Abstract
Soil health is a key factor for the global food security. One of the main bottlenecks for the efficient utilization of farmland soil is the accumulation of allelochemicals excreted by plant roots, leached and volatilized from plants, and released during the decomposition of plant residues. The negative impacts derived from allelochemicals include: decreasing soil fertility, breaking the balance of soil microbial community, reducing seed germination rate and crop yield, and interfering with the growth of neighboring or successive plants. Recently, biochar, a soil amendment with low cost and high performance, is extensively studied on its functions and effects on farmland systems. In this review, we focused on the potential role of biochar in controlling the environmental fate and risk of allelochemicals in soils. The source and potential toxicity of allelochemicals in farmland and the deficiencies in current methods for alleviating the allelopathy were summarized first, followed by the discussion on their sorption and degradation by biochar as well as the impact of biochar properties on the performance and mechanism. At the same time, the generation of allelochemicals regulated by biochar through improving plant growth conditions was analyzed. The modification of biochar for alleviating the allelopathy and then overcoming the obstacles of agricultural production was also addressed. Future research directions, such as targeted biochar production, the interactions between different allelochemicals and biochar investigated by simulations and modeling, negative effects and stability of biochar or modified biochar, were then proposed. This review will provide useful theoretical and technical supports for targeted biochar applications in soils with allelopathy.
Graphical abstract

Highlights
1. Biochar can regulate the generation and release of allelochemicals by plant and be more effective after modification;
2. Biochar alleviates the allelopathy by adsorbing or degrading allelochemicals and buffering ecological stress;
3. Biochar promotes biodegradation of allelochemicals by activating soil organisms and providing habitats for them.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
With the fast increase of the global population, the demand for food is also growing explosively. To ensure the grain yield and quality, intensive soil cultivation is promoted globally, resulting in the deterioration of soil quality and reduction of crop yields (Lu and Huang 2010; Zhou et al. 2017). The obstacle of crop rotation has become the main bottleneck of efficient soil utilization, which will exacerbate the competition between plants for resources in the exhausted soil. Even worse, the plants may secrete many chemicals to ensure themselves in the leading position of the competition. These competitions may reduce the soil fertility, seed germination rate and crop yields, or disturb the balance of microbial communities (Li et al. 2021). Most of the above negative impacts are associated with the accumulation of allelochemicals (Bennett et al. 2012). These chemicals are secondary metabolites generated by plants. They enter soil through volatilization from plant stems and leaves, secretion from the root, decomposition of plant residues, and leaching from leaves by rain or dew (Inderjit 2005; Weston and Mathesius 2013). The content of allelochemicals generated by plants is highly determined by the nutrients, pH value, humidity, microbial community structure and activity, planting structure and density, as well as the chemical stresses of the soil environment (Duke 2010). On one hand, allelochemicals could induce the colonization of beneficial microorganisms, and thus plant can take advantage in competing for resources (Qu and Wang 2008). On the other hand, these chemicals can inhibit the growth of surrounding plants by interfering with their photosynthesis, interrupting the synthesis of adenosine triphosphate (ATP), and disrupting the normal metabolism of plants (Kumar and Bais 2010; Weir et al. 2004). In high-density crop planting, the accumulated allelochemicals may exceed the defense ability of its own kind (Yang et al. 2015). Therefore, understanding the release and regulation of allelochemicals is crucial for improving the utilization efficiency of cultivated soil.
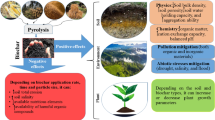
Generally, allelochemical release is heavily associated with the environmental stresses of plants. Biochar has been proven to improve soil environment and microbial activity (Fig. 1). Biochar is a kind of carbon-rich particle produced by pyrolyzing waste biomass under hypoxic conditions. Biochar application can increase soil porosity and pH, retain water and nutrient (Yang et al. 2016), and immobilize various contaminants (Kondrlova et al. 2018), which could reduce the environmental stress on crops and thus decrease the generation of allelochemicals. Moreover, biochar can provide habitats for microorganisms or establish a redox environment in soil, which promotes allelochemical degradation, biologically and/or chemically (Yan et al. 2021). Therefore, biochar displays a promising potential to alleviate the allelopathy as a soil amendment. However, some technical and practical barriers are reported in biochar application in large quantities, such as its relatively high cost, release of PAHs, and high redox potential (Das et al. 2020). Unfortunately, most previous work only focused on a certain process in soil, lacking attention on the comprehensive system with multiple processes, especially in complex environmental media.
Illustration of the interactions between biochar, plants, allelochemicals, and soil microorganisms. Biochar can adsorb and degrade allelochemicals, which is beneficial in decreasing the activities of allelochemicals. The microorganisms sheltered by biochar could enhance the biodegradation of allelochemicals. Generation of allelochemicals by plants may be decreased due to the favorite growth situation established by biochar and microorganisms
In this review, the occurrence and implications of allelochemicals in soil systems were first introduced. The feasibility of biochar for regulating the release and environmental fate of allelochemicals were examined and summarized. The interactions between biochar and microorganisms, plants, or allelochemicals were systematically analyzed and discussed. The modification of biochar with enhanced functions for alleviating allelopathy was also proposed. This review concludes with emphasizing the future challenges and opportunities of biochar application in mitigating allelopathic effects and eventually contributing to sustainable agriculture.
2 The negative impacts of allelochemicals in soil systems and the current preventive methods
2.1 The types and negative impacts of allelochemicals generated by plants
Many plants can generate allelochemicals (Wang et al. 2005). For example, allelopathic wheat generated allelochemical (2,4-dihydroxy-7-methoxy-1,4-benzoxazin-3-one) in roots with the concentration of 300 to 900 μg/g dry weight (Kong et al. 2018); three chemicals, veratric acid, maltol, and (−)-loliolide, were generated by crabgrass with the concentrations ranged from 0.16 to 8.10 μg/g (Zhou et al. 2013). The generated allelochemicals enter the soil systems through volatilization from plant stems and leaves, secretion and drip washing from the plant root, decomposition of plant residues, and leaching from leaves by rain or dew (Fig. 1). The allelochemical types and associated plant species, bioactive concentration range, and corresponding action modes are summarized in Table 1. Different allelochemicals may have different target physicochemical processes. For instance, ester allelochemicals can reduce the number of micronucleus in plant root tip cells (Duan et al. 2013); alkaloids can destroy electron transport and ATP synthesis (Veiga et al. 2008); quinones, benzoic acid derivatives, cinnamic acid derivatives, flavonoids, and terpenoids can inhibit the photosynthesis and respiration, nutrient accumulation, nutrient circulation, membrane permeability, enzyme activity, and hormone balance of plants (Jose and Gillespie 1998). Allelochemicals from plants can also cause diseases and pests to the competing species and enlarges their own territory (Yang et al. 2015). They may cause a substantial decrease in the quality and yield of other crops and even the death of other species in extreme cases (Aschehoug et al. 2016). The overall action mechanisms include the inhibition of cell differentiation by changing the cellular and submicroscopic structures, enhancement of cell membrane permeability, reduction of selective permeation, decrease of hormone and enzyme activity, inhibition of photosynthesis, and blockade of gene expression. Mostly, several mechanisms play a synergistic role in inhibiting the growth of plants, which is more pronounced when multiple allelochemicals coexist.
2.2 Deficiencies in current methods for alleviating the allelopathy
Recently, investigators have carried out various studies to alleviate the allelopathy in soil by regulating the biological conditions and environmental factors (Sujeeun and Thomas 2017; Zhang et al. 2018). For instance, Li et al. constructed a reductive system of soil disinfestation to effectively degrade the allelochemicals generated by Panax notoginseng and suppress the pathogens (Li et al. 2019b). However, the variety of allelochemicals produced by different plant species, combined with the complexity of soil systems, pose many difficulties in alleviating allelopathy (Yang et al. 2016). Cropping modes, such as crop rotation and intercropping, are helpful in alleviating allelopathy (Korai et al. 2018). They can maintain the crop yields for a short time and are economically preferred without additional financial investment. However, long-term crop rotation and intercropping may cause higher accumulation and diversity of allelochemicals in soil. Soil still faces the problems of fertility reduction, quality degradation, and microbial imbalance (Korai et al. 2018). The use of chemical fertilizer in soil can supplement nutrients in a short period of time, reduce the occurrence of pests and diseases, and thus alleviate the allelopathy (Ding et al. 2012). However, nutrients may be leached during rain events, and long-term use of chemical fertilizers will cause soil degradation and environmental pollution (Tong et al. 1997). Therefore, it is urgent to find a new method to regulate the generation and release of allelochemicals and release in soil systems and thus improve agricultural production.
3 Biochar application decreases the effective allelochemical concentration through sorption and degradation
Biochar may alleviate the negative impacts of allelochemicals in soil through sorption and degradation owing to its physicochemical properties. The sorption and degradation of allelochemicals by biochar released from plants are presented in Fig. 2.
The interaction mechanisms between biochar and allelochemicals. The sorption occurs with various mechanisms playing roles simultaneously, because of diverse compositions and structures of both allelochemicals and biochar; the degradation of allelochemicals is driven by biochar chemical reactivity-related structures (such as redox potential, persistent free radicals, and functional groups) or biochar mediated processes (such as biodegradation, photodegradation and electron shuttling)
3.1 Biochar properties affecting allelochemical sorption
The physical and chemical properties of biochar determining their sorption characteristics include specific surface area, particle size, porous structure, surface charge, and functional groups. Generally speaking, the higher the pyrolysis temperature, the larger the surface area of biochar, the more sorption sites for organic molecules, thus the greater sorption capacity to allelochemicals (Ahmad et al. 2014). The sorption irreversibility is another dimension that should be carefully evaluated in sorption studies. For example, micropores may play an important role in retaining pollutants (Ma et al. 2019). When allelopathic molecules enter pores with similar size, the pores will be expanded, accompanied by an exothermic process, which makes them difficult to desorb (Ma et al. 2019).
