Abstract
Introduction
To assess the 1-year outcomes of vertically fixated posterior chamber phakic intraocular lens implantation through a superior corneal incision.
Methods
This pilot study comprised 71 eyes of 43 consecutive patients undergoing vertically fixated implantable collamer lens (ICL) implantation through a superior corneal incision to correct moderate to high myopia and myopic astigmatism. We prospectively determined the safety, efficacy, predictability, stability, and adverse events preoperatively, and at 1 week and 1, 3, and 12 months postoperatively.
Results
The mean follow-up period was 10.4 ± 5.4 months. Uncorrected and corrected visual acuity were −0.20 ± 0.10 and −0.25 ± 0.07 logMAR, respectively, at 1 year postoperatively. At 1 year postoperatively, 98% and 100% of eyes were within 0.5 and 1.0 D, respectively, of the targeted correction. A nonsignificant change in manifest refraction of −0.01 ± 0.08 D occurred from 1 week to 1 year. The manifest astigmatism decreased significantly, from 0.69 ± 0.73 D preoperatively to 0.21 ± 0.27 D at 1 year postoperatively (Mann–Whitney U test, p < 0.001). No vision-threatening complications occurred at any time in this series.
Conclusions
According to our experience, the vertically fixated ICL through a superior incision achieved good results, without significant complications. Considering that younger patients requiring ICL surgery tend to have with-the-rule astigmatism, this surgical technique may be a viable option for reducing astigmatism without using toric ICLs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Why carry out this study? |
A superior corneal incision is beneficial for reducing astigmatism especially when using non-toric implantable collamer lenses (ICLs), and subsequent vertical ICL fixation may be surgically easier than horizontal fixation. Nevertheless, detailed outcomes of this new approach have not been investigated so far. |
What was learned from the study? |
Vertically fixated ICL implantation through a superior corneal incision achieved good results for the correction of moderate to high myopia and myopic astigmatism, with a significant reduction in refractive astigmatism. The results suggest that this promising approach is a viable option for such refractive candidates, especially when using the non-toric ICL models. |
Introduction
The EVO Visian implantable collamer lens (EVO Visian ICL™, STAAR Surgical, Monrovia, CA, USA), a posterior chamber phakic intraocular lens with central port technology, has been established as a long-term safe and effective means to correct moderate to high ametropia [1,2,3,4] and has also been shown to be more effective in reducing significant postoperative complications such as cataract formation than the conventional ICL without a central port [1,2,3,4,5], possibly resulting from the improved circulation of the aqueous humor to the anterior surface of the crystalline lens [6, 7]. The ICL is generally fixated to the horizontal plane through a temporal corneal incision according to the manufacturer’s recommendations. However, based on our findings that ICL implantation induces corneal astigmatism by an approximately 0.5 diopter (D) with-the-rule (WTR) shift [8, 9], a superior corneal incision may be clinically beneficial to reduce astigmatism, and subsequent vertical ICL fixation may be surgically easier than horizontal fixation when using a superior incision because ICL rotation is no longer required. Nevertheless, the clinical outcomes of this new surgical technique remain unknown. Therefore, an investigation of the outcomes of this procedure may give us essential insights into further improving good unaided outcomes of non-toric ICL surgery. The goal of this pilot study is to prospectively investigate the 1-year outcomes of vertically fixated ICL implantation through a superior corneal incision in patients with moderate to high ametropia. To the best of our knowledge, this is the first case series to assess the detailed outcomes of vertically fixated ICL implantation through a superior incision.
Methods
Study Population
The protocol was registered with the University Hospital Medical Information Network Clinical Trial Registry (000046332). This prospective study comprised consecutive patients who underwent implantation of the posterior chamber phakic intraocular lens with a 0.36-mm central port (EVO-ICL, KS-AP™, V4c and V5 models, STAAR Surgical) for the correction of moderate to high myopia and myopic astigmatism at Kitasato University Hospital from January to December 2020, and who completed at least a 3-month routine examination follow-up.
