Abstract
Introduction
The aim of this study was to investigate the safety and efficacy of a single injection of intravitreal faricimab (IVF) in patients with neovascular age-related macular degeneration (nAMD) who had a prior treatment history.
Methods
A retrospective analysis was conducted on a consecutive cohort of 80 eyes of 75 patients with nAMD who had a prior history of treatment with an injection of anti-vascular endothelial growth factor. Best-corrected visual acuity (BCVA), central subfield thickness (CST), and central choroidal thickness (CCT) were compared before the initial IVF injection and after a treatment interval matching the previous duration.
Results
Central choroidal thickness decreased significantly following the IVF injection, but there was no significant change in BCVA or CST. Mean (± standard deviation) BCVA changed from 0.34 ± 0.37 to 0.36 ± 0.40 (P = 0.29), CST changed from 242 ± 72 to 242 ± 82 µm (P = 0.99), and CCT changed from 189 ± 98 to 179 ± 97 µm (P < 0.0001). When the changes were evaluated according to the previous anti-VEGF agent administered, CCT was found to be significantly decreased by 8.7 ± 2.5 µm (P < 0.0001) in eyes previously treated with brolucizumab and by 13.1 ± 3.6 µm (P < 0.0001) in eyes previously treated with aflibercept. No adverse events were observed during the study period.
Conclusion
Intravitreal faricimab injection is a safe and effective treatment for nAMD in terms of short-term outcomes. Further long-term study is necessary.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Why carry out this study? |
Faricimab, a novel agent which targets both vascular endothelial growth factor (VEGF) and angiopoietin-2, was approved by the US Food and Drug Administration in 2022 based on the results of clinical trials showing that this agent may be more effective than aflibercept for treatment-naïve neovascular age-related macular degeneration (nAMD), even with long treatment intervals. |
Although previous reports have demonstrated that brolucizumab is superior to aflibercept for treating lesions in the sub-retinal pigment epithelium and for reducing choroidal thickness, there is limited information on the efficacy and safety of switching to other anti-VEGF agents. |
What was learned from this study? |
This study evaluated the efficacy and safety of intravitreal injection of faricimab in patients with nAMD and a prior history of treatment with aflibercept and brolucizumab. |
The findings suggest that a single intravitreal injection of faricimab has favorable efficacy and safety. |
Introduction
In 2022, a new bispecific antibody called faricimab, which targets both vascular endothelial growth factor (VEGF) and angiopoietin (Ang)-2, was approved by the US Food and Drug Administration and regulatory agencies in various countries [1, 2]. The results of two multicenter phase III clinical trials, TENAYA and LUCENE, showed that patients with treatment-naïve neovascular age-related macular degeneration (nAMD) who received intravitreal injection of faricimab (faricimab [IVF] injection) achieved best-corrected visual acuity (BCVA) that was non-inferior to that in patients treated with intravitreal injection of aflibercept (IVA injection). These results highlighted the potential effectiveness of IVF as a treatment for patients with nAMD, particularly in terms of maintaining BCVA outcomes, even with longer treatment intervals [3]. Interestingly, an analysis of the Japanese and Asian subgroups showed that the proportion of patients who were able to maintain extended fixed treatment intervals of 16 weeks was higher in the TENAYA study than in the pooled global TENAYA/LUCENE trials (66% vs. 45%, and 60% vs. 44%, respectively) [4, 5]. Short-term studies from Japan have also reported improved BCVA and retinal morphology during the loading phase of IVF treatment in patients with treatment-naïve nAMD [6, 7].
However, despite the favorable results observed with IVF injection in nAMD, several challenges still need to be addressed. First, the two clinical trials and previous studies focused on patients with treatment-naïve nAMD [3, 6, 7], so their results do not apply to patients with previously treated nAMD. Also, the previous trials only compared IVF injection and IVA injection, and there is a dearth of comparisons between IVF and intravitreal injection of brolucizumab (IVB injection). Another area where evidence is lacking is the efficacy and safety of switching treatments. While the short-term outcomes of studies from the USA have been favorable in terms of exudate and visual acuity in patients who receive IVF after prior treatment with aflibercept or bevacizumab [1, 8], reports from Japan have demonstrated that the efficacy of brolucizumab is superior to that of aflibercept in the treatment of sub-retinal pigment epithelium lesions and in the reduction of choroidal thickness [9,10,11,12,13,14,15]. Therefore, it is important to investigate the efficacy of faricimab in Japanese patients with a prior history of treatment for nAMD, who often have a higher prevalence of polypoidal choroidal vasculopathy and pachychoroid [16], and to conduct separate analyses for the various anti-VEGF agents previously used. Therefore, in this real-world study, we investigated the short-term safety and efficacy of IVF for nAMD in Japanese patients with a prior treatment history.
