Abstract
Purpose
Lymph node metastasis (LNM) is a vital prognosis factor in patients with papillary thyroid microcarcinoma (PTMC). The study tried to identify clinicopathological factors for LNM of PTMC.
Methods
The clinicopathological data of 1031 patients with PTMC were extracted and analyzed. Univariate and multivariate analyses were used to identify risk factors associated with cervical lymph node metastasis. ROC analysis was used to determine the ideal critical points of the sum of the maximum diameter of multifocal in a unilateral lobe.
Results
The probability of LNM, central lymph node metastasis (CLNM) and lateral lymph node metastasis(LLNM)of PTMC patients were 35.6, 33.7 and 5.6%, respectively. In addition, 1.9% PTMC had LLNM only. Male, age ≤ 40 years, tumor largest diameter ≥ 5 mm, multifocal, non-uniform echoic distribution, the sum of the maximum diameter of multifocal in a unilateral lobe ≥ 8.5 mm, tumors in the lower pole location were prone to CLNM. Ultrasound mix-echo, the sum of the maximum diameter of the multifocal ≥ 10.75 mm, tumors in the upper pole location were extremely prone to LLNM. T3 were prone to LLNM or skip LLNM.
Conclusions
According to the clinicopathological characteristics of PTMC, the cervical lymph nodes should be correctly evaluated to guide the surgical treatment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Papillary thyroid carcinoma (PTC) is the most frequent malignancy of the thyroid gland. The incidence of PTC rapidly increases worldwide in recent years and up to 43% of all these thyroid cancers are papillary thyroid microcarcinoma (PTMC) with a maximum diameter of 1.0 cm or less [1]. At present, it remains controversial whether PTMC needs surgery. Ito [2] does not recommend surgery for some PTMC patients. Sugitani et al. [3] suggest that the delayed surgery has no effect on outcomes. Similarly, the guideline of the American Thyroid Association (ATA) suggests that cautious observation is a safe and effective alternative to immediate surgical resection for PTMC [4]. However, PTC has a high metastasis rate especially central lymph node metastasis (CLNM), about 20 to 50% of PTC patients have CLNM [5]. Moreover, the CLNM can significantly affect the prognosis of thyroid cancer [5, 6]. PTMC is also potentially aggressive with a lymph node metastasis (LNM) rate of 37.3% [7]. The ultrasonic examination is important for the preoperative diagnosis of CLNM for PTMC patients, but this ultrasonic diagnosis can be difficult in some cases. We conducted this retrospective study of the clinicopathological features of PTMC to explore the lymph node metastasis of PTMC, and to provide evidence that is helpful for the clinical management of PTMC.
Materials and methods
General clinical materials
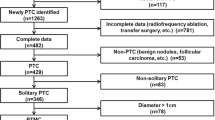
During the period from October 2010 to January 2017, 1031 cases of PTMC in the department of Surgical Oncology in Hangzhou First People’s Hospital were included in the study. Among these patients, 1010 cases were preoperatively diagnosed or suspected as PTMC by either fine needle aspiration(FNA) (518 cases) or B ultrasound (492 cases), and 21 cases were initially diagnosed as benign nodular goiter (19 cases) or hyperthyroidism (2 cases). All cases were confirmed to be PTMC successively by the intraoperative frozen and postoperative pathologic examination of resected tumor tissues. Criteria for admission: patients who had unilateral/bilateral PTMC in first surgery. Exclusion criteria: patients who had a history of other malignant tumors. This study was approved by the Ethics committee of our hospital and stayed in line with STROCSS criteria [8] with a unique identifying number (UIN: 3665) from the Research Registry. The data were analyzed in relation to gender, age at diagnosis, tumor largest diameter, capsular invasion, T staging, multifocal, the sum of the tumor diameter if multifocal, bilateral, tumor location (upper third, middle third, lower third and isthmus), B ultrasonography characteristics such as ultrasound intensity, composition, echoic distribution, tumor border, shape, calcification, aspect ratio, blood flow, and LNM that includes CLNM and lateral lymph node metastasis (LLNM). To evaluate the suspicious lateral lymph node metastasis, we used FNA for lymph node size larger than or equal to 1 cm and intraoperative frozen pathological examination for those smaller than 1 cm. The size and number of thyroid tumors, and the presence or absence of capsular invasion were recorded by high-frequency B ultrasonography. Each thyroid cancer patient was independently examined by two ultrasonographists. If two ultrasonographists had different opinions, the third ultrasonographist would participate in the examination to verify the data.