Inclusion and Exclusion Criteria
Inclusion criteria were 20 ≤ age < 50 years, stable refraction, moderate to high myopia and astigmatism, and no history of trauma or ocular surgery. Exclusion criteria were eyes with concomitant corneal disease such as keratoconus, pellucid marginal degeneration, irregular corneal astigmatism, or severe dry eye, or eyes with cataracts, glaucoma, uveitis, other concomitant eye diseases, or intentional undercorrection or monovision for middle-aged patients. We selected a superior corneal incision when conducting non-toric ICL surgery in eyes with refractive astigmatism of 0.75 D or more, or when performing non-toric or toric ICL surgery in eyes in which the predicted vertically fixated ICL vault was close to 500 µm using the KS formula [10].
The study was approved by the Institutional Review Board at Kitasato University Hospital (B21-196) and followed the tenets of the Declaration of Helsinki. Written informed consent for ICL surgery was obtained from all patients after explaining the possible consequences of the study.
Outcome Measures
Preoperatively, and 1 week and 1, 3, and 12 months postoperatively, we obtained the following parameters: the logarithm of the minimal angle of resolution (logMAR) of uncorrected distance visual acuity (UDVA) and corrected distance visual acuity (CDVA), the manifest spherical equivalent, the manifest cylinder, the intraocular pressure (IOP) using a non-contact tonometer, the endothelial cell density (ECD; preoperatively and 3 months and 1 year postoperatively) using a non-contact specular microscope, and the vault between the anterior surface of the crystalline lens and the posterior surface of the ICL using anterior segment optical coherence tomography (AS-OCT), in addition to routinely performed ophthalmic examinations. All available visit data were collected and grouped according to the closest time point. If more than one visit was available within a given grouping, we used the visit data most comparable to the given time point for this analysis.
Lens Power Calculation and Size Selection
We determined the ICL size (12.1, 12.6, 13.2, and 13.7 mm) based on the KS prediction formula using AS-OCT (CASIA, Tomey Corporation, Aichi, Japan). We also selected the ICL power using an online calculation and ordering system provided by the manufacturer based on a modified vertex formula [11, 12]. We chose the toric model ICL in eyes with manifest astigmatism of 1 D or more and the non-toric model ICL in eyes with less than 1 D.
Surgical Procedures
We described the details of the surgical procedures in our previous reports [4, 13,14,15,16]. In brief, on the day of surgery, dilating and topical anesthetic agents were applied. A model V4c or V5 ICL was implanted by one experienced surgeon (K.K.) through a 3-mm superior clear corneal incision after injection of a viscosurgical substance into the anterior chamber. Next, the ICL was inserted into the posterior chamber, the viscosurgical substance was replaced with a balanced salt solution, and a miotic agent was administered. We utilized topical antibiotic and steroidal medications four times daily for 1 week, reducing the dose gradually.
Statistical Analysis
We first checked the normality of all data samples using the Shapiro–Wilk test. Then, since no data fulfilled the criteria for normal distribution, we applied the Mann–Whitney U test to compare the pre- and the 1-year postsurgical data. We also used the Kruskal–Wallis test to evaluate the time course of the changes, with the Steel–Dwass test employed for multiple comparisons. Unless otherwise indicated, the results are expressed as mean ± standard deviation, and a value of p < 0.05 was deemed statistically significant.
Results
Study Population
A total of 71 eyes of 43 patients (20 men and 23 women) met the inclusion criteria of this study. Table 1 shows the preoperative baseline demographics of the study population. The number of eyes examined was 71 (100%), 71 (100%), 71 (100%), and 50 (70%) at 1 week and 1, 3, and 12 months postoperatively, respectively. The mean follow-up period was 10.4 ± 5.4 months. Non-toric and toric ICLs were used in 51 (72%) and 20 (28%) eyes, respectively.
