Abstract
Background
Gastroenteropancreatic neuroendocrine tumours (GEP-NET) are a rare, life-threatening type of cancer. The survival benefit of 177Lu-DOTATATE has been demonstrated in GEP-NET patients. Health technology assessment bodies require data on health-related utility impacts of treatment. A cancer-specific instrument, EORTC QLQ-C30, was used to collect the data for 177Lu-DOTATATE within clinical studies, but utility-based instruments were not included.
Objective
The main aim of this study was to compare EQ-5D-3L and QLU-C10D utilities obtained from EORTC QLQ-C30 using two different approaches. A secondary aim was to analyse the EQ-5D-3L and QLU-C10D utilities of patients treated with 177Lu-DOTATATE versus best supportive care. A supplementary aim was to evaluate the effect of 177Lu-DOTATATE on patients’ health-related utility over time.
Methods
Three datasets were used for the analysis. NETTER-1 is a clinical trial, whilst ERASMUS and Guy’s and St. Thomas (GStT) are real-world datasets. Two mapping algorithms (response mapping and ordinary least square regression) were applied to generate EQ-5D-3L utilities from EORTC QLQ-C30. An algorithm was used to obtain QLU-C10D utilities from EORTC QLQ-C30.
Results
In all studies, EQ-5D-3L utilities were higher than QLU-C10D utilities at most time points measured, although the magnitude of the differences was small. In NETTER-1, EQ-5D-3L and QLU-C10D utilities were higher in the 177Lu-DOTATATE arm compared with the octreotide long-acting release (LAR) arm, overall and pre-progression. In all studies, patients’ health-related utilities seem to be maintained over time.
Conclusion
There were small differences between EQ-5D-3L and QLU-C10D utilities, but these did not translate to relative differences over time or between groups. In NETTER-1, patients in the 177Lu-DOTATATE arm had higher health-related utilities than patients in the octreotide LAR arm. Health-related utility may at least remain maintained in patients with GEP-NET receiving 177Lu-DOTATATE.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The magnitude of the differences between EQ-5D-3L and QLU-C10D utilities generated from EORTC QLQ-C30 scores is small. |
Health-related utility may at least remain maintained in patients with GEP-NET receiving 177Lu-DOTATATE. |
1 Introduction
Gastroenteropancreatic neuroendocrine tumours (GEP-NET) are a life-threatening type of cancer caused by the growth of tumours that originated from neuroendocrine cells anywhere along the gastrointestinal tract and the pancreas [1]. GEP-NET cancer is considered a rare disease, although its prevalence has risen steadily over the last four decades. In the USA, the highest incidence rate between 2000 and 2012 was 3.56 per 100,000 [2], whilst in the UK, the incidence is 1.3 per 100,000 per year [3].
GEP-NET are characterised by their ability to produce a large number of peptide hormones and biogenic amines that can cause a broad symptomatology [4, 5]. Symptoms are not specific to one disease, which leads to misdiagnosis or a delay in diagnosis [6]. Patients may manifest symptoms such as abdominal pain, anorexia, diarrhoea, nausea, skin lesions and bleeding [4]. Although the anatomical location of the primary tumour can vary widely in GEP-NET patients, two main types are distinguished: pancreatic NET (P-NET) and gastrointestinal NET (GI-NET). These groups have distinct genetic and molecular characteristics, prognosis is different and treatment options may differ for progressing patients [7].
Due to tumour growth, hormonal function and late diagnosis, GEP-NET cancers are considered complex to treat [6]. The primary treatment for GEP-NET consists of curative surgery. However, because of diagnosis delay, only a minority of patients can have curative surgery, as most present with advanced, inoperable primary tumours at time of first diagnosis [8]. There is, therefore, a need for therapies to extend survival of these patients, whilst preserving or improving their quality of life.
