Abstract
Background
Parathyroidectomy (PTX) is often performed in dialysis patients when medical treatment fails to control secondary hyperparathyroidism (SHPT). PTX is viewed by many as a cost-containing measure for patients who have been treated with vitamin D analogs and calcimimetics. Yet, information about health resource utilization and costs before and after PTX is limited.
Methods
This retrospective cohort study used professional service and pharmacy claims to identify subjects on dialysis undergoing PTX from 1/1/2008-12/31/2010. Only subjects with at least six months of information before and after PTX were considered. Subjects with primary hyperparathyroidism or kidney transplant were excluded. Prescription use, physician encounters, and surgical complications were compared during the six months immediately before and after PTX.
Results
The mean (SD) age of the 181 study subjects was 51 (15) years; 59% female; and 80% insured by Medicare. Overall, the percentage of patients receiving medications to manage altered mineral metabolism increased from 67% before to 79% after PTX. Specifically, oral vitamin D use increased, while the utilization of cinacalcet decreased resulting in mean (SD) monthly medication charges decreasing from $486 (507) to $226 (288) (p < 0.01). The mean (SD) number of physician encounters rose from 15 (14) before to 21 (22) per 6 months after PTX (p < 0.01) resulting in the corresponding increase in mean (SD) monthly charges from $1531 (2150) to $1965 (3317) (p = 0.08). Hypocalcemia was the predominant diagnosis recorded for post-surgical physician encounters occurring in 31% of all subjects; 84% of hypocalcemic episodes were managed in acute care facilities.
Conclusions
The cost of medications to manage SHPT decreased after PTX largely due to reduction in cinacalcet use, whereas vitamin D use increased likely to manage hypocalcemia. The frequency and cost of physician encounters, especially in acute care settings, were higher in the 6 months after PTX attributable largely to episodes of severe hypocalcemia. Overall, the reduction in prescription costs during the 6 months after PTX is outweighed by the higher costs associated with physician care.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Secondary hyperparathyroidism (SHPT) associated with alterations in bone and mineral metabolism is common in patients with chronic kidney disease (CKD) [1–3]. SHPT progresses over time manifesting as increasing parathyroid gland hyperplasia and increasing synthesis of parathyroid hormone (PTH). In patients with CKD on dialysis, SHPT is characterized both by elevations in PTH as well as abnormalities in calcium and phosphorus levels. Previous studies [4, 5] have reported that these biochemical abnormalities are associated with adverse health outcomes, including elevated rates of bone fracture, cardiovascular disease, and death [6–8]. It is recommended that patients on dialysis should be monitored for serum calcium and phosphorus every 1–3 months and for PTH every 3–6 months [1], while the specific target levels of these biomarkers are still being debated [9–15].
Pharmacological intervention for SHPT includes vitamin D analogs, phosphate binders, and calcimimetics [3, 16, 17]. Total or partial surgical removal of parathyroid glands, ie, parathyroidectomy (PTX) is considered when patients fail to respond adequately to medical therapy and is recommended in patients with severe HPT defined by PTH > 800 pg/mL [17]. Parathyroidectomy is a viable option for such patients according to the current practice guidelines from the Kidney Disease: Improving Global Outcomes (KDIGO) [18]; it generally improves the short-term and long-term profile of biochemical markers [18–21]. However, the major disadvantage of sub-total PTX are the risks involved in operating in the anterior neck if the patient should need a second surgery; while the disadvantage of total PTX with autotransplant lies in the possibility of prolonged hypocalcemia during the waiting period for the auto transplanted parathyroid tissue to become functional [21]. In a recent, comprehensive review of therapeutic strategies, Stack, BC (2012) noted the decline in parathyroidectomy rates in the US in the 1990s and reported an observed favorable response five years post surgery. Yet, the mortality rate is doubled compared to non-operative SHPT patients and the risk of hypoparathyroidism is also increased, particularly for total PTX with auto-transplantation [19]. Long term consequences of PTX have not been studied sufficiently, mainly due to the ethical issues around potential randomization. However, Kestenbaum et al. (2004) in an observational study of Medicare claims found that patients had higher mortality within 90 days post PTX, but better overall survival 12 months after the surgery compared to those receiving medical therapy to control SHPT [22]. Also, improved survival post-PTX has been reported by Iwamoto et al. [23]. Chen et al. (1998) reported low mortality, short length of hospital stay, and high patient satisfaction among elderly patients undergoing PTX [24].