Safety and Efficacy Outcomes
Standard refractive surgery graphs are shown in Fig. 1. The corresponding graphs when using non-toric and toric models are presented in Supplementary Materials 1 and 2, respectively. At 1 week and 1, 3, and 12 months postoperatively, 100%, 100%, 97%, and 98% of eyes, and 96%, 93%, 96%, and 92% of eyes had UDVA of 20/20 and 20/16 or better, respectively (Fig. 1a). LogMAR UDVA was −0.22 ± 0.09, −0.20 ± 0.10, −0.21 ± 0.09, and −0.20 ± 0.10 at 1 week and 1, 3, and 12 months postoperatively, respectively (Kruskal–Wallis test, p = 0.681). We found a significant difference between preoperative UDVA and 1-year postoperative UDVA (Mann–Whitney U test, p < 0.001). The efficacy index (mean postoperative UDVA / mean preoperative CDVA) was 0.98 ± 0.19 at 1 year postoperatively.
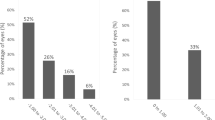
a Cumulative percentages of eyes attaining specified cumulative levels of uncorrected distance visual acuity (UDVA). b Changes in corrected distance visual acuity (CDVA). c A scatter plot of the attempted versus the achieved manifest spherical equivalent correction. d Distribution of spherical equivalent refractive accuracy. e Distribution of refractive astigmatism, and f time course of changes in manifest spherical equivalent
At 1 year postoperatively, 29 eyes (58%) showed no change in CDVA, 18 eyes (36%) gained one line, while three eyes (6%) lost one line, but no eyes had lost more than one line (Fig. 1b). LogMAR CDVA was −0.27 ± 0.06, −0.25 ± 0.07, −0.26 ± 0.06, and −0.25 ± 0.07 at 1 week and 1, 3, and 12 months postoperatively, respectively (p = 0.470). We found a significant difference between preoperative CDVA and 1-year postoperative CDVA (p = 0.016). The safety index (mean postoperative CDVA / mean preoperative CDVA) was 1.10 ± 0.18 at 1 year postoperatively.
Predictability and Stability Outcomes
Scatter plots of the attempted versus achieved manifest spherical equivalent correction, distribution of spherical equivalent refractive accuracy, and distribution of refractive astigmatism are shown in Fig. 1c, d, e, respectively. At 1 week and 1, 3, and 12 months postoperatively, 94%, 94%, 94%, and 90% of eyes, and 100%, 100%, 100%, and 98% of eyes were within ± 0.25 and 0.5 D, respectively, of the attempted spherical equivalent correction. All eyes were within ± 1.0 D at all time points.
The time-course change in the manifest spherical equivalent is shown in Fig. 1f. At 1 week and 1, 3, and 12 months postoperatively, the manifest spherical equivalent was −0.04 ± 0.15, −0.05 ± 0.16, −0.06 ± 0.16, and −0.08 ± 0.18 D, respectively (p = 0.685). At 1 week and 1, 3, and 12 months postoperatively, the manifest cylinder was 0.23 ± 0.31, 0.19 ± 0.28, 0.20 ± 0.27, and 0.21 ± 0.27 D, respectively (p = 0.887). The manifest astigmatism decreased significantly, from 0.69 ± 0.73 D preoperatively to 0.21 ± 0.27 D at 1 year postoperatively (Mann–Whitney U test, p < 0.001). It decreased significantly from 0.40 ± 0.44 D preoperatively to 0.22 ± 0.27 D postoperatively when using the non-toric models (p = 0.049), and from 1.43 ± 0.83 D preoperatively to 0.00 ± 0.00 D postoperatively when using the toric ICL models (p < 0.001).
Intraocular Pressure
The IOP was 14.2 ± 3.2, 13.0 ± 2.5, 13.4 ± 2.7, and 13.1 ± 2.8 mmHg at 1 week and 1, 3, and 12 months postoperatively, respectively (p = 0.052). No significant increase in IOP (> 25 mmHg) occurred in any case throughout the observation period.
Endothelial Cell Density
No significant change in ECD was observed; density decreased from 2800 ± 218 cells/mm2 preoperatively to 2762 ± 228 cells/mm2 at 3 months and 2740 ± 232 cells/mm2 at 1 year postoperatively (p = 0.462). The mean percentage of ECD loss was 1.6 ± 6.5% at 1 year postoperatively.
