Abstract
Understanding patterns of habitat selection and factors affecting space use is fundamental in animal conservation. In urban landscapes, such knowledge can be used to advise householders on how best to manage their gardens for wildlife. In this study, we tracked 28 West European hedgehogs (Erinaceus europaeus), a species of conservation concern in the UK, in an area of high-density housing using radio and GPS tags to quantify patterns of habitat use and identify factors associated with the proportion of time spent in individual gardens. Both males and females exhibited a preference for residential gardens, but there were subtle differences between the sexes in relation to house type and front versus back gardens. Hedgehogs spent significantly more time in gardens where artificial food was provided, where a compost heap was present, if foxes (Vulpes vulpes) were infrequent visitors, if it rained overnight and as daylength increased (i.e., shorter nights); garden use was not significantly associated with variables potentially likely to reflect invertebrate prey abundance. These data suggest that the primary positive action that householders can undertake for urban hedgehogs is providing supplementary food. However, householders often feed hedgehogs after they know they are already visiting their garden. Consequently, the presence of artificial food may make it difficult to identify other important influences affecting garden use. Finally, we report that a GPS fix acquisition rate < 60% likely had no major effect on the results of our analyses, but should be a consideration in future studies using this technique on this species and in this habitat.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanisation poses significant threats to biodiversity worldwide (Seto et al. 2012; Concepción et al. 2015), causing habitat loss, degradation and fragmentation (Theodorou et al. 2020), human–wildlife conflicts (Soulsbury and White 2015; Adams 2016), the introduction of non-native species (McKinney 2006; Gaertner et al. 2017), exposure to pollutants (Ditchkoff et al. 2006; Grimm et al. 2008) and wildlife–vehicle collisions (Wright et al. 2020). Urban landscapes can, nonetheless, provide key habitats for animals (Goddard et al. 2013; Löki et al. 2019; Soanes and Lentini 2019; Spotswood et al. 2021) as well as refuge from predators (Møller 2012) and access to abundant resources (Oro et al. 2013). As a result, some species are now found only in urban areas (Oliveira Hagen et al. 2017; Soanes and Lentini 2019), whereas other species, including some of conservation concern (e.g., Coleman and Barclay 2012; Orros and Fellowes 2015), can be found at substantially higher densities in towns and cities compared to rural landscapes (Blair 1996; Tryjanowski et al. 2007; Alexandre et al. 2010; Bateman and Fleming 2012; Kettel et al. 2019). Understanding how animals utilise urban spaces has, therefore, become increasingly important as urbanisation rates continue to rise (Ritchie 2018; Gonçalves-Souza et al. 2020).
One way to gain insight into how animals use urban spaces is through habitat selection analyses to assess habitat preferences and / or avoidances within the context of landscape-scale distribution or home range utilisation (Saunders et al. 1997; Dowding et al. 2010a; Thomas et al. 2014; Roberts et al. 2017; Mueller et al. 2018). However, understanding habitat use on a finer scale can yield greater benefits in conservation planning (Gilioli et al. 2018), particularly for species that perceive the environment at small spatial scales (Ritchie and Olff 1999), have limited dispersal ability (Gilioli et al. 2018) and / or which may be associated with specific habitats or microhabitats (Banks and Skilleter 2007). The way in which such individuals move within and between habitats will be dependent on intra- and inter-specific interactions, environmental conditions and site-based variables such as resource availability and quality (Morris 2003; Roberts et al. 2017; Bista et al. 2019).
Residential gardens within urban areas are favoured by a range of fauna (Saunders et al. 1997; Newman et al. 2003; Murgui 2009; Dowding et al. 2010a) and can collectively cover a substantial area. For example, private domestic gardens constitute 35–47% of greenspace in some British cities (Loram et al. 2007) and cover > 430,000 ha in the UK as a whole (Davies et al. 2009). On a finer scale, the structure of and features within individual gardens can vary markedly as the result of differences in garden size, householders’ gardening preferences and their management decisions (Gaston et al. 2005; Smith et al. 2006a; Goddard et al. 2013). Consequently, there is likely to be fine-scale variation in the functional value of different individual gardens both within and between species.
Assessing how animals use individual gardens, and identifying underlying causal factors can, however, be challenging. First, it requires gaining access to privately owned land, or recruiting homeowners to provide data; homeowners’ willingness to engage in such projects can be affected by pre-existing environmental interests (Dyson et al. 2019) and differences between project types, places and cultures (Sakurai et al. 2015). Second, within-garden characteristics are likely to fluctuate over time, sometimes over very short timescales. For example, invertebrate prey abundance has been shown to vary with weeding practices (Jaganmohan et al. 2013) and temporal and microclimatic parameters (Martay and Pearce-Higgins 2018), whilst anthropogenic food availability depends on the regularity of householders’ wildlife feeding habits and the volume of food supplied (Davies et al. 2012). Last, quantifying how animals use gardens necessitates the use of specialist equipment such as GPS or radio tracking devices, camera traps or microchip readers to monitor fine-scale movements (Galbraith et al. 2017; Van Helden et al. 2020). Each of these are, however, associated with their own advantages and disadvantages including cost, reliability, accuracy and welfare concerns (Coulombe et al. 2010; Wearn and Glover-Kapfer 2019). In light of these challenges, preliminary studies would be useful in identifying suitable methodologies for use in such investigations.
One species that is commonly associated with gardens, yet is challenging to study in urban landscapes, is the West European hedgehog (Erinaceus europaeus; hereafter ‘hedgehog’), a small (< 1.5 kg), nocturnal insectivore (Morris 2018). It is thought that rural hedgehog populations in Britain have declined markedly in recent decades (Harris et al. 1995; Mathews et al. 2018) probably due to habitat loss and fragmentation (Bearman-Brown et al. 2020), intensive agricultural practices and predation by or intraguild competition with the European badger (Meles meles) (Young et al. 2006; Trewby et al. 2014; Williams et al. 2018a). Hedgehogs nowadays occur in higher densities in urban settings (Hubert et al. 2011; van de Poel et al. 2015; Schaus et al. 2020) where the risk of predation by badgers appears to be comparatively low (Hubert et al. 2011; Pettett et al. 2017a) whilst the abundance of anthropogenic foods and nesting opportunities is likely high (Hubert et al. 2011; Pettett et al. 2017a; Gimmel et al. 2021). Nonetheless, urban areas are associated with a range of risks not typically evident in rural landscapes, including disturbance by humans or domestic animals (Rasmussen et al. 2019; Rast et al. 2019), exposure to urban-associated pollutants and pesticides (Dowding et al. 2010b; Taucher et al. 2020), barriers to movement created by built structures including roads (Rondinini and Doncaster 2002) and fences (Gazzard et al. 2021), and road traffic accidents (Wright et al. 2020).
In addition to sex, season and environmental conditions, hedgehog movement behaviour also varies between urban landscapes (Dowding et al. 2010a; Rasmussen et al. 2019; Schaus Calderón 2021) potentially due to differences in building density (Schaus Calderón 2021), road type (Rondinini and Doncaster 2002) and disturbance levels (Berger et al. 2020a). Irrespective of this, hedgehogs consistently favour back gardens (Baker and Harris 2007; Hof and Bright 2009; Dowding et al. 2010a; Williams et al. 2015, 2018b; Rasmussen et al. 2019; Gazzard and Baker 2020; Schaus Calderón 2021) and are thought to require access to around 13–14 back gardens per night (Rasmussen et al. 2019; Schaus Calderón 2021). However, only a minority of gardens available within a given area appear to be utilised (Williams et al. 2018b; Schaus Calderón 2021). Consequently, multiple studies have attempted to identify those factors affecting garden use (Baker and Harris 2007; Hof and Bright 2009; Williams et al. 2015, 2018b; Gazzard and Baker 2020) but these have relied on hedgehog presence / absence data such that they have not been able to identify whether animals were using gardens for, e.g. foraging versus simply passing through, nor differentiate between gardens based on intensity of use. Overall, therefore, our understanding of the extent to which different factors influence patterns of garden use is limited (Williams et al. 2018b; Schaus Calderón 2021).
Understanding which factors affect hedgehogs’ patterns of garden use would have clear conservation implications as members of the public could be advised on how to manage their gardens in a “hedgehog-friendly” way. This is particularly relevant in the UK where urban gardens may be increasingly important as the national hedgehog population declines. Within the UK at the current time, there is particular emphasis on the construction of high-density housing in urban locations, including on greenfield and brownfield sites (Department for Communities and Local Government 2017); this type of housing is, by definition, associated with smaller gardens than those evident in earlier periods of rapid housing development (Loram et al. 2007). Therefore, the aim of this study was to investigate patterns of habitat use and garden selection by hedgehogs in an area of high-density housing as these are likely to be the dominant form of new housing in the UK for the foreseeable future. In particular, we: (1) used global positioning system (GPS) and radio tracking data in compositional analyses (Aebischer et al. 1993) to identify key habitats and garden types; and (2) used GIS data and householder questionnaire surveys to investigate the effects of outside- and within-garden factors on the time hedgehogs spent in individual gardens. Last, GPS fix acquisition rates in mammal tracking studies are commonly < 100% (Hofman et al. 2019), including in previous studies of hedgehogs (Rodriguez Recio et al. 2013; Braaker et al. 2014), because tags with limited “sky-view” (e.g., tags under dense canopy cover) can fail to connect to sufficient satellites (Ironside et al. 2017); such “missing data” have the potential to confound the sorts of analyses outlined above if they are, for example, associated with particular habitat types and / or movement characteristics. Therefore, we (3) conducted a series of assessments of these missing fixes to identify whether they were likely to have impacted our results or not.