The particle size of biochar is associated with several other properties, such as the surface area and the abundance of surface functional groups, which generally shows a positive relationship with the sorption rate and capacity (Patel et al. 2021). Recently, studies have emphasized the importance of nano-sized biochar in the removal of organic contaminants and its superior performance compared to the bulk counterparts (Shen et al. 2020). The particles of nano-biochar share similar characteristics with engineered nanomaterials with great surface area and abundant functional groups, which have high sorption affinity to organics. It seems that the application of nano-sized biochar could be a more efficient technique to remove allelochemicals compared to bulk biochar (Rajput et al. 2022). However, it should be noted that the biochar particles of colloidal state tend to aggregate or move away from the original application spot, which likely reduces the effectiveness of the nano-size biochar. At the same time, a wide knowledge gap still exists between the laboratory tests of nano-biochar and its pilot application. Thus, the efficiency of nano-sized biochar for organic contaminant removal in soil should be systematically evaluated in pilot-scale studies.
The sorption strength is also highly dependent on the functional groups on biochar (Zhu et al. 2005). For example, oxygen-containing functional groups (OFGs) on biochar could adsorb organics through the formation of electron-donor-acceptor (EDA) complexes in binary and ternary sorption systems (Mohammad et al. 2018). The structures of allelochemicals and biochar may contain aromatic rings, carboxyl groups, hydroxyl groups, ester groups, and quinone groups. These functional groups can be paired to form binary complexes. The binding energies of the interactions can be calculated based on the density functional theory (DFT) calculations and they can be used for comparing the sorption strength (Chen et al. 2021). In these EDA interactions, the donors and acceptors are dependent on the actual sorption systems, which can be elucidated based on the frontier orbital theory in quantum chemical calculations (Chen et al. 2019). The molecular dynamics simulation is also an efficient approach to study the sorption of allelochemicals, by which the sorption process can be visualized and the sorption energy and mechanisms can be elucidated in statistics (Zuo et al. 2022). At the same time, hydrogen bonds including charge-assisted hydrogen bonds (Ahmad et al. 2014), hydrophobic effects, and partitioning could all contribute to allelochemical sorption on biochar (Dai et al. 2019). Notably, the ternary sorption, such as cation bridging formed between the negatively charged allelochemicals and biochar, could also increase the sorption (Xu et al. 2021b).
After their application in soil, biochar particles might be covered by soil organic matter or microbes forming an eco-corona (Wang et al. 2021). In this case, the sorption or degradation may be mainly mediated by the coated organic matter and soil microbes. If this situation is not properly considered, the sorption or degradation of allelochemicals by biochar may be misestimated, most likely overestimated. The contribution of biochar in these conditions may be quantitatively analyzed by measuring the effective surface area.
3.2 Biochar properties affecting allelochemical degradation
Recent studies pointed out that biochar could mediate the degradation of organic pollutants. The degradation of organic pollutants mediated by biochar is recognized as free radical pathway and non-radical pathway (Ruan et al. 2019; Yuan et al. 2017). The main contributors in the free radical pathway are the environmental persistent free radicals (EPFRs), hydroxyl radical (•OH), and superoxide radical (•O2−) (Fang et al. 2015b). The EPFRs on biochar may degrade organic pollutants after their direct contact, or induce •OH or •O2− generation from water or oxygen to degrade organic pollutants (Qin et al. 2018). In addition, it is noted that redox-active structures of biochar might exchange electrons with organic pollutants to promote degradation (Van der Zee and Cervantes 2009), which is known as a non-radical pathway. Especially, the phenolic and quinones functional groups on biochar are electron-donating functional groups and electron-accepting functional groups, respectively (Yuan et al. 2017). Many allelochemicals possess abundant phenolic hydroxyl groups, which are readily participating in redox reactions with biochar. In these cases, the organics were easily attacked by electrons, which promoted both transformation and biotransformation of the chemicals (Yuan et al. 2017). In addition, the strongly oxidized biochar will generate 1O2 through the energy transfer of the quinone structure (Zhang et al. 2021a). It has strong oxidizing properties and can degrade allelochemicals, which is also a non-radical degradation pathway.
Additionally, when biochar is added to soil, biodegradation of allelochemicals can be accelerated. For example, adding biochar to soil increased the diversity of fungi and reduced the diversity of bacteria, which accelerated the biodegradation of allelochemicals (Li et al. 2013). The addition of biochar can increase the biodegradation of phthalates by 36% (Ren et al. 2021). At the same time, the effective activity concentration or even the activity-related structures of the extracellular enzymes may be altered after they interacted with biochar. Therefore, the biodegradation of allelochemicals can be promoted or inhibited but it is heavily associated with the type of enzymes (Fang et al. 2021). Therefore, biochar could effectively alleviate allelopathy in the soil if the chemical or biological degradation could be properly mediated.
3.3 Allelochemical types affecting biochar-allelochemical interactions
The hydrophobic properties, polarity, and molecular size of allelochemicals regulate their interactions with biochar. For example, hydrophilic allelochemicals (p-hydroxybenzoic acid) are more easily adsorbed by biochar (Teixido et al. 2011). The hydrophobic allelochemicals (such as phthalates) could be adsorbed through the hydrophobic effect or van der Waals forces (Dickenson and Inyang 2015). The allelochemicals with larger molecules (such as ginsenosides) are retained in mesopores, while allelochemicals of smaller molecules (such as citric acid) are more easily protected by the micropores of biochar (Ma et al. 2019). It is thus a comprehensive task to alleviate allelopathy using biochar, and it is also enlightening to specifically design biochar to remove a class of targeted allelochemicals. Therefore, how to specifically alleviate the allelopathy produced by different allelochemicals requires more in-depth exploration. In addition, if biochar-allelochemicals interaction mechanisms could be revealed on how allelochemicals complementarily occupy different sorption sites, a biochar designed for a broad-spectrum sorption and degradation ability might be a promising way, which will be discussed in the subsequent section.
4 Biochar application decreases allelochemical generation through improving plant growth condition
The generation of allelochemicals is dependent on the physiological requirement of the plant. Generally, physical or chemical stresses will promote the generation of allelochemicals, while a low amount of allelochemicals will be excreted if the plant is in a preferred condition. The application of biochar may improve soil fertility or productivity, but at the same time, may pose some additional stress to plants. As presented in Fig. 3, the apparent impact of biochar on allelochemical generation may be a combination of various processes, which will be individually discussed in the following subsections.
Positive (green areas below the gray interface) and negative (pink areas above the gray interface) effects of biochar on allelochemical generation. Overall, the addition of biochar induces more positive than negative effects. Specifically, biochar can adsorb or release nutrients, shelter microorganisms, adsorb or release stressful chemicals, and change soil physiochemical/biological properties. The above processes are strongly associated with the release of allelochemicals
4.1 Nutrients released from or retained by biochar
When crops are densely grown, they compete for the limited nutrients (Aschehoug et al. 2016). According to the statistics of intraspecies and interspecies interaction studies in recent years, 67% of these studies showed negative outcomes because of intraspecific and interspecific competitions for the nutrients (Adler et al. 2018). Biochar application may greatly alleviate these competitions, and thus reduce the generation of allelochemicals.
Biochar itself can release a variety of nutrients, such as C, N, P, and K, which can alleviate the nutrient stress, and thus reduce the release of allelochemicals (Lou et al. 2016). The elemental composition and content of biochar largely depend on the raw materials and pyrolysis temperatures. Hair and chicken feather-derived biochar has a high content of N, while animal manure-derived biochar has high contents of P and K (Domingues et al. 2017; Hass et al. 2012). When the pyrolysis temperature increased between 300 and 600 °C, the content of total P and total K in wood and herbal biochar increased accordingly (Zheng et al. 2013). However, it should be noted that not all these nutrients are available to plants. Only soluble components can be readily used for plant growth. As a matter of fact, available P and N decreased with the increasing temperature (300–600 °C) (Zheng et al. 2013). The reason is that P was converted to less soluble minerals at high temperatures, and N was lost as gas products at high temperatures or converted into heterocyclic-N (Zheng et al. 2013). Soil chemistry conditions also play an important role in the nutrient availability of biochar. Taken P as an example, P release in 24 h increased from 20 mmol/kg to 87 mmol/kg as pH decreased from 8.9 to 4.5, which was comparable to conventional P fertilizers (Silber et al. 2010). Therefore, adding biochar to soil can effectively reduce the nutrient limits to plants and decrease the generation and release of allelochemicals.
Biochar can also adsorb nutrients in the soil and release them slowly to function as a slow-release fertilizer. The nutrient detaining by biochar is closely related to the feedstock. For example, rice husk biochar could strongly retain more nutrients than camellia oil shell biochar, because the pores of rice husk biochar were well developed (Zhao et al. 2021). In wheat cultivation experiments, corn stalk biochar was a better slow-release fertilizer matrix than wheat stalk biochar, because of its larger cation exchange capacity (CEC) and higher sorption to nutrients (Zhao et al. 2021).