177Lu-DOTATATE (Lutathera®) is a novel therapy that has been developed as a treatment for GEP-NET. 177Lu-DOTATATE is a peptide receptor radionuclide therapy that works by binding to the somatostatin receptors of the GEP-NET. Lutetium (177Lu) oxodotreotide, the active substance in 177Lu-DOTATATE, generates radioactivity that kills the tumour cell, without harming the healthy neighbouring cells [9]. The survival benefit of 177Lu-DOTATATE has been demonstrated in the Neuroendocrine Tumour Therapy (NETTER-1) and ERASMUS studies [10, 11]. The time to deterioration (TTD) analysis of NETTER-1, which used a minimally important difference of 10 points in each EORTC QLQ-C30 domain score, revealed that 177Lu-DOTATATE prolonged the median time to quality-of-life deterioration compared with high-dose octreotide long-acting release (LAR) (60 mg every 4 weeks) [12]. The benefit of 177Lu-DOTATATE was particularly apparent for the EORTC QLQ-C30 global health status (median TTD 28.8 vs 6.1 months; HR 0.406), as well as for the following symptoms, recognised as disease relevant: diarrhoea, pain and fatigue [12].
177Lu-DOTATATE was granted market authorization by the European Medical Agency in 2017 and by the USA Food and Drug Administration in 2018. In addition, 177Lu-DOTATATE has received reimbursement from several health technology assessment (HTA) bodies, including NICE in England, SMC in Scotland and CADTH in Canada.
Many HTA agencies require health outcomes to be expressed as quality-adjusted life-years (QALYs) when making decisions about allocation of healthcare resources [13]. This is because QALYs are a generic measure, and their use allows for a direct and consistent comparison across disease areas. Also, QALYs are preference-based and reflect the general public’s preferences towards the avoidance of states of ill health. One of the most widely used preference-based patient-reported outcome instruments is the EQ-5D [14].
When the EQ-5D instrument is not administered in a clinical trial, health-related outcomes needed for HTA submissions can be obtained using mapping. Mapping involves the development and use of a statistical algorithm to generate health-related utilities from other measures of health outcomes, usually non-preference-based condition-specific measures [15].
One widely used condition-specific measure of health-related quality of life (HRQoL) is the European Organisation for Research and Treatment Quality of Life Questionnaire Core 30 (EORTC QLQ-C30), which was developed to capture disease-specific symptomatology and changes in symptomatology over time [16]. Similarly to most condition-specific measures, EORTC QLQ-C30 is non-preference based and cannot be used to calculate QALYs [17]. However, although not developed specifically as a preference-based measure, a health state classification system (QLU-C10D) that generates utility data from EORTC QLQ-C30 scores is available.
It has been suggested that condition-specific measures of HRQoL may be more sensitive or responsive than generic measures such as EQ-5D, and therefore may be preferred for use in economic evaluations [18]. It is thus important to understand the impact on utility estimates of using a generic-based approach versus a condition-specific-based approach.
Our study sought to serve multiple aims. The first aim was to compare EQ-5D-3L and QLU-C10D utilities obtained by using different approaches: mapping from EORTC QLQ-C3O to EQ-5D-3L and using an algorithm that generates QLU-C10D utilities from EORTC QLQ-C30 scores. The secondary aim was to analyse the EQ-5D-3L and QLU-C10D utilities of patients treated with 177Lu-DOTATATE versus octreotide LAR. An additional aim was to evaluate the effect of 177Lu-DOTATATE on patients’ HRQoL and derived utilities in three different studies.
2 Methods
2.1 Instruments
2.1.1 EQ-5D-3L
The EQ-5D is a well-established, generic, preference-based instrument that was developed and validated in the 1980s by the EuroQol Group [14]. The EQ-5D has been widely used to measure HRQoL across a variety of diseases due to its validity and generality [19]. The EQ-5D-3L, which represents the original version of the EQ-5D, evaluates patients’ HRQoL on five dimensions (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression), each being characterised by three levels of severity (no problems, some/moderate problems, extreme problems). In our study, the EQ-5D-3L was scored using the UK valuation set, which generates health utilities that range between − 0.594, which represents a health state worse than death, and 1, which indicates full health.
