Abstract
Background
Treatment-resistant depression (TRD) is a chronic illness requiring long-term treatment. Esketamine nasal spray (ESK) has been studied in several long-term trials of patients with TRD, including SUSTAIN-1 (NCT02493868) and SUSTAIN-3 (NCT02782104). This subgroup analysis of SUSTAIN-3 evaluated patients with TRD who received a second induction (IND) and maintenance treatment with ESK plus oral antidepressant (AD) after a relapse in SUSTAIN-1.
Methods
Patients aged 18–64 years who achieved stable remission or response with ESK and subsequently relapsed after randomization to continue ESK or switch to placebo nasal spray (PBO) in SUSTAIN-1 and entered the IND phase of SUSTAIN-3 were included in this interim analysis. Response (≥50% improvement in total score from baseline for Montgomery–Åsberg Depression Rating Scale [MADRS] and Patient Health Questionnaire 9-item [PHQ-9]), remission (MADRS score ≤12; PHQ-9 total score <5), changes in depression rating scores (measured as mean change from baseline), and safety were evaluated (incidence of treatment-emergent and serious adverse events [AE]).
Results
Of the 96 eligible patients who entered IND in SUSTAIN-3, 32 (33.3%) were taking ESK+AD at the time of relapse in SUSTAIN-1 and 64 (66.7%) were taking AD+PBO. Substantial improvements in depressive symptoms were observed over the second IND phase in both groups and were maintained over the optimization/maintenance (OP/M) phase. MADRS response rates following a second IND were 71.9% and 73.4% for previously relapsed (PR) ESK+AD and PR-AD+PBO, respectively; remission rates were 62.5% and 60.9%, respectively. During the IND and OP/M phases, 58.3% and 83.3% of patients experienced a treatment-emergent AE, respectively. No patients discontinued due to an AE during the second IND.
Conclusions
Patients with TRD benefitted from receiving a second IND and maintenance treatment with ESK and no new safety signals were identified.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This subgroup analysis evaluated efficacy and safety outcomes in patients with treatment-resistant depression who relapsed while taking or after discontinuing esketamine nasal spray (ESK) plus an oral antidepressant during SUSTAIN-1 and received a second induction and maintenance treatment with ESK in SUSTAIN-3. |
Patients with treatment-resistant depression who previously responded to ESK and experienced a relapse benefitted from receiving a second induction and maintenance treatment with ESK. |
ESK was generally well tolerated and no new safety signals were identified. |
1 Introduction
Major depression is one of the leading causes of disability worldwide, affecting approximately 8.9 million adults with medication-treated major depressive disorder (MDD) in the United States, including approximately 2.8 million adults (30.9%) who exhibit treatment-resistant symptoms [1]. Individuals with treatment-resistant depression (TRD) often experience prolonged depressive episodes, which can lead to treatment discontinuation prior to reaching recovery [2]. These prolonged or recurrent depressive episodes can result in difficulties with social and occupational function, decline of physical health, suicidal thoughts, and increased health care utilization [3, 4]. While there is no universal definition, TRD is commonly defined as the absence of a response to two or more oral antidepressants (ADs) of adequate dose and duration [4, 5]. Response and remission are commonly used outcome criteria for the treatment of depression with the goal of achieving remission because these outcomes can be predictors of long-term stability [5, 7]. Therefore, identifying and optimizing treatment options for patients with TRD represents a substantial unmet need [4, 8].
Esketamine nasal spray (ESK) is a noncompetitive N-methyl-d-aspartate (NMDA) receptor antagonist approved by the United States Food and Drug Administration, as well as health authorities in other countries, in conjunction with an oral AD, for the treatment of TRD in adults and for the treatment of depressive symptoms in adults with MDD with acute suicidal ideation or behavior [9, 10]. Several real-world studies have demonstrated the benefit of ESK treatment in adult patients with TRD [11, 12]. However, in some cases, after a reduction in frequency of ESK dosing or treatment discontinuation, patients may experience a relapse or a worsening of depressive symptoms [8]; in these scenarios, clinicians may consider restarting treatment with ESK if clinically appropriate. A recent study found that a subset of patients ≥65 years of age, who were nonresponders to ESK treatment after completing the double-blind induction (IND) phase of a randomized 4-week efficacy study and subsequently entered the IND phase of an open-label, long-term, safety and efficacy study of ESK (SUSTAIN-2), received clinical benefit from a second IND of ESK treatment [13]. Understanding the outcomes of patients with TRD who previously responded to ESK, experienced a relapse, and subsequently received a second IND and maintenance treatment with ESK is of clinical interest due to the complex nature of treating patients with TRD [3, 4].