The intensive tillage may result in both nutrient shortage and allelochemical accumulation (Dong et al. 2018). The application of biochar could introduce some additional nutrients and partly alleviate the nutrient shortage (Rajkovich et al. 2012). At the same time, biochar could adsorb or degrade allelochemicals (Gámiz et al. 2021). Both processes may be associated with the apparent decrease of the detected allelochemicals. Clearly, previous studies did not carefully identify these two processes. How to distinguish these processes will need a specifically designed experiment. The investigators suggested to add a reference experiment with the addition of biochar supernatant to quantify the impact of biochar nutrients on allelochemical release.
4.2 Improvement of soil physical and chemical properties by biochar
The addition of biochar to soil can improve soil effective water content, increase soil porosity, promote soil aggregates, enhance soil CEC, and thus develop a more suitable environment for plant growth, and reduce the generation of allelochemicals (Abel et al. 2013). Studies have shown that effective water content was increased by about 22% after the application of biochar, which can reduce the drought stress on plants (Peake et al. 2014). Soil porosity was also increased by 20% after biochar application, which alleviated the pressure caused by soil compaction on plant growth (Laird et al. 2010). The addition of biochar can increase soil CEC by up to 35.4% due to the lower original content of exchangeable Ca2+ compared with biochar (Hailegnaw et al. 2019). The addition of biochar can also increase soil aggregation by 8–36% because of the increased organic matter contents (Oguntunde et al. 2004). Moreover, biochar is effective to increase soil pH value of acidic soils, thus alleviating soil acidification stress to plants (Wu et al. 2018). Considering the highly heterogeneous structures of biochar, biochar with different properties may have different advantages to alleviate one of the stresses. If a systematic comparison could be conducted on the strength of the individual stresses to trigger the generation and release of allelochemicals, the biochar could be site-specifically designed to overcome the environmental stresses of a certain remediation site.
4.3 Pollutants immobilized or removed by biochar
Soil pollution, such as heavy metals, organic pollutants, has a major negative effect on plant growth. When the pollution exceeds plant tolerance, their self-protective system will be activated and allelochemicals will be secreted to detoxicate the pollution. For example, the secretion of citric acid, malic acid, and succinic acid in the wheat root system increased significantly under Al stress (de Andrade et al. 2011). These allelochemicals could form relatively stable complexes with heavy metals, which reduce the bioavailability and stress of heavy metals on plants (Riaz et al. 2018). Under the stress of organic pollutants, such as phenanthrene, the secretion of organic acids in ryegrass was significantly stimulated (Xie et al. 2008). These allelochemicals can promote the growth of bacteria (Sphingobacteriia and Actinobacteria) that degrade phenanthrene in the rhizosphere, and alleviate the stress of organic pollutants on plants (Li et al. 2019a).
After biochar addition, the bioavailability of heavy metals in the soil will be reduced through sorption, complexation, precipitation, or even redox reactions. For example, some anions in biochar, such as OH−, CO32−, and SiO32−, could form insoluble salts and reduce the content of exchangeable heavy metals (Zhu et al. 2015). Moreover, with the increase of soil pH, the negative charge on the surface of clay minerals also increased, which promoted the sorption of heavy metals by soil particles (Chao et al. 2018). The investigators summarized that the addition of biochar in the soil will reduce the mobility of Cu, Cd, Pb, and Zn by 36.4%, 25.6%, 26.1%, and 16.5%, respectively (Liu et al. 2021). Similarly, organic pollutants can be immobilized through surface sorption, matrix partitioning, sequestration, or degradation (Ruan et al. 2019; Van der Zee and Cervantes 2009). In addition, biochar may accelerate organic pollutant degradation, mediated by biochar redox potential or PFRs as discussed earlier.
It is generally accepted that the generation of allelochemicals stimulated by pollution could be greatly decreased after biochar application. However, the following points need to be carefully evaluated when assessing pollutant-biochar interactions. (1) It is unknown how to estimate the release of the adsorbed pollutants from biochar, especially with the change of soil chemistry conditions. It is easy to understand that the primary process for pollutant detoxification is retention or degradation, not the apparent sorption. Therefore, the batch sorption experiments provide only limited information to understand biochar function. (2) Biochar properties should be considered dynamically in soil systems. The active sites may easily interact with microorganisms or soil chemical compositions, likely being deactivated. Thus, biochar-pollutant interactions are also changed. Only limited studies were carried out to investigate biochar aging and its implications. (3) The sorption of chemicals by biochar may reduce the apparent bioavailability of allelochemicals. However, if the allelochemicals were just adsorbed on biochar without degradation or decomposition, their allelopathy was not affected largely. Even worse, the adsorbed allelochemicals will be accumulated to increase their contact probabilities with soil organisms, including microorganisms and soil animals on the surface of biochar, resulting in the deterioration of soil health.
4.4 Improvement of soil biological/microbial compositions by biochar
The impacts of biochar on soil biological compositions or activities could be understood from enzymes, microorganisms, and animals. The activity of soil enzyme is one of the important indicators for soil quality and determines the decomposition of organic matter and nutrient cycling. Some enzymes are directly related to allelochemical excretion. For example, the activities of rhizosphere enzymes (such as protease, urease, phosphodiesterase, arylsulfatase, and β-glucosidase) are closely associated with the secretion of small molecular organic acids by regulating the C-, N-, S-, and N-cycling (Das et al. 2017). Biochar application could improve enzyme activities in soils through stimulating soil microbial biomass (Khadem and Raiesi 2017) or protect enzymes from the attack of organisms and reactive chemicals (Joseph et al. 2021).
It is widely reported that biochar could alter the microbial communities of soils, and it is also well-known that allelochemical generation could determine or be determined by plant-microorganism communications (Scavo et al. 2019). The porous structure and carbon-based properties of biochar as well as the nutrients and water retained by biochar are all beneficial for microorganism growth, especially in soil with low organic matters (Joseph et al. 2013). Studies have shown that biochar applied to the soil increased the microbial diversity by 1.25 times and the colonization rate of arbuscular mycorrhiza (AM) by 20% (Matsubara et al. 2008). The increase in AM colonization rate is beneficial to increasing the absorption capacity of nutrients and water by plant roots, thereby reducing the secretion of plant allelochemicals.
It should be noted that biochar application does not change only one microorganism species. Investigators generally agreed that biochar application enhanced the abundance of microorganisms, but reported different results on the diversity of the microbial community as affected by biochar addition (Xu et al. 2021a). It is still unknown how biochar-motivated microbial community change is related to allelochemical release by plants because of the complex soil conditions and biochar properties. In addition, biochar not only promotes the growth of beneficial microorganisms but also promotes the growth of harmful microorganisms. It is hardly investigated how to balance the different species for a long term.
The activity of animals in the soil is also important for soil productivity. However, the interactions among soil animals, allelochemicals, and biochar application have not been studied yet. It was reported that allelochemicals could pose significant toxicity to soil animals, including earthworms (Liu et al. 2020) and nematodes (Hamaguchi et al. 2019). Biochar may have dual roles in benefiting soil animal activity. For example, biochar could adsorb or degrade allelochemicals to alleviate their toxic impacts. At the same time, biochar may maintain preferential habitats for soil animals by improving the soil quality (Sanchez-Hernandez et al. 2019).
4.5 The flip side of applying biochar to regulate allelopathy
Volatile organic compounds (VOCs), EPFRs, heavy metals and polycyclic aromatic hydrocarbons (PAHs) contained in biochar may induce oxidative stress in plant roots, destroy the integrity of root tip cell membranes, and stimulate the generation of allelochemicals (Gupta et al. 2011). Fortunately, the contents of these endogenous pollutants in biochar are normally low. The negative effects of biochar may be limited, especially when considering that biochar is generally applied below 5%.
Additionally, it may not be wise to over-inhibit allelochemical release. Some allelochemicals have specific ecological functions and are effective in pollutant detoxification or beneficial microorganism colonization. The concentration range of allelochemicals for the beneficial impacts is still unknown because the literature mostly focused on the implications of allelochemicals, let alone the biochar addition strategies to ensure this beneficial concentration range. Extensive studies of the beneficial concentration ranges of allelochemicals will be useful for synthesizing or producing herbicides based on natural structures (Zhao et al. 2019).
5 Biochar modification to mitigate allelopathy in agriculture
Because of the diversity of biochar feedstock and high heterogeneity of its structures, biochar may not always be selective or effective enough to its targeted application goals. A modification can be designed for a targeted biochar application. Fortunately, biochar modification has been widely studied. Researchers have established a variety of biochar modification technologies, including physical, chemical, biological, and combined methods (Zhang et al. 2021b). For example, the surface area of biochar could be enlarged by acid modification, alkali, or CO2 activations; the intensity of PFRs in biochar can be enhanced by loading metals or metal oxides, N-doping, or incorporation of functional groups; the magnetism can be imparted to biochar by loading magnetic metal oxides on biochar through co-precipitation or hydrothermal method; the relative content of surface functional groups can be manipulated by using oxidizers or reductants (Zhang et al. 2021b). As shown in Fig. 4, biochar can be modified for different applications, all aiming at alleviating allelopathy from different aspects.
Alleviation of allelopathy by modified biochar. Biochar could moderate the stresses from the environment or pollution, and thus plants may generate a lower amount of allelochemicals. At the same time, biochar may adsorb or degrade allelochemicals to decrease their effective concentrations. Biochar can be specifically modified with improved properties to decrease allelochemical generation or regulate their behavior and activity
5.1 Biochar modification to increase allelochemical immobilization or removal
Allelochemical removal includes both sorption and degradation. The enhanced sorption generally correlated with the increased specific surface area and pore volume. The targeted modification methods include steam activation and acid treatment. When the biochar is purged by high-temperature CO2, it will be thermally altered with CO generation, which will increase the micropores. This method is cheap with high feasibility (Rajapaksha et al. 2016). Phosphoric acid is a commonly used modifier. Phosphoric acid is dehydrated during activation of biochar and forms polyphosphates or phosphates cross-linkers. They stay in the porous structure inside the biochar, preventing the pores from shrinking during the pyrolysis (Fierro et al. 2010). When the ash content of the biochar is washed, their internal micropores increase greatly. The increased surface area and porosity all contribute to the improved sorption capacity to allelochemicals.
