Abstract
This study investigated the lead ion removal of the multi-walled carbon nanotubes (MWCNTs) and oxidized multi-walled carbon nanotubes (MWCNT-COOH). The main purpose of this work is to study the possibilities on the removal of Pb(II) ions from aqueous solutions using MWCNTs and MWCNT-COOH surfaces as adsorbents. Removal of Pb(II) ions was investigated using solutions with different concentrations in the range 10 to 100 mg/L. In this study, removal of Pb(II) ions on surfaces has been investigated by atomic absorption spectrophotometry. The microstructure of carbon nanotubes were characterized using scanning electron microscopy. Three different kinetic theories were applied to experimental data. The kinetic rates were modeled using the pseudo-first-order, four-type linear pseudo-second-order, and intraparticle diffusion. The pseudo-second-order model was found to explain the adsorption kinetics most effectively. The results indicated a significant potential of the multi-walled carbon nanotube as an adsorbent for Pb(II) ion removal.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Exposure to heavy metal ions can cause a variety of adverse health impacts, including serious damages to the immune system, central nervous system, and reproductive system [1]. The contamination of groundwater by heavy metal ions is a major problem in many countries around the world [2]. For example, Pb(II) is a major harmful pollutant to the biosphere, and even trace amounts of it pose a detrimental risk to human health. Although lead is an essential element for the normal metabolism of many living organisms, it is believed that excess lead may be harmful to mankind. So the determination of heavy metal ions is still an intensively active research area [3–5]. Lead is one of the most harmful heavy metals. It has toxic effects on human, animals, and the plants. The main lead pollution resources are battery manufacturing, acid metal plating, ceramic and glass industries, tetraethyl lead manufacturing, and other industries. Lead has fatal effects on the kidney, the nervous system, the liver, and the brain [6]. Therefore, lead must be removed from water. Adsorption is one of the commonly used purification technologies. It is easy to apply and is an economical process. Carbon nanotubes (CNTs) are carbon allotropes consisting of sp2-hybridized carbons, like fullerene and graphene as well as graphite. Since CNTs were discovered by Iijima in 1991, they have existed in a wide variety of forms, i.e., single-walled carbon nanotubes, double-walled carbon nanotubes, few-walled carbon nanotubes, and multi-walled carbon nanotubes (MWCNTs) [7]. Because of so many outstanding performances, CNTs exhibit a great promise for potential applications in many technological fields such as hydrogen storage, catalyst supports, chemical sensors, and nanoelectronic devices [8]. The known ability of CNTs to establish electrostatic interactions and their large surface areas can facilitate the adsorption of many kinds of pollutants from water, such as aniline, phenol, and their substitutes and sodium chloride, as well as several divalent metal ions [9–11]. Surface modifications of CNTs have been applied recently to enhance the dispersion property and adsorption capacities of CNTs. Oxidation of CNTs have been widely reported [12]. During oxidation, the surface characteristics are altered due to the introduction of new functional groups (e.g., COOH, OH) [12]. Many other functional groups could also be appropriate for metal ion adsorption [13, 14]. The goals of this research were to evaluate the removal of behavior of Pb(II) by the MWCNTs and oxidized multi-walled carbon nanotubes (MWCNT-COOH). Three different kinetic theories were applied to experimental data. The kinetic rates were modeled using the pseudo-first-order, four-type linear pseudo-second-order, and intraparticle diffusion.
Methods
MWCNT (purity, >95%; diameter 1 to 2 nm; length, 5 to 30 nm; surface area, approximately 400 m2/g; and manufacturing method, catalytic chemical vapor deposition) and MWCNT-COOH (content of COOH: 1 to 6 wt.%; purity, >95%; average diameter 1 to 2 nm; length 5 to 30 nm, and SSA, 400 m2/g) were purchased from NanoAmor, Nanostructured & Amorphous Materials, Inc, Houston, TX, USA. Doubly distilled water was used and all adsorbents were washed before using. Figure 1 presents SEM images of MWCNTs and MWCNT-COOH. Pb(NO3)2 (molecular weight, 331.20 g/mol) were supplied by Merck, Darmstadt, Germany (maximum purity available). All solutions were prepared with deviations of less than ±0.1% from the desired concentrations.
