Abstract
Background
Regorafenib belongs to a sub-group of small-molecule multi-targeted tyrosine kinase inhibitors(TKIs). In various studies with respect to the side-effect of regorafenib, drug-associated proteinuria standardly qualified to be defined as nephrotic syndrome was rarely reported as well as the relation of regorafenib with the occurrence and development of thrombotic microangiopathy (TMA).
Case presentation
In this case report and literature review, we presented a 62-year-old patient receiving regorafenib for metastatic colon cancer, manifesting abundant proteinuria, in which TMA was also diagnosed through renal biopsy. As far as we were concerned, this was the first reported in terms of regorafenib-induced TMA confirmed by renal biopsy.
Conclusion
This case indicates that regorafenib, a kind of TKIs may result in TMA, which is a rare but life-threatening complication of cancer treatment drug. Insights from this case might help physicians diagnose rare forms of TMA and adjust treatment for patients in a timely manner.
Similar content being viewed by others
Background
Regorafenib is an orally-taken small-molecule multi-targeted tyrosine kinase inhibitors(TKIs)that block the activities of various protein kinases which are supposed to activate vascular endothelial growth factor receptors 1-3(VEGFR 1-3), platelet-derived growth factor receptors and fibroblast growth factor receptors by targeting tyrosine receptor kinases, and thus influence cellular metabolism, proliferation and distant migration [1]. Because of its anti-tumoral function, regorafenib has been applied around the world in metastatic colorectal cancer (mCRC). However, since proteinuria and hypertension are common side effects among all drug therapies targeting VEGF signaling and its downstream pathways, major adverse effects (AEs) should be considered with the use of such drugs [2]. In the past decade, reports have suggested that TKIs might exert multiple effects on patients’ kidney, inducing hypertension, proteinuria, acute kidney injury (AKI), and thrombotic microangiopathy (TMA) [3,4,5,6]. Among patients receiving regorafenib, hypertension occurs in 32.4% of the population, while all-grade and high-grade proteinuria are found in 7.0 and 1.4% of them respectively [7, 8]. A previous phase 3 clinical trial showed regorafenib-induced proteinuria was more frequent in Japanese individuals compared with other populations, but was generally manageable [9]. Other studies assessing regorafenib-associated proteinuria found out proteinuria rarely reached the diagnostic standard of nephrotic syndrome, and it remained unknown whether regorafenib was associated with the development of TMA. Meanwhile, studies investigating the associations between TKIs and nephrotic syndrome were scarce.
In order to supplement research on regorafenib use and morbidity of TMA, we presented a 62-year-old female receiving regorafenib for metastatic colon cancer, who developed abundant proteinuria caused by TMA (determined pathologically).
Case presentation
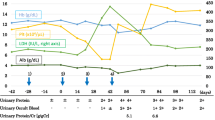
A 62-year-old woman was diagnosed with stage IV colon cancer at hepatic flexure and lung metastasis according to the 7th edition of the American Joint Committee on Cancer (AJCC) tumor node metastasis (TNM) staging system two years ago (May 4th, 2016), and have been treated with combined neoadjuvant chemotherapy (imatinib, folinic acid and fluorouracil) as the primary drug regimen. Three weeks later (May 25th, 2016), she was admitted to the Department of Gastrointestinal Surgery for relieving her symptoms, during which tumor removal with palliative right hemi-colectomy was performed. Afterwards, FOLFIRI (5-fluorouracil, leucovorin, irinotecan) regimen which included leucovorin at 500 mg d1, fluorouracil at 500 mg d1, fluorouracil at 1700 mg civ/23h d1-2 Q2W, and irinotecan at 260 mg d1, was administered for 6 courses (July 14th, 2016 - Nov 24th, 2016). However, she still manifested progressive cancer, lung metastasis was determined as right lung biopsy showed moderately differentiated intestinal adenocarcinoma with RAS (rat sarcoma virus) genetic mutations. Therefore, bevacizumab (anti-VEGF-A antibody) (400 d1 q3w) plus 8 courses of XELOX which included oxaliplatin at 150 mg d1 and capecitabine at 1500 mg bid d1-d14 (Jan 5th, 2017 - Sep 25th, 2017) was initiated after the abovementioned treatment. Radiotherapy for pulmonary metastasis was also carried out 