Abstract
Here, we report the novel use of the ethonolic leaf extract of Piper betle for gold nanoparticle (AuNP) synthesis. The successful formation of AuNPs was confirmed by UV-visible spectroscopy, and different parameters such as leaf extract concentration (2%), gold salt concentration (0.5 mM), and time (18 s) were optimized. The synthesized AuNPs were characterized with different biophysical techniques such as transmission electron microscopy (TEM), Fourier transform infrared (FT-IR) spectroscopy, X-ray diffraction (XRD), and energy-dispersive X-ray spectroscopy (EDX). TEM experiments showed that nanoparticles were of various shapes and sizes ranging from 10 to 35 nm. FT-IR spectroscopy revealed that AuNPs were functionalized with biomolecules that have primary amine group –NH2, carbonyl group, –OH groups, and other stabilizing functional groups. EDX showed the presence of the elements on the surface of the AuNPs. FT-IR and EDX together confirmed the presence of biomolecules bounded on the AuNPs. Cytotoxicity of the AuNPs was tested on HeLa and MCF-7 cancer cell lines, and they were found to be nontoxic, indicating their biocompatibility. Thus, synthesized AuNPs have potential for use in various biomedical applications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Synthesis of gold nanoparticles (AuNPs) has gained immense significance during the last few years due to their catalytic, optical, and electrical properties [1]. The existing chemical and physical methods are successful in producing well-defined AuNPs, but these processes usually require use of toxic chemicals. Therefore, the synthesized nanoparticles are not useful in medical and biological applications [2–4]. For this reason, the synthesis of AuNPs using an eco-friendly method is important to address the growing concerns on the overall toxicity of nanoparticles for medical and biotechnological applications. AuNPs have been considered important due to their unique and tunable surface plasmon resonance (SPR) property and their applications in biomedical science including drug delivery, tissue/tumor imaging, photo thermal therapy, immune chromatography, and identification of pathogens in clinical specimens [5]. It is known that the physico-chemical properties of AuNPs are strongly dependent upon their interaction with capping agent molecules [6]. Indeed, the surface chemistry of AuNPs can modify their interaction with external systems [7]. This study describes an eco-friendly method for the synthesis of AuNPs using Piper betle as the reducing and stabilizing agent.
P. betle Linn (family: Piperaceae) leaves are widely used as a post-meal mouth freshener, and the crop is extensively grown in India, Sri Lanka, Malaysia, Thailand, Taiwan, and other Southeast Asian countries. The leaves of this plant are economically and medicinally important and have been traditionally used in India, China. In Thailand leaves are used to prevent oral malodor since it has an antibacterial activity against obligate oral anaerobes responsible for halitosis [8]. Aqueous extracts of P. betle have also been shown to reduce the adherence of early dental plaque bacteria [9]. The leaves of P. betle have a strong pungent and aromatic flavor and are used as a mouth freshener, in wound healing [10], as a digestive and pancreatic lipase stimulant [11], antioxidant [12, 13], antifungal, antibacterial [1, 14–16], anti-inflammatory, bioprotective [17], and antidiabetic [18] agent. In Chinese folk medicine, it is used for curing wind-cold cough, bronchial asthma, rheumatism, stomachalgia, and pregnancy edema [19]. P. betel leaves contain a significant amount of antioxidants such as hydroxychavicol, eugenol, ascorbic acid, and β-carotene [20]. We have successfully synthesized AuNPs by hypothesizing that the presence of strong antioxidants and flavonoids would assist in the reduction of gold ions to AuNPs. To aid this process, we applied microwave (MW) irradiation, which has the advantages of homogeneous heating that directly influence the nucleation process of AuNPs synthesis [21, 22]. Cytotoxicity tests were done to examine the effect of the synthesized AuNPs on the proliferation of two human cancer cell lines and revealed that the synthesized AuNPs were biocompatible. Therefore, AuNPs synthesized using this method can potentially be used in biomedical applications.
Methods
Materials
P. betel was collected from a local market in Guwahati, India. The reagents were of analytical grade obtained either from Merck (Mumbai, India) or Sisco Research Laboratories (Mumbai, India). 3,4,5-Dimethylthiazol-2-yl-2-5-diphenyltetrazolium bromide (MTT) was purchased from Hi Media (Bangalore, India). Cell lines were obtained from the National Centre for Cell Sciences (Pune, India). Cell culture-related plasticware were obtained from Sigma-Aldrich (Bangalore, India).