2.1.2 EORTC QLQ-C30
The EORTC QLQ-C30 is a cancer-specific HRQoL measure that has been widely used in cancer clinical trials due to its reliability and validity [20]. The EORTC QLQ-C30 comprises 30 questions that assess five functional domains (physical, emotional, social, role, cognitive), eight symptoms (fatigue, pain, nausea/vomiting, constipation, diarrhoea, insomnia, dyspnoea, and appetite loss), global health status/quality of life and financial impact [16]. Most items have a four-point scale (not at all, a little, quite a bit and very much), except the two items on global health status and quality of life, which have seven levels, ranging from ‘very poor’ to ‘excellent’. The scores for each of the 15 dimensions are converted to a 0–100 scale, with a higher score indicating a better quality of life for the functioning domains and global health status, but a poorer quality of life on the symptomatic scale [19].
2.1.3 QLU-C10D
The QLU-C10D is a health state classification system that was derived from the EORTC QLQ-C30, as a basis for a multi-attribute preference-based instrument [21]. The QLU-C10D contains 13 questions of the QLQ-C30’s 30 questions, which are combined into 10 items evaluating four functional domains (physical, role, social, emotional) and six symptoms (pain, fatigue, sleep, appetite, nausea, bowel problems). Out of the 10 QLU-C10D items, seven items are single and have a four-point scale (not at all, a little, quite a bit and very much), whereas the remaining three are composite [21]. In our study, the QLU-C10D was scored using the UK valuation set, which generates health utilities that range between − 0.083, which is considered slightly worse than death, and 1, which represents full health [22].
2.2 Datasets
Three datasets were used for the analysis. NETTER-1 was a clinical trial of 177Lu-DOTATATE, whilst ERASMUS and Guy’s and St. Thomas (GStT) are real-world evidence (RWE) studies of patients treated with 177Lu-DOTATATE.
2.2.1 NETTER-1
The NETTER-1 study was an international, multicentre, open-label, phase III trial, conducted in eight countries worldwide, which assessed the efficacy of 177Lu-DOTATATE in 229 progressing patients with midgut NET that had metastasized or that were inoperable. Progression was defined based on imaging with the use of the RECIST criteria. Patients were randomly assigned to receive either four infusions of 177Lu-DOTATATE every 8 weeks plus best supportive care, including octreotide LAR (30 mg), or octreotide LAR alone (60 mg) every 4 weeks. The primary endpoint was progression-free survival. Our analysis included 214 patients who completed the EORTC QLQ-C30 questionnaire at baseline and then every 12 weeks throughout the study. The NETTER-1 study is described in more detail elsewhere [10].
2.2.2 ERASMUS
The ERASMUS study was an open-label, single-arm study which evaluated the efficacy and safety of 177Lu-DOTATATE in progressing patients with GEP-NET and bronchial NET. A total of 1214 patients were treated with 177Lu-DOTATATE from January 2000 to January 2015 at a single centre in the Netherlands. Of these, 443 patients were included in the efficacy analysis. A total of 331 Dutch patients with GEP-NET included in the efficacy dataset, and who completed the EORTC QLQ-C30 questionnaire at one or more timepoints throughout the study (baseline, 3 weeks, 3 months, 6 months and biannually) form the basis of our analysis. More details about the ERASMUS study can be found elsewhere [11].
2.2.3 Guy’s and St. Thomas (GStT)
The GStT study is a real-world study that was conducted in a single UK centre. Data from patients with various subtypes of NET and treated with 177Lu-DOTATATE at Guy’s and St. Thomas Hospital, London, UK, were collected. The study followed up 62 patients who were administered the EORTC QLQ-C30 questionnaire at various timepoints in order to assess the impact of 177Lu-DOTATATE on their quality of life. Our analysis included only patients with GEP-NET in accordance with the marketing authorisation for 177Lu-DOTATATE (N = 47).
2.3 Statistical Analysis
2.3.1 Socio-Demographic and Patient-Reported Outcome Data
Summary information on socio-demographic characteristics and EORTC QLQ-C30 data for the NETTER-1, ERASMUS and GStT datasets are presented in Sect. 3.1. However, detailed information on the socio-demographic and baseline clinical characteristics of the NETTER-1 and ERASMUS studies can be found elsewhere [12, 23].