Methods
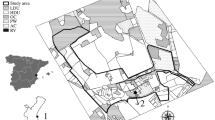
The study was conducted in a 5.8 km2 area of Earley, Reading, UK (51°25’N, 0°55’W; population > 33,000), bordered by a University of Reading campus, major A-roads and the River Loddon. Earley is a residential area that falls within the urban sprawl of Reading Town, having undergone rapid urbanisation in the late twentieth century (Earley Town Council 2017). Earley is now characterised by a series of medium- and high-density housing developments (approximately 20.5 houses ha−1 across the entire survey area, but with some estates reaching 38.1 houses ha−1: Schaus Calderón 2021) constructed predominantly during the 1970s–1990s and which consist of streets and cul-de-sacs of detached, semi-detached and terraced houses with their associated gardens (Ward 2004; Wokingham Borough Council 2012; Earley Town Council 2017). Median garden size is 167 m2, below the national average of 226 m2 (Office for National Statistics 2021).
Tracking data
Hedgehogs were captured by hand during nocturnal transect surveys undertaken between June and October 2016–2018 inclusive. Trained surveyors walked along public footpaths using torches to systematically search for hedgehogs. On occasion, local householders contacted the surveyors to notify us of active hedgehogs in gardens, in which case surveyors would be granted access to the garden to record and tag hedgehogs as appropriate. Captured individuals were weighed, sexed and uniquely marked by securing short sections of numbered heat shrink tubing over approximately five spines posterior to the head (Reeve et al. 2019a). Suitable healthy adults weighing > 600 g were fitted with either very high-frequency radio (VHF; TW-3, Lotek UK, 10.8 g) or GPS (PinPoint 250 VHF Swift, Lotek UK, 10 g) tags (hereafter, “tags”), ensuring tag weight was < 5% of the animal’s body mass (Sikes and Gannon 2011). Tags were attached to a clipped area (~ 2 cm2) of spines below the hedgehog’s rump in a central position using epoxy resin. Hedgehogs were released at the point of capture typically within 5–10 min and monitored until they moved away to ensure individuals were not impeded by the tags.
Hedgehogs were tracked during June–October of 2016–2018 inclusive for 1–9 full nights (excluding nights on which the tags were attached or detached); one individual was tracked in more than 1 year (in 2017 and 2018), with the tag removed between years. Location fixes were recorded every 5 min from 22:00 to 04:00 British Summer Time (BST), yielding up to 73 fixes per night. VHF tagged animals were tracked on foot by triangulation with a VHF Sika receiver and hand-held three-element Yagi antenna (Lotek UK). Both tag types were retrieved by tracing their radio signal either when they became detached (e.g., when the hedgehog moved through small gaps under garden fences) or by recapturing the animal at the end of the study. All handling, tagging and tracking procedures were performed under ethical approval by the University of Reading and under licence by Natural England (refs: 20130866-0-0-0-5 and 2017-29687-SCI-SCI).
Outlying data points within the GPS dataset were identified and removed by examining consecutive fixes for implausible locations and speed of travel. In line with Braaker et al. (2014), fixes were excluded where they indicated speed of movement was > 1 m per second; none of the VHF fixes indicated speeds greater than this. The remaining fixes from each full tracking session were used to construct 100% minimum convex polygons in QGIS 3.4.4, representing nightly areas ranged (NAR) by each hedgehog. Differences in mean NAR between the sexes and between tag types were tested using independent t-tests: if a hedgehog was tracked for > 1 night, then their nightly MCPs were first averaged before analyses; if the variances of the two samples were unequal, a Welch–Satterthwaite type correction was applied (Ruxton 2006). For each full night of tracking, the total number of front and back gardens used (defined as a garden where ≥ 1 fixes were recorded) was also counted in QGIS with the aid of satellite imagery and an OS Mastermap® Topography Layer (© 2020 Ordnance Survey) to define garden boundaries.
Each hedgehog location was assigned to one of the following eight habitat categories: front gardens of (1) detached, (2) semi-detached, (3) terraced houses; back gardens of (4) detached, (5) semi-detached, (6) terraced houses; (7) amenity grassland; and (8) other habitats (roads and other areas of hardstanding, scrub, woodland and freshwater). Habitats within the study area were digitised in QGIS based upon satellite imagery, the OS Mastermap® Topography Layer (© 2020 Ordnance Survey) and land class datasets available through the UK Centre for Ecology & Hydrology (Morton et al. 2020).
Missing GPS fixes
Due to changes in tag availability and budget, hedgehogs were fitted with VHF tags in 2017 and GPS tags in 2016 and 2018. These tag types produce movement data of comparable accuracies that generate similar home range estimates (Coulombe et al. 2006; Glasby and Yarnell 2013), and thus data from both tag types were used in our analyses. However, since GPS tags can sometimes fail to connect to sufficient satellites to generate a location fix (Ironside et al. 2017), we assessed the patterns of missing GPS fixes to determine whether they were associated with abnormal movements or atypical habitats in five ways.
First, we collated the frequency with which different numbers of consecutive fixes were or were not recorded to identify whether missing fixes tended to occur in large groups. Second, we quantified the proportion of programmed fixes that were recorded versus not recorded, and how these varied throughout the 6-h tracking regimen. This would help identify whether missing fixes tended to occur at specific times of the night. Third, the size of nightly range areas (see below) was compared between those animals tracked using VHF tags, where all fixes were recorded every night, versus those tracked using GPS tags to see whether missing fixes in the latter resulted in significantly smaller nightly range estimates. Fourth, the minimum straight-line distances moved between different blocks of consecutive fixes where the intervening locations were fully or partially recorded versus those where all intervening locations had been missed were quantified (see Supplementary Figure S1); these comparisons enabled us to determine whether hedgehogs had tended to move significantly further across blocks of missing data or not. Finally, the possible effect of hedgehogs moving into different habitats where fixes may be particularly prone to be missed was investigated by comparing the habitat composition of pooled home range areas (see below) against the habitat composition of pooled home ranges after a 100-m buffer zone had been added to the points immediately preceding and following a block (of any size) of missing fixes.
All of the above analyses indicated that there was little evidence that missing GPS locations were associated with unusual movements or atypical habitats (see Supplementary Information). Instead, the pattern of missing fixes appeared to be consistent with the assumption that GPS tagged hedgehogs were moving normally within habitats but were periodically in proximity to structures that could potentially block their GPS signal, such as fences or buildings, but also perhaps underneath structures such as decking and sheds where they may be resting. Overall, therefore, we do not believe that there are likely to be any significant biases in the habitat types nor individual gardens where the positions of GPS tagged hedgehogs were and were not recorded. Consequently, the analyses outlined below are based on the assumption that the locations of GPS tagged animals are a random sub-sample of those of the hedgehogs tracked. The implications of this assumption are considered in the Discussion.
Compositional analysis of habitat use
Compositional analysis was implemented using the adehabitatHS package in R 4.0.3 with 1000 iterations of the randomisation test (Aebischer et al. 1993; Calenge 2006); this compares the log ratios of each individual’s “used” versus “available” habitats to indicate whether a habitat was used more (or less) than others based on their aerial availability (Aebischer et al. 1993; Calenge 2006). A ranking matrix was then constructed to display the differences in log ratios of all possible pairs of habitat categories. Wilks’ lambda (Λ) was used to test whether the difference between the proportion of habitat selected / used versus available differed significantly from zero (i.e., whether habitat use was “non-random”). Since there are known to be marked spatial differences in the areas ranged by male and female hedgehogs both on a nightly basis but also over the course of several nights (Kristiansson 1984; Reeve 1982; Rondini and Doncaster 2002; Dowding et al. 2010a; Morris 2018), and to identify any contrasts in habitat preference between the sexes, habitat use was analysed separately for males and females.
Habitat selection was evaluated at two levels (Johnson 1980). First, the selection of habitats in the context of the positioning of ranges within the wider landscape was quantified by comparing the habitat composition of individual ranges with that of the study site. Individual’s overall ranges were calculated as the MCP encompassing all of their known tracking locations (hereafter ‘pooled range area’: PRA). The study area was defined by a 500 m buffer surrounding all PRAs (Sparks et al. 2005; Dickson et al. 2012; Pettett et al. 2017a); this buffer was based upon existing hedgehog movement data (Kristiansson 1984; Reeve 1982; Rondini and Doncaster 2002; Dowding et al. 2010a; Morris 2018) and previous research that identified habitat within 500 m was of relevance to hedgehog occupancy in gardens (Gazzard and Baker 2020). Second, habitat use by each hedgehog within their PRA was assessed by comparing the proportion of fixes recorded in each habitat to the proportion of total habitat available within the range. For both analyses, values of zero of “used” habitats were substituted with 0.01, as recommended by Aebischer et al. (1993). Buildings were considered inaccessible to hedgehogs and were excluded from all analyses.
Factors affecting proportionate garden use
Factors affecting the use of back gardens were further investigated using generalised linear mixed models (GLMMs) with binomial distributions (Warton and Hui 2011). The response variable comprised the proportion of a tracking night known to have been spent in an individual garden (hereafter ‘proportionate garden use’: PGU). PGU was calculated by dividing the number of location fixes recorded in a single garden by 73 (the maximum number of fixes that could have been recorded each night).