Based on the literature, PTX appears effective in lowering PTH levels albeit not without risks. However, the current literature remains scarce on the impact of parathyroid surgery on healthcare utilization and costs. The objective of our study was to better understand healthcare utilization and charges before and after PTX among CKD patients on dialysis.
Methods
This retrospective cohort study was based on the data from private practice provider medical claims (Centers for Medicare and Medicaid Services [CMS]; N = 1500 records), and National Council for Prescription Drug Programs prescription claims (NCPDP v5.2) in the IMS database. The database contains approximately one billion submitted professional services claims per year, and pharmacy claims for dispensed prescriptions from 50%-60% of retail pharmacies across various geographic regions in the United States. All patient-related information is encrypted, de-identified, and compliant with the regulations of the Health Insurance Portability, Affordability and Accountability 1996 (HIPAA 1996).
To be included in the study, patients were required to have at least two professional service claims with ICD-9-CM diagnoses for CKD stage 5 or end stage renal disease (ESRD) at least 30 days apart, and one service claim with the Current Procedural Terminology (CPT) code of 60500, 60502 or 60505 for PTX procedure(s) during January 2008 and December 2010. The index date was the first procedure date of PTX. Further, patients were excluded from the study if they were younger than 18 years of age as of index date, had primary hyperparathyroidism, or kidney transplant, or fewer than six months of observation before and after PTX.
We analyzed, during the six months before and after PTX, the pharmacy claims for drug prescriptions, physician encounters and corresponding charges, and surgical complications recorded in the professional service claims including hematoma or bleeding, vocal cord paralysis or recurrent laryngeal nerve injury, hypocalcemia, wound infection and seroma formation. Descriptive statistics, i.e. means, standard deviation (SD) and median, were reported for continuous variables; the number and percentage of patients were reported for categorical variables. The paired t-tests and McNemar tests were performed to assess the differences before and after PTX. All the statistical analyses were executed with Stata 12.1 (StataCorp LP. College Station, TX).
Results
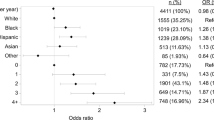
The present study identified 181 patients on dialysis undergoing PTX between 2008 and 2010. Their mean (SD) age was 51.1 (14.7) years. Female patients accounted for 59.1% of the study cohort. Almost half of the study cohort inhabited the South census region and 80.1% were insured by Medicare (Table 1).
Overall the study patients incurred the mean (SD) charges of $4529 (2902) for physician care on the index date – the date receiving PTX. (Table 2) The charges for physician care on index date were mostly billed by surgeon (80.7%) and from inpatient acute care facilities (80.7%).
Based on prescription filling activities in the pharmacy claims, more patients received pharmacotherapy to manage SHPT-related biochemical abnormalities after PTX than before (66.9% pre vs 75.8% post; p < 0.01) (Table 3). The higher medication use post-surgery was mainly attributed to the increased use of oral vitamin D (calcitriol). The percentage of patients filling scripts for vitamin D increased from 11.6% pre- to 59.1% post-surgery (p < 0.01). In contrast, the use of calcimimetics deceased from 34.8% pre- to 11.6% post-surgery (p < 0.01). The use of phosphate binders remained stable before and after PTX. While the study patients filled more prescriptions on average post-surgery compared to pre-surgery, their monthly charges in prescriptions filled to manage SHPT decreased from $486 (SD = 507) pre- to $226 (SD = 288) post-surgery (p < 0.01).
Patients had more all-cause physician encounters after PTX than before PTX (Table 4). The mean number (SD) of all-cause physician encounters during a six-month period increased from 15.1 (14.1) pre-surgery to 20.7 (22.0) post-surgery (p < 0.01). A higher percentage of patients received acute inpatient care attended by their physician care team post-surgery than pre-surgery (79.0% post vs 43.1% pre; p < 0.01); the mean (SD) of six-month physician encounters in the acute inpatient setting increased from 4.5 (9.3) pre-surgery to 10.5 (16.1) post-surgery (p < 0.01). However, physician encounters in the acute inpatient setting accounted for a significant increase, whereas fewer patients had encounters in physician offices or outpatient clinics after the surgery (70.2% post vs. 89.0% pre; p < 0.01). The average physician encounters in the offices or outpatient clinics is fewer during the six-month period of post-surgery than prior to the surgery (3.5, SD = 4.6 post vs 4.3, SD = 5.1; pre: p = 0.03). Healthcare charges billed by physicians also had a marked increase after PTX (Table 5). The mean(SD) monthly charges for all-cause encounters (excluding the actual surgery costs) rose from $1531 (2150) pre-surgery to $1965 (3317) post surgery (p = 0.08). The mean (SD) charges in the acute inpatient setting increased significantly from $681 (1474) pre-surgery to $1209 (1706) post-surgery on average (p < 0.01). Furthermore, approximately one-third of patients had evidence of possible surgery-related complications identified in the professional service claims. Hypocalcaemia was the most frequently diagnosed condition related to surgery and accounted for 90% of patients with possible surgery-related complications (Table 6).