28 times in 2 months (Jan 8th, 2017 - Sep 22nd, 2017). Nevertheless, the tumor was not able to be contained either and continued to progress. Thus, regorafenib(80 mg qd d1-d21) was added on Jan 11th, 2018.In the beginning, she had normal renal function (serum creatinine, 45 μmol/L) without proteinuria or hypertension. Bevacizumab administration was performed for 8 courses spanning 8 months with transient liver dysfunction (Fig. 1A). After discontinuing bevacizumab, liver function returned to normal. However, serum creatinine gradually increased after regorafenib treatment initiation, her blood test showed hypercholesterolemia (7.11 mmol/L) and hypertriglyceridemia (2.04 mmol/L) (Fig. 1B). Moreover, she progressively developed nephrotic syndrome (serum creatinine, 91 μmol/L) in recent three months, and was thus transferred to the Department of Nephrology. On admission, her 24-hour urinary protein was 4.6 g/24h, and serum albumin was 29.1 g/L (Table 1). Regarding antibodies related to membranous nephropathy, anti-phospholipase A2 (PLA2) antibody was negative. While anti-thrombospondin type-1domain-containing 7A (THSD7A) antibody was positive. Coombs test, haptoglobin and also ADAMTS-13 activity were negative. Kidney biopsy showed fibrin thrombi in glomerular capillaries, endothelial injury with thickening, and mesangiolysis (Fig. 2A, B, C, D) revealing TMA. Meanwhile, Immunofluorescence showed IgM, kappa and lambda deposition in glomerular mesangium and glomerular capillary loops (Fig. 3A, B, C) with weak deposition of C4 and C1q. Electron microscopy revealed diffuse endothelial thickening with obliterated capillary lumina and ubendothelial fluffy materials or fibrin tactoids deposition (Fig. 4). Pathological examination indicated a diagnosis of renal-limited TMA caused by anti-VEGF drugs. After careful consultation with oncologists, regorafenib therapy was suspended. Two years following discharge from our hospital, the patient received irbesartan treatment and responded with gradual improvement of her clinical manifestation, with urinary protein 2+ and serum albumin of 33.9 g/L (Table 1).
Discussion and conclusion
The patient’s other causes of nephrotic syndrome including membranous nephropathy and minimal change nephropathy were ruled out according to the results of renal biopsy. Because of the patients’ renal injury appeared aftefr regorafenib treatment, combing with the results of renal biopsy, we also exclude a paraneoplastic nephrotic syndrome. The current case indicated that kidney damage associated with regorafenib coule lead to nephrotic-range proteinuria and histopathological TMA limited to the kidney unlike systemic TMA, which was associated with thrombotic thrombocytopenic purpura [10]. Most recently, kidney-related adverse event of regorafenib had been reported as lupus-like glomerulonephritis, which presented with acute kidney injury, proteinuria, and hematuria [6]. However, nephrotic syndrome with or without TMA characteristics has seldom been reported as a severe side effect of TKIs in adults with cancer.
It was widely acknowledged that solid tumors were characterized by rapid growth and abundant blood supply. Therefore, it was a recommended therapeutic to inhibit vascular development in order to treat solid tumors. The main targets of anti-angiogenesis were VEGFs, VEGF receptors (VEGFRs) and molecules in their downstream signaling pathways [11]. Current drugs with approved by the US Food and Drug Administration (FDA) were primarily VEGF antagonists (e.g., bevacizumab and sunitinib), multi-target TKIs (targeting VEGFR-3, including sunitinib and regorafenib) and VEGFR inhibitors such as cetuximab, erlotinib, and gefitinib. TKIs mainly inhibited angiogenesis, which was required for cancer cell survival and proliferation. All-grade and high-grade proteinuria reported in 33 clinical studies using VEGFR and TKIs were found in 18.7 and 2.4% of the individuals respectively [12]. Regorafenib was tolerated by Japanese individuals (n = 100) in the CORRECT (patients with metastatic colorectal cancer treated with regorafenib or placebo after failure of standard therapy) trial at a level generally comparable to other Asians; however, Japanese population showed elevated incidence rates of treatment-associated all-grade hypertension (60 vs. 23%), proteinuria (40 vs. 2%) and thrombocytopenia (39 vs. 9%), and elevated lipase amounts (25 vs. 2%) in comparison with non-Japanese cases [9].
