Abstract
Background
Gabapentin has been adopted in Enhanced Recovery After Surgery protocols as a means to reduce opioid consumption while maintaining adequate post-operative analgesia. The purpose of our study was to review and compare changes in length of stay, opioid use, and patient reported pain scores after the addition of gabapentin into five, distinct pain protocols for posterior spinal fusion in adolescent idiopathic scoliosis.
Methods
A retrospective review was completed using a database of electronic medical data from a single pediatric orthopedic healthcare system that was queried for patients with adolescent idiopathic scoliosis who underwent first-time posterior spinal fusion. Perioperative data including demographics, hospital length of stay, surgical details, opioid use, patient reported pain scores, and non-opioid analgesic use were collected.
Results
From December 2012 to February 2019, 682 hospitalizations for posterior spinal fusion in adolescent idiopathic scoliosis were identified with complete inpatient data; 49% were administered gabapentin. For the gabapentin cohort, the system saw no statistically significant effect on length of stay or pain averaged over POD#0–3. Opioid use was statistically lower averaged over POD#0–3. Individual sites saw variation on length of stay and opioid use compared to the system.
Conclusion
In conclusion, system-wide data showed gabapentin containing protocols reduced opioid use while maintaining clinically equivalent analgesia. However, variations of individual site results make it difficult to conclude the degree to which gabapentin were responsible for this effect.
Similar content being viewed by others
Background
Enhanced recovery after surgery (ERAS) protocols were first published nearly 20 years ago. Since then, a multitude of guidelines have been published for a variety of surgical procedures. The underlying foundation of ERAS is to use a multidisciplinary and multimodal approach to minimize the stress response following surgery in order to optimize patient recovery [1]. One of the goals of ERAS protocols is to reduce the dependence on opioids for post-operative analgesia. Opioids carry several side effects including nausea, vomiting, sedation, pruritus, and urinary retention [2]. The American Society of Pain recommends gabapentinoids (gabapentin, pregabalin) be considered as part of a multi-modal approach to analgesia [3]. It is thought that gabapentinoids decrease acute post-operative pain by inhibiting the release of excitatory neurotransmitters from primary afferent nerve fibers thus reducing hyper-excitability of dorsal horn neurons following tissue injury during surgery [4].
Several studies have looked at the analgesic efficacy of gabapentin for posterior spinal fusion in adolescent idiopathic scoliosis. One study, which looked at the use of a single loading dose of gabapentin did not change post-operative pain or opioid use [5]. However, a series of four recent studies which used both a loading dose plus inpatient maintenance dosing showed deceases in opioid use and some improvement in post-operative pain [6,7,8,9].
In this study, we focus on the implementation of gabapentin as part of pain protocols in five geographically diverse, pediatric orthopedic institutions. Each autonomously managing their clinical protocols but operating under an umbrella healthcare system. We reviewed changes in length of stay, post-operative opioid use, and post-operative patient reported pain scores after the addition of gabapentin into pain protocols.
The purpose of our study is to compare the results at individual sites with the findings of the aforementioned studies. Our future goal is to use these findings to develop a standardized, system wide pain protocol.
Methods
This study was reviewed by an Institutional Review Board and determined to be exempt as defined under federal regulation 45 CFR 46. A database containing de-identified electronic medical record data was queried from December 2012 to February 2019 for all patients with adolescent idiopathic scoliosis (AIS) who underwent first-time posterior spinal fusion (PSF). Data from nine, affiliated, pediatric orthopedic institutions across the United States, was returned. Pain protocols were designed and implemented independently at each site. This included the dosing protocol of gabapentin, use of other non-opioid analgesics, and opioid administration protocol. Sites using neuroaxial (intrathecal opioid or epidural infusions) anesthesia as part of their pain protocol were removed. Specific gabapentin dosing protocols during individual visits were not available. Table 1 provides the median loading and maintenance dose of gabapentin at each site to approximate the most common dosing protocol at each site.
Perioperative data for this population; including demographics, hospital length of stay, surgical details, opioid use, patient reported pain scores, and non-opioid analgesic use; was collected. The primary outcome measures were length of stay, opioid use from post-operative day (POD) #0 to #3, and patient reported pain scores from POD#0 to POD#3.
