Abstract
Background
Multiple forms of electrical stimulation (ES) potentially offer widely varying clinical benefits. Diminished function commonly associated with acute and chronic pain lessens productivity and increases medical costs. This review aims to compare the relative effects of various forms of ES on functional and pain outcomes.
Methods
A comprehensive literature search focused on studies of commonly marketed forms of ES used for treatment of pain and improvement of function. Peer-reviewed manuscripts were categorized as “Important” (systematic review or meta-analysis, randomized controlled trial, observational cohort study) and “Minor” (retrospective case series, case report, opinion review) for each identified form of ES.
Results and Discussion
Varying forms of ES have markedly different technical parameters, applications, and indications, based on clinically meaningful impact on pain perception, function improvement, and medication reduction. Despite being around for decades, there is limited quality evidence for most forms of ES, although there are several notable exceptions for treatment of specific indications. Neuromuscular electrical stimulation (NMES) has well-demonstrated beneficial effects for rehabilitation of selective spinal cord injured (SCI), post-stroke, and debilitated inpatients. Functional electrical stimulation (FES) has similarly shown effectiveness in rehabilitation of some stroke, SCI, and foot drop outpatients. H-Wave® device stimulation (HWDS) has moderate supportive evidence for treatment of acute and refractory chronic pain, consistently demonstrating improvements in function and pain measures across diverse populations. Interestingly, transcutaneous electrical nerve stimulation (TENS), the most widely used form of ES, demonstrated insignificant or very low levels of pain and functional improvement.
Conclusion
Ten of 13 reviewed forms of ES have only limited quality evidence for clinically significant reduction of pain or improvement of function across different patient populations. NMES and FES have reasonably demonstrated effectiveness, albeit for specific clinical rehabilitation indications. HWDS was associated with the most clinically significant outcomes, in terms of functional improvement combined with reduction of pain and medication use. More rigorous long-term clinical trials are needed to further validate appropriate use and specific indications for most forms of ES.
Level of Evidence
II.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Evidence quality remains low for clinically significant effectiveness of many forms of electrical stimulation (ES). |
Insignificant or low levels of improvements in pain and function have been consistently reported for some forms of ES [e.g., transcutaneous electrical nerve stimulation (TENS)]. |
Neuromuscular electrical stimulation (NMES) and functional electrical stimulation (FES) are supported by moderate quality evidence for rehabilitation of stroke, spinal cord injury (SCI), debilitation, and nerve dysfunction. |
H-Wave® device stimulation (HWDS) has moderate quality evidence for functional improvement with reduction of pain and medication use in acute and chronic pain conditions. |
Introduction
Chronic pain, defined as a minimum of 3 months duration, is a major source of worldwide suffering, interfering with activities of daily living and overall functioning [1, 2]. The US Centers for Disease Control has estimated the national prevalence of chronic pain to possibly be as high as 40% [3, 4]. While perception of acute pain appropriately notifies the body of imminent danger, chronic pain involves disruptive neural signals which compromise function and lead to debilitation. Chronic pain often results in inability to work and lowers self-esteem, leading to disturbing life events, including a high incidence of divorce and even suicide [5,6,7,8]. Within the US an estimated US$560 billion per year in lost productivity has been attributed to the effects of chronic pain [9].
Widely applied treatment regimens for chronic pain promote medications like non-steroidal anti-inflammatory drugs and opioids [10], while non-pharmacological treatment options include physical therapy, exercise, cognitive behavioral therapy, invasive interventions like nerve blocks/ablation, and complementary and alternative medicine, including acupuncture [11,12,13,14,15]. Multimodal regimens are reasonably effective through various treatment combinations. Some pharmacological treatments have disturbing side effects, particularly addiction and overdose with long-term opioids use [10, 16]. With the North American “opioid epidemic”, there is a dire need for less toxic treatment options which target primary sources of pain generation [17, 18].
Several forms of ES stimulate muscles that may be atrophied by disuse or neurological compromise and are intended primarily for functional rehabilitation and training. These include neuromuscular electrical stimulation (NMES), functional electrical stimulation (FES), and electrical muscle stimulation (EMS). Numerous other forms of ES emit widely varying electrical pulses and current into and beneath the skin over a painful anatomical target, to reduce pain and/or improve function in a potentially safer and more physiological manner than with drugs. These electrotherapies, primarily intended for pain and/or to improve function and applied stand-alone or as adjuncts, include transcutaneous electrical nerve stimulation (TENS), H-Wave® device stimulation (HWDS), interferential current (IFC) or therapy (IFT), noninvasive interactive neurostimulation (NIN), pulsed magnetic field therapy (PMFT) or pulsed electromagnetic field therapy (PEMF) or pulsed electrical stimulation (PES), galvanic current stimulation (GCT) or direct current (DC), microcurrent electrical nerve stimulation (MENS), percutaneous electrical nerve stimulation (PENS), percutaneous neuromodulation therapy (PNT) or peripheral nerve stimulation (PNS) or peripheral nerve field stimulation (PNFS), and transcranial direct current stimulation (tDCS).
Most of these forms of ES are non-invasive or subcutaneous modalities, each designed to either reduce pain and improve function or to assist with rehabilitation and training [19]. There is an urgent need for such innovative modalities and treatment paradigms, based on best available clinical evidence, to improve and maximize overall patient outcomes and healthcare. The purpose of this scoping, comparative review is to critically analyze the literature regarding the relative effectiveness of currently available forms of ES, particularly focusing on pain and functional improvement. Basic recommendations will follow regarding appropriate use for different conditions and patient populations.
