin levels to remain stable over 2 years of DMF treatment [17].
The RESPOND (ClinicalTrials.gov identifier: NCT01903291) study was conducted to evaluate DMF in patients with RMS after suboptimal response to GA in a real-world clinical setting. In the overall study population, DMF was associated with a 78% decrease in ARR 12 months after versus the 12 months before the initiation of DMF, and with stable or improved patient-reported outcomes (PROs) from baseline to 12 months after initiation [18]. The objective of this manuscript is to report on a post hoc analysis of RESPOND that evaluated the effectiveness of DMF in patients whose only prior MS therapy was GA (“first switch” patients), including patients with early active MS (“early MS switch” patients [patients early in MS disease course based on disability and relapse activity, who switch to DMF]).
Methods
RESPOND was a phase IV, prospective, multicenter, open-label, single-arm, 12-month observational study in patients with RMS in the USA who were administered oral DMF 240 mg twice daily between August 2013 and February 2016.
Patients eligible for inclusion in this study were ≥ 18 years of age and experiencing a suboptimal response to GA or had discontinued treatment with GA for RMS following a suboptimal response within 30 days of enrollment. A suboptimal response was defined as perceived suboptimal efficacy, intolerance, or poor adherence to GA, as determined by the prescribing physician. The decision to initiate treatment with DMF was made before study enrollment. DMF was acquired through patients’ insurance and not provided by the study sponsor. DMF treatment was initiated within 60 days of enrollment and administered per the US Prescribing Information [19] unless otherwise directed by the prescriber. Patients were considered not eligible if they presented with major comorbid conditions as determined by the prescribing physician, had a history of malignancy and/or current serious infections, were enrolled in other studies, or had received prior treatment with DMF.
As described in Fig. 1, endpoints were evaluated in one subset consisting of the first switch patients, and in three subsets of this subset of patients: (1) low disability (early MS switch patients, as assessed by the Patient-Reported Expanded Disability Status Scale [PR-EDSS]); (2) low relapse; and (3) both low disability and low relapse. The primary endpoint was ARR at 12 months. Relapse data were collected from medical records, including hospitalizations due to MS relapses as well as MS relapses associated with intravenous or oral corticosteroid use over the 12-month study duration, with the aim to assess clinical effectiveness over the 12 months before and after initiation of DMF. Relapses were defined as new or recurrent neurologic symptoms not associated with fever, lasting ≥ 24 h. New or recurrent neurologic symptoms that evolved gradually over months were considered disease progression, not an acute relapse. New or recurrent neurologic symptoms that occurred < 30 days following the onset of a relapse as defined above were considered part of the same relapse.
Patient subsets in the RESPOND study. GA Glatiramer acetate, ITT intent-to-treat, MS multiple sclerosis, PR-EDSS Patient-Reported Expanded Disability Status Scale. aRatzker et al. [37] noted that the average physician patient discrepancy in EDSS assessment is not uniform for EDSS score ≤ 3.5 vs. > 3.5; hence, we used this threshold to dichotomize the PR-EDSS
Secondary endpoints included the change from baseline to 12 months in PROs and health economic–related outcomes after treatment initiation. PROs were recorded before and at 6 and 12 months after initiation of DMF. PROs included: (1) the 36-item Short Form Health Survey (SF-36), a general health survey with eight domains: vitality, physical functioning, bodily pain, general health, physical role functioning, emotional role functioning, social role functioning, and mental health; (2) the 5-item Modified Fatigue Impact Scale (MFIS-5), which assesses the effects of fatigue on physical, cognitive, and psychosocial functioning; (3) the 14-item Treatment Satisfaction Questionnaire for Medication (TSQM-14), which measures patient satisfaction with current medication [20]; (4) the Work Productivity and Activity Impairment Questionnaire: Multiple Sclerosis (WPAI-MS), which measures impairment in work and overall activity due to MS [21]; (5) the 7-item Beck Depression Inventory (BDI-7), which assesses depressive symptoms; and (6) the PR-EDSS, which quantifies disabilities in patients with MS.