Length of stay was calculated from the day of surgery to day of discharge. POD#0 was defined as arrival time in post-anesthesia care unit until midnight on the day of surgery. Subsequent post-operative days were defined as intervals from midnight to midnight. Opioid use (intravenous and oral) and pain scores (0–10 numeric rating system) were collected from POD#0 to POD#3. Individual patient opioid use was converted to oral morphine equivalents, summed, and normalized by weight for this time-period while pain scores were averaged. Statistical analysis we done using SPSS v27 (IBM Corporation, Armonk NY, USA). Fischer exact test and Mann–Whitney U test were used for non-normally distributed data. Statistical significance was defined as p < 0.05.
Results
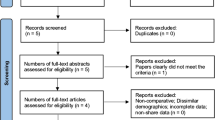
From December 2012 to February 2019, 682 hospitalizations for PSF in AIS were identified with complete inpatient data on length of stay, opioid use, and patient reported pain scores across the five sites (Fig. 1). For these 682 patients, 336 (49%) were administered gabapentin.
Initial query returned 1221 visits for first-time PSF in AIS. After review of data, 149 visits were removed due to inaccurate or incomplete data. Another 390 were removed from institutions using neuroaxial techniques as part of their pain protocol. Final study group consisted of 682 visits. Site-specific breakdown given with total number of visits followed by number of patients administered and not administered gabapentin during hospitalization
Table 2 provides demographic, surgical, and combined system data. The gabapentin administration group had statistically longer surgical times (Yes 409 min v No 394 min, p = 0.016) and more female patients (Yes 84% v No 77%, p = 0.033). The system combined gabapentin cohort saw no statistically significant effect on length of stay (Yes 5.2 v No 5.2, p = 1) or average pain (from POD#0–3 Yes 3.8 v No 3.9, p = 0.527). Opioid use was statistically lower averaged over POD#0–3 (Yes 4.2 v No 4.8, p < 0.001); with all days except POD#0 seeing a statistically significant decrease.
Demographic, surgical, and site-specific data for each of the three end-points is reported in Additional file 1. Site 4 saw a statistically different distribution in the number of levels fused with the gabapentin group having more surgeries of greater complexity and increased surgical time. The gabapentin group was younger in Site 3. Body mass index was greater for the gabapentin group in Site 2.
Three sites (Site 2, Site 3, Site 5) saw a statistically significant decrease in length of stay. Decrease in pain averaged over POD#0–3 was insignificant across all sites. Change in opioid usage was mixed. Three sites saw a statistically significant decrease (Site 1, Site 3, Site 5) in opioid use. Individual days across sites saw statistically significant fluctuations in both directions.
Discussion
This was a single healthcare system, retrospective database review looking at the effect of gabapentin on hospital length of stay, total opioid use normalized to weight, and average patient reported pain across five different sites; each with a unique but similar pain protocol. Our results showed, when looking at combined system data, the addition of gabapentin to pain protocols decreased average opioid use from immediately after surgery until POD#3. No effect was seen on average pain or average length of stay. These findings on length of stay and opioid use were not reflected at each of the five sites.
When comparing our gabapentin findings with existing current literature, one must first examine the gabapentin dosing protocols in existing studies. Table 3 provides the gabapentin regimen for four existing studies.
These studies had various protocols for loading dose between 10–15 mg/kg and maintenance dosing between 6 to 30 mg/kg/day [6,7,8,9]. Dosing protocols varied at our individual sites (Table 1), but median dosing trended lower than the described study protocols. These differences make comparing the efficacy of gabapentin difficult and points to the need for defining and standardizing an optimal gabapentin dosing protocol.
In looking at our three primary end-points, our system in total did not see a significant change in length of stay. This resembles Choudhry et al. finding no significant decrease in hours to discharge in the gabapentin group [6]. However, three sites (Site 2, Site 3, Site 5) saw gabapentin patients, on average, discharged sooner. In addition to pain control, several factors are responsible for hospital length of stay including ambulation, lack of surgical complications, and home readiness. We did not have information on discharge processes and criteria changes over time at our institutions. Without being to compare discharge criteria, it is difficult to conclude the effect of gabapentin on length of stay.