The SUSTAIN-3 study (ClinicalTrials.gov identifier NCT02782104) is a long-term, open-label, phase III study evaluating the efficacy, safety, and tolerability of ESK [14,15,16]. The SUSTAIN-3 study provides an opportunity to assess a subset of patients who relapsed in a previous study (SUSTAIN-1; NCT02493868) and were eligible to receive a second induction in SUSTAIN-3 [8]. Patients in SUSTAIN-1 could have experienced relapse on oral AD + placebo (PBO) or ESK + AD (e.g., following treatment discontinuation or a reduction in ESK dosing frequency). Overall, the SUSTAIN-1 study demonstrated that continued treatment with ESK+AD resulted in a significant and clinically meaningful delay in relapse compared with AD+PBO in patients with TRD. This subgroup analysis of the ongoing SUSTAIN-3 study evaluated efficacy and safety outcomes in patients who relapsed while taking or after discontinuing ESK during SUSTAIN-1 and received a second induction followed by maintenance treatment with ESK.
2 Methods
2.1 Study Design and Treatment
The SUSTAIN-1 relapse prevention study had five phases: a 4-week screening phase, a 4-week open-label (OL) IND phase, a 12-week optimization phase, a variable-duration maintenance phase, and a 2-week follow-up phase. After the 4-week IND phase and 12-week optimization phase (in which all patients received ESK+AD), patients who achieved stable remission or stable response were randomly assigned 1:1 to continue ESK treatment or switch to PBO. ESK dosing of 56 or 84 mg in SUSTAIN-1 was flexible, twice weekly for 4 weeks in IND (with dosing frequency subsequently individualized based on depression severity in the optimization and maintenance phases), and in conjunction with an oral AD. Full study design and inclusion and exclusion criteria for SUSTAIN-1 are published elsewhere [8].
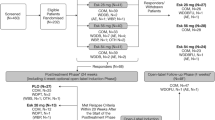
SUSTAIN-3 is an ongoing, multicenter, phase III, OL extension study to evaluate the efficacy, safety, and tolerability of ESK, given in conjunction with an oral AD, in adults with TRD. Data collected from June 21, 2016, to December 1, 2020, were used in this interim analysis with patients having up to 48 months of treatment at data cutoff. The study comprises two phases: a 4-week IND phase and a variable-duration optimization/maintenance (OP/M) phase (Fig. 1). Dosing of 56 or 84 mg of ESK in SUSTAIN-3 was flexible, twice weekly for 4 weeks in IND. During the OP/M phase, ESK was self-administered as a flexible-dose regimen of 56 or 84 mg, with adjustments according to efficacy and tolerability based on the Clinical Global Impression-Severity (CGI-S) score. Patients could enter the IND phase of SUSTAIN-3 from SUSTAIN-1 if they attained stable remission or response status with ESK after an initial 16 weeks of treatment and subsequently experienced relapse in the maintenance phase of SUSTAIN-1 (defined as Montgomery–Åsberg Depression Rating Scale [MADRS] [17] total score ≥22 for two consecutive assessments separated by 5–15 days and/or hospitalization for worsening depression or any other clinically relevant event).
SUSTAIN-3 study design. aStable remission was defined as MADRS total score ≤12 for at least 3 of the last 4 weeks of the optimization phase; stable response was defined as ≥50% reduction in the MADRS total score from baseline in each of the last 2 weeks of the optimization phase but does not meet criteria for stable remission. bBased on CGI-S and tolerability. AD antidepressant, CGI-S Clinical Global Impression-Severity, ESK esketamine nasal spray, OL open label, PR-AD+PBO previously relapsed on an oral AD plus placebo, PR-ESK+AD previously relapsed on ESK plus an oral AD
2.2 Patient Population
Adult patients 18–64 years of age determined to be medically stable on the basis of a physical examination, vital signs, and 12-lead electrocardiogram performed on the day of the first treatment session and according to the investigator’s judgment and knowledge of the participant’s stability in SUSTAIN-1 were eligible. Patients were also required to meet the Diagnostic and Statistical Manual of Mental Disorders—Fifth Edition (DSM-5) criteria for recurrent MDD, without psychotic features, confirmed by the Mini International Neuropsychiatric Interview. Patients were not eligible if the benefits versus risk of continued ESK treatment was not favorable in the opinion of the investigator or if the patient had suicidal ideation with intent to act since the last study visit in SUSTAIN-1. Patients who had a neurodegenerative disorder, evidence of mild cognitive impairment, ongoing evidence of uncontrolled hypertension, or a positive test result for drugs of abuse on the day of the first treatment session (pre-dose) were excluded. Patients were also excluded if they had a current or prior DSM-5 diagnosis of a psychotic disorder, bipolar or related disorders, obsessive compulsive disorder (current only), intellectual disability, autism spectrum disorder, borderline personality disorder, antisocial personality disorder, histrionic personality disorder, or narcissistic personality disorder.