Statistical Analysis
Unadjusted ARR (total number of relapses divided by patient-years in the study, excluding data obtained after patients switched to alternative medications for MS) at 12 months before and after initiation of DMF was calculated as the total number of relapses over the time a patient was known to be treated, divided by the total number of patient-years of follow-up. The 95% confidence interval (CI) of unadjusted ARRs and ARR rate ratio was based on an empirical (robust) standard error from a generalized estimating equation using an unadjusted Poisson regression model. The proportion of patients relapse-free at 12 months was estimated using the Kaplan–Meier method, based on the time-to-first-relapse survival distribution. For the analysis of PRO data, missing data were not included. Mean changes in PROs from baseline to 12 months were assessed using a Wilcoxon signed-rank test.
The study was conducted in accordance with relevant US federal regulations, the Declaration of Helsinki, and the International Council on Harmonisation Guideline for Good Clinical Practice. Approvals were granted by relevant institutional ethics committees (See full list of Institutional review board approvals as Electronic Supplementary Material) for study protocol and amendments, and written assent and consent forms were obtained from each patient and his or her parent or legal guardian.
Results
Patients
A total of 318 enrolled, eligible patients received at least one dose of DMF; among them were 231 first switch patients (120 with low disability, 219 with low relapse, 114 with both low disability and low relapse) (Fig. 1). Patient demographics and MS disease characteristics at baseline are described in Table 1. The mean (standard deviation [SD]) age of patients enrolled in RESPOND was 47.6 (10.9) years and in the first switch subset was 47.4 (11.2 years), which was older compared with patients participating in the DEFINE and CONFIRM trials (integrated analysis; mean [SD] age: 37.9 [9.2] years) [7]. The mean (SD) time on most recent GA treatment before switching to DMF was 51.3 (49.1) months. Patients in the early MS switch, low disability cohorts had an average disease duration between 5.7 and 7.6 years.
Of the 318 patients in the RESPOND intent-to-treat population (n = 318), 71 patients (22.3%) discontinued treatment. Reasons for discontinuation included adverse event (18.6%, n = 59/318), lost to follow-up (0.3%, n = 1/318), efficacy reasons (1.9%, n = 6/318), death (0.3%, n = 1/318), and other (1.3%, n = 4/318). Discontinuation rates were consistent across the early MS subsets (first switch 22.9%, n = 53/231; low disability 22.5%, n = 27/120; low relapse 22.4%, n = 49/219; low disability and relapse 21.9%, n = 25/114).
Annualized Relapse Rate
Among first switch patients, the estimated unadjusted ARR was 0.48 (95% CI 0.40–0.58) for the 12 months before initiation of DMF and 0.11 (95% CI 0.06–0.18) for the 12 months after initiation of DMF, which represents a 78% decrease in the ARR.
Among early MS switch patients with baseline PR-EDSS score ≤ 3.5, ≤ 1 relapses in the prior year, or both, the unadjusted ARRs for the 12 months before initiation of DMF were 0.47 (95% CI 0.37–0.59), 0.37 (0.32–0.44), and 0.39 (0.31–0.49), respectively; the ARRs estimated for the 12 months after initiation of DMF were 0.06 (95% CI 0.02–0.19), 0.09 (0.05–0.17), and 0.06 (0.02–0.20), respectively, which represents an 87, 75, and 83% decrease in the ARR (Fig. 2).
Proportion of Patients Without Relapse
Estimates of the proportion of patients who were relapse-free 12 months after initiation of DMF versus 12 months before were 94 versus 59% in first switch patients and 97 versus 58%, 94 versus 63%, and 97 versus 61% in early MS switch patients in the low disability, low relapse, and low disability and relapse subsets, respectively. Kaplan–Meier estimates of the proportion of patients who were relapse-free 12 months after initiation of DMF were 92.5% for first switch patients and 96.1, 93.2, and 95.9% for early MS switch patients in the low disability, low relapse, or low disability and relapse subsets, respectively.