Methods
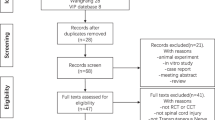
A scoping literature search of all published studies from 2000 to 2023 was conducted using PubMed, Web of Science, Embase, and the Cochrane Library, with the intention of providing an extensive and comprehensive overview and categorization of various forms of available ES, something currently unavailable in the literature. Inclusion criteria included studies primarily focusing on the use of ES for treatment of pain and improvement of functional status. ES was defined as use of a medical device to send electrical pulses to an anatomical target (i.e., nerve, muscle, vessel) with the intention to influence nerve conduction and/or muscle contraction. Some forms of ES have been described in the literature by several different names, so every effort was made to thoroughly cover available related technologies. Articles retrieved from the searches were evaluated independently by one reviewer using predefined standardized data extraction forms, and then data were evaluated again by a second independent reviewer. Following article retrieval, studies were categorized as “Important” (systematic review or meta-analysis, randomized controlled trial, observational cohort study) and “Minor” (retrospective case series, case report, opinion review). This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Results
Evidence for each form of ES is categorized and recorded in Table 1.
Marketed Primarily for Neurological or Disuse Functional Rehabilitation and Training
Neuromuscular Electrical Stimulation (NMES)
NMES alternates depolarization and repolarization primarily to decrease muscle spasm and prevent disuse atrophy [20]. NMES emits a mid-frequency rectangular waveform with pulse duration around 500 µs [21]. This form of ES stimulates muscles to repeatedly contract, simulating real exercise, which can lead to rapid fatigue and tetany when applied for longer periods [30]. NMES is prescribed for physical rehabilitation, mostly for patients with prolonged inpatient immobilization and for muscle strengthening in patients with spinal cord injuries [20,21,22]. A meta-analysis using NMES for cerebral palsy children suggested improvements in gross motor function (sitting and standing), although studies were of low quality [23]. Systematic reviews of NMES for muscle weakness in hospitalized adults with advanced disease indicated general effectiveness in combination with other rehabilitation methods, with proven safety for critically ill patients when properly applied [24, 25]. The effectiveness of NMES for pain relief, noting isolated reports of reduced neck pain in cervical spondylosis patients compared to sham treatment, remains questionable, with the majority of clinical studies being of low quality and failing to report adverse outcomes related to tetanic muscle contractions [26, 27].
Functional Electrical Stimulation (FES)
FES emits various programmed sequences of nerve stimulation eliciting distinct muscle contractions, which resemble some normal functional movements, for stroke recovery and highly motivated spinal cord injury patients (SCI) [28, 29]. A systematic review of 34 clinical trials concluded that FES was superior in improving movement scores of stroke patients with upper limb dysfunction when compared to other modalities including TENS, NMES, and tDCS [30]. FES cycling as an adjunct therapy for chronic SCI patients demonstrated a marked increase in daily functional measures compared to stretching, in contrast to outcomes of another multi-center trial in acquired brain injury patients which failed to demonstrate any improved mobility [31, 32]. FES is most utilized to stimulate muscles of the anterior compartment of the leg, causing functional dorsiflexion for patients with peroneal nerve palsy, effectively resulting in increased walking speed [33, 34]. The addition of FES to a conventional foot drop rehabilitation program showed greater reduction of lower extremity spasticity, with motor strength recovery being superior to use of an ankle–foot orthosis [29, 35, 36]. FES is not currently indicated as a treatment modality for pain, with more research needed to understand long-term therapeutic effects [28, 30].
Electrical Muscle Stimulation (EMS)
EMS has a similar design to TENS but with stronger stimulation intensity which causes muscle contractions. The most common application of EMS is for facial muscles (e.g., Bell’s palsy), although it has been proposed to treat neck and upper back conditions. A systematic review noted an extremely low quality of evidence, which has suggested ineffectiveness compared to placebo, with no reduction of disability or pain [37].
Marketed Primarily for Pain Relief and/or Function Improvement
Transcutaneous Electrical Nerve Stimulation (TENS)
TENS is a widely promoted intervention for chronic pain and spasticity, most commonly being prescribed for temporary relief of neck and low back pain. A TENS device emits variable electrical impulses, with most applications using higher frequencies between 40 and 150 Hz, with a short rectangular-shaped pulse duration typically around 50 µs. Low frequencies between 1 and 4 Hz with high intensity are less commonly used in a theoretical attempt to inhibit peripheral large-diameter, nociceptive transmission neurons [38,39,40]. Given the variability of application protocols, TENS evidence continues to be compromised by inconsistency in parameters, dosing, and testing populations, which have not been standardized in reported trials [41]. A 2019 systematic review of TENS for chronic pain found the quality of the evidence to be incredibly low, while it has not been demonstrated to be harmful or beneficial for pain control, disability, health-related quality of life (QoL), or in reducing use of pain-relieving medications [42]. Meta-analyses reported that TENS negligibly improved symptoms of chronic back pain relative to control treatments, but was less effective than other neurostimulation device forms [16, 43]. Another meta-analysis of TENS for chronic low back pain reported only very slight pain relief (< 1 on 1–10 pain scale), also noting little beneficial effect over 5 weeks of treatment [41]. One systematic review and meta-analysis suggested that the addition of TENS, as an adjunct to a multi-modal pain protocol following total knee arthroplasty, might help reduce reported pain and narcotic use during the first 24 h, although no beneficial effect was noted at 2-weeks [44].
H-Wave® Device Stimulation (HWDS)
HWDS, a unique form of ES, has different pain-relieving high-frequency (60 Hz) effects than TENS, with primary low-frequency (2 Hz) muscle effects being notably distinct from NMES. HWDS has a much different waveform compared to other forms of ES, emitting a very prolonged (5000 µs) pulse width/duration, being orders of magnitude longer than with other devices. While high-frequency mode effects can produce effective anesthesia/analgesia, low-frequency mode does not cause muscle weakness or tetany with extended use like NMES [45]. Pre-clinical studies demonstrated that the exponentially decaying biphasic waveform causes non-fatiguing muscle contractions and positive physiological effects on blood and lymph flow (vasodilation, angiogenesis), as well as nerve function [46, 47]. A 2021 systematic review of HWDS clinical trials found that low- to moderate-quality studies consistently reported clinically significant improvements in pain and function, as well as a reduction in pain medication use over a spectrum of acute and chronic pain disorders. Noting an excellent safety profile and relative cost-effectiveness, consideration of HWDS as part of a multimodal pain management program was suggested [45]. A cohort survey study of first responders using HWDS made available at the job site demonstrated multiple work-related performance improvements [48]. A large observational cohort study of end-stage workers’ compensation chronic (mean 8 years) pain patients found that HWDS treatment led to significant QoL gains, in terms of improved function, reported pain, and decreased medication use [49].