2.3 Efficacy and Safety Assessments
Clinician- and patient-reported disease severity measures, including the MADRS and the Patient Health Questionnaire 9-item (PHQ-9) [18], were administered. The MADRS was assessed by site clinicians at baseline, on days 8, 15, 22, and 28 of the IND phase, and every 4 weeks during the OP/M phase. The PHQ-9 was completed by patients at baseline, on days 15 and 28 of the IND phase, and every 4 weeks during the OP/M phase. The proportions of patients who attained response or remission status were determined with both MADRS and PHQ-9 for day 28 of the IND phase and weeks 24 and 52 of the OP/M phase. For MADRS, response was defined as ≥50% improvement in total score from baseline, and remission was defined as a score ≤12 [19, 20]. For PHQ-9, response and remission were defined as ≥50% improvement in total score from baseline and total score <5 (normal), respectively, which are commonly used clinical definitions [21,22,23,24]. Efficacy outcome results are summarized descriptively. The incidence of treatment-emergent adverse events (TEAEs) and serious adverse events (SAEs) were also evaluated.
3 Results
3.1 Patients
This analysis included a total of 96 patients who had relapsed during the SUSTAIN-1 study and subsequently entered the IND phase in SUSTAIN-3; most patients (98%) continued to the OP/M phase of SUSTAIN-3. Of these patients, 32 had been taking ESK+AD and 64 had been taking AD+PBO at time of relapse in the maintenance phase of SUSTAIN-1; these patient groups are henceforth referred to as PR-ESK+AD (previously relapsed on ESK plus an oral AD) and PR-AD+PBO (previously relapsed on an oral AD plus PBO after receiving ESK). Mean [SD] disease severity scores at baseline of the second IND phase (SUSTAIN-3) were consistent with moderate to severe disease for MADRS (PR-ESK+AD: 26.5 [9.13]; PR-AD+PBO: 29.2 [6.53]) and PHQ-9 (PR-ESK+AD: 12.3 [4.65]; PR-AD+PBO: 13.7 [5.00]). Mean age at baseline was 46.0 and 44.5 years for PR-ESK+AD and PR-AD+PBO, respectively; 75.0% and 67.2% were female, and 90.6% of patients in each group were White (Table 1).
3.2 Prior Exposure to Study Medication
Following randomization, during the maintenance phase of SUSTAIN-1, the median extent of exposure to ESK was 9.1 weeks in the PR-ESK+AD group and the median exposure to PBO was 3.8 weeks in the PR-AD+PBO group. The median duration between relapse in SUSTAIN-1 and the beginning of the second IND in SUSTAIN-3 was 22.0 days and 16.0 days in the PR-ESK+AD group and PR-AD+PBO group, respectively. Most patients (71.9%) in the PR-ESK+AD group were taking 84 mg of ESK at the end of the SUSTAIN-1 optimization phase. Prior to relapse, most patients were dosed weekly in both the PR-ESK+AD group (66.7%) and the PR-AD+PBO group (68.4%).