Patient-Reported Outcomes
Statistically significant improvements in mean (SD) scores from baseline to 12 months were observed in first switch patients and in early MS switch patients in the low relapse subset for the SF-36 physical component score (P = 0.0197 and P = 0.0186) and mental component score (P = 0.0010 and P = 0.0025), the MFIS-5 (P = 0.0003 and P = 0.0030), and the BDI-7 (both P = 0.0043) (Table 2). In the early MS switch low disability subset and low disability and relapse subset, statistically significant improvements from baseline to 12 months were observed for the SF-36 mental component score (P = 0.0140 and P = 0.0166, respectively) (Fig. 3). All patient subsets had significantly improved TSQM-14 treatment satisfaction scores (P < 0.0001) (Fig. 4).
Discussion
Here, we report a post hoc analysis of RESPOND, which was a prospective analysis of real-world clinical data on the impact of DMF on clinical measures and PROs in patients whose only prior MS therapy was GA (first switch patients), including patients with early active MS (early MS switch patients) experiencing a suboptimal response to GA. Of note, RESPOND was not designed to compare the clinical efficacy of DMF versus GA.
Among the subset of patients analyzed in this study, the ARR after 12 months of DMF treatment was statistically significantly lower than the ARR reported for the 12 months before the initiation of DMF. At 12 months after DMF treatment, 94% of patients were relapse-free compared with 59% before the initiation of DMF. The majority of PRO score improvements from baseline to 12 months after initiation of DMF treatment also reached statistical significance; no worsening was seen with any PRO after the switch to DMF. These results are consistent with the previous findings in the overall RESPOND study population and with those of the integrated phase III clinical trials DEFINE and CONFIRM [3, 4, 8]. As noted previously, the average age of patients in the overall RESPOND population and other US phase IV trials was 10 years older than patients participating in the DEFINE/CONFIRM pivotal trials [22,23,24]. It is possible that the lower ARR observed in RESPOND may be in part owing to the inclusion of younger patients who have more inflammatory activity and more relapses [25], or that the switch to DMF had a better treatment effect on ARR because of increased convenience and overall satisfaction than the daily or thrice-weekly injections required of GA, as reflected by the TSQM-14 data.
PROs involving quality-of-life measures, such as physical functioning, mental functioning, fatigue, and depression, were all perceived by patients on DMF as improved compared with their previous injectable GA treatment, demonstrating that the switch to oral DMF enhanced the mental and physical well-being of patients. There is growing evidence to support the use of PROs in both clinical practice and clinical studies to determine the effectiveness of treatment and to enhance the patient–clinician relationship [26]. Patient involvement in treatment management decisions may enhance the patient’s participatory role and also increase the likelihood that physicians’ treatment plans are aligned with patient-perceived needs [27].
Although GA is an effective treatment for MS that reduces the rate of relapses and new brain lesions observed using magnetic resonance imaging, some patients do not respond to this injectable platform therapy because of a need for greater effectiveness and/or convenience [28]. In a pivotal trial, DMF treatment resulted in a larger numerical reduction in relapse rates compared with GA (as an active open-label comparator), although this study was not powered to show a statistical difference between the two agents [4].
Because RMS is a chronic disease, the overall aims of treatment are to modify the course of the disease, preventing or delaying disability, while managing symptoms to improve quality of life [29]. Data in newly diagnosed and early MS patients support the premise that treatment with an anti-inflammatory disease-modifying therapy individualized according to disease- and patient-specific preferences should start as early as possible. Brain atrophy as a result of the MS disease process is progressive and accelerates over time [30] and, therefore, early intervention to prevent and/or reduce the risk of further irreversible brain damage is crucial [31]. Switching to oral DMF from injectable GA is a reasonable transition strategy that may work for many patients with RMS, particularly if it improves adherence and is of potentially superior efficacy [18]. There is always a need to proactively and consistently inquire about the adherence-jeopardizing side effects of disease-modifying therapies, and to be prepared to switch to alternative therapy to prevent disease breakthrough.