Lens Vault
The ICL vault was 456 ± 197, 465 ± 192, 435 ± 193, and 382 ± 171 µm at 1 week and 1, 3, and 12 months postoperatively, respectively (p = 0.075). Figure 2 shows the postoperative distribution of the ICL vault. No excessive-low vault (< 50 µm) or excessive-high vault (> 1000 µm) requiring ICL exchange was found in any case throughout the observation period.
Secondary Surgeries/Adverse Events
There were no obvious intraoperative complications, such as an upside-down ICL insertion or traumatic cataract formation, and all surgeries were uneventful in this study cohort. Complaints of mild symptomatic glare or halo were reported in four eyes at 3 months postoperatively, but these symptoms gradually recovered over time without any additional interventions. Otherwise, there were no vision-threatening complications, such as symptomatic or asymptomatic cataract formation, pigment dispersion glaucoma, pupillary block, retinal detachment, or significant endothelial cell loss (≥ 15%), throughout the follow-up period in any eye.
Discussion
In the present study, our 1-year findings demonstrated that vertically fixated ICL implantation through a superior corneal incision achieved good results in terms of the safety, efficacy, predictability, and stability for the correction of moderate to high myopia and myopic astigmatism and that no serious intraoperative or postoperative complication occurred in any case throughout the 1-year observation period. These results suggest that this promising approach represents a viable and feasible option for such refractive candidates, especially when using non-toric ICL models.
A superior corneal incision may be beneficial for reducing the amplitude of astigmatism and obtaining good unaided vision, especially when using non-toric ICLs, since most younger refractive candidates have WTR astigmatism. Our results also showed that the manifest astigmatism was significantly decreased even after non-toric ICL implantation, although the amplitude of reduction in astigmatism was small (approximately 0.25 D). This is in agreement with our recent study in which standard ICL surgery resulted in a change in mean arithmetic surgically induced astigmatism (SIA) of approximately 0.5 D in both the temporal and the superior incision groups [8, 9], and the magnitude of the centroid SIA decreased to approximately 50% of the mean arithmetic SIA in the temporal incision group, but approximately 80% in the superior incision group [9], implying that the effect of a 3-mm incision in the former group was more prominent than that in the latter group. Hence, we believe that this promising approach is clinically beneficial for the refractive surgeon during preoperative planning and for the ICL manufacturer to improve the astigmatic outcomes of non-toric ICL implantation. It should be emphasized that the SIA of ICL implantation is small in amount but not necessarily negligible, because ICL implantation is one of the refractive surgeries that aim to provide the best correction not only of spherical errors but also of cylindrical errors.
Predicting the optimal ICL vault is essential, especially when considering the safety of ICL surgery. It has been demonstrated that the ideal ICL vault is identical to the central corneal thickness (approximately 500 µm) [10, 15, 16]. There are still ongoing concerns about selecting the optimal ICL size, even when we employ the recommended ICL sizing nomogram provided by the ICL manufacturer, based on the white-to-white distance and the anterior chamber depth measurements. Angle closure and subsequent intraocular pressure rise can occur when selecting a larger ICL size, whereas cataract formation or toric ICL rotation can arise when selecting a smaller ICL size. We determined the optimal ICL size based on the KS formula (version 3) using AS-OCT. We found no excessive-low vault or excessive-high vault requiring ICL exchange in any eye, but the postoperative ICL vault tended to be slightly low in this study population. We recently reported that a machine learning technique has advantages over conventional prediction nomograms for obtaining the ideal ICL vault and subsequent optimal ICL size [16, 17]. However, it has been shown that the vertical sulcus-to-sulcus diameter is longer than the horizontal sulcus-to-sulcus diameter [18]. Accordingly, it is reasonable that the ICL vault in a vertical position in this study was smaller than that in a horizontal position in previous studies. Indeed, it was recently demonstrated that the horizontally fixated ICL vault was significantly higher than the vertically fixated ICL vault [19]. We assume that further customization of these preexisting formulas is still necessary to select the optimal ICL size in a vertical position.