Preparation of leaf extract
P. betel leaves were washed with deionized water to remove adsorbed dirt. The leaves were chopped into small pieces (2 × 2 cm) and dried at room temperature (25°C) under shade. The dried leaves were powered in a mixer grinder (Bajaj Model GX 11, Mumbai, India). Five grams of powder was dissolved in 50 ml of ethanol and kept at 4°C for 1 week to get the leaf extract. The P. betle leaf extract (PLE) was filtered using a Whatman (50 mm; Sigma, Bangalore, India) filter paper, and the filtrate was stored at 4°C for various experiments.
Synthesis of AuNPs
The synthesis of AuNPs was carried out by varying the PLE concentration (0.5% to 4%) against 0.5 mM HAuCl4 in a total volume of 2 ml made up with double distilled water. The resulting mixtures were placed in a domestic microwave oven (900 W, 2.45 GHz, LG MO- MC-767 W/WS, LG Corp., Noida, India) and irradiated for 15 s. To obtain the optimum concentration of HAuCl4, the experiments were carried out by varying the gold solution concentration against 2% of PLE. The optimum time for synthesis was determined by incubating 2% of PLE with 0.5 mM by varying the MW irradiation time from 12 to 30 s with an interval of 2 s.
Characterization of AuNPs
UV-visible spectroscopy
All UV-visible (UV–vis) spectroscopic measurements of the synthesized AuNPs were carried out on a Cary 100 BIO UV–vis spectrophotometer (Varian, Palo Alto, CA, USA).
Transmission electron microscope
Colloidal solution (5 ml) of synthesized AuNPs was centrifuged twice at 20,000 rpm for 20 min to remove the non-covalently bounded molecules on the their surfaces. The resulting pellet was redispersed in 1 ml of distilled water, a few drops were placed over a carbon-coated copper grid, and the water was evaporated in a hot air oven (Daihan Labtech Co.Ltd. model LDO-150 F, New Delhi, India) at 60°C for 4 h. Transmission electron microscope (TEM) measurements were performed on a TEM instrument (JEOL model 2100, JEOL Ltd., Tokyo, Japan) operated at 190 V of 200 kV.
XRD, FT-IR, and EDX analyses
To obtain the X-ray diffraction (XRD) pattern, AuNP solution was placed on a microscope glass slide and allowed to dry in a hot air oven at 50°C, and the process was repeated to form a layer on the glass slide. The dried samples were analyzed with the help of an XRD instrument (Bruker Advance D8 XRD machine, Bruker, Madison, WI, USA) with a Cu source at 1.5406 Å wavelength in thin film mode.
AuNP colloidal solution (50 ml) was synthesized with optimum parameters (2% of PLE, 0.5 mM HAuCl, and 18 s) and centrifuged at 20,000 rpm for 20 min, and the process was repeated twice to remove non-covalently bounded molecules. The resulting pellet was resuspended in 5 ml of distilled water and lyophilized (Christ Gefriertrocknungsanlagen GmbH Model 1–4, Osterode, Germany) for 16 h. Infrared spectra were recorded using a Fourier transform infrared (FT-IR) spectroscope (Spectrum One, Perkin Elmer, Waltham, MA, USA) from 4,000/cm to 450/cm, with a resolution of 2 cm and five scans/sample by using 1 mg of finely powdered AuNPs prepared with 200 mg of KBr.
The elemental composition of the AuNPs was obtained by using energy-dispersive X-ray spectroscopy (EDX; LEO 1430 VP, Carl Zeiss AG, Oberkochen, Germany) at variable pressure and a scanning electron microscope equipped with INCA Oxford EDX facility at an acceleration voltage of 10 keV.
Cytotoxicity studies
To maintain the HeLa (human cervical cancer) and MCF-7 (human breast cancer) cells, we used the minimal essential medium (MEM) containing 1.0 mM sodium pyruvate, 0.1 mM nonessential amino acids, 1.5 g/L sodium bicarbonate, 2 mM L-glutamine supplemented with 10% FBS (heat inactivated), and 1% antibiotic-antimycotic solution (1,000 U/mL penicillin G, 10 mg/mL streptomycin sulfate, 5 mg/mL gentamycin, and 25 μg/mL amphotericin B). The cells were cultured at 37°C in a humidified incubator (HF 160 W, Heal Force, Shanghai, China) supplemented with 5% CO2.