3.3 Clinician- and Patient-Rated Depressive Symptoms
Substantial improvements in depressive symptoms were observed over the second 4-week IND phase in both groups, with mean (SD) changes from baseline in MADRS total scores of −14.6 (9.84) and −19.1 (9.16), respectively, for PR-ESK+AD and PR-AD+PBO; these improvements were maintained over the course of the OP/M phase (Fig. 2). A similar trend was observed for PHQ-9 scores, with mean (SD) changes from baseline of −5.3 (4.92) and −8.2 (5.77) for PR-ESK+AD and PR-AD+PBO, respectively, with improvements maintained over the OP/M phase (Fig. 3). Similar MADRS and PHQ-9 results were also observed in the combined PR-ESK+AD and PR-AD+PBO groups, with mean (SD) changes from baseline over the second 4-week IND phase of −17.6 (9.59) for MADRS and −7.3 (5.65) for PHQ-9 (Supplemental Fig. 1, see electronic supplementary material [ESM]). The PR-ESK+AD group had numerically higher mean MADRS and PHQ-9 total scores (last observation carried forward [LOCF]) at the end of the 4-week IND phase (IND endpoint) and at weeks 24 and 52 in the OP/M phase compared with the PR-AD+PBO group (Table 2).
Mean MADRS total scores (LOCF) during the reinduction and optimization/maintenance phases of SUSTAIN-3 among patients who previously experienced relapse in SUSTAIN-1. IND induction, LOCF last observation carried forward, MADRS Montgomery-Åsberg Depression Rating Scale, OP/M optimization/maintenance, PR-AD+PBO previously relapsed on an oral antidepressant plus placebo, PR-ESK+AD previously relapsed on esketamine nasal spray plus an oral antidepressant
Mean PHQ-9 total scores (LOCF) during the reinduction and optimization/maintenance phases of SUSTAIN-3 among patients who previously experienced relapse in SUSTAIN-1. IND induction, LOCF last observation carried forward, OP/M optimization/maintenance, PHQ-9 Patient Health Questionnaire 9-item, PR-AD+PBO previously relapsed on an oral antidepressant plus placebo, PR-ESK+AD previously relapsed on esketamine nasal spray plus an oral antidepressant
3.4 Response and Remission
In the PR-ESK+AD group, 71.9% and 62.5% of patients achieved response and remission based on MADRS, respectively, at the IND phase endpoint; response and remission rates were 37.5% and 40.6% at week 24 in the OP/M phase and 40.6% and 37.5% at week 52 in the OP/M phase, respectively (Table 3). Corresponding response and remission rates based on PHQ-9 scores were 46.9% and 21.9% at the IND phase endpoint, 31.3% and 28.1% at week 24 in the OP/M phase, and 31.3% and 28.1% at week 52 in the OP/M phase.
In the PR-AD+PBO group, 73.4% and 60.9% of patients achieved response and remission based on MADRS, respectively, at the IND phase endpoint; response and remission rates were 69.4% and 58.1% at week 24 in the OP/M phase and 54.8% and 41.9% at week 52 in the OP/M phase, respectively (Table 3). Corresponding response and remission rates based on PHQ-9 scores were 63.5% and 53.1% at the IND phase endpoint, 57.4% and 41.9% at week 24 in the OP/M phase, and 49.2% and 33.9% at week 52 in the OP/M phase.
3.5 Safety
During the IND and OP/M phases, 58.3% and 83.3% of patients experienced a TEAE, respectively; the most common TEAEs in the IND phase were dissociation (17.7%), dizziness (13.5%), vertigo (13.5%), and dysgeusia (12.5%). The most common TEAEs in the OP/M phase included dissociation (27.1%), headache (25.0), dizziness (21.9%), vertigo (20.8%), and somnolence (18.8%; the full list of the most common [≥10%] TEAEs is shown in Table 4). No patients experienced an SAE during the IND phase; 12 patients (12.8%) experienced an SAE during the OP/M phase. The most common SAEs were psychiatric disorders (3.1%; including affect lability, depressed mood, depression, suicidal ideation, and major depression); neoplasms benign, malignant, and unspecified (2.1%; including prostate cancer and esophageal squamous cell carcinoma); and nervous system disorders (2.1%; including loss of consciousness and metabolic encephalopathy). Overall, no new safety signals were identified from this analysis.
During the OP/M phase, four patients (4.3%) withdrew from the study due to a TEAE (one patient who experienced a TEAE of major depression in SUSTAIN-1 withdrew from SUSTAIN-3 due to major depression; no other patients had overlapping TEAEs in both studies). No patients withdrew from the IND phase due to a TEAE. Overall, rates of discontinuation during the OP/M phase were similar among groups (PR-ESK+AD: 28.1%; PR-AD+PBO: 22.6%) over a period of up to 52 weeks of follow-up. Other reasons for discontinuation during the OP/M phase included withdrawal by subject (n = 3, 3.2%), lack of efficacy (n = 2, 2.1%), loss to follow-up (n = 1, 1.1%), and death (n = 1, 1.1%; patient died from COVID-19 disease, which was not considered related to ESK treatment by the investigator).