Methods
Ethical Approval and Consent
This study had a single-center retrospective design and was approved by the institutional review board of Jichi Medical University (JICHI20-127) and adhered to the tenets of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study procedures followed institutional guidelines, and all patients provided oral informed consent before the procedures were performed.
Procedure
This study enrolled a total of 80 eyes from 75 consecutive patients with nAMD who had previously been treated with either aflibercept or brolucizumab. These patients received their first IVF injection at Jichi Medical University Hospital between May 2022 and September 2022. Macular neovascularization (MNV) was diagnosed based on fundus examination, fundus photography, fluorescein angiography (FA), indocyanine green angiography (ICGA), or swept-source optical coherence tomography (SS-OCT; performed by HT, SI, or RT). FA/ICGA had been performed before the first previous anti-VEGF injection. Color photographs of the fundus were obtained using a commercially available fundus camera system (model VX-10; Kowa Co., Ltd., Nagoya, Japan). In the present study, FA/ICGA was performed using a confocal scanning laser ophthalmoscope (Heidelberg Retina Angiography; Heidelberg Engineering, Heidelberg, Germany). Cross-sectional images of the macula were obtained using SS-OCT (DRI OCT Triton; Topcon, Tokyo, Japan).
At baseline (immediately before switching treatment), all patients underwent an ophthalmic assessment with refraction, BCVA testing, slit-lamp biomicroscopy with or without contact lenses, indirect ophthalmoscopy, color fundus photography, and SS-OCT.
To evaluate the impact of IVF injections, patients were instructed to maintain their regular visit intervals (± 1 week) after the initial IVF injection, regardless of the presence or absence of exudative changes on that day. There was no loading therapy administered upon switching. Our hypothesis was that if the previous anti-VEGF agent had a comparable effect to faricimab, no significant variation would be observed in the extent of exudative changes and choroidal thickness. We also predicted that a superior effect would lead to enhanced suppression of exudation and choroidal thickness, while a weaker effectiveness would result in increased changes in both parameters.
Inclusion and Exclusion Criteria
The inclusion criteria were patients who had been following a treat-and-extend (TAE) regimen and who could visit during the period we indicated. The TAE regimen is a proactive treatment strategy in which the date of the next injection is decided according to the presence or absence of exudative changes or hemorrhage at the time of injection. The treatment interval had been extended up to 20 weeks, with adjustment intervals of 2 or 4 weeks, prior to the IVF injection. Patients who experienced a recurrence of nAMD after being stable during a follow-up period of ≥ 6 months and/or had first IVF injection as a reactive treatment were excluded from the study. We identified 117 eyes of 109 patients with nAMD who had previously been treated with other anti-VEGF drugs and received their first IVF injection between inclusion periods. Patients were excluded from the study for the following reasons: (1) 14 eyes from 14 patients had received prior anti-VEGF treatment for a duration of ≥ 6; (2) four patients (5 eyes) sought earlier visits due to decreased vision; (3) 12 patients (13 eyes) had extended their visits by 2 weeks due to personal scheduling conflicts; (4) three eyes from three patients had missing data due to OCT malfunction during the examination; (5) one patient (1 eye) could not visit due to illness; and (6) one eye from one patient received laser photocoagulation for the treatment of a polypoidal lesion in conjunction with IVF. One patient had multiple reasons for exclusion.
Outcome Measures
Outcome measures were changes in BCVA, central subfield thickness (CST), and central choroidal thickness (CCT) after the initial IVF injection and after a treatment interval matching the previous duration. As a safety outcome evaluation, we assessed potential adverse effects, including intraocular inflammation, retinal vasculitis, retinal pigment epithelial (RPE) tear, severe visual loss (≥ 15 letters) due to any causes, procedure-related adverse effects, such as traumatic cataract formation and retinal tear, and any systemic adverse effects reported by the patient.