To examine the cytotoxicity of AuNPs in cancer cells, monocultures of the HeLa and MCF-7 cell lines were incubated for 24 h with increasing concentrations of filter (0.2 micron)-sterilized AuNPs, and the cell viability was estimated by MTT dye conversion assay. Cells not exposed to AuNPs were taken as control. Cells were seeded and maintained (1 × 104) in a 96-well plate (Cell Bind, Corning Inc., Corning, NY, USA) using MEM medium with serum. After 24 h of growth, the medium was replaced with the serum-free medium that contained varied concentrations of AuNPs (10 to 100 μM). The medium was removed after 24 h of treatment, and cells were washed with phosphate-buffered saline (0.01 M, pH 7.2). This was followed by the addition of 100 μl of MTT (0.5 mg/mL) prepared in serum-free medium to each well and incubation for 4 h at 37°C. After incubation, the medium was removed, and 100 μl of dimethyl sulfoxide (DMSO) was added to each well to solubilize the formazan crystals, and the concentration of formazan was determined by measuring its absorbance at 570 nm using a multiwell plate reader (Tecon micro plate reader, model 680, Tecon Inc., San Clemente, CA, USA). The cell viability was calculated with the following equation:
where Atreated and Acontrol are the absorbances of the treated and untreated cells, respectively.
Results and discussion
Effect of PLE and HAuCl4 on the AuNP synthesis
The formation of AuNPs was monitored with the naked eye by observing the appearance of the ruby red color, as shown in Figure 1. P. betel has a high content of phenolic acids, antioxidants, and flavonoids. The abundant hydroxyl groups available in these compounds could participate in the gold bioreduction. The bioreduction of gold could occur through the oxidation of hydroxyl (R–OH) to carbonyl groups (R–C = O), as shown in following equation:
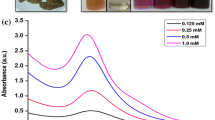
The SPR peak of 2% PLE against 0.5 mM gold solution showed intense and narrow peak centered around 544 nm, as shown in Figure 2a, whereas the SPR peaks above the 2% PLE concentration were flattening and red-shifted, indicating the formation of large-size AuNPs and those below the 2% concentration were considered as suboptimal. Thus, 2% PLE was considered as optimal for the synthesis of AuNPs. Figure 2b shows the UV–Vis spectra of products obtained by reacting 2% PLE with various concentrations of HAuCl4 (0.1 to 1 mM), which displayed an intense peak at 547 nm for 0.5 mM HAuCl4. However, the SPR peaks displayed by the 0.3 mM HAuCl4 were suboptimal, whereas those of 0.7 and 1 mM HAuCl4 were red-shifted. Since there was no significant formation of the peak with 0.1 mM, 0.5 mM HAuCl4 was taken as the optimum for the synthesis of the AuNPs. UV-visible spectral analysis of the different reaction products exhibited sharp SPR peaks presented in Table 1, indicating the successful formation of AuNPs.
UV-Visible absorption spectra of AuNPs. Synthesized by (a) different concentrations of PLE (a = 0.5%, b = 1%, c = 1.5%, d = 2%, e = 2.5%, f = 3%, g = 3.5%, and h = 4%) for 15 s against 0.5 mM HAuCl4, and (b) varying concentrations of HAuCl4 (a = 0.1 mM, b = 0.3 mM, c = 0.5 mM, d = 0.7 mM, and e = 1 mM) against 2% PLE.
Time-dependent synthesis of AuNPs
Figure 3 shows the various experiments carried out to determine the optimum time for the synthesis of AuNPs by incubating the 2% PLE and 0.5 mM gold solution at different time intervals (12 to 30 s) of microwave irradiation. We found that the peaks above 18 s were flattening, indicating no further significant formation of AuNPs. The peaks below 18 s were considered as suboptimal as they displayed poor intensity. This revealed that 18 s was sufficient to reduce the 0.5 mM gold solution to AuNPs, thereby proving it as optimum for the synthesis.