As discussed earlier, the apparent sorption may not completely inhibit the bioavailability of allelochemicals because of the desorption. Therefore, the strong interaction between biochar and allelochemicals is essential for allelochemical immobilization. A modification method to introduce specifical functional groups, such as oxygen-containing functional groups, especially the carboxyl group, will help to enhance the formation of stable chemical bonds between the biochar and allelochemicals (such as covalent bonds and hydrogen bonds) (Xue et al. 2012).
The doping of biochar is also a promising way to improve their sorption and degradation for allelochemicals. For example, the N-doped biochar exhibited a high catalytic degradation ability for organics owing to the abundant unpaired electrons on N atoms (Chen et al. 2019; Wan et al. 2020); N-doped biochar also can enhance the sorption strength and affinity for sulfamethazine because it could provide both π electrons and unpaired electrons for sorption. Biochar loaded with positively charged inorganic nanoparticles can increase the surface charge of biochar and promote the electrostatic interactions and surface complexations between biochar and allelochemicals (Zhang et al. 2013).
Recent studies have also suggested that biochar is reactive and could mediate the degradation of organics. Some modifications could increase the redox potential of biochar. For example, the modification by borohydride could increase the phenolic hydroxyl group of biochar, and the loading of metals or phenolic compounds on biomass could increase the concentration of PFRs in the biochar (Fang et al. 2015a; Zhong et al. 2019). These modifications could accelerate the degradation of allelochemicals by biochar. However, this degradation of allelochemicals by biochar may not be complete mineralization. It is known that incomplete degradation of organic contaminants could be accompanied by the generation of even more toxic byproducts (Sun et al. 2020). This type of degradation evaluation is still absent in allelochemical-biochar interaction studies.
If biochar is modified to benefit microbial colonization, the degradation of allelochemicals by microorganisms could also be enhanced. The biochar coated with multi-layer conductive structure MXene served as the cathode of microbial electrosynthesis (MES). This modification not only enhanced the conductivity of biochar but also enriched microorganisms (such as Proteobacteria and Firmicutes), which enhanced the chemical and biodegradation of allelochemicals (Tahir et al. 2021). More effectively, some specific allelochemical-degrading microorganisms could be inoculated and protected in biochar for field application.
5.2 Biochar modification to decrease allelochemical generation and release
The decreased allelochemical generation is generally related to favorable growth conditions for plants, such as decreased pollutant stress, increased nutrients, and a balanced microorganism system. According to the previous discussion, it will be much easier to develop modification methods if the purpose is certain. It would be more preferred to modify biochar with one method but with multiple advantages to improve plant growth conditions. For example, acid modification (phosphoric acid, nitric acid, or sulfuric acid) can mostly decompose aromatic substances and lignocellulose, and increase the specific surface area and porosity of biochar (Fierro et al. 2010). In order to achieve the purpose of reducing soil bulk density, increasing soil moisture content, enhancing soil porosity, and reducing the pressure caused by drought compaction on crop growth, phosphoric acid and nitric acid modification can increase P and N contents in biochar and provide a food source for microorganisms (Abd El-Mageed et al. 2021). Sulfuric acid modification can also improve the sorption capacity to Na+, reduce the content of soluble Na+ and salt in the soil, and alleviate the salt stress in soil (Duan et al. 2021). Acidic methanol modified biochar can appropriately increase the specific surface area of biochar, and increase the number of esters and hydroxyl groups, as well as the electron density of oxygen atoms and pH value (Jing et al. 2014). Therefore, the sorption performance of biochar for pollutants is significantly improved.
However, it should be noted that sometimes the modification may not always be beneficial to plant growth. For example, biochar is always washed after modification, and the content of dissolvable organic matter in the biochar will be reduced. The nutrient value of the biochar could be also reduced. In addition, it is possible that some modifying reagents are retained in the pores, which may pose negative effects on the growth of plants. Loading metal oxides on biochar can also cause certain environmental risks. Although the content of the loaded metal oxides is low, the release or dissolution of the metals may impart heavy metal pollutants into soil systems.
6 Conclusions and future perspectives
The accumulation of allelochemicals in soil may inhibit the germination and growth of plants, even reduce crop yields. Biochar could regulate the behavior and fate of allelochemicals according to the following three aspects: 1) it partly overcomes the allelopathy in soil by adsorbing and degrading allelochemicals; 2) it reduces the generation of allelochemicals by buffering environmental or ecological stress for plants; and 3) it stimulates the activity of soil organisms and promotes biodegradation of allelochemicals. Specifically modified biochar could be more effective in alleviating allelopathy because of their enhanced properties in sorption or degradation of allelochemicals, decreasing the generation of allelochemical by plants, and improving the soil conditions targeted, which are all beneficial in decreasing the accumulation of allelochemicals in the soil.
A thorough evaluation should be carefully carried out before the mass application of biochar for allelopathy control. For example, biochar provides habitats or refuges for both beneficial and harmful microorganisms. How to balance the effects on microorganisms in a long term? Another example is that sorption of allelochemicals by biochar leads to their enrichment on the biochar particles. Does this process increase the contact between allelochemicals and microorganisms? How to mitigate the by the application of modified biochar? The regulation of the secretion of plant allelochemicals is a complicated process. If biochar is to be used in solving allelopathy problems, the following studies need to be considered:
-
a.
Because allelochemical generation is determined by various factors, it is important to identify the contribution of different functions of biochar to alleviate allelopathy. The above-mentioned three pathways may be related to different biochar properties. Thus, targeted biochar production is highly dependent on our understanding of the relationship between biochar properties and its functions.
-
b.
Both allelochemicals and biochar are diverse in their types and properties. It may not be easy to establish a general standard to optimize their interactions. It is thus necessary to explore the interactions between different allelochemicals and biochar, summarizing the best combinations to immobilize or degrade allelochemicals by biochar for a certain plant species. It should be noted that it is not possible to test all the combinations. Hence, the integrated investigations of computational simulations, quantitative structure–activity relationship (QSAR) modeling, and meta-analysis might be an efficient way to reduce the experimental workload.
-
c.
The application of modified biochar can be an efficient way to mitigate the generation and release of allelochemicals. The targeted design and preparation of modified biochar should be considered to effectively regulate the concentration range of allelochemicals for beneficial impacts. Nanosization could be a promising technique for biochar modification, but the interactions between nano-biochar and soil animals, allelochemicals, soil microbes, and soil organic matter should be explored. The balance between cost and efficiency of field application of nano-biochar must be accounted.
-
d.
The possible negative effects of biochar or modified biochar should be taken into full consideration before and during application. The amount and type of biochar, the growth phase of plants, and soil characteristics should all be considered to establish a sustainable strategy for biochar application.
-
e.
The stability of biochar or modified biochar in soil should be paid more attention. The decomposition mediated by soil microorganisms and physiochemical aging may change the surface properties and thus the function of biochar, but the pros and cons of plant growth and allelochemical generation are not fully understood.
In summary, the application of biochar in alleviating allelopathy may have significant environmental and ecological benefits. Considering that biochar could be produced using the residual biomass from farmland, a sustainable agroecosystem could be established if biochar functions in relation to the behavior and activity of allelochemicals in soil are fully understood.
Availability of data and materials
Data sharing not applicable to this review since the manuscript does not have any.
associated data.