Agents targeting VEGF attracted increasing attention in the treatment of late-stage cancer. VEGF was also a key molecule in the glomerular filtration barrier [13]. Normally, glomerular epithelial cells (podocytes) expressed VEGF, which acted on endothelial cells to maintain the structure and function of the glomerular filtration barrier. Animal experiments have shown that blocking VEGF could lead to endothelial cell damage, coagulation pathway activation, glomerular filtration barrier damaging, proteinuria promotion and TMA occurrence [12]. Glomerular microangiopathy caused by anti-VEGF treatment typically manifested as segmental glomerular capillary microaneurysms and hyalinosis, rarely with concomitant fibrin/platelet thrombi or broken red blood cells. Such microangiopathy was characterized by diffuse glomerular distribution, sparing pre-glomerular vessels, and morphological features of chronic TMA.
In recent years, the rapid development of new anti-tumor biological agents, such as ones with therapeutic inhibition of VEGF signaling, has brought about improved medical efficacy and provided patients with more choices, which however might also affect the functions of multiple organs, including the kidney. Many nephrologists were unfamiliar with kidney damage caused by these drugs, and oncologists had a hard time with it as well. With the necessity of novel anti-cancer drugs and increasing morbidity of renal problems associated with these agents, the need of establishing cross-discipline medical team was urgent, and nephrologists and oncologists should be encouraged to learn from and cooperate with each other in order to treat patients better. Specifically, the renal toxicity of anti-tumor biological agents should be listed as a discussion topic. At the same time, nephrologists should understand the prevention, clinical manifestations and management of cancerous lesions. While using bevacizumab, routine urine, blood pressure, liver and kidney function tests should be performed regularly. A reduction in immune tolerance and immune escape should be recorded as well, and the drug amounts should be reduced, in order to decrease the risk of albuminuria, nephrotic syndrome or renal insufficiency. However, the optimal way to monitor such patients still remained unknown. How susceptibility to VEGF suppression is regulated remains largely undefined. In addition, that an individual tolerating long-term treatment with bevacizumab develops TMA readily upon switching to regorafenib is enigmatic
In conclusion, this case indicates that regorafenib, a kind of TKIs may result in TMA, which is a rare but life-threatening complication of cancer treatment drug. Insights from this case might help physicians diagnose rare forms of TMA and adjust treatment for patients in a timely manner.
Availability of data and materials
The datasets used in this study are available from the corresponding author on reasonable request.
Abbreviations
- TKIs:
-
Tyrosine kinase inhibitors
- TMA:
-
Thrombotic microangiopathy
- RTKs:
-
Receptor tyrosine kinases
- VEGF:
-
Vascular endothelial growth factor
- VEGFR:
-
Vascular endothelial growth factor receptors
- mCRC:
-
Metastatic colorectal cancer
- PLA2:
-
Phospholipase A2
- THSD7A:
-
Thrombospondin type-1domain-containing 7A
- FDA:
-
Food and Drug Administration
References
Wilhelm SM, Dumas J, Adnane L, Lynch M, Carter CA, Schutz G, et al. Regorafenib (BAY 73-4506): a new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity[J]. Int J Cancer. 2011;129(1):245–55. https://doi.org/10.1002/ijc.25864.
Izzedine H, Rixe O, Billemont B, Baumelou A, Deray G. Angiogenesis inhibitor therapies: focus on kidney toxicity and hypertension[J]. Am J Kidney Dis. 2007;50(2):203–18. https://doi.org/10.1053/j.ajkd.2007.04.025.
Jhaveri KD, Flombaum CD, Kroog G, Glezerman IG. Nephrotoxicities associated with the use of tyrosine kinase inhibitors: a single-center experience and review of the literature[J]. Nephron Clin Pract. 2011;117(4):c312–9. https://doi.org/10.1159/000319885.