Surgical procedures and Pathological examination
The PTMC patients in this study underwent open surgeries by a surgical team. The primary lesion was performed simultaneously with the cervical lymph node dissection. Ipsilateral lobe and isthmus resection was performed for unilateral primary lesions. Total thyroidectomy was performed for unilateral primary lesions requiring iodine 131 treatment. We generally performed the lymph node dissection of ipsilateral cervical central VI, which is recommended in China. The mean number of removed lymph nodes of the central compartment is 9.2. Lymph node dissections of contralateral cervical central VI and bilateral lateral compartments were performed only if there was lymph node metastasis in these compartments. As for all bilateral primary lesions, total thyroidectomy and bilateral cervical central VI lymph node dissection were performed, and lateral lymph node dissection was performed only for those having lateral lymph node metastasis. Additional lateral compartment neck dissections including levels II, III, IV and Vb would be performed if metastases were present in the lateral compartment. Level I and Va lymph nodes were spared unless there were clinically positive lymph nodes. Two expert pathologists independently examined the surgical specimens. Specifically, the histological type, number, largest diameter, and the presence/absence of capsular invasion in the primary thyroid tumor and the lymph node were determined.
Follow-up
In the present study, each patient was followed up for more than a year. 92% patients (949 cases) were followed up from 15 to 90 months and none of them had distant metastasis. However, six patients suffered from local recurrence (one case of lateral compartment, and five cases of contralateral glands).
Statistical analysis
The clinicopathologic features were compared using the Pearson Chi-square test. In multivariate analysis, the odds ratio (OR) and the 95% confidence interval (CI) for relationships between each significant variable and lymph node metastasis were calculated using backward stepwise binary logistic regression. P < 0.05 was considered to indicate a statistically significant difference. The receiver-operating characteristic (ROC) analysis was used to identify the cutoff point of the sum largest diameter of multifocal tumors for defining the risk of CLNM and LLNM. All analyses were performed using IBM SPSS, version 20.
Results
Clinicopathological characteristics of PTMC
There were 1031 patients with PTMC involved in this study. The mean age of these patients is 45.99 ± 10.94 yr (ranged from 14 to 74). 35.6% (367/1031) of cases exhibited neck lymph node metastasis including central compartment and lateral compartment. 33.7% (347/1031) had CLNM and 5.6% (58/1031) had LLNM. We further found that 38 patients (3.7%) had both CLNM and LLNM, the situation of which should be considered to be relatively severe. In addition, to our surprise, 1.9% (20/1031) presented only LLNM (Table 1).
The serum parathyroid hormone and calcium levels were assayed to assess the postoperative parathyroid function. There was no permanent hypoparathyroidism, which was defined as persistently low blood calcium levels (< 2 mmol/L) and subnormal parathyroid hormone levels by 6 months after surgery. The presence of recurrent laryngeal nerve (RLN) injury was determined by reviewing the surgical records and postoperative laryngoscopy reports. 16 patients presented transient RLN paralyses and recovered within 2–3 months of surgery. There was no permanent RLN paralysis. The morbidities of hypoparathyroidism and RLN injury in this study were lower than the literatures (permanent hypoparathyroidism:3.5–6.3%; permanent RLN injury: 1.3–3.6%) [9,10,11,12]. In the study, 0.39% (4/1031) patients exhibited pleura rupture (which was recognized during surgery and repaired), which is relatively higher than the literatures [13, 14]. The reason may be that there are more patients compared to the literatures undergoing dissections of lymph nodes posterior to the right recurrent laryngeal nerve. Some studies have reported that the incidence of chylous leakage in patients was from 0.5 to 8.3% [15,16,17]. In our study, 2.04% (21/1031) patients presented chylous leakage which resolved within 2–3 weeks of surgery. Horner’s syndrome, a very rare complication with less than 30 cases so far reported in the literature [18], was not observed in this study.