Abbreviations
- BC:
-
Biochar
- ATP:
-
Adenosine triphosphate
- OFGs:
-
Oxygen functional groups
- π-π EDA:
-
π-π electron-donor-acceptor
- DFT:
-
Density function theory
- EPFRs:
-
Environmental persistent free radicals
- CEC:
-
Cation exchange capacity
- AM:
-
Arbuscular mycorrhiza
- VOCs:
-
Volatile organic compounds
- PAHs:
-
Polycyclic aromatic hydrocarbons
- MES:
-
Microbial electrosynthesis
- QSAR:
-
Quantitative structure–activity relationship
References
Abd El-Mageed TA, Belal EE, Rady MOA, Abd El-Mageed SA, Mansour E, Awad MF, Semida WM (2021) Acidified biochar as a soil amendment to drought stressed (Vicia faba L.) plants: influences on growth and productivity, nutrient status, and water use efficiency. Agronomy 11(7):1290. https://doi.org/10.3390/agronomy11071290
Abel S, Peters A, Trinks S, Schonsky H, Facklam M, Wessolek G (2013) Impact of biochar and hydrochar addition on water retention and water repellency of sandy soil. Geoderma 202:183–191. https://doi.org/10.1016/j.geoderma.2013.03.003
Abrahim D, Takahashi L, Kelmer-Bracht AM, Ishii-Iwamoto EL (2003) Effects of phenolic acids and monoterpenes on the mitochondrial respiration of soybean hypocotyl axes. Allelopathy J 11(1):21–30
Abu-Reidah IM, Arraez-Roman D, Segura-Carretero A, Fernandez-Gutierrez A (2013) Profiling of phenolic and other polar constituents from hydro-methanolic extract of watermelon (Citrullus lanatus) by means of accurate-mass spectrometry (HPLC–ESI–QTOF–MS). Food Res Int 51(1):354–362. https://doi.org/10.1016/j.foodres.2012.12.033
Adler PB, Smull D, Beard KH, Choi RT, Furniss T, Kulmatiski A, Meiners JM, Tredennick AT, Veblen KE (2018) Competition and coexistence in plant communities: intraspecific competition is stronger than interspecific competition. Ecol Lett 21(9):1319–1329. https://doi.org/10.1111/ele.13098
Ahmad M, Rajapaksha AU, Lim JE, Ming Z, Bolan N, Mohan D, Vithanage M, Sang SL, Yong SO (2014) Biochar as a sorbent for contaminant management in soil and water: a review. Chemosphere 99:19–33. https://doi.org/10.1016/j.chemosphere.2013.10.071
Anaya AL, Pelayo-Benavides HR (1997) Allelopathic potential of Mirabilis Jalapa L. (Nyctaginaceae): effects on germination, growth and cell division of some plants. Allelopathy J 4(1):57–68
Aschehoug ET, Brooker R, Atwater DZ, Maron JL, Callaway RM (2016) The mechanisms and consequences of interspecific competition among plants. Annu Rev Ecol Evol S 47(1):263–281. https://doi.org/10.1146/annurev-ecolsys-121415-032123
Bennett AJ, Bending GD, Chandler D, Hilton S, Mills P (2012) Meeting the demand for crop production: the challenge of yield decline in crops grown in short rotations. Biol Rev 87(1):52–71. https://doi.org/10.1111/j.1469-185X.2011.00184.x
Carlsen SCK, Kudsk P, Laursen B, Mathiassen SK, Mortensen AG, Fomsgaard IS (2009) Allelochemicals in rye (Secale cereale L.): cultivar and tissue differences in the production of benzoxazinoids and phenolic acids. Nat Prod Commun 4(2):199–208. https://doi.org/10.1177/1934578X0900400206
Carlsen SCK, Pedersen HA, Spliid NH, Fomsgaard IS (2014, 2012) Fate in soil of flavonoids released from white clover (Trifolium repens L.). Appl Environ Soil Sci:743413. https://doi.org/10.1155/2012/743413
Chao X, Qian X, Han-Hua Z, Shuai W, Qi-Hong Z, Dao-You H, Yang-Zhu Z (2018) Effect of biochar from peanut shell on speciation and availability of lead and zinc in an acidic paddy soil. Ecotox Environ Safe 164:554–561. https://doi.org/10.1016/j.ecoenv.2018.08.057
Chen Q, Wang X, Yi P, Zhang P, Zhang L, Wu M, Pan B (2021) Key roles of electron cloud density and configuration in the adsorption of sulfonamide antibiotics on carbonaceous materials: molecular dynamics and quantum chemical investigations. Appl Surf Sci 536:147757. https://doi.org/10.1016/j.apsusc.2020.147757
Chen Q, Zheng J, Xu J, Dang Z, Zhang L (2019) Insights into sulfamethazine adsorption interfacial interaction mechanism on mesoporous cellulose biochar: coupling DFT/FOT simulations with experiments. Chem Eng J 356:341–349. https://doi.org/10.1016/j.cej.2018.09.055
Chen Y, Yan X (2007) The role of glucosinolates in plant-biotic environment interactions. Acta Ecol Sin 27(6):2584–2593
Chen Y, Zhang L, Wang J (2011) Effects of coumarin application on plant growth and nitrogen metabolism in leaves of Medicago sativa. Allelopathy J. 28(1):105–114
Chung IM, Park SK, Thiruvengadam M, Lee JH, Kim SH, Rajakumar G (2018) Review of the biotechnological applications of rice allelopathy in agricultural production. Weed Biol Manag 18(2):63–74. https://doi.org/10.1111/wbm.12145
Dai Y, Zhang N, Xing C, Cui Q, Sun Q (2019) The adsorption, regeneration and engineering applications of biochar for removal organic pollutants: a review. Chemosphere 223:12–27. https://doi.org/10.1016/j.chemosphere.2019.01.161
Das S, Chou ML, Jean JS, Yang HJ, Kim PJ (2017) Arsenic-enrichment enhanced root exudates and altered rhizosphere microbial communities and activities in hyperaccumulator Pteris vittata. J Hazard Mater 325:279–287. https://doi.org/10.1016/j.jhazmat.2016.12.006
Das SK, Ghosh GK, Avasthe R (2020) Application of biochar in agriculture and environment, and its safety issues. Biomass Convers Bior 1114:1–11. https://doi.org/10.1007/s13399-020-01013-4
de Andrade LR, Ikeda M, do Amaral, L.I., Ishizuka, J. (2011) Organic acid metabolism and root excretion of malate in wheat cultivars under aluminium stress. Plant Physiol Biochem 49(1):55–60. https://doi.org/10.1016/j.plaphy.2010.09.023
Dickenson E, Inyang M (2015) The potential role of biochar in the removal of organic and microbial contaminants from potable and reuse water: a review. Chemosphere 134:232–240. https://doi.org/10.1016/j.chemosphere.2015.03.072
Dinelli G, Bonetti A, Marotti I, Minelli M, Busi S, Catizone P (2007) Root exudation of diclofop-methyl and triasulfuron from foliar-treated durum wheat and ryegrass. Weed Res 47(1):25–33. https://doi.org/10.1111/j.1365-3180.2007.00549.x
Ding H, Wang Z, Chen X, Zhang P, Xu F, Xu Y, Li M, Chen Y (2018) New triterpenoid saponin C-20 epimers from the alkaline-degradation products of ginsenoside re and their cytotoxic activities. Chem Nat Comp 54(3):490–495. https://doi.org/10.1007/s10600-018-2386-z
Ding L, Lin-Lin QI, Jing HW, Feng QS, Juan LI (2008) Preliminary evaluation of the allelopathic potential of four ent-kaurene diterpenes. J Northwest Nor Uni 44(6):74–78
Ding X, Han X, Yao L, Qiao Y, Li L, Na L (2012) Changes in soil organic carbon pools after 10 years of continuous manuring combined with chemical fertilizer in a Mollisol in China. Soil Till Res 122:36–41. https://doi.org/10.1016/j.still.2012.02.002
Domingues RR, Trugilho PF, Silva CA, Melo I, Melo LCA, Magriotis ZM, Sanchez-Monedero MA (2017) Properties of biochar derived from wood and high-nutrient biomasses with the aim of agronomic and environmental benefits. PLoS One 12(5):e0176884. https://doi.org/10.1371/journal.pone.0176884
Dong L, Xu J, Li Y, Fang H, Niu W, Li X, Zhang Y, Ding W, Chen S (2018) Manipulation of microbial community in the rhizosphere alleviates the replanting issues in Panax ginseng. Soil Biol Biochem 125:64–74. https://doi.org/10.1016/j.soilbio.2018.06.028
Duan LJ, Wang HX, Gao LC, Tang ZM, Zong-Hui JI, Fan QT, Cui LX (2013) Effect of phthalate esters on micronuclei rates in vicia faba root tip cells. Carcino Terato Muta 25(6):478–477
Duan M, Liu G, Zhou B, Chen X, Wang Q, Zhu H, Li Z (2021) Effects of modified biochar on water and salt distribution and water-stable macro-aggregates in saline-alkaline soil. J Soils Sediments 21(6):2192–2202. https://doi.org/10.1007/s11368-021-02913-2
Duke SO (2010) Allelopathy: current status of research and future of the discipline: a commentary. Allelopathy J. 25(1):17–29
Ervin G, Wetzel N, Robert G (2000) Allelochemical autotoxicity in the emergent wetland macrophyte Juncus effusus (Juncaceae). Am J Bot 87(6):853–860. https://doi.org/10.2307/2656893
Fang G, Liu C, Gao J, Dionysiou DD, Zhou D (2015a) Manipulation of persistent free radicals in biochar to activate persulfate for contaminant degradation. Environ Sci Technol 49(9):5645–5653. https://doi.org/10.1021/es5061512
Fang GD, Zhu CY, Dionysiou DD, Gao J, Zhou DM (2015b) Mechanism of hydroxyl radical generation from biochar suspensions: implications to diethyl phthalate degradation. Bioresour Technol 176:210–217. https://doi.org/10.1016/j.biortech.2014.11.032