Data from the ongoing, multinational, prospective ESTEEM study evaluating the long-term safety and effectiveness of DMF in clinical practice support DMF as an effective treatment option for patients with RMS, including those who are newly diagnosed, early in their disease course, or switching from interferon or GA [32]. In an interim analysis, patients treated with DMF (n = 2025 in the overall interim analysis population) had significantly lower ARRs at 12 months after initiation of DMF compared with 12 months prior to treatment initiation (78% decrease in ARR; P < 0.0001); in addition, the clinical benefit observed in newly diagnosed, early MS, and interferon/GA switch patients was shown to be consistent with the findings in the overall ESTEEM population.
Our real-world results demonstrate discontinuation rates lower than those previously reported in comparative efficacy studies by Vollmer et al. and Hersh et al. (22.3 vs. 47.1% and 41.3%, respectively) [33, 34]. Patients participating in the Vollmer and Hersh studies initiated DMF treatment in 2013—the year DMF was approved by the US Food and Drug Administration—before optimization of gastrointestinal side effect mitigation strategies, recommendations, and patient counseling to improve adherence to DMF treatment. To further illustrate this point, a recent paper by Sejbaek et al. reported a substantial variance in discontinuation rates between two different MS centers, 17.3 versus 42.7% [35]; reportedly, the clinic with the lower discontinuation rate prospectively planned management of gastrointestinal events and flushing, and set patient expectations. Our data are supported by another real-world, single-center study where discontinuation rates for FTY and DMF during the first 12 months of treatment were similar (11 and 15%, respectively) [36].
Our study consists of an analysis of the clinical effectiveness of DMF in real-world clinical practice, specifically in patients with early MS switching from injectable therapies, the situation most often encountered by neurologists and healthcare professionals. However, this study does have limitations, such as a lack of randomization (because patients were self-selected for switching rather than having a randomized or serial selection protocol) and potential bias because of regression to the mean (for the patients who switched due to relapse). In this type of switch study, there may be a bias in favor of the switch therapy if patients and clinicians have decided on an alternative drug because of suboptimal efficacy or tolerability issues with the original therapy. Furthermore, the follow-up for patients initiated on DMF was 12 months, thus not providing longer term follow-up, which is relevant for a chronic disease such as RMS. There was also a lack of an active comparator control. Lastly, although a definition of relapse was provided to each clinician/investigator, independent verification was not obtained. Despite these potential shortcomings, we believe that these analyses provide valuable insights that may further guide clinical decision-making in the management of patients with early RMS, particularly those requiring a change in disease-modifying therapies.
As more MS disease-modifying therapies become available, there is an increasing need to investigate reasonable treatment switching strategies, to guide both clinicians and patients through the evolving MS therapeutic landscape.
Conclusions
These results suggest that DMF may be an effective treatment option in early MS patients with RMS who experience a suboptimal response to GA therapy. DMF was associated with a lower relapse rate in patients after 12 months of therapy compared with the 12 months before initiation of DMF, with improvement in PROs over the same time frame.
References
Wootla B, Eriguchi M, Rodriguez M. Is multiple sclerosis an autoimmune disease? Autoimmune Dis. 2012;2012:969657.
Orton SM, Herrera BM, Yee IM, et al. Sex ratio of multiple sclerosis in Canada: a longitudinal study. Lancet Neurol. 2006;5:932–6.
Gold R, Kappos L, Arnold DL, et al. Placebo-controlled Phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J Med. 2012;367:1098–107.
Fox RJ, Miller DH, Phillips JT, et al. Placebo-controlled Phase 3 study of oral BG-12 or glatiramer in multiple sclerosis. N Engl J Med. 2012;367:1087–97.
Kita M, Fox RJ, Phillips JT, et al. Effects of BG-12 (dimethyl fumarate) on health-related quality of life in patients with relapsing–remitting multiple sclerosis: findings from the CONFIRM study. Mult Scler. 2014;20:253–7.