Discussions
This study presented the estimates of short-term healthcare utilization and expenses before and after PTX among patients on dialysis in the U.S. The results from our study suggest that after PTX, the overall cost of medications to manage SHPT decreases largely because of the reduction in the use of cinacalcet. However, vitamin D use increases post-surgery, although the net effect on cost is downward because of a shift to greater use of calcitriol as compared to more expensive vitamin D analogues, presumably for the management of hypocalcaemia. The frequency and cost of physician encounters, especially in the acute care setting, were higher in the 6-month period post surgery. Hypocalcaemia was the dominant post-surgical complication, and it was associated with additional physician care, often provided in the acute care setting. Overall, the cost reduction in prescriptions during the 6-month post surgical period was outweighed by the cost increase associated with physician care.
From the clinical standpoint, parathyroidectomy is often reserved for the patients who have failed to respond to medical therapy for SHPT. While clinical practice worldwide is variable with respect to indications for and frequency of utilization of PTX, one recent study reports that the PTX rate in the US has fluctuated between 1992 and 2007 from the lowest recorded in 1998 (7.0 per 1000 patient years) to the highest during 2002 (12.8 per 1000 patient years) [25], which could reflect changing treatment patterns and guidelines.
Parathyroidectomy may also seem appealing from the economic standpoint arguably because of the ability to save downstream costs of pharmacotherapies [26–28]. Our study highlights important short term clinical and economic consequences of PTX which have not been described previously. Although PTX is effective in lowering serum PTH [29–34], it also commonly results in persistent, inappropriately low PTH levels with the inherent risk of hypocalcemia and adynamic bone disease [35–39]. Additionally, about 10-30% of CKD patients undergoing either subtotal or total PTX still experienced persistent or recurrent SHPT after the surgery [22–24, 26, 27, 29].
Limitations of study
This study has several limitations. Parathyroidectomy is not a very common surgery within the general or CKD population. The study design required at least six months of information before and after PTX; the post PTX cost may have been underestimated. The short study duration was selected because there is no comparator group outside of PTX and the design assumes the intervention of interest. In addition, the study utilized the CPT codes of 60500, 60502 and 60505 to identify patients undergoing PTX. These CPT codes cannot differentiate surgical procedures between total PTX with autotransplantation, and subtotal PTX.
Race, or ethnicity, is generally not available in the professional, medical service claims or pharmacy claims. Thus, it is difficult to evaluate the differences of received treatment and care utilization among different racial or ethnic groups in this study.
This study did not find any claims activities related to the use of IV vitamin D in private practitioner claims or calcium supplement in the pharmacy claims. This is because such patients often receive injectable medications at the dialysis centers making it likely to be recorded in the medical charts in the centers or institutional claims (CMS-1450 form) rather than in professional medical service claims (CMS-1500 form) which was used in this study. Calcium supplements are often available over-the-counter and not included in the prescription formularies.
This study also attempted to bridge the information in the medical and pharmacy claims with laboratory results from a national laboratory provider in order to better understand the changes of clinical endpoints such as serum calcium, phosphorus, PTH and vitamin D before and after PTX. However, there were only few patients who had information available in both claims and laboratory data. It is conceivable that most dialysis patients’ routine blood work is captured within the dialysis centers rather than in facilities outside dialysis centers. Furthermore, the laboratory results are under-reported in the professional service claims (CMS-1500 form) due to the fact most of the laboratory results are not required for reimbursement of professional services.
The professional service and pharmacy claims largely reflect the charges and payments for delivered health services. Neither are designed for research needs to provide patient medical history to assess the disease severity and progression, nor to evaluate cost as these claims tend to be non-adjudicated claims. Finally, the continuum of care may not be fully observed in the claims-based study since treatment may be provided at a healthcare site not reported in the claims or at another practice site.