Interferential Current (IFC) or Therapy (IFT)
IFC, sometimes called IFT, uses differing medium-frequency electrical currents to reduce tissue impedance, theoretically allowing higher penetration into target tissues to inhibit the nervous system, supposedly resulting in muscle relaxation, pain suppression, and accelerated healing [50]. However, IFC has provided no additional clinical benefits for first-line conservative treatment of patients with low back or lower extremity pain, or for subacromial impingement syndrome [51,52,53]. While two older systematic reviews suggested marginal benefit, non-standardized protocols, heterogeneity, and other methodological limitations have significantly undermined the quality of IFC evidence in musculoskeletal pain and knee osteoarthritis [54, 55]. A more recent IFC systematic review found no statistically significant reduction in treatment of acute and chronic pain [56].
Noninvasive Interactive Neurostimulation (NIN)
NIN reportedly stimulates A, delta, and C nerve fibers, emitting larger currents through areas of lower impedance [57, 58]. Specialized attachments can be interchanged to access different nerve regions, including a probe, an elastic band with sensors, and a cranium comb. Compared to TENS and IFC, this form of ES has a higher amplitude of stimulation, where it has been suggested that NIN therapy may supplement a standard rehabilitation protocol to decrease pain over the short term in post-TKA patients [59]. One meta-analysis of different treatment modalities for plantar fasciitis reported that NIN seemed to be the most effective in reducing pain [60]. However, this product is not easily accessible and quite expensive, the mechanism of action is poorly understood, and there are insufficient trials demonstrating long-term effectiveness [58, 60, 61].
Pulsed Magnetic Field Therapy (PMFT) or Pulsed Electromagnetic Field therapy (PEMF) or Pulsed Electrical Stimulation (PES)
PMFT, also called pulsed electromagnetic field therapy or pulsed electrical stimulation, emits subsensory pulsed electric potentials that supposedly alter the homeostatic balance of cartilage degradation and synthesis, stimulating repair in degenerative conditions like osteoarthritis [19, 62, 63]. One systematic review concluded that PMFT might result in mild benefit for some knee osteoarthritis patients [64]. Another systematic review and meta-analysis reported that PMFT might have limited effectiveness in improving physical function in knee osteoarthritis patients, but there was no effect on pain reduction, with the authors acknowledging that the strength of evidence was low for physical function and extremely low for pain [62]. A Comparative Effectiveness Review by the Agency for Healthcare Research and Quality concluded that PMFT failed to improve disability (low evidence) or pain (moderate evidence) [65]. Concerns have also been reported about possible excessive non-ionizing radiation exposure with several European PMFT systems [66].
Galvanic Current Stimulation (GCT) or Direct Current (DC)
GCT, also called direct current, has been experimented with over several centuries and is still used in some treatments of the skin, being promoted for inhibiting nociceptor activity. It is also believed to help transport ionized substances through the skin, particularly topical anti-inflammatory drugs (iontophoresis). Systematic reviews have reported low-quality evidence that GCT does not reduce disability or pain compared to placebo [37].
Microcurrent Electrical Neuromuscular Stimulation (MENS)
MENS is a sub-sensory very low-level current that is purported to mimic naturally occurring electrical body impulses to decrease pain and to facilitate healing processes. Despite some brief duration improvement in grip strength and heel rise in elderly patients compared to sham, no subsequent clinical improvement was noted [67]. This ES form has a notable paucity of evidence in the literature.
Percutaneous Electrical Nerve Stimulation (PENS)
PENS, unlike TENS, uses needles that are inserted around or immediately adjacent to the targeted nerve, making it indistinguishable from electro-acupuncture. Randomized controlled trials of PENS have reported limited clinical improvement in migraine and neck pain patients, but less so for low back and knee pain [68,69,70,71]. PENS lacks quality evidence demonstrating any sustained benefit, so when trialed should only be with a licensed and trained therapist after more conventional treatments have failed [72].
Percutaneous Neuromodulation Therapy (PNT) or Peripheral Nerve Stimulation (PNS) or Peripheral Nerve Field Stimulation (PNFS)
PNT and its variations including PNS and PNFS, involve electrodes placed percutaneously or deeper in the region of targeted peripheral nerves for treatment of postoperative pain, neuropathic pain, or complex regional pain syndrome. Clinical evidence for this form of ES has been mostly limited to case reports/series and retrospective reviews, with insufficient evidence to support safety and effectiveness, with reported complications when older open nerve exposure techniques were used for electrode placement, resulting in multiple revisions from lead migration or failure [73, 74]. A more recent systematic review of PNS, typically using ultrasound lead placement, specifically noted a lack of high-quality RCTs with methodological limitations and heterogeneity, but indicated some potential benefit for chronic migraines, cluster headaches, post-amputation pain, chronic pelvic pain, and possibly for chronic low back and lower extremity pain [75].
Transcranial Direct Current Stimulation (tDCS)
tDCS uses surface cranial leads to stimulate the sensorimotor cortex, reportedly altering gamma-aminobutyric acid neurotransmission and resting membrane potential, resulting in either depolarization or hyperpolarization, depending on flow and axon orientation. This ES form contrasts with the efferent and afferent peripheral nerve stimulation of many other ES forms [76, 77]. Studies have reported cathode tDCS improvement in QoL and arm function in stroke patients, but only tentative evidence for pain reduction in fibromyalgia patients [78, 79]. A 2020 meta-analysis concluded that existing evidence was underwhelming and that tDCS was ineffective for reducing chronic back pain [80]. Previous systematic reviews of non-invasive brain stimulation techniques highlighted that tDCS had minimal short-term effects on chronic pain, noting multiple sources of research bias [81, 82]. Another systematic review questioned whether tDCS had any effect at all, noting only one demonstrable effect on a single neurophysiological monitoring measure, motor-evoked potential amplitude [83].