Statistical Analysis
Categorical data were assessed using Pearson’s Chi-squared (χ2) test or Fisher’s direct probability test. After confirming normal distribution using the Shapiro–Wilk test, Student’s t-test, paired t-test, and one-way analysis of variance (ANOVA) were utilized to evaluate continuous variables. Repeat tests were not performed, so no correction was made for multiple comparisons. All statistical analyses were performed using JMP Pro software version 17.0.0 (SAS Institute Inc., Cary, NC, USA). Statistical significance was defined by a P value of < 0.05. Results are shown as the mean value ± standard deviation (SD).
Results
Patient Demographics
The study included 80 eyes of 75 patients (49 men, 65%) with a mean (± SD) age of 76.7 ± 8.9 years (Table 1). Of these eyes, 33 were classified with MNV (type 1, n = 21; type 2, n = 7; type 3, n = 5) and 47 with polypoidal choroidal vasculopathy. The patients had received a mean of 22.4 ± 14.3 injections of other anti-VEGF agents, and the follow-up before their first IVF injection was 71.0 ± 46.9 months. Throughout the follow-up period, the patients were managed with a TAE regimen, resulting in a well-controlled baseline CST of 242 ± 72 µm and a CCT of 189 ± 97 µm.
Baseline characteristics were comparable between the aflibercept and brolucizumab groups before switching, except for the injection interval, which was longer in the brolucizumab group than in the aflibercept group (10.9 ± 3.5 vs. 8.8 ± 3.6 weeks; P = 0.015).
Changes in BCVA, CST, and CCT
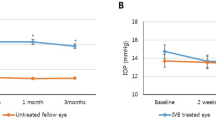
Table 2 shows the changes in BCVA, CST, and CCT after the initial switch in treatment. BCVA did not change significantly after switching to IVF (P = 0.29), and no significant exudative changes, subretinal hemorrhage, or adverse events were observed. CCT decreased significantly after the switch (P < 0.001), but there was no significant change in CST (P = 0.99) or BCVA (P = 0.29). Similar results were observed when evaluating the changes in CCT according to each prior anti-VEGF agent (Table 3). CCT decreased by 8.7 ± 2.5 µm (P < 0.0001) in the brolucizumab group (a representative case is shown in Fig. 1) and by 13.1 ± 3.6 µm (P < 0.0001) in the aflibercept group (a representative case is shown in Fig. 2). However, the difference in the CCT decrease between the two groups was not statistically significant (P = 0.32). No adverse events were observed during the course of the study.
Representative case of a decrease in CCT in the brolucizumab group. The patient was a 75-year-old man with type 3 macular neovascularization at baseline. He had received 14 anti-vascular endothelial growth factor injections (brolucizumab, n = 3; aflibercept, n = 11) over 47 months. The treatment interval immediately before the initial IVF injection was 20 weeks. a LogMAR BCVA, CST, and CCT were 0.15, 322 µm, and 184 µm, respectively. Optical coherence tomography and imaging of the fundus revealed subretinal hemorrhage and pigment epithelial detachment with subretinal fluid and intraretinal fluid. After the IVF injection, the patient's next visit was scheduled for 20 weeks, in accordance with the treatment protocol. b Twenty weeks after the initial intravitreal faricimab injection, the logMAR BCVA, CST, and CCT were 0, 218 µm, and 129 µm, respectively. Optical coherence tomography revealed decreases in pigment epithelial detachment, subretinal fluid, and intraretinal fluid. BCVA Best-corrected visual acuity, CCT central choroidal thickness, CST central subfield macular thickness, IVF intravitreal faricimab, logMAR logarithm of the minimum angle of resolution
Representative case of a decrease in CCT in the aflibercept group. The patient was a 74-year-old man with type 1 macular neovascularization at baseline. He had received 16 anti-vascular endothelial growth factor injections (aflibercept, n = 15; ranibizumab, n = 1) over 48 months. The treatment interval immediately before the initial IVF injection was 10 weeks. a LogMAR BCVA, CST, and CCT were − 0.079, 205 µm, and 340 µm, respectively. Optical coherence tomography and imaging of the fundus revealed pigment epithelial detachment without subretinal hemorrhage, subretinal fluid, or intraretinal fluid. b Twelve weeks after the initial IVF injection, his logMAR BCVA, CST and CCT were 0, 238 µm, and 264 µm, respectively. Optical coherence tomography revealed pigment epithelial detachment with subretinal fluid
Discussion
Unlike conventional anti-VEGF agents, faricimab suppresses Ang-2-modulated pathology and is expected to provide superior disease control [17, 18]. This study assessed outcomes before the initial IVF injection and after a treatment interval matching the previous duration. We found that there was no significant change in CST or BCVA after switching from IVA or IVB to IVF. However, there was a significant decrease in CCT, indicating a positive response to IVF. No adverse events were noted in patients who had previously received either aflibercept or brolucizumab.