Fang J, Jin L, Meng Q, Wang D, Lin D (2021) Interactions of extracellular DNA with aromatized biochar and protection against degradation by DNase I. J Environ Sci 101:205–216. https://doi.org/10.1016/j.jes.2020.08.017
Fierro V, Muñiz G, Basta AH, El-Saied H, Celzard A (2010) Rice straw as precursor of activated carbons: activation with ortho-phosphoric acid. J Hazard Mater 181(1–3):27–34. https://doi.org/10.1016/j.jhazmat.2010.04.062
Gámiz B, López-Cabeza R, Velarde P, Spokas KA, Cox L (2021) Biochar changes the bioavailability and bioefficacy of the allelochemical coumarin in agricultural soils. Pest Manag Sci 77(2):834–843. https://doi.org/10.1002/ps.6086
Gupta B, Pathak GC, Pandey N (2011) Induction of oxidative stress and antioxidant responses in Vigna mungo by zinc stress. Russ J Plant Phys 58(1):85–91. https://doi.org/10.1134/S1021443711010079
Hailegnaw NS, Mercl F, Pračke K, Száková J, Tlustoš P (2019) Mutual relationships of biochar and soil pH, CEC, and exchangeable base cations in a model laboratory experiment. J Soils Sediments 19(5):2405–2416. https://doi.org/10.1007/s11368-019-02264-z
Hamaguchi T, Sato K, Vicente CS, Hasegawa K (2019) Nematicidal actions of the marigold exudate α-terthienyl: oxidative stress-inducing compound penetrates nematode hypodermis. Biol Open 8(4):bio038646. https://doi.org/10.1242/bio.038646
Hartwig UA, Joseph CM, Phillips DA (1991) Flavonoids released naturally from alfalfa seeds enhance growth rate of rhizobium meliloti. Plant Physiol 95(3):797–803. https://doi.org/10.1104/pp.95.3.797
Hass A, Gonzalez JM, Lima IM, Godwin HW, Halvorson JJ, Boyer DG (2012) Chicken manure biochar as liming and nutrient source for acid appalachian soil. J Environ Qual 41(4):1096–1106. https://doi.org/10.2134/jeq2011.0124
Huang YQ, Han XR, Yang JF, Liang CH, Zhan XM (2013) Autotoxicity of peanut and identification of phytotoxic substances in rhizosphere soil. Allelopathy J. 31(2):297–308
Inderjit (2005) Soil microorganisms: An important determinant of allelopathic activity. Plant Soil 274(1/2):227–236 https://www.jstor.org/stable/24129045
Jennings JA, Nelson CJ (1998) Influence of soil texture on alfalfa autotoxicity. Agron J 90(1):54–58. https://doi.org/10.2134/agronj1998.00021962009000010010x
Jing XR, Wang YY, Liu WJ, Wang YK (2014) Enhanced adsorption performance of tetracycline in aqueous solutions by methanol-modified biochar. Chem Eng J 248:168–174. https://doi.org/10.1016/j.cej.2014.03.006
Jose S, Gillespie AR (1998) Allelopathy in black walnut (Juglans nigra L.) alley cropping. II. Effects of juglone on hydroponically grown corn (Zea mays L.) and soybean (Glycine max L. Merr.) growth and physiology. Plant Soil 203(2):199–205. https://doi.org/10.1023/a:1004353326835
Joseph S, Cowie AL, Van Zwieten L, Bolan N, Budai A, Buss W, Cayuela ML, Graber ER, Ippolito JA, Kuzyakov Y, Luo Y, Ok YS, Palansooriya KN, Shepherd J, Stephens S, Weng Z, Lehmann J (2021) How biochar works, and when it doesn't: a review of mechanisms controlling soil and plant responses to biochar. GCB Bioenergy 13(11):1731–1764. https://doi.org/10.1111/gcbb.12885
Joseph S, Graber ER, Chia C, Munroe P, Donne S, Thomas T, Nielsen S, Marjo C, Rutlidge H, Pan GX (2013) Shifting paradigms: development of high-efficiency biochar fertilizers based on nano-structures and soluble components. Carbon Manag 4(3):323–343. https://doi.org/10.4155/cmt.13.23
Jrg Z, Stephan S, Ranju C, Jens M, Christoph B, Nadine S, Dierk S, Steffen A (2016) Non-targeted profiling of semi-polar metabolites in Arabidopsis root exudates uncovers a role for coumarin secretion and lignification during the local response to phosphate limitation. J Exp Bot 5:1421–1432. https://doi.org/10.1093/jxb/erv539
Kato-Noguchi H, Ino T, Sata N, Yamamura S (2002) Isolation and identification of a potent allelopathic substance in rice root exudates. Physiol Plant 115(3):401–405. https://doi.org/10.1034/j.1399-3054.2002.1150310.x
Kato-Noguchi H, Thi HL, Sasaki H, Suenaga K (2012) A potent allelopathic substance in cucumber plants and allelopathy of cucumber. Acta Physiol Plant 34(5):2045–2049. https://doi.org/10.1007/s11738-012-0997-8
Khadem A, Raiesi F (2017) Influence of biochar on potential enzyme activities in two calcareous soils of contrasting texture. Geoderma 308:149–158. https://doi.org/10.1016/j.geoderma.2017.08.004
Kocacaliskan I, Ceylan M, Terzi I (2009) Effects of juglone on seedling growth in intact and coatless seeds of cucumber (Cucumis sativus cv. Beith Alpha). Sci Res Essays 4(1):39–41. https://doi.org/10.5897/SRE.9000433
Kondrlova E, Horak J, Igaz D (2018) Effect of biochar and nutrient amendment on vegetative growth of spring barley (Hordeum vulgare L. var. Malz). Australian. J Crop Sci 12(02):178–184. https://doi.org/10.21475/ajcs.18.12.02.pne476
Kong C-H, Zhang S-Z, Li Y-H, Xia Z-C, Yang X-F, Meiners SJ, Wang P (2018) Plant neighbor detection and allelochemical response are driven by root-secreted signaling chemicals. Nat Commun 9(1):3867. https://doi.org/10.1038/s41467-018-06429-1
Korai PK, Xia X, Liu XY, Bian RJ, Omondi MO, Nahayo A, Pan GX (2018) Extractable pool of biochar controls on crop productivity rather than greenhouse gas emission from a rice paddy under rice-wheat rotation. Sci Rep 8:1–9. https://doi.org/10.1038/s41598-018-19331-z
Kumar AS, Bais HP (2010) Allelopathy and exotic plant invasion [M]. In: Plant communication from an ecological perspective. Springer, Berlin, Heidelberg, pp 61–74. https://doi.org/10.1007/978-3-642-12162-3_4
Laird DA, Fleming P, Davis DD, Horton R, Wang B, Karlen DL (2010) Impact of biochar amendments on the quality of a typical Midwestern agricultural soil. Geoderma 158:443–449. https://doi.org/10.1016/j.geoderma.2010.05.013
Li B, Zhang Q, Chen Y, Su Y, Sun S, Chen G (2021) Different crop rotation systems change the rhizosphere bacterial community structure of Astragalus membranaceus (Fisch). Bge var mongholicus (Bge) Hsiao Appl Soil Ecol 166:104003. https://doi.org/10.1016/j.apsoil.2021.104003
Li J, Luo C, Zhang D, Cai X, Jiang L, Zhao X, Zhang G (2019a) Diversity of the active phenanthrene degraders in PAH-polluted soil is shaped by ryegrass rhizosphere and root exudates. Soil Biol Biochem 128:100–110. https://doi.org/10.1016/j.soilbio.2018.10.008
Li XJ, Xia ZC, Kong CH, Xu XH (2013) Mobility and microbial activity of allelochemicals in soil. J Agric Food Chem 61(21):5072–5079. https://doi.org/10.1021/jf400949m
Li Y, Wang B, Chang Y, Yang Y, Yao C, Huang X, Zhang J, Cai Z, Zhao J (2019b) Reductive soil disinfestation effectively alleviates the replant failure of Sanqi ginseng through allelochemical degradation and pathogen suppression. Appl Microbiol Biot 103(8):3581–3595. https://doi.org/10.1007/s00253-019-09676-4
Li ZH, Wang Q, Ruan X, Pan CD, Jiang DA (2010) Phenolics and plant allelopathy. Molecules 15(12):8933–8952. https://doi.org/10.3390/molecules15128933
Liu DL, Lovett JV (1993) Biologically active secondary metabolites of barley. II. Phytotoxicity of barley allelochemicals. J Chem Ecol 19(10):2231. https://doi.org/10.1007/BF00979660
Liu J, Sun K, Han L (2021) Effect of biochar on soil heavy metal speciation and bioavailability: a review. Environ Chem 40(6):1643–1658. https://doi.org/10.7524/j.issn.0254-6108.2021011402
Liu J, Xu G, Yin L, Xu X, Armitage DW, Dong T (2020) Invasive plants exert disproportionately negative allelopathic effects on the growth and physiology of the earthworm Eisenia fetida. Sci Total Environ 747:141534. https://doi.org/10.1016/j.scitotenv.2020.141534
Liu P, Liu ZH, Wang CB, Guo F, Wang M, Zhang YF, Dong L, Wan SB (2012) Effects of three long-chain fatty acids present in peanut (Arachis hypogaea L.) root exudates on its own growth and the soil enzymes activities. Allelopathy J. 29(1):13–24
Lou YM, Joseph S, Li LQ, Graber ER, Liu XY, Pan GX (2016) Water extract from straw biochar used for plant growth promotion: An initial test. BioResources 11(1):249–266
Lu X, Huang S (2010) Barriers and solutions to China’s cultivated land protection. Int J Environ Stud 67(2):223–232. https://doi.org/10.1080/00207231003696343
Ma S, Jing F, Sohi SP, Chen J (2019) New insights into contrasting mechanisms for PAE adsorption on millimeter, micron- and nano-scale biochar. Environ Sci Pollut Res 26(18):18636–18650. https://doi.org/10.1007/s11356-019-05181-3
Macias FA (1994) Allelopathy in the search for natural herbicide models [M]. 582, pp 310–329. https://doi.org/10.1021/bk-1995-0582.ch023