Risk factors of LNM
First, we studied the relationship between these risk factors and LNM. And univariate analysis showed that LNM was significantly associated with tumor characteristics of age, gender, tumor largest diameter, capsular invasion, T staging, multifocality, bilateral, echoic distribution, calcification, and blood flow (Table 2). However, when these variables were included in multivariate analysis, bilateral was not a statistically significant predictor of the LNM. Some subgroups of the variables left were also excluded. Finally, male, young age (≤ 40 years), tumor largest diameter greater than 7 mm, T3, and multifocality were independent risk factors with odds ratios of 1.954, 1.916, 3.964, 1.924, and 1.858, respectively (Table 2).
Risk factors of CLNM
After the integral analysis of LNM, we further studied the risk factors of CLNM. Univariate analysis showed that PTMC patients with CLNM were significantly associated with tumor characteristics of male, age, tumor largest diameter, capsular invasion, multifocality, bilateral, T staging, echoic distribution, calcification and blood flow (Table 2). However, when these variables were included in multivariate analysis, five factors were excluded. As a result, male, young age (≤ 40 years), tumor largest diameter greater than 5 mm, the multifocality, and non-uniform echoic distribution were independent risk factors with odds ratios of 1.961, 1.888, 1.917, 1.976, and 1.367 respectively (Table 2).
Risk factors of LLNM
Univariate analysis showed that PTMC patients with LLNM were significantly associated with clinicopathological characteristics of tumor largest diameter, capsular invasion, T staging, ultrasound intensity, shape, and blood flow (Table 2). Nevertheless, after being included in multivariate analysis, tumor largest diameter, capsular invasion, ultrasound shape, and blood flow were excluded. T3 and ultrasound mix-echo were independent risk factors with odds ratios of 3.575, and 4.729, respectively (Table 2).
LNM is related to the location of the gland in PTMC patients
In this study, there were 803 PTMC patients with single lesions in a unilateral lobe, and we divided the gland into four parts according to the upper pole, the middle pole, the lower pole, and the isthmus. The CLNM rates of the lesions in the upper pole, the middle pole, the lower pole, and the isthmus were 25.30, 29.40, 39.70, and 28.30%, respectively (P = 0.011). However, PTMC patients with lesions in the upper pole were most prone to LLNM, with a metastasis rate of 8.10% (P = 0.013). The probabilities of CLNM and LLNM in PTMC patients with lesions in the isthmus were 28.30 and 1.90%, respectively (Table 3).
LNM is related to the multifocal in PTMC patients
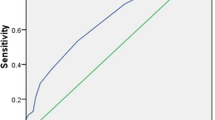
In this study, there were 140 unilateral lobes with multifocal PTMC. We not only found that patients with multifocal PTMC were associated with CLNM, but also found that the sum of the maximum diameter of multifocal in a unilateral lobe was closely related to CLNM and LLNM. The ROC analysis showed that the areas under the ROC curves for the prediction of CLNM and LLNM by the risk factor sum ≥ 8.5 and ≥ 10.75 mm were 0.644, 0.687, respectively (Fig. 1a, b).