Measures of PGU could be obtained from the same hedgehog but for different gardens in a single night and / or the same garden over different nights. To account for any parallels between data collected from the same individual, therefore, hedgehog ID was included as a random effect. Similarly, it was possible for an individual garden to appear more than once in the dataset if, for example, they were used by different hedgehogs on the same night or the same hedgehog on different nights. However, we elected to not treat garden ID as a grouping / random effect since we were interested in quantifying the effects of garden differences, rather than controlling for this.
Explanatory variables included factors specific to (a) tagged hedgehogs, (b) gardens, (c) environmental conditions at the time of garden use and (d) alternative habitats present within 500 m of the garden (Table 1). Continuous variables were z-transformed so that their effects could be easily compared (Schielzeth 2010). Habitat data were digitised and measured using Natural Environment Research Council land class datasets (Morton et al. 2020). Environmental data (temperature and rainfall) were obtained from the University of Reading’s Whiteknights campus weather station (Met Office 2012), which borders the study site; daylength data were taken from the Benson weather station, 18 km north (Thorsen 2021). Therefore, these variables represent general climatic conditions at the time of tracking rather than specific microclimatic parameters within individual gardens. For the variables describing individual garden characteristics, data were collected during door-to-door householder questionnaire surveys undertaken on site during 2016. In some instances, householders were not contactable, or did not wish to take part, and tagged hedgehogs inevitably utilised areas outside of the surveyed households. Therefore, the data that were available for this analysis represent a subsample of gardens (N = 49) that were known to be used by hedgehogs. Although red foxes (Vulpes vulpes) were present on the study site, badgers were not.
Multicollinearity checks were performed by examining the correlation structure of the explanatory variables, and further checking variance inflation factors (Fox and Monette 1992). The modelling process entailed successively adding variables and comparing Akaike’s Information Criterion (AIC) values (Burnham and Anderson 1998). In some cases, where a variable marginally worsened or did not affect model fit, but which was considered meaningful to interpretation, the variable was retained even if non-significant. Models were constructed with a log link function and fitted by Laplace approximation, using the lme4 package in R; global and final models were checked for overdispersion. Odds ratio values (Rita and Komonen 2008) and marginal and conditional R2 values (Nakagawa et al. 2017) are provided for the final model: marginal R2 represents the proportion of variance explained by fixed effects; conditional R2 represents the proportion of variance explained by both fixed and random effects.
Results
Including all GPS and VHF tagged hedgehogs, 28 individuals (13 males and 15 females) were tracked over 98 complete nights, generating 2920 fixes via VHF tags (73 fixes collected for all hedgehogs on all nights) and 2254 via GPS tags (nightly mean ± SD: 40 ± 10). Mean (± SD) NAR areas of males (3.54 ± 3.06 ha) were significantly larger than those of females (0.71 ± 0.31 ha: Table 2; t12 = 3.33, p < 0.01); however, mean NAR areas did not differ significantly between tag types for either males (GPS: 5.37 ± 4.66 ha; VHF: 3.00 ± 3.24 ha; t11 = − 1.21, p = 0.25) or females (GPS: 0.87 ± 0.31 ha; VHF: 0.61 ± 0.29 ha; t13 = 1.69, p = 0.11). The mean number of back gardens used per night was 8.1 ± 5.3 (range: 0–27). Furthermore, on only two occasions did hedgehogs fail to use gardens completely; in both instances, the animals roamed relatively small areas (≤ 0.25 ha) and spent most of the night foraging in hedgerows adjacent to houses. The mean number of front and back gardens used nightly by an individual did not significantly differ between males and females (back gardens: t27 = 1.10, p = 0.28; front gardens: t27 = 0.24, p = 0.82). However, back gardens were visited more frequently by both sexes; males used a mean of 3.4 (± 3.3) front and 8.7 (± 6.8) back gardens compared to 4.5 (± 2.7) and 6.8 (± 3.5) for females, respectively.
Habitat selection
Front and back gardens comprised 10.1% and 20.7% of the study area, respectively, with 13.6% of land occupied by buildings, 10.8% by roads, 16.3% by amenity grassland and the remaining 28.5% by other habitat types (see Supplementary Table S2 for a full breakdown). The selection of habitats within each hedgehog’s PRA relative to their availability across the study site was non-random for males (Λ = 0.07, p < 0.01) and females (Λ = 0.19, p = 0.02). At this scale, front and back gardens of terraced houses ranked first and second most favoured habitats for both sexes (Table 3). Amenity grassland occurred the least in PRAs relative to its overall availability.
Within their ranges, males exhibited a preference for the back gardens of detached houses, followed by the back gardens of semi-detached houses (Λ = 0.07, p = 0.02; Table 4). Conversely, for females, the front gardens of detached houses and the back gardens of detached and semi-detached houses were equally top-ranking, although, overall, habitats within their ranges did not appear to be used in a statistically non-random manner (Λ = 0.15, p = 0.14).
Factors affecting proportionate garden use
Data from 118 occasions of garden use were used in the GLMM analysis; these included data from a total of 49 gardens and 7 hedgehogs (4 males, 3 females) tracked in June–September 2017 or September 2018. The variables AMENITY500m and BACKGARDENS500m were excluded from the analysis as they were strongly correlated with garden AREA; similarly, BUILDINGS500m was excluded as it was correlated with WOODLAND500m.
When random effects (individual hedgehogs) were accounted for, proportionate garden use was significantly negatively linked to: the presence of front-to-back access into the garden; fox sightings; the presence of a pond; whether it had rained on the tracking night; and the quantity of garden habitat present within 500 m of the used garden (Table 5). Conversely, hedgehogs appeared to spend significantly more time in gardens where food was provided, where a compost heap was present and when day length was longer (i.e., in the summer).
Discussion
The use of GPS tags to record animal movements has become increasingly popular in ecological research (Nielson et al. 2009) as they can produce large quantities of continuous data periods whilst reducing surveyor effort and minimising disturbance (Adams et al. 2013; Glasby and Yarnell 2013). However, GPS tags are often associated with constraints relating to cost, battery life, location error and fix success rate (Adams et al. 2013; Glasby and Yarnell 2013; Hofman et al. 2019). Missing or erroneous location fixes resulting from restricted “sky-view” (Ironside et al. 2017) are not unusual when GPS tracking animals in urban areas (e.g., van Heezik et al. 2010; Hanmer et al. 2017), including hedgehogs: for 18 hedgehogs tracked in Zurich, < 50% of all possible GPS fixes were obtained (Braaker et al. 2014). Similarly, following data cleaning, a minimum of 46% of fixes were retained for hedgehogs tracked in Berlin (Berger et al. 2020b) and, in a GPS hedgehog tracking project in Regents Park, London, average fix success rate was 41% (Reeve et al. 2019b). In the current study, only 57% of all possible GPS locations were successfully recorded (see Supplementary Information).
Failure to record GPS locations could be attributed to several factors. Hedgehogs often navigate landscapes by travelling parallel to linear structures (Hof et al. 2012) such as hedgerows, fences or walls (Yarnell et al. 2014) which might block or reflect satellite signals (Adams et al. 2013). Buildings in particular are thought to create difficulties for GPS tracking in urban locations (Rose et al. 2005), yet their effect on fix success rate is likely to be more pronounced where building height is greater, i.e. in highly urbanised centres (Adams et al. 2013); in our study, houses were typically two stories high. Alternatively, the use of artificial features such as garden decking, cavities beneath garden sheds, hedgehog nesting / hibernation boxes and other refugia for shelter during the night (Morris 2018) could impede satellite connection by GPS tags. Hedgehogs also use “feeding stations” (typically wooden, brick or plastic boxes) installed by householders in gardens so that they can supply artificial foods for hedgehogs whilst simultaneously preventing non-target species such as domestic cats (Felis catus) or foxes from accessing the food, and / or to protect hedgehogs from predators such as badgers (e.g., Rasmussen et al. 2019; Finch et al. 2020). Consequently, loss of satellite connection could be linked to movements alongside built structures as well as periods of sheltering or feeding under cover, all of which are normal behaviours for hedgehogs in gardens (Morris 2018).
Overall, it is considered unlikely that the missing fixes reported in this study were linked to atypical movements or habitat use. This is because of the following: (1) the duration of missing fixes primarily comprised short bouts of 1–5 consecutive misses and mirrored the pattern of non-missing fixes; (2) the distance moved during blocks of missing fixes was broadly similar, or lower, to that observed for straight-line distances moved between known locations; (3) there was no significant difference in mean nightly area ranged for hedgehogs equipped with VHF versus those fitted with GPS tags and (4) the addition of a 100-m buffer around all fixes preceding or following a block of missing fixes did not significantly affect the habitat composition of individual pooled ranges. We have therefore assumed that the unrecorded (and recorded) fixes were a random sample of the locations of each hedgehog’s movement trajectory over the course of the night, and such random missing data should not have any substantive effect on the results of our habitat use analyses (see Nielson et al. 2009).
Habitat selection
Hedgehogs exhibited a preference for residential gardens: gardens of terraced houses were present disproportionately more in hedgehog ranges relative to their availability in the study site and gardens of detached and semi-detached houses were selected over other habitat types within ranges. This is consistent with previous studies (Dowding et al. 2010a; Schaus Calderón 2021) and underlines the potential importance of gardens in future conservation efforts for this species.