When considering effects on patient reported pain following surgery, it is important to first consider what a clinically significant change in pain score is. Research into pain scores has shown that in a 0–10 numeric rating scale (NRS) system, for patients experiencing moderate pain, a minimal change of 1.3 is needed by patients to report improvement [10]. Similarly in a 0–100 visual analogue scale (VAS) system, again for moderate pain (VAS of 31–70), a change of at least 10 points was deemed meaningful by patients [11]. In prior studies, only Rusy et al. reported a statistically and clinically significant decrease immediately following surgery (2.5 v 6) and first pain score the morning after surgery (3.2 v 5) [7]. In our data, the two groups had no clinically significant decrease averaged over POD#0–3 or on specific days. This lack of change in patient reported pain scores was not surprising. We had thought patients and nursing would maintain pain at the same tolerable levels before and after introduction of gabapentin. All previous studies showed gabapentin decreasing opioid usage approximately 30% with the effect greatest the first day or two after surgery [6,7,8,9]. In our system, the gabapentin group experienced a statistically significant decrease in opioid use averaged from POD#0–3 and each day except POD#1. However, only three sites saw a statistically significant decrease averaged over POD#0–3 (Site 1 14%, Site 2 25%, Site 5 20%). Daily statistically significant increases and decreases in opioid usage were seen scattered across the individual sites. When analyzing opioid use, it is important to consider if opioids were administered on a schedule, or if dosed as needed by patient request. Gabapentin groups may have required less opioids but were still administered opioids according to a scheduled protocol. Unfortunately, our database was unable to capture if opioids were given at a scheduled interval or as needed by patients. We hope to re-examine opioid administration protocols in the future as our database matures.
As a retrospective database review, there is an inherent limitation related to possible erroneously documented data in the electronic medical records. In an attempt to mitigate this effect, all data in this study was manually reviewed and outliers were discarded if unable to be corrected. Additionally, we lacked precise details on the degree of surgical correction, attempting to use duration of surgery and billing codes as proxy. Any changes in discharge processes over time at the individual institutions was unknown.
Further, variations in surgical technique approach and equipment likely exist in and between sites. These surgical variables should be considered in future multi-modal analgesic work.
Our five sites adopted different gabapentin dosing protocols to their underlying multi-modal pain protocols. This indicates, as prior studies have concluded, that more research needs to be done to determine the optimal gabapentin dosing protocol which balances analgesic potency with adverse side effects (sedation, dizziness, fall risk). Further, the efficacy of gabapentin needs to be studied in relation to other multi-modal analgesic techniques (scheduled/as need opioid dosing, methadone, non-opioid analgesics, neuroaxial anesthesia) in order to create an optimal multi-modal analgesic protocol.
Conclusion
Our system-wide data suggests the addition of gabapentin to multi-modal pain protocols can decrease the use of opioids while maintaining clinically equivalent analgesia. The heterogeneity in gabapentin dosing and underlying pain protocols, along with the varying individual site results, makes it difficult to conclude the degree to which gabapentin were responsible for this effect. Our more pertinent finding is the need for institutions to verify the desired clinical effect of new or modified components of multi-modal analgesic protocols by defining, collecting, and analyzing objective metrics of quality improvement.
Availability of data and materials
Restrictions apply to the availability of these data. Data are available upon request from the corresponding author for researchers who meet the criteria for access to confidential data.
Abbreviations
- ERAS:
-
Enhanced recovery after surgery
- AIS:
-
Adolescent idiopathic scoliosis
- PSF:
-
Posterior spinal fusion
- POD:
-
Post-operative day
- NRS:
-
Numeric rating scale
- VAS:
-
Visual analogue scale
References
Brindle M, Nelson G, Lobo DN, Ljungqvist O, Gustafsson UO. Recommendations from the ERAS® Society for standards for the development of enhanced recovery after surgery guidelines. BJS open. 2020;4(1):157–63. https://doi.org/10.1002/bjs5.50238.
Dolin SJ, Cashman JN. Tolerability of acute postoperative pain management: nausea vomiting sedation pruritis and urinary retention. Evidence from published data. Br J Anaesth. 2005;95(5):584–91 S0007091217349905. https://doi.org/10.1093/bja/aei227.
Chou R, Gordon DB, de Leon-Casasola OA, Rosenberg JM, Bickler S, Brennan T, Carter T. Guidelines on the management of postoperative pain. J Pain. 2016;17(2):131–57. https://doi.org/10.1016/j.jpain.2015.12.008.
Tiippana EM, Hamunen K, Kontinen VK, Kalso E. Do surgical patients benefit from perioperative gabapentin/pregabalin? A systematic review of efficacy and safety. Anesth Analg. 2007;104(6):1545–56. https://doi.org/10.1213/01.ane.0000261517.27532.80.
Mayell A, Srinivasan I, Campbell F, Peliowski A. Analgesic effects of gabapentin after scoliosis surgery in children: a randomized controlled trial. Pediatr Anesth. 2014;24(12):1239–44. https://doi.org/10.1111/pan.12524.