Our present findings, particularly in terms of BCVA and CST, are inconsistent with those reported previously which suggested that switching from IVA to IVF leads to improved visual and anatomic outcomes in patients with nAMD [1, 2, 8]. These discordant results may be attributed to differences in baseline characteristics between the studies. Specifically, the baseline BCVA was worse in previous studies than in our study; in addition, CST was greater in one of the previous studies than in our study. However, given that all patients were in the maintenance phase and their disease activities were already well-controlled prior to the switch, it is reasonable to expect that there would be no substantial alteration observed in CST during the current study [3].
Interestingly, in the present study, we found that CCT can decrease after the first IVF injection in patients with a history of treatment with aflibercept or brolucizumab. Treatment with aflibercept leads to a decrease in subfoveal choroidal thickness in eyes with treatment-naïve nAMD [19, 20]. Furthermore, brolucizumab has also been associated with significant reduction in the CCT in patients with nAMD, regardless of prior aflibercept treatment [21]. While the magnitude of the decrease in CCT in our study was smaller than that in patients with treatment-naïve nAMD who received anti-VEGF therapy, our findings suggest that IVF may still have a positive impact on CCT regardless of prior treatments, although the extent of the effect might differ from that in treatment-naïve patients [19].
When considering the accumulating evidence on the relationship between pachychoroid and angiopoietins, our results can be interpreted in an intriguing manner. In Asian countries, a significant proportion of patients with nAMD are considered to have pachychoroid features, and the phenotype of nAMD is known to differ from that of Caucasian patients [16, 22,23,24]. Previous studies found that VEGF concentrations in the aqueous humor were lower in eyes with pachychoroid neovascularization than in those with drusen-associated MNV [25, 26], whereas Ang-2 is elevated in eyes with pachychoroid features [26]. Moreover, knockout of Ang-1 in mice leads to a pachychoroid phenotype, indicating the role of angiopoietins in choroidal remodeling [27]. Taken together, our results support the hypothesis that dual Ang-2/VEGF-A inhibition potentiates choroidal vascular remodeling, which might give better results for Asian patients with nAMD, especially those associated with the pachychoroid phenotype. Although further studies are needed to investigate the effect of faricimab on the choroid, understanding the interplay between Ang-1, Ang-2, and VEGF-A could shed light on the mechanisms underlying pachychoroid-related disorders and guide targeted therapeutic approaches for restoring the integrity of the choroid.
This study has some limitations. First, all patients included in this study were Japanese, which may limit the generalizability of its findings to other ethnic populations. The impact of ethnic and genetic variations on treatment responses and outcomes cannot be fully elucidated based on this study alone. Second, the sample size was relatively small, which might have affected our ability to detect subtle differences or associations that could have been present but not apparent as a consequence of limited statistical power. A larger sample size would provide more statistical power and enhance the generalizability of the results. Finally, this study is a short retrospective analysis. Long-term real-world follow-up data will be crucial to understand the usefulness of faricimab in the future.
Conclusion
In this study, there was no significant change in CST or BCVA following a switch from IVA or IVB to a single IVF injection. However, a potential decrease in CCT was observed. These findings suggest that IVF is generally safe and effective in terms of improving the choroidal vascular circulation.
References
Rush RB, Rush SW. Intravitreal faricimab for aflibercept-resistant neovascular age-related macular degeneration. Clin Ophthalmol. 2022;16:4041–6.