2.3.2 EQ-5D Utilities Generation
Two mapping approaches were applied to obtain EQ-5D-3L health-related utility values from EORTC QLQ-C30 scores. A systematic and critical review of mapping algorithms from EORTC QLQ-C30 to EQ-5D-3L has been published [24]. The authors assessed the external validity and established the generalizability of ten mapping algorithms using various criteria. Out of these, two performed particularly well: an ordinary least-square (OLS) regression [25], and a response mapping algorithm [26]. Given its external validity and the fact that the UK population valuation set can be applied, the response mapping algorithm published by Longworth et al. [26] was selected for our analysis.
Apart from the response mapping, Longworth et al. [26] also examined the efficiency of other commonly used mapping approaches, including OLS regression, which was ranked as the second-best performing algorithm. Since the population included in the Longworth et al. study [26] (breast, lung and multiple myeloma cancer) had a different QLQ-C30 profile compared with the GEP-NET patients from our studies, we also predicted EQ-5D-3L utilities using the OLS model developed by Longworth et al. [26].
In the indirect (response) mapping approach, separate multinomial logistic regressions are fitted to each of the five dimensions of the EQ-5D-3L to estimate the probability of being in each of the three response levels. The expected EQ-5D utility values are calculated by multiplying the probability of being in each response level by the corresponding value in the UK tariff, as shown in Formula 1. In the OLS model, it is assumed that the relationship between the dependent variable (EQ-5D) and the independent variable (EORTC QLQ-C30) can be expressed as a linear function [26]. In our study, we used the best performing OLS (OLS model 8) examined by Longworth et al. [26], which uses age as a covariate. All statistical analyses were conducted using Stata® version 15.1 (StataCorp LP, College Station, TX, USA).
where PM2 and PM3 are the probabilities of being in mobility level 2 and 3, respectively, and similarly for the other EQ-5D dimensions, self-care (SC), usual activities (UA), pain/discomfort (PD), anxiety/depression (AD).
2.3.3 QLU-C10D Utilities Generation
The QLU-C10D utilities were generated from EORTC QLQ-C30 scores by firstly deriving the level responses of the QLU-C10D items from the EORTC QLQ-C30 and secondly by applying UK utility weights. A more detailed description is offered below.
Firstly, the level responses of the QLU-C10D items (seven single and three composite) are derived from the corresponding 13 EORTC QLQ-C30 items using the algorithm developed by King et al. [27]. All seven single QLU-C10D items are equal to the level responses of the corresponding EORTC QLQ-C30 items. However, each of the three composite items (physical functioning, social functioning, and bowel problems) of the QLU-C10D use two different EORTC QLQ-C30 items, as shown in Formula 2. The physical functioning domain of the QLU-C10D combines the response levels of questions 2 and 3 of the EORTC QLQ-C30, referring to troubles taking a long and a short walk, respectively. The level response of the social functioning domain of the QLU-C10D is calculated as the maximum value of the response level of either question 26 or question 27 of the EORTC QLQ-C30, which evaluate how much family life and social activities, respectively, are affected by the condition. The QLU-C10D bowel problems item uses the maximum value of the response level of either question 16 or question 18 of the EORTC QLQ-C30, which refer to constipation and diarrhoea, respectively.
where PF and SF are the physical and social functioning domains of QLU-C10D, respectively; BP is the bowel problems item of QLU-C10D.
q2, q3, q26, q27, q16 and q18 are questions 2, 3, 26, 27, 16 and 18, respectively, of the EORTC QLQ-C30. 1, 2, 3 and 4 are all the possible level responses.
Secondly, utility decrements for each level response down from 1 (i.e., no problems) for each of the 10 QLU-C10D items are generated. For our study, these were retrieved from model 2 developed by Norman et al. [22] who generated utility weights for the QLU-C10D from a UK general population study. For each health state, the utility score is 1 minus the sum of each utility decrement calculated for each of the 10 items of the QLU-C10D.
2.3.4 Comparison Between EQ-5D and QLU-C10D Utilities
Comparisons between the EQ-5D-3L and QLU-C10D utilities were based on descriptions of the summary statistics such as mean, standard deviation, confidence intervals and via visual inspection.