Madronich, M.B., Guenther, A.B., Wessman, C.A., 2011. Monoterpene emissions from a ponderosa pine forest. Does age matter? Agu fall meeting abstracts. B51E-0440
Matsubara Y, Hasegawa N, Fukui H (2008) Incidence of fusarium root rot in asparagus seedlings infected with arbuscular mycorrhizal fungus as affected by several soil amendments. J Japan Soc Hortic Sci 2002 71(3):370–374. https://doi.org/10.2503/jjshs.71.370
Mohammad B, Ahmed J, Zhou H, Ngo M (2018) Sorption of hydrophobic organic contaminants on functionalized biochar: protagonist role of π-π electron-donor-acceptor interactions and hydrogen bonds. J Hazard Mater 360:270–278. https://doi.org/10.1016/j.jhazmat.2018.08.005
Natividad CL, de la Cruz IF, Carlos J, Gallego A (2019) Autotoxicity of diterpenes present in leaves of cistus ladanifer L. Plants 8(2):27. https://doi.org/10.3390/plants8020027
Nicol RW, Yousef L, Traquair JA, Bernards MA (2003) Ginsenosides stimulate the growth of soilborne pathogens of American ginseng. Phytochemistry 64(1):257–264. https://doi.org/10.1016/S0031-9422(03)00271-1
Oguntunde PG, Fosu M, Ajayi AE, Giesen NVD (2004) Effects of charcoal production on maize yield, chemical properties and texture of soil. Biol Fer Soil 39(4):295–299. https://doi.org/10.1007/s00374-003-0707-1
Patel M, Kumar R, Pittman CU, Mohan D (2021) Ciprofloxacin and acetaminophen sorption onto banana peel biochars: environmental and process parameter influences. Environ Res 201:111218. https://doi.org/10.1016/j.envres.2021.111218
Peake LR, Reid BJ, Tang X (2014) Quantifying the influence of biochar on the physical and hydrological properties of dissimilar soils. Geoderma 235:182–190. https://doi.org/10.1016/j.geoderma.2014.07.002
Peng W, Ying W, ChuiHua K (2008) Allelopathy of plant volatile monoterpenes mediated by soil: a case study of Ambrosia trifida L. Acta Ecol Sin 28(1):62–68
Porter PM, Banwart WL, Hassett JJ (1986) Phenolic acids and flavonoids in soybean root and leaf extracts. Environ Exp Bot 26(1):65–73. https://doi.org/10.1016/0098-8472(86)90054-7
Qin Y, Li G, Gao Y, Zhang L, Ok YS, An T (2018) Persistent free radicals in carbon-based materials on transformation of refractory organic contaminants (ROCs) in water: a critical review. Water Res 137:130–143. https://doi.org/10.1016/j.watres.2018.03.012
Qu XH, Wang JG (2008) Effect of amendments with different phenolic acids on soil microbial biomass, activity, and community diversity. Appl Soil Ecol 39(2):172–179. https://doi.org/10.1016/j.apsoil.2007.12.007
Queiroz SCN, Cantrell CL, Duke SO, Wedge DE, Nandula VK, Moraes RM, Cerdeira AL (2012) Bioassay-directed isolation and identification of phytotoxic and fungitoxic acetylenes from conyza canadensis. J Agr Food Chem 60(23):5893–5898. https://doi.org/10.1021/jf3010367
Rajapaksha AU, Chen SS, Tsang DC, Zhang M, Vithanage M, Mandal S, Gao B, Bolan NS, Ok YS (2016) Engineered/designer biochar for contaminant removal/immobilization from soil and water: potential and implication of biochar modification. Chemosphere 148:276–291. https://doi.org/10.1016/j.chemosphere.2016.01.043
Rajkovich S, Enders A, Hanley K, Hyland C, Zimmerman AR, Lehmann J (2012) Corn growth and nitrogen nutrition after additions of biochars with varying properties to a temperate soil. Biol Fert Soil 48(3):271–284. https://doi.org/10.1007/s00374-011-0624-7
Rajput VD, Minkina T, Ahmed B, Singh VK, Mandzhieva S, Sushkova S, Bauer T, Verma KK, Shan S, van Hullebusch ED, Wang B (2022) Nano-biochar: a novel solution for sustainable agriculture and environmental remediation. Environ Res 210:112891. https://doi.org/10.1016/j.envres.2022.112891
Ren X, He XF, Zhang ZF, Yan ZQ, Jin H, Li XZ, Qin B (2015) Isolation, identification, and autotoxicity effect of allelochemicals from rhizosphere soils of flue-cured tobacco. J Agr Food Chem 63(41):8975–8980. https://doi.org/10.1021/acs.jafc.5b03086
Ren X, Tang J, Wang L, Sun H (2021) Combined effects of microplastics and biochar on the removal of polycyclic aromatic hydrocarbons and phthalate esters and its potential microbial ecological mechanism. Front Microbiol 12:647766. https://doi.org/10.3389/fmicb.2021.647766
Riaz M, Yan L, Wu X, Hussain S, Aziz O, Jiang C (2018) Mechanisms of organic acids and boron induced tolerance of aluminum toxicity: a review. Ecotoxicol Environ Saf 165:25–35. https://doi.org/10.1016/j.ecoenv.2018.08.087
Robles MR, Wang N, Kim R, Choi BH (2015) Cytotoxic effects of repin, a principal sesquiterpene lactone of Russian knapweed. J Neuro Res 47(1):90–97 (https://doi.org/10.1002/(SICI)1097-4547(19970101)47:1<90::AID-JNR10>3.0.CO;2-E)
Ruan X, Sun Y, Du W, Tang Y, Liu Q, Zhang Z, Doherty W, Frost RL, Qian G, Tsang DCW (2019) Formation, characteristics, and applications of environmentally persistent free radicals in biochars: a review. Bioresour Technol 281:457–468. https://doi.org/10.1016/j.biortech.2019.02.105
Sanchez-Hernandez JC, Ro KS, Díaz FJ (2019) Biochar and earthworms working in tandem: research opportunities for soil bioremediation. Sci The Total Environ 688:574–583. https://doi.org/10.1016/j.scitotenv.2019.06.212
Scavo A, Abbate C, Mauromicale G (2019) Plant allelochemicals: agronomic, nutritional and ecological relevance in the soil system. Plant Soil 442(1):23–48. https://doi.org/10.1007/s11104-019-04190-y
Shao H, Huang X, Zhang Y, Zhang C (2013) Main alkaloids of Peganum harmala L. and their different effects on dicot and monocot crops. Molecules 18(3):2623–2634. https://doi.org/10.3390/molecules18032623
Shen Y, Tang H, Wu W, Shang H, Zhang D, Zhan X, Xing B (2020) Role of nano-biochar in attenuating the allelopathic effect from Imperata cylindrica on rice seedlings. Environ Sci Nano 7(1):116–126. https://doi.org/10.1039/c9en00828d
Silber A, Levkovitch I, Graber ER (2010) pH-dependent mineral release and surface properties of cornstraw biochar: agronomic implications. Enviro Sci Technol 44(24):9318–9323. https://doi.org/10.1021/es101283d
Sujeeun L, Thomas SC (2017) Potential of biochar to mitigate allelopathic effects in tropical island invasive plants. Trop Conserv Sci 10:194008291769726. https://doi.org/10.1177/1940082917697264
Sun H, He F, Choi W (2020) Production of reactive oxygen species by the reaction of periodate and hydroxylamine for rapid removal of organic pollutants and waterborne bacteria. Environ Sci Technol 54(10):6427–6437. https://doi.org/10.1021/acs.est.0c00817
Suo MR, Yang JS (2006) Survey in studies on chemical constituents of sesquiterpene and their physiological activities in plants of Helianthus L. Chin Trad Herbal Drug 37(1):135–140
Tahir K, Miran W, Jang J, Maile N, Shahzad A, Moztahida M, Ghani AA, Kim B, Jeon H, Lee DS (2021) MXene-coated biochar as potential biocathode for improved microbial electrosynthesis system. Sci Total Environ 773:145677. https://doi.org/10.1016/j.scitotenv.2021.145677
Teixido M, Pignatello JJ, Beltran JL, Granados M, Peccia J (2011) Speciation of the Ionizable antibiotic sulfamethazine on black carbon (biochar). Environ Sci Technol 45(23):10020–10027. https://doi.org/10.1021/es202487h
Tong Y, Emteryd O, Lu D, Grip H (1997) Effect of organic manure and chemical fertilizer on nitrogen uptake and nitrate leaching in a Eum-orthic anthrosols profile. Nutr Cycl Agroecos 48(3):225–229. https://doi.org/10.1023/A:1009730632387
Tyski S, Markiewicz M, Gulewicz K, Twardowski T (1988) The effect of lupin alkaloids and ethanol extracts from seeds of lupinus angustifolius on selected bacterial strains. J Plant Physiol 133(2):240–242. https://doi.org/10.1016/S0176-1617(88)80144-5
Van der Zee FP, Cervantes FJ (2009) Impact and application of electron shuttles on the redox (bio) transformation of contaminants: a review. Biotechnol Adv 27(3):256–277. https://doi.org/10.1016/j.biotechadv.2009.01.004
Veiga TAM, Gonzalez-Vazquez R, Braga PAC, Silva M, King-Diaz B, Lotina-Hennsen B (2008) Alkaloids isolated from Hortia superba (Rutaceae) interact with spinach thylakoids inhibiting the electron transport chain. Allelopathy J. 21(1):133–143
Wan Z, Sun Y, Tsang DCW, Khan E, Yip ACK, Ng YH, Rinklebe J, Ok YS (2020) Customised fabrication of nitrogen-doped biochar for environmental and energy applications. Chem Eng J 401:126136. https://doi.org/10.1016/j.cej.2020.126136
Wang L, Gao C, Yang K, Sheng Y, Xu J, Zhao Y, Lou J, Sun R, Zhu L (2021) Effects of biochar aging in the soil on its mechanical property and performance for soil CO2 and N2O emissions. Sci Total Environ 782:146824. https://doi.org/10.1016/j.scitotenv.2021.146824