Stanga PE, Valentin-Bravo FJ, Stanga SEF, Reinstein UI, Pastor-Idoate S, Downes SM. Faricimab in neovascular AMD: first report of real-world outcomes in an independent retina clinic. Eye (Lond); 2023. https://doi.org/10.1038/s41433-023-02505-z.
Heier JS, Khanani AM, Quezada Ruiz C, et al. Efficacy, durability, and safety of intravitreal faricimab up to every 16 weeks for neovascular age-related macular degeneration (TENAYA and LUCERNE): two randomised, double-masked, phase 3, non-inferiority trials. Lancet. 2022;399:729–40.
Mori R, Honda S, Gomi F, et al. Efficacy, durability, and safety of faricimab up to every 16 weeks in patients with neovascular age-related macular degeneration: 1-year results from the Japan subgroup of the phase 3 TENAYA trial. Jpn J Ophthalmol. 2023;67:301–10.
Takahashi K, Cheung CMG, Iida T, et al. Efficacy, durability, and safety of faricimab in patients from Asian countries with neovascular age-related macular degeneration: 1-Year subgroup analysis of the TENAYA and LUCERNE trials. Graefes Arch Clin Exp Ophthalmol 2023:1–13. https://doi.org/10.1007/s00417-023-06071-8.
Matsumoto H, Hoshino J, Nakamura K, Nagashima T, Akiyama H. Short-term outcomes of intravitreal faricimab for treatment-naive neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2023. https://doi.org/10.1007/s00417-023-06116-y.
Mukai R, Kataoka K, Tanaka K, et al. Three-month outcomes of faricimab loading therapy for wet age-related macular degeneration in Japan. Sci Rep. 2023;13:8747.
Khanani AM, Aziz AA, Khan H, et al. The real-world efficacy and safety of faricimab in neovascular age-related macular degeneration: the TRUCKEE study—6 month results. Eye (Lond). 2023. https://doi.org/10.1038/s41433-023-02553-5.
Hoshino J, Matsumoto H, Mukai R, Nakamura K, Akiyama H. Intravitreal aflibercept versus brolucizumab for treatment-naive neovascular age-related macular degeneration with type 1 macular neovascularization: comparison of short-term outcomes. Ophthalmologica. 2022;245:413–20.
Tanaka K, Koizumi H, Tamashiro T, et al. Short-term results for brolucizumab in treatment-naive neovascular age-related macular degeneration: a Japanese multicenter study. Jpn J Ophthalmol. 2022;66:379–85.
Ito A, Maruyama-Inoue M, Kitajima Y, Ikeda S, Inoue T, Kadonosono K. One-year outcomes of intravitreal brolucizumab injections in patients with polypoidal choroidal vasculopathy. Sci Rep. 2022;12:7987.
Matsumoto H, Hoshino J, Mukai R, Nakamura K, Akiyama H. Short-term outcomes of intravitreal brolucizumab for treatment-naive neovascular age-related macular degeneration with type 1 choroidal neovascularization including polypoidal choroidal vasculopathy. Sci Rep. 2021;11:6759.
Kikushima W, Sakurada Y, Sugiyama A, Tanabe N, Kume A, Iijima H. Comparison of initial treatment between 3-monthly intravitreal aflibercept monotherapy and combined photodynamic therapy with single intravitreal aflibercept for polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol. 2017;255:311–6.
Yamamoto A, Okada AA, Kano M, et al. One-year results of intravitreal aflibercept for polypoidal choroidal vasculopathy. Ophthalmology. 2015;122:1866–72.
Morimoto M, Matsumoto H, Mimura K, Akiyama H. Two-year results of a treat-and-extend regimen with aflibercept for polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol. 2017;255:1891–7.
Maruko I, Iida T, Saito M, Nagayama D, Saito K. Clinical characteristics of exudative age-related macular degeneration in Japanese patients. Am J Ophthalmol. 2007;144:15–22.
Akwii RG, Sajib MS, Zahra FT, Mikelis CM. Role of angiopoietin-2 in vascular physiology and pathophysiology. Cells. 2019;8:471.
Saharinen P, Eklund L, Alitalo K. Therapeutic targeting of the angiopoietin-TIE pathway. Nat Rev Drug Discov. 2017;16:635–61.