3 Results
3.1 Descriptive Statistics
Table 1 presents the socio-demographic data of patients in all studies. Patients’ mean age was similar in all three studies, with the lowest and highest mean ages in ERASMUS (58 years old) and NETTER-1 (62 years old). Gender distribution was comparable across studies, with GStT having the highest percentage of males (55%) and NETTER-1 the lowest (48%). In all studies, most patients were diagnosed with GI-NET. However, there are important differences in the type of tumours included. NETTER-1 only included patients with mid-gut NET, whereas ERASMUS and GStT also included patients with foregut and hindgut tumours as well as pancreatic tumours.
Summary statistics of the EORTC QLQ-C30 data for NETTER-1, ERASMUS and GStT studies are presented in Tables 2, 3 and 4, respectively. The data suggests that in all studies, global health functioning is the most affected aspect of patients’ life, whereas physical, cognitive or social functioning are the least affected. In addition, in all studies, the most frequent symptoms experienced by patients are diarrhoea and fatigue, whilst nausea, vomiting and constipation are the least frequently reported.
3.2 Comparison Between EQ-5D-3L and QLU-C10D Utilities
Table 5 presents summary data for EQ-5D-3L and QLU-C10D health-related utilities in all three studies.
In NETTER-1, mapping to the EQ-5D-3L using both response mapping and OLS regression yielded higher utility values than using the QLU-C10D in all comparisons, although the magnitude of the differences was small. Moreover, the EQ-5D-3L utilities generated using response mapping and OLS model were remarkably similar, with the greatest difference in the 177Lu-DOTATATE arm, post-progression (0.78 vs 0.74, respectively). In the ERASMUS study, utilities derived by mapping to EQ-5D-3L (using both response mapping and OLS model) were slightly higher than those generated using the QLU-C10D algorithm. There was also little or no difference in utilities pre- and post-progression using either approach to obtain utilities. In the GStT study, utilities obtained by mapping to the EQ-5D-3L using both approaches were higher than QLU-C10D utilities, overall and pre-progression, but not post-progression. However, these findings are subject to uncertainty considering the small number of patients with progressive disease. Patients without disease progression had a higher average utility score than patients with progressive disease using either approach.
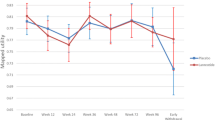
Figure 1 illustrates the trend of the EQ-5D-3L utilities obtained using both mapping approaches and QLU-C10D utilities over time in all studies. The small difference between EQ-5D-3L and QLU-C10D utilities is further strengthened by the similarity of their distribution, as shown in Fig. 2. Additionally, it can be seen that both response mapping and OLS regression yielded utilities with a remarkably similar distribution.
3.3 Comparison Between 177Lu-DOTATATE and Octreotide Long-Acting Release
In NETTER-1, the mean utility values generated using all approaches were higher in the 177Lu-DOTATATE arm compared with the octreotide LAR arm overall and pre-progression. For patients with progressive disease, the EQ-5D-3L utilities obtained by OLS regression and the QLU-C10D utilities were slightly higher in the octreotide LAR arm compared with the 177Lu-DOTATATE arm. However, we note that data for patients with progressive disease was limited.
3.4 The impact of 177Lu-DOTATATE on the HRQoL of GEP-NET patients
Figure 1 indicates that 177Lu-DOTATATE may maintain patients’ HRQoL over time. Moreover, Tables 6 and 7 present the EQ-5D-3L and QLU-C10D utilities, respectively, at each visit for the GStT study. The results are consistent between approaches and indicate that utilities followed a small upward trend over time. This suggests that the HRQoL of GEP-NET patients treated with 177Lu-DOTATATE in a real-world setting may at least remain maintained for a substantial time period following treatment. In addition, Fig. 3 is a visual representation of the above findings. Nevertheless, it is worth noting that since GStT is a small study, the results are subject to uncertainty, as reflected by the size of the confidence intervals.