Conclusion
Our study showed that cost of medications to manage SHPT decreased after PTX largely due to reduction in cinacalcet use, whereas vitamin D (calcitriol) use increased likely to manage hypocalcemia. The frequency and cost of physician encounters, especially in acute care settings, were higher in the 6 months after PTX attributable largely to episodes of severe hypocalcemia. Overall, the reduction in prescription costs during the 6 months after PTX is outweighed by higher costs associated with physician care. The impact of PTX on postoperative healthcare utilization and charges may be a consideration in the treatment decisions for dialysis patients for whom all medical options have been exhausted.
Abbreviations
- CKD:
-
Chronic kidney disease
- CMS:
-
Center for Medicare and Medicaid Services
- CPT:
-
Current procedural terminology
- ESRD:
-
End-stage renal disease
- ICD-9:
-
International classification of diseases, ninth edition, clinical modification
- IV:
-
Intravenous
- PTH:
-
Parathyroid Hormone
- PTX:
-
Parathyroidectomy
- SHPT:
-
Secondary hyperparathyroidism
- SD:
-
Standard deviation.
References
Moe SM, Drüeke TB, Block GA, et al: KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl. 2009, 76: S1-S130.
Tomasello S: Secondary Hyperparathyroidism and Chronic Kidney Disease. Diabetes Spectrum. 2008, 21: 19-25. 10.2337/diaspect.21.1.19.
De Francisco ALM: Secondary Hyperparathyroidism: Review of the Disease and Its Treatment. Clinical Therapeutics. 2004, 26: 12.
Levin A, Bakris GL, Molitch M, Smulders M, Tian J, Williams LA, Andress DL: Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int. 2007, 71: 31-38. 10.1038/sj.ki.5002009.
Vassalotti JA, Uribarri J, Chen S-C, Li S, Wang C, Collins AJ, Calvo MS, Whaley-Connell AT, McCullough PA, Norris KC: Trends in mineral metabolism: Kidney Early Evaluation Program (KEEP) and the National Health and Nutrition Examination Survey (NHANES) 1999–2004. Am J Kidney Dis. 2008, 51: S56-S68. 10.1053/j.ajkd.2007.12.018.
Block GA, Martin KJ, De Francisco ALM, Turner SA, Avram MM, Suranyi MG, Hercz G, Cunningham J, Abu-Alfa AK, Messa P, Coyne DW, Locatelli F, Cohen RM, Evenepoel P, Moe SM, Fournier A, Braun J, McCary LC, Zani VJ, Olson KA, Drüeke TB, Goodman WG: Cinacalcet for secondary hyperparathyroidism in patients receiving hemodialysis. N Engl J Med. 2004, 350: 1516-1525. 10.1056/NEJMoa031633.
Danese MD, Kim J, Doan QV, Dylan M, Griffiths R, Chertow GM: PTH and the risks for hip, vertebral, and pelvic fractures among patients on dialysis. Am J Kidney Dis. 2006, 47: 149-156. 10.1053/j.ajkd.2005.09.024.
Kalantar-Zadeh K, Kuwae N, Regidor DL, Kovesdy CP, Kilpatrick RD, Shinaberger CS, McAllister CJ, Budoff MJ, Salusky IB, Kopple JD: Survival predictability of time-varying indicators of bone disease in maintenance hemodialysis patients. Kidney Int. 2006, 70: 771-780. 10.1038/sj.ki.5001514.
Cassidy MJ, Owen JP, Ellis HA, Dewar J, Robinson CJ, Wilkinson R, Ward MK, Kerr DN: Renal osteodystrophy and metastatic calcification in long-term continuous ambulatory peritoneal dialysis. Q J Med. 1985, 54: 29-48.
Fernandez-Reyes MJ, Auxiliadora Bajo M, Robles P, Selgas R, Oliver J, Del Peso G, Garcia G, Jimenez C, Garcia-Gallego F: Mitral annular calcification in CAPD patients with a low degree of hyperparathyroidism. An analysis of other possible risk factors. Nephrol Dial Transplant. 1995, 10: 2090-2095.
Goodman WG, Goldin J, Kuizon BD, Yoon C, Gales B, Sider D, Wang Y, Chung J, Emerick A, Greaser L, Elashoff RM, Salusky IB: Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N Engl J Med. 2000, 342: 1478-1483. 10.1056/NEJM200005183422003.
Maher ER, Young G, Smyth-Walsh B, Pugh S, Curtis JR: Aortic and mitral valve calcification in patients with end-stage renal disease. Lancet. 1987, 2: 875-877.
Ribeiro S, Ramos A, Brandão A, Rebelo JR, Guerra A, Resina C, Vila-Lobos A, Carvalho F, Remédio F, Ribeiro F: Cardiac valve calcification in haemodialysis patients: role of calcium-phosphate metabolism. Nephrol Dial Transplant. 1998, 13: 2037-2040. 10.1093/ndt/13.8.2037.