Miscellaneous ES Forms Unrelated to Pain and Function
The forms of ES selected for this review have focused strictly on devices that have been designed and marketed to improve pain and/or function, where there has been some medical evidence to draw reasonable conclusions regarding relative effectiveness and clinical indications. Literature searches led to numerous other FDA-cleared ES device forms for other unrelated indications, primarily for use by physical and wound therapists or for treatment of psychological disorders. Physical and wound therapy-related examples include the following: Iontophoresis, Faradic ES (muscle reeducation/atrophy), Russian ES (e.g., Olympic muscle enhancement), EMG-triggered stimulation (partial functioning nerves), reciprocal EMG-triggered stimulation (over-stimulated nerves), high-voltage ES (wound healing), and high volt pulsed current (edema, decubitus wounds). Examples of ES forms used for psychological disorders include the following: electroconvulsive therapy (severe depression), deep brain stimulation (severe depression, neurological disorders), repetitive transcranial magnetic stimulation (depression, severe OCD), and vagus nerve stimulation (depression, PTSD).
Discussion
Comparison of ES Types and General Recommendations
Function
Functional loss can occur because of direct neurological and/or motor deficit resulting from stroke, SCI, nerve injury, and muscle disease or disuse. Another common reason for functional compromise is from any sustained perception of pain, which similarly leads to guarding and relative disuse. As such, clinical research which focuses on objective functional outcome measures probably represents a more reliable indicator of overall effectiveness than basic recording of various subjective pain measures. Some forms of ES that address recovery of muscle or nerve dysfunction do not simultaneously provide pain relief. This review highlighted that several of the more effective ES forms had a greater impact on functional measures than reported pain.
Of the three recommended forms of ES, NMES has proven effectiveness in the rehabilitation of SCI and immobilized/debilitated inpatients, as well as for cerebral palsy patients, but does not seem to reduce pain significantly [20,21,22,23,24,25,26,27]. FES is another effective ES tool for stroke recovery and with highly motivated SCI outpatients, as well as for functional treatment of foot drop (peroneal nerve palsy), although no pain relief should be expected [28,29,30, 33, 34]. Several moderate-quality HWDS studies have shown statistically significant gains in functional measures, being somewhat higher by percentage than the recorded reductions in pain measures [45, 48, 49].
While many forms of ES preclude recommendation, two warrant mention for preliminary findings related to functional improvement. PMFT might have limited effectiveness in improving physical function in knee osteoarthritis patients [62, 64], and cathode tDCS seemed to result in improvement in QoL and arm function in stroke patients [78, 79].
Pain
While pain measurements (e.g., VAS, BPI) remain problematically subjective, recorded reduction of pain medication use has more potential for objectivity when comparing ES effectiveness. Of the three recommended ES forms, only HWDS demonstrated statistically significant reductions in pain measures, as well as medication reduction [45, 49]. Several other non-recommended ES forms had marginal effects on reported pain. TENS, which is widely implemented for short-term pain relief, reduced reported pain by < 1 on a 1–10 pain scale, and has reportedly been helpful as an early post-operative adjunct following total knee surgery, as part of a multimodal pain control protocol [41, 44]. NIN therapy may similarly help decrease pain over the short term in post-TKA patients, and shows some promise for plantar fascia pain [59, 60]. Limited clinical improvement in migraine and neck pain patients has been reported with PENS [68,69,70,71]. PNS may have some potential benefit for chronic migraines and cluster headaches, post-amputation pain, chronic pelvic pain, and possibly for chronic low back and lower extremity pain [75].
Confusion in the Literature and Marketplace
Given the defined scope of this comparative review of ES forms, focused on effectiveness for improving function and decreasing pain, it is important to understand how much misinformation and lack of standardization exists, particularly in marketing materials directed to consumers and providers. Even the multiple names for a single form of ES further obfuscate collective understanding and differentiation between available devices. Further confusion results from insurance coverage decisions, which often reflect an attitude of rejection of all ES devices, regardless of merit, based on best available medical evidence, or common mis-grouping of much different ES forms. The FDA clearance process creates another twist, where a system of broad classifications (buckets) is used. Even within one specific classification, it is widely acknowledged in the healthcare community that each class contains significant variation and diversity between cleared devices/drugs, while the FDA never attempts to differentiate or to provide guidance regarding relative clinical or cost-effectiveness between cleared devices/drugs.
In summary, this scoping comparative review presents a thorough overview, currently unavailable in the literature, but has not attempted to present a myriad of technical aspects for each and every distinct ES form. For those more interested, there are many significant design differences, primarily related to waveform, pulse frequency, and pulse duration. Waveforms are commonly DC, AC, burst or modulated AC, and low-intensity DC, among others, including “exponentially decaying.” Pulse frequency (in Hz) is typically low, middle, or high, while pulse duration (in µs) can be short, mid, long, or ultralong. Based on such varying reported clinical outcomes, it seems reasonable to conclude that such notable differences in electrical parameters are important for effective ES form design.
Conclusion
Most forms of ES are used as second-line therapies, following the failure of other conventional treatments. When considered as a whole, the majority of ES forms for treatment of pain and loss of function lack highly rigorous research. This may be at least partly attributed to the fact that most are FDA-cleared as Class II medical devices, which have less demanding research requirements than for higher safety risk Class III devices. Nevertheless, several forms of ES appear to be relatively effective for rehabilitation of function, as well as reduction of pain and medication use. This comparative analysis of 13 distinct forms of ES found disappointing clinical results for 10 of them. NMES and FES have demonstrated reasonable effectiveness for the functional rehabilitation of some stroke, SCI, and patients with certain types of nerve compromise. HWDS appears to have the most promising clinical outcomes, in terms of functional improvement combined with reduction of pain and medication use. More higher-quality long-term clinical trials are needed to further validate appropriate use and to identify specific indications for most forms of ES.