Synthesized AuNPs were characterized with various biophysical techniques
We have employed the TEM technique to visualize the size and shape of formed AuNPs. Figure 4a shows the typical bright-field TEM image of AuNPs obtained with optimum reaction conditions and reveals that the majority of AuNPs formed were circular in shape. The size of the synthesized AuNPs ranges from 10 to 35 nm, and the average size was found to be 6 nm, as shown in Figure 4b. Figure 5a shows the high-resolution TEM (HRTEM) image showing clear lattice fringes of 0.22 nm, revealing that the growth of the AuNPs occurred preferentially on the (1 1 1) plane. The inter-planer distance of the Au (1 1 1) plane was in agreement with the (1 1 1) d-spacing of bulk Au (0.2355 nm) [23, 24]. The crystalline nature of the synthesized AuNPs was confirmed from the selected area electron diffraction (SAED) pattern with bright circular rings corresponding to the (1 1 1), (2 0 0), and (2 2 0) planes, as shown in Figure 5b. [23, 25]. Also, we observed the significant change in the peak position (red-shifted peaks) of the reaction mixture obtained by using 4% PLE and 0.7 mM HAuCl4 for 30 s, indicating the formation of large-size AuNPs, which was confirmed using TEM technique. The TEM images shown in Figure 6 revealed the formation of large-size AuNPs, which agreed with the results obtained in Figure 2.
Figure 7 shows the XRD (Bruker D8 ADVANCE X-ray powder diffractometer, Bruker AXS Inc, Madison, WI, USA) pattern of the AuNPs that showed three prominent Bragg reflections, which were indexed on the basis of the fcc structure of gold. The intensity of the (1 1 1), (2 0 0), and (2 2 0) diffraction peaks corresponding to 38.1°, 44.5°, and 64.8°, respectively, confirmed that the synthesized AuNPs were of crystalline nature [23, 26, 27].
Possibility of functional groups being involved in the synthesis of AuNPs
The involvement of functional groups in synthesized AuNPs was studied by FT-IR. The FT-IR spectra of PLE, as shown in Figure 8a, showed peaks at 3,495 cm−1 (hydroxyl group arising from alcohols and phenolic compounds), 2,865 cm−1 (secondary amine), 1,651 cm−1 (amide I bond of proteins), 1,422 cm−1 (C = C groups from alkenes), 1,328 cm−1 (C–CH3 group), and 1,083 cm−1 ( C–N stretching vibrations of amide bond), whereas the AuNPs showed peaks at 3,402, 2,865, 1,637, 1,365, and 1,046 cm−1, as indicated in Figure 8b[26, 27]. The main significant difference between the spectrum of intact PLE before and after gold reduction is the disappearance peak at 1,328 cm−1 (curve a) and appropriate alteration of 1,422 to 1,365 cm−1. This phenomenon revealed that biomolecules in the PLE, such as antioxidants, phenols, and flovonoids, that contain abundant aromatic C = C groups (1,422 cm−1) may play an important role in the bioreduction and stabilization of AuNPs and may be responsible for the appearance of the significant peak at 1,365 cm−1 corresponding to the C–C bond. The three absorption peaks of AuNPs at 1,637, 2,865, and 3,042 cm−1 are attributed to the primary amines, alkyl, and OH groups, respectively. The reappearance of these groups in the spectrum of AuNPs suggested the attachment of some N–H, N = H, and OH groups onto AuNPs during the biosynthesis process. Hence, it was deduced that AuNPs might be capped and stabilized by biomolecules (antioxidants, flavonoids, and polyphenols) originating from PLE.
EDX analysis
Figure 9 shows the EDX profile of AuNPs, which revealed a strong signal for gold and Cu along with very strong carbon, nitrogen, and oxygen peaks. The signal for Cu originated from the grid used for the analysis. The resulting carbon and oxygen peaks may have originated from the antioxidants/flavonoids/phenolic or organic biomolecules bound to the surface of the AuNPs. The EDX and FT-IR together suggested the presence of biomolecules on the surface of the synthesized AuNPs.