This study suffers from several limitations. Firstly, as this was a pilot study, the sample size was relatively small, with a median follow-up of approximately 1 year. Therefore, a larger patient sample with a long-term observation period is still necessary to confirm our preliminary results. Secondly, manifest astigmatism does not necessarily reflect the precise refractive status of the eye, since it can be affected by the accommodation. However, in our previous report, we confirmed that corneal astigmatism was also significantly decreased after ICL surgery through a superior corneal incision [9]. Thirdly, we included both eyes of the same patient undergoing ICL implantation, although only one eye per patient should be used for statistical analysis. We found no significant inter-eye correlations of logMAR UDVA, logMAR CDVA, the manifest cylinder, the IOP, or the vault at 1 year postoperatively, although correlations were observed for the manifest spherical equivalent and the ECD, possibly due to the small sample size. We also confirmed similar outcomes when only one eye was selected randomly from each patient, as shown in Supplementary Material 3.
Conclusion
In summary, our pilot study demonstrated that a vertically fixated ICL through a superior corneal incision achieved good results in terms of safety, efficacy, predictability, and stability for the correction of moderate to high myopia and myopic astigmatism, without significant complications during the 1-year observation period, and that a significant reduction in astigmatism was found not only after toric ICL implantation but also after non-toric ICL implantation. These findings indicate that this new approach is a viable surgical option for correcting moderate to high ametropia, especially for the clinical application of non-toric ICL surgery. A longer, careful follow-up in a large cohort of patients is still necessary to confirm our preliminary findings.
Change history
26 May 2023
A Correction to this paper has been published: https://doi.org/10.1007/s40123-023-00732-x
References
Shimizu K, Kamiya K, Igarashi A, Kobashi H. Long-term comparison of posterior chamber phakic intraocular lens with and without a central hole (hole ICL and conventional ICL) implantation for moderate to high myopia and myopic astigmatism: consort-compliant article. Medicine (Baltimore). 2016;95: e3270.
Alfonso JF, Fernández-Vega-Cueto L, Alfonso-Bartolozzi B, Montés-Micó R, Fernández-Vega L. Five-year follow-up of correction of myopia: posterior chamber phakic intraocular lens with a central port design. J Refract Surg. 2019;35:169–76.
Fernández-Vega-Cueto L, Alfonso-Bartolozzi B, Lisa C, Madrid-Costa D, Alfonso JF. Seven-year follow-up of posterior chamber phakic intraocular lens with central port design. Eye Vis (Lond). 2021;8:23.
Kamiya K, Shimizu K, Takahashi M, Ando W, Hayakawa H, Shoji N. Eight-year outcomes of implantation of posterior chamber phakic intraocular lens with a central port for moderate to high ametropia. Front Med (Lausanne). 2021. https://doi.org/10.3389/fmed.2021.799078.
Packer M. The implantable collamer lens with a central port: review of the literature. Clin Ophthalmol. 2018;12:2427–38.
Fujisawa K, Shimizu K, Uga S, Suzuki M, Nagano K, Murakami Y, Goseki H. Changes in the crystalline lens resulting from insertion of a phakic IOL(ICL) into the porcine eye. Graefe’s Arch Clin Exp Ophthalmol. 2007;245:114–22.
Shiratani T, Shimizu K, Fujisawa K, Uga S, Nagano K, Murakami Y. Crystalline lens changes in porcine eyes with implanted phakic IOL (ICL) with a central hole. Graefes Arch Clin Exp Ophthalmol. 2008;246:719–28.
Kamiya K, Shimizu K, Aizawa D, Igarashi A, Komatsu M. Surgically induced astigmatism after posterior chamber phakic intraocular lens implantation. Br J Ophthalmol. 2009;93(12):1648–51.