Bollee G, Pate N, Cazajous G, Robert C, Goujon JM, Fakhouri F, et al. Thrombotic microangiopathy secondary to VEGF pathway inhibition by sunitinib[J]. Nephrol Dial Transplant. 2009;24(2):682–5. https://doi.org/10.1093/ndt/gfn657.
Wallace E, Lyndon W, Chumley P, Jaimes EA, Fatima H. Dasatinib-induced nephrotic-range proteinuria[J]. Am J Kidney Dis. 2013;61(6):1026–31. https://doi.org/10.1053/j.ajkd.2013.01.022.
Strasma A, Coke H, Mamlouk O, Tchakarov A, Mandayam S. Lupus-like glomerulonephritis associated with regorafenib, a multikinase inhibitor[J]. Kidney Med. 2021;3(2):294–8. https://doi.org/10.1016/j.xkme.2020.11.011.
Liu B, Ding F, Liu Y, Xiong G, Lin T, He D, et al. Incidence and risk of hypertension associated with vascular endothelial growth factor receptor tyrosine kinase inhibitors in cancer patients: a comprehensive network meta-analysis of 72 randomized controlled trials involving 30013 patients[J]. Oncotarget. 2016;7(41):67661–73. https://doi.org/10.18632/oncotarget.11813.
Zhang ZF, Wang T, Liu LH, Guo HQ. Risks of proteinuria associated with vascular endothelial growth factor receptor tyrosine kinase inhibitors in cancer patients: a systematic review and meta-analysis[J]. PLoS One. 2014;9(3):e90135. https://doi.org/10.1371/journal.pone.0090135.
Yoshino T, Komatsu Y, Yamada Y, Yamazaki K, Tsuji A, Ura T, et al. Randomized phase III trial of regorafenib in metastatic colorectal cancer: analysis of the CORRECT Japanese and non-Japanese subpopulations[J]. Invest New Drugs. 2015;33(3):740–50. https://doi.org/10.1007/s10637-014-0154-x.
Jha PK, Vankalakunti M, Siddini V, Bonu R, Prakash GK, Babu K, et al. Sunitinib induced nephrotic syndrome and thrombotic microangiopathy[J]. Indian J Nephrol. 2013;23(1):67–70. https://doi.org/10.4103/0971-4065.107215.
Ferrara N. VEGF and the quest for tumour angiogenesis factors[J]. Nat Rev Cancer. 2002;2(10):795–803. https://doi.org/10.1038/nrc909.
Izzedine H, Mangier M, Ory V, Zhang SY, Sendeyo K, Bouachi K, et al. Expression patterns of RelA and c-mip are associated with different glomerular diseases following anti-VEGF therapy[J]. Kidney Int. 2014;85(2):457–70. https://doi.org/10.1038/ki.2013.344.
Pfister F, Amann K, Daniel C, Klewer M, Buttner A, Buttner-Herold M. Characteristic morphological changes in anti-VEGF therapy-induced glomerular microangiopathy[J]. Histopathology. 2018;73(6):990–1001. https://doi.org/10.1111/his.13716.
Acknowledgments
We appreciate the patient for participating in this study.
Funding
This study was supported by the 1•3•5 Project for Disciplines of Excellence, West China Hospital, Sichuan University (No. ZYGD18027). The funding body guided the design of the study and revised the manuscript before submission. The funder also supported all costs associated with the study, including the fees upon acceptance for publication.
Author information
Authors and Affiliations
Contributions
HZ and PF conceived, designed and supervised the entire study. XZ collected all the data, HX and SL made pathological pictures and legend, QY and NG wrote the manuscript. Each author contributed important intellectual content during manuscript drafting and revision. All the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
A family of the participant signed a letter of informed consent to allow her data to be stored, as required by West China Hospital.
Consent for publication
Written informed consent was obtained from the patient for the publication of this case report. A copy of the written consent is available for review by the Editor of this journal.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yin, Q., Guo, N., Zhou, X. et al. Regorafenib-induced renal-limited thrombotic microangiopathy: a case report and review of literatures. BMC Nephrol 23, 112 (2022). https://doi.org/10.1186/s12882-021-02656-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12882-021-02656-9