Velentzas C, Meindok H, Oreopoulos DG, Meema HE, Rabinovich S, Jones M, Sutton D, Rapoport A, De Veber GA: Visceral calcification and the CaXP product. Adv Exp Med Biol. 1978, 103: 195-201. 10.1007/978-1-4684-7758-0_21.
Massry SG, Coburn JW, et al: K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am. J. Kidney Dis. 2003, 42: S1-S201.
Cunningham J, Locatelli F, Rodriguez M: Secondary Hyperparathyroidism: Pathogenesis, Disease Progression, and Therapeutic Options. Clin J Am Soc Nephrol. 2011, 6: 913-921. 10.2215/CJN.06040710.
Stack BC: Secondary Hyperparathyroidism. British Medical Journal (BMJ) Point of Care. 2012, https://online.epocrates.com/u/29111107/Secondary+hyperparathyroidism.
Neonakis E, Wheeler MH, Krishnan H, Coles GA, Davies F, Woodhead JS: Results of surgical treatment of renal hyperparathyroidism. Arch Surg. 1995, 130: 643-648. 10.1001/archsurg.1995.01430060081015.
Gagné ER, Ureña P, Leite-Silva S, Zingraff J, Chevalier A, Sarfati E, Dubost C, Drüeke TB: Short- and long-term efficacy of total parathyroidectomy with immediate autografting compared with subtotal parathyroidectomy in hemodialysis patients. J Am Soc Nephrol. 1992, 3: 1008-1017.
Gasparri G, Camandona M, Abbona GC, Papotti M, Jeantet A, Radice E, Mullineris B, Dei Poli M: Secondary and tertiary hyperparathyroidism: causes of recurrent disease after 446 parathyroidectomies. Ann Surg. 2001, 233: 65-69. 10.1097/00000658-200101000-00011.
Jofré R, López Gómez JM, Menárguez J, Polo JR, Guinsburg M, Villaverde T, Pérez Flores I, Carretero D, Rodríguez Benitez P, Pérez García R: Parathyroidectomy: whom and when?. Kidney Int Suppl. 2003, S97-S100.
Kestenbaum B, Andress DL, Schwartz SM, Gillen DL, Seliger SL, Jadav PR, Sherrard DJ, Stehman-Breen C: Survival following parathyroidectomy among United States dialysis patients. Kidney Int. 2004, 66: 2010-2016. 10.1111/j.1523-1755.2004.00972.x.
Iwamoto N, Sato N, Nishida M, Hashimoto T, Kobayashi H, Yamasaki S, Ono T, Nishimura M, Tokoro T, Sakoda C, Murakawa M, Okino K, Okamoto Y, Imai R, Adachi N, Ninomiya K, Mabuchi H, Koyama M, Nakanouchi T, Iseki K: Total parathyroidectomy improves survival of hemodialysis patients with secondary hyperparathyroidism. J Nephrol. 2012, 25: 755-763. 10.5301/jn.5000056.
Chen H, Parkerson S, Udelsman R: Parathyroidectomy in the elderly: do the benefits outweigh the risks?. World J Surg. 1998, 22: 531-535. 10.1007/s002689900431. discussion 535–536
Li S, Chen Y, Peng YW, et al: Trends in Parathyroidectomy Rates in US Hemodialysis Patients From 1992 to 2007. Am J Kidney Dis. 2011, 57 (4): 602-611. 10.1053/j.ajkd.2010.10.041.
Narayan R, Perkins RM, Berbano EP, Yuan CM, Neff RT, Sawyers ES, Yeo FE, Vidal-Trecan GM, Abbott KC: Parathyroidectomy Versus Cinacalcet Hydrochloride–Based Medical Therapy in the Management of Hyperparathyroidism in ESRD: A Cost Utility Analysis. Am J Kidney Dis. 2007, 49: 801-813. 10.1053/j.ajkd.2007.03.009.
Schneider R, Kolios G, Koch BM, Fernández ED, Bartsch DK, Schlosser K: An economic comparison of surgical and medical therapy in patients with secondary hyperparathyroidism—the German perspective. Surgery. 2010, 148: 1091-1099. 10.1016/j.surg.2010.09.009.