PLE provided a nontoxic coating on the surface of AuNPs
The cytotoxicity of AuNPs under in vitro conditions in HeLa and MCF-7 cells was examined by MTT assay for 24 h in terms of effect of AuNPs on cell proliferation. Only cells that are viable after 24-h exposure to the AuNPs can metabolize MTT efficiently and produce purple colored crystal which is soluble in DMSO. Figure 10 shows unaltered cellular morphologies of two cell lines after treatment with AuNPs, suggesting that treatment with AuNPs did not induce any cytotoxic effect causing significant damage or death of the treated cells. Figure 11 shows the viability of both HeLa and MCF-7 cancer cell lines after 24 h post treatment and showed excellent viability as high as 100 μmol/L of AuNPs. This indicated that the PLE provided a nontoxic coating on the surface of AuNPs. This revealed that the synthesized AuNPs were of insignificant toxicity on cancer line, providing an opportunity for application in drug delivery and molecular imaging.
Conclusions
The method of AuNP synthesis described here has advantages such as rapidness, use of biologically benign biomaterial (PLE), and most importantly, resulting in biocompatible AuNPs. The synthesized AuNPs have insignificant toxicity at a maximum dose of 100 μM; thus, the particles could be very effective as a drug delivery tool and for other biomedical applications.
Submission declaration
This is to declare that the present work is not published anywhere and submitted elsewhere for the publication. All authors materially participated in the present research work, and there will not be any conflicts among authors in the future. Therefore, I request you to consider the present article entitled ‘Piper betle-mediated green synthesis of biocompatible gold nanoparticles’ for publication in the peer-reviewed journal International Nano Letters. Utpal Bora.
Abbreviations
- AuNPs:
-
gold nanoparticles
- PLE:
-
Piper betle leaf extract
- MW:
-
microwave irradiation
- TEM:
-
transmission electron microscopy
- FT-IR:
-
Fourier transforms infrared spectroscopy
- XRD:
-
X-ray diffraction
- EDX:
-
energy-dispersive X-ray spectroscopy.
References
Kasthuri J, Kathiravan K, Rajendiran NJ: Phyllanthin-assisted biosynthesis of silver and gold nanoparticles: a novel biological approach. Nanopart. Res. 2009, 11: 1075–1085. 10.1007/s11051-008-9494-9
Catherine M, Man G, Catherine D, Cusker M, Tuna Y, Rotello VM: Toxicity of gold nanoparticles functionalized with cationic and anionic side chains. Bioconjugate Chem. 2004, 15: 897–900. 10.1021/bc049951i
Singh S, Nalwa HS: Nanotechnology and health safety toxicity and risk assessments of nanostructured materials on human health. J Nanosci Nanotechnol 2007, 7: 3048–3070. 10.1166/jnn.2007.922
Kattumuri V, Katti K, Bhaskaran S, Boote EJ, Casteel SW, Fent GM: Gum arabic as a phytochemical construct for the stabilization of gold nanoparticles: in vivo pharmacokinetics and X-ray-contrast-imaging studies. Small 2007, 3: 333–341. 10.1002/smll.200600427
Gleiter H: Nanostructured materials: basic concepts and microstructure. Acta Materialia. 2000, 48: 1–29. 10.1016/S1359-6454(99)00285-2
Bradley JS, Schmid GE: Cluster and Colloids: From Theory to Application. Wiley, Weinheim; 1994.
Elechiguerra JL, Burt JL, Morones RJ, Camacho A, Gao X, Lara HH: Interaction of silver nanoparticles with HIV-1. J Nanobiotechnol. 2005, 3: 1–10. 10.1186/1477-3155-3-1
Niranjan R, Nivedita R, Ritu I, Chandrasekaran S: Phenolic antibacterials from Piper betle in the prevention of halitosis. J Ethnopharmacol 2002, 83: 149–152. 10.1016/S0378-8741(02)00194-0
Razak FA, Othman RY, Haji ZHA: Evaluation of antihistaminic activity of Piper betel leaf in guinea pig. J Oral Sci. 2006, 48: 71–75. 10.2334/josnusd.48.71
Santhanam G, Nagarjan S: Development and validation of UV spectroscopic methods for the quick estimation of Piper betle leaf (PBL) extract. Fitoterapia 1990, 61: 458–459.
Prabhu MS, Patel K, Saraawathi G, Srinivasan K: Effect of orally administered betel leaf (Piper betle leaf Linn.) on digestive enzymes of pancreas and intestinal mucosa and on bile production in rats. Indian J Exp Biol 1995, 33: 752–756.