Data Availability
The data used in this review are available from multiple sources, including public databases PubMed, Web of Science, Embase, and the Cochrane Library and are included in the references section of this article.
References
Treede RD, Rief W, Barke A, et al. Chronic pain as a symptom or a disease: the IASP Classification of Chronic Pain for the International Classification of Diseases (ICD-11). Pain. 2019;160(1):19–27. https://doi.org/10.1097/j.pain.0000000000001384.
Mäntyselkä P, Kumpusalo E, Ahonen R, et al. Pain as a reason to visit the doctor: a study in Finnish primary health care. Pain. 2001;89:175–80.
Cohen SP, Vase L, Hooten WM. Chronic pain: an update on burden, best practices, and new advances. The Lancet. 2021;397(10289):2082–97. https://doi.org/10.1016/s0140-6736(21)00393-7.
Dahlhamer J, Lucas J, Zelaya C, et al. Prevalence of chronic pain and high-impact chronic pain among adults. MMWR Morb Mortal Wkly Rep. 2018;67:1001–6.
Landefeld JC, Miaskowski C, Tieu L, et al. Characteristics and factors associated with pain in older homeless individuals: Results from the Health Outcomes in people experiencing homelessness in older middle age (hope home) study. J Pain. 2017;18(9):1036–45. https://doi.org/10.1016/j.jpain.2017.03.011.
Fisher R, Ewing J, Garrett A, Harrison EK, Lwin KKT, Wheeler DW. The nature and prevalence of chronic pain in homeless persons: an observational study. F1000Research. 2013;2:164. https://doi.org/10.12688/f1000research.2-164.v1.
Morasco BJ, Gritzner S, Lewis L, Oldham R, Turk DC, Dobscha SK. Systematic review of prevalence, correlates, and treatment outcomes for chronic non-cancer pain in patients with comorbid substance use disorder. Pain. 2011;152(3):488–97. https://doi.org/10.1016/j.pain.2010.10.009.
Tang N, Crane C. Suicidality in chronic pain: a review of the prevalence, risk factors and psychological links. Psychol Med. 2006;36(5):575–86. https://doi.org/10.1017/S0033291705006859.
Steglitz J, Buscemi J, Ferguson MJ. The future of pain research, education, and treatment: a summary of the IOM report “Relieving pain in America: a blueprint for transforming prevention, care, education, and research.” Transl Behav Med. 2012;2(1):6–8. https://doi.org/10.1007/s13142-012-0110-2.
American Society of Anesthesiologists. (2022 Mar 23). Chronic Pain. https://www.asahq.org/madeforthismoment/pain-management/types-of-pain/chronic/. Accessed 25 Jan 2023.
Geneen LJ, Moore RA, Clarke C, Martin D, Colvin LA, Smith BH. Physical activity and exercise for chronic pain in adults: an overview of Cochrane Reviews. Cochrane Database Syst Rev. 2017. https://doi.org/10.1002/14651858.CD011279.pub3.
Williams AC, Fisher E, Hearn L, Eccleston C. Psychological therapies for the management of chronic pain (excluding headache) in adults. Cochrane Database Syst Rev. 2020. https://doi.org/10.1002/14651858.CD007407.pub. (Accessed 26 January 2023).
Harvard Health Publishing (2021 Sep 16). Pain. Available at: https://www.health.harvard.edu/topics/pain. Accessed 25 Jan 2023.
Li J, Szabova A. Ultrasound-guided nerve blocks in the head and neck for chronic pain management: the anatomy, sonoanatomy, and procedure. Pain Physician. 2021;24(8):533–48.
Bauer BA, Tilburt JC, Sood A, Li GX, Wang SH. Complementary and alternative medicine therapies for chronic pain. Chin J Integr Med. 2016;22(6):403–11. https://doi.org/10.1007/s11655-016-2258-y. (Epub 2016 May 26).
Voon P, Karamouzian M, Kerr T. Chronic pain and opioid misuse: a review of reviews. Subst Abuse Treat Prev Policy. 2017. https://doi.org/10.1186/s13011-017-0120-7.
Swedish Council on Health Technology Assessment. Methods of Treating Chronic Pain: A Systematic Review [Internet]. Stockholm: Swedish Council on Health Technology Assessment (SBU) Yellow Report. 2006, 177, 1–2.
Ekholm O, Diasso PDK, Davidsen M, Kurita GP, Sjøgren P. Increasing prevalence of chronic non-cancer pain in Denmark 360 from 2000 to 2017: a population-based survey. Eur J Pain. 2022;26:624–33.
White P. Faculty opinions recommendation of electrical stimulation for pain relief in knee osteoarthritis: Systematic review and network meta-analysis. In: Faculty Opinions–Post-Publication Peer Review of the Biomedical Literature. 2017. https://doi.org/10.3410/f.725277008.793540054.
Seifert HM, Fuglevand AJ. Restoration of movement using functional electrical stimulation and Bayes’ theorem. J Neurosci. 2002;22(21):9465–74.
Glaviano NR, Saliba S. Can the use of neuromuscular electrical stimulation be improved to optimize quadriceps strengthening? Sports Health. 2016;8(1):79–85.
Doucet BM, Lam A, Griffin L. Neuromuscular electrical stimulation for skeletal muscle function. Yale J Biol Med. 2012;85(2):201–15.
Salazar AP, Pagnussat AS, Pereira GA, Scopel G, Lukrafka JL. Neuromuscular electrical stimulation to improve gross motor function in children with cerebral palsy: a meta-analysis. Braz J Phys Ther. 2019;23(5):378–86. https://doi.org/10.1016/j.bjpt.2019.01.006. (Epub 2019 Jan 23).