Kita M, Fox RJ, Gold R, et al. Effects of delayed-release dimethyl fumarate (DMF) on health-related quality of life in patients with relapsing-remitting multiple sclerosis: an integrated analysis of the Phase 3 DEFINE and CONFIRM studies. Clin Ther. 2014;36:1958–71.
Viglietta V, Miller D, Bar-Or A, et al. Efficacy of delayed-release dimethyl fumarate in relapsing-remitting multiple sclerosis: integrated analysis of the Phase 3 trials. Ann Clin Transl Neurol. 2015;2:103–18.
Gold R, Giovannoni G, Phillips JT, et al. Efficacy and safety of delayed-release dimethyl fumarate in patients newly diagnosed with relapsing-remitting multiple sclerosis (RRMS). Mult Scler. 2015;21:57–66.
Gold R, Arnold DL, Bar-Or A, et al. Long-term effects of delayed-release dimethyl fumarate in multiple sclerosis: interim analysis of ENDORSE, a randomized extension study. Mult Scler. 2017;23:253–65.
Buron MD, Chalmer TA, Sellebjerg F, et al. Comparative effectiveness of teriflunomide and dimethyl fumarate: a nationwide cohort study. Neurology. 2019;92:e1811–20.
Braune S, Grimm S, van Hövell P, et al. Comparative effectiveness of delayed-release dimethyl fumarate versus interferon, glatiramer acetate, teriflunomide, or fingolimod: results from the German NeuroTransData registry. J Neurol. 2018;265:2980–92.
Ontaneda D, Nicholas J, Carraro M, et al. Comparative effectiveness of dimethyl fumarate versus fingolimod and teriflunomide among MS patients switching from first-generation platform therapies in the US. Mult Scler Relat Disord. 2019;27:101–11.
Kalincik T, Kubala Havrdova E, Horakova D, et al. Comparison of fingolimod, dimethyl fumarate and teriflunomide for multiple sclerosis. J Neurol Neurosurg Psychiatry. 2019;90:458–68.
Prosperini L, Lucchini M, Haggiag S, et al. Fingolimod vs dimethyl fumarate in multiple sclerosis: a real-world propensity score-matched study. Neurology. 2018;91:e153–61.
Cutter G SM, Betts M, Nambiar S, Fahrbach K, Sarri G, Rock M, Debray T. (2019) Comparative effectiveness of delayed-release dimethyl fumarate vs. other disease-modifying therapies in patients with multiple sclerosis: a network meta-analysis of real-world evidence. Presented at European Committee for Treatment & Research in Multiple Sclerosis (ECTRIMS). Stockholm. https://onlinelibrary.ectrims-congress.eu/ectrims/2019/stockholm/278594/gary.cutter.comparative.effectiveness.of.delayed-release.dimethyl.fumarate.vs.html. Accessed 16 Sept 2019.
Sejbaek T, Nielsen HH, Penner N, et al. Dimethyl fumarate decreases neurofilament light chain in CSF and blood of treatment naïve relapsing MS patients. J Neurol Neurosurg Psychiatry. 2019;90:1324–30.
Longbrake E, Mao-Draayer Y, Cascione M, et al. Dimethyl fumarate treatment shifts the immune environment toward an anti-inflammatory cell profile while maintaining protective humoral immunity. Mult Scler. 2020. https://doi.org/10.1177/1352458520937282.
Kresa-Reahl K, Repovic P, Robertson D, Okwuokenye M, Meltzer L, Mendoza JP. Effectiveness of delayed-release dimethyl fumarate on clinical and patient-reported outcomes in patients with relapsing multiple sclerosis switching from glatiramer acetate: RESPOND, a prospective observational study. Clin Ther. 2018;40:2077–87.
US Food and Drug Administration (2017) Highlights of prescribing information. TECFIDERA. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/204063s017lbl.pdf. Accessed 6 Feb 2020.
Atkinson MJ, Sinha A, Hass SL, et al. Validation of a general measure of treatment satisfaction, the Treatment Satisfaction Questionnaire for Medication (TSQM), using a national panel study of chronic disease. Health Qual Life Outcomes. 2004;2:12.
Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics. 1993;4:353–65.
Cohan SL, Moses H, Calkwood J, et al. Clinical outcomes in patients with relapsing-remitting multiple sclerosis who switch from natalizumab to delayed-release dimethyl fumarate: a multicenter retrospective observational study (STRATEGY). Mult Scler Relat Disord. 2018;22:27–34.
Chinea A, Amezcua L, Vargas W, et al. Real-world safety and effectiveness of dimethyl fumarate in Hispanic or Latino patients with multiple sclerosis: 3-year results from ESTEEM. Neurol Ther. 2020. https://doi.org/10.1007/s40120-020-00192-6.
Williams MJ, Amezcua L, Okai A, et al. Real-world safety and effectiveness of dimethyl fumarate in black or African American patients with multiple sclerosis: 3-year results from ESTEEM. Neurol Ther. 2020. https://doi.org/10.1007/s40120-020-00193-5.
Khademi M, Dring AM, Gilthorpe JD, et al. Intense inflammation and nerve damage in early multiple sclerosis subsides at older age: a reflection by cerebrospinal fluid biomarkers. PLoS ONE. 2013;8:e63172.
Khurana V, Sharma H, Afroz N, Callan A, Medin J. Patient-reported outcomes in multiple sclerosis: a systematic comparison of available measures. Eur J Neurol. 2017;24:1099–107.
D’Amico E, Leone C, Patti F. Disability may influence patient willingness to participate in decision making on first-line therapy in multiple sclerosis. Funct Neurol. 2016;31:21–3.
Pereira VC, Malfetano FR, Meira ID, et al. Clinical response to interferon beta and glatiramer acetate in multiple sclerosis patients: a Brazilian cohort. Arq Neuropsiquiatr. 2012;70:774–9.
National Institute for Health and Care Excellence. Multiple sclerosis. https://bnf.nice.org.uk/treatment-summary/multiple-sclerosis.html. Accessed 5 Feb 2020.
Andravizou A, Dardiotis E, Artemiadis A, et al. Brain atrophy in multiple sclerosis: mechanisms, clinical relevance and treatment options. Autoimmun Highlights. 2019;10:1–25.
Cerqueira JJ, Compston DAS, Geraldes R, et al. Time matters in multiple sclerosis: can early treatment and long-term follow-up ensure everyone benefits from the latest advances in multiple sclerosis? J Neurol Neurosurg Psychiatry. 2018;89:844–50.
Giles K, Hanna J, Wu F, et al editors. Efficacy of delayed-release dimethyl fumarate in newly diagnosed and other early MS patients, and patients switching from interferon or glatiramer acetate, in routine medical practice: interim results from ESTEEM. Los Angeles: American Academy of Neurology; 2018.
Vollmer B, Nair KV, Sillau SH, Corboy J, Vollmer T, Alvarez E. Comparison of fingolimod and dimethyl fumarate in the treatment of multiple sclerosis: two-year experience. Mult Scler J Exp Transl Clin. 2017;3:2055217317725102.
Hersh CM, Love TE, Bandyopadhyay A, et al. Comparative efficacy and discontinuation of dimethyl fumarate and fingolimod in clinical practice at 24-month follow-up. Mult Scler J Exp Transl Clin. 2017;3:2055217317715485.
Sejbaek T, Nybo M, Petersen T, Illes Z. Real-life persistence and tolerability with dimethyl fumarate. Mult Scler Relat Disord. 2018;24:42–6.
Repovic P, Park Y, Herrera V, editors. Treatment discontinuation and clinic resource utilization among patients treated with fingolimod and dimethyl fumarate in the real-world setting. London: European Committee for Treatment and Research in Multiple Sclerosis; 2016.
Ratzker PK, Feldman JM, Scheinberg LC, et al. Self-assessment of neurologic impairment in multiple sclerosis. Neurorehabil Neural Repair. 1997;11:207–11.