Outcomes of receiver-operating characteristic (ROC) curve analyses. a ROC curve analysis for the sum of the maximum diameter of multifocal in a unilateral lobe of CLNM: An index point ≥ 8.5 mm of the sum of the maximum diameter of multifocal was found to be the optimal point to distinguish between PTMC with and without CLNM. The sensitivity and specificity were 64.4 and 59.3%, respectively, with an AUC of 0.644. b ROC curve analysis for the sum of the maximum diameter of multifocal in a unilateral lobe of LLNM: An index point ≥ 10.75 mm of the sum of the maximum diameter of multifocal was found to be the optimal point to distinguish between PTMC with and without LLNM. The sensitivity and specificity were 58.3 and 74.2%, respectively, with an AUC of 0.687
Discussion
Generally most of PTMC has a good prognosis, but some PTMC patients can suffer from cancer death or poor quality of life. These poor outcome cases mainly occur in PTMC with metastatic lymph node or/and invasion of thyroid peripheral tissues, such as recurrent laryngeal nerve, internal jugular vein, trachea or esophagus. Compared with the intra-thyroid PTC group, the PTC group with LNM showed higher recurrence rates (5.2 vs. 31.5%), and higher disease-specific mortality (1.3 vs. 12.6%) [19]. In this study, no distant metastasis was found in all 1031 PTMC patients enrolled. Therefore, the distant metastasis is rare in PTMC and we focused on the association of LNM of PTMC patients with the clinicopathological characteristics and B ultrasonography characteristics. Our data analysis indicated a significant correlation between LNM and several risk factors.
Some studies used a tumor largest diameter of 5, 5.5, 7 mm to indicate the aggressiveness of PTMC [20,21,22,23]. In the present study, the tumor largest diameter ≥ 5 mm was an independent risk factor for CLNM. We suggested that patients with PTMC greater than 5 mm must be paid attention to the CLNM. Furthermore, the tumor largest diameter ≥ 7 mm was an independent risk factor for LNM. Therefore, for patients with PTMC larger than 7 mm, we should pay attention not only to CLNM, but also to LLNM.
The association of PTMC with gender and age remains controversial. Jeon et al. [24] showed that age ≥ 45 years was at a higher risk of LNM while male was not an independent risk factor. On the contrary, some studies revealed that both male and younger age were risk factors [21, 22, 25, 26]. In our study, male was an independent risk factor for LNM and CLNM, rather than for LLNM. We also found that age ≤ 40 years was an independent risk factor for LNM and CLNM, rather than for LLNM. These findings suggested that male and younger PTMC patients were prone to cervical LNM, especially CLNM (Table 2).
Additionally, we found that the multifocal lesion in PTMC was an important risk factor in CLNM. This finding is consistent with the data reported [7, 20, 21, 26,27,28,29]. Furthermore, with the increase of the sum of the maximum diameter of multifocal in a unilateral lobe, not only was the probability of CLNM increased, but also the probability of LLNM (Table 4). In the group with the sum of the maximum diameter > 8 mm, the rate of CLNM was 47.50%. The ROC analysis showed that the optimal point was 8.5 mm of the sum of the maximum diameter of multifocal. The sensitivity and specificity were 64.4 and 59.3%, respectively. In the group with the sum of the maximum diameter > 11 mm, the rate of LLNM was 19.40%. The ROC analysis showed that the optimal point was 10.75 mm of the sum of the maximum diameter of multifocal. The sensitivity and specificity were 58.3 and 74.2%, respectively (Fig. 1a, b). Therefore, we should pay more attention to the PTMC patients with multifocal lesion and their sum of the maximum diameter.
In clinical practice, we used to observe a phenomenon with small tumor lesion but invaded extra thyroid tissues. There were 59 PTMC patients with T3 in our study. We found that T3 was associated with LLNM (P = 0.001, OR = 3.575), which was similar to Lu et al. [26] At the same time, we found 20 PTMC patients with only LLNM. Of these, four cases belonged to stage T3, and 16 cases were intra-thyroid lesions. The only LLNM rates of PTMC patients with T3 and with intra-thyroid lesion were 6.78% and 1.66%, respectively (P < 0.05), the former being 4.08 times more than the latter. The above results demonstrated that PTMC patients with T3 were prone to having LLNM or skip LLNM. LLNM shall be considered as a risk factor for distant metastatic spread and for cancer-related death [30]. Thus, we suggest that attention should be paid to the PTMC patients with T3 (Table 2).