Dasgupta N, De B: Antioxidant activity of Piper betel L. leaf extract in vitro. Food Chem 2004, 88: 219–224. 10.1016/j.foodchem.2004.01.036
Choudhury D, Kale RK: Antioxidant and non-toxic properties of Piper betle leaf extract: in vitro and in vivo studies. Phytother Res. 2002, 16: 461–466. 10.1002/ptr.1015
Tappayuthpijarn P, Dejatiwongse Q, Pongpech P, Leelaporn A: Antibacterial activity of extracts of Piper betle leaf. Thai J Pharmacol. 1982, 4: 205–212.
Boonyaratanakornkit L, Pothiyanont P, Noppakun N, Sinhaseni P, Laorpaksa A, Virunhaphol S: Activity of betle leaf ointment on skin diseases. Thai J Pharm Sci. 1990, 15: 277–287.
Pongpech P, Prasertsilpe VJ: The study of antimicrobial activity of Piper betle cream and gel against some fungi, yeast and bacteria. GPO. 1993, 19: 8–22.
Bhattacharya S, Subramanian M, Roychowdhury S, Bauri AK, Kamat JP, Chattopadhyay SJ: Radioprotective property of the ethanolic extract of Piper betel leaf. Radiat Res 2005, 46: 165–171. 10.1269/jrr.46.165
Arambewela LSR, Arawwawala LDAM, Ratnasooriya WD: Antidiabetic activities of aqueous and ethanolic extracts of Piper betle leaves in rats. J Ethnopharmacol 2005, 102: 239–245. 10.1016/j.jep.2005.06.016
Nanjing Zhongyiyao University: The Dictionary of Traditional Chinese Medicines. Shanghai Science and Technology Press, Shanghai; 2006.
Capdevielle-Pardies P, David J, Miquel JL, Le Bras M: Quid of betel. Med Trop. 1985, 45: 299–307.
Patel K, Kapoor S, Dave DP: Synthesis of nanosized silver colloids by microwave dielectric heating. J Chem Sci 2005, 117: 53–60. 10.1007/BF02704361
Yin HB, Yamamoto T, Wada YJ: Large-scale and size-controlled synthesis of silver nanoparticles under microwave irradiation. Materials Chemi and Phys. 2004, 83: 66–70. 10.1016/j.matchemphys.2003.09.006
Shankar SS, Rai A, Ahmad A, Sastry M: Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. J Colloid Interface Sci 2004, 275: 496–502. 10.1016/j.jcis.2004.03.003
Kannan P, John SA: Synthesis of mercaptothiadiazole-functionalized gold nanoparticles and their self-assembly on Au substrates. Nanotechnology 2008, 19: 085602. 10.1088/0957-4484/19/8/085602
Shankar SS, Rai A, Ankamwar B, Singh A, Ahmad A, Sastry M: Biological synthesis of triangular gold nanoprisms. Nat Mater 2004, 3: 482–489. 10.1038/nmat1152
Babu PJ, Sharma P, Kalita MC, Bora U: Green synthesis of biocompatible gold nanoparticles using Fagopyrum esculentum leaf extract. Front Mater Sci 2011, 5: 379–387. 10.1007/s11706-011-0153-1
Babu PJ, Sharma P, Borthakur BB, Das RK, Bora U: Synthesis of gold nanoparticles using Mentha arvensis leaf extract. International Journal of Green Nanotechnology: Physics and Chemistry 2010, 2: 62–68. 10.1080/19430876.2010.532443
Acknowledgments
We thank the Department of Science and Technology, Government of India, for funding this work (Project No. R & D/07/1479/ENV/P/UB/04). PJB thanks MHRD for providing research fellowship and PS thanks DBT Biotech Hub for the post doctoral trainee ship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
UB put forward the hypothesis for this work JBP, UB, and RT interpreted the results and prepared the manuscript. JBP, SS, and PS prepared the samples, conducted the biophysical studies such as TEM, XRD, FT-IR, etc. All authors read and approved the final manuscript.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Punuri, J.B., Sharma, P., Sibyala, S. et al. Piper betle-mediated green synthesis of biocompatible gold nanoparticles. Int Nano Lett 2, 18 (2012). https://doi.org/10.1186/2228-5326-2-18
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/2228-5326-2-18