Adsorption studies
In this research, the 1,000 mg/L stock solution of Pb(II) was prepared by dissolving Pb(NO3)2 in deionized water and adding water to 1,000 mL. The experimental solutions were prepared by diluting the stock solution with deionized water when necessary. To evaluate the equilibrium properties, we first prepared various solutions with initial lead ion concentration from 10 to 100 mg/L and then added 0.05 g MWCNTs and MWCNT-COOH for each, as adsorbent to the solution. These samples were then mounted on a shaker (HZQ-C, Hangzhou Chincan Trading Co., Ltd, Hangzhou, China) and shaken continuously for 5 to 45 min at 298 K. Ultrasonic bath (71020-DTH-E; Model 1510 DTH, 220V; EMS Company,) was used to prevent the particles to aggregate and form a bulk. Then, the samples were centrifuged at 450 rpm, and the supernatant was filtered by 0.2-μm filtered papers for subsequent analysis of lead ion concentration. The filtrates were then immediately examined using atomic absorption spectrophotometry (Perkin-Elmer AAnalyst 700, Waltham, MA, USA) in order to measure the ion concentration. The difference between the initial and the equilibrium ion concentration determines the amount of ion adsorbed onto MWCNT-COOH and MWCNT surfaces. All the experiments were performed in triplicate, and only the mean values are reported. As shown previously, the adsorption of ion on the bottle wall is ignorable. The amount of lead adsorbed was calculated using the following equation:
where C0 and C e (mg/L) are the liquid-phase concentrations of lead at initial and equilibrium concentrations, respectively. V is the volume of the solution (L) and m is the mass of adsorbent used (g). All the experiments were performed in triplicate, and the mean values have been used in calculations.
Results and discussion
Effect of contact time
In order to determine the time required for removal of lead ions by MWCNT-COOH and MWCNT surfaces, 10 mL of 100 mg/L solution was shaken with 0.05 g MWCNTs. The equilibrium studies were performed at the selected interval of time ranging from 5 to 60 min. As shown in Figure 2, after 35 min of stirring the solution, the removal efficiency did not increase. Therefore, the optimum value of stirring time was found to be 35 min. The change in the rate of lead ion removal might be due to the fact that initially all adsorbent sites were vacant and the solute concentration gradient is high. Afterwards, the lead ion uptake rate by the MWCNT-COOH and MWCNT surfaces decreased significantly due to the decrease in adsorption sites. On the basis of these results, a 35-min shaking period was selected for all further studies.
Adsorption kinetics
Kinetic modeling not only allows estimation of sorption rates but also leads to suitable rate expressions characteristic of possible reaction mechanisms. In this respect, several kinetic models including the pseudo-first-order kinetics model, pseudo-second-order kinetics models (four linear forms), and the intraparticle diffusion model were investigated.
Pseudo-first-order model
A pseudo-first-order kinetic equation is given as [15]:
where q t is the amount of lead ions removed at time t (mg/g), q e is the adsorption capacity at equilibrium (mg/g), k1 is the pseudo-first-order rate constant (1/min), and t is the contact time (min). Using Equation 2, ln (q e − q t ) versus t was plotted as shown in Figure 3. The pseudo-first-order rate constant (k1) determined from the model is presented. Results are presented in Tables 1 and 2 for the removal of lead ions by MWCNTs and MWCNT-COOH, respectively, at 25°C.
Pseudo-second-order model
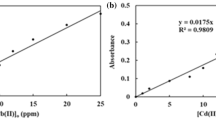
In this model, the rate-limiting step is the surface adsorption that involves chemisorption, where the removal from a solution is due to physicochemical interactions between the two phases [16]. The model is usually represented by its linear form as shown in Figure 2 and Table 3. Fitted pseudo-second-order-model kinetic parameters for the removal of lead ions by MWCNTs and MWCNT-COOH at 25°C are presented in Tables 4 and 5, respectively.
Where k2 (g/mg/min) is the pseudo-second-order rate constant of adsorption. The plot is shown in Figures 4,5,6 and 7.
Pseudo-second-order kinetic model for removal of lead ions by MWCNTs (a) and MWCNT-COOH surfaces (b). (for Equation 2).
Intraparticle diffusion model
The intraparticle diffusion model describes adsorption processes, where the rate of adsorption depends on the speed at which adsorbate diffuses towards adsorbent (i.e., the process is diffusion-controlled), which is presented by Equation 7.
where, k3 is the rate constant of the intraparticle transport (g/mg/min) and c is the intercept [17–20]. The intraparticle diffusion model as fitted with the experimental data is presented in the plot of q t versus t1/2 depicted in Figure 8a,b, and the values of ki and correlation coefficients are given in Tables 6 and 7.
Comparing the results of the kinetics models reveals that the pseudo-second-order model has the best agreement with the experimental data.
Conclusions
The present investigation showed that the MWCNTs and MWCNT-COOH surfaces were effective in the removal of lead ions from the aqueous solution. The surface area of the MWCNT-COOH was relatively high with large-pore-volume MWCNT surfaces as adsorbents The adsorption of Pb(II) ion amount increased with increasing time by both surfaces. Kinetics for the removal of lead ions were obtained and fitted to different kinetics models. Kinetics data were best fitted by the pseudo-second-order model and the result indicated that this adsorbent is excellent in the removal of lead from aqueous solution at variable concentrations.