The location of PTMC was related to the compartment of cervical lymph node metastasis [31]. We found that location in the lower third of the thyroid lobe conferred a higher risk for CLNM (P < 0.05), and that the location of upper third was related to a higher risk for LLNM (P < 0.05). However, the PTMC patients with lesions in the thyroid isthmus were prone to CLNM and rare to LLNM (Table 3). Thus, we should pay attention to estimate the compartment of cervical lymph node metastasis according to the location of the solitary primary tumor in the thyroid.
With the development of ultrasound technology, the diagnosis of PTMC has been improved. However, limited studies were performed to correlate LNM with ultrasound characteristics. A previous report [22] has reported that blood flow (present vs. absent) may improve the efficacy of predicting LNM (P < 0.001, OR = 5.3). In our study, blood flow was not a significant predictor of the LNM, CLNM or LLNM (P > 0.05). The present result indicated that PTMC patients with blood flow had limited value in predicting cervical LNM. But in this study, we found that the probability of CLNM of PTMC patients with non-uniform or uniform echoic distribution was significantly different (P = 0.003). It showed non-uniform echoic distribution was an independent predictive factor for CLNM (OR = 1.367). As for ultrasound intensity, we found that strong correlations of PTMC patients with mix-echo and LLNM (P < 0.01, OR = 4.729) (Table 2). These phenomenon suggested that PTMC patients with features of non-uniform echoic distribution and mix-echo should be paid attention to their respective the cervical CLNM and LLNM. In addition, other ultrasound features such as shape, composition, tumor border, calcification, and aspect ratio, were not associated with the cervical LNM.
There are some limitations in our study. First, the study is a single center study though it has a large sample size. Second, we did not divide the subtypes of PTMC so there may be some statistical bias. Third, it may omit few occult lymph node metastases because we did not perform lymph node dissection in patients not identified having contralateral lymph node metastasis. Finally, due to the difficulty in standardization of preoperative imaging diagnostic techniques of the lymph node assessment in the central compartment, the proportion of the suspicious lymph nodes in the central compartment was not calculated. Additional studies are required to address these issues.
Conclusion
We found that the probability of cervical LNM was closely related to the clinical and pathological characteristics of PTMC. Male, age ≤ 40 years, tumor largest diameter ≥ 5 mm, multifocal, non-uniform echoic distribution, the sum of the maximum diameter of multifocal in a unilateral lobe ≥ 8.5 mm, tumors in the lower pole location were prone to CLNM. Ultrasound mix-echo, the sum of the maximum diameter of the multifocal ≥ 10.75 mm, tumors in the upper pole location were extremely prone to LLNM. Patients with T3 were prone to LLNM or skip LLNM. Therefore, according to the clinical and pathological characteristics of PTMC, the regional lymph nodes should be correctly evaluated to guide the surgical treatment of cervical LNM in PTMC.