4 Discussion
Patients with TRD who previously responded to treatment with ESK and relapsed experienced improvement in depressive symptoms and functioning during a second course of IND therapy; this improvement was sustained during maintenance treatment. Patients appeared to benefit from a second IND of ESK treatment regardless of whether they experienced relapse after discontinuation of ESK (PR-AD+PBO) or while continuing treatment with ESK (PR-ESK+AD). Overall, ESK was well tolerated with no new safety signals identified. No patients discontinued due to a TEAE during a second IND; however, four patients discontinued due to TEAEs during the OP/M phase. The overall rates of discontinuation during the OP/M phase were similar between groups (PR-ESK+AD: 28.1%; PR-AD+PBO: 22.6%) over a period of up to 52 weeks of follow-up. To provide context, a long-term open-label study of vortioxetine for up to 52 weeks reported that 310/834 (37.2%) patients discontinued treatment early for a variety of reasons [25].
The patient trajectories in this study (i.e., discontinuing ESK treatment or relapsing while on ESK) could be reflective of real-world clinical scenarios. Approximately one-third of the patients included in this analysis had relapsed while receiving ESK on a maintenance schedule during SUSTAIN-1, a scenario that could occur outside of a clinical trial setting. The other two-thirds relapsed after discontinuing ESK, which would likely occur in clinical settings if patients opted not to continue ESK on a maintenance schedule.
In SUSTAIN-1, both patient groups received an initial 16 weeks of ESK treatment, were randomly assigned to either continue on ESK treatment or switch to PBO treatment, and subsequently relapsed during the maintenance phase. Notably, patients from SUSTAIN-1 had a similar trend in mean MADRS scores over time compared with the subgroup of patients in this analysis, suggesting that a second course of ESK IND and maintenance treatment is comparably beneficial to patients [8]. Additionally, when examining the combined PR-ESK+AD and PR-AD+PBO groups, substantial improvements were observed over the second 4-week IND phase, with mean (SD) changes from baseline of −17.6 (9.59) for MADRS and −7.3 (5.65) for PHQ-9. Overall, these findings provide evidence to help guide clinical decision making regarding the potential benefit of a second ESK IND and treatment course in both the PR-ESK+AD group and the PR-AD+PBO group.
Several limitations should be noted when considering this analysis. First, SUSTAIN-3 was an open-label study with no comparator group. Second, the generalizability of the study findings may be limited by the exclusion of participants with significant psychiatric or medical comorbidities or substance use disorder. Third, there may be an inherent variability associated with PHQ-9 scores as they are patient reported. Finally, the small sample size of the subgroups may limit the statistical power and interpretation of results. Of note, the results in the PR-ESK+AD group were driven by a very small number of patients (n = 32).
5 Conclusions
This subgroup analysis found that ESK was generally well tolerated and patients with TRD who previously responded to ESK and experienced a relapse benefited from receiving a second induction and maintenance treatment with ESK. These findings support the potential benefit of a second ESK IND and treatment course in patients with TRD who relapsed while taking or after discontinuing ESK and can help guide clinical decision making in the future.
References
Zhdanava M, Pilon D, Ghelerter I, Chow W, Joshi K, Lefebvre P, et al. The prevalence and national burden of treatment-resistant depression and major depressive disorder in the United States. J Clin Psychiatry. 2021;82(2):20m13699. https://doi.org/10.4088/JCP.20m13699.
Voineskos D, Daskalakis ZJ, Blumberger DM. Management of treatment-resistant depression: challenges and strategies. Neuropsychiatr Dis Treat. 2020;16:221–34. https://doi.org/10.2147/NDT.S198774.
Burcusa SL, Iacono WG. Risk for recurrence in depression. Clin Psychol Rev. 2007;27(8):959–85. https://doi.org/10.1016/j.cpr.2007.02.005.
Al-Harbi KS. Treatment-resistant depression: therapeutic trends, challenges, and future directions. Patient Prefer Adherence. 2012;6:369–88. https://doi.org/10.2147/PPA.S29716.
Gaynes BN, Asher G, Gartlehner G, Hoffman V, Green J, Boland E, et al. Definition of Treatment-Resistant Depression in the Medicare Population. Rockville: Agency for Healthcare Research and Quality (US); 2018.