4 Discussion
GEP-NET are life-threatening tumours that originate from the neuroendocrine cells along the gastrointestinal tract and pancreas. Until recently, treatment options, apart from curative surgery, were very limited [8].
Our study compared EQ-5D-3L and QLU-C10D utilities by two different approaches: mapping from EORTC QLQ-C30 scores to EQ-5D-3L utilities using response mapping and OLS regression and applying an algorithm to generate QLU-C10D utilities from EORTC QLQ-C30 scores. The data used in this study was retrieved from a clinical trial of 177Lu-DOTATATE and two real-world evidence studies.
The findings show that EQ-5D-3L utilities (generated using both mapping approaches) were higher than QLU-C10D utilities in all studies and at most time points measured. Nevertheless, the magnitude of the differences was small and did not translate to relative differences over time or between groups. This finding is further supported by the similarity of the standard deviations of the EQ-5D-3L and QLU-C10D utilities. However, based on the size of the standard deviations, it is worth noting that the utilities generated were associated with uncertainty.
In addition, our study suggests that in NETTER-1, a benefit in terms of patients’ quality of life was observed in the 177Lu-DOTATATE arm compared with the octreotide LAR arm, overall and pre-progression. These results are in line with the findings available in the literature [12].
An additional aim of this study was to assess the impact of 177Lu-DOTATATE on the HRQoL of patients with GEP-NET. All three studies indicated that the HRQoL of patients with GEP-NET receiving 177Lu-DOTATATE may at least remain maintained for a substantial period following treatment. In addition, all three studies suggested that global health functioning is the most affected aspect of patients’ life, whereas the most frequently experienced symptoms by GEP-NET patients are fatigue and diarrhoea. We highlight that our results are consistent with the findings of previous work [12]. We note that although the EQ-5D potentially captures the impact of symptoms within dimensions such as usual activities and pain/discomfort, the QLU-C10D directly captures all the most frequently experienced symptoms by GEP-NET patients.
Given that GEP-NET tumours are a rare disease, the use of three separate datasets is one of the advantages of our study as it strengthens the validity and generalizability of the findings. Moreover, although the GStT study is uncontrolled and hence not able to provide information on comparative HRQoL effects, it shows evidence that patients treated in a real-world setting with 177Lu-DOTATATE report appreciable HRQoL. In addition, the use of three separate datasets provided a large overall sample, which increases the accuracy of the results. It is also worth mentioning that the algorithms used have been previously validated in other external datasets [24], which further enhances the reliability of our findings.
We acknowledge some limitations of the study. Firstly, the number of observations for patients with progressive disease was limited in all three studies, particularly in NETTER-1 and GStT. This makes the comparison of the utilities between patients without and with progressive disease subject to a high degree of uncertainty. However, data for patients with progressive disease is often limited in clinical trials in oncology [28]. Secondly, we recognise that the mapping algorithms have higher errors when predicting EQ-5D values for patients with poorer health compared with those with better health. Nevertheless, this is commonly observed in mapping studies. Another limitation of the study is the uncertainty introduced by the mapping approaches themselves. As the EQ-5D-3L values generated by the mapping algorithms are predicted rather than observed, they are associated with more uncertainty than if EQ-5D-3L data had been directly collected within the clinical trials.
Using three separate datasets, we have shown that the HRQoL of GEP-NET patients treated with 177Lu-DOTATATE may at least remain maintained for a substantial period following treatment. The effectiveness and cost effectiveness of 177Lu-DOTATATE have been widely recognised by several HTA agencies [29, 30]. We believe our work adds to the existing body of evidence for the effectiveness of 177Lu-DOTATATE based on the results presented for the GStT study. To our knowledge, our study is the first to compare mapping to EQ-5D-3L and QLU-C10D scores from EORTC QLQ-C30 data. Although the predicted EQ-5D-3L and QLU-C10D utilities were comparable, in a cost-effectiveness analysis, the utility difference could lead to a distinct incremental cost-effectiveness ratio, which would have implications for reimbursement decision making. More research would be beneficial in validating our results using external datasets. Moreover, further research could include demonstrating the impact of 177Lu-DOTATATE using mapping approaches from EORTC QLQ-C30 data to the EQ-5D-5L. Additionally, research into the inclusion of the GINET-21 module of the EORTC QLQ-C30 into the mapping algorithm may be beneficial, as GINET-21 has been developed and validated specifically for patients with GEP-NET, and thus may provide better predictive ability in this patient population [31].