In contrast, areas of amenity grassland, which were typically present as private or public sports fields, were the least preferred habitat. These are typically highly managed and are often also used for exercising dogs (Canis lupus familiaris) throughout the day and early night. Consequently, they are associated with high levels of disturbance and little natural prey (Martay and Pearce-Higgins 2018), although earthworms may be relatively abundant on warm wet nights. However, foxes are also attracted to sports fields on such nights (Saunders et al. 1997) and hedgehogs may seek to avoid these (see below). Furthermore, such greenfield sites are increasingly being developed to meet housing demands in UK towns and cities. Paradoxically, therefore, although such developments may be controversial, their conversion to housing and associated gardens could represent a net gain in resources for urban hedgehog populations.
Factors affecting the use of back gardens
The proportional use of residential back gardens was significantly affected by a range of biotic and abiotic factors as well as within- and outside-garden factors, the former being under the control of individual householders whereas the latter are not. In the context of within-garden variables, garden use was positively associated with the provisioning of artificial food and the presence of a compost heap, and negatively associated with front garden to back garden access, the presence of a pond; four non-significant variables were also retained to improve model fit: the presence of either a garden shed and / or artificial lighting, the proportion of the garden covered by lawn and other plantings, and garden area.
Although definitive data are lacking, the supplemental feeding of hedgehogs does appear to have become increasingly common amongst UK householders (Morris 1985, 2018; Gimmel et al. 2021). However, it appears that, in many cases, householders only start to put food out once they know that hedgehogs are already visiting their garden. As such, artificial food cannot be a factor that originally attracted hedgehogs to the garden but, once present, it may become a major influence. In the context of the objectives of our study, this may, therefore, represent a significant confounding effect (i.e., the presence of anthropogenic food may reduce the ability to identify other factors that initially attracted hedgehogs to those gardens). Disentangling these factors would require some form of experimental manipulation (e.g., temporally withdrawing supplementary food) but this is likely to be logistically difficult given the perceived importance of artificial food by those householders that do feed hedgehogs.
However, it is worth noting that the odds ratio associated with the presence of food (OR = 1.384; Table 5) was not that large in comparison with other factors that also positively affected garden use. As outlined above, it is plausible that we may have underestimated the amount of time spent in gardens where artificial food was available if GPS fixes were missed when hedgehogs were inside feeding shelters, but it is also possible that there are limitations associated with using “time spent in the garden” as the metric by which to judge garden quality. For example, in contrast with natural prey, anthropogenic foods are typically predictable and abundant. Therefore, they require little foraging effort and hedgehogs may be able to obtain their entire daily energy requirement in one location in a relatively short space of time. This negative relationship between anthropogenic food intake and foraging time could also potentially indicate that our approach to measuring garden use would under-estimate the relative importance of gardens where these foods are available.
In addition, supplementary feeding could be associated with a range of negative impacts including changes in hibernation behaviour (Gazzard and Baker 2020), increased disease transmission risk (Rasmussen et al. 2019) and / or a reduction in dietary quality (Gimmel et al. 2021). For example, commercially available dry hedgehog foods in Switzerland contained high quantities of cereals that would otherwise not be part of the hedgehog’s natural diet (Gimmel et al. 2021). Additionally, the consumption of soft foods (e.g., canned pet or hedgehog foods) has been linked to tartar formation in hedgehogs (Sainsbury et al. 1996; Bexton and Couper 2019) which may have harmful consequences for oral health (Gimmel et al. 2021), though this has not been corroborated. Nonetheless, anthropogenic food is likely to be a key driver of hedgehog abundance in urban areas (Hubert et al. 2011) and could provide critical sustenance for vulnerable individuals (Reeve 1994). Further studies are needed, therefore, to examine the quality and quantity of food supplied by householders and to determine how this benefits and / or impacts local hedgehog populations such that appropriate advice can be given to householders.
Hedgehogs also spent more time in gardens where compost heaps were present; these are thought to be attractive to hedgehogs (Williams et al. 2015; Taucher et al. 2020) either by providing nest sites / material (Molony et al. 2006; Pettett et al. 2017a), or as a source of invertebrate prey (Curds 1985). Conversely, the proportion of the garden covered by lawn and other plantings was unimportant, whereas garden ponds were significantly negatively correlated with proportionate garden use. This is perhaps surprising as both can support a diversity of invertebrate prey species (Smith et al. 2006b; Ancillotto et al. 2019) and the creation of ponds is recommended by hedgehog conservation groups. One possible explanation for this negative relationship is that householders who feed hedgehogs also typically put out water for them to drink. Consequently, it may be that hedgehogs attracted to gardens where householders are putting out food are also able to access sufficient water, implying that they then do not necessarily need to visit other gardens with ponds. Furthermore, the metrics used here do not necessarily give any indication of the quality or management practices of ponds or green habitats (such as mowing regimes) within gardens, which may influence the use of gardens by hedgehogs via, e.g., driving invertebrate abundance (Smith et al. 2006b).
Access into and between gardens has been highlighted as a potentially major form of habitat fragmentation for urban hedgehog populations and is the focus of the Hedgehog Street campaign (run by the People’s Trust for Endangered Species and British Hedgehog Preservation Society) which aims to persuade householders to create holes in or under their fences to improve inter-garden connectivity (Gazzard et al. 2021). Although access into neighbouring back gardens (ACCESS) did not affect proportionate garden use, hedgehogs spent less time in gardens where access from the back garden to the front garden (FRONTTOBACK) was possible. The underlying reason for this is not immediately obvious. At one level, this could indicate a fragmentation effect (i.e., they are spending more time in gardens where they are not able to leave via the front garden), but this seems unlikely given that they were able to access, and presumably leave, that garden via other routes. Similarly, spending less time in back gardens where access to the front garden was available may reflect the absence of a fragmentation effect, but could conversely indicate a preference for front gardens. In fact, both sexes used the front gardens of detached houses to a much greater extent than their aerial availability and females also exhibited a stronger preference for the front gardens of other types of housing than males. It is possible, therefore, that females may be using front gardens to avoid competition with and / or harassment from males, or simply competition with other conspecifics. Identifying whether this is the case would necessitate detailed observations of the behaviour of animals within both front and back gardens; this could be achieved using trail cameras, CCTV cameras or even security cameras installed by householders as a deterrent to criminal activity. These sorts of recording devices would be unlikely to affect hedgehog behaviour, especially as artificial lighting did not affect garden use in this study and has also been shown not to affect foraging behaviour at artificial feeding stations (Finch et al. 2020).
Further variables retained in the final model were related to factors beyond the control of individual householders: the total area of gardens (GARDENS500) and woodland (WOODLAND500) within a 500-m radius of the focal garden, and focal garden area (AREA). Of these, only the former was significant, indicating that hedgehogs spent less time in individual gardens when the area of gardens in the surrounding landscape increased. This possibly reflects the fact that, as garden availability increases, then the reliance on individual gardens decreases. Conversely, the absence of a significant effect of garden size is surprising, given that previous studies have highlighted differences in engagement with wildlife-friendly gardening activities with garden size (Gaston et al. 2007; Loram et al. 2008; Goddard et al. 2013), and that both male and female hedgehogs exhibited a preference for the back gardens of detached houses within their ranges, which typically tend to comprise larger gardens. The latter is marginally different to the results of Dowding et al. (2010a) who suggested that female hedgehogs in Bristol potentially avoided the back gardens of detached houses because of the potential presence of badgers. In our study, however, badgers were not reported by any of the householders surveyed indicating that these were unimportant in this district of Reading; in other districts, the presence of badgers does appear to significantly reduce the use of gardens by hedgehogs (Williams et al. 2018b).
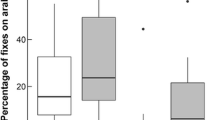
Although badgers were absent on the study site, many participants had observed foxes using their back gardens. Urban foxes are widespread in the UK (Scott et al. 2014) and typically occur in higher densities in towns and cities than in rural locations (Bateman and Flemming 2012). Foxes will predate hedgehogs (Morris 2018; Rasmussen et al. 2019) but also compete with them for a range of different foods, including that put out by householders (Pettett et al. 2017b). Consequently, hedgehog distribution has been negatively linked to fox abundance in the UK (Pettett et al. 2017b). In contrast to other studies where no significant association between hedgehog occupancy and the presence of foxes was observed (Hof and Bright 2009; Williams et al. 2018a), we found that hedgehogs were likely to spend more time in gardens where foxes were less frequent visitors (< monthly) and less time where foxes visited at least weekly. This suggests that whilst hedgehogs are not completely deterred by foxes, they may seek to reduce their risk of predation and competition by spending less time in gardens which foxes also utilise frequently. Hedgehogs might also exhibit similar behavioural responses to domestic dogs (Williams et al. 2018b; Rasmussen et al. 2019), although many pet dogs are secured inside their owner’s home for much of the night during which hedgehogs are active. We were not able to investigate this in this study as too few householders owned pet dogs.