Riedel M, Moller HJ, Obermeier M, Schennach-Wolff R, Bauer M, Adli M, et al. Response and remission criteria in major depression—a validation of current practice. J Psychiatr Res. 2010;44(15):1063–8. https://doi.org/10.1016/j.jpsychires.2010.03.006.
Mendlewicz J. Towards achieving remission in the treatment of depression. Dialogues Clin Neurosci. 2008;10(4):371–5. https://doi.org/10.31887/DCNS.2008.10.4/jmendlewicz.
Daly EJ, Trivedi MH, Janik A, Li H, Zhang Y, Li X, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiat. 2019;76(9):893–903. https://doi.org/10.1001/jamapsychiatry.2019.1189.
SPRAVATO® (esketamine) nasal spray, CIII [prescribing information]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 07/2020.
Takahashi N, Yamada A, Shiraishi A, Shimizu H, Goto R, Tominaga Y. Efficacy and safety of fixed doses of intranasal esketamine as an add-on therapy to Oral antidepressants in Japanese patients with treatment-resistant depression: a phase 2b randomized clinical study. BMC Psychiatry. 2021;21(1):526. https://doi.org/10.1186/s12888-021-03538-y.
Martinotti G, Vita A, Fagiolini A, Maina G, Bertolino A, Dell’Osso B, et al. Real-world experience of esketamine use to manage treatment-resistant depression: a multicentric study on safety and effectiveness (REAL-ESK study). J Affect Disord. 2022;15(319):646–54. https://doi.org/10.1016/j.jad.2022.09.043.
Brendle M, Ahuja S, Valle MD, Moore C, Thielking P, Malone DC, et al. Safety and effectiveness of intranasal esketamine for treatment-resistant depression: a real-world retrospective study. J Comp Eff Res. 2022;11(18):1323–36. https://doi.org/10.2217/cer-2022-0149.
Wajs E, Aluisio L, Holder R, Daly EJ, Lane R, Lim P, et al. Esketamine nasal spray plus oral antidepressant in patients with treatment-resistant depression: assessment of long-term safety in a phase 3, open-label study (SUSTAIN-2). J Clin Psychiatry. 2020. https://doi.org/10.4088/JCP.19m12891.
Zaki N, Fu D, Daly E, Kevo S, Lopena O, Turkoz I. Long-term efficacy of esketamine nasal spray in adults with treatment-resistant depression: a subgroup analysis of the ongoing SUSTAIN-3 study. Neuroscience Education Institute (NEI) Congress; 2021 November 4–7, 2021; Colorado Springs, CO; 2021.
Zaki N, Fu D, Daly E, Kevo S, Reilly K, Turkoz I, et al. Long-Term Safety of Esketamine Nasal Spray in Adults With Treatment-Resistant Depression: A Subgroup Analysis of the SUSTAIN-3 Study. Neuroscience Education Institute (NEI) Congress; 2021 November 4–7, 2021; Colorado Springs, CO; 2021.
Zaki N, Chen LN, Lane R, Doherty T, Drevets WC, Morrison RL, et al. Long-term safety and maintenance of response with esketamine nasal spray in participants with treatment-resistant depression: interim results of the SUSTAIN-3 study. Neuropsychopharmacology. 2023. https://doi.org/10.1038/s41386-023-01577-5.
Williams JB, Kobak KA. Development and reliability of a structured interview guide for the Montgomery Asberg Depression Rating Scale (SIGMA). Br J Psychiatry. 2008;192(1):52–8. https://doi.org/10.1192/bjp.bp.106.032532.
Spitzer RL, Kroenke K, Williams JB. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA. 1999;282(18):1737–44. https://doi.org/10.1001/jama.282.18.1737.
Montgomery SA, Möller HJ. Is the significant superiority of escitalopram compared with other antidepressants clinically relevant? Int Clin Psychopharmacol. 2009;24(3):111–8. https://doi.org/10.1097/YIC.0b013e32832a8eb2.
Kennedy SH, Andersen HF, Lam RW. Efficacy of escitalopram in the treatment of major depressive disorder compared with conventional selective serotonin reuptake inhibitors and venlafaxine XR: a meta-analysis. J Psychiatry Neurosci. 2006;31(2):122–31.
Coley RY, Boggs JM, Beck A, Hartzler AL, Simon GE. Defining success in measurement-based care for depression: a comparison of common metrics. Psychiatr Serv. 2020;71(4):312–8. https://doi.org/10.1176/appi.ps.201900295.