Jones S, Man WD, Gao W, Higginson IJ, Wilcock A, Maddocks M. Neuromuscular electrical stimulation for muscle weakness in adults with advanced disease. Cochrane Database System Rev. 2016. https://doi.org/10.1002/14651858.CD009419.pub3.
Sachetti A, Carpes MF, Dias AS, Sbruzzi G. Safety of neuromuscular electrical stimulation among critically ill patients: systematic review. Revista Brasileira de Terapia Intensiva. 2018;30(2):219–25. https://doi.org/10.5935/0103-507X.20180036.
Miao Q, Qiang JH, Jin YL. Effectiveness of percutaneous neuromuscular electrical stimulation for neck pain relief in patients with cervical spondylosis. Medicine. 2018. https://doi.org/10.1097/md.0000000000011080.
Martimbianco ALC, Torloni MR, Andriolo BN, Porfírio GJ, Riera R. Neuromuscular electrical stimulation (NMES) for patellofemoral pain syndrome. Cochrane Database Syst Rev. 2017;12(12):CD11289. https://doi.org/10.1002/14651858.CD011289.pub2.
Vafadar AK, Côté JN, Archambault PS. Effectiveness of functional electrical stimulation in improving clinical outcomes in the upper arm following stroke: A systematic review and meta-analysis. BioMed Res Int. 2015. https://doi.org/10.1155/2015/729768.
Pilutti LA, Motl RW. Functional electrical stimulation cycling exercise for people with multiple sclerosis. Curr Treat Opt Neurol. 2019. https://doi.org/10.1007/s11940-019-0597-7.
Tang Y, Wang L, He J, Xu Y, Huang S, Fang Y. Optimal method of electrical stimulation for the treatment of upper limb dysfunction after stroke: a systematic review and bayesian network meta-analysis of randomized controlled trials. Neuropsychiatr Dis Treat. 2021;15(17):2937–54. https://doi.org/10.2147/NDT.S332967.
Sadowsky CL, Hammond ER, Strohl AB, et al. Lower extremity functional electrical stimulation cycling promotes physical and functional recovery in chronic spinal cord injury. J Spinal Cord Med. 2013;36(6):623–31. https://doi.org/10.1179/2045772313Y.0000000101.
de Sousa DG, Harvey LA, Dorsch S, Leung J, Harris W. Functional electrical stimulation cycling does not improve mobility in people with acquired brain injury and its effects on strength are unclear: a randomised trial. J Physiother. 2016;62(4):203–8. https://doi.org/10.1016/j.jphys.2016.08.004.
Springer S, Vatine JJ, Lipson R, Wolf A, Laufer Y. Effects of dual-channel functional electrical stimulation on gait performance in patients with hemiparesis. ScientificWorldJournal. 2012. https://doi.org/10.1100/2012/530906.
Marsden J, Stevenson V, McFadden C, Swain I, Taylor P. The effects of functional electrical stimulation on walking in hereditary and spontaneous spastic paraparesis. Neuromodulation. 2013;16(3):256–60. https://doi.org/10.1111/j.1525-1403.2012.00494.x. (discussion 260).
Sabut SK, Sikdar C, Kumar R, Mahadevappa M. Dorsiflexor muscle: effects on dorsiflexor strength, plantarflexor spasticity, and motor recovery in stroke patients. NeuroRehabilitation. 2011;29(4):393–400. https://doi.org/10.3233/NRE-2011-0717.
van Swigchem R, van Duijnhoven HJ, den Boer J, Geurts AC, Weerdesteyn V. Effect of peroneal electrical stimulation versus an ankle-foot orthosis on obstacle avoidance ability in people with stroke-related foot drop. Phys Ther. 2012;92(3):398–406. https://doi.org/10.2522/ptj.20100405.
Kroeling P, Gross A, Graham N, et al. Electrotherapy for neck pain. Cochrane Database Syst Rev. 2013. https://doi.org/10.1002/14651858.CD004251.pub5.
Johnson MI, Paley CA, Jones G, Mulvey MR, Wittkopf PG. Efficacy and safety of transcutaneous electrical nerve stimulation (TENS) for acute and chronic pain in adults: a systematic review and meta-analysis of 381 studies (the meta-TENS study). BMJ Open. 2022;12(2): e051073. https://doi.org/10.1136/bmjopen-2021-051073.
Levin MF, Hui-Chan CW. Conventional and acupuncture-like transcutaneous electrical nerve stimulation excite similar afferent fibers. Arch Phys Med Rehabil. 1993;74(1):54–60.
Sivaramakrishnan A, Solomon JM, Manikandan N. Comparison of transcutaneous electrical nerve stimulation (TENS) and functional electrical stimulation (FES) for spasticity in spinal cord injury—a pilot randomized cross-over trial. J Spinal Cord Med. 2017;41(4):397–406. https://doi.org/10.1080/10790268.2017.1390930.
Jauregui JJ, Cherian JJ, Gwam CU, et al. A meta-analysis of transcutaneous electrical nerve stimulation for chronic low back pain. Surg Technol Int. 2016;28:296–302.
Gibson W, Wand BM, Meads C, Catley MJ, O’Connell NE. Transcutaneous electrical nerve stimulation (TENS) for chronic pain—an overview of Cochrane Reviews. Cochrane Database of Syst Rev. 2019. https://doi.org/10.1002/14651858.CD011890.pub3.
Wu LC, Weng PW, Chen CH, Huang YY, Tsuang YH, Chiang CJ. Literature review and meta-analysis of transcutaneous electrical nerve stimulation in treating chronic back pain. Reg Anesth Pain Med. 2018;43(4):425–33. https://doi.org/10.1097/aap.0000000000000740.
Zhu Y, Feng Y, Peng L. Effect of transcutaneous electrical nerve stimulation for pain control after total knee arthroplasty: a systematic review and meta-analysis. J Rehabil Med. 2017;49(9):700–4. https://doi.org/10.2340/16501977-2273.