Abiotic parameters such as temperature and rainfall can also influence the spatial behaviour of mammals (van Beest et al. 2012; Maestri and Marinho 2014). In hedgehogs, nightly ranges and activity levels have been recorded to increase with higher temperatures, although only after midnight when vehicular and pedestrian traffic is reduced (Dowding et al. 2010a). In our study, minimum nightly temperature did not impact garden use possibly because it was not reflective of temperature change throughout the night or specific temperatures within gardens, implying that future studies need to record microclimatic conditions within individual gardens more intensively. Conversely, hedgehogs did spend significantly less time in gardens during nights when it had rained. As outlined above, studies of urban foxes (Saunders et al. 1997) have noted an increased tendency to utilise playing fields under these conditions because of increased earthworm availability, but they will also do the same in residential gardens (P. Baker, pers. obs.). Therefore, it might be expected that rainfall would increase hedgehog activity within gardens contrary to what we observed. However, the relative abundance of earthworms in gardens versus playing fields is not well known. One alternative possibility is that the reduced use of gardens on rainy nights was associated with hedgehogs seeking shelter (and potentially impacting the satellite connection with GPS tags).
Conclusions and recommendations
Studying urban wildlife poses its own particular set of problems, not least because much of the landscape is privately owned and difficult to observe directly from publicly accessible space. Consequently, there is the need to develop novel approaches to address key questions. In this study, we used radio and GPS tracking in combination with a priori questionnaire surveys of householders to identify factors that affected patterns of habitat and individual garden use by West European hedgehogs. Two major limitations were encountered: first, only 57% of planned GPS locations were recorded; second, we were only able to quantify the characteristics of 49 gardens subsequently visited by tracked hedgehogs. Consequently, we were not able to perfectly map the movement trajectory of tracked animals. As such, our results should be regarded as preliminary, with future studies required to validate or refute them.
We, therefore, recommend that authors should routinely publish the percentage of scheduled GPS fixes which are missed and consider the implications of these missing data on the research questions being considered. In this study, there was no evidence that missing GPS fixes affected estimates of the nightly area ranged by hedgehogs or patterns of habitat use. We postulate that most missing fixes were associated with hedgehogs’ tendency to skirt linear features such as fences when travelling, but also to be inside feeding stations or sheltering in refugia under, e.g., sheds and decking. This may mean, therefore, that we have under-estimated the relative time spent in gardens where artificial food was supplied by householders.
Residential gardens were preferred habitats although there were differences in the relative rankings of front and back gardens of different house types between male and female hedgehogs. This suggests possible differential uses of resources, and / or patterns of selection and avoidance. The proportion of time spent in back gardens was associated with a range of biotic and abiotic factors both within and outside those gardens. In particular, hedgehogs were more likely to have spent a greater proportion of their time in back gardens where artificial food was available, where compost heaps were present and where householders perceived foxes were uncommon visitors, but spent less time in these gardens on nights when it rained, where access to the front garden was possible and where foxes were perceived as frequent visitors. It was not possible to consider any effects of badgers as these were absent from the study site.
Surprisingly, time spent in gardens was significantly negatively associated with the presence of a pond and not significantly affected by the proportion of garden covered by microhabitats that might support invertebrate prey, factors which are highlighted by conservation NGOs as being of benefit to hedgehogs. It is possible, however, that the absence of any positive benefits from these factors may be obscured by the presence of artificial food and water but also by limited knowledge of how invertebrate prey abundance varies between gardens and within gardens over time. Therefore, we have three further recommendations for future studies. First, the relative contribution of food provided by householders to the total food intake of urban hedgehogs needs to be determined. This is of importance as hedgehogs may potentially be especially dependent on the behaviour of a relatively small number of householders, but also because they may have higher rates of contact with conspecifics at feeding stations: this could increase competition and the risk of disease transfer. In addition, if hedgehogs are obtaining most of their food from householders, this is likely to make it difficult to identify the relative importance of other within-garden factors on patterns of garden use without some form of experimental manipulation. Second, video recordings of hedgehog activity within gardens would enable a much more detailed analysis of how the within-garden factors considered in this study influence their behaviour. In addition, such recordings would also enable us to consider patterns of interactions between individual hedgehogs, but also between hedgehogs and other species including domestic cats and dogs, foxes and badgers. Third, invertebrate surveys are required to determine how the availability of key prey groups varies spatially and temporally within urban gardens.
Data availability
The raw data are available from the corresponding author upon request.
References
Adams CE (2016) Urban wildlife management, 3rd edn. CRC Press, Florida
Adams AL, Dickinson KJM, Robertson BC, van Heezik Y (2013) An evaluation of the accuracy and performance of lightweight GPS collars in a suburban environment. PLoS ONE 8:e68496. https://doi.org/10.1371/journal.pone.0068496
Aebischer NJ, Robertson PA, Kenward RE (1993) Compositional analysis of habitat use from animal radiotracking data. Ecology 74:1313–1325. https://doi.org/10.2307/1940062
Alexandre CV, Esteves KE, de Moura e Mello MAM (2010) Analysis of fish communities along a rural–urban gradient in a neotropical stream (Piracicaba River Basin, São Paulo, Brazil). Hydrobiologia 641:97–114. https://doi.org/10.1007/s10750-009-0060-y
Ancillotto L, Bosso K, Salinas-Ramos VB, Russo D (2019) The importance of ponds for the conservation of bats in urban landscapes. Landsc Urban Plan 190:103607. https://doi.org/10.1016/j.landurbplan.2019.103607
Baker PJ, Harris S (2007) Urban mammals: what does the future hold? An analysis of the factors affecting patterns of use of residential gardens in Great Britain. Mammal Rev 37:297–315. https://doi.org/10.1111/j.1365-2907.2007.00102.x
Banks SA, Skilleter GA (2007) The importance of incorporating fine-scale habitat data into the design of an intertidal marine reserve system. Biol Conserv 138:13–29. https://doi.org/10.1016/j.biocon.2007.03.021
Barnett KL, Facey SL (2016) Grasslands, invertebrates, and precipitation: a review of the effects of climate change. Front Plant Sci 7:1196. https://doi.org/10.3389/fpls.2016.01196
Bateman PW, Fleming PA (2012) Big city life: carnivores in urban environments. J Zool 287:1–23. https://doi.org/10.1111/j.1469-7998.2011.00887.x
Bearman-Brown LE, Baker PJ, Scott D, Uzal A, Evans L, Yarnell RW (2020) Over-winter survival and nest site selection of the West-European hedgehog (Erinaceus europaeus) in arable dominated landscapes. Animals 10:1449. https://doi.org/10.3390/ani10091449
Berger A, Barthel LMF, Rast W, Hofer H, Gras P (2020a) Urban hedgehog behavioural responses to temporary habitat disturbance versus permanent fragmentation. Animals 10:2109. https://doi.org/10.3390/ani10112109
Berger A, Lozano B, Barthel LMF, Schubert N (2020b) Moving in the Dark—evidence for an Influence of Artificial Light at Night on the Movement Behaviour of European Hedgehogs (Erinaceus europaeus). Animals 10:1306. https://doi.org/10.3390/ani10081306
Bexton S, Couper D (2019) Veterinary care of free-living hedgehogs. In Pract 41:420–432. https://doi.org/10.1136/inp.l5944
Bista D, Paudel PK, Jnawali SR, Sherpa AP, Shrestha S, Acharya KP (2019) Red panda fine-scale habitat selection along a Central Himalayan longitudinal gradient. Ecol Evol 9:5260–5269. https://doi.org/10.1002/ece3.5116
Blair RB (1996) Land use and avian species diversity along an urban gradient. Ecol Appl 6:506–519. https://doi.org/10.2307/2269387