Kamiya K, Ando W, Takahashi M, Shoji N. Comparison of magnitude and summated vector mean of surgically induced astigmatism vector according to incision site after phakic intraocular lens implantation. Eye Vis (Lond). 2021;8(1):32.
Igarashi A, Shimizu K, Kato S, Kamiya K. Predictability of the vault after posterior chamber phakic intraocular lens implantation using anterior segment optical coherence tomography. J Cataract Refract Surg. 2019;45(8):1099–104.
Sanders DR, Vukich JA, Gaston M. Implantable contact lens in treatment of myopia study group. U.S. Food and Drug Administration clinical trial of the Implantable Contact Lens for moderate to high myopia. Ophthalmology. 2003;110:255–66.
Sanders DR, Doney K, Poco M, ICL in Treatment of Myopia Study Group. United States Food and Drug Administration clinical trial of the Implantable Collamer Lens (ICL) for moderate to high myopia: three-year follow-up. Ophthalmology. 2004;111:1683–92.
Shimizu K, Kamiya K, Igarashi A, Shiratani T. Early clinical outcomes of implantation of posterior chamber phakic intraocular lens with a central hole (Hole ICL) for moderate to high myopia. Br J Ophthalmol. 2012;96:409–12.
Kamiya K, Shimizu K, Igarashi A, Kitazawa Y, Kojima T, Nakamura T, Oka Y, Matsumoto R. Posterior chamber phakic intraocular lens implantation: comparative, multicentre study in 351 eyes with low-to-moderate or high myopia. Br J Ophthalmol. 2018;102:177–81.
Ando W, Kamiya K, Hayakawa H, Takahashi M, Shoji N. Comparison of phakic intraocular lens vault using conventional nomogram and prediction formulas. J Clin Med. 2020;9(12):4090.
Kamiya K, Ryu IH, Yoo TK, Kim JS, Lee IS, Kim JK, Ando W, Shoji N, Yamauchi T, Tabuchi H. Prediction of phakic intraocular lens vault using machine learning of anterior segment optical coherence tomography metrics. Am J Ophthalmol. 2021;226:90–9.
Kang EM, Ryu IH, Lee G, Kim JK, Lee IS, Jeon GH, Song H, Kamiya K, Yoo TK. Development of a web-based ensemble machine learning application to select the optimal size of posterior chamber phakic intraocular lens. Transl Vis Sci Technol. 2021;10(6):5.
Oh J, Shin HH, Kim JH, Kim HM, Song JS. Direct measurement of the ciliary sulcus diameter by 35-megahertz ultrasound biomicroscopy. Ophthalmology. 2007;114(9):1685–8.
Kim BK, Chung YT. Clinical results of Visian implantable collamer lens implantation according to various sizes and implantation angles. Eur J Ophthalmol. 2021. https://doi.org/10.1177/11206721211033468.
Acknowledgements
We thank all study participants for their involvement in the study.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. No funding was received for the publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Kazutaka Kamiya, Wakako Ando, Hideki Hayakawa, and Nobuyuki Shoji were involved in the design and conduct of the study, Kazutaka Kamiya, Wakako Ando, Hideki Hayakawa, and Satoshi Gotoda were involved in collection, management, analysis, and interpretation of data, all authors were involved in preparation, review, and final approval of the manuscript.
Disclosures
Kazutaka Kamiya, Wakako Ando, Hideki Hayakawa, Satoshi Gotoda and Nobuyuki Shoji declare that they have no conflict of interest related to this work.
Compliance with Ethics Guidelines
This study was approved by the Institutional Review Board at Kitasato University Hospital (B21-196) and followed the tenets of the Declaration of Helsinki. Written informed consent for ICL surgery was obtained from all patients after explaining the possible consequences of the study.
Data Availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original online version of this article was revised to correct some data in Abstract and Results section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Kamiya, K., Ando, W., Hayakawa, H. et al. Vertically Fixated Posterior Chamber Phakic Intraocular Lens Implantation Through a Superior Corneal Incision. Ophthalmol Ther 11, 701–710 (2022). https://doi.org/10.1007/s40123-022-00470-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-022-00470-6