Williamson TK, Rodriguez HC, Gonzaba A, Poddar N, Norwood SM, Gupta A. H-Wave® device stimulation: a critical review. J Pers Med. 2021;11(11):1134. https://doi.org/10.3390/jpm11111134.
Smith TL, Callahan MF, Blum K, Dinubile NA, Chen TJH, Waite RL. H-Wave® effects on blood flow and angiogenesis in longitudinal studies in rats. J Surg Orthop Adv. 2011;20(4):255–9.
Julka IS, Alvaro M, Kumar D. Beneficial effects of electrical stimulation on neuropathic symptoms in diabetes patients. J Foot Ankle Surg. 1998;37(3):191–4.
Williamson TK, Rodriguez HC, Han D, Norwood SM, Gupta A. Job-related performance and quality of life benefits in first responders given access to H-Wave® device stimulation: a retrospective cohort study. J Pers Med. 2022;12(10):1674. https://doi.org/10.3390/jpm12101674.
Trinh A, Williamson TK, Han D, Hazlewood JE, Norwood SM, Gupta A. Clinical and quality of life benefits for end-stage workers’ compensation chronic pain claimants following H-Wave® device stimulation: a retrospective observational study with mean 2-year follow-up. J Clin Med. 2023;12(3):1148. https://doi.org/10.3390/jcm12031148.
Dias LV, Cordeiro MA, Schmidt de Sales R, et al. Immediate analgesic effect of transcutaneous electrical nerve stimulation (TENS) and interferential current (IFC) on chronic low back pain: Randomised placebo-controlled trial. J Bodywork Mov Ther. 2012;27:181–90. https://doi.org/10.1016/j.jbmt.2021.03.005.
Menezes MA, Mendonça Araújo F, Lima LV, Souza TAS, Carvalho EAN, Melo DJ. Interferential current intensity influences the hypoalgesic response in healthy subjects under mechanically-induced pain: a randomized controlled trial. Physiother Theory Pract. 2022;3:1–13. https://doi.org/10.1080/09593985.2022.2068095.
Werners R, Pynsent PB, Bulstrode CJ. Randomized trial comparing interferential therapy with motorized lumbar traction and massage in the management of low back pain in a primary care setting. Spine. 1999;24(15):1579–84.
Jarit GJ, Mohr KJ, Waller R, Glousman RE. The effects of home interferential therapy on post-operative pain, edema, and range of motion of the knee. Clin J Sport Med. 2003;13(1):16–20.
Fuentes JP, Armijo Olivo S, Magee DJ, Gross DP. Effectiveness of interferential current therapy in the management of musculoskeletal pain: a systematic review and meta-analysis. Phys Ther. 2010;90(9):1219–38. https://doi.org/10.2522/ptj.20090335.
Zeng C, Li H, Deng Z, Yang Y, Zhang Y, Lei GH. Electrical stimulation for pain relief in knee osteoarthritis: systematic review and network meta-analysis. Osteoarth Cartil. 2015;23(2):189–202. https://doi.org/10.1016/j.joca.2014.11.014.
Almeida CC, Silva VZMD, Júnior GC, Liebano RE, Durigan JLQ. Transcutaneous electrical nerve stimulation and interferential current demonstrate similar effects in relieving acute and chronic pain: a systematic review with meta-analysis. Braz J Phys Ther. 2018;22(5):347–54. https://doi.org/10.1016/j.bjpt.2017.12.005. (Epub 2018 Feb 2).
Razzano C, Izzo R, Savastano R, Colantuoni C, Carbone S. Noninvasive interactive neurostimulation therapy for the treatment of low-grade lateral ankle sprain in the professional contact sport athlete improves the short-term recovery and return to sport: a randomized controlled trial. J Foot Ankle Surg. 2019;58(3):441–6. https://doi.org/10.1053/j.jfas.2018.09.009. (Epub 2019 Mar 23).
Razzano C, Carbone S, Mangone M, Iannotta MR, Battaglia A, Santilli V. Treatment of chronic plantar fasciitis with noninvasive interactive neurostimulation: a prospective randomized controlled study. J Foot Ankle Surg. 2017;56(4):768–72. https://doi.org/10.1053/j.jfas.2017.02.015.
Nigam AK, Taylor DM, Valeyeva Z. Non-invasive interactive neurostimulation (InterX™) reduces acute pain in patients following total knee replacement surgery: a randomised, controlled trial. J Orthop Surg Res. 2011;24(6):45. https://doi.org/10.1186/1749-799X-6-45.
Li X, Zhang L, Gu S, et al. Comparative effectiveness of extracorporeal shock wave, ultrasound, low-level laser therapy, noninvasive interactive neurostimulation, and pulsed radiofrequency treatment for treating plantar fasciitis: a systematic review and network meta-analysis. Medicine (Baltimore). 2018;97(43): e12819. https://doi.org/10.1097/MD.0000000000012819.
Grazzi L, Toppo C, D’Amico D, et al. Non-pharmacological approaches to headaches: non-invasive neuromodulation, nutraceuticals, and behavioral approaches. Int J Environ Res Public Health. 2021;18(4):1503. https://doi.org/10.3390/ijerph18041503.
Negm A, Lorbergs A, Macintyre NJ. Efficacy of low frequency pulsed subsensory threshold electrical stimulation vs placebo on pain and physical function in people with knee osteoarthritis: systematic review with meta-analysis. Osteoarth Cartil. 2013;21(9):1281–9. https://doi.org/10.1016/j.joca.2013.06.015.
Fary RE, Carroll GJ, Briffa TG, Briffa NK. The effectiveness of pulsed electrical stimulation in the management of osteoarthritis of the knee: results of a double-blind, randomized, placebo-controlled, repeated-measures trial. Arthritis Rheum. 2011;63(5):1333–42. https://doi.org/10.1002/art.30258.
Li S, Yu B, Zhou D, He C, Zhuo Q, Hulme JM. Electromagnetic fields for treating osteoarthritis. Cochrane Database Syst Rev. 2013. https://doi.org/10.1002/14651858.CD003523.pub2.