Braaker S, Moretti M, Boesch R, Ghazoul J, Obrist MK, Bontadina F (2014) Assessing habitat connectivity for ground-dwelling animals in an urban environment. Ecol Appl 24:1583–1595. https://doi.org/10.1890/13-1088.1
Burnham KP, Anderson DR (1998) Model selection and inference, 2nd edn. Springer, New York
Calenge C (2006) The package “adehabitat” for the R software: a tool for the analysis of space and habitat use by animals. Ecol Modell 197:516–519. https://doi.org/10.1016/j.ecolmodel.2006.03.017
Coleman JL, Barclay RMR (2012) Urbanization and the abundance and diversity of Prairie bats. Urban Ecosyst 15:87–102. https://doi.org/10.1007/s11252-011-0181-8
Concepción ED, Moretti M, Altermatt F, Nobis MP, Obrist MK (2015) Impacts of urbanisation on biodiversity: the role of species mobility, degree of specialisation and spatial scale. Oikos 124:1571–1582. https://doi.org/10.1111/oik.02166
Coulombe ML, Massé A, Côté SD (2006) Quantification and accuracy of activity data measured with VHF and GPS telemetry. Wildl Soc Bull 34:81–92. https://doi.org/10.2193/0091-7648(2006)34[81:QAAOAD]2.0.CO;2
Curds T (1985) An experimental ecological study of a garden compost heap. J Biol Educ 19:71–78. https://doi.org/10.1080/00219266.1985.9654690
Davies ZG, Fuller RA, Loram A, Irvine KN, Sims V, Gaston KJ (2009) A national scale inventory of resource provision for biodiversity within domestic gardens. Biol Conserv 142:761–771. https://doi.org/10.1016/j.biocon.2008.12.016
Davies ZG, Fuller RA, Dallimer M, Loram A, Gaston KJ (2012) Household factors influencing participation in bird feeding activity: a national scale analysis. PLoS ONE 7:e39692. https://doi.org/10.1371/journal.pone.0039692
Department for Communities and Local Government (2017) Housing white paper. Ministry of Housing, Communities & Local Government. https://www.gov.uk/government/collections/housing-white-paper. Accessed 22 September 2021
Dickson BG, Jennes JF, Beier P (2012) Influence of vegetation, topography, and roads on cougar movement in Southern California. J Wildl Manage 69:264–276. https://doi.org/10.2193/0022-541X(2005)069%3c0264:IOVTAR%3e2.0.CO;2
Ditchkoff SS, Saalfeld ST, Gibson CJ (2006) Animal behavior in urban ecosystems: modifications due to human-induced stress. Urban Ecosyst 9:5–12. https://doi.org/10.1007/s11252-006-3262-3
Dowding CV, Harris S, Poulton S, Baker PJ (2010a) Nocturnal ranging behaviour of urban hedgehogs, Erinaceus europaeus, in relation to risk and reward. Anim Behav 80:13–21. https://doi.org/10.1016/j.anbehav.2010.04.007
Dowding CV, Shore RF, Worgan A, Baker PJ, Harris S (2010b) Accumulation of anticoagulant rodenticides in a non-target insectivore, the European hedgehog (Erinaceus europaeus). Environ Pollut 158:161–166. https://doi.org/10.1016/j.envpol.2009.07.017
Dyson K, Ziter C, Fuentes TL, Patterson MS (2019) Conducting urban ecology research on private property: advice for new urban ecologists. J Urban Ecol 5:juz001. https://doi.org/10.1093/jue/juz001
Earley Town Council (2017) Earley Town Guide. http://www.earley-tc.gov.uk/wp-content/uploads/2017/12/30.11.17-Interactive-Earley-Town-Guide-2017.pdf. Accessed 20 August 2021
Finch D, Smith BR, Marshall C, Coomber FG, Kubasiewicz LM, Anderson M, Wright PGR, Mathews F (2020) Effects of artificial light at night (ALAN) on European hedgehog activity at supplementary feeding stations. Animals 10:768. https://doi.org/10.3390/ani10050768
Fox J, Monette G (1992) Generalized collinearity diagnostics. J Am Stat Assoc 87:178–183. https://doi.org/10.1080/01621459.1992.10475190
Gaertner M, Wilson JRU, Cadotte MW, Maclvor JS, Zenni RD, Richardson DM (2017) Non-native species in urban environments: patterns, processes, impacts and challenges. Biol Invasions 19:3461–3469. https://doi.org/10.1007/s10530-017-1598-7
Galbraith JA, Jones DN, Beggs JR, Parry K, Stanley MC (2017) Urban Bird feeders dominated by a few species and individuals. Front Ecol Evol 5:81. https://doi.org/10.3389/fevo.2017.00081
Garcês A, Soeiro V, Lóio S, Sargo R, Sousa L, Silva F, Pires I (2020) Outcomes, mortality causes, and pathological findings in European Hedgehogs (Erinaceus europeus, Linnaeus 1758): a seventeen year retrospective analysis in the North of Portugal. Animals 10:1305. https://doi.org/10.3390/ani10081305
Gaston KJ, Warren PH, Thompson K, Smith RM (2005) Urban domestic gardens (IV): the extent of the resource and its associated features. Biodivers Conserv 14:3327–3349. https://doi.org/10.1007/s10531-004-9513-9
Gaston KJ, Fuller RA, Loram A, MacDonald C, Power S, Dempsey N (2007) Urban domestic gardens (XI): variation in urban wildlife gardening in the United Kingdom. Biodivers Conserv 16:3227–3238. https://doi.org/10.1007/s10531-007-9174-6
Gazzard A, Baker PJ (2020) Patterns of feeding by householders affect activity of hedgehogs (Erinaceus europaeus) during the hibernation period. Animals 10:1344. https://doi.org/10.3390/ani10081344
Gazzard A, Boushall A, Brand E, Baker PJ (2021) An assessment of a conservation strategy to increase garden connectivity for hedgehogs that requires cooperation between immediate neighbours: a barrier too far? PLoS ONE 16:e0259537. https://doi.org/10.1371/journal.pone.0259537
Gilioli KC, Kéry M, Guimarães M (2018) Unravelling fine-scale habitat use for secretive species: when and where toads are found when not breeding. PLoS ONE 13:e0205304. https://doi.org/10.1371/journal.pone.0205304
Gimmel A, Eulenberger U, Liesegang A (2021) Feeding the European hedgehog (Erinaceus europaeus L.)—risks of commercial diets for wildlife. J Anim Physiol Anim Nutr 105:91–96. https://doi.org/10.1111/jpn.13561
Glasby L, Yarnell RW (2013) Evaluation of the performance and accuracy of Global Positioning System bug transmitters deployed on a small mammal. Eur J Wildlife Res 59:915–919. https://doi.org/10.1007/s10344-013-0770-3
Goddard MA, Dougill AJ, Benton TG (2013) Why garden for wildlife? Social and ecological drivers, motivations and barriers for biodiversity management in residential landscapes. Ecol Econ 86:258–273. https://doi.org/10.1016/j.ecolecon.2012.07.016
Gonçalves-Souza D, Verburg PH, Dobrovolksi R (2020) Habitat loss, extinction predictability and conservation efforts in the terrestrial ecoregions. Biol Conserv 246:108579. https://doi.org/10.1016/j.biocon.2020.108579
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM (2008) Global change and the ecology of cities. Science 319:756–760. https://doi.org/10.1126/science.1150195
Haigh A, O’Riordan RM, Butler F (2013) Habitat selection, philopatry and spatial segregation in rural Irish hedgehogs (Erinaceus europaeus). Mammalia 77:163–172. https://doi.org/10.1515/mammalia-2012-0094
Hanmer HJ, Thomas RL, Fellowes MDE (2017) Urbanisation influences range size of the domestic cat (Felis catus): consequences for conservation. J Urban Ecol 3:1–11. https://doi.org/10.1093/jue/jux014
Harris S, Morris P, Wray S, Yalden D (1995) A review of British mammals: population estimates and conservation status of British mammals other than cetaceans. JNCC, Peterborough
Hedgehog Street (2020) Hedgehogs After Dark: Fighting. https://www.hedgehogstreet.org/hadfighting/. Accessed 4 December 2021
Hedgehog Street (2021) Garden hazards for hedgehogs. https://www.hedgehogstreet.org/garden-hazards/. Accessed 20 November 2021
Hof AR, Bright PW (2009) The value of green spaces in built-up areas for Western hedgehogs. Lutra 52:69–82
Hof AR, Snellenberg J, Bright PW (2012) Food or fear? Predation risk mediates edge refuging in an insectivorous mammal. Anim Behav 4:1099–1106. https://doi.org/10.1016/j.anbehav.2012.01.042
Hofman MGP, Hayward MW, Heim M, Marchand P, Rolandsen CM, Mattisson J, Urbano F, Heurich M, Mysterud A, Melzheimer J, Morellet N, Voigt U, Allen BL, Gher B, Ruco C, Ullmann W, Holland Ø, Jørgensen NH, Steinheim G, Cagnacci F, Kroeschel M, Kaczenksy P et al (2019) Right on track? Performance of satellite telemetry in terrestrial wildlife research. PLoS ONE 14:e0216223. https://doi.org/10.1371/journal.pone.0216223
Hubert P, Julliard R, Biagianti S, Poulle M-L (2011) Ecological factors driving the higher hedgehog (Erinaceus europeaus) density in an urban area compared to the adjacent rural area. Landsc Urban Plan 103:34–43. https://doi.org/10.1016/j.J.landurbanplan.2011.05.010
Ironside KE, Mattson DJ, Choate D, Stoner D, Arundel T, Hansen J, Theimer T, Holton B, Jansen B, Sexton JO, Longshore K, Edwards TCE Jr, Peters M (2017) Variable terrestrial GPS telemetry detection rates: addressing the probability of successful acquisitions. Wildlife Soc Bull 41:329–341. https://doi.org/10.1002/wsb.758
Jaganmohan M, Vailshery LS, Nagendra H (2013) Patterns of insect abundance and distribution in urban domestic gardens in Bangalore, India. Diversity 5:767–778. https://doi.org/10.3390/d5040767