Koizumi H, Kano M, Yamamoto A, et al. Subfoveal choroidal thickness during aflibercept therapy for neovascular age-related macular degeneration: twelve-month results. Ophthalmology. 2016;123:617–24.
Koizumi H, Kano M, Yamamoto A, et al. Short-term changes in choroidal thickness after aflibercept therapy for neovascular age-related macular degeneration. Am J Ophthalmol. 2015;159:627–33.
Tamashiro T, Tanaka K, Itagaki K, et al. Subfoveal choroidal thickness after brolucizumab therapy for neovascular age-related macular degeneration: a short-term multicenter study. Graefes Arch Clin Exp Ophthalmol. 2022;260:1857–65.
Matsumoto H, Hoshino J, Mukai R, Nakamura K, Kishi S, Akiyama H. Clinical characteristics and pachychoroid incidence in Japanese patients with neovascular age-related macular degeneration. Sci Rep. 2022;12:4492.
Mitchell P, Wang JJ, Foran S, Smith W. Five-year incidence of age-related maculopathy lesions: the Blue Mountains Eye Study. Ophthalmology. 2002;109:1092–7.
Klein R, Klein BE, Tomany SC, Meuer SM, Huang GH. Ten-year incidence and progression of age-related maculopathy: the Beaver Dam eye study. Ophthalmology. 2002;109:1767–79.
Hata M, Yamashiro K, Ooto S, et al. Intraocular vascular endothelial growth factor levels in pachychoroid neovasculopathy and neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci. 2017;58:292–8.
Inoda S, Takahashi H, Inoue Y, et al. Cytokine profiles of macular neovascularization in the elderly based on a classification from a pachychoroid/drusen perspective. Graefes Arch Clin Exp Ophthalmol. 2022;260:747–58.
Liu P, Lavine JA, Fawzi A, Quaggin SE, Thomson BR. Angiopoietin-1 is required for vortex vein and choriocapillaris development in mice. Arterioscler Thromb Vasc Biol. 2022;42:1413–27.
Acknowledgements
Author Contribution
Satoru Inoda conceptualized the study, conducted the statistical analysis, acquired and interpreted the data, and drafted the work. Hidenori Takahashi, Hidetoshi Kawashima, and Yasuo Yanagi reviewed the analyses and revised the draft. Ryota Takahashi, Yuto Hashimoto, Hana Yoshida, Takuya Takayama, and Hironori Takahashi acquired the data. All authors reviewed and approved the manuscript.
Funding
No funding or sponsorship was received for this study. The journal’s Rapid Service fees were funded by the authors.
Medical Writing and Editorial Assistance
Editorial assistance in the preparation of this article was provided by ThinkSCIENCE, Inc. (Tokyo, Japan) and was funded by Jichi Medical University.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval
This study was approved by the institutional review board of Jichi Medical University (JICHI20-127) and adhered to the tenets of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study procedures followed the guidelines of the institution, and all patients provided informed consent prior to the procedures.
Conflict of Interest
Ryota Takahashi, Yuto Hashimoto, Hana Yoshida, Hironori Takahashi, and Takuya Takayama declare that they have no competing interest. Satoru Inoda declares lecturer’s fees from Kowa, Novartis, Chugai and DeepEyeVision, Inc., and grants from Novartis outside this work. Hidenori Takahashi was a founder of DeepEyeVision, Inc., and declares patents outside this work, lecturer’s fees from Santen, Kowa, Senju, Novartis and Bayer outside this work and grants from Senju, Novartis and Bayer outside this work. Hidetoshi Kawashima declares lecturer’s fees from Otsuka, Senju, Mitsubishi-Tanabe, Kowa, Santen, Novartis and Zeiss outside this work and grants from Senju, Linical, DeepEyeVision, HOYA, Santen, Heiwa-Iyou and Bayer outside this work. Yasuo Yanagi declares grants from Santen and Novartis outside this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Inoda, S., Takahashi, H., Takahashi, R. et al. Visual and Anatomical Outcomes After Initial Intravitreal Faricimab Injection for Neovascular Age-Related Macular Degeneration in Patients with Prior Treatment History. Ophthalmol Ther 12, 2703–2712 (2023). https://doi.org/10.1007/s40123-023-00779-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-023-00779-w