5 Conclusion
EQ-5D-3L utilities obtained using response mapping and OLS regression were higher than QLU-C10D utilities in all studies and in most instances. However, the differences were small and associated with substantial standard deviations. The NETTER-1 study showed that both EQ-5D-3L and QLU-C10D utilities were higher in the 177Lu-DOTATATE arm compared with the octreotide LAR arm, overall and pre-progression. In addition, our study indicated that the HRQoL of GEP-NET patients treated with 177Lu-DOTATATE may at least remain maintained for a substantial period following treatment. Future research could include generating health-related utilities by mapping from the EORTC QLQ-C30 to the EQ-5D-5L and assessing whether the inclusion of the GINET-21 module within the mapping algorithm reduces uncertainty in the predicted values for this patient population.
References
Díez M, Teulé A, Salazar R. Gastroenteropancreatic neuroendocrine tumors: diagnosis and treatment. Ann Gastroenterol. 2013;26(1):29.
Dasari A, et al. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017;3(10):1335–42.
Genus T, et al. Incidence and prevalence of neuroendocrine tumours in England. In: UKI NETS 15th national conference. BioScientifica. 2017.
Oladejo A. Gastroenteropancreatic neuroendocrine tumors (GEP-NETs)-approach to diagnosis and management. Ann Ibadan Postgrad Med. 2009;7(2):29–33.
Fraenkel M, et al. Incidence of gastroenteropancreatic neuroendocrine tumours: a systematic review of the literature. Endocr Relat Cancer. 2014;21(3):R153–63.
Basuroy R, et al. Delays and routes to diagnosis of neuroendocrine tumours. BMC Cancer. 2018;18(1):1122.
Yalcin S, et al. Gastroenteropancreatic neuroendocrine tumors: recommendations of Turkish multidisciplinary neuroendocrine tumor study group on diagnosis, treatment and follow-up. Arch Med Sci AMS. 2017;13(2):271.
Öberg KE. The management of neuroendocrine tumours: current and future medical therapy options. Clin Oncol. 2012;24(4):282–93.
Hennrich U, Kopka K. Lutathera®: the First FDA-and EMA-approved radiopharmaceutical for peptide receptor radionuclide therapy. Pharmaceuticals. 2019;12(3):114.
Strosberg J, et al. Phase 3 trial of 177Lu-Dotatate for midgut neuroendocrine tumors. N Engl J Med. 2017;376(2):125–35.
Brabander T, et al. Long-term efficacy, survival, and safety of [177Lu-DOTA0, Tyr3] octreotate in patients with gastroenteropancreatic and bronchial neuroendocrine tumors. Clin Cancer Res. 2017;23(16):4617–24.
Strosberg J, et al. Health-related quality of life in patients with progressive midgut neuroendocrine tumors treated with 177Lu-dotatate in the phase III NETTER-1 trial. J Clin Oncol. 2018;36(25):2578.
Devlin NJ, Lorgelly PK. QALYs as a measure of value in cancer. J Cancer Policy. 2017;11:19–25.
Devlin NJ, Brooks R. EQ-5D and the EuroQol group: past, present and future. Appl Health Econ Health Policy. 2017;15(2):127–37.
Longworth L, Rowen D. NICE DSU technical support document 10: the use of mapping methods to estimate health state utility values. 2011. Available from http://www.nicedsu.org.uk.
Aaronson NK, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. JNCI J Natl Cancer Inst. 1993;85(5):365–76.
Brazier J, Tsuchiya A. Preference-based condition-specific measures of health: what happens to cross programme comparability? Health Econ. 2010;19(2):125–9.
Versteegh MM, et al. Condition-specific preference-based measures: benefit or burden? Value Health. 2012;15(3):504–13.