Acknowledgements
Funding
This study and the journal’s rapid service fee were sponsored by Biogen (Cambridge, MA, USA).
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
Study concept and design: JP Mendoza. Acquisition, analysis, or interpretation: P Repovic, D Robertson, K Kresa-Reahl, SL Cohan, R Su, R Avila, I Koulinska, JP Mendoza. Drafting: P Repovic, D Robertson, K Kresa-Reahl, SL Cohan, R Su, R Avila, I Koulinska, JP Mendoza. Critical revision of the manuscript for important intellectual content: P Repovic, D Robertson, K Kresa-Reahl, SL Cohan, R Su, R Avila, I Koulinska, JP Mendoza. Statistical analysis: R Su. Obtained funding: K Kresa-Reahl, JP Mendoza. Administrative, technical, or material support: R Su, JP Mendoza. Study supervision: JP Mendoza. R Su and JP Mendoza had full access to all the data in the study, and take responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing and/or Editorial Assistance
Biogen provided funding for medical writing support in the development of this manuscript. Katherine Ayling-Rouse from Excel Scientific Solutions wrote the first draft of the manuscript based on input from authors, and Nathaniel Hoover from Excel Scientific Solutions copyedited and styled the manuscript per journal requirements. Biogen reviewed the manuscript and provided feedback to the authors. The authors had full editorial control of the manuscript and provided their final approval of all content.
Disclosure
Pavle Repovic has been a consultant to Alexion, Biogen, Celgene, EMD Serono, Genentech, Genzyme, Novartis, and Viela; and served on speaker bureaus for Alexion, Biogen, Celgene, EMD Serono, Genentech, Genzyme, and Viela. Derrick Robertson has served as a consultant for Alexion, Biogen, Celgene, EMD Serono, Genentech, Novartis, Sanofi-Genzyme, and Teva Neuroscience; served on speaker bureaus for Acorda, Alexion, Biogen, Celgene, EMD Serono, Genentech, Mallinckrodt, Novartis, Sanofi-Genzyme, and Teva Neuroscience; and received grant support from Actelion, Biogen, EMD Serono, Genentech, Mallinckrodt, MedDay, Novartis, PCORI, Sanofi-Genzyme, and TG. Kiren Kresa-Reahl is currently employed as a medical director for Atara; has served on speaker bureaus for Biogen, EMD Serono, Genzyme, Novartis, Pfizer, and Teva; and has received research funding from Biogen, Genzyme, Mallinckrodt, and Novartis. Stanley L. Cohan has received research support from AbbVie, Adamas, Alexion, Biogen, MedDay, Novartis, Roche-Genentech, and Sanofi-Genzyme; served on steering committees/advisory boards for Biogen, EMD Serono, Novartis, Pear, Roche-Genentech, and Sage; and received speaking honoraria from Biogen, Sanofi-Genzyme, and Roche-Genentech. Ray Su, Robin Avila, Irene Koulinska, and Jason P. Mendoza are employees of and hold stock/stock options in Biogen.
Compliance with Ethics Guidelines
The study was conducted in accordance with relevant US federal regulations, the Declaration of Helsinki, and the International Council on Harmonisation Guideline for Good Clinical Practice. Approvals were granted by relevant institutional ethics committees (See full list of Institutional review board approvals as supplementary material) for study protocol and amendments, and written assent and consent forms were obtained from each patient and his or her parent or legal guardian. RESPOND was registered at ClinicalTrials.gov (NCT01903291).
Data Availability
All data generated or analyzed during this analysis are included in this published article/as supplementary information files.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Repovic, P., Robertson, D., Kresa-Reahl, K. et al. Effectiveness of Dimethyl Fumarate in Patients With Relapsing Multiple Sclerosis Switching After Suboptimal Response to Glatiramer Acetate, Including Patients With Early Multiple Sclerosis: Subgroup Analysis of RESPOND. Neurol Ther 10, 169–182 (2021). https://doi.org/10.1007/s40120-020-00223-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40120-020-00223-2