Johnson DH (1980) The comparison of usage and availability measurements for evaluating resource preference. Ecology 61:65–71. https://doi.org/10.2307/1937156
Jones SA, Champman S (2020) The Ethics and Welfare Implications of Keeping Western European Hedgehogs (Erinaceus europaeus) in Captivity. J Appl Anim Welf Sci 23:467–483. https://doi.org/10.1080/10888705.2019.1672553
Kettel ES, Gentle LK, Yarnell RW, Quinn JL (2019) Breeding performance of an apex predator, the peregrine falcon, across urban and rural landscapes. Urban Ecosyst 22:117–125. https://doi.org/10.1007/s11252-018-0799-x
Kristiansson H (1984) Ecology of a hedgehog Erinaceus europaeus population in southern Sweden. PhD thesis, University of Lund
Löki V, Deák B, Balázs Lukács A, Molnár VA (2019) Biodiversity potential of burial places—a review on the flora and fauna of cemeteries and churchyards. Glob Ecol Conserv 18:e00614. https://doi.org/10.1016/j.gecco.2019.e00614
Loram A, Tratalos J, Warren PH, Gaston KJ (2007) Urban domestic gardens (X): the extent & structure of the resource in five major cities. Landsc Ecol 22:601–615. https://doi.org/10.1007/s10980-006-9051-9
Loram A, Warren P, Gaston KJ (2008) Urban domestic gardens (XIV): the characteristics of gardens in five cities. Environ Manage 42:361–376. https://doi.org/10.1007/s00267-008-9097-3
Maestri R, Marinho JR (2014) Singing in the rain. Rainfall and moonlight affect daily activity patterns of rodents in a Neotropical forest. Acta Theriol 59:427–433. https://doi.org/10.1007/s13364-014-0177-x
Martay B, Pearce-Higgins JW (2018) Using data from schools to model variation in soil invertebrates across the UK: the importance of weather, climate, season and habitat. Pedobiologia 67:1–9. https://doi.org/10.1016/j.pedobi.2018.01.002
Mathews F, Kubasiewicz LM, Gurnell J, Harrower CA, McDonald RA, Shore RF (2018) A review of the population and conservation status of British mammals. A report by the Mammal Society under contract to Natural England, Natural Resources Wales and Scottish Natural Heritage. http://publications.naturalengland.org.uk/publication/5636785878597632. Accessed 19 December 2021
McKinney ML (2006) Urbanization as a major cause of biotic homogenization. Biol Conserv 127:247–260. https://doi.org/10.1016/j.biocon.2005.09.005
Met Office (2012) Met Office Integrated Data Archive System (MIDAS) Land and Marine Surface Stations Data (1853-current). NCAS British Atmospheric Data Centre, 2021. http://catalogue.ceda.ac.uk/uuid/220a65615218d5c9cc9e4785a3234bd0. Accessed 28 October 2021
Møller AP (2012) Urban areas as refuges from predators and flight distance of prey. Behav Ecol 23:1030–1035. https://doi.org/10.1093/beheco/ars067
Morris P (1985) The effect of supplementary feeding on movements of hedgehogs (Erinaceus europaeus). Mammal Rev 15:23–32. https://doi.org/10.1111/j.1365-2907.1985.tb00383.x
Morris DW (2003) Toward an ecological synthesis: a case for habitat selection. Oecologia 136:1–13. https://doi.org/10.1007/s00442-003-1241-4
Morris P (2018) Hedgehog. William Collins, London
Morton RD, Marston CG, O’Neil AW, Rowland CS (2020) Land Cover Map 2019 (land parcels, GB). NERC Environmental Information Data Centre. https://doi.org/10.5285/44c23778-4a73-4a8f-875f-89b23b91ecf8. Accessed 18 October 2021
Mueller MA, Drake D, Allen ML (2018) Coexistence of coyotes (Canis latrans) and red foxes (Vulpes vulpes) in an urban landscape. PLoS ONE 13:e0190971. https://doi.org/10.1371/journal.pone.0190971
Murgui E (2009) Seasonal patterns of habitat selection of the House Sparrow Passer domesticus in the urban landscape of Valencia (Spain). J Ornithol 150:85. https://doi.org/10.1007/s10336-008-0320-z
Nakagawa S, Johnson PCD, Schielzeth H (2017) The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J R Soc Interface 14:20170213. https://doi.org/10.1098/rsif.2017.0213
Newman TJ, Baker PJ, Simcock E, Saunders G, White PCL, Harris S (2003) Changes in red fox habitat preference and rest site fidelity following a disease-induced population decline. Acta Theriol 48:79–91
Nielson RM, Manly BFJ, McDonald LL, Sawyer H, McDonald TL (2009) Estimating habitat selection when GPS fix success is less than 100%. Ecology 90:2956–2962. https://doi.org/10.1016/j.anbehav.2010.04.007
Office for National Statistics (2020) “Explore access to gardens (and their typical size) in your neighbourhood”. https://www.ons.gov.uk/economy/environmentalaccounts/articles/oneineightbritishhouseholdshasnogarden/2020-05-14. Accessed 20 August 2021
Oliveira Hagen E, Hagen O, Ibáñez-Álamo JD, Petchey OL, Evans KL (2017) Impacts of urban areas and their characteristics on avian functional diversity. Front Ecol Evol 5:84. https://doi.org/10.3389/fevo.2017.00084
Oro D, Genovart M, Tavecchia G, Fowler MS, Martínez-Abraín A (2013) Ecological and evolutionary implications of food subsidies from humans. Ecol Lett 16:1501–1514. https://doi.org/10.1111/ele.12187
Orros M, Fellowes M (2015) Widespread supplementary feeding in domestic gardens explains the return of reintroduced Red Kites Milvus milvus to an urban area. Ibis 157:230–238. https://doi.org/10.1111/ibi.12237
Pettett CE, Moorhouse TP, Johnson PJ, Macdonald DW (2017a) Factors affecting hedgehog (Erinaceus europaeus) attraction to rural villages in arable landscapes. Eur J Wildl Res 63:1–12. https://doi.org/10.1007/s10344-017-1113-6
Pettett CE, Johnson PJ, Moorhouse TP, Macdonald DW (2017b) National predictors of hedgehog Erinaceus europaeus distribution and decline in Britain. Mammal Rev 48:1–6. https://doi.org/10.1111/mam.12107
Rasmussen SL, Berg TB, Dabelsteen T, Jones OR (2019) The ecology of suburban juvenile European hedgehogs (Erinaceus europaeus) in Denmark. Ecol Evol 9:13174–13187. https://doi.org/10.1002/ece3.5764
Rast W, Barthel LMF, Berger A (2019) Music festival makes hedgehogs move: how individuals cope behaviorally in response to human-induced stressors. Animals 9:455. https://doi.org/10.3390/ani9070455
Rautio A, Isomursu M, Valtonen A, Hirvelä-Koski V, Kunnasranta M (2016) Mortality, diseases and diet of European hedgehogs (Erinaceus europaeus) in an urban environment in Finland. Mammal Res 61:161–169. https://doi.org/10.1007/s13364-015-0256-7
Reeve N (1982) The home range of the hedgehog as revealed by a radio tracking study. Symp Zool Soc London 49:207–230
Reeve N (1994) Hedgehogs. Academic Press, London
Reeve NJ, Huijser MP (1999) Mortality factors affecting wild hedgehogs: a study of records from wildlife rescue centres. Lutra 42:7–24
Reeve N, Bowen C, Gurnell J (2019a) An improved identification marking method for hedgehogs. Mammal Commun 5:1–5
Reeve N, Bowen C, Gurnell J (2019b) A critical look at the use of GPS tags to study the movements of hedgehogs in a London park. https://www.royalparks.org.uk/managing-the-parks/conservation-and-improvement-projects/hedgehogs/hedgehog-research-reports Accessed 19 December 2021
Riber AB (2006) Habitat use and behaviour of European hedgehog Erinaceus europaeus in a Danish rural area. Acta Theriol 51:363–371
Rita H, Komonen A (2008) Odds ratio: an ecologically sound tool to compare proportions. Ann Zool Fenn 45:66–72. https://doi.org/10.5735/086.045.0106
Ritchie H (2018) Urbanization. University of Oxford. https://ourworldindata.org/urbanization. Accessed 29 December 2020
Ritchie ME, Olff H (1999) Spatial scaling laws yield a synthetic theory of biodiversity. Nature 400:557–560. https://doi.org/10.1038/23010
Roberts CP, Cain JW III, Cox RD (2017) Identifying ecologically relevant scales of habitat selection: diel habitat selection in elk. Ecosphere 8:e02013. https://doi.org/10.1002/ecs2.2013
Rodriguez Recio M, Mathieu R, Latham MC, Latham DM, Seddon PJ (2013) Quantifying fine-scale resource selection by introduced European hedgehogs (Erinaceus europaeus) in Ecologically sensitive areas. Biol Invasions 15:1807–1818. https://doi.org/10.1007/s10530-013-0410-6
Rondinini C, Doncaster C (2002) Roads as barriers to movement for hedgehogs. Funct Ecol 16:504–509. https://doi.org/10.1046/j.1365-2435.2002.00651.x
Rose E, Nagel P, Haag-Wackernagel D (2005) Suitability of using the global positioning system (GPS) for studying Feral Pigeons Columba livia in the urban habitat. Bird Study 52:145–152. https://doi.org/10.1080/00063650509461384
Ruxton GD (2006) The unequal variance t-test is an underused alternative to Student’s t-test and the Mann-Whitney U test. Behav Ecol 17:688–690
Sainsbury AW, Cunningham AA, Morris PA, Kirkwood JK, Macgregor SK (1996) Health and welfare of rehabilitated juvenile hedgehogs (Erinaceus europaeus) before and after release into the wild. Vet Rec 138:61–65. https://doi.org/10.1136/vr.138.3.61
Sakurai R, Kobori H, Nakamura M, Kikuchi T (2015) Factors influencing public participation in conservation activities in urban areas: a case study in Yokohama, Japan. Biol Conserv 184:424–430. https://doi.org/10.1016/j.biocon.2015.02.012