Choudhry DK, Brenn BR, Sacks K, Shah S. Evaluation of gabapentin and clonidine use in children following spinal fusion surgery for idiopathic scoliosis: a retrospective review. J Pediatr Orthop. 2019;39(9):e687–93. https://doi.org/10.1097/BPO.0000000000000989.
Rusy LM, Hainsworth KR, Nelson TJ, Czarnecki ML, Tassone JC, Thometz JG, Lyon RM, Berens RJ, Weisman SJ. Gabapentin use in pediatric spinal fusion patients: a randomized, double-blind, controlled trial. Anesth Analg. 2010;110(5):1393–8. https://doi.org/10.1213/ANE.0b013e3181d41dc2.
Anderson DE, Duletzke NT, Pedigo EB, Halsey MF. Multimodal pain control in adolescent posterior spinal fusion patients: a double-blind, randomized controlled trial to validate the effect of gabapentin on postoperative pain control, opioid use, and patient satisfaction. Spine Deformity. 2020;8(2):177–85. https://doi.org/10.1007/s43390-020-00038-z.
Trzcinski S, Rosenberg RE, Vasquez Montes D, Sure A, Zhou P, Tishelman J, Mansky R, McLeod L, Tracy J, Buckland AJ, Errico TJ. Use of gabapentin in posterior spinal fusion is associated with decreased postoperative pain and opioid use in children and adolescents. Clinical Spine Surgery. 2019;32(5):210–4. https://doi.org/10.1097/BSD.0000000000000783.
Cepeda MS, Africano JM, Polo R, Alcala R, Carr DB. What decline in pain intensity is meaningful to patients with acute pain? Pain. 2003;105(1–2):151–7. https://doi.org/10.1016/s0304-3959(03)00176-3.
Bodian CA, Freedman G, Hossain S, Eisenkraft JB, Beilin Y. The visual analog scale for pain: clinical significance in postoperative patients. J American Soc Anesthesiol. 2001;95(6):1356–61. https://doi.org/10.1097/00000542-200112000-00013.
Acknowledgements
We thank Carole Tucker, PT, PhD for her technical assistance in querying the database.
Shiners Spine Study Group institutional affiliations
Michelle Diu 1, Graham Fedorak 2, Purnendu Gupta 3, Matthew Kuestner 2, Cheryl Lawing 4, Scott Luhmann 5, Robert Moore 5, Sarah Oswald 3, Rolando Roberto 6, Casey Stondell 6, Vishwas Talwalkar 7, Pravin Taneja 8, Richard Vax 4, Polina Voronov 3, Michelle Welborn 1.
1 Shriners Children's Portland; 3101 SW Sam Jackson Park Rd, Portland, OR 97239, USA
2 Shriners Children's Salt Lake City; 1275 E Fairfax Rd, Salt Lake City, UT 84103, USA
3 Shriners Children's Chicago; 2211 N Oak Park Ave, Chicago, IL 60707, USA
4 Shriners Children's Florida; 12,502 USF Pine Drive, Tampa, FL 33612, USA
5 Shriners Children's St. Louis; 4400 Clayton Ave, St. Louis, MO 63110, USA
6 Shriners Children's Northern California; 2425 Stockton Blvd, Sacramento, CA 95817, USA
7 Shriners Children's Lexington; 110 Conn Terrace, Lexington, KY 40508, USA
8 Shriners Children's Philadelphia; 3551 N Broad St, Philadelphia, PA 19140, USA
Funding
This research received no external funding.
Author information
Authors and Affiliations
Consortia
Contributions
Conceptualization, D.Z. and S.P.; methodology, D.Z.; software, D.Z.; validation, D.Z.; formal analysis, D.Z. and B.B.; investigation, D.Z.; resources, R.C. and A.S.; data curation, D.Z.; writing—original draft preparation, D.Z.; writing—review and editing, all authors; visualization, D.Z. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All methods were carried out in accordance with relevant guidelines and regulations. All experimental protocols were approved by Shriners Hospitals for Children Department of Research Programs and Western Institutional Review Board’s IRB Affairs Department. Patient consent was waived by Western Institutional Review Board’s (WIRB’s) IRB Affairs Department under 45 CFR § 46.104(d)(4).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests related to this research.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Demographic, surgical, and outcome data for each individual site is presented.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, DA., Brenn, B., Cho, R. et al. Effect of gabapentin on length of stay, opioid use, and pain scores in posterior spinal fusion for adolescent idiopathic scoliosis: a retrospective review across a multi-hospital system. BMC Anesthesiol 23, 10 (2023). https://doi.org/10.1186/s12871-022-01965-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12871-022-01965-2