Kennedy-Martin M, et al. Which multi-attribute utility instruments are recommended for use in cost-utility analysis? A review of national health technology assessment (HTA) guidelines. Eur J Health Econ. 2020;21:1245–57.
Velikova G, et al. Health-related quality of life in EORTC clinical trials—30 years of progress from methodological developments to making a real impact on oncology practice. Eur J Cancer Suppl. 2012;10(1):141–9.
King M, et al. QLU-C10D: a health state classification system for a multi-attribute utility measure based on the EORTC QLQ-C30. Qual Life Res. 2016;25(3):625–36.
Norman R, et al. UK utility weights for the EORTC QLU-C10D. Health Econ. 2019;28(12):1385–401.
Khan S, et al. Quality of life in 265 patients with gastroenteropancreatic or bronchial neuroendocrine tumors treated with [177Lu-DOTA0, Tyr3] octreotate. J Nucl Med. 2011;52(9):1361–8.
Doble B, Lorgelly P. Mapping the EORTC QLQ-C30 onto the EQ-5D-3L: assessing the external validity of existing mapping algorithms. Qual Life Res. 2016;25(4):891–911.
Versteegh MM, et al. Mapping Qlq-C30, Haq, and Msis-29 on Eq-5d. Med Decis Mak. 2012;32(4):554–68.
Longworth L, et al. Use of generic and condition-specific measures of health-related quality of life in NICE decision-making: a systematic review, statistical modelling and survey. Health Technol Assess. 2014;18(9):1–224.
King MT, et al. Australian utility weights for the EORTC QLU-C10D, a multi-attribute utility instrument derived from the cancer-specific quality of life questionnaire, EORTC QLQ-C30. Pharmacoeconomics. 2018;36(2):225–38.
Brogan AP, et al. Payer perspectives on patient-reported outcomes in health care decision making: oncology examples. J Manag Care Spec Pharm. 2017;23(2):125–34.
Palmer J, Leeuwenkamp OR. Cost-effectiveness of lutetium (177Lu) oxodotreotide vs everolimus in gastroenteropancreatic neuroendocrine tumors in Norway and Sweden. World J Clin Cases. 2020;8(20):4793.
Leeuwenkamp O, et al. Cost-effectiveness of Lutetium [177Lu] oxodotreotide versus best supportive care with octreotide in patients with midgut neuroendocrine tumors in France. J Med Econ. 2020;23(12):1534–41.
Yadegarfar G, et al. Validation of the EORTC QLQ-GINET21 questionnaire for assessing quality of life of patients with gastrointestinal neuroendocrine tumours. Br J Cancer. 2013;108(2):301–10.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
PHMR received financial support from Advanced Accelerator Application, a Novartis company, for conducting the study, including data analysis and preparation of the manuscript.
Conflict of interest
A. Soare, and Dr L. Longworth are full-time employees at PHMR. PHMR received financial support from Advanced Accelerator Application, a Novartis company, for the work, including data analysis and preparation of the manuscript. Oscar Leeuwenkamp is a full-time employee of Advanced Accelerator Application, a Novartis company, which funded the work.
Ethics approval
Not applicable (reanalysis of existing datasets based on studies that have previously received ethical approvals).
Consent to participate
Not applicable (reanalysis of existing datasets based on studies that have previously received ethical approvals).
Availability of data and material (data transparency)
Not available.
Code availability (software application or custom code)
Not available.
Consent for publication
Not applicable.
Author contributions
AS: led the drafting of the manuscript, contributed to the analysis, study design and interpretation. OL: contributed to the study design, interpretation and drafting of the manuscript. LL: led the study design and interpretation, contributed to the analysis and drafting of the manuscript. Oversight of the project.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Soare, IA., Leeuwenkamp, O. & Longworth, L. Estimation of Health-Related Utilities for 177Lu-DOTATATE in GEP-NET Patients Using Utilities Mapped from EORTC QLQ-C30 to EQ-5D-3L and QLU-C10D Utilities. PharmacoEconomics Open 5, 715–725 (2021). https://doi.org/10.1007/s41669-021-00280-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41669-021-00280-9