Schueller SM, Kwasny MJ, Dear BF, Titov N, Mohr DC. Cut points on the Patient Health Questionnaire (PHQ-9) that predict response to cognitive-behavioral treatments for depression. Gen Hosp Psychiatry. 2015;37(5):470–5. https://doi.org/10.1016/j.genhosppsych.2015.05.009.
National Committee for Quality Assurance. HEDIS depression measures for electronic clinical data. 2022 January 8, 2022 [cited 2022 01/18/2022].
Zimmerman M, Walsh E, Friedman M, Boerescu DA, Attiullah N. Identifying remission from depression on 3 self-report scales. J Clin Psychiatry. 2017;78(2):177–83. https://doi.org/10.4088/JCP.16m10641.
Alam MY, Jacobsen PL, Chen Y, Serenko M, Mahableshwarkar AR. Safety, tolerability, and efficacy of vortioxetine (Lu AA21004) in major depressive disorder: results of an open-label, flexible-dose, 52-week extension study. Int Clin Psychopharmacol. 2014;29(1):36–44. https://doi.org/10.1097/YIC.0000000000000010.
Acknowledgements
The authors thank Lindsey Kirkland, PhD, Soniya Patel, PhD, and Kevin O’Regan, PhD (ApotheCom, Yardley, PA, USA), for editorial and medical writing assistance on the outline, first draft, and all subsequent drafts, which was funded by Janssen Scientific Affairs, LLC. The data from this study were previously presented at the Anxiety and Depression Association of America (ADAA) Conference in March 2022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research was supported by Janssen Scientific Affairs, LLC.
Conflict of interest
Samuel Wilkinson discloses receiving contract research funding from Janssen, Oui Therapeutics, and Sage Therapeutics for the conduct of clinical trials (administered through Yale University); he also discloses receiving consulting fees from Janssen, Oui Therapeutics, and Sage Therapeutics. Meredith Castro, Mary Pat Petrillo, Brianne Brown, and Patricia Cabrera are employees of Janssen Scientific Affairs, LLC, and hold stock in Johnson & Johnson, Inc. Naim Zaki, Stephane Borentain, Dong Jing Fu, and Ibrahim Turkoz are employees of Janssen Research & Development, LLC, and hold stock in Johnson & Johnson, Inc. Rayan Al Jurdi receives research funding from Janssen Pharmaceuticals, Inc. and Neurolief, Inc. and also serves on the Treatment Resistant Depression (TRD) Speaker Bureau, which is financially supported by Janssen Pharmaceuticals, Inc. Liping Sun is an employee of Cytel, Inc.
Ethics approval
This study (ClinicalTrials.gov identifier: NCT02782104) was approved by Independent Ethics Committees or Institutional Review Boards. This study was conducted in accordance with the ethical principles that have their origin in the Declaration of Helsinki and that are consistent with Good Clinical Practices and applicable regulatory requirements.
Consent to participate
All patients signed informed consent for participation in this study.
Consent for publication
Not applicable.
Availability of data and material
The data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.janssen.com/clinical-trials/transparency. As noted on this site, requests for access to the study data can be submitted through Yale Open Data Access (YODA) Project site at http://yoda.yale.edu.
Code availability
Not applicable.
Author contributions
Conceptualization: SW, MC, SB, DJF, IT. Methodology: SW, MC, SB, DJF, IT, LS, BB. Software: IT, LS. Validation: IT, LS. Formal analysis: NZ, IT, LS. Investigation: RAJ, MC, DJF. Resources/funding acquisition: MC. Data curation: MC, IT. Writing, reviewing, and editing: SW, RAJ, MC, MPP, NZ, SB, DJF, IT, LS, BB, PC. Visualization: MC, IT. Supervision/project administration: MC. All authors contributed to drafting several versions of the manuscript and revised it critically for important intellectual content, have read and approved the final submitted manuscript, and agree to be accountable for the work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Castro, M., Wilkinson, S.T., Al Jurdi, R.K. et al. Efficacy and Safety of Esketamine Nasal Spray in Patients with Treatment-Resistant Depression Who Completed a Second Induction Period: Analysis of the Ongoing SUSTAIN-3 Study. CNS Drugs 37, 715–723 (2023). https://doi.org/10.1007/s40263-023-01026-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-023-01026-3