Shamliyan TA, Wang SY, Olson-Kellogg B, Kane RL. Physical Therapy Interventions for Knee Pain Secondary to Osteoarthritis [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2012 Nov. Report No.: 12(13)-EHC115-EF.
Jaermann T, Suter F, Osterwalder D, Luechinger R. Measurement and analysis of electromagnetic fields of pulsed magnetic field therapy systems for private use. J Radiol Prot. 2011;31(1):107–16. https://doi.org/10.1088/0952-4746/31/1/007.
Kwon DR, Kim J, Kim Y, et al. Short-term microcurrent electrical neuromuscular stimulation to improve muscle function in the elderly: a randomized, double-blinded, sham-controlled clinical trial. Medicine. 2017;96(26): e7407. https://doi.org/10.1097/MD.0000000000007407.
Weiner DK, Perera S, Rudy TE, Glick RM, Shenoy S, Delitto A. Efficacy of percutaneous electrical nerve stimulation and therapeutic exercise for older adults with chronic low back pain: a randomized controlled trial. Pain. 2008;140(2):344–57. https://doi.org/10.1016/j.pain.2008.09.005.
He DP, Zhang J, Bai ZF. Percutaneous electrical nerve stimulation for chronic knee pain: a randomized, sham-controlled trial. Altern Ther Health Med. 2019;25(2):30–4.
Miao Q, Qiang JH, Jin YL. Effectiveness of percutaneous neuromuscular electrical stimulation for neck pain relief in patients with cervical spondylosis. Medicine. 2018;97(26): e11080. https://doi.org/10.1097/MD.0000000000011080.
Li H, Xu QR. Effect of percutaneous electrical nerve stimulation for the treatment of migraine. Medicine. 2017;96(39): e8108. https://doi.org/10.1097/MD.0000000000008108.
Yokoyama M, Sun X, Oku S, et al. Comparison of percutaneous electrical nerve stimulation with transcutaneous electrical nerve stimulation for long-term pain relief in patients with chronic low back pain. Anesth Analg. 2004;98(6):1552–6.
Hofmeister M, Memedovich A, Brown S, et al. Effectiveness of neurostimulation technologies for the management of chronic pain: a systematic review. Neuromodulation. 2020;23(2):150–7. https://doi.org/10.1111/ner.13020.
Banks GP, Winfree CJ. Evolving techniques and indications in peripheral nerve stimulation for pain. Neurosurg Clin N Am. 2019;30(2):265–73. https://doi.org/10.1016/j.nec.2018.12.011.
Xu J, Sun Z, Wu J, Rana M, Garza J, Zhu AC, Chakravarthy KV, Abd-Elsayed A, Rosenquist E, Basi H, Christo P, Cheng J. Peripheral nerve stimulation in pain management: a systematic review. Pain Physician. 2021;24(2):E131–52.
Knotkova H. Evidence-based review of transcranial direct current stimulation (tdcs) for chronic pain syndromes. Brain Stimul. 2017;10(2):403. https://doi.org/10.1016/j.brs.2017.01.196.
Lefaucheur JP, Antal A, Ayache SS, et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin Neurophysiol. 2017;128(1):56–92. https://doi.org/10.1016/j.clinph.2016.10.087.
Elsner B, Kwakkel G, Kugler J, Mehrholz J. Transcranial direct current stimulation (tdcs) for improving capacity in activities and arm function after stroke: A network meta-analysis of Randomised Controlled Trials. J NeuroEng Rehabil. 2017. https://doi.org/10.1186/s12984-017-0301-7.
Lloyd DM, Wittkopf PG, Arendsen LJ, Jones AKP. Is transcranial direct current stimulation (tDCS) effective for the treatment of pain in fibromyalgia? A systematic review and meta-analysis. J Pain. 2020;21(11–12):1085–100. https://doi.org/10.1016/j.jpain.2020.01.003.
Alwardat M, Pisani A, Etoom M, et al. Is transcranial direct current stimulation (tDCS) effective for chronic low back pain? A systematic review and meta-analysis. J Neural Transm. 2020;127:1257–70. https://doi.org/10.1007/s00702-020-02223-w.
O’Connell NE, Wand BM, Marston L, Spencer S, Desouza LH. Non-invasive brain stimulation techniques for chronic pain. Cochrane Database Syst Rev. 2014. https://doi.org/10.1002/14651858.CD008208.pub3.
Boldt I, Eriks-Hoogland I, Brinkhof MW, de Bie R, Joggi D, von Elm E. Non-pharmacological interventions for chronic pain in people with spinal cord injury. Cochrane Database Syst Rev. 2014. https://doi.org/10.1002/14651858.CD009177.pub2.
Horvath JC, Forte JD, Carter O. Evidence that transcranial direct current stimulation (tDCS) generates little-to-no reliable neurophysiologic effect beyond MEP amplitude modulation in healthy human subjects: a systematic review. Neuropsychologia. 2015;66:213–36. https://doi.org/10.1016/j.neuropsychologia.2014.11.021.
Funding
No funding or sponsorship was received for this study or publication of this article. The journal’s rapid service fee was funded by Electronic Waveform Lab, Inc. (Huntington Beach, CA, USA).
Author information
Authors and Affiliations
Contributions
Ashim Gupta conceptualized the study. The first draft of the manuscript was written by Christian B. Allen and Tyler K. Williamson. Stepehen M. Norwood and Ashim Gupta commented on the previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
Stephen M. Norwood and Ashim Gupta are consultants for Electronic Waveform Lab, Inc. (Huntington Beach, CA, USA). The other authors declare that they have no competing interests.
Ethical Approval
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Allen, C.B., Williamson, T.K., Norwood, S.M. et al. Do Electrical Stimulation Devices Reduce Pain and Improve Function?—A Comparative Review. Pain Ther 12, 1339–1354 (2023). https://doi.org/10.1007/s40122-023-00554-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40122-023-00554-6