Saunders G, White PCL, Harris S (1997) Habitat utilisation by urban foxes (Vulpes vulpes) and the implications for rabies control. Mammalia 61:497–510
Schaus Calderón J (2021) Responses of the European hedgehog to urbanisation: impact on population dynamics, animal movement and habitat selection. PhD thesis, Nottingham Trent University
Schaus J, Uzal A, Gentle LK, Baker PJ, Bearman-Brown L, Bullion S, Gazzard A, Lockwood H, North A, Reader T, Scott DM, Sutherland CS, Yarnell RW (2020) Application of the Random Encounter Model in citizen science projects to monitor animal densities. Remote Sens Ecol Conserv 6:514–528. https://doi.org/10.1002/rse2.153
Schielzeth H (2010) Simple means to improve the interpretability of regression coefficients. Methods Ecol Evol 1:103–113
Scott DM, Berg MJ, Tolhurst BA, Chauvenet ALM, Smith GC, Neaves K, Lochhead J, Baker PJ (2014) Changes in the Distribution of Red Foxes (Vulpes vulpes) in Urban Areas in Great Britain: findings and limitations of a media-driven nationwide survey. PLoS ONE 9:e99059. https://doi.org/10.1371/journal.pone.0099059
Seto KC, Güneralp B, Hutyra LR (2012) Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. PNAS 109:16083–16088. https://doi.org/10.1073/pnas.1211658109
Sikes RS, Gannon WL (2011) Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J Mammal 92:235–253. https://doi.org/10.1644/10-MAMM-F-355.1
Smith RM, Thompson K, Hodgson JG, Warren PH, Gaston KJ (2006a) Urban domestic gardens (IX): composition and richness of the vascular plant flora, and implications for native biodiversity. Biol Conserv 129:312–322. https://doi.org/10.1016/j.biocon.2005.10.045
Smith RM, Gaston KJ, Warren PH, Thompson K (2006b) Urban domestic gardens (VIII): environmental correlates of invertebrate abundance. Biodivers Conserv 15:2515–2545. https://doi.org/10.1007/s10531-005-2784-y
Soanes K, Lentini PA (2019) When cities are the last chance for saving species. Front Ecol Environ 17:225–231. https://doi.org/10.1002/fee.2032
Soulsbury CD, White PCL (2015) Human–wildlife interactions in urban areas: a review of conflicts, benefits and opportunities. Wildl Res 42:541–553. https://doi.org/10.1071/WR14229
Sparks DW, Ritzi CM, Duchamp JE, Whitaker JO (2005) Foraging habitat of the Indiana bat (Myotis sodalis) at an urban-rural interface. J Mammal 86:713–718. https://doi.org/10.1644/1545-1542(2005)086[0713:FHOTIB]2.0.CO;2
Spotswood EN, Beller EE, Grossinger R, Grenier JL, Heller NE, Aronson MFJ (2021) The biological deserts fallacy: cities in their landscapes contribute more than we think to regional biodiversity. Bioscience 71:148–160. https://doi.org/10.1093/biosci/biaa155
Stone EL, Harris S, Jones G (2015) Impacts of artificial lighting on bats: a review of challenges and solutions. Mamm Biol 80:213–219. https://doi.org/10.1016/j.mambio.2015.02.004
Taucher AL, Gloor S, Dietrich A, Geiger M, Hegglin D, Bontadina F (2020) Decline in distribution and abundance: urban hedgehogs under pressure. Animals 10:1606. https://doi.org/10.3390/ani10091606
Theodorou P, Herbst SC, Kahnt B, Landaverde-González P, Baltz LM, Osterman J, Paxton RJ (2020) Urban fragmentation leads to lower floral diversity, with knock-on impacts on bee biodiversity. Sci Rep 10:21756. https://doi.org/10.1038/s41598-020-78736-x
Thomas RL, Baker PJ, Fellowes MDE (2014) Ranging characteristics of the domestic cat (Felis catus) in an urban environment. Urban Ecosyst 17:911–921. https://doi.org/10.1007/s11252-014-0360-5
Thorsen S (2021) Reading, England, United Kingdom—Sunrise, Sunset, and Daylength. https://www.timeanddate.com/sun/uk/reading. Accessed 28 October 2021
Trewby ID, Young R, McDonald RA, Wilson GJ, Davison J, Walker N, Robertson A, Doncaster CP, Delahay RJ (2014) Impacts of removing badgers on localised counts of hedgehogs. PLoS ONE 9:e95477. https://doi.org/10.1371/journal.pone.0095477
Tryjanowski P, Sparks TH, Biaduń W, Brauze T, Hetmański T, Martyka R, Skórka P, Indykiewicz P, Myczko Ł, Kunysz P, Kawa P, Czyż S, Czechowski P, Polakowski M, Zduniak P, Jerzak L, Janiszewski T, Goławski A, Dudu L, Nowakowski JJ, Wuczyński A, Wysocki D (2015) Winter bird assemblages in rural and urban environments: a national survey. PLoS ONE 10:e0130299. https://doi.org/10.1371/journal.pone.0130299
van Beest FM, Van Moorter B, Milner JM (2012) Temperature-mediated habitat use and selection by a heat-sensitive northern ungulate. Anim Behav 84:723–735. https://doi.org/10.1016/j.anbehav.2012.06.032
van Heezik Y, Smyth A, Adams A, Gordon J (2010) Do domestic cats impose an unsustainable harvest on urban bird populations? Biol Conserv 143:121–130. https://doi.org/10.1016/j.biocon.2009.09.013
van Heezik Y, Freeman C, Porter S, Dickinson KJM (2013) Garden size, householder knowledge, and socio-economic status influence plant and bird diversity at the scale of individual gardens. Ecosystems 16:1442–1454. https://doi.org/10.1007/s10021-013-9694-8
Van Helden BE, Close PG, Stewart BA, Speldewinde PC, Comer SJ (2020) Critically Endangered marsupial calls residential gardens home. Anim Conserv 24:445–456. https://doi.org/10.1111/acv.12649
van de Poel JL, Dekker J, van Langevelde F (2015) Dutch hedgehogs Erinaceus europaeus are nowadays mainly found in urban areas, possibly due to the negative effects of badgers Meles meles. Wildlife Biol 21:51–55. https://doi.org/10.2981/wlb.00072
Ward SV (2004) Planning and urban change, 2nd edn. SAGE, London
Warton DI, Hui FKC (2011) The arcsine is asinine: the analysis of proportions in ecology. Ecology 92:3–10. https://doi.org/10.1890/10-0340.1
Wearn OR, Glover-Kapfer P (2019) Snap happy: camera traps are an effective sampling tool when compared with alternative methods. R Soc Open Sci 6:181748. https://doi.org/10.1098/rsos.181748
Williams RL, Stafford R, Goodenough AE (2015) Biodiversity in urban gardens: assessing the accuracy of citizen science data on garden hedgehogs. Urban Ecosyst 18:819–833. https://doi.org/10.1007/s11252-014-0431-7
Williams B, Baker PJ, Thomas E, Wilson G, Judge J, Yarnell RW (2018a) Reduced occupancy of hedgehogs (Erinaceus europaeus) in rural England and Wales: the influence of habitat and an asymmetric intraguild predator. Sci Rep 8:12156. https://doi.org/10.1038/s41598-018-30130-4
Williams B, Mann N, Neumann JL, Yarnell RW, Baker PJ (2018b) A prickly problem: developing a volunteer-friendly tool for monitoring populations of a terrestrial urban mammal, the West European hedgehog (Erinaceus europaeus). Urban Ecosyst 21:1075–1086. https://doi.org/10.1007/s11252-018-0795-1
Wokingham Borough Council (2012) Borough Design Guide. https://www.wokingham.gov.uk/planning-policy/planning-policy-information/supplementary-planning-guidance-and-documents/. Accessed 20 August 2021
Wright PGR, Coomer FG, Bellamy CC, Perkins SE, Mathews F (2020) Predicting hedgehog mortality risks on British roads using habitat suitability modelling. PeerJ 7:e8154. https://doi.org/10.7717/peerj.8154
Yarnell RW, Pacheco M, Williams B, Neumann JL, Rymer DJ, Baker PJ (2014) Using occupancy analysis to validate the use of footprint tunnels as a method for monitoring the hedgehog Erinaceus europaeus. Mammal Rev 44:3–4. https://doi.org/10.1111/mam.12026
Young RP, Davison J, Trewby ID, Wilson GJ, Delahay RJ, Doncaster CP (2006) Abundance of hedgehogs (Erinaceus europaeus) in relation to the density and distribution of badgers (Meles meles). J Zool 269:349–356. https://doi.org/10.1111/j.1469-7998.2006.00078.x
Acknowledgements
We are grateful to all the householders who took part in our questionnaire surveys, as well as those university students who assisted with fieldwork; particular thanks go to Samuel Nutt, Lane Matthews, Emma Brand and Anne Boushall for their support during radio tracking sessions.
Funding
This study was supported by the People’s Trust for Endangered Species and British Hedgehog Preservation Society (PTES/2117).
Author information
Authors and Affiliations
Contributions
AG and PJB designed the study. AG conducted the fieldwork, GIS data collection and householder surveys, and processed and analysed the data. PJB analysed patterns of missing GPS fixes. AG and PJB wrote the manuscript with comments from RWY. AG, PJB and RWY reviewed and edited the final draft.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
All ecological surveys were conducted under licence from Natural England, and face-to-face questionnaire surveys were granted approval by the ethical review panel of the School of Biological Sciences at the University of Reading.
Additional information
Handling editor: Juan Carranza.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gazzard, A., Yarnell, R.W. & Baker, P.J. Fine-scale habitat selection of a small mammalian urban adapter: the West European hedgehog (Erinaceus europaeus). Mamm Biol 102, 387–403 (2022). https://doi.org/10.1007/s42991-022-00251-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-022-00251-5