Wang P, Bi SP, Ma LP, Han WY (2006) Aluminum tolerance of two wheat cultivars (Brevor and Atlas66) in relation to their rhizosphere pH and organic acids exuded from roots. J Agr Food Chem 54(26):10033–10039. https://doi.org/10.1021/jf0611769
Wang R, Zhou B, Zhang F, Zhang Q (2005) Allelopathic effects of root extracts of egg plants on Verticillium wilt (Verticillium dahliae). Allelopathy J. 15:75–83
Weir TL, Park SW, Vivanco JM (2004) Biochemical and physiological mechanisms mediated by allelochemicals. Curr Opin Plant Biol 7(4):472–479. https://doi.org/10.1016/j.pbi.2004.05.007
Weston LA, Mathesius U (2013) Flavonoids: their structure, biosynthesis and role in the rhizosphere, including allelopathy. J Chem Ecol 39(2):283–297. https://doi.org/10.1007/s10886-013-0248-5
Willis RJ (2000) Juglans spp, juglone and allelopathy. Allelopathy J. 7(1):1–55
Wu L, Kobayashi Y, Wasaki J, Koyama H (2018) Organic acid excretion from roots: a plant mechanism for enhancing phosphorus acquisition, enhancing aluminum tolerance, and recruiting beneficial rhizobacteria. Soil Sci Plant Nutr 64(6):697–704. https://doi.org/10.1080/00380768.2018.1537093
Xie M, Yan C, Ye J (2008) Effect of phenanthrene on the secretion of low molecule weight organic compounds by ryegrass root. Ecol Environ 17(2):576–579
Xu N, Wei M, Wang C, Shi W, Tian FM, Wang XF (2013) Composition of welsh onion (Allium fistulosum L.) root exudates and their allelopathy on cucumber sprouts and fusarium oxysporum f.sp cucumerinum. Allelopathy J. 32(2):243–256
Xu W, Whitman WB, Gundale MJ, Chien C-C, Chiu C-Y (2021a) Functional response of the soil microbial community to biochar applications. GCB Bioenergy 13(1):269–281. https://doi.org/10.1111/gcbb.12773
Xu Y, Yu X, Xu B, Peng D, Guo X (2021b) Sorption of pharmaceuticals and personal care products on soil and soil components: influencing factors and mechanisms. Sci Total Environ 753:141891. https://doi.org/10.1016/j.scitotenv.2020.141891
Xue Y, Gao B, Yao Y, Inyang M, Zhang M, Zimmerman AR, Ro KS (2012) Hydrogen peroxide modification enhances the ability of biochar (hydrochar) produced from hydrothermal carbonization of peanut hull to remove aqueous heavy metals: batch and column tests. Chem Eng J 200:673–680. https://doi.org/10.1016/j.cej.2012.06.116
Yamamoto Y (2009) Movement of allelopathic compound coumarin from plant residue of sweet vernalgrass (Anthoxanthum odoratumL.) to soil. Grassl Sci 55(1):36–40. https://doi.org/10.1111/j.1744-697X.2009.00135.x
Yan T, Xue J, Zhou Z, Wu Y (2021) Biochar-based fertilizer amendments improve the soil microbial community structure in a karst mountainous area. Sci Total Environ 794:148757. https://doi.org/10.1016/j.scitotenv.2021.148757
Yang D, Liu Y, Liu S, Li Z, Tan X, Huang X, Zeng G, Lu Z, Zheng B (2016) Biochar to improve soil fertility. A review. Agron Sustain Dev 36(2):1–18. https://doi.org/10.1007/s13593-016-0372-z
Yang M, Zhang X, Xu Y, Mei X, Jiang B, Liao J, Yin Z, Zheng J, Zhao Z, Fan L (2015) Autotoxic ginsenosides in the rhizosphere contribute to the replant failure of panax notoginseng. PLoS One 10(2):e0118555. https://doi.org/10.1371/journal.pone.0118555
Yeasmin R, Motoki S, Yamamoto S, Nishihara E (2013) Allelochemicals inhibit the growth of subsequently replanted asparagus (Asparagus officinalisL.). Biol Agric Hortic 29(3):165–172. https://doi.org/10.1080/01448765.2013.796570
Yongvanich T, Juntarajumnong W, Nakapong S, Chulalaksananukul W (2002) Allelopathic effect of flavonoids from Typha angustifolia on seed growth of Mimosa pigra. Thai J Agric Sci 2007:361–367
Yuan Y, Bolan N, Prevoteau A, Vithanage M, Biswas JK, Ok YS, Wang H (2017) Applications of biochar in redox-mediated reactions. Bioresour Technol 246:271–281. https://doi.org/10.1016/j.biortech.2017.06.154
Yuansen HU, Cuixiang LI, Guoyin DU, Yafeng LIU, Xincheng JIA (2007) Identification of allelochemicals in cucumber root exudates and its allelopathy to radicle and fusarium oxysporum. Ecol Environ 16(3):954–957
Zhang K, Sun P, Khan A, Zhang Y (2021a) Photochemistry of biochar during ageing process: reactive oxygen species generation and benzoic acid degradation. Sci Total Environ 765:144630. https://doi.org/10.1016/j.scitotenv.2020.144630
Zhang M, Gao B, Varnoosfaderani S, Hebard A, Yao Y, Inyang M (2013) Preparation and characterization of a novel magnetic biochar for arsenic removal. Bioresour Technol 130:457–462. https://doi.org/10.1016/j.biortech.2012.11.132
Zhang P, Duan W, Peng H, Pan B, Xing B (2021b) Functional biochar and its balanced design. ACS Environl Au 2(2):115–127. https://doi.org/10.1021/acsenvironau.1c00032
Zhang Z, Chen L, Wang J, Yao J, Li J (2018) Biochar preparation fromSolidago canadensisand its alleviation of the inhibition of tomato seed germination by allelochemicals. RSC Adv 8(40):22370–22375. https://doi.org/10.1039/c8ra03284j
Zhao H-H, Kong C-H, Xu X-H (2019) Herbicidal efficacy and ecological safety of an allelochemical-based benzothiazine derivative. Pest Manag Sci 75(10):2690–2697. https://doi.org/10.1002/ps.5377
Zhao Z, Wang X, Li H, Ren S, Chen J, Wang L (2021) Slow-release property and soil remediation mechanism of biochar-based fertilizers. J Plant Nutr Ferti 27(5):886–897
Zheng H, Wang Z, Deng X, Zhao J, Xing B (2013) Characteristics and nutrient values of biochars produced from giant reed at different temperatures. Bioresour Technol 130(2):463–471. https://doi.org/10.1016/j.biortech.2012.12.044
Zhiqiang Y, Liming Z, Hui J, Bo Q (2015) Potential ecological roles of flavonoids fromStellera chamaejasme. Plant Signa Behav 10(3):e1001225. https://doi.org/10.1080/15592324.2014.1001225
Zhong D, Jiang Y, Zhao Z, Wang L, Chen J, Ren S, Liu Z, Zhang Y, Tsang DCW, Crittenden JC (2019) pH dependence of arsenic oxidation by rice-husk-derived biochar: roles of redox-active moieties. Environ Sci Technol 53(15):9034–9044. https://doi.org/10.1021/acs.est.9b00756
Zhou B, Kong C-H, Li Y-H, Wang P, Xu X-H (2013) Crabgrass (Digitaria sanguinalis) allelochemicals that interfere with crop growth and the soil microbial community. J Agr Food Chem 61(22):5310–5317. https://doi.org/10.1021/jf401605g
Zhou D, Song X, Zhao F, Gu B (2017) Soil environment and pollution remediation. Pedosphere 27(3):387–388. https://doi.org/10.1016/s1002-0160(17)60359-x
Zhu DQ, Kwon S, Pignatello JJ (2005) Adsorption of single-ring organic compounds to wood charcoals prepared under different thermochemical conditions. Environ Sci Technol 39(11):3990–3998. https://doi.org/10.1021/es050129e
Zhu Q, Wu J, Wang L, Yang G, Zhang X (2015) Effect of biochar on heavy metal speciation of paddy soil. Water Air Soil Pollut 226(12):1–10. https://doi.org/10.1007/s11270-015-2680-3
Zunino M, Zygadlo J (2004) Effect of monoterpenes on lipid oxidation in maize. Planta 219(2):303–309. https://doi.org/10.1007/s00425-004-1216-7
Zuo X, Yi P, Chen Q, Wu M, Zhang L, Pan B, Xing B (2022) Inter-molecular interactions of phthalic acid esters and multi-stage sorption revealed by experimental investigations and computation simulations. Chem Eng J 431:134018. https://doi.org/10.1016/j.cej.2021.134018
Acknowledgments
The authors thank the valuable comments of anonymous reviewers and Editor.
Funding
This work was supported by the National Natural Science Foundation of China (41725016, 42067055, and 41907322), a joint fund between NSFC-NCN (41961134002), and USDA Hatch Program (MAS 00549).
Author information
Authors and Affiliations
Contributions
Quan Chen: Investigation, writing-original draft, writing-review & editing, funding acquisition; Pinyao Lan: Investigation, writing-original draft; Min Wu: Investigation, writing-original draft, funding acquisition; Meng Lu: Investigation; Bo Pan: writing-original draft, writing-review & editing, funding acquisition; Baoshan Xing: writing-original draft, writing-review & editing, funding acquisition. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Agree.
Consent for publication
Agree.
Competing interests
Bo Pan and Baoshan Xing are the editorial board member for Carbon Research and was not involved in the editorial review, or the decision to publish, this article. All authors declare that there are no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Q., Lan, P., Wu, M. et al. Biochar mitigates allelopathy through regulating allelochemical generation from plants and accumulation in soil. carbon res 1, 6 (2022). https://doi.org/10.1007/s44246-022-00003-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44246-022-00003-7