References
Leenhardt L, Grosclaude P, Chérié-Challine L (2004) Increased incidence of thyroid carcinoma in France: a true epidemic or thyroid nodule management effects? Report from the French Thyroid Cancer Committee. Thyroid 14:1056–1060
Ito Y, Uruno T, Nakano K, Takamura Y, Miya A, Kobayashi K, Yokozawa T, Matsuzuka F, Kuma S, Kuma K, Miyauchi A (2003) An observation trial without surgical treatment in patients with papillary microcarcinoma of the thyroid. Thyroid 13:381–387
Sugitani I, Toda K, Yamada K, Yamamoto N, Ikenaga M, Fujimoto Y (2010) Three distinctly different kinds of papillary thyroid microcarcinoma should be recognized: our treatment strategies and outcomes. World J Surg 34:1222–1231
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, Pacini F, Randolph GW, Sawka AM, Schlumberger M, Schuff KG, Sherman SI, Sosa JA, Steward DL, Tuttle RM, Wartofsky L (2016) 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: the American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26:1–133
Kim YS, Park WC (2012) Clinical predictors of right upper paraesophageal lymph node metastasis from papillary thyroid carcinoma. World J Surg Oncol 10:164
Podnos Y, Smith D, Wagman L, Ellenhorn J (2005) The implication of lymph node metastasis on survival in patients with well-differentiated thyroid cancer. Am Surg 71:731–734
Zhao Q, Ming J, Liu C, Shi L, Xu X, Nie X, Huang T (2013) Multifocality and total tumor diameter predict central neck lymph node metastases in papillary thyroid microcarcinoma. Ann Surg Oncol 20:746–752
Agha RA, Borrelli MR, Vella-Baldacchino M, Thavayogan R, Orgill DP (2017) The STROCSS statement: strengthening the reporting of cohort studies in surgery. Int J Surg 46:198–202
Kim SK, Woo JW, Lee JH, Park I, Choe JH, Kim JH, Kim JS (2016) Prophylactic central neck dissection might not be necessary in papillary thyroid carcinoma: analysis of 11,569 cases from a single Institution. J Am Coll Surg 222:853–864
Zhao W, You L, Hou X, Chen S, Ren X, Chen G, Zhao Y (2017) The effect of prophylactic central neck dissection on locoregional recurrence in papillary thyroid cancer after total thyroidectomy: a systematic review and meta-analysis: pCND for the locoregional recurrence of papillary thyroid cancer. Ann Surg Oncol 24(8):2189–2198. https://doi.org/10.1245/s10434-016-5691-4
Barczyński M, Konturek A, Stopa M, Nowak W (2013) Prophylactic central neck dissection for papillary thyroid cancer. Br J Surg 100:410–418
Zetoune T, Keutgen X, Buitrago D, Aldailami H, Shao H, Mazumdar M, Rd FT, Zarnegar R (2010) Prophylactic central neck dissection and local recurrence in papillary thyroid cancer: a meta-analysis. Ann Surg Oncol 17:3287
Lee SJ, Lee DJ, Kim MC, Im UJ (2010) Pneumothorax in a post-anesthetic care unit after right thyroidectomy with left neck dissection -A case report. Korean J Anesthesiol 59:429–432
Koeppen M, Scott B, Morabito J, Fiegel M, Eckle T (2017) Pneumomediastinum and bilateral pneumothoraces causing respiratory failure after thyroid surgery. Case Rep Anesthesiol 2017. https://doi.org/10.1155/2017/8206970
Roh JL, Kim DH, Park CI (2008) Prospective identification of chyle leakage in patients undergoing lateral neck dissection for metastatic thyroid cancer. Ann Surg Oncol 15:424–429
PhD GWM, PhD XCM, PhD YXM, Wei HM, Koch WM (2012) Prospective randomized trial of high versus low negative pressure suction in management of chyle fistula after neck dissection for metastatic thyroid carcinoma. Head Neck 34:1711–1715
Kupferman ME, Patterson DM, Mandel SJ, Livolsi V, Weber RS (2004) Safety of modified radical neck dissection for differentiated thyroid carcinoma. Laryngoscope 114:403–406