References
Cho HH, Wepasnick K, Smith BA, Bangash FK, Fairbrother DH, Ball WP, Ball WP: Sorption of aqueous Zn[II] and Cd[II] by multiwall carbon nanotubes: the relative roles of oxygen-containing functional groups and graphenic carbon. Langmuir 2010, 26: 967–981. 10.1021/la902440u
Hyung H, Kim JH: Natural organic matter (NOM) adsorption to multi walled carbon nanotubes: effect of NOM characteristics and water quality parameters. Environ. Sci. Technol. 2008, 42: 4416–4421. 10.1021/es702916h
Xu D, Tan X, Chen CL, Wang XK: Removal of Pb(II) from aqueous solution by oxidized multiwalled carbon nanotubes. J. Hazard. Mater. 2008, 154: 407–416. 10.1016/j.jhazmat.2007.10.059
Sheng GD, Li JX, Shao DD, Hu J, Chen CL, Chen Y, Wang X: Adsorption of copper(II) on multiwalled carbon nanotubes in the absence and presence of humic or fulvic acids. J. Hazard. Mater. 2010, 178: 333–340. 10.1016/j.jhazmat.2010.01.084
Schierz A, Zaenker H: Aqueous suspensions of carbon nanotubes: surface oxidation, colloidal stability and uranium sorption. Environ. Pollut. 2009, 157: 1088–1094. 10.1016/j.envpol.2008.09.045
Giannakopoulos E, Christoforidis KC, Tsipis A, Jerzykiewicz M, Deligiannakis Y: Influence of Pb(II) on the radical properties of humic substances and model compounds. J. Phys. Chem. A 2005, 109: 2223–2232. 10.1021/jp045121q
Sadeghian Z: Large-scale production of multi-walled carbon nanotubes by low cost spray pyrolysis of hexane. New. Carbon. Mater. 2009, 24: 33–38. 10.1016/S1872-5805(08)60034-7
Shao D, Hu J, Wang X: Plasma induced grafting multiwalled carbon nanotube with chitosan and its application for removal of UO 2 2+, Cu2+, and Pb2+ from aqueous solutions. Plasma. Process. Polym. 2010, 7: 977–985. 10.1002/ppap.201000062
Wang P, Shi QH, Liang HJ, Steuerman DW, Stucky GD, Keller AA: Enhanced environmental mobility of carbon nanotubes in the presence of humic acid and their removal from aqueous solution. Small 2008, 4: 2166–2170. 10.1002/smll.200800753
Ren XM, Chen CL, Nagatsu M, Wang XK: Carbon nanotubes as adsorbents in environmental pollution management: a review. Chem. Eng. J. 2011, 170: 395–410. 10.1016/j.cej.2010.08.045
Petersen EJ, Zhang LW, Mattison NT, O’Carroll DM, Whelton AJ, Uddin N, Nguyen T, Huang QG, Henry TB, Holbrook RD, Chen KL: Potential release pathways, environmental fate, and ecological risks of carbon nanotubes. Environ. Sci. Technol. 2011, 45: 9837–9856. 10.1021/es201579y
Zhang D, Pan B, Zhang H, Ning P, Xing BS: Contribution of different sulfamethoxazole species to their overall adsorption on functionalized carbon nanotubes. Environ. Sci. Technol. 2010, 44: 3806–3811. 10.1021/es903851q
Mauter MS, Elimelech M: Environmental applications of carbon-based nanomaterials. Environ. Sci. Technol. 2008, 42: 5843–5859. 10.1021/es8006904
Tian Y, Gao B, Wang Y, Morales VL, Carpena RM, Huang Q, Yang L: Deposition and transport of functionalized carbon nanotubes in water saturated sand columns. J. Hazard. Mater. 2012, 213–214: 265–272.
Li YH, Wang S, Luan Z, Ding J, Xu C, Wu D: Adsorption of Cadmium( II) from aqueous solution by surface oxidized carbon nanotubes. Carbon 2003, 41: 1057–1062. 10.1016/S0008-6223(02)00440-2
Wang H, Zhou A, Peng F, Yu H, Yang J: Mechanism study on adsorption of acidified multi-walled carbon nanotubes to Pb(II). J. Colloid Interface Sci. 2007, 316: 277–283. 10.1016/j.jcis.2007.07.075
Schierz A, Zanker H: Aqueous suspensions of carbon nanotubes: Surface oxidation, colloidal stability and uranium sorption. Envi. Pollution 2009, 157: 1088–1094. 10.1016/j.envpol.2008.09.045
Hameed BH, Daud FBM: Adsorption studies of basic dye on activated carbon derived from agricultural waste: Hevea brasiliensis seed coat. Chem. Eng. J. 2008, 139: 48–55. 10.1016/j.cej.2007.07.089
Ho YS, McKay G: Pseudo-second order model for sorption processes. Process. Biochem. 1999, 34: 451–465. 10.1016/S0032-9592(98)00112-5
Weber TW, Chakkravorti RK: Pore and solid diffusion models for fixed-bed adsorbers. AICHE J. 1974, 20: 228–232. 10.1002/aic.690200204
Acknowledgements
The author would like to thank the Islamic Azad, Islamshahr Branch for their financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The author declares that he has no competing interests.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Robati, D. Pseudo-second-order kinetic equations for modeling adsorption systems for removal of lead ions using multi-walled carbon nanotube. J Nanostruct Chem 3, 55 (2013). https://doi.org/10.1186/2193-8865-3-55
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/2193-8865-3-55