Komaba H, Moriwaki K, Goto S, Yamada S, Taniguchi M, Kakuta T, Kamae I, Fukagawa M: Cost-Effectiveness of Cinacalcet Hydrochloride for Hemodialysis Patients With Severe Secondary Hyperparathyroidism in Japan. Am J Kidney Dis. 2012, 60: 262-271. 10.1053/j.ajkd.2011.12.034.
Coulston JE, Egan R, Willis E, Morgan JD: Total parathyroidectomy without autotransplantation for renal hyperparathyroidism. Br J Surg. 2010, 97: 1674-1679. 10.1002/bjs.7192.
Low T-HH, Clark J, Gao K, Eris J, Shannon K, O’Brien C: Outcome of parathyroidectomy for patients with renal disease and hyperparathyroidism: predictors for recurrent hyperparathyroidism. ANZ J Surg. 2009, 79: 378-382. 10.1111/j.1445-2197.2009.04893.x.
Puccini M, Carpi A, Cupisti A, Caprioli R, Iacconi P, Barsotti M, Buccianti P, Mechanick J, Nicolini A, Miccoli P: Total parathyroidectomy without autotransplantation for the treatment of secondary hyperparathyroidism associated with chronic kidney disease: clinical and laboratory long-term follow-up. Biomed Pharmacother. 2010, 64: 359-362. 10.1016/j.biopha.2009.06.006.
Rayes N, Seehofer D, Schindler R, Reinke P, Kahl A, Ulrich F, Neuhaus P, Nüssler NC: Long-term results of subtotal vs total parathyroidectomy without autotransplantation in kidney transplant recipients. Arch Surg. 2008, 143: 756-761. 10.1001/archsurg.143.8.756. discussion 761
Schneider R, Slater EP, Karakas E, Bartsch DK, Schlosser K: Initial parathyroid surgery in 606 patients with renal hyperparathyroidism. World J Surg. 2012, 36: 318-326. 10.1007/s00268-011-1392-0.
Stracke S, Keller F, Steinbach G, Henne-Bruns D, Wuerl P: Long-term outcome after total parathyroidectomy for the management of secondary hyperparathyroidism. Nephron Clin Pract. 2009, 111: c102-c109. 10.1159/000191200.
Atsumi K, Kushida K, Yamazaki K, Shimizu S, Ohmura A, Inoue T: Risk factors for vertebral fractures in renal osteodystrophy. Am J Kidney Dis. 1999, 33: 287-293. 10.1016/S0272-6386(99)70302-1.
Coco M, Rush H: Increased incidence of hip fractures in dialysis patients with low serum parathyroid hormone. Am J Kidney Dis. 2000, 36: 1115-1121. 10.1053/ajkd.2000.19812.
Hercz G, Pei Y, Greenwood C, Manuel A, Saiphoo C, Goodman WG, Segre GV, Fenton S, Sherrard DJ: Aplastic osteodystrophy without aluminum: the role of “suppressed” parathyroid function. Kidney Int. 1993, 44: 860-866. 10.1038/ki.1993.323.
Taal MW, Masud T, Green D, Cassidy MJ: Risk factors for reduced bone density in haemodialysis patients. Nephrol Dial Transplant. 1999, 14: 1922-1928. 10.1093/ndt/14.8.1922.
Wang M, Hercz G, Sherrard DJ, Maloney NA, Segre GV, Pei Y: Relationship between intact 1–84 parathyroid hormone and bone histomorphometric parameters in dialysis patients without aluminum toxicity. Am J Kidney Dis. 1995, 26: 836-844. 10.1016/0272-6386(95)90453-0.
Pre-publication history
The pre-publication history for this paper can be accessed here:http://www.biomedcentral.com/1472-6963/13/248/prepub
Acknowledgements
We would like to thank Innokentiy Kasatkin for his assistance in data extraction and management and Ajita De for her assistance in manuscript preparation as well as Holly Tomlin (employee and stockholder, Amgen Inc) for her editing and journal formatting assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The study was funded by Amgen Inc. VB and KC are employees and shareholders of Amgen. GH and CLC are employees of IMS Health.
Authors’ contributions
VB and KC conceived the study. All authors contributed to study design. CLC directed the data extraction and management and performed the statistical analyses in consultation with VB, KC and GH. CLC drafted the manuscript and all authors contributed to critically revising the paper. Finally, all authors read and approved the submitted manuscript.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Belozeroff, V., Cooper, K., Hess, G. et al. Healthcare use and costs before and after parathyroidectomy in patients on dialysis. BMC Health Serv Res 13, 248 (2013). https://doi.org/10.1186/1472-6963-13-248
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1472-6963-13-248