Harding JL, Sywak MS, Sidhu S, Delbridge LW (2004) Horners syndrome in association with thyroid and parathyroid disease. ANZ J Surg 74:442
Liu F, Kuo S, Hsueh C, Chao T, Lin J (2015) Postoperative recurrence of papillary thyroid carcinoma with lymph node metastasis. J Surg Oncol 112:149–154
Vasileiadis I, Karatzas T, Vasileiadis D, Kapetanakis S, Charitoudis G, Karakostas E, Kouraklis G (2014) Clinical and pathological characteristics of incidental and nonincidental papillary thyroid microcarcinoma in 339 patients. Head Neck 36:564–570
Tao Y, Wang C, Li L, Xing H, Bai Y, Han B, Liu Z, Yang X, Zhu S (2017) Clinicopathological features for predicting central and lateral lymph node metastasis in papillary thyroid microcarcinoma: analysis of 66 cases that underwent central and lateral lymph node dissection. Mol Clin Oncol 6:49–55
Zhao C, Jiang W, Gao Y, Niu W, Zhang X, Xin L (2017) Risk factors for lymph node metastasis (LNM) in patients with papillary thyroid microcarcinoma (PTMC): role of preoperative ultrasound. J Int Med Res 45:1221–1230
Lee KJ, Cho YJ, Kim SJ, Lee SC, Kim JG, Ahn CJ, Lee DH (2011) Analysis of the clinicopathologic features of papillary thyroid microcarcinoma based on 7-mm tumor size. World J Surg 35:318–323
Jeon MJ, Kim WG, Choi YM, Kwon H, Lee YM, Sung TY, Yoon JH, Chung KW, Hong SJ, Kim TY, Shong YK, Song DE, Kim WB (2016) Features predictive of distant metastasis in papillary thyroid microcarcinomas. Thyroid 26:161–168
Sapuppo G, Palermo F, Russo M, Tavarelli M, Masucci R, Squatrito S, Vigneri R, Pellegriti G (2017) Latero-cervical lymph node metastases (N1b) represent an additional risk factor for papillary thyroid cancer outcome. J Endocrinol Invest 40:1–9
Lu ZZ, Zhang Y, Wei SF, Li DS, Zhu QH, Sun SJ, Li M, Li LI (2015) Outcome of papillary thyroid microcarcinoma: study of 1990 cases. Mol Clin Oncol 3:672–676
Wu YL, Sun JM, Zhang JJ, Cui QX, Zheng WH, Li XR (2017) Clinicopathological characteristics of papillary thyroid microcarcinoma and risk factors for central lymph node metastasis. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 52:426–429
Chen C, Chen HZ, Lyu CH, Xue S, Yu NK, Chen X (2016) Clinicopathological features of thyroid papillary microcarcinoma of which the maximum diameter is less than 5 mm: a series of 487 cases. Zhonghua Wai Ke Za Zhi 54:870–874
Gui CY, Qiu SL, Peng ZH, Wang M (2017) Clinical and pathologic predictors of central lymph node metastasis in papillary thyroid microcarcinoma: a retrospective cohort study. J Endocrinol Invest 41:403–409
Sapuppo G, Tavarelli M, Russo M, Malandrino P, Belfiore A, Vigneri R, Pellegriti G (2018) Lymph node location is a risk factor for papillary thyroid cancer-related death. J Endocrinol Invest 1–5. https://doi.org/10.1007/s40618-018-0865-5
Zhang L, Wei WJ, Ji QH, Zhu YX, Wang ZY, Wang Y, Huang CP, Shen Q, Li DS, Wu Y (2012) Risk factors for neck nodal metastasis in papillary thyroid microcarcinoma: a study of 1066 patients. J Clin Endocrinol Metab 97:1250–1257
Acknowledgements
The authors are grateful to all members of the Department of Surgical Oncology at Hangzhou First People’s Hospital for assistance with surgical treatment of PTMC and clinical data collection.
Funding
This work was supported by the Key Project of Scientific and Technological Innovation in Hangzhou (20131813A08).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Luo, Y., Zhao, Y., Chen, K. et al. Clinical analysis of cervical lymph node metastasis risk factors in patients with papillary thyroid microcarcinoma. J Endocrinol Invest 42, 227–236 (2019). https://doi.org/10.1007/s40618-018-0908-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-018-0908-y